南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (7): 1451-1459.doi: 10.12122/j.issn.1673-4254.2025.07.11
收稿日期:2025-02-22
出版日期:2025-07-20
发布日期:2025-07-17
通讯作者:
周佩洋
E-mail:781893244@qq.com;zhoupeiyang@126.com
作者简介:王静娴,在读硕士研究生,E-mail: 781893244@qq.com
基金资助:
Jingxian WANG( ), Zijing REN, Peiyang ZHOU(
), Zijing REN, Peiyang ZHOU( )
)
Received:2025-02-22
Online:2025-07-20
Published:2025-07-17
Contact:
Peiyang ZHOU
E-mail:781893244@qq.com;zhoupeiyang@126.com
摘要:
目的 探讨鞘氨醇1-磷酸受体5(S1PR5)对氧糖剥夺及复氧复糖(OGD/R)诱导的小鼠脑微血管内皮细胞屏障功能的影响及相关机制。 方法 体外培养小鼠脑微血管内皮细胞bEnd.3,使用OGD/R诱导屏障功能障碍,分别使用S1PR5特异性激动剂A971432、敲低S1PR5的siRNA及过表达S1PR5的慢病毒进行干预。设置对照组:bEnd.3正常培养;OGD/R组:bEnd.3进行OGD/R;A971432组:OGD/R组+A971432;siNC组:转染siNC+OGD/R;siS1PR5组:转染siS1PR5+OGD/R;OE NC组:感染慢病毒LV5-NC+OGD/R;OE S1PR5组:感染慢病毒LV5-S1PR5+OGD/R。RT-qPCR法分别检测敲低或过表达S1PR5的效率;采用CCK-8检测在不同培养条件下bEnd.3细胞的活力;采用FITC-dextran渗透法检测内皮屏障渗透性;采用细胞免疫荧光法检测蛋白的定位及表达;DCFH-DA探针法检测细胞内活性氧水平;Western blotting法检测蛋白的表达水平。 结果 CCK-8结果显示激动S1PR5可以增加OGD/R下调的细胞活力(P<0.0001),FITC-dextran渗透法结果显示激动和过表达S1PR5可减少FITC-dextran的渗漏(P<0.001),而敲低S1PR5增加FITC-dextran的渗漏(P<0.001)。Western blotting及免疫荧光结果显示,与OGD/R组相比,激动和过表达S1PR5可以增加屏障蛋白ZO-1(P<0.05)、Occludin(P<0.05)的表达,而敲低S1PR5会下调ZO-1和Occludin(P<0.05)的表达。ROS检测结果显示,激动和过表达S1PR5能减少ROS的产生,而敲低S1PR5会增加ROS产生。Western blotting检测发现过表达S1PR5可以增加抗氧化蛋白Nrf2(P<0.0001)、HO-1(P<0.0001)、SOD2(P<0.01)的表达。 结论 S1PR5受体的激动剂干预及基因过表达可显著改善OGD/R模型诱导的活力减少及通透性增加,而基因敲低S1PR5则加剧OGD/R诱导的功能损伤,其机制可能与减少ROS,上调抗氧化蛋白表达相关。
王静娴, 任自敬, 周佩洋. S1PR5激动与过表达通过调控氧化应激增强脑微血管内皮细胞屏障功能抵抗氧糖剥夺/复氧复糖损伤[J]. 南方医科大学学报, 2025, 45(7): 1451-1459.
Jingxian WANG, Zijing REN, Peiyang ZHOU. S1PR5 activation or overexpression enhances barrier function of mouse brain microvascular endothelial cells against OGD/R injury by modulating oxidative stress[J]. Journal of Southern Medical University, 2025, 45(7): 1451-1459.
| Gene | Targeting sequence (5'-3') |
|---|---|
| siRNA1 | Sense: GCUAUUGCUCGGGGCUGCUATT Antisense: UAGCAGCCCCGAGCAAUAGCTT |
| siRNA2 | Sense: GCUCUACGCCAAGGCCUAUTT Antisense: AUAGGCCUUGGCGUAGAGCTT |
| siRNA3 | Sense: GCUCUACGCCAAGGCCUAUTT Antisense: ACGUGACACGUUCGGAGAATT |
| siNC | Sense: UUCUCCGAACGUGUCACGUTT Antisense: ACGUGACACGUUCGGAGAATT |
表1 S1PR5 siRNA靶向序列
Tab.1 S1PR5-specifc siRNA sequences used in this study
| Gene | Targeting sequence (5'-3') |
|---|---|
| siRNA1 | Sense: GCUAUUGCUCGGGGCUGCUATT Antisense: UAGCAGCCCCGAGCAAUAGCTT |
| siRNA2 | Sense: GCUCUACGCCAAGGCCUAUTT Antisense: AUAGGCCUUGGCGUAGAGCTT |
| siRNA3 | Sense: GCUCUACGCCAAGGCCUAUTT Antisense: ACGUGACACGUUCGGAGAATT |
| siNC | Sense: UUCUCCGAACGUGUCACGUTT Antisense: ACGUGACACGUUCGGAGAATT |
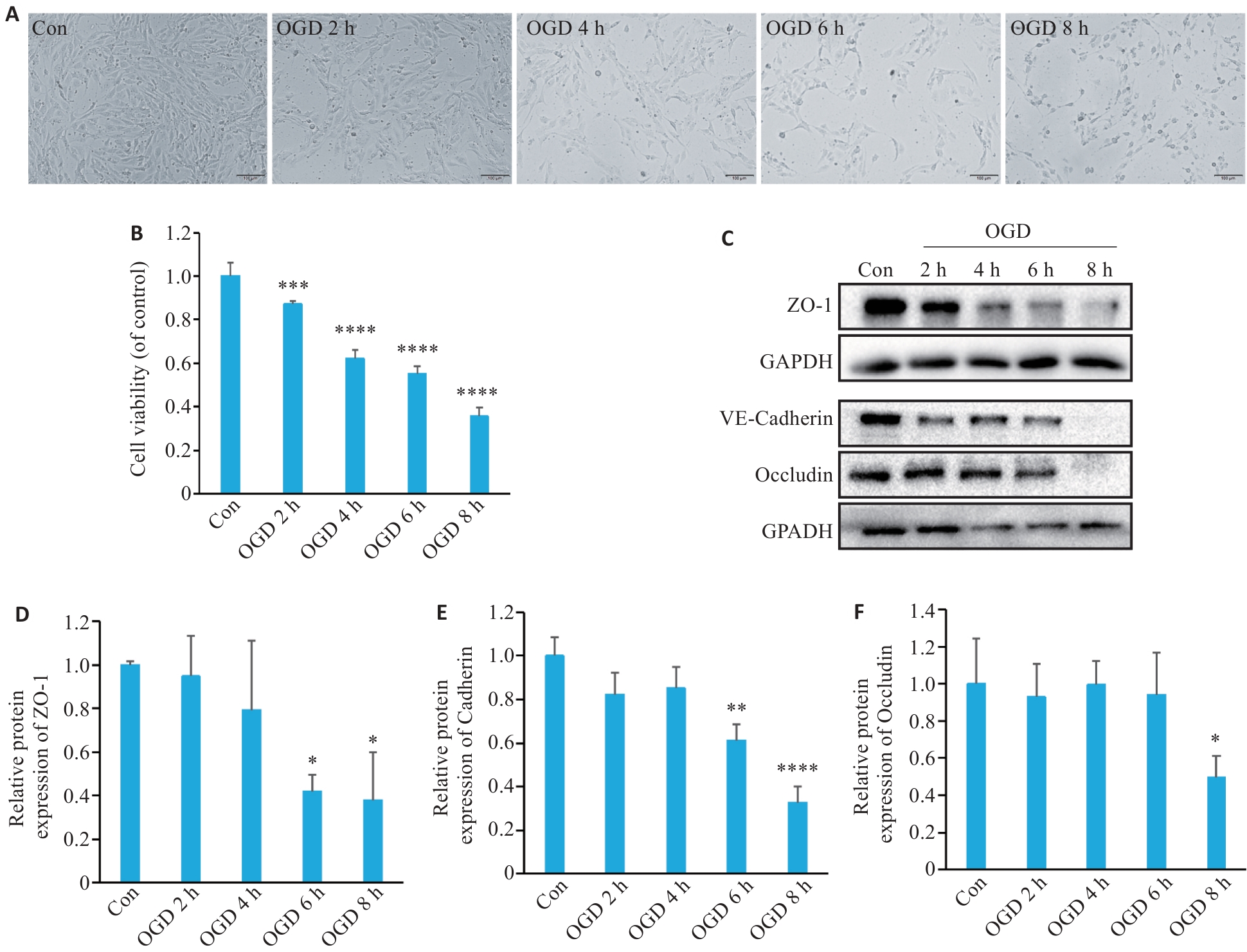
图1 OGD处理降低bEnd.3细胞活性并下调细胞间紧密连接蛋白的表达
Fig.1 Oxygen-glucose deprivation (OGD) decrease viability of bEnd.3 cells and down-regulate expressions of intercellular tight junction proteins. A: Representative images of bEnd.3 cells after OGD treatment. B: Viability of b.End3 cells assessed with CCK-8 assay (n=6). C: Western blotting of tight junction proteins (ZO-1 and occludin) and VE-cadherin after OGD. D-F: Quantification of ZO-1, VE-cadherin, and occludin protein expression levels (n=3). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 vs Con.
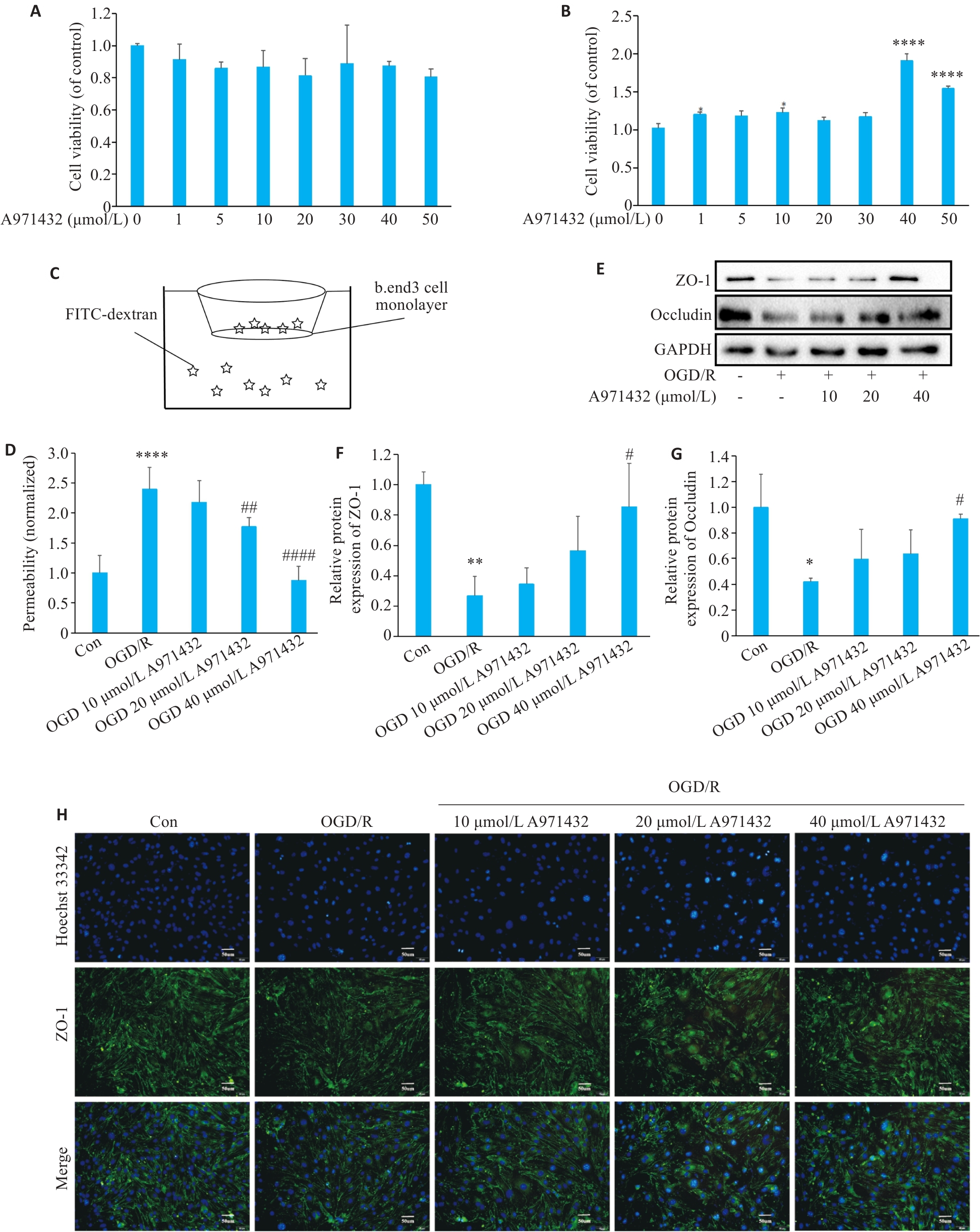
图2 激动S1PR5有助于恢复OGD下调的细胞活性并上调细胞间紧密连接蛋白的表达
Fig.2 Activation of S1PR5 restores viability of OGD-damaged cells and up-regulates expressions of intercellular tight junction proteins. A: CCK8 assay for assessing viability of cultured bEnd.3 cells treated with different concentrations of A971432. B: CCK8 assay for assessing viability of bEnd.3 cells exposed to OGD after treatment with different concentrations of A971432 (****P<0.0001 vs 0 μmol/L A971432). C: Schematic diagram for detecting endothelial permeability of bEnd.3 cells using FITC-Dextran. D: Detection of endothelial permeability using FITC-Dextran. E: Western blotting of ZO-1 and occluding expressions in bEnd.3 cells with OGD/R after S1PR5 activation. F, G: Quantitative analysis of the expression levels of ZO-1 and occludin. H: Changes in the fluorescence intensity of tight junction proteins caused by activation of S1PR5 in bEnd.3 cells with OGD/R (scale bar= 50 μm). *P<0.05, **P<0.01, ****P<0.0001 vs Con; #P<0.05, ##P<0.01, ####P<0.0001 vs OGD/R.
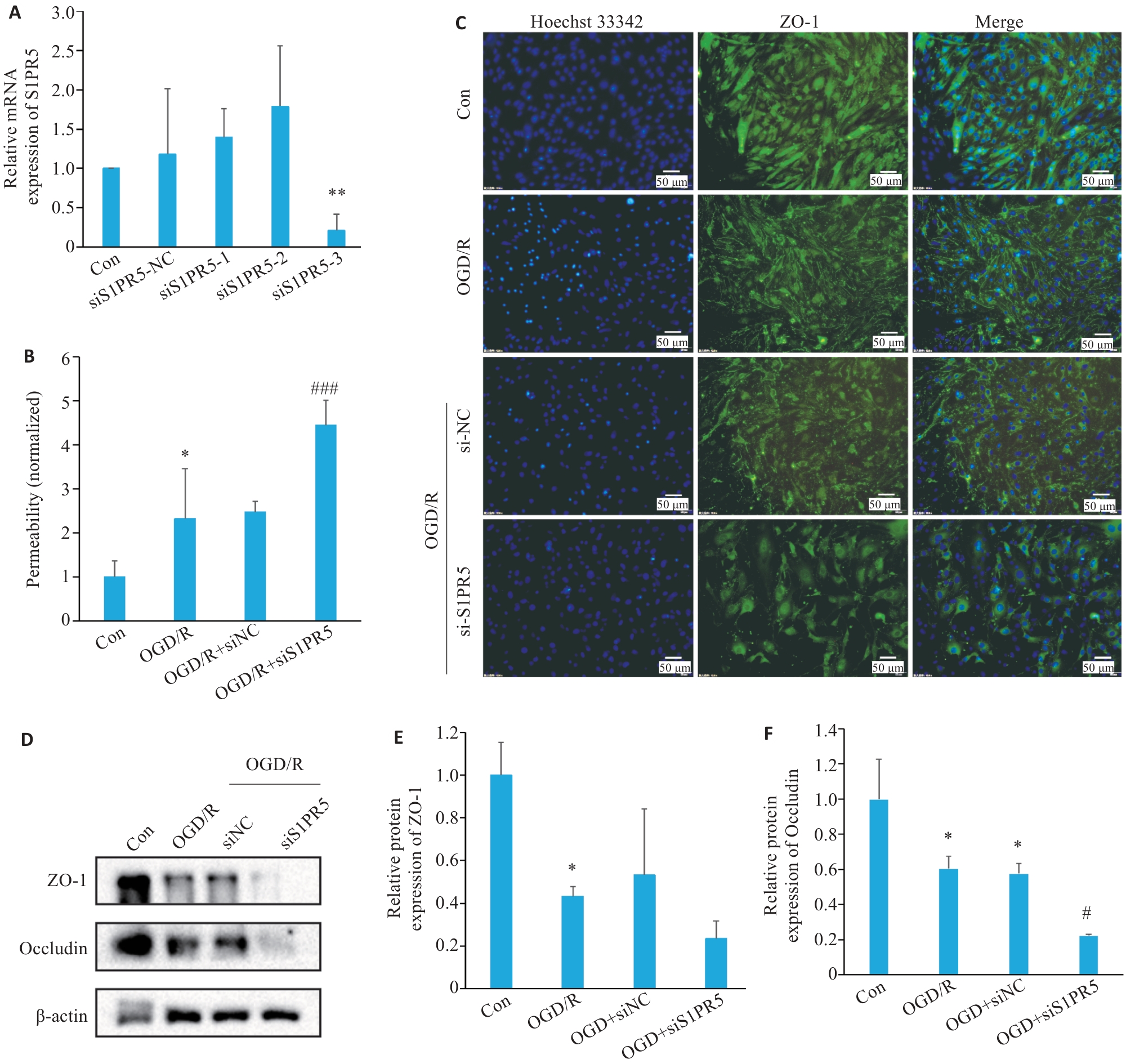
图3 敲低S1PR5加剧OGD诱导的细胞活力下降及紧密连接蛋白的减少
Fig.3 S1PR5 knockdown exacerbates the reduction of cell viability and tight junction protein expressions in OGD-induced bEnd.3 cells. A: Efficiency of S1PR5 knockdown assessed by RT-qPCR. B: Endothelial permeability of the transfected cells detected by FITC-Dextran. C: Alterations in the fluorescence intensity of the tight junction proteins in bEnd.3 cells with S1PR5 knockdown following OGD/R. D-F: Western blotting for detecting the expression levels of ZO-1 and occluding in bEnd.3 cells with OGD/R after S1PR5 knockdown. *P<0.05, **P<0.01 vs Con; #P<0.05, ###P<0.001 vs OGD/R.
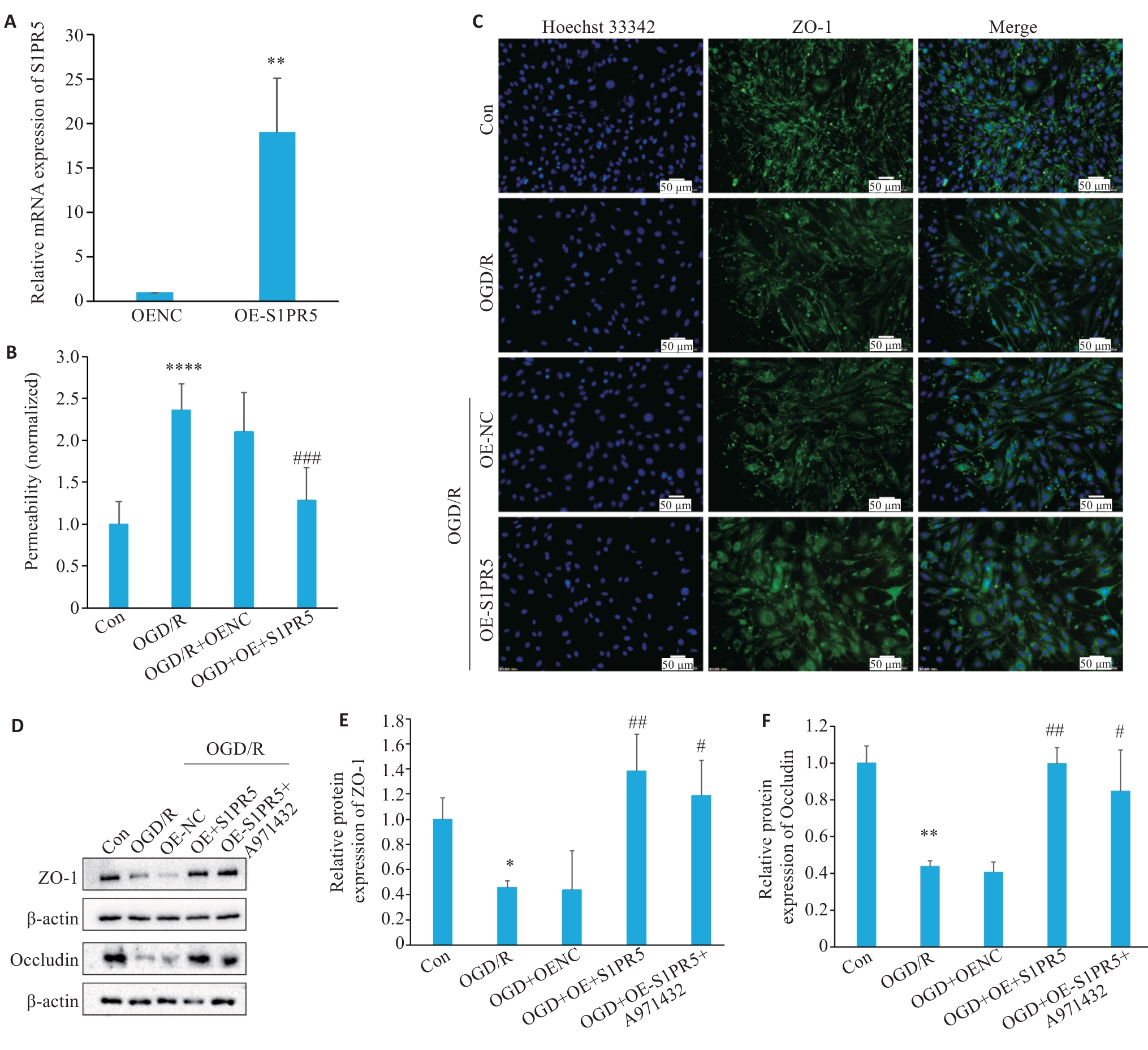
图4 过表达S1PR5抑制OGD诱导的细胞活力下降及紧密连接蛋白的减少
Fig.4 Overexpression of S1PR5 enhances viability and tight junction protein expressions in OGD-induced bEnd.3 cells. A: RT-qPCR for assessing efficiency of lentivirus-mediated S1PR5 overexpression (**P<0.01 vs OENC). B: Endothelial permeability of the cells detected by FITC-Dextran. C: Changes in the fluorescence intensity of tight junction proteins in OGD-induced bEnd.3 cells with S1PR5 overexpression. D-F: Western blotting for detecting expression levels of ZO-1 and occludin in OGD-induced bEnd.3 cells with S1PR5 overexpression. *P<0.05, **P<0.01, ****P<0.0001 vs Con; #P<0.05, ##P<0.01, ###P<0.001 vs OGD/R.
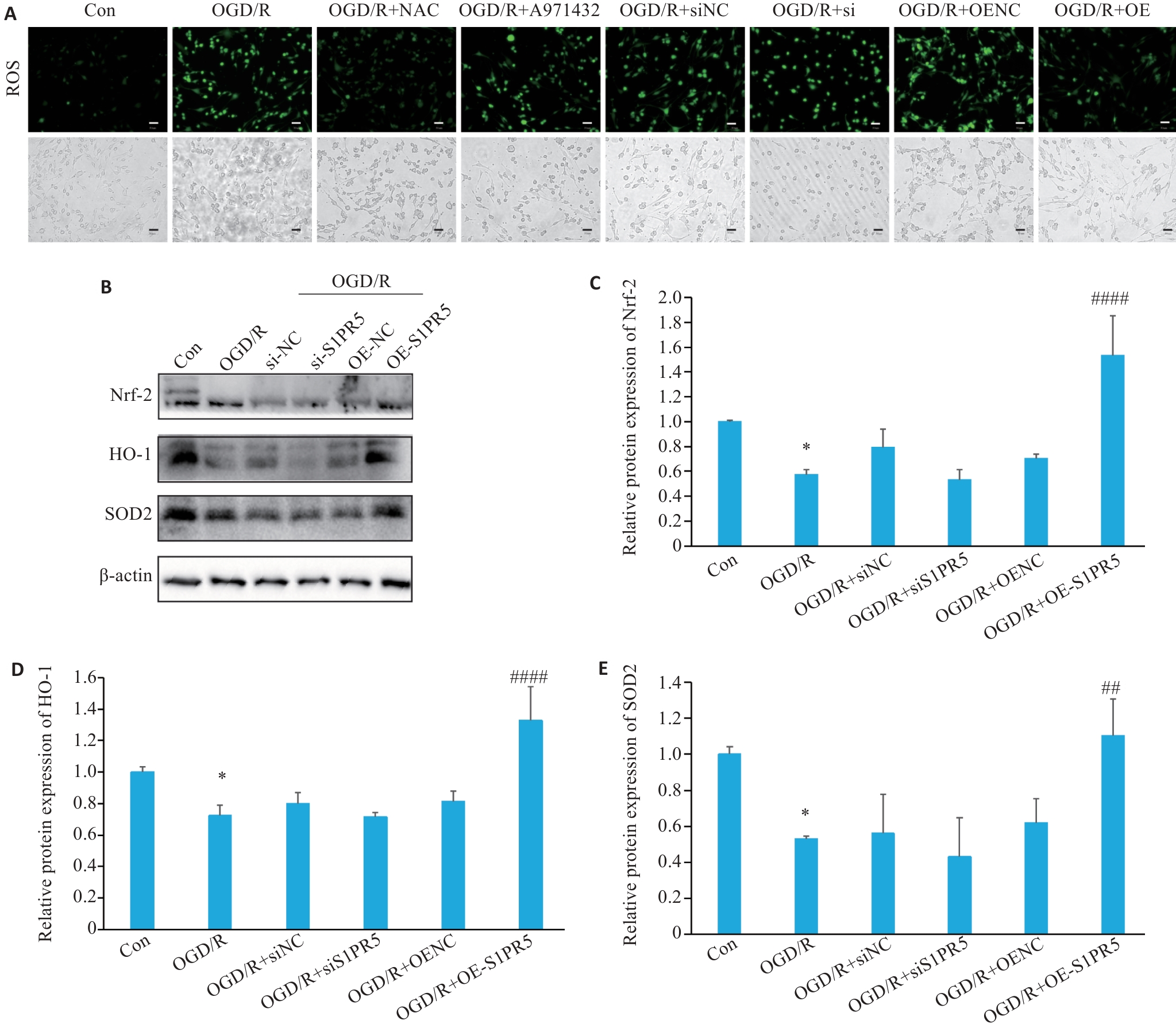
图5 S1PR5介导Nrf-2/HO-1通路发挥抗氧化作用
Fig.5 S1PR5 overexpression produces antioxidant effect in OGD-induced bEnd.3 cells via the Nrf-2/HO-1 pathway. A: Effects of S1PR5 knockdown and overexpression on intracellular reactive oxygen species in OGD-induced bEnd.3 cells (scale bar=50 μm). B-E: Western blotting for detecting expression levels of Nrf2, HO-1 and SOD2 in OGD-induced bEnd.3 cells with S1PR5 knockdown or overexpression. *P<0.05 vs Con, ##P<0.01, ####P<0.0001 vs OGD/R.
| [1] | Zhao Z, Nelson AR, Betsholtz C, et al. Establishment and dysfunction of the blood-brain barrier[J]. Cell, 2015, 163(5): 1064-78. doi:10.1016/j.cell.2015.10.067 |
| [2] | Ding QQ, Liu SW, Yao YD, et al. Global, regional, and national burden of ischemic stroke, 1990-2019[J]. Neurology, 2022, 98(3): e279-90. doi:10.1212/wnl.0000000000013115 |
| [3] | Munji RN, Soung AL, Weiner GA, et al. Profiling the mouse brain endothelial transcriptome in health and disease models reveals a core blood-brain barrier dysfunction module[J]. Nat Neurosci, 2019, 22(11): 1892-902. doi:10.1038/s41593-019-0497-x |
| [4] | Cartier A, Hla T. Sphingosine 1-phosphate: Lipid signaling in pathology and therapy[J]. Science, 2019, 366(6463): eaar5551. doi:10.1126/science.aar5551 |
| [5] | Obinata H, Hla T. Sphingosine 1-phosphate and inflammation[J]. Int Immunol, 2019, 31(9): 617-25. doi:10.1093/intimm/dxz037 |
| [6] | van Doorn R, Lopes Pinheiro MA, Kooij G, et al. Sphingosine 1-phosphate receptor 5 mediates the immune quiescence of the human brain endothelial barrier[J]. J Neuroinflammation, 2012, 9: 133. doi:10.1186/1742-2094-9-133 |
| [7] | Di Pardo A, Castaldo S, Amico E, et al. Stimulation of S1PR5 with A-971432, a selective agonist, preserves blood-brain barrier integrity and exerts therapeutic effect in an animal model of Huntington's disease[J]. Hum Mol Genet, 2018, 27(14): 2490-501. doi:10.1093/hmg/ddy153 |
| [8] | 李星阅, 任自敬, 王 越, 等. S1PR5激动剂减轻H2O2诱导的脑微血管内皮细胞高通透性及氧化应激损伤[J]. 医学研究生学报, 2022, 35(8): 820-6. |
| [9] | Matsumoto N, Yamashita T, Shang JW, et al. Up-regulation of sphingosine-1-phosphate receptors and sphingosine kinase 1 in the peri-ischemic area after transient middle cerebral artery occlusion in mice[J]. Brain Res, 2020, 1739: 146831. doi:10.1016/j.brainres.2020.146831 |
| [10] | Cao CC, Dai L, Mu JY, et al. S1PR2 antagonist alleviates oxidative stress-enhanced brain endothelial permeability by attenuating p38 and Erk1/2-dependent cPLA2 phosphorylation[J]. Cell Signal, 2019, 53: 151-61. doi:10.1016/j.cellsig.2018.09.019 |
| [11] | Jiang XY, Andjelkovic AV, Zhu L, et al. Blood-brain barrier dysfunction and recovery after ischemic stroke[J]. Prog Neurobiol, 2018, 163/164: 144-71. doi:10.1016/j.pneurobio.2017.10.001 |
| [12] | Qiu Y, Shen JY, Jiang WL, et al. Sphingosine 1-phosphate and its regulatory role in vascular endothelial cells[J]. Histol Histopathol, 2022, 37(3): 213-25. |
| [13] | Qiao N, An ZH, Fu ZY, et al. Kinsenoside alleviates oxidative stress-induced blood-brain barrier dysfunction via promoting Nrf2/HO-1 pathway in ischemic stroke[J]. Eur J Pharmacol, 2023, 949: 175717. doi:10.1016/j.ejphar.2023.175717 |
| [14] | Prager B, Spampinato SF, Ransohoff RM. Sphingosine 1-phosphate signaling at the blood-brain barrier[J]. Trends Mol Med, 2015, 21(6): 354-63. doi:10.1016/j.molmed.2015.03.006 |
| [15] | Thompson J, Yang YR, Duval K, et al. Progressive disruption of sphingosine-1-phosphate receptor 1 correlates with blood-brain barrier leakage in a rat model of chronic hypoxic hypoperfusion[J]. Aging Dis, 2024, 16(2): 1099-119. |
| [16] | Xiang P, Chew WS, Seow WL, et al. The S1P2 receptor regulates blood-brain barrier integrity and leukocyte extravasation with implications for neurodegenerative disease[J]. Neurochem Int, 2021, 146: 105018. doi:10.1016/j.neuint.2021.105018 |
| [17] | Fan XH, Chen HP, Xu C, et al. S1PR3, as a core protein related to ischemic stroke, is involved in the regulation of blood-brain barrier damage[J]. Front Pharmacol, 2022, 13: 834948. doi:10.3389/fphar.2022.834948 |
| [18] | Wu JL, Liang Y, Fu PF, et al. Sphingosine-1-phosphate receptor 3 induces endothelial barrier loss via ADAM10-mediated vascular endothelial-cadherin cleavage[J]. Int J Mol Sci, 2023, 24(22): 16083. doi:10.3390/ijms242216083 |
| [19] | Hansen LN, Lohfink N, Vutukuri R, et al. Endothelial sphingosine-1-phosphate receptor 4 regulates blood-brain barrier permeability and promotes a homeostatic endothelial phenotype[J]. J Neurosci, 2022, 42(10): 1908-29. doi:10.1523/jneurosci.0188-21.2021 |
| [20] | Spampinato SF, Merlo S, Sano Y, et al. Protective effect of the sphingosine-1 phosphate receptor agonist siponimod on disrupted blood brain barrier function[J]. Biochem Pharmacol, 2021, 186: 114465. doi:10.1016/j.bcp.2021.114465 |
| [21] | Wang ZF, Higashikawa K, Yasui H, et al. FTY720 protects against ischemia-reperfusion injury by preventing the redistribution of tight junction proteins and decreases inflammation in the subacute phase in an experimental stroke model[J]. Transl Stroke Res, 2020, 11(5): 1103-16. doi:10.1007/s12975-020-00789-x |
| [22] | Yang CJ, Hawkins KE, Doré S, et al. Neuroinflammatory mechanisms of blood-brain barrier damage in ischemic stroke[J]. Am J Physiol Cell Physiol, 2019, 316(2): C135-53. doi:10.1152/ajpcell.00136.2018 |
| [23] | Gao CL, Xu YZ, Liang ZZ, et al. A novel PGAM5 inhibitor LFHP-1c protects blood-brain barrier integrity in ischemic stroke[J]. Acta Pharm Sin B, 2021, 11(7): 1867-84. doi:10.1016/j.apsb.2021.01.008 |
| [24] | Li Y, Liao J, Xiong LY, et al. Stepwise targeted strategies for improving neurological function by inhibiting oxidative stress levels and inflammation following ischemic stroke[J]. J Control Release, 2024, 368: 607-22. doi:10.1016/j.jconrel.2024.02.039 |
| [25] | Sun YY, Zhu HJ, Zhao RY, et al. Remote ischemic conditioning attenuates oxidative stress and inflammation via the Nrf2/HO-1 pathway in MCAO mice[J]. Redox Biol, 2023, 66: 102852. doi:10.1016/j.redox.2023.102852 |
| [26] | Martín-Montañez E, Pavia J, Valverde N, et al. The S1P mimetic fingolimod phosphate regulates mitochondrial oxidative stress in neuronal cells[J]. Free Radic Biol Med, 2019, 137: 116-30. doi:10.1016/j.freeradbiomed.2019.04.022 |
| [27] | Colombo E, Bassani C, De Angelis A, et al. Siponimod (BAF312) activates Nrf2 while hampering NFκB in human astrocytes, and protects from astrocyte-induced neurodegeneration[J]. Front Immunol, 2020, 11: 635. doi:10.3389/fimmu.2020.00635 |
| [28] | Chen HW, Chen K, Huang WJ, et al. Structure of S1PR2-hetero-trimeric G13 signaling complex[J]. Sci Adv, 2022, 8(13): eabn0067. doi:10.1126/sciadv.abn0067 |
| [29] | Bellezza I, Giambanco I, Minelli A, et al. Nrf2-Keap1 signaling in oxidative and reductive stress[J]. Biochim Biophys Acta Mol Cell Res, 2018, 1865(5): 721-33. doi:10.1016/j.bbamcr.2018.02.010 |
| [30] | Fan W, Chen HP, Li M, et al. NRF2 activation ameliorates blood-brain barrier injury after cerebral ischemic stroke by regulating ferroptosis and inflammation[J]. Sci Rep, 2024, 14(1): 5300. doi:10.1038/s41598-024-53836-0 |
| [1] | 张安邦, 孙秀颀, 庞博, 吴远华, 时靖宇, 张宁, 叶涛. 电针预处理通过调节肠道-大脑轴及Nrf2/HO-1信号通路抑制铁死亡减轻大鼠脑缺血再灌注损伤[J]. 南方医科大学学报, 2025, 45(5): 911-920. |
| [2] | 高志, 吴傲, 胡仲翔, 孙培养. 类风湿性关节炎中氧化应激与免疫浸润的生物信息学分析[J]. 南方医科大学学报, 2025, 45(4): 862-870. |
| [3] | 黄鹏伟, 陈洁, 邹金虎, 高雪锋, 曹虹. 槲皮素促进应激颗粒G3BP1解聚改善HIV-1 gp120诱导的星形胶质细胞神经毒性[J]. 南方医科大学学报, 2025, 45(2): 304-312. |
| [4] | 张钰明, 夏士程, 张淋淋, 陈梦茜, 刘晓婧, 高琴, 叶红伟. 金银花提取物对小鼠阿霉素肝脏损伤的保护作用[J]. 南方医科大学学报, 2024, 44(8): 1571-1581. |
| [5] | 任志军, 刁建新, 王奕婷. 芎归汤通过抑制氧化应激诱导的心肌凋亡减轻小鼠心梗后心衰引起的心肌损伤[J]. 南方医科大学学报, 2024, 44(7): 1416-1424. |
| [6] | 梁国新, 唐红悦, 郭畅, 张明明. miR-224-5p调控PI3K/Akt/FoxO1轴抑制氧化应激减轻缺氧/复氧诱导的心肌细胞损伤[J]. 南方医科大学学报, 2024, 44(6): 1173-1181. |
| [7] | 陈国栋, 罗素新. 秋水仙碱通过激活AMPK减轻小鼠心肌缺血再灌注损伤[J]. 南方医科大学学报, 2024, 44(2): 226-235. |
| [8] | 凌旭光, 徐雯雯, 庞观来, 洪旭星, 刘凤芹, 李 洋. 茶多酚通过抑制NLRP3炎症小体改善脓毒症小鼠的急性肺损伤[J]. 南方医科大学学报, 2024, 44(2): 381-386. |
| [9] | 孙晓鹏, 史 航, 张 磊, 刘 中, 李克威, 钱玲玲, 朱星宇, 杨康佳, 付 强, 丁 华. 外胚层间充质干细胞来源的外泌体通过控制炎症和氧化损伤减少M1型小胶质细胞并促进H2O2处理后PC12细胞的存活[J]. 南方医科大学学报, 2024, 44(1): 119-128. |
| [10] | 叶红伟, 张钰明, 云 琦, 杜若丽, 李 璐, 李玉萍, 高 琴. 白藜芦醇可减轻高糖诱导的心肌细胞肥大:基于促进SIRT1表达维持线粒体稳态[J]. 南方医科大学学报, 2024, 44(1): 45-51. |
| [11] | 蒙 诺, 杨慧莹, 陈金凤, 覃颖颖, 雷 宇, 黄振宁, 唐国都. 和厚朴酚减轻高甘油三酯血症急性胰腺炎大鼠模型的氧化应激:基于激活SIRT3-MnSOD2通路[J]. 南方医科大学学报, 2023, 43(3): 405-411. |
| [12] | 陈 兴, 王开万, 储德海, 朱 羽, 张文兵, 曹慧萍, 谢文宇, 鲁传豪, 李 侠. 连翘酯苷B抑制小鼠脑缺血/再灌注引起的氧化应激损伤:基于激活AMPK/DAF-16/FOXO3通路[J]. 南方医科大学学报, 2023, 43(2): 199-205. |
| [13] | 郑冬晓, 陈琳琳, 韦其慧, 朱梓然, 刘子略, 金 琳, 杨观玉, 谢 曦. 褐藻素通过调控Nrf2/Keap1通路缓解糖尿病大鼠心肌肥大[J]. 南方医科大学学报, 2022, 42(5): 752-759. |
| [14] | 朱妍妍, 王桐生, 戴 宁, 邓梦云, 刘红娟, 童小慧, 李 莉. 金丝桃苷通过激活Keap1/Nrf2/HO-1通路保护小鼠GC-2细胞的氧化损伤[J]. 南方医科大学学报, 2022, 42(5): 673-680. |
| [15] | 邵晓丽, 余江毅, 倪卫惠. 汉黄芩苷通过上调SIRT1表达减轻糖尿病视网膜病变引起的细胞和组织损伤[J]. 南方医科大学学报, 2022, 42(4): 463-472. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||