南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 803-815.doi: 10.12122/j.issn.1673-4254.2026.04.09
• • 上一篇
雷艳萍1( ), 宋嘉晟2, 徐乐吾2, 刘睿2, 赵岳2(
), 宋嘉晟2, 徐乐吾2, 刘睿2, 赵岳2( )
)
收稿日期:2025-10-26
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
赵岳
E-mail:leiyanping1988@163.com;1530718558@qq.com
作者简介:雷艳萍,博士,讲师,E-mail: leiyanping1988@163.com
基金资助:
Yanping LEI1( ), Jiasheng SONG2, Lewu XU2, Rui LIU2, Yue ZHAO2(
), Jiasheng SONG2, Lewu XU2, Rui LIU2, Yue ZHAO2( )
)
Received:2025-10-26
Online:2026-04-20
Published:2026-04-24
Contact:
Yue ZHAO
E-mail:leiyanping1988@163.com;1530718558@qq.com
摘要:
目的 探讨麦冬皂苷D(OD)是否通过调控β-catenin/FUNDC1/线粒体自噬信号轴减轻阿霉素(Dox)诱导的心肌肥厚。 方法 通过Dox诱导心肌肥厚,分组如下:对照组,Dox刺激组,Dox刺激+OD治疗组,Dox刺激+感染AAV-β-catenin组,Dox刺激+感染AAV(腺相关病毒载体)组。细胞RNA测序分析Dox刺激组与对照组基因表达差异;Western blotting检测β-catenin,活化β-catenin,FUNDC1,LC3,p62,β-MHC(β肌球蛋白重链),α-actin;免疫组化/荧光检测β-catenin,FUNDC1;透射电镜检测线粒体损伤;染色质免疫共沉淀(ChIP)及双荧光素酶报告基因检测β-catenin对FUNDC1的转录调节作用。 结果 相比对照组,Dox刺激显著抑制β-catenin信号(P<0.001)和FUNDC1介导的线粒体自噬(P<0.001),导致线粒体损伤和心肌细胞肥大(P<0.001)。相比Dox组,OD处理可逆转上述效应,恢复β-catenin信号(P<0.001),增强FUNDC1转录与表达(P<0.001),并促进线粒体自噬,减轻心肌肥厚(P<0.001)。过表达β-catenin或FUNDC1均模拟了OD的心肌保护作用,而敲低β-catenin则加重心肌肥厚,且FUNDC1过表达可逆转该表型。机制上,β-catenin直接结合FUNDC1启动子并激活其转录。 结论 OD通过激活β-catenin/FUNDC1/线粒体自噬轴,增强线粒体质量控制,从而缓解Dox诱导的心肌肥厚。
雷艳萍, 宋嘉晟, 徐乐吾, 刘睿, 赵岳. 麦冬皂苷D通过激活β-catenin/FUNDC1/线粒体自噬轴减轻阿霉素诱导的小鼠心肌肥厚[J]. 南方医科大学学报, 2026, 46(4): 803-815.
Yanping LEI, Jiasheng SONG, Lewu XU, Rui LIU, Yue ZHAO. Ophiopogonin D alleviates doxorubicin-induced myocardial hypertrophy in mice by activating the β-catenin/FUNDC1/mitophagy axis[J]. Journal of Southern Medical University, 2026, 46(4): 803-815.
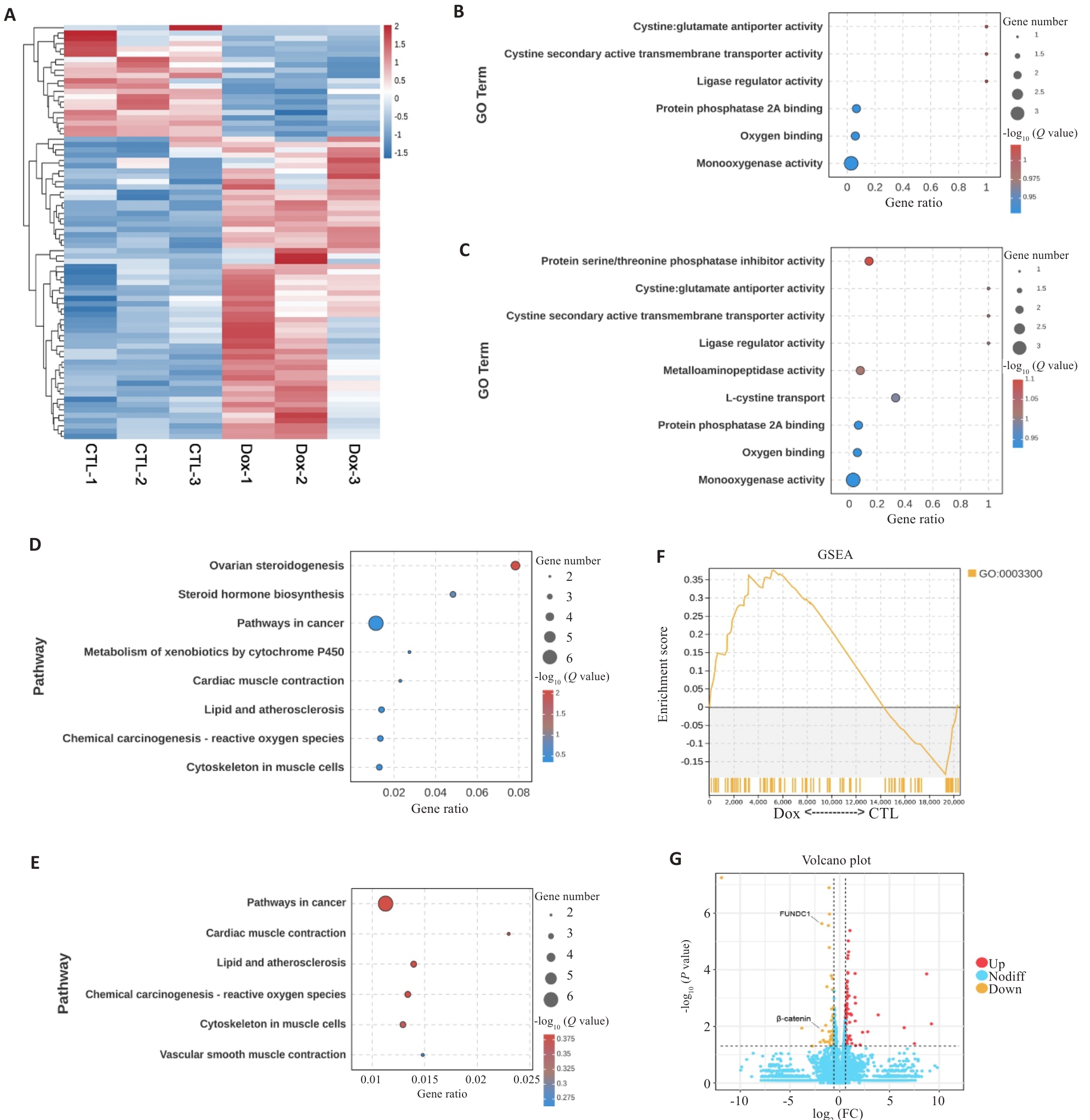
图1 Dox致心肌损伤与线粒体自噬及心肌肥大相关
Fig.1 Dox-induced myocardial injury is associated with mitophagy and myocardial hypertrophy. A: Heatmap of gene expression profiles in doxorubicin (Dox)-treated cardiomyocytes as compared with control (CTL) group. B: Bubble plot of Gene Ontology (GO) enrichment analysis for mitophagy-related genes. C: Bubble plot of GO enrichment analysis for myocardial hypertrophy-related genes. D: Bubble plot of Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis for mitophagy-related pathways. E: Bubble plot of KEGG enrichment analysis for myocardial hypertrophy-related pathways. F: Gene Set Enrichment Analysis (GSEA) plot for myocardial hypertrophy-related gene set (GO:0003300), showing enrichment scores and distribution of genes in Dox and CTL groups. G: Volcano plot showing differential mRNA expression profile in Dox and CTL groups.
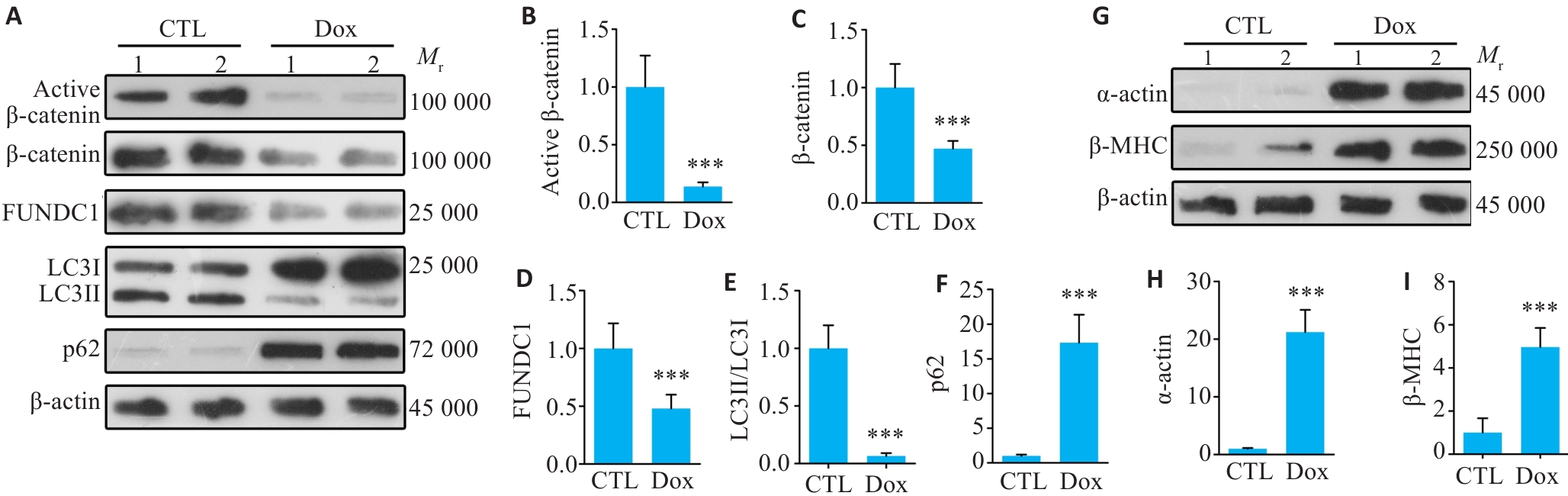
图2 Dox抑制心肌细胞中β-catenin信号及线粒体自噬, 促进心肌肥大
Fig.2 Dox inhibits β-catenin activation and mitochondrial autophagy in cardiomyocytes and promotes cardiomyocyte hypertrophy. A: Western blot bands of active β‑catenin, total β‑catenin, FUNDC1, LC3II/LC3I and p62 protein levels of CTL (control) and Dox (doxorubicin)-treated primary cardiomyocytes. Lanes 1 and 2 on the Western blotting indicated sample 1 and sample 2, respectively, within the group. B-F: Quantitative analysis of the protein levels (Mean±SD, n=6; ***P<0.001 vs CTL group). G-I: Western blot bands and quantitative analysis of α-actin (H) and β-MHC (I) protein levels in CTL and Dox-treated groups. Data are presented as Mean±SD (n=6). ***P<0.001 vs CTL group.
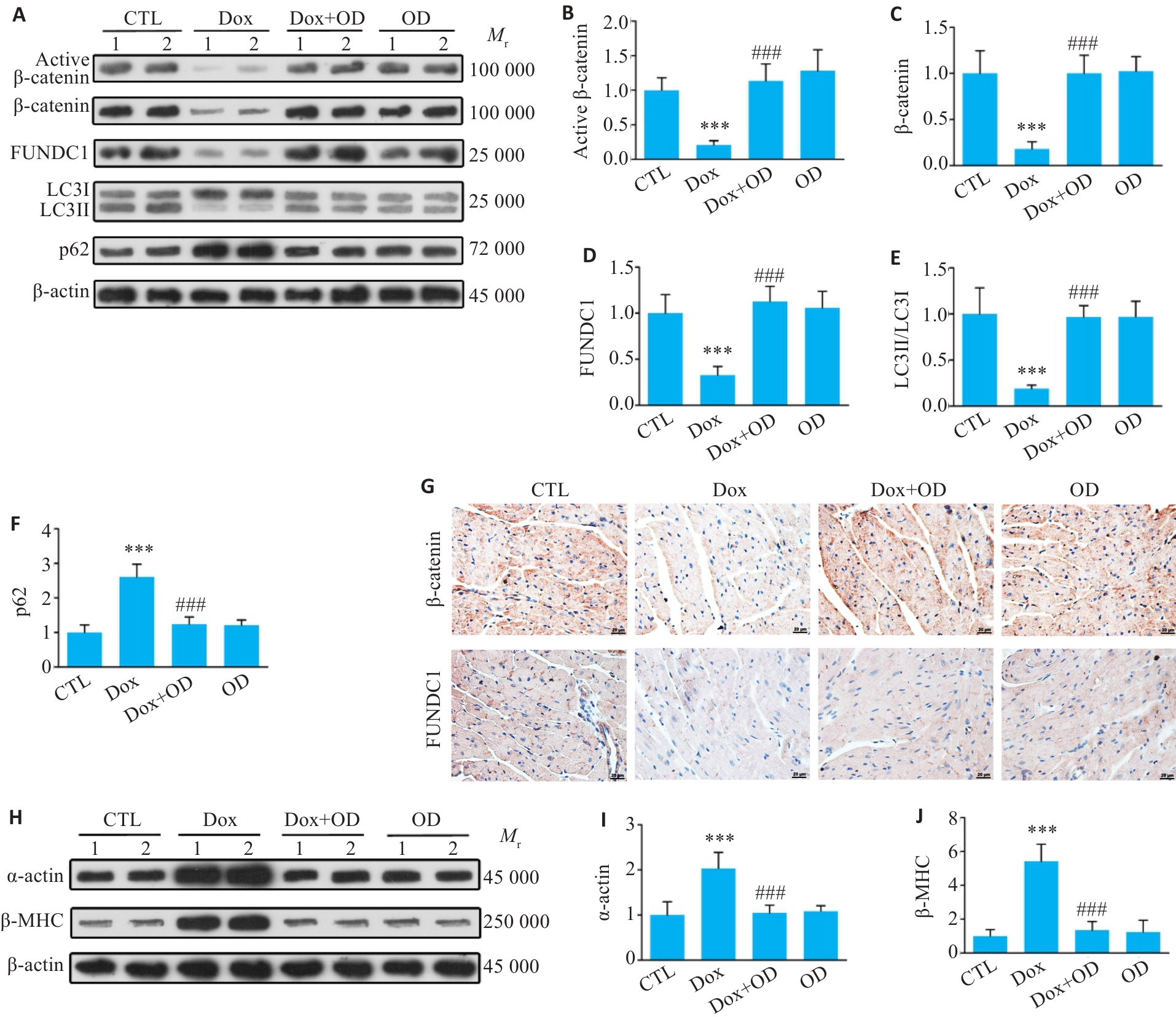
图3 OD激活β-catenin信号通路并增强线粒体自噬,缓解Dox诱导的心肌肥大
Fig.3 Ophiopogonin D (OD) activates β-catenin signaling and enhances mitophagy to improve Dox-induced cardiac hypertrophy. A: Western blotting bands of active β-catenin, total β-catenin, FUNDC1, LC3II/LC3I and p62 in the heart tissues of mice treated with Dox and OD. B-F: Quantitative analysis of the protein levels. G: Immunohistochemical staining for β‑catenin and FUNDC1 in the myocardial sections (Scale bar=20 μm). H-J: Western blotting bands and quantitative analysis of α-actin and β-MHC protein levels in the heart tissues of the mice in different groups. Data are presented as Mean±SD (n=6). ***P<0.001 vs CTL group; ###P<0.001 vs Dox group.
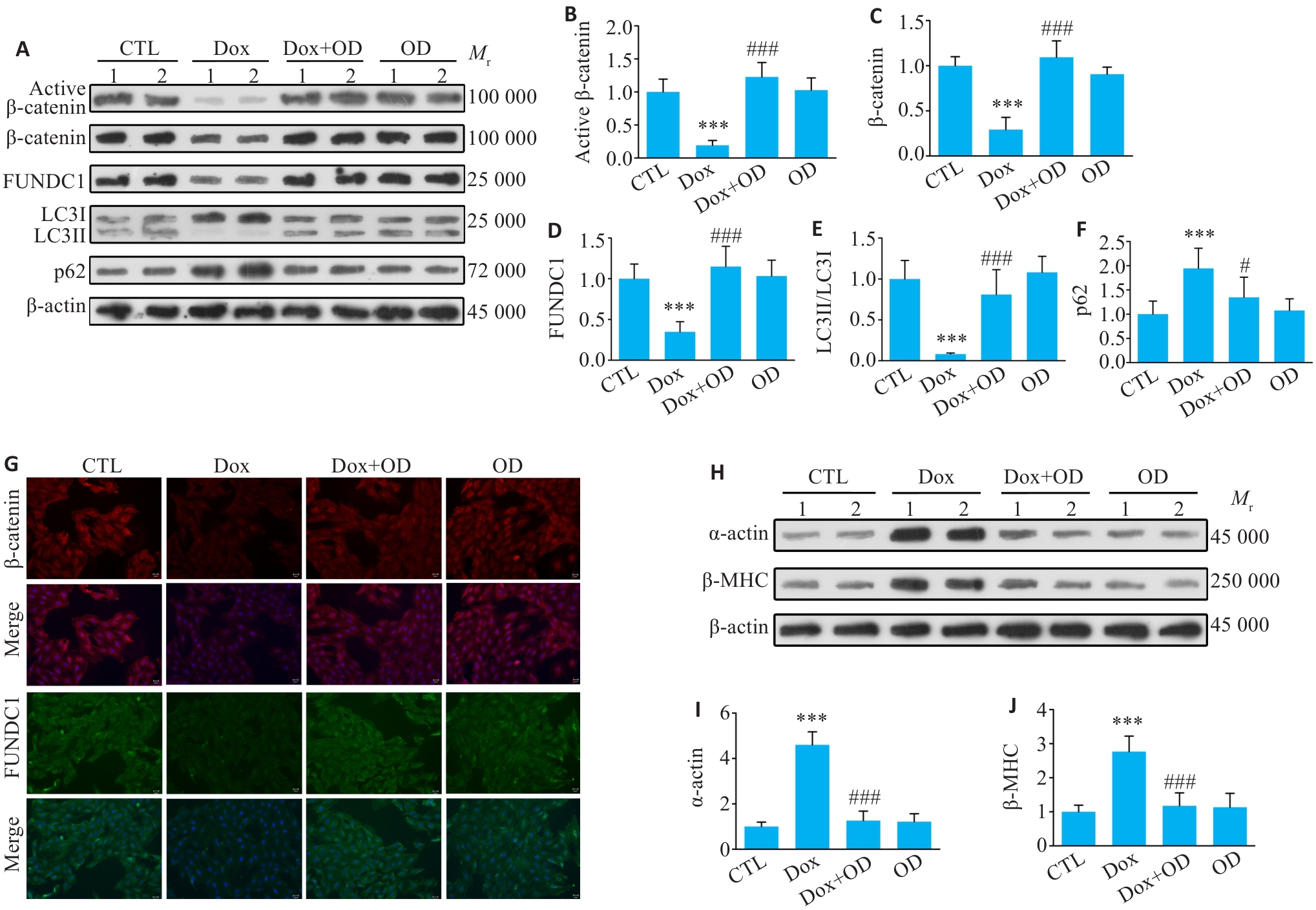
图4 OD激活β-catenin信号并增强线粒体自噬,对抗Dox诱导的心肌细胞肥大
Fig.4 OD activates β-catenin signaling and enhances mitophagy to counteract Dox-induced cardiomyocyte hypertrophy. A-F: Western blot bands of active β-catenin, total β-catenin, FUNDC1, LC3II/LC3I and p62 proteins and quantitative analysis of their expression levels in primary cardiomyocytes from different treatment groups. G: Immunofluorescence staining for β-catenin (red) and FUNDC1 (green) in the cells from each group (Scale bar=25 μm). The cell nuclei were stained with DAPI (blue). H-J: Western blotting bands of α-actin and β-MHC protein and their expression levels in the cells from each group. Data are presented as Mean±SD (n=6). ***P<0.001 vs CTL group; #P<0.05, ###P<0.001 vs Dox group.
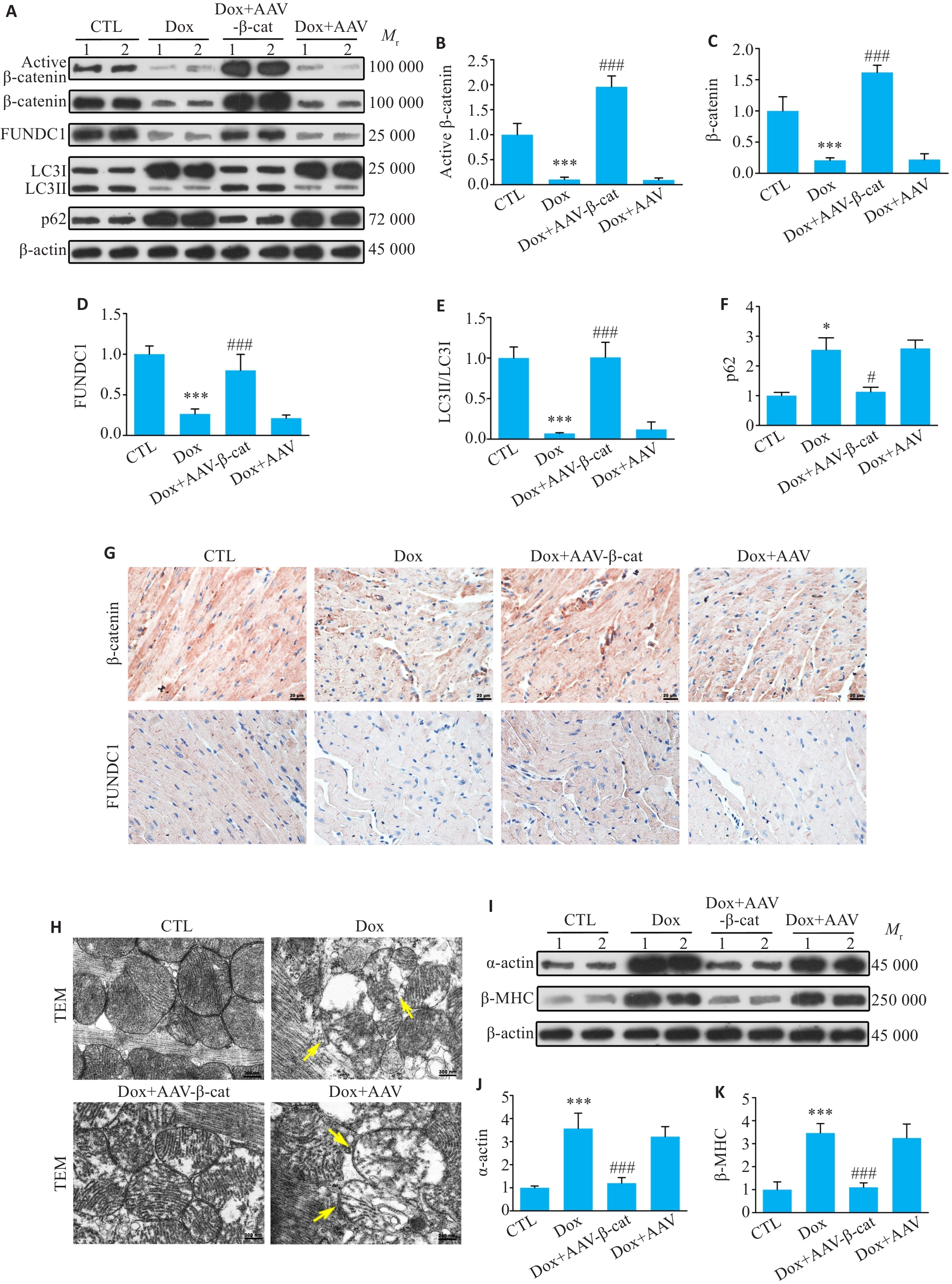
图5 体内实验显示心脏过表达β-catenin恢复线粒体自噬, 并改善Dox诱导的心肌肥厚
Fig.5 Overexpression of β-catenin in the heart restores mitophagy and alleviates Dox-induced myocardial hypertrophy in mice. A-F: Western blotting bands of activated β-catenin, total β-catenin, FUNDC1, LC3II/LC3I and p62 proteins and their expression levels in cardiac tissues of the mice in each group. G: Immunohistochemistry for β-catenin and FUNDC1 in the myocardial tissues in each group (Scale bar=20 μm). H: Transmission electron microscopy of the myocardial tissues in each group. Yellow arrows indicates damaged mitochondria (Scale bar=300 nm). I-K: Western blotting bands of α-actin and β-MHC proteins and their expression levels in each group. Data are presented as Mean±SD (n=6). *P<0.05, ***P<0.001 vs CTL group; #P<0.05, ###P<0.001 vs Dox group.
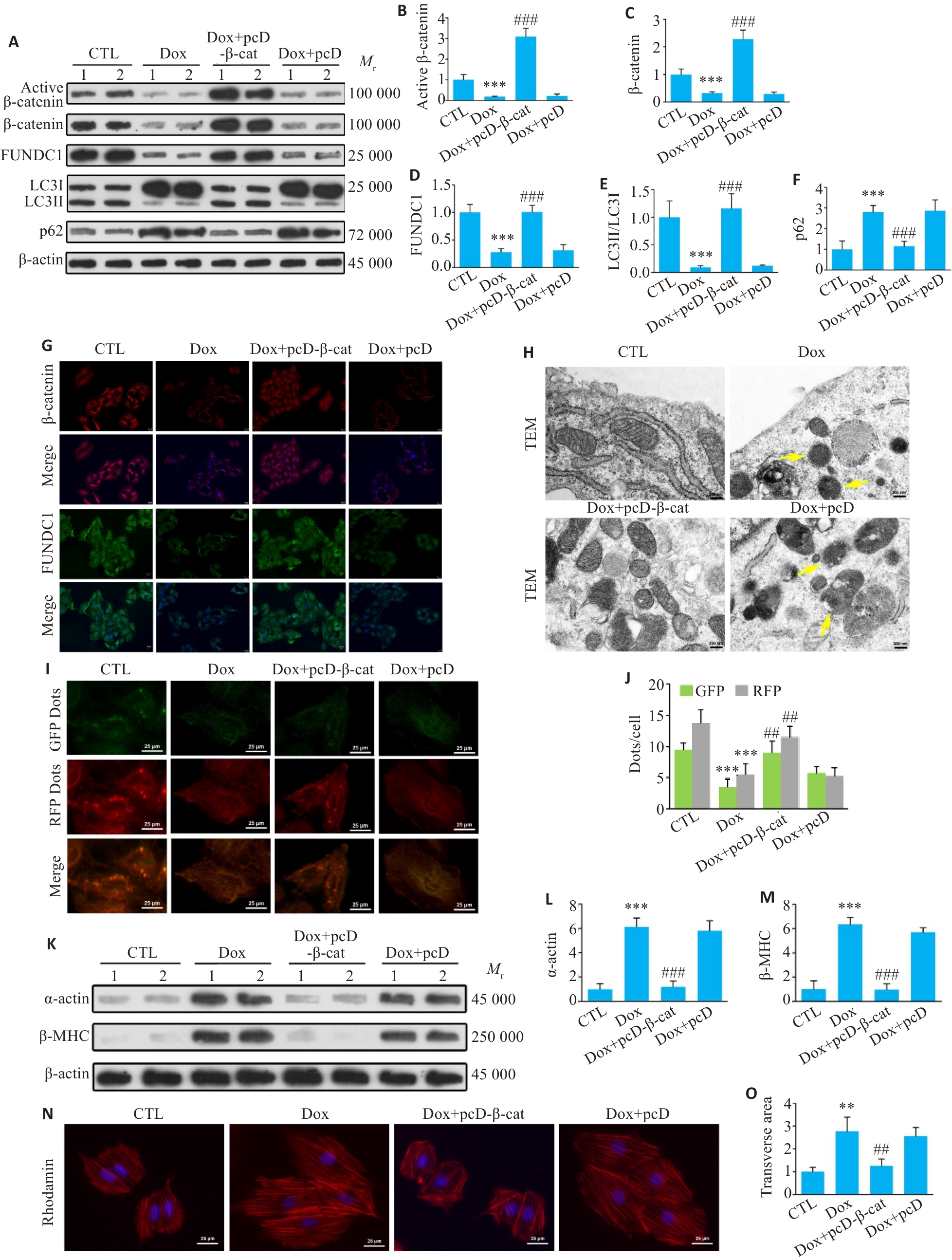
图6 体外实验显示, 心肌细胞内过表达β-catenin恢复线粒体自噬, 并改善Dox诱导的心肌细胞肥大
Fig.6 Cardiomyocyte-specific overexpression of β-catenin restores mitophagy and ameliorates Dox-induced hypertrophy in H9c2 cells. A-F: Western blotting bands of active β-catenin, total β-catenin, FUNDC1, LC3II/LC3I and p62 proteins and their expression levels in H9c2 cells in each group. G: Immunofluorescence staining for β-catenin and FUNDC1 in H9c2 cells (Scale bar=25 μm). H: Transmission electron microscopy of H9c2 cells in each group. Yellow arrows indicate damaged mitochondria (Scale bar=300 nm). I: Detection of autophagic flux in each group of cells using the mRFP-GFP-LC3 adenovirus probe (Scale bar=25 μm). Yellow spots represent autophagosomes (GFP+/mRFP+), and red spots represent autolysosomes (GFP-/mRFP+). J: Green and red fluorescent points in the indicated groups. K-M: Western blotting bands of α-actin and β-MHC proteins and their expression levels in each group of cells. N: Rhodamine staining of H9c2 cells and cross-sectional area of the cells in each group (×40). O: Statistical analysis of cross-sectional area of myocardial cells. Data are presented as Mean±SD (n=6).**P<0.01, ***P<0.001 vs CTL group; ##P<0.01, ###P<0.001 vs Dox group. pcD-β-cat: pcDNA-β-catenin; pcD: pcDNA.
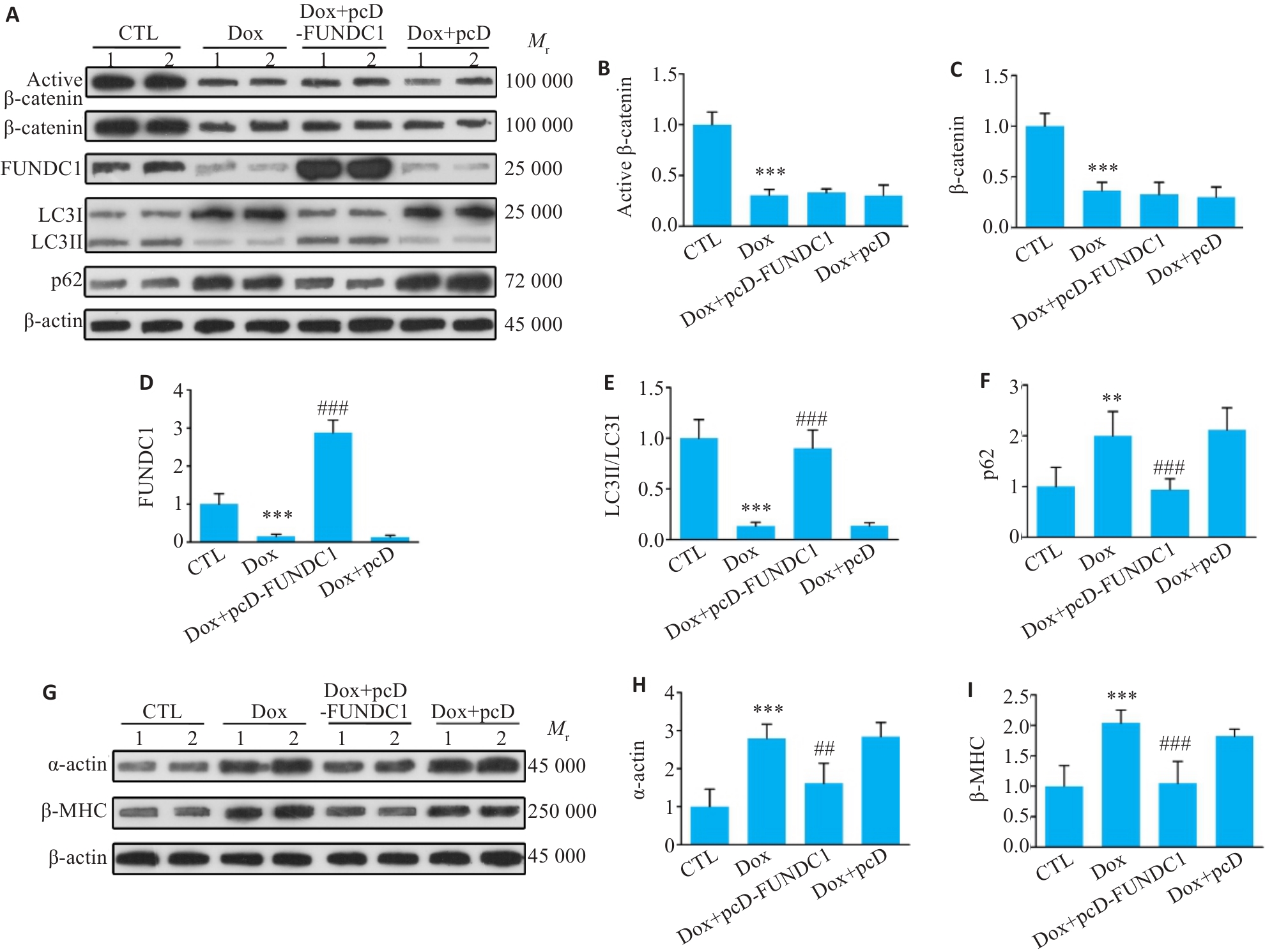
图7 过表达FUNDC1促进线粒体自噬,逆转Dox诱导的心肌细胞肥厚
Fig.7 Overexpression of FUNDC1 promotes mitophagy and reverses Dox-induced cardiomyocyte hypertrophy. A-F: Western blotting bands of active β-catenin, total β-catenin, FUNDC1, LC3II/LC3I and p62 proteins and their expression levels in H9c2 cells in different treatment groups. G-I: Western blotting bands of α-actin and β-MHC proteins and their expression levels in each group. Data are presented as Mean±SD (n=6). **P<0.01, ***P<0.001 vs CTL group; ##P<0.01, ###P<0.001 vs Dox group. pcD-FUNDC1: pcDNA-FUNDC1; pcD: pcDNA.
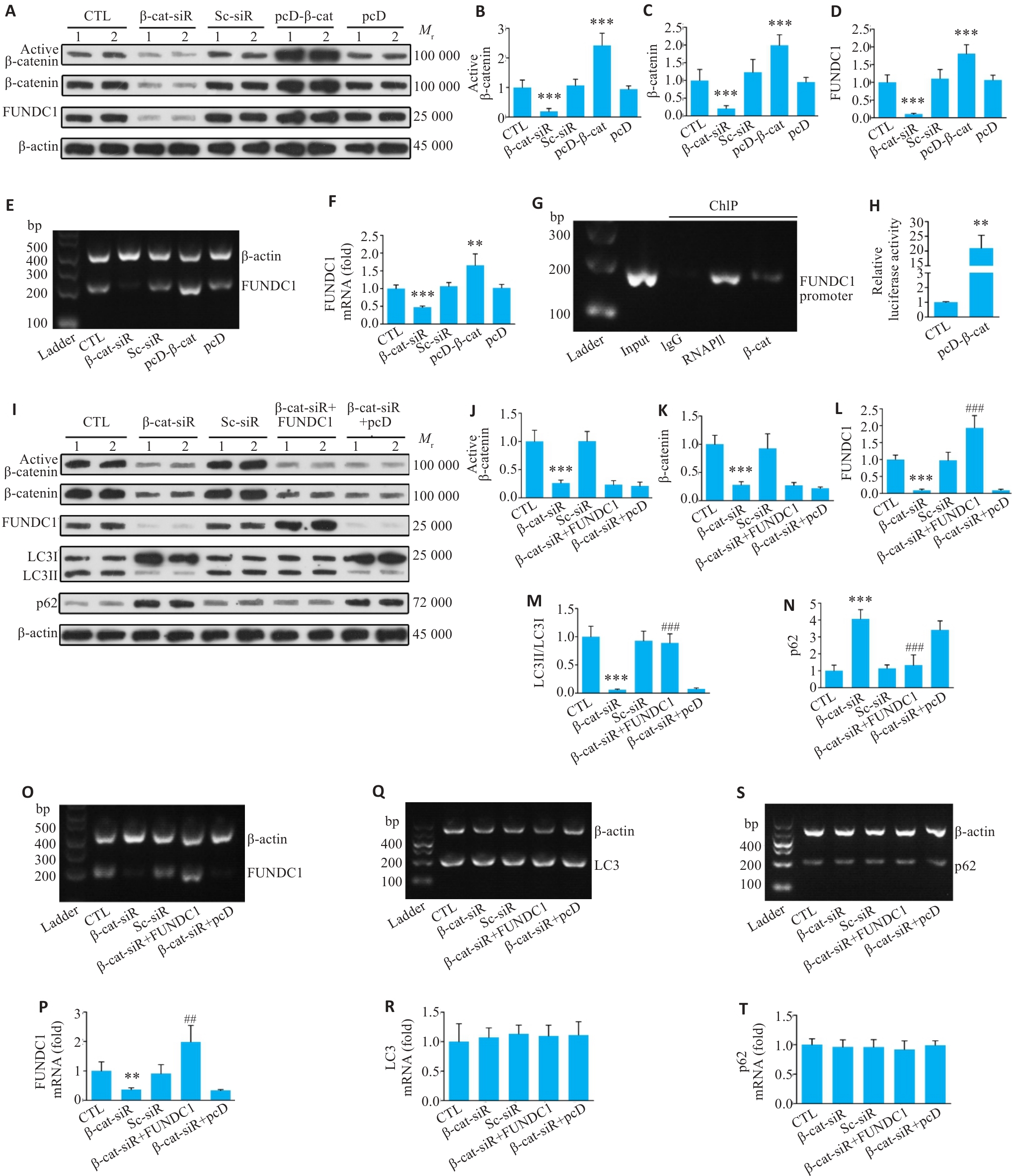
图8 β-catenin作为转录因子直接调控 FUNDC1 介导的线粒体自噬
Fig.8 β‑catenin, as a transcription factor, directly regulates FUNDC1-mediated mitophagy. A-D: Western blotting of active β-catenin, β‑catenin and FUNDC1 proteins in H9c2 transfected with β‑catenin-siRNA and pcDNA‑β‑catenin. E, F: RT-PCR for detection of FUNDC1 mRNA levels in each group of cells. G: Chromatin immunoprecipitation (ChIP) analysis showing the binding of β‑catenin protein to the promoter region of the FUNDC1 gene. H: Dual-luciferase reporter gene assay analysis showing that overexpression of β‑catenin significantly enhances transcriptional activity of the FUNDC1 promoter. I-N: Results of Western blotting of active β‑catenin, β‑catenin, FUNDC1, LC3II/LC3I and p62 protein levels in the cardiomyocytes with β‑catenin knockdown after overexpression of FUNDC1. O, P: RT-PCR for detection of FUNDC1 mRNA levels in each group. Q, R: RT-PCR for detecting LC3 mRNA levels in each group. S, T: RT-PCR for detecting p62 mRNA levels in each group. Data are presented as Mean±SD (n=6). **P<0.01, ***P<0.001 vs CTL group; ##P<0.01, ###P<0.001 vs β-catenin-siRNA group. β-cat-siR: β-catenin-siRNA; Sc-siR: Scramble-siRNA; pcD-β-cat: pcDNA-β-catenin; pcD: pcDNA; FUNDC1: pcDNA-FUNDC1.
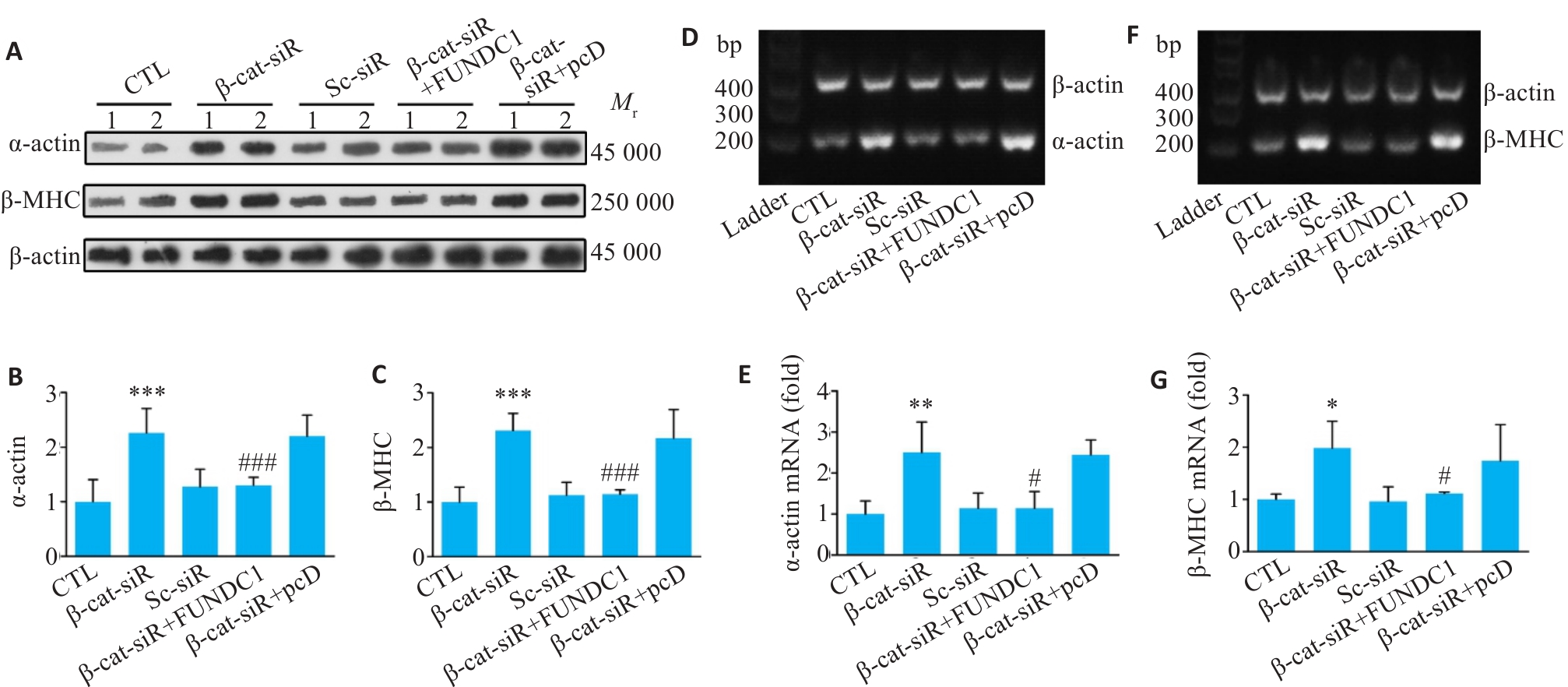
图9 β-catenin/FUNDC1 轴调控心肌细胞肥大
Fig.9 The β-catenin/FUNDC1 axis regulates cardiomyocyte hypertrophy. A-C: Western blotting of α-actin and β-MHC proteins in cardiomyocytes after overexpression of FUNDC1 on the basis of β-catenin knockdown. D, E: RT-PCR for detecting α-actin mRNA levels in each group of the cells. F, G: RT-PCR for detecting β-MHC mRNA levels in each group. Data are presented as Mean±SD (n=6).*P<0.05, **P<0.01, ***P<0.001 vs CTL group; #P<0.01, ###P<0.001 vs β-catenin-siRNA group.
| [1] | Nsairat H, Lafi Z, Abualsoud BM, et al. Vitamin C as a cardioprotective agent against doxorubicin-induced cardiotoxicity[J]. J Am Heart Assoc, 2025, 14(16): e042534. doi:10.1161/jaha.125.042534 |
| [2] | Zhang S, Xu HF, Zhang ZX, et al. Research on doxorubicin-induced cardiotoxicity mechanism and its forensic application[J]. Fa Yi Xue Za Zhi, 2025, 41(2): 120-6. |
| [3] | Goje ID, Goje GI, Ordodi VL, et al. Doxorubicin-induced cardiotoxicity and the emerging role of SGLT2 inhibitors: from glycemic control to cardio-oncology[J]. Pharmaceuticals (Basel), 2025, 18(5): 681. doi:10.3390/ph18050681 |
| [4] | Ito-Hagiwara K, Hagiwara J, Endo Y, et al. Cardioprotective strategies against doxorubicin-induced cardiotoxicity: a review from standard therapies to emerging mitochondrial transplantation[J]. Biomed Pharmacother, 2025, 189: 118315. doi:10.1016/j.biopha.2025.118315 |
| [5] | Yeh ETH, Chang HM. Anthracycline-induced cardiotoxicity: mechanism and prevention[J]. Trans Am Clin Climatol Assoc, 2025, 135: 184-95. |
| [6] | Chang ZS, Wang L, Zhou JX, et al. FoxO3 activation alleviates doxorubicin-induced cardiomyopathy by enhancing autophagic flux and suppressing mTOR/ROS signalling[J]. J Cell Mol Med, 2025, 29(15): e70775. doi:10.1111/jcmm.70775 |
| [7] | Liu PH, Xu TH, Luo YJ, et al. SIRT3 attenuates sepsis-induced EndMT and cardiac remodeling by facilitating mitophagy process via PINK1/Parkin signaling[J]. Int Immunopharmacol, 2025, 164: 115377. doi:10.1016/j.intimp.2025.115377 |
| [8] | Li SJ, Li XY. Mitophagy in hypertensive cardiac hypertrophy: mechanisms and therapeutic implications[J]. J Clin Hypertens (Greenwich), 2025, 27(8): e70127. doi:10.1111/jch.70127 |
| [9] | Shen Y, Gao X, Xiang Y, et al. Exploiting mitochondria by triggering a faulty unfolded protein response leads to effective cardioprotection[J]. Int J Med Sci, 2025, 22(1): 188-96. doi:10.7150/ijms.100523 |
| [10] | Wang J, Zhuang H, Jia L, et al. Nuclear receptor subfamily 4 group A member 1 promotes myocardial ischemia/reperfusion injury through inducing mitochondrial fission factor-mediated mitochondrial fragmentation and inhibiting FUN14 domain containing 1-depedent mitophagy[J]. Int J Biol Sci, 2024, 20(11): 4458-75. doi:10.7150/ijbs.95853 |
| [11] | Liu JQ, Xiao Q, Xiao JN, et al. Wnt/β-catenin signalling: function, biological mechanisms, and therapeutic opportunities[J]. Signal Transduct Target Ther, 2022, 7(1): 3. doi:10.1038/s41392-021-00762-6 |
| [12] | Lei YP, Hu HJ, Tang HF, et al. Homocysteine promotes cardiomyocyte hypertrophy through inhibiting β-catenin/FUNDC1 mediated mitophagy[J]. Sci Rep, 2025, 15(1): 22207. doi:10.1038/s41598-025-06772-6 |
| [13] | Xiang SL, Tang XW. Interfering with AQP1 alleviates ferroptosis, improves mitochondrial function and energy metabolic disorder in hypoxia/reoxygenation-induced H9c2 cardiomyocytes via Wnt/β-catenin pathway[J]. Microvasc Res, 2025, 160: 104821. doi:10.1016/j.mvr.2025.104821 |
| [14] | Deng YG, Hou MZ, Wu YB, et al. SIRT3-PINK1-PKM2 axis prevents osteoarthritis via mitochondrial renewal and metabolic switch[J]. Bone Res, 2025, 13(1): 36. doi:10.1038/s41413-025-00413-4 |
| [15] | Li HY, Leung JCK, Yiu WH, et al. Tubular β-catenin alleviates mitochondrial dysfunction and cell death in acute kidney injury[J]. Cell Death Dis, 2022, 13(12): 1061. doi:10.1038/s41419-022-05395-3 |
| [16] | Zhang YQ, Chen B, Zhang H, et al. Extraction, purification, structural characterization, bioactivities, modifications and structure-activity relationship of polysaccharides from Ophiopogon japonicus: a review[J]. Front Nutr, 2024, 11: 1484865. doi:10.3389/fnut.2024.1484865 |
| [17] | Chen YT, Ma LL, Yan YZ, et al. Ophiopogon japonicus polysaccharide reduces doxorubicin-induced myocardial ferroptosis injury by activating Nrf2/GPX4 signaling and alleviating iron accumulation[J]. Mol Med Rep, 2025, 31(2): 36. doi:10.3892/mmr.2024.13401 |
| [18] | Lei YP, Xu LW, Liu R, et al. Ophiopogonin D mitigates doxorubicin-induced cardiomyocyte ferroptosis through the β‑catenin/GPX4 pathway[J]. Front Pharmacol, 2025, 16: 1586937. doi:10.3389/fphar.2025.1586937 |
| [19] | Li XP, Luo WB, Tang Y, et al. Semaglutide attenuates doxorubicin-induced cardiotoxicity by ameliorating BNIP3-Mediated mitocho-ndrial dysfunction[J]. Redox Biol, 2024, 72: 103129. doi:10.1016/j.redox.2024.103129 |
| [20] | Wallace KB, Sardão VA, Oliveira PJ. Mitochondrial determinants of doxorubicin-induced cardiomyopathy[J]. Circ Res, 2020, 126(7): 926-41. doi:10.1161/circresaha.119.314681 |
| [21] | Liu XQ, Hussain R, Mehmood K, et al. Mitochondrial-endoplasmic reticulum communication-mediated oxidative stress and autophagy[J]. Biomed Res Int, 2022, 2022: 6459585. doi:10.1155/2022/6459585 |
| [22] | Liu RX, Xu CL, Zhang WL, et al. FUNDC1-mediated mitophagy and HIF1α activation drives pulmonary hypertension during hypoxia[J]. Cell Death Dis, 2022, 13(7): 634. doi:10.1038/s41419-022-05091-2 |
| [23] | He WB, Sun ZC, Tong G, et al. FUNDC1 alleviates doxorubicin-induced cardiotoxicity by restoring mitochondrial-endoplasmic reticulum contacts and blocked autophagic flux[J]. Theranostics, 2024, 14(9): 3719-38. doi:10.7150/thno.92771 |
| [24] | Haybar H, Khodadi E, Shahrabi S. Wnt/β‑catenin in ischemic myocardium: interactions and signaling pathways as a therapeutic target[J]. Heart Fail Rev, 2019, 24(3): 411-9. doi:10.1007/s10741-018-9759-z |
| [25] | Tao H, Yang JJ, Shi KH, et al. Wnt signaling pathway in cardiac fibrosis: New insights and directions[J]. Metabolism, 2016, 65(2): 30-40. doi:10.1016/j.metabol.2015.10.013 |
| [26] | Wang XY, He K, Ma LL, et al. Puerarin attenuates isoproterenol-induced myocardial hypertrophy via inhibition of the Wnt/β-catenin signaling pathway[J]. Mol Med Rep, 2022, 26(4): 306. doi:10.3892/mmr.2022.12822 |
| [27] | Huo PP, Wang SJ, Li ZN, et al. ACSS3 protein macromolecule regulates glycolysis in keloid through Wnt/β‑catenin signaling pathway: Bioinformatics, machine learning, and experimental validation[J]. Cell Signal, 2025, 135: 112056. doi:10.1016/j.cellsig.2025.112056 |
| [28] | Zhang HY, Kang XZ, Ruan J, et al. Ophiopogonin D improves oxidative stress and mitochondrial dysfunction in pancreatic β cells induced by hydrogen peroxide through Keap1/Nrf2/ARE pathway in diabetes mellitus[J]. Chin J Physiol, 2023, 66(6): 494-502. doi:10.4103/cjop.cjop-d-23-00069 |
| [29] | Huang XY, Wang YG, Wang Y, et al. Ophiopogonin D reduces myocardial ischemia-reperfusion injury via upregulating CYP2J3/EETs in rats[J]. Cell Physiol Biochem, 2018, 49(4): 1646-58. doi:10.1159/000493500 |
| [30] | Zhang YY, Meng C, Zhang XM, et al. Ophiopogonin D attenuates doxorubicin-induced autophagic cell death by relieving mitochondrial damage in vitro and in vivo [J]. J Pharmacol Exp Ther, 2015, 352(1): 166-74. doi:10.1124/jpet.114.219261 |
| [1] | 张国栋, 魏思昂, 王弘, 谢艳丽, 何晖, 李嵘. 有氧运动通过氧化应激-炎症-Hippo/YAP通路轴改善心力衰竭小鼠心功能[J]. 南方医科大学学报, 2026, 46(4): 753-760. |
| [2] | 梁芷晴, 潘富珍, 邓利强, 麦哲芬, 马云, 施传坚, 付卫明. 生脉散通过调控乳酸/Wnt/β-catenin/LDHA通路改善非小细胞肺癌奥希替尼耐药的作用机制[J]. 南方医科大学学报, 2026, 46(3): 523-531. |
| [3] | 林心君, 何昱霖, 施红, 刘佳绣, 胡海霞. 石斛合剂通过调控Sirt3介导的线粒体自噬通路缓解大鼠糖尿病心肌病[J]. 南方医科大学学报, 2026, 46(1): 47-54. |
| [4] | 闫爱丽, 罗梦瑶, 常晋瑞, 李新华, 朱娟霞. 橙皮素通过调控AMPK/NLRP3通路减轻阿霉素诱导的小鼠心肌毒性[J]. 南方医科大学学报, 2025, 45(9): 1850-1858. |
| [5] | 云琦, 杜若丽, 贺玉莹, 张贻欣, 王佳慧, 叶红伟, 李正红, 高琴. 肉桂酸通过抑制TLR4减轻阿霉素诱导的小鼠心肌损伤铁死亡的发生[J]. 南方医科大学学报, 2025, 45(9): 1946-1958. |
| [6] | 莫艳秀, 舒洋, 莫钰兰, 刘峻彤, 徐欧欧, 邓华菲, 王岐本. 敲除CDC20可明显抑制宫颈癌细胞的增殖及侵袭转移[J]. 南方医科大学学报, 2025, 45(6): 1200-1211. |
| [7] | 蔡蕊, 黄卓, 贺文霞, 艾添红, 宋晓伟, 胡淑婷. 剪接因子HNRNPH1通过调控Circ-MYOCD的反向剪接影响心肌肥厚的发生[J]. 南方医科大学学报, 2025, 45(3): 587-594. |
| [8] | 廖茗, 钟文华, 张冉, 梁娟, 徐文陶睿, 万文珺, 吴超, 李曙. 源自蛇毒的蛋白C激活剂通过调控HIF-1α抑制BNIP3活性氧生成保护人脐静脉内皮细胞免受缺氧-复氧损伤[J]. 南方医科大学学报, 2025, 45(3): 614-621. |
| [9] | 陈镝, 吕莹, 郭怡欣, 张怡荣, 王蕊璇, 周小若, 陈雨欣, 武晓慧. 双氢青蒿素可显著增强阿霉素诱导的三阴性乳腺癌细胞凋亡:基于负向调控STAT3/HIF-1α通路[J]. 南方医科大学学报, 2025, 45(2): 254-260. |
| [10] | 夏士程, 韦慧芳, 洪维灿, 张钰明, 尹菲玚, 张贻欣, 张淋淋, 高琴, 叶红伟. 金银花提取物对阿霉素诱导的小鼠心肌损伤的保护作用及其机制[J]. 南方医科大学学报, 2025, 45(12): 2527-2540. |
| [11] | 宋博文, 周仁杰, 徐盈, 施金冉, 张志郅, 李静, 耿志军, 宋雪, 王炼, 王月月, 左芦根. TMCO1在胃癌中高表达与患者不良预后相关并通过抑制调亡促进肿瘤恶性进展[J]. 南方医科大学学报, 2025, 45(11): 2385-2393. |
| [12] | 姜雪凝, 黄晴晴, 徐盈, 王舜印, 张小凤, 王炼, 王月月, 左芦根. 高表达YEATS2通过激活Wnt/β-catenin通路促进胃癌细胞上皮-间质转化进程[J]. 南方医科大学学报, 2025, 45(11): 2416-2426. |
| [13] | 易明, 罗烨, 吴露, 吴泽衡, 蒋翠平, 陈史钰, 柯晓. 黄芪甲苷通过抑制PINK1/Parkin通路调控细胞线粒体自噬减轻D-半乳糖诱导的内皮细胞衰老[J]. 南方医科大学学报, 2025, 45(11): 2427-2436. |
| [14] | 郭克磊, 李颖利, 宣晨光, 侯紫君, 叶松山, 李林运, 陈丽平, 韩立, 卞华. 益气养阴化浊通络方通过调控miR-21a-5p/FoxO1/PINK1介导的线粒体自噬减轻糖尿病肾病小鼠的足细胞损伤[J]. 南方医科大学学报, 2025, 45(1): 27-34. |
| [15] | 展俊平, 黄硕, 孟庆良, 范围, 谷慧敏, 崔家康, 王慧莲. 缺氧微环境下补阳还五汤通过抑制BNIP3-PI3K/Akt通路抑制类风湿关节炎滑膜成纤维细胞的线粒体自噬[J]. 南方医科大学学报, 2025, 45(1): 35-42. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||