南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 532-540.doi: 10.12122/j.issn.1673-4254.2026.03.07
• 基础研究 • 上一篇
宋淇乐1( ), 苗益恺2, 冯小桐1, 王一凡1, 刘伟1, 魏琪1, 于新汝1, 陈文文3(
), 苗益恺2, 冯小桐1, 王一凡1, 刘伟1, 魏琪1, 于新汝1, 陈文文3( ), 付晓艳4(
), 付晓艳4( )
)
收稿日期:2025-09-23
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
陈文文,付晓艳
E-mail:sqlbangong@163.com;wen-860521@163.com;txyfu66@163.com
作者简介:宋淇乐,在读硕士研究生,E-mail: sqlbangong@163.com
基金资助:
Qile SONG1( ), Yikai MIAO2, Xiaotong FENG1, Yifan WANG1, Wei LIU1, Qi WEI1, Xinru YU1, Wenwen CHEN3(
), Yikai MIAO2, Xiaotong FENG1, Yifan WANG1, Wei LIU1, Qi WEI1, Xinru YU1, Wenwen CHEN3( ), Xiaoyan FU4(
), Xiaoyan FU4( )
)
Received:2025-09-23
Online:2026-03-20
Published:2026-03-26
Contact:
Wenwen CHEN, Xiaoyan FU
E-mail:sqlbangong@163.com;wen-860521@163.com;txyfu66@163.com
Supported by:摘要:
目的 评价硒代胱氨酸(SeC)体外抑制结肠癌细胞生长的分子机制。 方法 体外培养RKO、HCT-116、LoVo结肠癌细胞,SeC处理细胞24 h和48 h,分为正常对照组和SeC低、中、高剂量组(SeC为5、10、20 μmol/L)。MTT检测结肠癌细胞的活性。划痕实验检测结肠癌细胞迁移能力。流式细胞仪PI染色检测细胞周期阻滞和凋亡情况。荧光探针检测自由基产生、线粒体结构和膜电位变化。铁死亡标志物如丙二醛(MDA)、谷胱甘肽(GSH)、亚铁离子(Fe2+)分析铁死亡的水平,Western blotting检测蛋白表达情况。 结果 SeC低、中、高剂量组均抑制结肠癌细胞增殖和迁移(P<0.05);SeC不同剂量处理组中细胞周期相关蛋白CDK2、CDK4表达下调,并且凋亡蛋白PARP和Caspase9发生活化(P<0.05);此外,Western blotting结果显示SeC诱导铁死亡蛋白FTH1,xCT表达下降和DMT1表达升高;SeC不同剂量处理组中MDA,Fe2+水平增加和GSH水平降低(P<0.05)。荧光结果显示线粒体结构损伤、活性氧(ROS)生成增多;Western blotting结果显示氧化损伤蛋白的磷酸化表达升高和NRF2/HO-1蛋白表达下调(P<0.05);ROS清除剂显著逆转结肠癌细胞中SeC对DMT1、PARP、p-H2A.X蛋白的上调作用(P<0.05)。 结论 SeC可通过诱导ROS生成启动氧化应激损伤诱导结肠癌细胞凋亡和铁死亡,进而抑制结肠癌细胞生长,是结肠癌潜在的化疗剂。
宋淇乐, 苗益恺, 冯小桐, 王一凡, 刘伟, 魏琪, 于新汝, 陈文文, 付晓艳. 硒代胱氨酸通过诱导活性氧产生启动氧化应激损伤抑制结肠癌细胞生长[J]. 南方医科大学学报, 2026, 46(3): 532-540.
Qile SONG, Yikai MIAO, Xiaotong FENG, Yifan WANG, Wei LIU, Qi WEI, Xinru YU, Wenwen CHEN, Xiaoyan FU. Selenocystine inhibits colon cancer cell growth by promoting reactive oxygen species generation to trigger oxidative damage[J]. Journal of Southern Medical University, 2026, 46(3): 532-540.
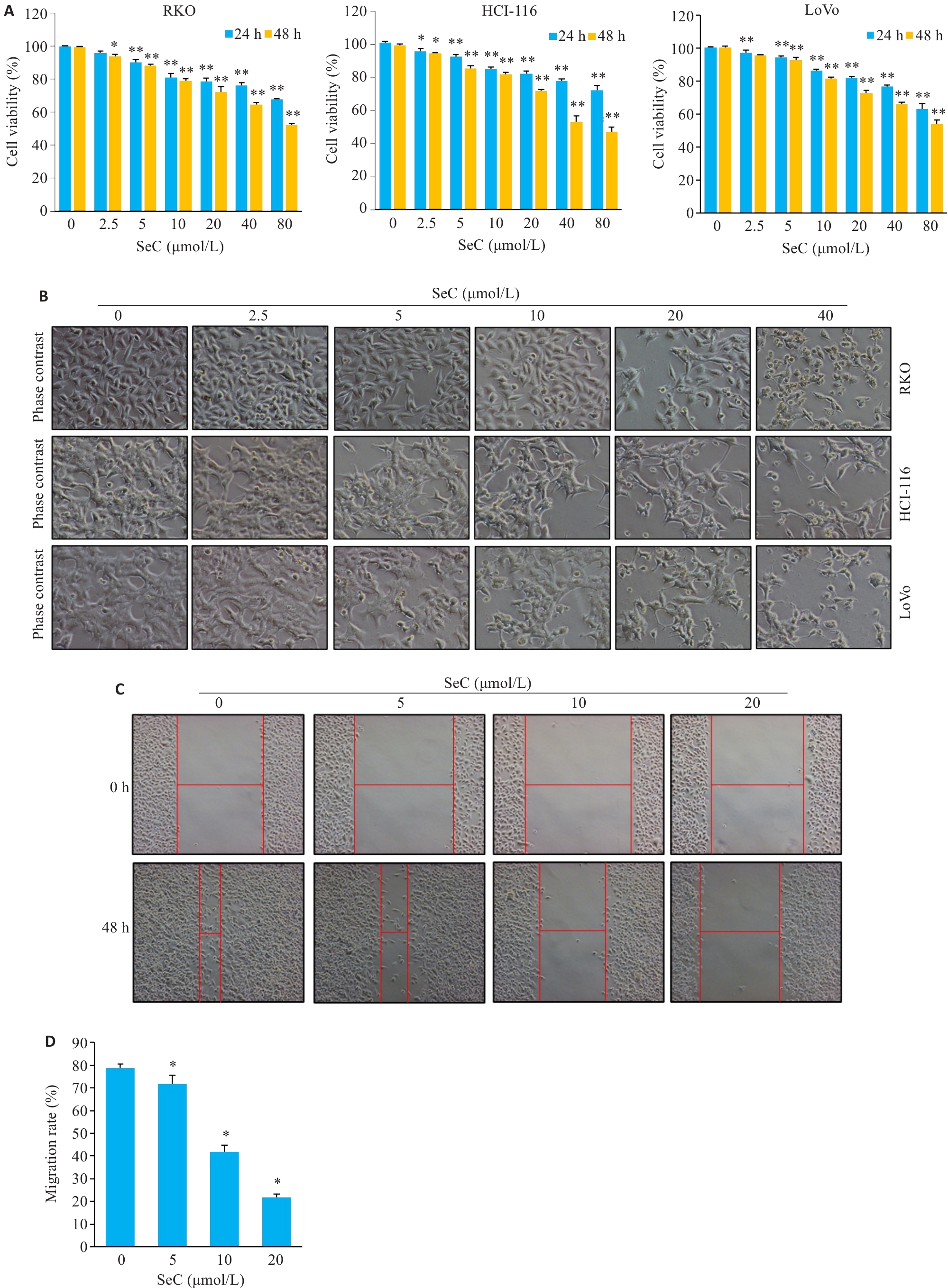
图1 SeC剂量依赖性抑制结肠癌细胞的生长
Fig.1 SeC inhibits growth of colon cancer cells in a dose-dependent manner. A: SeC inhibits viability of RKO, HCT-116 and LoVo cells detected by MTT assay. B: Changes in cell morphology (Original magnification: ×200). C: SeC inhibits the migration of RKO cells detected by wound healing assay. (×100). D: Quantitative analysis of cell migration. All experiments were repeated 3 times (n=3). All data were presented as Mean±SD. *P<0.05, **P<0.01 vs SeC 0 μmol/L.
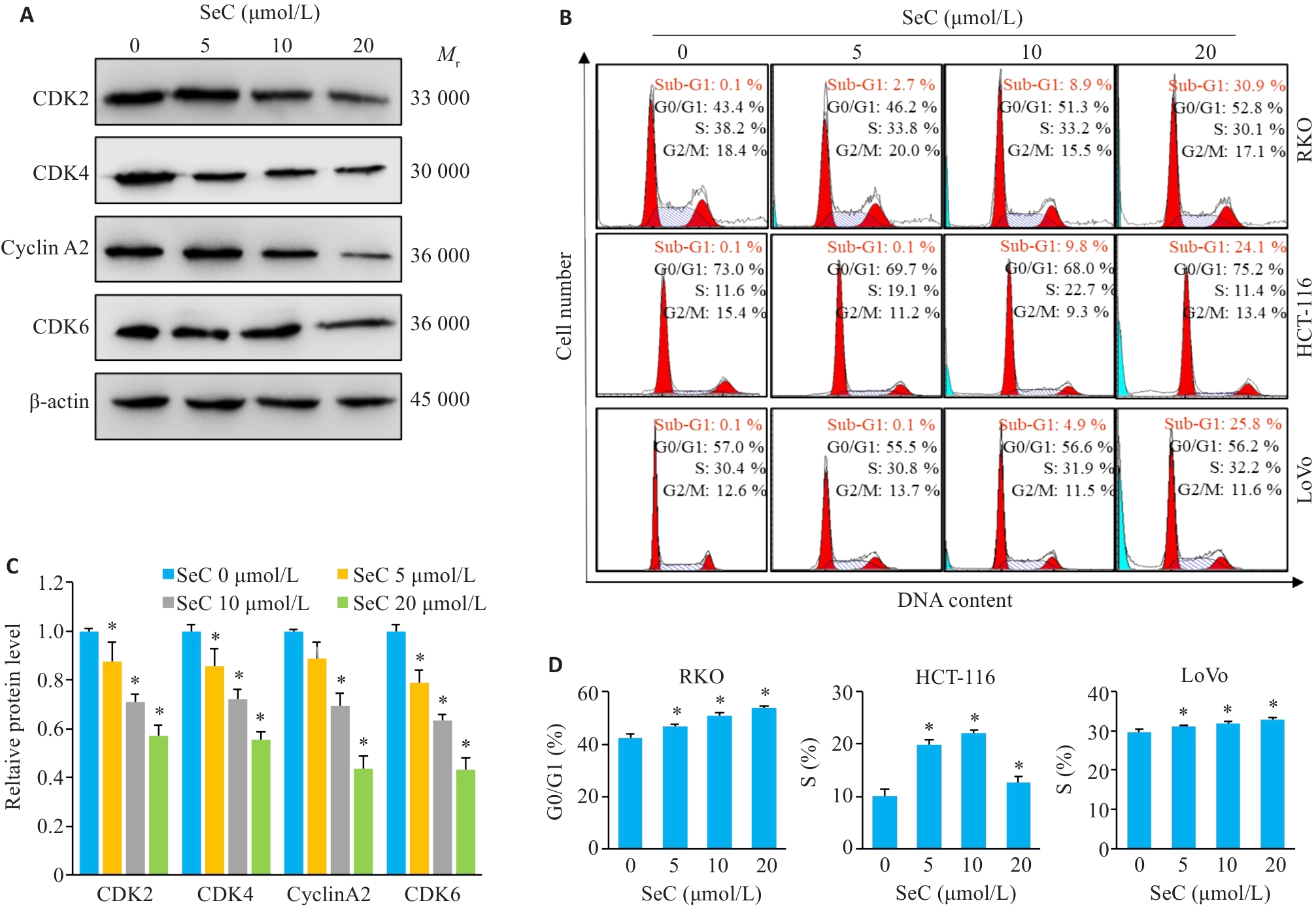
图2 SeC诱导结肠癌细胞周期阻滞和凋亡
Fig.2 SeC induces cell cycle arrest and apoptosis in colon cancer cells. A: SeC inhibits expressions of RKO cell cycle-related proteins detected by Western blotting. B: SeC-induced cell cycle arrest and apoptosis in RKO, HCT-116, and LoVo cells detected by flow cytometry with PI staining. C: Quantitative analysis of cell cycle proteins. D: Quantitative analysis of cell cycle arrest in RKO, HCT-116, and LoVo cells. All experiments were repeated three times (n=3). All data are presented as Mean±SD. *P<0.05 vs SeC 0 μmol/L.
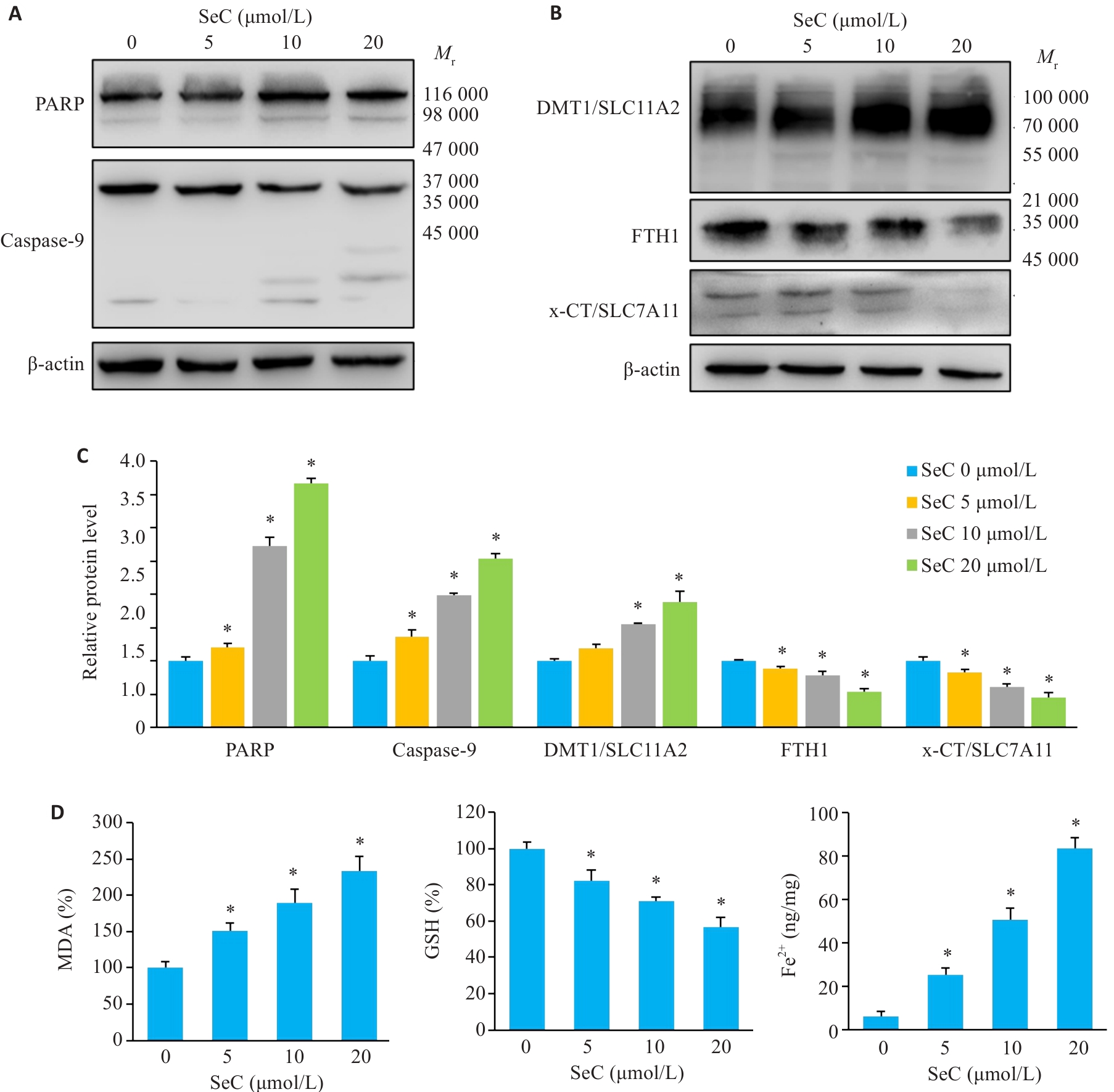
图3 SeC诱导结肠癌细胞凋亡和铁死亡
Fig.3 SeC induces apoptosis and ferroptosis in colon cancer cells. A: PARP cleavage and caspase-9 activation. B: Effect of SeC on ferroptosis-related proteins in RKO cells detected by Western blotting. C: Quantitative analysis of apoptotic and ferroptosis-related proteins. D: Measurement of MDA, GSH and Fe2+ in RKO cells after SeC treatment. All experiments were repeated three times (n=3). All data are presented as Mean±SD. *P<0.05 vs SeC 0 μmol/L.
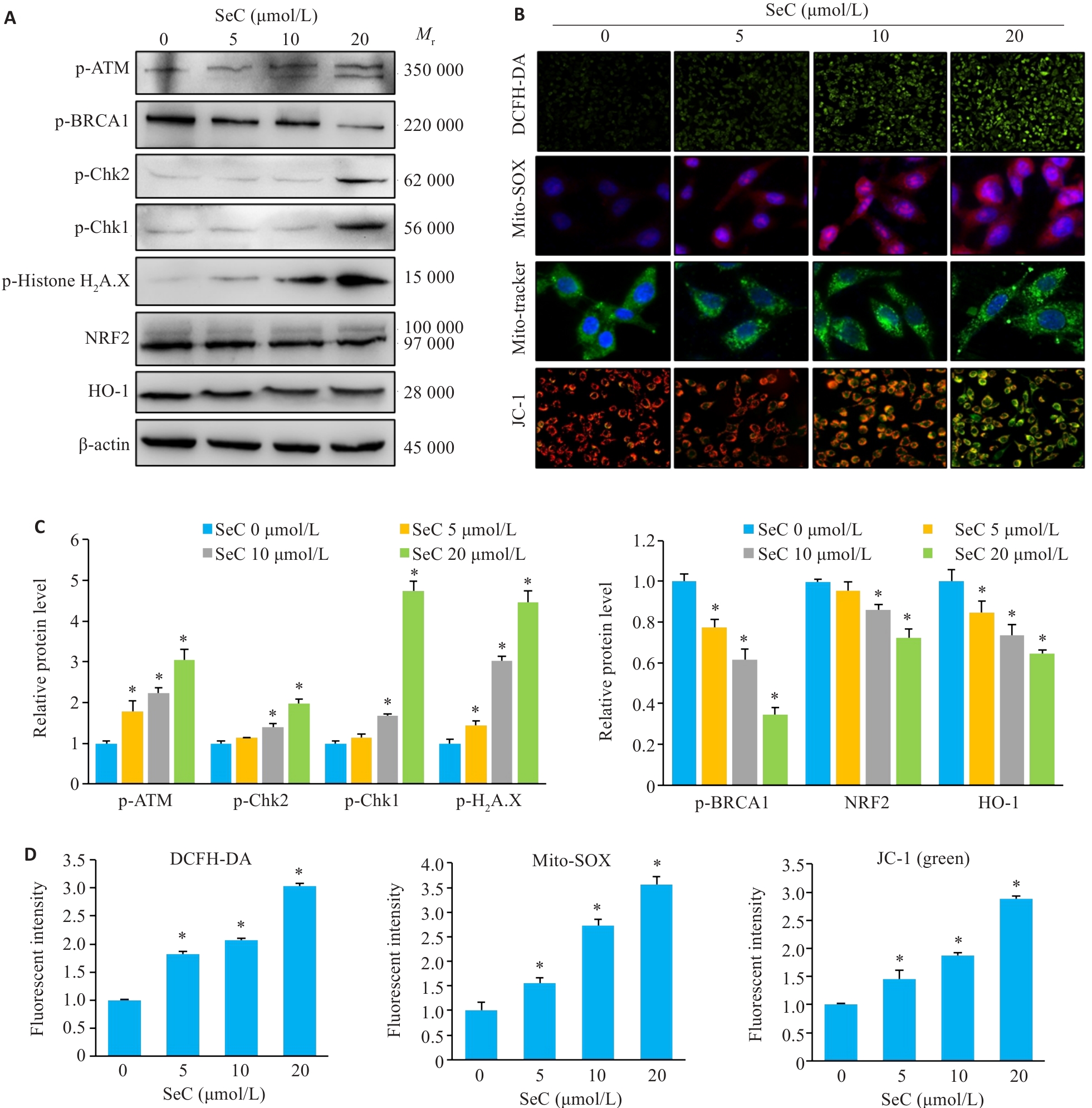
图4 SeC诱导结肠癌细胞线粒体功能障碍和氧化应激损伤
Fig.4 SeC induces mitochondrial dysfunction and oxidative stress damage in colon cancer cells. A: Effects of SeC on oxidative stress-related proteins in RKO cells detected by Western blotting. B: SeC induces ROS accumulation and mitochondrial dysfunction in RKO cells. DCFH-DA fluorescent probe was used to detect ROS production (×100). Mito-SOX fluorescent probe was used to detect mitochondrial superoxide production (×400). Mito-Tracker fluorescent probe was used to detect mitochondrial structural damage (×400). JC-1 fluorescent probe was used to detect mitochondrial membrane potential changes (×200). C: Quantitative analysis of oxidative stress-related protein and DNA damage-related protein results. D: Quantitative analysis of ROS, Mito-SOX and JC-1 fluorescence results. All experiments were repeated 3 times (n=3). All data are presented as Mean±SD. *P<0.05 vs SeC 0 μmol/L.
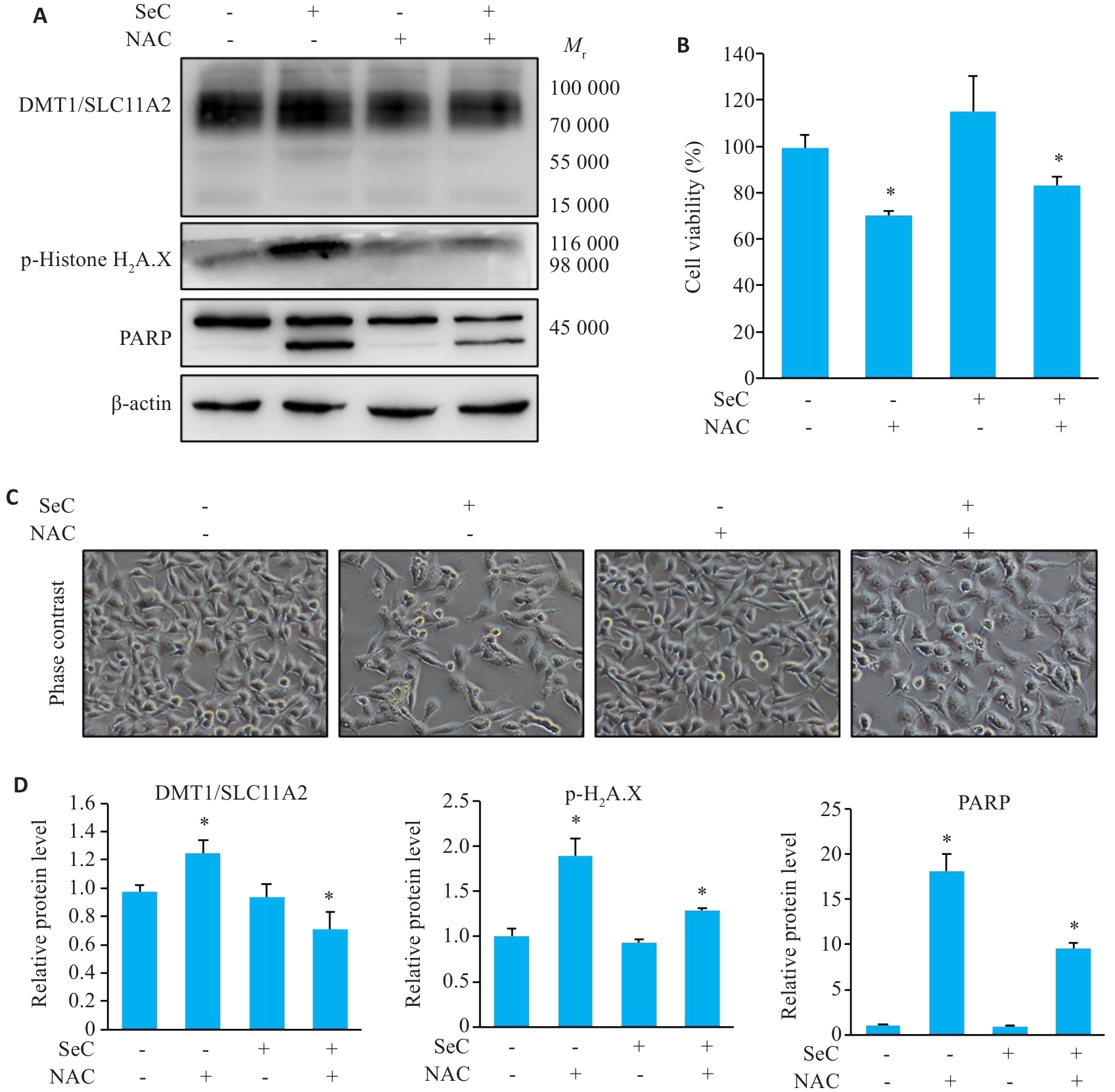
图5 ROS清除减弱SeC诱导的凋亡和铁死亡
Fig.5 ROS elimination attenuates SeC-induced apoptosis and ferroptosis. A: NAC pretreatment inhibits SeC-induced DNA damage, apoptosis, and ferroptosis detected by Western blotting. B: NAC pretreatment reverses the effects of SeC on colon cancer cells. C: Changes in cell morphology (×200). D: Quantitative analysis of apoptosis-related proteins, ferroptosis-related proteins, and DNA damage-related proteins. All experiments were repeated 3 times (n=3). All data are presented as Mean±SD. *P<0.05 vs SeC 0 μmol/L.
| [1] | Malki A, ElRuz RA, Gupta I, et al. Molecular Mechanisms of Colon Cancer Progression and Metastasis: Recent Insights and Advancements[J]. Int J Mol Sci, 2020, 22(1): 130. doi:10.3390/ijms22010130 |
| [2] | Peters KM, Carlson BA, Gladyshev VN, et al. Selenoproteins in colon cancer[J]. Free Radic Biol Med, 2018, 127: 14-25. doi:10.1016/j.freeradbiomed.2018.05.075 |
| [3] | Audisio A, Fazio R, Dapra V, et al. Neoadjuvant chemotherapy for early-stage colon cancer[J]. Cancer Treat Rev, 2024, 123:102676. doi:10.1016/j.ctrv.2023.102676 |
| [4] | Benarba B, Pandiella A. Colorectal cancer and medicinal plants: Principle findings from recent studies[J]. Biomed Pharmacother, 2018, 107: 408-23. doi:10.1016/j.biopha.2018.08.006 |
| [5] | Aiello P, Sharghi M, Mansourkhani SM, et al. Medicinal Plants in the Prevention and Treatment of Colon Cancer[J]. Oxid Med Cell Longev, 2019, 2019: 2075614. doi:10.1155/2019/2075614 |
| [6] | Thiengsusuk A, Boonprasert K, Na-Bangchang K. A Systematic Review of Drug Metabolism Studies of Plants With Anticancer Properties: Approaches Applied and Limitations[J]. Eur J Drug Metab Pharmacokinet, 2020, 45(2): 173-225. doi:10.1007/s13318-019-00582-8 |
| [7] | Winther KH, Rayman MP, Bonnema SJ, et al. Selenium in thyroid disorders-essential knowledge for clinicians[J]. Nat Rev Endocrinol, 2020, 16(3): 165-76. doi:10.1038/s41574-019-0311-6 |
| [8] | Wichman J, Winther KH, Bonnema SJ, et al. Selenium Supplementation Significantly Reduces Thyroid Autoantibody Levels in Patients with Chronic Autoimmune Thyroiditis: A Systematic Review and Meta-Analysis[J]. Thyroid, 2016, 26(12):1681-92. doi:10.1089/thy.2016.0256 |
| [9] | Mojadadi A, Au A, Salah W, et al. Role for Selenium in Metabolic Homeostasis and Human Reproduction[J]. Nutrients, 2021, 13(9):3256. doi:10.3390/nu13093256 |
| [10] | Hsu WL, Wang CM, Yao CL, et al. Blockage of Nrf2 and autophagy by L-selenocystine induces selective death in Nrf2-addicted colorectal cancer cells through p62-Keap-1-Nrf2 axis[J]. Cell Death Dis, 2022, 13(12): 1060. doi:10.1038/s41419-022-05512-2 |
| [11] | Long M, Wu J, Hao J, et al. Selenocystine-induced cell apoptosis and S-phase arrest inhibit human triple-negative breast cancer cell proliferation[J]. In Vitro Cell Dev Biol Anim, 2015, 51(10):1077-84. doi:10.1007/s11626-015-9937-4 |
| [12] | Bartolini D, Sancineto L, Fabro De Bem A, et al. Selenocompounds in Cancer Therapy: An Overview[J]. Adv Cancer Res, 2017, 136:259-302. doi:10.1016/bs.acr.2017.07.007 |
| [13] | Wahyuni EA, Yii CY, Liang HL, et al. Selenocystine induces oxidative-mediated DNA damage via impairing homologous recombination repair of DNA double-strand breaks in human hepatoma cells[J]. Chem Biol Interact, 2022, 365: 110046. doi:10.1016/j.cbi.2022.110046 |
| [14] | Chen T, Wong YS. Selenocystine induces reactive oxygen species-mediated apoptosis in human cancer cells[J]. Biomed Pharmacother, 2009, 63(2): 105-13. doi:10.1016/j.biopha.2008.03.009 |
| [15] | LeFort KR, Rungratanawanich W, Song BJ. Contributing roles of mitochondrial dysfunction and hepatocyte apoptosis in liver diseases through oxidative stress, post-translational modifications, inflamm-ation, and intestinal barrier dysfunction[J]. Cell Mol Life Sci, 2024, 81(1): 34. doi:10.1007/s00018-023-05061-7 |
| [16] | Yu W, Liu T, Yuan Z, et al. Polymer-engineered liposome-delivered Ebselen against tumor through GSH/H2O2-responsive disruption of redox homeostasis and direct p53 activation[J]. Biomaterials, 2026, 326: 123698. doi:10.1016/j.biomaterials.2025.123698 |
| [17] | Dai C, He Y, Lu H, et al. Nanoquadruplex-driven hydrogen therapy: NIR-controlled release for targeted cancer ferroptosis[J]. Biomaterials, 2026, 326: 123635. doi:10.1016/j.biomaterials.2025.123635 |
| [18] | Riedl SJ, Shi Y. Molecular mechanisms of caspase regulation during apoptosis[J]. Nat Rev Mol Cell Biol, 2004, 5(11): 897-907. doi:10.1038/nrm1496 |
| [19] | Bock FJ, Tait SWG. Mitochondria as multifaceted regulators of cell death[J]. Nat Rev Mol Cell Biol, 2020, 21(2): 85-100. doi:10.1038/s41580-019-0173-8 |
| [20] | Bou-Teen D, Kaludercic N, Weissman D, et al. Mitochondrial ROS and mitochondria-targeted antioxidants in the aged heart[J]. Free Radic Biol Med, 2021, 167: 109-24. doi:10.1016/j.freeradbiomed.2021.02.043 |
| [21] | Sun CY, Cong KP, Zhao DD, et al. 8-Nitrotryptanthrin inhibits colorectal cancer progression via TGF-beta/SMAD and PI3K/AKT/mTOR pathways[J]. Front Pharmacol, 2025, 16: 1576673. doi:10.3389/fphar.2025.1576673 |
| [22] | Wang R, Ha KY, Dhandapani S, et al. Biologically synthesized black ginger-selenium nanoparticle induces apoptosis and autophagy of AGS gastric cancer cells by suppressing the PI3K/Akt/mTOR signaling pathway[J]. J Nanobiotechnology, 2022, 20(1): 441. doi:10.1186/s12951-022-01576-6 |
| [23] | Wang M, Xu H, Xiong X, et al. Antiproliferative activity of selenium-enriched coumarin derivatives on the SK-N-SH neuroblastoma cell line: Mechanistic insights[J]. Eur J Med Chem, 2025, 286: 117322. doi:10.1016/j.ejmech.2025.117322 |
| [24] | Dixon SJ, Lemberg KM, Lamprecht MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death[J]. Cell, 2012, 149(5):1060-72. doi:10.1016/j.cell.2012.03.042 |
| [25] | Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease[J]. Nat Rev Mol Cell Biol, 2021, 22(4):266-82. doi:10.1038/s41580-020-00324-8 |
| [26] | Chen J, Wang Y, Wu J, et al. The Potential Value of Targeting Ferroptosis in Early Brain Injury After Acute CNS Disease[J]. Front Mol Neurosci, 2020, 13: 110. doi:10.3389/fnmol.2020.00110 |
| [27] | Sun Y, Li Q, Guo H, et al. Ferroptosis and Iron Metabolism after Intracerebral Hemorrhage[J]. Cells, 2022, 12(1): 90. doi:10.3390/cells12010090 |
| [28] | Choi JA, Lee EH, Cho H, et al. High-Dose Selenium Induces Ferroptotic Cell Death in Ovarian Cancer[J]. Int J Mol Sci, 2023, 24(3): 1918. doi:10.3390/ijms24031918 |
| [29] | Chen Y, Liu X, Chen B, et al. Selenium nanoparticles decorated by fucoidan induce ferroptosis in HepG2 cells[J]. Int J Biol Macromol, 2025, 289: 138841. doi:10.1016/j.ijbiomac.2024.138841 |
| [30] | Wang B, Wang Y, Zhang J, et al. ROS-induced lipid peroxidation modulates cell death outcome: mechanisms behind apoptosis, autophagy, and ferroptosis[J]. Arch Toxicol, 2023, 97(6): 1439-51. doi:10.1007/s00204-023-03476-6 |
| [31] | Tang Q, Chen H, Mai Z, et al. Bim- and Bax-mediated mitochondrial pathway dominates abivertinib-induced apoptosis and ferroptosis[J]. Free Radic Biol Med, 2022, 180: 198-209. doi:10.1016/j.freeradbiomed.2022.01.013 |
| [32] | Qiao R, Yuan Z, Yang M, et al. Selenium-Doped Nanohetero-junctions for Highly Efficient Cancer Radiosensitization[J]. Adv Sci (Weinh), 2024, 11(29): e2402039. doi:10.1002/advs.202402039 |
| [33] | Ren M, Liang S, Lin S, et al. Design, synthesis and biological evaluation of artesunate-Se derivatives as anticancer agents by inducing GPX4-mediated ferroptosis[J]. Bioorg Chem, 2024, 152:107733. doi:10.1016/j.bioorg.2024.107733 |
| [34] | Chen QM. Nrf2 for protection against oxidant generation and mitochondrial damage in cardiac injury[J]. Free Radic Biol Med, 2022, 179: 133-43. doi:10.1016/j.freeradbiomed.2021.12.001 |
| [35] | Qin Q, Zhang H, Lai M, et al. Sulfasalazine induces ferroptosis in osteosarcomas by regulating Nrf2/SLC7A11/GPX4 signaling axis[J]. Sci Rep, 2025, 15(1): 30197. doi:10.1038/s41598-025-13324-5 |
| [1] | 彭煜策, 姜毅, 马丹, 何岸, 吕鼎一, 罗明昊, 罗素新. 异牡荆素通过促进SIRT3表达减轻糖尿病小鼠的心肌氧化应激损伤[J]. 南方医科大学学报, 2026, 46(2): 316-324. |
| [2] | 尹林, 张可妮, 乔通, 牛民主, 殷丽霞, 刘馨悦, 耿志军, 李静, 胡建国. 藜芦酸通过激活Nrf2/HO-1信号通路减轻氧化应激缓解葡聚糖硫酸钠诱导的小鼠实验性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 403-411. |
| [3] | 乔通, 尹林, 张可妮, 牛民主, 黄菊, 耿志军, 李静, 胡建国. 茯苓新酸A通过调节AMPK/mTOR介导的自噬来减轻葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2026, 46(1): 131-140. |
| [4] | 张淑芬, 黄添容, 杨灿洪, 陈家镒, 吕田明, 张嘉发. 莱菔硫烷通过抑制Aβ42寡聚体激活的U87细胞中MAPK/NF-κB信号通路降低反应性星形胶质细胞介导的SH-SY5Y凋亡[J]. 南方医科大学学报, 2026, 46(1): 191-199. |
| [5] | 赵锦燕, 彭娇, 林明和, 朱晓勤, 黄彬, 林久茂. 清解扶正颗粒通过抑制线粒体依赖的凋亡、激活AMPK-PGC-1α通路缓解5-氟尿嘧啶引起的骨骼肌损伤[J]. 南方医科大学学报, 2026, 46(1): 94-103. |
| [6] | 何榕茂, 方泽扬, 张芸芸, 吴友谅, 梁世秀, 计涛, 陈科全, 王斯琪. 铁死亡相关基因对溃疡性结肠炎具有诊断预测价值[J]. 南方医科大学学报, 2025, 45(9): 1927-1937. |
| [7] | 云琦, 杜若丽, 贺玉莹, 张贻欣, 王佳慧, 叶红伟, 李正红, 高琴. 肉桂酸通过抑制TLR4减轻阿霉素诱导的小鼠心肌损伤铁死亡的发生[J]. 南方医科大学学报, 2025, 45(9): 1946-1958. |
| [8] | 王莹, 李静, 王伊迪, 华明钰, 胡玮彬, 张晓智. 原发性肝癌患者的临床结局与治疗反应预测模型:基于失巢凋亡和免疫基因[J]. 南方医科大学学报, 2025, 45(9): 1967-1979. |
| [9] | 陈丹丹, 任乾千, 吕梦林, 张宝文, 刘醒然, 张蒙, 王阳, 寇现娟. 天麻钩藤饮通过抑制坏死性凋亡通路改善帕金森病小鼠的运动功能障碍[J]. 南方医科大学学报, 2025, 45(8): 1571-1580. |
| [10] | 欧泽金, 李瀛, 陈诗, 王梓译, 何美仪, 陈志成, 唐侍豪, 孟晓静, 王致. 抑制铁死亡减轻敌草快引起的斑马鱼急性肾损伤的机制[J]. 南方医科大学学报, 2025, 45(8): 1743-1750. |
| [11] | 李军仪, 陈思源, 谢力遥, 王劲, 程奥, 张绍伟, 林继瑜, 方志涵, 潘一锐, 崔翀鹤, 陈庚鑫, 张超, 李栎. 益智仁提取物谷甾醇通过抑制铁死亡中的ETS-5基因表达延长秀丽隐杆线虫的寿命[J]. 南方医科大学学报, 2025, 45(8): 1751-1757. |
| [12] | 常笑语, 张瀚文, 曹红亭, 侯玲, 孟鑫, 陶虹, 罗彦, 李光华. 热应激对大鼠胸主动脉内皮细胞生物钟基因 Bmal1和细胞周期蛋白表达水平的影响[J]. 南方医科大学学报, 2025, 45(7): 1353-1362. |
| [13] | 陈鑫源, 吴成挺, 李瑞迪, 潘雪芹, 张耀丹, 陶俊宇, 林才志. 双术汤通过P53/SLC7A11/GPX4通路诱导胃癌细胞铁死亡[J]. 南方医科大学学报, 2025, 45(7): 1363-1371. |
| [14] | 王静娴, 任自敬, 周佩洋. S1PR5激动与过表达通过调控氧化应激增强脑微血管内皮细胞屏障功能抵抗氧糖剥夺/复氧复糖损伤[J]. 南方医科大学学报, 2025, 45(7): 1451-1459. |
| [15] | 张梦影, 赵晨玲, 田丽伟, 余郭芳, 杨文明, 董婷. 肝豆扶木汤通过GPX4/ACSL4/ALOX15通路抑制铁死亡改善Wilson病小鼠的肝脏脂肪变性[J]. 南方医科大学学报, 2025, 45(7): 1471-1478. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||