Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (2): 271-277.doi: 10.12122/j.issn.1673-4254.2026.02.04
Lifan LI( ), Weijian YU(
), Weijian YU( ), Meitao TAN, Yunting LI, Anfei LIU, Xiaojing MENG(
), Meitao TAN, Yunting LI, Anfei LIU, Xiaojing MENG( )
)
Received:2025-08-27
Online:2026-02-20
Published:2026-03-10
Contact:
Xiaojing MENG
E-mail:Lilifan5256@163.com;yuweijianliam@163.com;xiaojingmeng@smu.edu.cn
Lifan LI, Weijian YU, Meitao TAN, Yunting LI, Anfei LIU, Xiaojing MENG. Lactobacillus plantarum alleviates lead exposure-induced learning and memory impairment in mice by regulating bile acid metabolism and inhibiting hippocampal NLRP3 expression[J]. Journal of Southern Medical University, 2026, 46(2): 271-277.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.02.04
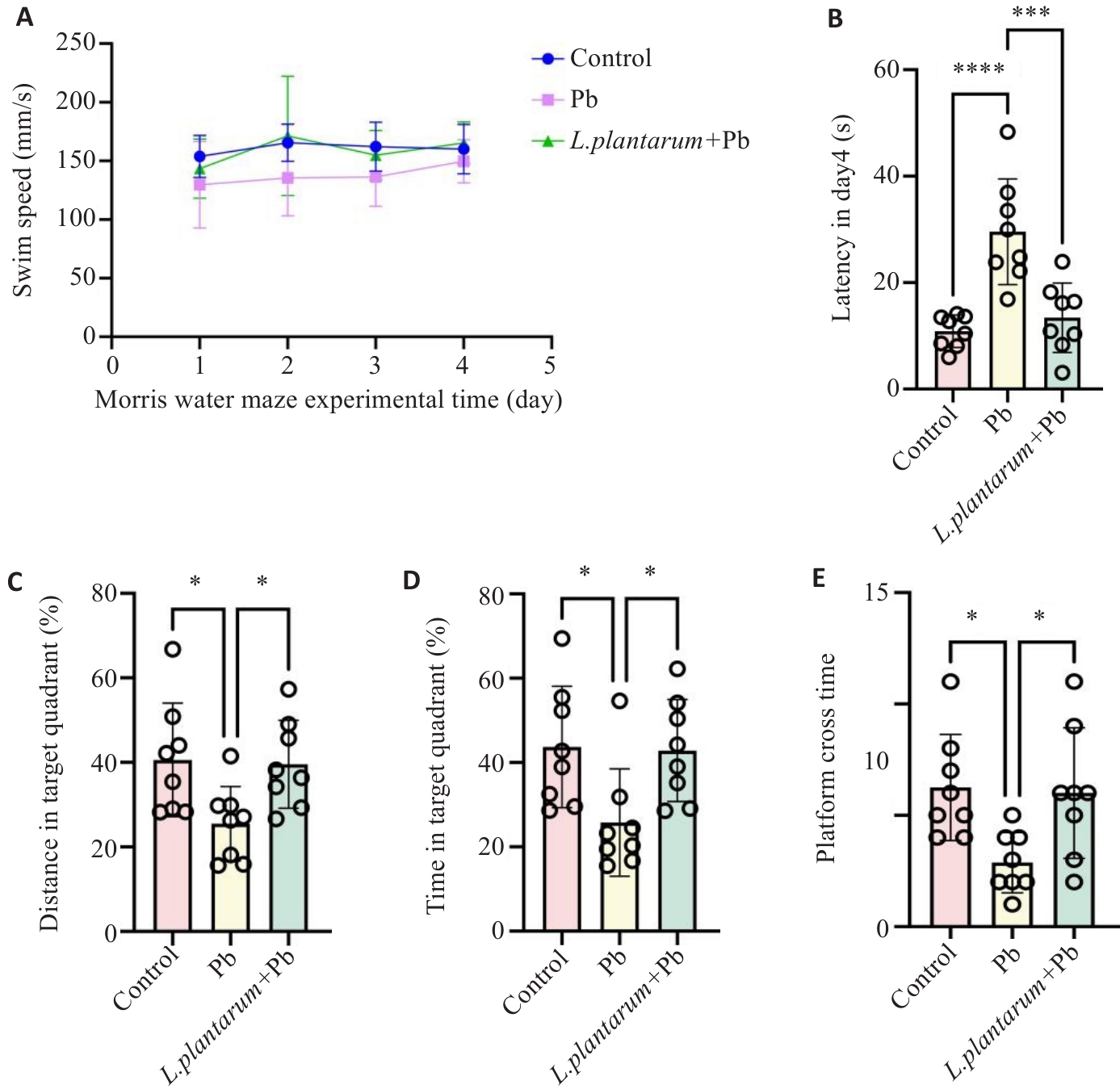
Fig.1 L.plantarum supplementation alleviates lead-induced learning and memory impairments in mice as shown by performance in Morris water maze test. A: Swimming speed. B: Escape latency on day 4. C: Percentage of swimming distance in the target quadrant. D: Percentage of swimming time in the target quadrant. E: Number of crossings over the target platform. n=8, *P<0.05, ***P<0.001, ****P<0.0001.
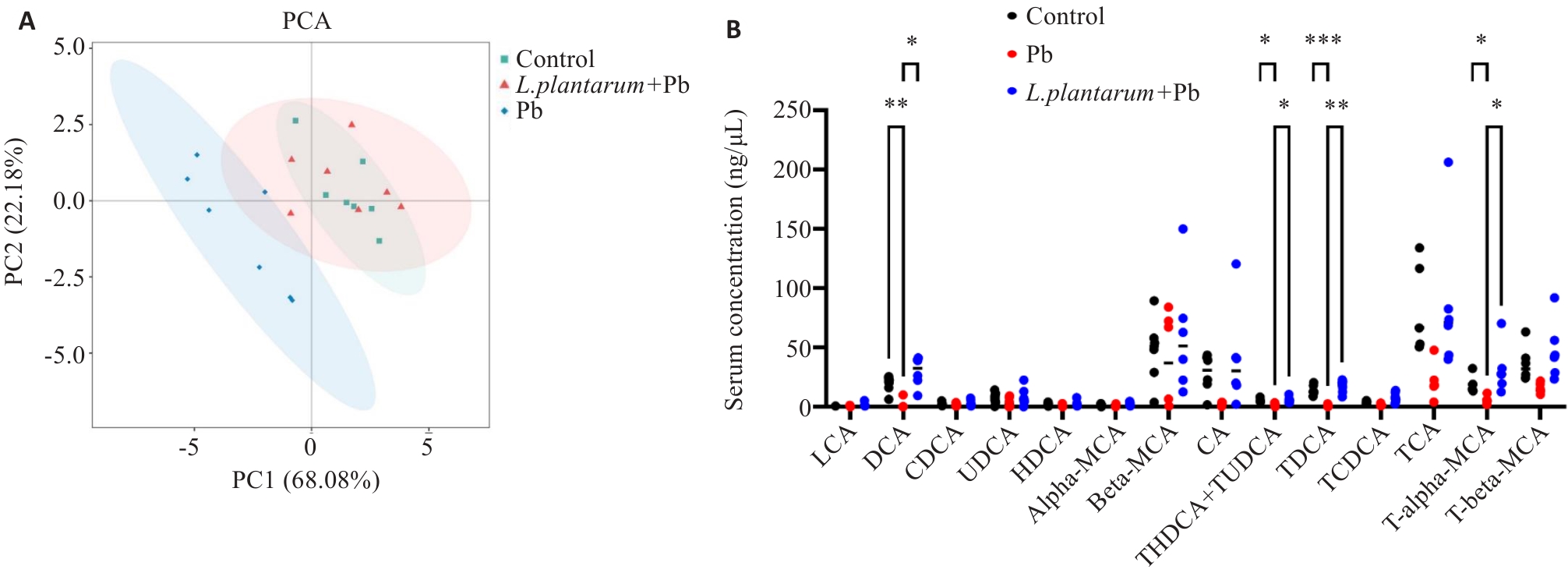
Fig.3 L.plantarum treatment partially restores lead-induced disorders in bile acid metabolism. A: Principal component analysis of serum bile acid targeted metabolic profiles in different groups. B: Quantitative comparison of serum bile acid levels among the 3 groups. *P<0.05, **P<0.01, ***P<0.001.
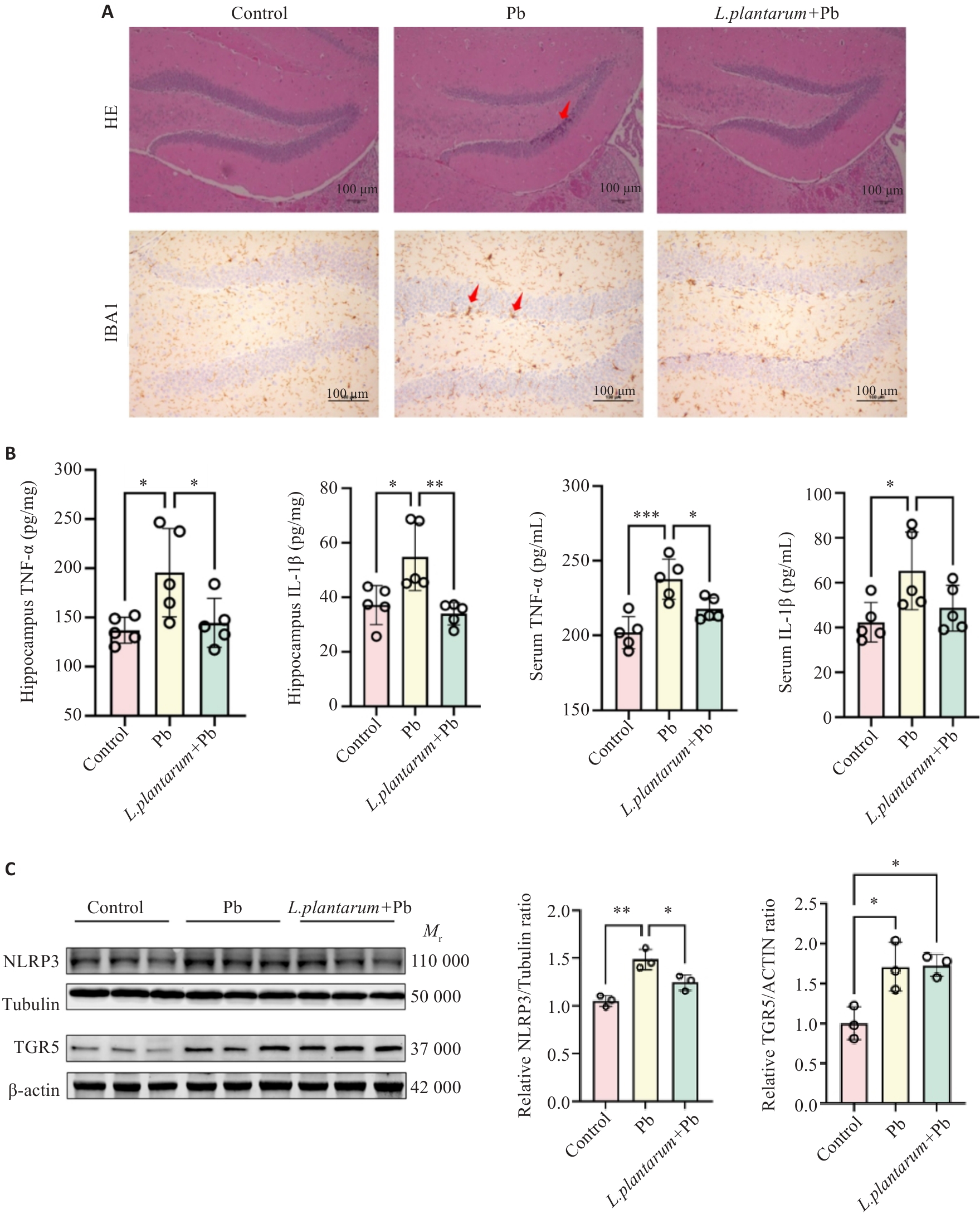
Fig.4 L.plantarum supplementation alleviates lead-induced neuroinflammation by inhibiting NLRP3 inflammasome. A: HE staining and immunohistochemistry for IBA1 expression in the hippocampal DG region. The red arrow in the upper panel indicates hippocampal pathology, and the arrows in the lower panel indicate activated microglia. B: Levels of IL-1β and TNF-α in the hippocampal tissue and serum of the mice detected using ELISA (n=5). C: Western blotting for detecting TGR5 and NLRP3 expression levels in the hippocampus (n=3). *P<0.05, **P<0.01, ***P<0.001.
| [1] | Tong S, von Schirnding YE, Prapamontol T. Environmental lead exposure: a public health problem of global dimensions[J]. Bull World Health Organ, 2000, 78(9): 1068-77. |
| [2] | 国家统计局https://data.stats.gov.cn/easyquery.htm?cn=A01&zb=A02091F&sj=202504 [Z]. 2024 |
| [3] | Gottesfeld P, Cherry CR. Lead emissions from solar photovoltaic energy systems in China and India[J]. Energy Policy, 2011, 39(9): 4939-46. doi:10.1016/j.enpol.2011.06.021 |
| [4] | Wei BG, Yang LS. A review of heavy metal contaminations in urban soils, urban road dusts and agricultural soils from China[J]. Microchem J, 2010, 94(2): 99-107. doi:10.1016/j.microc.2009.09.014 |
| [5] | Patrick L. Lead toxicity part II: the role of free radical damage and the use of antioxidants in the pathology and treatment of lead toxicity[J]. Altern Med Rev, 2006, 11(2): 114-27. |
| [6] | Virgolini MB, Aschner M. Molecular mechanisms of lead neurotoxicity[J]. Adv Neurotoxicol, 2021, 5: 159-213. doi:10.1016/bs.ant.2020.11.002 |
| [7] | Shvachiy L, Geraldes V, Amaro-Leal Â, et al. Intermittent low-level lead exposure provokes anxiety, hypertension, autonomic dysfunction and neuroinflammation[J]. NeuroToxicology, 2018, 69: 307-19. doi:10.1016/j.neuro.2018.08.001 |
| [8] | Xie J, Xie L, Wei H, et al. Dynamic regulation of DNA methylation and brain functions[J]. Biology: Basel, 2023, 12(2): 152. doi:10.3390/biology12020152 |
| [9] | Kim M, Delgado E, Ko S. DNA methylation in cell plasticity and malignant transformation in liver diseases[J]. Pharmacol Ther, 2023, 241: 108334. doi:10.1016/j.pharmthera.2022.108334 |
| [10] | Huang DB, Chen LX, Ji QY, et al. Lead aggravates Alzheimer's disease pathology via mitochondrial copper accumulation regulated by COX17[J]. Redox Biol, 2024, 69: 102990. doi:10.1016/j.redox.2023.102990 |
| [11] | Pikor D, Hurła M, Słowikowski B, et al. Calcium ions in the physiology and pathology of the central nervous system[J]. Int J Mol Sci, 2024, 25(23): 13133. doi:10.3390/ijms252313133 |
| [12] | Liu AF, Li YT, Li LF, et al. Bile acid metabolism is altered in learning and memory impairment induced by chronic lead exposure[J]. J Hazard Mater, 2024, 471: 134360. doi:10.1016/j.jhazmat.2024.134360 |
| [13] | Shannon M, Graef J, Lovejoy FH. Efficacy and toxicity of d-penicillamine in low-level lead poisoning[J]. J Pediatr, 1988, 112(5): 799-804. doi:10.1016/s0022-3476(88)83212-8 |
| [14] | Tizabi Y, Bennani S, El Kouhen N, et al. Interaction of heavy metal lead with gut microbiota: implications for autism spectrum disorder[J]. Biomolecules, 2023, 13(10): 1549. doi:10.3390/biom13101549 |
| [15] | Yu XM, Wei M, Yang D, et al. Lactiplantibacillus plantarum strain FLPL05 promotes longevity in mice by improving intestinal barrier[J]. Probiotics Antimicrob Proteins, 2023, 15(5): 1193-205. doi:10.1007/s12602-022-09933-5 |
| [16] | Liu YW, Liong MT, Tsai YC. New perspectives of Lactobacillus plantarum as a probiotic: the gut-heart-brain axis[J]. J Microbiol, 2018, 56(9): 601-13. doi:10.1007/s12275-018-8079-2 |
| [17] | Nordström EA, Teixeira C, Montelius C, et al. Lactiplantibacillus plantarum 299v (LP299V®): three decades of research[J]. Benef Microbes, 2021, 12(5): 441-65. doi:10.3920/bm2020.0191 |
| [18] | Lu CS, Chang HC, Weng YH, et al. The add-on effect of Lactobacillus plantarum PS128 in patients with Parkinson's disease: a pilot study[J]. Front Nutr, 2021, 8: 650053. doi:10.3389/fnut.2021.650053 |
| [19] | Liao JF, Cheng YF, You ST, et al. Lactobacillus plantarum PS128 alleviates neurodegenerative progression in 1-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine-induced mouse models of Parkinson's disease[J]. Brain Behav Immun, 2020, 90: 26-46. doi:10.1016/j.bbi.2020.07.036 |
| [20] | Beltrán-Velasco AI, Reiriz M, Uceda S, et al. Lactiplantibacillus (Lactobacillus) plantarum as a complementary treatment to improve symptomatology in neurodegenerative disease: a systematic review of open access literature[J]. Int J Mol Sci, 2024, 25(5): 3010. doi:10.3390/ijms25053010 |
| [21] | Wang L, Li SY, Jiang Y, et al. Neuroprotective effect of Lactobacillus plantarum DP189 on MPTP-induced Parkinson's disease model mice[J]. J Funct Foods, 2021, 85: 104635. doi:10.1016/j.jff.2021.104635 |
| [22] | Xu Z, Zhang J, Wu J, et al. Lactobacillus plantarum ST-III culture supernatant ameliorates alcohol-induced cognitive dysfunction by reducing endoplasmic reticulum stress and oxidative stress[J]. Front Neurosci, 2022, 16: 976358. doi:10.3389/fnins.2022.976358 |
| [23] | Zhai Q, Liu Y, Wang C, et al. Lactobacillus plantarum CCFM8661 modulates bile acid enterohepatic circulation and increases lead excretion in mice[J]. Food Funct, 2019, 10(3): 1455-64. doi:10.1039/c8fo02554a |
| [24] | Li YT, Liu AF, Chen LX, et al. Lactobacillus plantarum WSJ-06 alleviates neurobehavioral injury induced by lead in mice through the gut microbiota[J]. Food Chem Toxicol, 2022, 167: 113308. doi:10.1016/j.fct.2022.113308 |
| [25] | Gadaleta RM, Cariello M, Crudele L, et al. Bile salt hydrolase-competent probiotics in the management of IBD: unlocking the “bile acid code”[J]. Nutrients, 2022, 14(15): 3212. doi:10.3390/nu14153212 |
| [26] | Zhu J, Zhou F, Zhou Q, et al. NLRP3 activation in microglia contributes to learning and memory impairment induced by chronic lead exposure in mice[J]. Toxicol Sci, 2023, 191(1): 179-91. doi:10.1093/toxsci/kfac115 |
| [27] | Su P, Wang D, Cao Z, et al. The role of NLRP3 in lead-induced neuroinflammation and possible underlying mechanism[J]. Environ Pollut, 2021, 287: 117520. doi:10.1016/j.envpol.2021.117520 |
| [28] | Nagpal AG, Brodie SE. Supranormal electroretinogram in a 10-year-old girl with lead toxicity[J]. Documenta Ophthalmol, 2009, 118(2): 163-6. doi:10.1007/s10633-008-9144-7 |
| [29] | Lidsky TI, Schneider JS. Lead neurotoxicity in children: basic mechanisms and clinical correlates[J]. Brain, 2003, 126(pt 1): 5-19. doi:10.1093/brain/awg014 |
| [30] | Xiao J, Wang T, Xu Y, et al. Long-term probiotic intervention mitigates memory dysfunction through a novel H3K27me3-based mechanism in lead-exposed rats[J]. Transl Psychiatry, 2020, 10(1): 25. doi:10.1038/s41398-020-0719-8 |
| [31] | Tian FW, Zhai QX, Zhao JX, et al. Lactobacillus plantarum CCFM8661 alleviates lead toxicity in mice[J]. Biol Trace Elem Res, 2012, 150(1): 264-71. doi:10.1007/s12011-012-9462-1 |
| [32] | Hu SP, Tang B, Lu C, et al. Lactobacillus rhamnosus GG ameliorates triptolide-induced liver injury through modulation of the bile acid-FXR axis[J]. Pharmacol Res, 2024, 206: 107275. doi:10.1016/j.phrs.2024.107275 |
| [33] | McMillin M, Frampton G, Tobin R, et al. TGR5 signaling reduces neuroinflammation during hepatic encephalopathy[J]. J Neurochem, 2015, 135(3): 565-76. doi:10.1111/jnc.13243 |
| [34] | Yanguas-Casás N, Barreda-Manso MA, Nieto-Sampedro M, et al. TUDCA: an agonist of the bile acid receptor GPBAR1/TGR5 with anti-inflammatory effects in microglial cells[J]. J Cell Physiol, 2017, 232(8): 2231-45. doi:10.1002/jcp.25742 |
| [35] | Jena PK, Sheng L, Di Lucente J, et al. Dysregulated bile acid synthesis and dysbiosis are implicated in Western diet-induced systemic inflammation, microglial activation, and reduced neuroplasticity[J]. FASEB J, 2018, 32(5): 2866-77. doi:10.1096/fj.201700984rr |
| [1] | Aili YAN, Mengyao LUO, Jinrui CHANG, Xinhua LI, Juanxia ZHU. Hesperetin alleviates doxorubicin-induced cardiotoxicity by regulating the AMPK/NLRP3 pathway [J]. Journal of Southern Medical University, 2025, 45(9): 1850-1858. |
| [2] | Shanyu LUO, Qiang ZHU, Yufei YAN, Zonghong JI, Huajie ZOU, Ruixia ZHANG, Yinggui BA. NLRP3 signaling pathway promotes hepatocyte pyroptosis in mice with nonalcoholic steatohepatitis in hypoxic environment [J]. Journal of Southern Medical University, 2025, 45(9): 2026-2033. |
| [3] | Qingge WANG, Xiaohui ZHAO, Yuxuan HE, Feixiang LIU, Yunke ZHANG. Qixiong Zuogui Granules enhance synaptic plasticity in aging rats by regulating the BDNF/TrkB signaling pathway [J]. Journal of Southern Medical University, 2025, 45(8): 1589-1598. |
| [4] | Chuyu DENG, Xueying WANG, Lixiang GAN, Dayu WANG, Xiaoyan ZHENG, Chunzhi TANG. Electroacupuncture at Zusanli improves blood lipid disorders in hyperlipidemic mice by improving gut microbiota structure [J]. Journal of Southern Medical University, 2025, 45(8): 1633-1642. |
| [5] | Chenfei LIU, Wei ZHANG, Yao ZENG, Yan LIANG, Mengting WANG, Mingfang ZHANG, Xinyuan LI, Fengchao WANG, Yanqing YANG. 2,6-dimethoxy-1,4-benzoquinone alleviates dextran sulfate sodium-induced ulcerative colitis in mice by suppressing NLRP3 inflammasome activation [J]. Journal of Southern Medical University, 2025, 45(8): 1654-1662. |
| [6] | Feifei SHANG, Xiaoke SHI, Yao ZENG, Xunqian TAO, Tianzhen LI, Yan LIANG, Yanqin YANG, Chuanwang SONG. Avitinib suppresses NLRP3 inflammasome activation and ameliorates septic shock in mice [J]. Journal of Southern Medical University, 2025, 45(8): 1697-1705. |
| [7] | Haiyi ZHOU, Siyi HE, Ruifang HAN, Yongge GUAN, Lijuan DONG, Yang SONG. Moxibustion promotes endometrial repair in rats with thin endometrium by inhibiting the NLRP3/pyroptosis axis via upregulating miR-223-3p [J]. Journal of Southern Medical University, 2025, 45(7): 1380-1388. |
| [8] | Zhihua TIAN, Qingqing YANG, Xin CHEN, Fangfang ZHANG, Baimao ZHONG, Hong CAO. Spermine suppresses GBP5-mediated NLRP3 inflammasome activation in macrophages to relieve vital organ injuries in neonatal mice with enterovirus 71 infection [J]. Journal of Southern Medical University, 2025, 45(5): 901-910. |
| [9] | Fenlan BIAN, Shiyao NI, Peng ZHAO, Maonanxing QI, Bi TANG, Hongju WANG, Pinfang KANG, Jinjun LIU. Asiaticoside alleviates myocardial ischemia-reperfusion injury in rats by inhibiting NLRP3 inflammasome-mediated pyroptosis [J]. Journal of Southern Medical University, 2025, 45(5): 977-985. |
| [10] | Yalei SUN, Meng LUO, Changsheng GUO, Jing GAO, Kaiqi SU, Lidian CHEN, Xiaodong FENG. Amentoflavone alleviates acute lung injury in mice by inhibiting cell pyroptosis [J]. Journal of Southern Medical University, 2025, 45(4): 692-701. |
| [11] | Zhengwang ZHU, Linlin WANG, Jinghan ZHAO, Ruixue MA, Yuchun YU, Qingchun CAI, Bing WANG, Pingsheng ZHU, Mingsan MIAO. Tuihuang Mixture improves α‑naphthylisothiocyanate-induced cholestasis in rats by inhibiting NLRP3 inflammasomes via regulating farnesoid X receptor [J]. Journal of Southern Medical University, 2025, 45(4): 718-724. |
| [12] | Jingyu CHEN, Jinhu ZOU, Bingliang ZHOU, Xuefeng GAO, Pengwei HUANG, Hong CAO. Indole-3-acetic acid alleviates Cryptococcus neoformans-induced pyroptosis in cerebral microvascular endothelial cells by regulating stress granule-mediated NLRP3 inflammasome activation [J]. Journal of Southern Medical University, 2025, 45(12): 2679-2689. |
| [13] | Shuxian LIN, Lina GUO, Yan MA, Yao XIONG, Yingxi HE, Xinzhu XU, Wen SHENG, Suhua XU, Feng QIU. Lactobacillus plantarum ZG03 alleviates oxidative stress via its metabolites short-chain fatty acids [J]. Journal of Southern Medical University, 2025, 45(10): 2223-2230. |
| [14] | Mingyuan LI, Wei ZHANG, Mengqing HUA. Bardoxolone methyl alleviates acute liver injury in mice by inhibiting NLRP3 inflammasome activation [J]. Journal of Southern Medical University, 2024, 44(9): 1662-1669. |
| [15] | Huaixiang TAO, Jinguang LUO, Zhiyuan WEN, Genming YU, Xiao SU, Xinwei WANG, Han GUAN, Zhijun CHEN. High STING expression exacerbates renal ischemia-reperfusion injury in mice by regulating the TLR4/NF-κB/NLRP3 pathway and promoting inflammation and apoptosis [J]. Journal of Southern Medical University, 2024, 44(7): 1345-1354. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||