南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 753-760.doi: 10.12122/j.issn.1673-4254.2026.04.04
• • 上一篇
张国栋1( ), 魏思昂2,3, 王弘1,2, 谢艳丽1, 何晖1, 李嵘1(
), 魏思昂2,3, 王弘1,2, 谢艳丽1, 何晖1, 李嵘1( )
)
收稿日期:2025-10-01
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
李嵘
E-mail:610089513@qq.com;liliu990316@163.com
作者简介:张国栋,博士,副教授, E-mail: 610089513@qq.com
基金资助:
Guodong ZHANG1( ), Siang WEI2,3, Hong WANG1,2, Yanli XIE1, Hui HE1, Rong LI1(
), Siang WEI2,3, Hong WANG1,2, Yanli XIE1, Hui HE1, Rong LI1( )
)
Received:2025-10-01
Online:2026-04-20
Published:2026-04-24
Contact:
Rong LI
E-mail:610089513@qq.com;liliu990316@163.com
Supported by:摘要:
目的 探讨有氧运动通过调控氧化应激、炎症与Hippo-YAP信号通路改善主动脉弓缩窄(TAC)诱导心力衰竭的作用机制。 方法 30只C57BL/6J雄性小鼠随机分为假手术组(Sham)、TAC模型组(TAC)及TAC+有氧运动组(TACE),10只/组。TACE组进行4周跑台有氧训练(强度递增)。干预后采用超声心动图评估心功能,Masson染色观察心肌纤维化,ELISA检测血清BNP、炎症因子(TNF-α、IL-6、IL-1β)及氧化应激指标(MDA、SOD、GSH-Px)。RT‑PCR与Western blotting分别检测心肌组织Hippo‑YAP通路相关基因及蛋白表达。通过生物信息学分析各指标相关性。 结果 与TAC组相比,TACE组心脏质量与心质量指数降低(P<0.01),左心室射血分数(LVEF)、左心室缩短分数(LVFS)及E/A比值升高(P<0.05),心肌纤维化减轻(P<0.05),血清与心肌BNP表达下降(P<0.05)。TACE组血清TNF‑α、IL‑6、IL‑1β及MDA水平降低(P<0.05),SOD与GSH‑Px活性升高(P<0.05)。RT‑PCR结果显示,TACE组心肌Mst1、Lats1、Lats2及YAP1 mRNA表达较TAC组下调(P<0.05)。Western blotting结果显示,TACE组心肌Mst1/2、Lats1/2及YAP总蛋白表达降低,而p‑Lats1/2与p‑YAP水平升高(P<0.05)。相关性分析提示氧化应激、炎症指标与Hippo‑YAP通路表达显著相关。 结论 有氧运动可改善TAC诱导的心力衰竭小鼠心功能,减轻心肌肥厚与纤维化,其保护作用可能与抑制氧化应激、炎症反应及调控Hippo‑YAP信号通路有关。
张国栋, 魏思昂, 王弘, 谢艳丽, 何晖, 李嵘. 有氧运动通过氧化应激-炎症-Hippo/YAP通路轴改善心力衰竭小鼠心功能[J]. 南方医科大学学报, 2026, 46(4): 753-760.
Guodong ZHANG, Siang WEI, Hong WANG, Yanli XIE, Hui HE, Rong LI. Aerobic exercise produces cardioprotective effects in mice by regulating the oxidative stress-inflammation-Hippo/YAP signaling axis[J]. Journal of Southern Medical University, 2026, 46(4): 753-760.
| Gene name | Forward primer | Reverse primer |
|---|---|---|
| BNP | GAGGTCACTCCTATCCTCTGG | GCCATTTCCTCCGACTTTTCTC |
| Mst1 | CTCACCACTGAATGACTTCCAG | AAGGCCCGACAGTCCAGAA |
| Last1 | AAAGCCAGAAGGGTACAGACA | CCTCAGGGATTCTCGGATCTC |
| Lats2 | GGACCCCAGGAATGAGCAG | CCCTCGTAGTTTGCACCACC |
| YAP1 | ACCCTCGTTTTGCCATGAAC | TGTGCTGGGATTGATATTCCGTA |
| GAPDH | TGGCCTTCCGTGTTCCTAC | GAGTTGCTGTTGAAGTCGCA |
表1 各基因的引物序列
Tab.1 Primer sequences for RT-PCR
| Gene name | Forward primer | Reverse primer |
|---|---|---|
| BNP | GAGGTCACTCCTATCCTCTGG | GCCATTTCCTCCGACTTTTCTC |
| Mst1 | CTCACCACTGAATGACTTCCAG | AAGGCCCGACAGTCCAGAA |
| Last1 | AAAGCCAGAAGGGTACAGACA | CCTCAGGGATTCTCGGATCTC |
| Lats2 | GGACCCCAGGAATGAGCAG | CCCTCGTAGTTTGCACCACC |
| YAP1 | ACCCTCGTTTTGCCATGAAC | TGTGCTGGGATTGATATTCCGTA |
| GAPDH | TGGCCTTCCGTGTTCCTAC | GAGTTGCTGTTGAAGTCGCA |
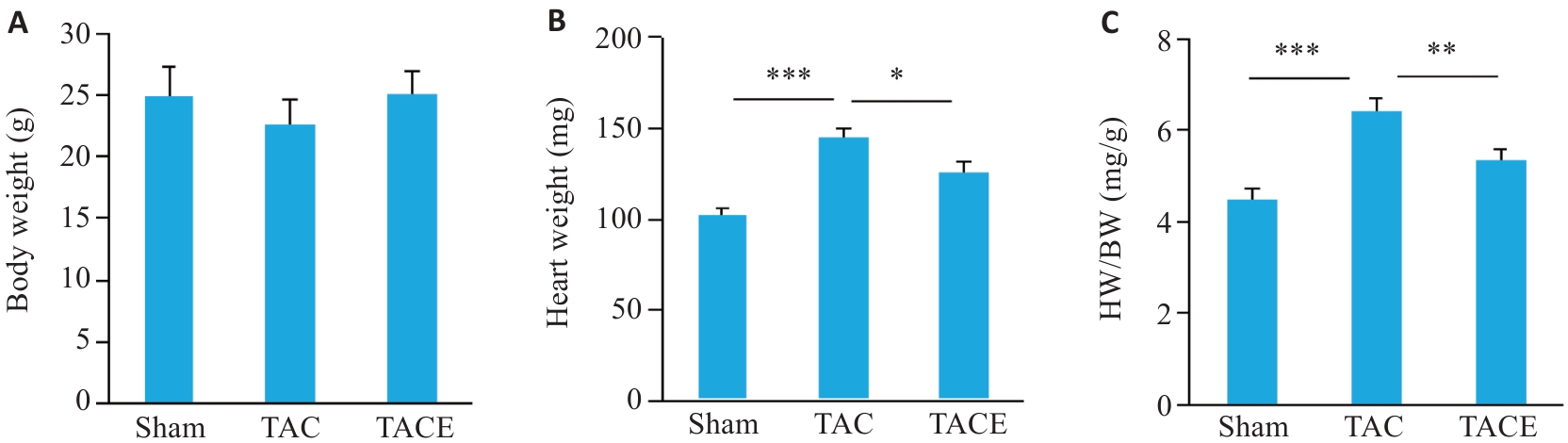
图1 有氧运动对TAC 手术小鼠体质量、心脏质量、心脏质量/体质量比的影响
Fig.1 Effects of aerobic exercise on body weight (A), heart weight (B), and HW/BW ratio (C) in mice following transverse aortic constriction (TAC) surgery (Mean±SD, n=10). *P<0.05, **P<0.01, ***P<0.001.
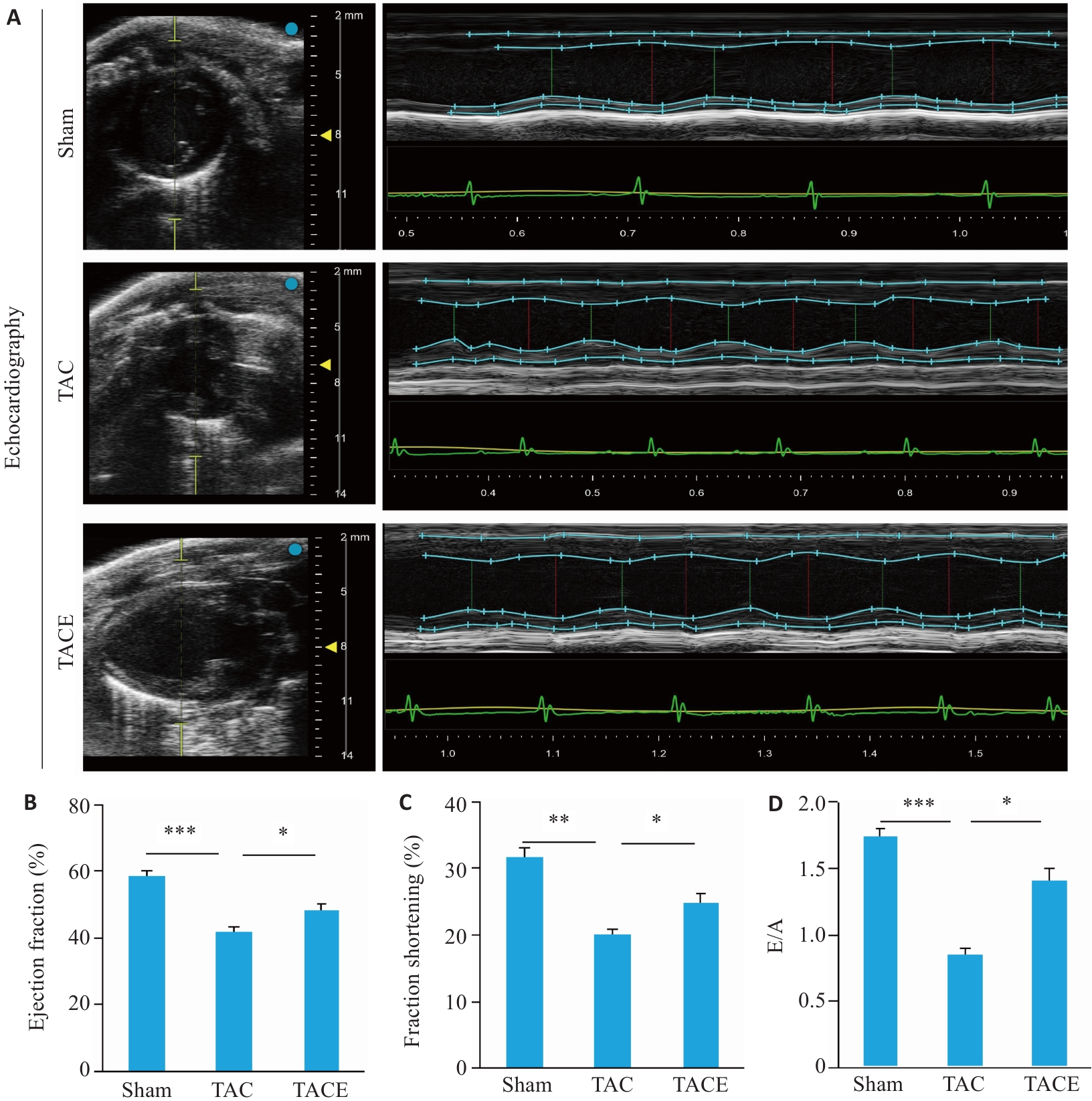
图2 有氧运动对TAC手术小鼠心脏功能的影响
Fig.2 Effects of aerobic exercise on cardiac function in TAC mice (Mean±SD, n=10). A: Echocardiography. B: Ejection fraction (%). C: Fraction shortening (%). D: E/A ratio. *P<0.05, **P<0.01, ***P<0.001.
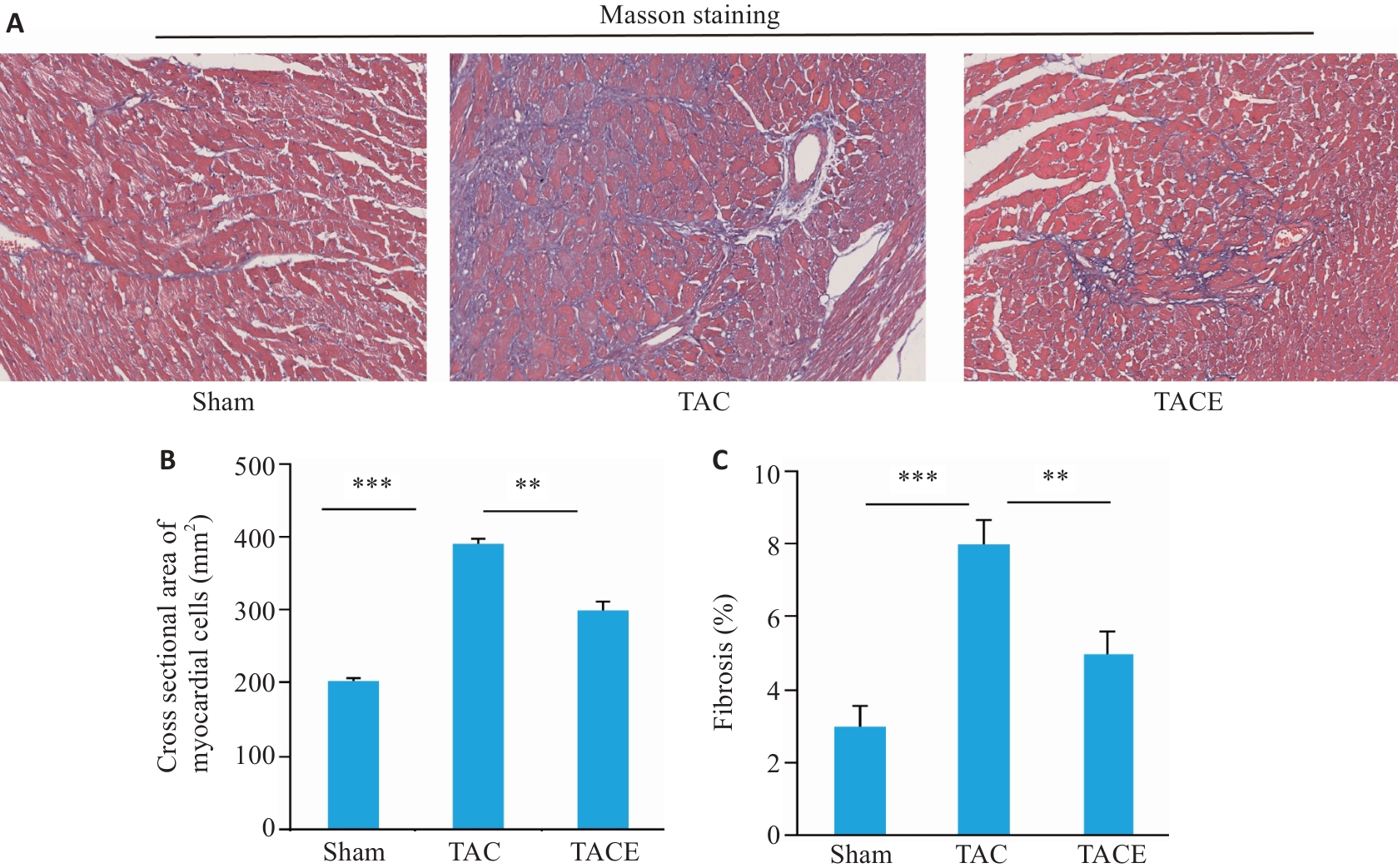
图3 有氧运动对 TAC 手术小鼠心肌细胞横截面积、纤维化的影响
Fig.3 Effects of aerobic exercise on cross sectional area and fibrosis of cardiomyocytes in TAC mice (Mean±SD, n=10). A: Mass staining of the myocardium in each group (Original magnification: ×200). B: Cross sectional areas of myocardial cells in each group. C: Percentage of cardiac fibrosis (%) in each group. **P<0.01, ***P<0.001.
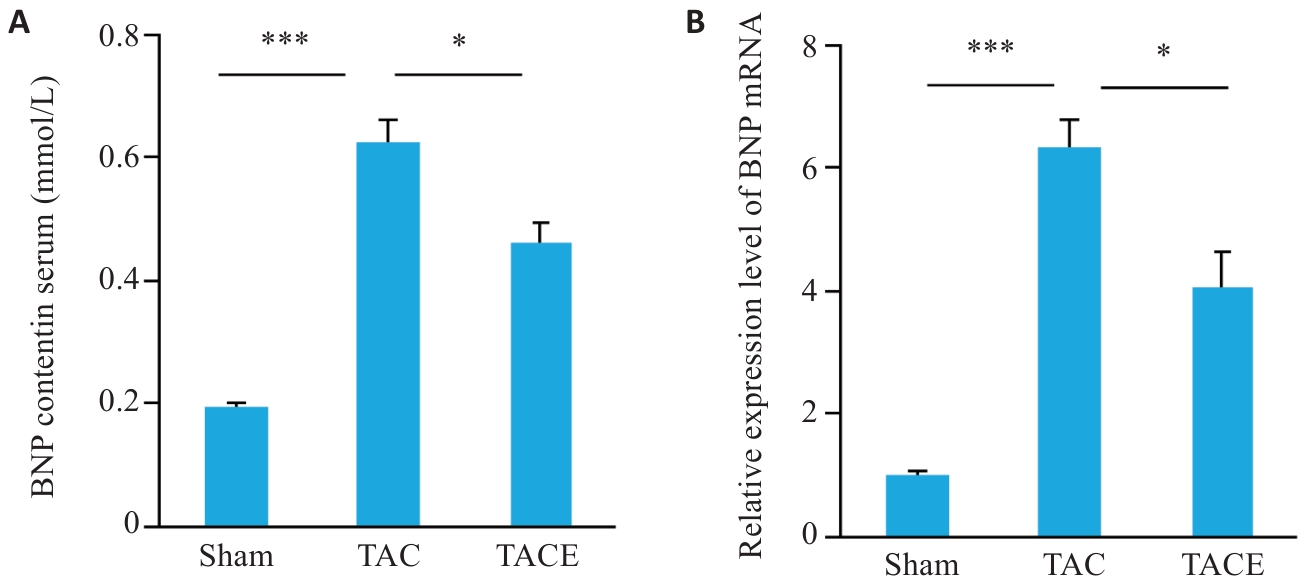
图4 有氧运动对TAC小鼠BNP变化的结果
Fig.4 Effect of aerobic exercise on BNP changes in TAC mice (Mean±SD, n=10). A: Serum BNP content. B: Relative BNP mRNA expression levels in the heart. *P<0.05, ***P<0.001.
| Group | MDA (ng/mL) | SOD (pg/mL) | GSH-Px (μg/mL) |
|---|---|---|---|
| Sham | 7.93±0. 54 | 161.97±11.34 | 10.72±0.87 |
| TAC | 15.35±0.83* | 92.68±8.71* | 4.27±0.57* |
| TACE | 10.37±0.62*# | 135.41±9.38*# | 9.24±0.92# |
表2 各组小鼠血清 MDA, SOD 和 GSH-Px 的水平
Tab.2 Levels of MDA, SOD, and GSH-Px in serum of the mice in each group (Mean±SD, n=10)
| Group | MDA (ng/mL) | SOD (pg/mL) | GSH-Px (μg/mL) |
|---|---|---|---|
| Sham | 7.93±0. 54 | 161.97±11.34 | 10.72±0.87 |
| TAC | 15.35±0.83* | 92.68±8.71* | 4.27±0.57* |
| TACE | 10.37±0.62*# | 135.41±9.38*# | 9.24±0.92# |
| Group | TNF-α (pg/mL) | IL-6 (pg/mL) | IL-1β (pg/mL) |
|---|---|---|---|
| Sham | 9.27±0.82 | 19.37±1.27 | 79.61±4.89 |
| TAC | 36.78±2.34* | 68.46±3.09* | 164.97±13.28* |
| TACE | 18.69±1.02*# | 35.34±2.56*# | 134.26±9.63*# |
表3 各组小鼠血清 TNF-α, IL-1β 和 IL-6 的表达水平
Tab.3 Serum levels of TNF-α, IL-1β, and IL-6 in the mice in each group (Mean±SD, n=10)
| Group | TNF-α (pg/mL) | IL-6 (pg/mL) | IL-1β (pg/mL) |
|---|---|---|---|
| Sham | 9.27±0.82 | 19.37±1.27 | 79.61±4.89 |
| TAC | 36.78±2.34* | 68.46±3.09* | 164.97±13.28* |
| TACE | 18.69±1.02*# | 35.34±2.56*# | 134.26±9.63*# |
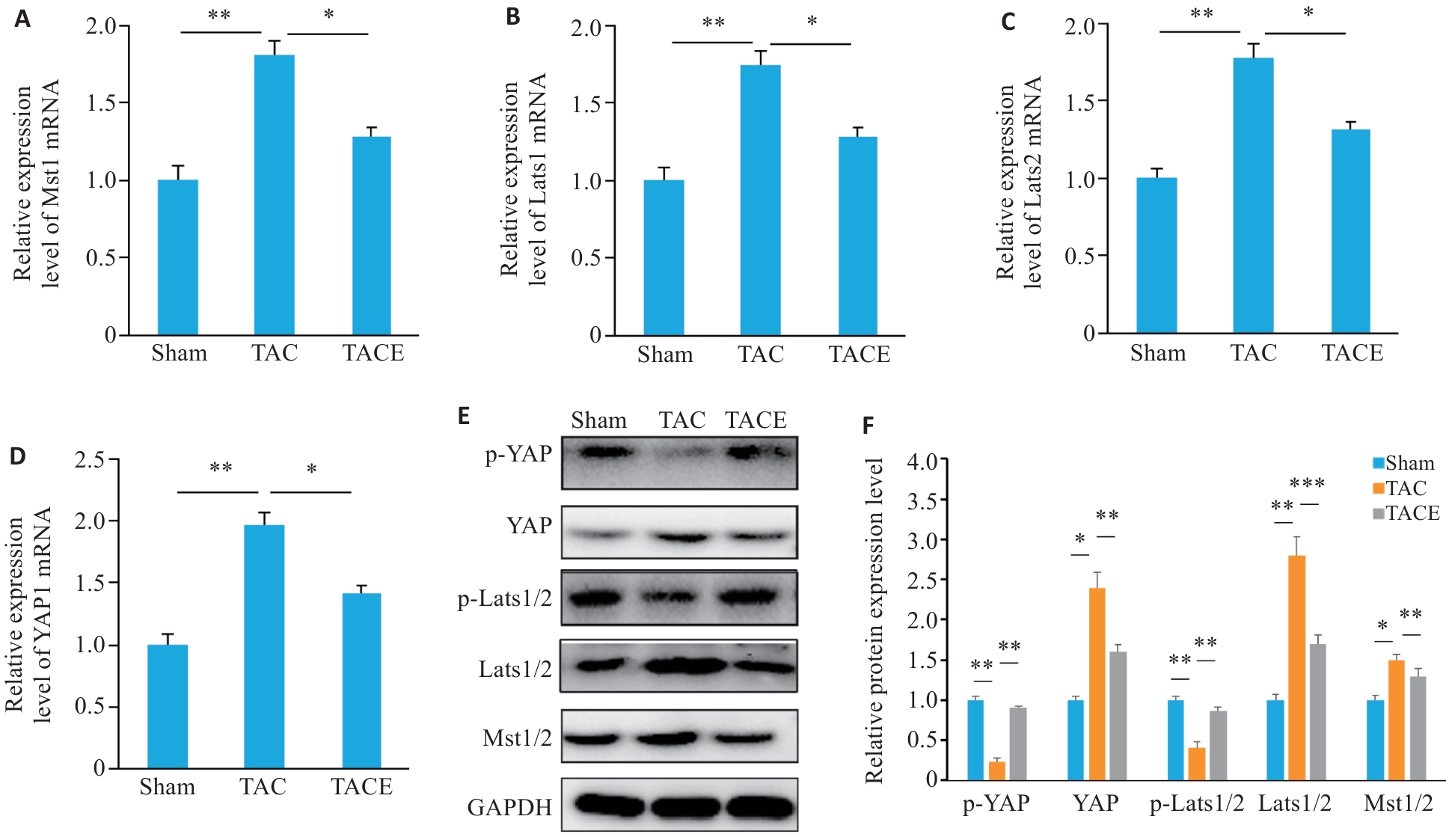
图5 有氧运动对TAC小鼠心肌组织中Hippo-YAP信号通路蛋白表达水平的影响
Fig.5 Effects of aerobic exercise on expression levels of Hippo YAP signaling pathway proteins in the myocardial tissue of TAC mice (Mean±SD, n=10). A: Relative Mst1 mRNA expression levels. B: Relative Lats1 mRNA expression level. C: Relative Lats2 mRNA expression level. D: Relative YAP mRNA expression level. E: Western blotting bands of Mst1/2, Lats1/2, p-Lats1/2, YAP, and p-YAP proteins. F: Quantitative analysis of Mst1/2, Lats1/2, p-Lats1/2, YAP, and p-YAP protein levels. *P<0.05, **P<0.01, ***P<0.001.
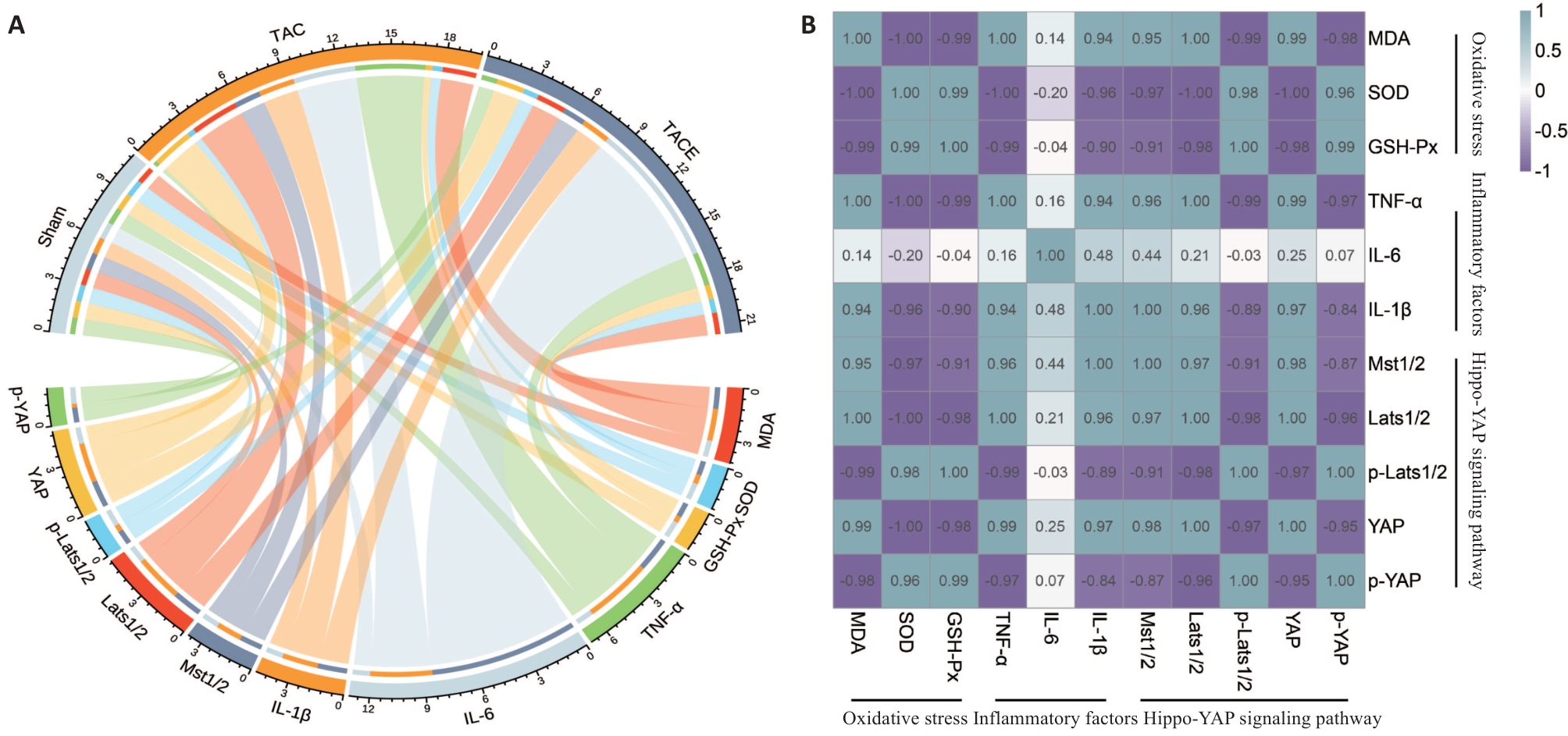
图6 各项指标相关性
Fig.6 Correlation of the indicators (Mean±SD, n=10). A: Delivery results of oxidative stress, inflammatory factors, and Hippo YAP signaling pathway in each group of mice. B: Correlation heatmap of various indicators. The intensity of colors in a heatmap indicates the degree of correlation.
| [1] | Groenewegen A, Rutten FH, Mosterd A, et al. Epidemiology of heart failure[J]. Eur J Heart Fail, 2020, 22(8): 1342-56. doi:10.1002/ejhf.1858 |
| [2] | Savarese G, Becher PM, Lund LH, et al. Global burden of heart failure: a comprehensive and updated review of epidemiology[J]. Cardiovasc Res, 2023, 118(17): 3272-87. doi:10.1093/cvr/cvac013 |
| [3] | Caturano A, Vetrano E, Galiero R, et al. Cardiac hypertrophy: from pathophysiological mechanisms to heart failure development[J]. Rev Cardiovasc Med, 2022, 23(5): 165. doi:10.31083/j.rcm2305165 |
| [4] | Hoque MM, Gbadegoye JO, Hassan FO, et al. Cardiac fibrogenesis: an immuno-metabolic perspective[J]. Front Physiol, 2024, 15: 1336551. doi:10.3389/fphys.2024.1336551 |
| [5] | Sachdev S, Ansari SA, Ansari MI, et al. Abiotic stress and reactive oxygen species: generation, signaling, and defense mechanisms[J]. Antioxidants, 2021, 10(2): 277. doi:10.3390/antiox10020277 |
| [6] | Sadiq IZ. Free radicals and oxidative stress: signaling mechanisms, redox basis for human diseases, and cell cycle regulation[J]. Curr Mol Med, 2023, 23(1): 13-35. doi:10.2174/1566524022666211222161637 |
| [7] | Aimo A, Castiglione V, Borrelli C, et al. Oxidative stress and inflammation in the evolution of heart failure: From patho-physiology to therapeutic strategies[J]. Eur J Prev Cardiol, 2020, 27(5): 494-510. doi:10.1177/2047487319870344 |
| [8] | Wijnker PJM, Sequeira V, Kuster DWD, et al. Hypertrophic cardiomyopathy: a vicious cycle triggered by sarcomere mutations and secondary disease hits[J]. Antioxid Redox Signal, 2019, 31(4): 318-58. doi:10.1089/ars.2017.7236 |
| [9] | Sugden PH, Clerk A. Oxidative stress and growth-regulating intracellular signaling pathways in cardiac myocytes[J]. Antioxid Redox Signal, 2006, 8(11/12): 2111-24. doi:10.1089/ars.2006.8.2111 |
| [10] | Kango-Singh M, Singh A. Regulation of organ size: insights from the Drosophila Hippo signaling pathway[J]. Dev Dyn, 2009, 238(7): 1627-37. doi:10.1002/dvdy.21996 |
| [11] | Zhao B, Lei QY, Guan KL. The Hippo-YAP pathway: new connections between regulation of organ size and cancer[J]. Curr Opin Cell Biol, 2008, 20(6): 638-46. doi:10.1016/j.ceb.2008.10.001 |
| [12] | Piccolo S, Dupont S, Cordenonsi M. The biology of YAP/TAZ: hippo signaling and beyond[J]. Physiol Rev, 2014, 94(4): 1287-312. doi:10.1152/physrev.00005.2014 |
| [13] | Zhao CX, Zeng CM, Ye S, et al. Yes-associated protein (YAP) and transcriptional coactivator with a PDZ-binding motif (TAZ): a nexus between hypoxia and cancer[J]. Acta Pharm Sin B, 2020, 10(6): 947-60. doi:10.1016/j.apsb.2019.12.010 |
| [14] | Zhong ZX, Jiao ZH, Yu FX. The Hippo signaling pathway in development and regeneration[J]. Cell Rep, 2024, 43(3): 113926. doi:10.1016/j.celrep.2024.113926 |
| [15] | Zheng AC, Chen QS, Zhang L. The Hippo-YAP pathway in various cardiovascular diseases: Focusing on the inflammatory response[J]. Front Immunol, 2022, 13: 971416. doi:10.3389/fimmu.2022.971416 |
| [16] | Chen XQ, Li YL, Luo JD, et al. Molecular mechanism of hippo-YAP1/TAZ pathway in heart development, disease, and regeneration[J]. Front Physiol, 2020, 11: 389. doi:10.3389/fphys.2020.00389 |
| [17] | Katzmarzyk PT, Staiano AE. Relationship between meeting 24-hour movement guidelines and cardiometabolic risk factors in children[J]. J Phys Act Health, 2017, 14(10): 779-84. doi:10.1123/jpah.2017-0090 |
| [18] | Fiuza-Luces C, Santos-Lozano A, Joyner M, et al. Exercise benefits in cardiovascular disease: beyond attenuation of traditional risk factors[J]. Nat Rev Cardiol, 2018, 15(12): 731-43. doi:10.1038/s41569-018-0065-1 |
| [19] | Wu ZJ, Han C, Wang ZY, et al. Combined training prescriptions for improving cardiorespiratory fitness, physical fitness, body composition, and cardiometabolic risk factors in older adults: Systematic review and meta-analysis of controlled trials[J]. Sci Sports, 2023. DOI:10.1016/j.scispo.2022.03.015 . |
| [20] | Bakker EA, Lee DC, Hopman MTE, et al. Dose-response association between moderate to vigorous physical activity and incident morbidity and mortality for individuals with a different cardiovascular health status: a cohort study among 142, 493 adults from the Netherlands[J]. PLoS Med, 2021, 18(12): e1003845. doi:10.1371/journal.pmed.1003845 |
| [21] | Liu YJ, Zhu JH, Yu JZ, et al. Cardiorespiratory fitness and metabolic risk in Chinese population: evidence from a prospective cohort study[J]. BMC Public Health, 2024, 24(1): 522. doi:10.1186/s12889-024-17742-4 |
| [22] | Petrosino JM, Heiss VJ, Maurya SK, et al. Graded maximal exercise testing to assess mouse cardio-metabolic phenotypes[J]. PLoS One, 2016, 11(2): e0148010. doi:10.1371/journal.pone.0148010 |
| [23] | 蔡 蕊, 黄 卓, 贺文霞, 等. 剪接因子HNRNPH1通过调控Circ-MYOCD的反向剪接影响心肌肥厚的发生[J]. 南方医科大学学报, 2025, 45(3): 587-94. |
| [24] | 黄 卓, 曾振宇, 李 佳, 等. 心力衰竭患者Circ-PALLD的高表达受转录因子GATA4的转录调控[J]. 南方医科大学学报, 2023, 43(8): 1371-8. doi:10.12122/j.issn.1673-4254.2023.08.14 |
| [25] | Liu JM, Tian XQ, Zhang M, et al. Advances in pharmacological research on myocardial remodeling agents: a decade in review[J]. Medicine, 2025, 104(23): e42757. doi:10.1097/md.0000000000042757 |
| [26] | Li JY, Chen L, Wang LC. Impact of different exercise modalities on physical function and quality of life in patients with heart failure[J]. J Multidiscip Healthc, 2024, 17: 2551-9. doi:10.2147/jmdh.s465578 |
| [27] | Patel L, Dhruve R, Keshvani N, et al. Role of exercise therapy and cardiac rehabilitation in heart failure[J]. Prog Cardiovasc Dis, 2024, 82: 26-33. doi:10.1016/j.pcad.2024.01.002 |
| [28] | Shah AK, Bhullar SK, Elimban V, et al. Oxidative stress as a mechanism for functional alterations in cardiac hypertrophy and heart failure[J]. Antioxidants, 2021, 10(6): 931. doi:10.3390/antiox10060931 |
| [29] | Donia T, Khamis A. Management of oxidative stress and inflammation in cardiovascular diseases: mechanisms and challenges[J]. Environ Sci Pollut Res Int, 2021, 28(26): 34121-53. doi:10.1007/s11356-021-14109-9 |
| [30] | Singh A, Kukreti R, Saso L, et al. Mechanistic insight into oxidative stress-triggered signaling pathways and type 2 diabetes[J]. Molecules, 2022, 27(3): 950. doi:10.3390/molecules27030950 |
| [31] | Guan XH, Hong X, Zhao N, et al. CD38 promotes angiotensin II-induced cardiac hypertrophy[J]. J Cell Mol Med, 2017, 21(8): 1492-502. doi:10.1111/jcmm.13076 |
| [32] | Lanser L, Fuchs D, Kurz K, et al. Physiology and inflammation driven pathophysiology of iron homeostasis: mechanistic insights into Anemia of inflammation and its treatment[J]. Nutrients, 2021, 13(11): 3732. doi:10.3390/nu13113732 |
| [33] | Zhong YG, Chen LY, Li MF, et al. Dangshen erling decoction ameliorates myocardial hypertrophy via inhibiting myocardial inflammation[J]. Front Pharmacol, 2022, 12: 725186. doi:10.3389/fphar.2021.725186 |
| [34] | Zhao MM, Zhang JS, Xu Y, et al. Selective inhibition of NLRP3 inflammasome reverses pressure overload-induced pathological cardiac remodeling by attenuating hypertrophy, fibrosis, and inflammation[J]. Int Immunopharmacol, 2021, 99: 108046. doi:10.1016/j.intimp.2021.108046 |
| [35] | Khaper N, Bryan S, Dhingra S, et al. Targeting the vicious inflammation-oxidative stress cycle for the management of heart failure[J]. Antioxid Redox Signal, 2010, 13(7): 1033-49. doi:10.1089/ars.2009.2930 |
| [36] | Zhang ZS, Guo JW. Deciphering oxidative stress in cardiovascular disease progression: a blueprint for mechanistic understanding and therapeutic innovation[J]. Antioxidants, 2024, 14(1): 38. doi:10.3390/antiox14010038 |
| [37] | Chen QY, Jiang YN, Guan X, et al. Aerobic exercise attenuates pressure overload-induced myocardial remodeling and myocardial inflammation via upregulating miR-574-3p in mice[J]. Circ Heart Fail, 2024, 17(3): e010569. doi:10.1161/circheartfailure.123.010569 |
| [1] | 宋淇乐, 苗益恺, 冯小桐, 王一凡, 刘伟, 魏琪, 于新汝, 陈文文, 付晓艳. 硒代胱氨酸通过诱导活性氧产生启动氧化应激损伤抑制结肠癌细胞生长[J]. 南方医科大学学报, 2026, 46(3): 532-540. |
| [2] | 彭煜策, 姜毅, 马丹, 何岸, 吕鼎一, 罗明昊, 罗素新. 异牡荆素通过促进SIRT3表达减轻糖尿病小鼠的心肌氧化应激损伤[J]. 南方医科大学学报, 2026, 46(2): 316-324. |
| [3] | 尹林, 张可妮, 乔通, 牛民主, 殷丽霞, 刘馨悦, 耿志军, 李静, 胡建国. 藜芦酸通过激活Nrf2/HO-1信号通路减轻氧化应激缓解葡聚糖硫酸钠诱导的小鼠实验性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 403-411. |
| [4] | 李楠, 张亮, 郭俏凤, 周越, 刘长江. 有氧运动通过调控miR-221-3p介导的脂肪组织巨噬细胞极化改善小鼠胰岛素抵抗[J]. 南方医科大学学报, 2026, 46(1): 74-82. |
| [5] | 王静娴, 任自敬, 周佩洋. S1PR5激动与过表达通过调控氧化应激增强脑微血管内皮细胞屏障功能抵抗氧糖剥夺/复氧复糖损伤[J]. 南方医科大学学报, 2025, 45(7): 1451-1459. |
| [6] | 张安邦, 孙秀颀, 庞博, 吴远华, 时靖宇, 张宁, 叶涛. 电针预处理通过调节肠道-大脑轴及Nrf2/HO-1信号通路抑制铁死亡减轻大鼠脑缺血再灌注损伤[J]. 南方医科大学学报, 2025, 45(5): 911-920. |
| [7] | 高志, 吴傲, 胡仲翔, 孙培养. 类风湿性关节炎中氧化应激与免疫浸润的生物信息学分析[J]. 南方医科大学学报, 2025, 45(4): 862-870. |
| [8] | 蔡蕊, 黄卓, 贺文霞, 艾添红, 宋晓伟, 胡淑婷. 剪接因子HNRNPH1通过调控Circ-MYOCD的反向剪接影响心肌肥厚的发生[J]. 南方医科大学学报, 2025, 45(3): 587-594. |
| [9] | 黄鹏伟, 陈洁, 邹金虎, 高雪锋, 曹虹. 槲皮素促进应激颗粒G3BP1解聚改善HIV-1 gp120诱导的星形胶质细胞神经毒性[J]. 南方医科大学学报, 2025, 45(2): 304-312. |
| [10] | 卢晓宇, 刘智慧, 刘烨, 庞天霄, 卞蓉, 郭玲, 何学红. 参芪泄浊饮通过调控Rap1/MAPK/FoxO3a信号通路改善氧化应激及炎症反应延缓大鼠肾纤维化[J]. 南方医科大学学报, 2025, 45(12): 2585-2597. |
| [11] | 林淑娴, 郭丽娜, 马燕, 熊尧, 何盈犀, 许欣筑, 盛雯, 许素哗, 邱峰. 植物乳植杆菌ZG03通过其代谢物短链脂肪酸缓解斑马鱼的氧化应激[J]. 南方医科大学学报, 2025, 45(10): 2223-2230. |
| [12] | 龙秀鹏, 陶顺, 阳绅, 李素云, 饶利兵, 李莉, 张哲. 槲皮素通过抑制MAPK信号通路改善心力衰竭[J]. 南方医科大学学报, 2025, 45(1): 187-196. |
| [13] | 陈志亮, 杨永刚, 黄霞, 成彦, 瞿媛, 衡琪琪, 符羽佳, 李可薇, 顾宁. 外泌体miRNA差异表达可作为诊断慢性心力衰竭合并高尿酸血症患者新型分子标志物及靶基因功能分析[J]. 南方医科大学学报, 2025, 45(1): 43-51. |
| [14] | 张钰明, 夏士程, 张淋淋, 陈梦茜, 刘晓婧, 高琴, 叶红伟. 金银花提取物对小鼠阿霉素肝脏损伤的保护作用[J]. 南方医科大学学报, 2024, 44(8): 1571-1581. |
| [15] | 任志军, 刁建新, 王奕婷. 芎归汤通过抑制氧化应激诱导的心肌凋亡减轻小鼠心梗后心衰引起的心肌损伤[J]. 南方医科大学学报, 2024, 44(7): 1416-1424. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||