Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 479-488.doi: 10.12122/j.issn.1673-4254.2026.03.01
Chaoqun LIU1,2( ), Ziyan NING2(
), Ziyan NING2( ), Jianghua WU1,2, Weiwei LIU2, Chuang LIN1, Jiawei XU3, Rui ZHOU1,2(
), Jianghua WU1,2, Weiwei LIU2, Chuang LIN1, Jiawei XU3, Rui ZHOU1,2( ), Liang ZHAO1,2(
), Liang ZHAO1,2( )
)
Received:2025-10-28
Online:2026-03-20
Published:2026-03-26
Contact:
Rui ZHOU, Liang ZHAO
E-mail:2436965229@qq.com;13277483682@163.com;yaruisunny@sina.com;liangsmu@foxmail.com
Supported by:Chaoqun LIU, Ziyan NING, Jianghua WU, Weiwei LIU, Chuang LIN, Jiawei XU, Rui ZHOU, Liang ZHAO. Tumor-secreted dentin sialophosphoprotein induces oxaliplatin resistance in colorectal cancer through an integrin αvβ3-dependent pathway[J]. Journal of Southern Medical University, 2026, 46(3): 479-488.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.01
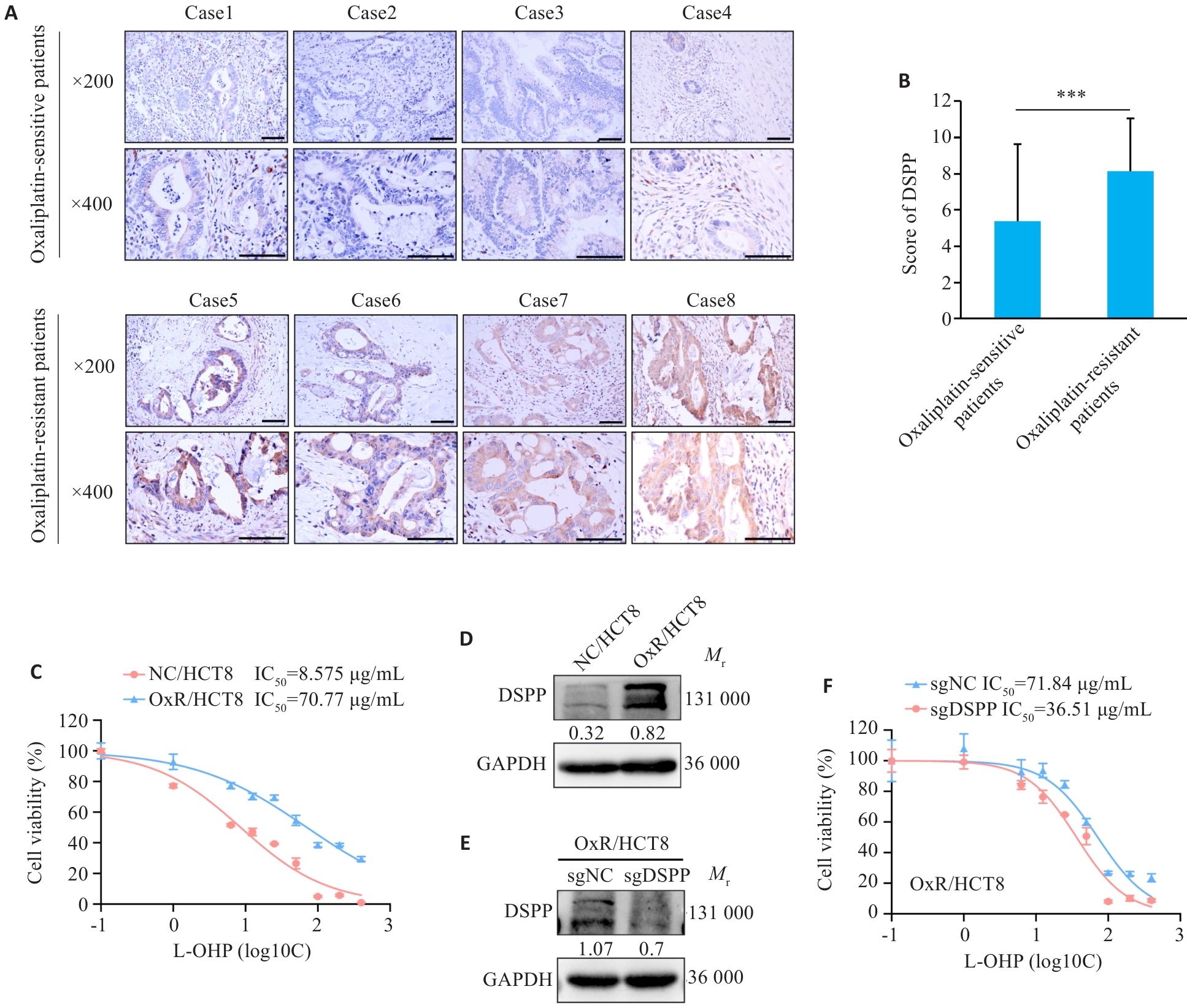
Fig.1 DSPP correlates with oxaliplatin responsiveness in colorectal cancer (CRC). A: Immunohistochemical (IHC) images showing DSPP expression in tumors from oxaliplatin (L-OHP)‑sensitive and ‑resistant CRC patients (***P<0.001). B: Quantification of DSPP IHC scores in L-OHP-sensitive and -resistant CRC tumors (n=30). C: CCK-8 assay for assessing viability of L-OHP-resistant HCT8 cell line (n=4). D: Western blotting of DSPP in L-OHP-sensitive HCT8 and L-OHP-resistant HCT8 cells. E: Western blotting confirming stable DSPP knockout in HCT8 cells. F: CCK-8 assay showing increased L-OHP sensitivity in DSPP-knockout HCT8 cells (n=4).
| Characteristic | Level | Oxaliplatin-sensitive group (n=30) | Oxaliplatin-resistant group (n=30) | P |
|---|---|---|---|---|
Tab.1 Baseline characteristics of oxaliplatin-sensitive and oxaliplatin-resistant colorectal cancer (CRC) samples
| Characteristic | Level | Oxaliplatin-sensitive group (n=30) | Oxaliplatin-resistant group (n=30) | P |
|---|---|---|---|---|
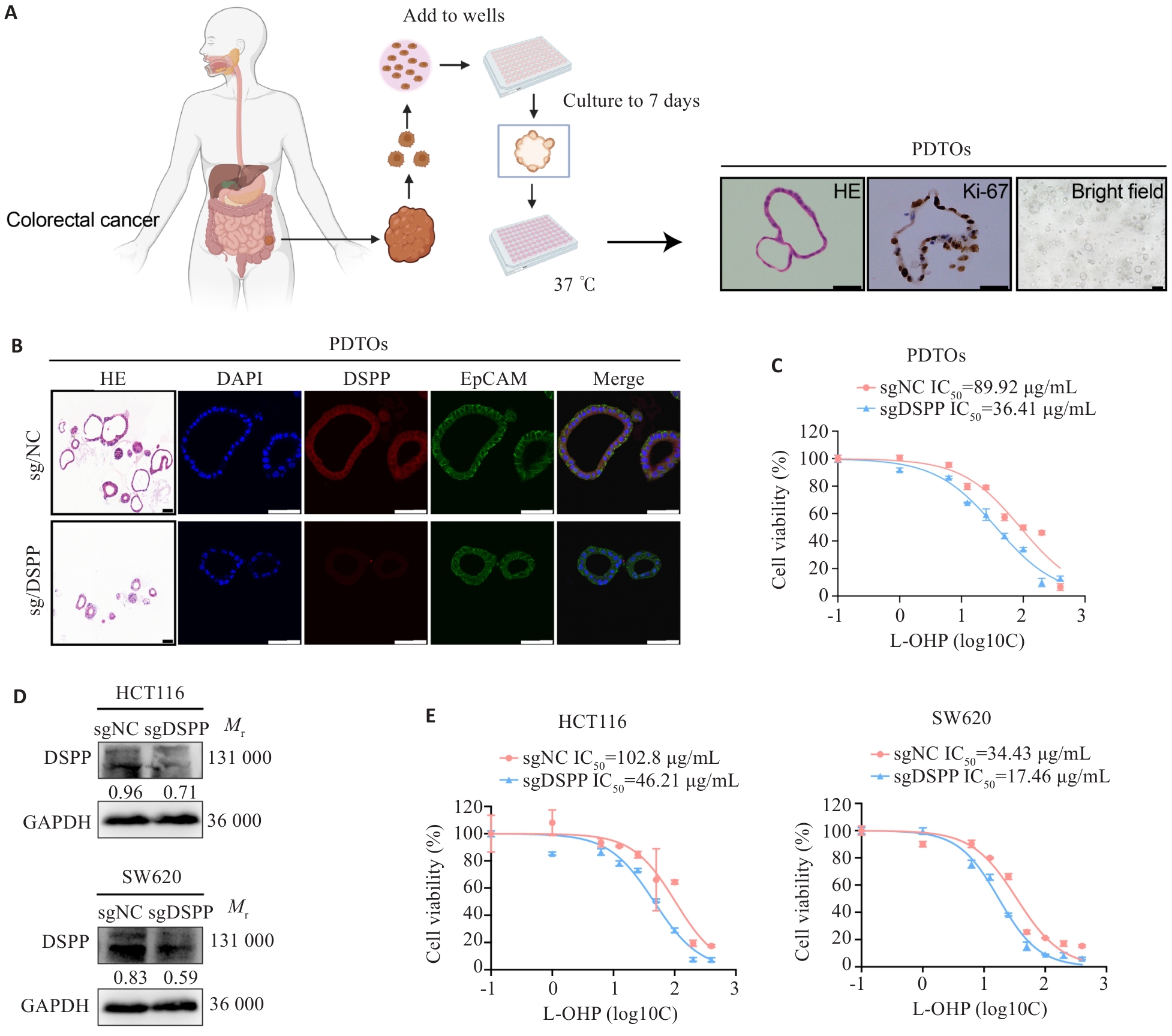
Fig.2 DSPP depletion enhances oxaliplatin resistance in human CRC organoids and cell lines. A: Representative schematic of the establishment and characterization of patient-derived tumor organoids (PDTOs). Scale bar=100 μm. B: HE and immunofluorescence staining showing DSPP expression in edited PDTOs. Scale bar=100 μm. C: CCK-8 assay showing significantly increased L-OHP sensitivity in DSPP-knockout PDTOs (n=4). D: Western blotting showing the efficiency of DSPP knockout in HCT116 and SW620 cells. E: CCK-8 assays showing increased L-OHP sensitivity in DSPP knockout HCT116 and SW620 cells (n=4).
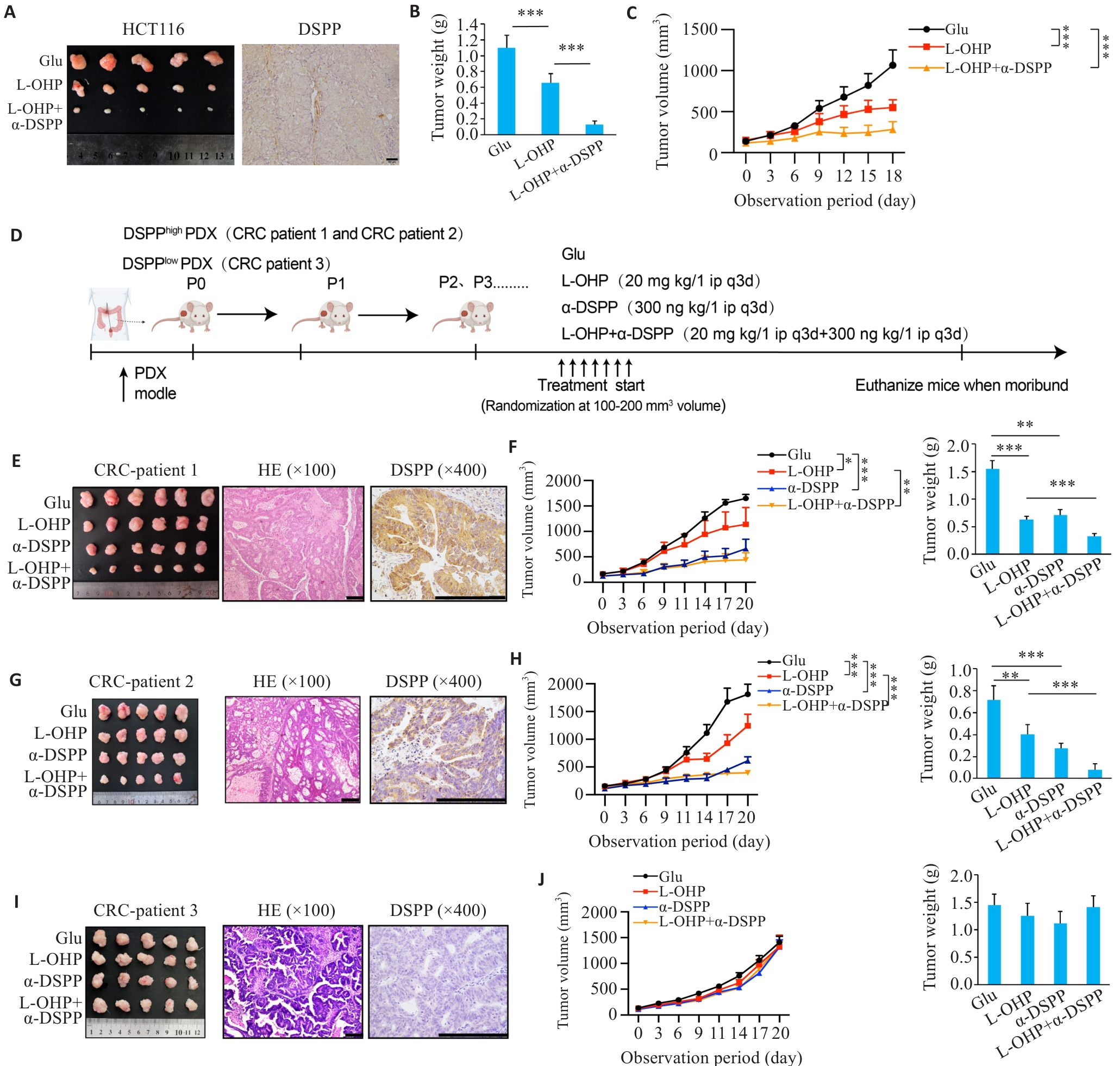
Fig.3 Targeting DSPP augments oxaliplatin efficacy in xenograft and PDX models.A: Representative subcutaneous xenograft tumors and IHC demonstrating the efficacy of combining L-OHP with a DSPP-targeting therapy (n=5). Scale bar=50 μm. B: Endpoint tumor weights in each group (n=5). C: Tumor growth curves over time in each group (n=5). D: Schematic diagram of patient-derived xenograft (PDX) establishment and treatment regimens. E, G, I: Representative macroscopic tumors, HE staining, and DSPP IHC from 3 independent PDX cases (n=5 or 6). Scale bar=200 μm. F, H, J: Tumor growth curves and tumor weights in the corresponding PDX cases. *P<0.05, **P<0.01, ***P<0.001.
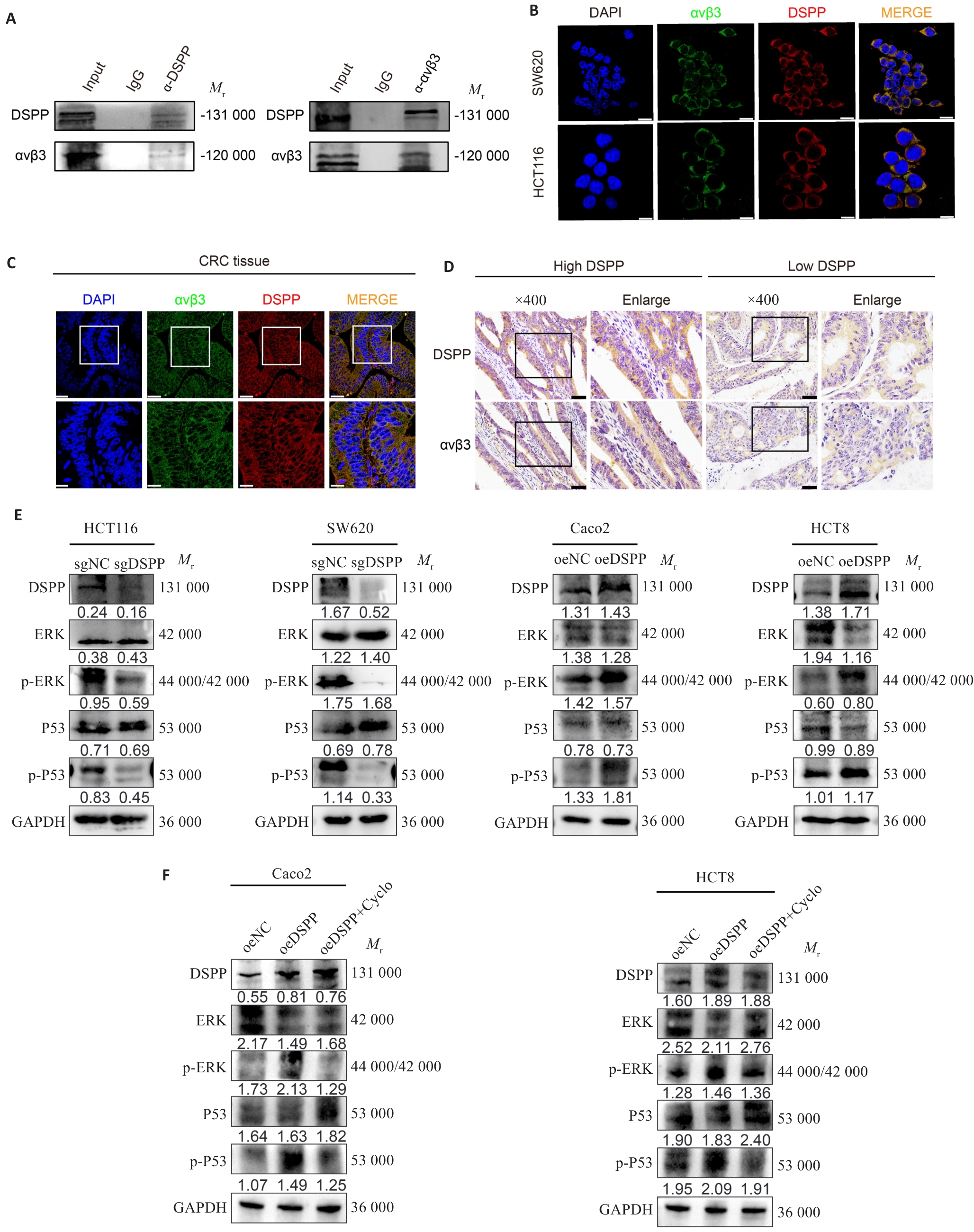
Fig.4 DSPP interacts with integrin αvβ3 and modulates MAPK signaling. A, B: Co-immunoprecipitation (Co-IP) and immunofluorescence images showing the interaction and subcellular co-localization of DSPP and integrin αvβ3 in CRC cells. Scale bar=10 μm. C: Immunofluorescence images showing co-localization of DSPP and αvβ3 in CRC tissues. Scale bar=50 μm. D: IHC showing the correlation between DSPP and integrin αvβ3 expression in human CRC specimens. Scale bar=50 μm. E, F: Western blotting showing that DSPP regulates MAPK/ERK and p53 pathways in the indicated CRC cell lines, and the integrin-targeted inhibitor Cyclo (at a concentration of 38.33 ng/mL) reverses DSPP-induced upregulation of ERK and P53 phosphorylation levels.
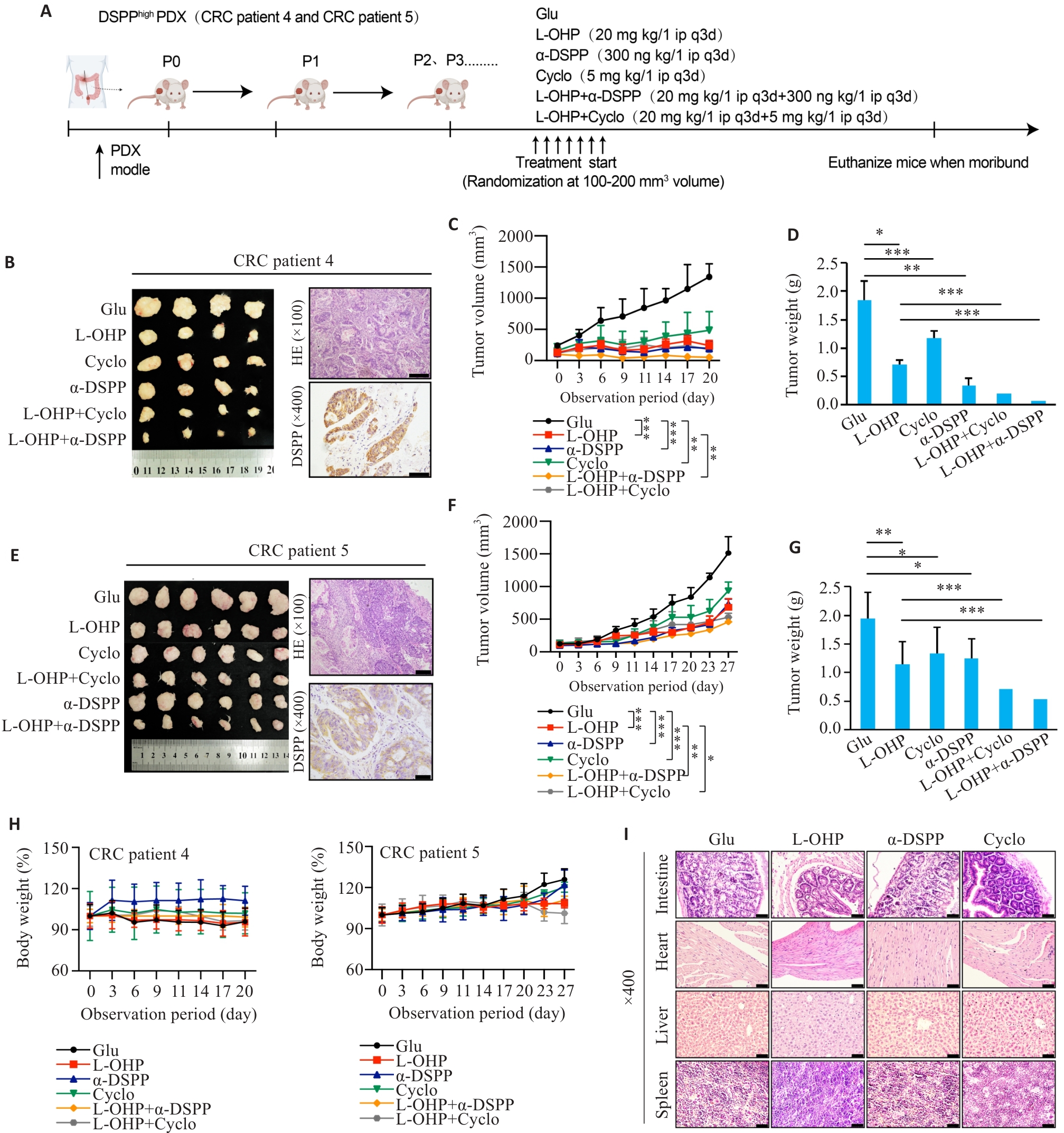
Fig.5 Targeting DSPP or integrin αvβ3 enhances oxaliplatin efficacy in CRC PDX models. A: Schematic diagram of the PDX experimental design for evaluating L-OHP combined with DSPP-targeting antibody or αvβ3 inhibitor Cyclo(-RGDfK). B, E: Representative macroscopic tumors, HE staining, and DSPP IHC from two independent PDX cases (n=4-6). Scale bar=100 μm. C, F: Tumor growth curves for the indicated treatment groups (n=4-6). D, G: Endpoint tumor weights in each group (n=4-6). H: Body-weight monitoring of PDX mice across different treatment groups, showing no significant toxicity (n=4-6). I: HE staining of major organs (intestine, liver, heart, and spleen) from PDX mice across the treatment groups. Scale bar=50 μm. *P<0.05, **P<0.01, ***P<0.001.
| [1] | Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025[J]. CA A Cancer J Clin, 2025, 75(1): 10-45. doi:10.3322/caac.21871 |
| [2] | Filho AM, Laversanne M, Ferlay J, et al. The GLOBOCAN 2022 cancer estimates: Data sources, methods, and a snapshot of the cancer burden worldwide[J]. Int J Cancer, 2025, 156(7): 1336-46. doi:10.1002/ijc.35278 |
| [3] | Han BF, Zheng RS, Zeng HM, et al. Cancer incidence and mortality in China, 2022[J]. J Natl Cancer Cent, 2024, 4(1): 47-53. doi:10.1016/j.jncc.2024.01.006 |
| [4] | Ciardiello F, Ciardiello D, Martini G, et al. Clinical management of metastatic colorectal cancer in the era of precision medicine[J]. CA Cancer J Clin, 2022, 72(4): 372-401. doi:10.3322/caac.21728 |
| [5] | Van Cutsem E, Cervantes A, Adam R, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer[J]. Ann Oncol, 2016, 27(8): 1386-422. |
| [6] | Bellahcène A, Castronovo V, Ogbureke KUE, et al. Small integrin-binding ligand N-linked glycoproteins (SIBLINGs): multifunctional proteins in cancer[J]. Nat Rev Cancer, 2008, 8(3): 212-26. doi:10.1038/nrc2345 |
| [7] | Fan SC, Gao HS, Sun LZ, et al. Knockdown of DSPP inhibits the migration and invasion of glioma cells[J]. Pathol Res Pract, 2018, 214(12): 2025-30. doi:10.1016/j.prp.2018.09.024 |
| [8] | Gu Y, Taifour T, Bui T, et al. Osteopontin is a therapeutic target that drives breast cancer recurrence[J]. Nat Commun, 2024, 15(1): 9174. doi:10.1038/s41467-024-53023-9 |
| [9] | Aseervatham J, Ogbureke KUE. Effects of DSPP and MMP20 silencing on adhesion, metastasis, angiogenesis, and epithelial-mesenchymal transition proteins in oral squamous cell carcinoma cells[J]. Int J Mol Sci, 2020, 21(13): 4734. doi:10.3390/ijms21134734 |
| [10] | Nikitakis NG, Gkouveris I, Aseervatham J, et al. DSPP-MMP20 gene silencing downregulates cancer stem cell markers in human oral cancer cells[J]. Cell Mol Biol Lett, 2018, 23: 30. doi:10.1186/s11658-018-0096-y |
| [11] | Liu D, Luo M, Hu J, et al. Osteopontin enhances cisplatin resistance of human A549 lung cancer cells via stimulating the PI3K signaling pathway and upregulating ERCC1 expression[J]. Transl Cancer Res, 2020, 9(5): 3258-65. doi:10.21037/tcr.2020.03.60 |
| [12] | Panda VK, Mishra B, Nath AN, et al. Osteopontin: a key multifaceted regulator in tumor progression and immunomodulation[J]. Biomedicines, 2024, 12(7): 1527. doi:10.3390/biomedicines12071527 |
| [13] | Qin YC, Yan X, Yuan XL, et al. Osteopontin promotes gastric cancer progression via phosphatidylinositol-3-kinase/protein kinase B/mammalian target of rapamycin signaling pathway[J]. World J Gastrointest Oncol, 2023, 15(9): 1544-55. doi:10.4251/wjgo.v15.i9.1544 |
| [14] | Gu M, Zheng XY. Osteopontin and vasculogenic mimicry formation are associated with response to neoadjuvant chemotherapy in advanced breast cancer[J]. OncoTargets Ther, 2017, 10: 4121-7. doi:10.2147/ott.s129414 |
| [15] | Göthlin Eremo A, Lagergren K, Othman L, et al. Evaluation of SPP1/osteopontin expression as predictor of recurrence in tamoxifen treated breast cancer[J]. Sci Rep, 2020, 10(1): 1451. doi:10.1038/s41598-020-58323-w |
| [16] | Qian J, LeSavage BL, Hubka KM, et al. Cancer-associated mesothelial cells promote ovarian cancer chemoresistance through paracrine osteopontin signaling[J]. J Clin Invest, 2021, 131(16): e146186. doi:10.1172/jci146186 |
| [17] | Sanchis P, Sabater A, Lechuga J, et al. PKA-driven SPP1 activation as a novel mechanism connecting the bone microenvironment to prostate cancer progression[J]. Oncogene, 2025, 44(38): 3568-79. doi:10.1038/s41388-025-03511-z |
| [18] | Fu YL, Zhang Y, Lei Z, et al. Abnormally activated OPN/integrin αVβ3/FAK signalling is responsible for EGFR-TKI resistance in EGFR mutant non-small-cell lung cancer[J]. J Hematol Oncol, 2020, 13(1): 169. doi:10.1186/s13045-020-01009-7 |
| [19] | Gkouveris I, Nikitakis NG, Aseervatham J, et al. The tumorigenic role of DSPP and its potential regulation of the unfolded protein response and ER stress in oral cancer cells[J]. Int J Oncol, 2018, 53(4): 1743-51. doi:10.3892/ijo.2018.4484 |
| [20] | Faraji SN, Nejatollahi F, Tamaddon AM, et al. Generation and characterization of a specific single-chain antibody against DSPP as a prostate cancer biomarker: Involvement of bioinformatics-based design of novel epitopes[J]. Int Immunopharmacol, 2019, 69: 217-24. doi:10.1016/j.intimp.2019.01.016 |
| [21] | Chaplet M, Waltregny D, Detry C, et al. Expression of dentin sialophosphoprotein in human prostate cancer and its correlation with tumor aggressiveness[J]. Int J Cancer, 2006, 118(4): 850-6. doi:10.1002/ijc.21442 |
| [22] | Mascha EJ, Vetter TR. Significance, errors, power, and sample size: the blocking and tackling of statistics: erratum[J]. Anesth Analg, 2018, 126(4): 1429. doi:10.1213/ane.0000000000002887 |
| [23] | Zhong Y, Long T, Gu CS, et al. MYH9-dependent polarization of ATG9B promotes colorectal cancer metastasis by accelerating focal adhesion assembly[J]. Cell Death Differ, 2021, 28(12): 3251-69. doi:10.1038/s41418-021-00813-z |
| [24] | Liu CQ, Pan ZH, Chen Q, et al. Pharmacological targeting PTK6 inhibits the JAK2/STAT3 sustained stemness and reverses chemoresistance of colorectal cancer[J]. J Exp Clin Cancer Res, 2021, 40(1): 297. doi:10.1186/s13046-021-02059-6 |
| [25] | Lieu C, Kennedy EB, Bergsland E, et al. Duration of oxaliplatin-containing adjuvant therapy for stage III colon cancer: ASCO clinical practice guideline[J]. J Clin Oncol, 2019, 37(16): 1436-47. doi:10.1200/jco.19.00281 |
| [26] | Kee JX, Yau JNN, Kumar Muthuramalingam RP, et al. Colorectal cancer at the crossroads: the good, the bad, and the future of platinum-based drugs[J]. Chem Rev, 2025, 125(21): 10248-341. doi:10.1021/acs.chemrev.5c00041 |
| [27] | Liu XW, Zhang SM, An Y, et al. USP10/XAB2/ANXA2 axis promotes DNA damage repair to enhance chemoresistance to oxaliplatin in colorectal cancer[J]. J Exp Clin Cancer Res, 2025, 44(1): 94. doi:10.1186/s13046-025-03357-z |
| [28] | Ding DB, Li WB, Ren JN, et al. SPP1(high) macrophage-induced T-cell stress promotes colon cancer liver metastasis through SPP1/CD44/PI3K/AKT signaling[J]. J Immunother Cancer, 2025, 13(10): e012330. doi:10.1136/jitc-2025-012330 |
| [29] | Zhang H, Guo M, Chen JH, et al. Osteopontin knockdown inhibits αv, β3 integrin-induced cell migration and invasion and promotes apoptosis of breast cancer cells by inducing autophagy and inactivating the PI3K/Akt/mTOR pathway[J]. Cell Physiol Biochem, 2014, 33(4): 991-1002. doi:10.1159/000358670 |
| [30] | Hidalgo M, Bruckheimer E, Rajeshkumar NV, et al. A pilot clinical study of treatment guided by personalized tumorgrafts in patients with advanced cancer[J]. Mol Cancer Ther, 2011, 10(8): 1311-6. doi:10.1158/1535-7163.mct-11-0233 |
| [31] | Aparicio S, Hidalgo M, Kung AL. Examining the utility of patient-derived xenograft mouse models[J]. Nat Rev Cancer, 2015, 15(5): 311-6. doi:10.1038/nrc3944 |
| [32] | Liu GK, Fan XY, Tang M, et al. Osteopontin induces autophagy to promote chemo-resistance in human hepatocellular carcinoma cells[J]. Cancer Lett, 2016, 383(2): 171-82. doi:10.1016/j.canlet.2016.09.033 |
| [33] | Ding K, Fan L, Chen SJ, et al. Overexpression of osteopontin promotes resistance to cisplatin treatment in HCC[J]. Oncol Rep, 2015, 34(6): 3297-303. doi:10.3892/or.2015.4306 |
| [34] | Di YQ, Zhang X, Wen XQ, et al. MAPK signaling-mediated RFNG phosphorylation and nuclear translocation restrain oxaliplatin-induced apoptosis and ferroptosis[J]. Adv Sci, 2024, 11(38): 2402795. doi:10.1002/advs.202402795 |
| [35] | Lin JF, Liu ZX, Chen DL, et al. Nucleus-translocated GCLM promotes chemoresistance in colorectal cancer through a moonlighting function[J]. Nat Commun, 2025, 16(1): 263. doi:10.1038/s41467-024-55568-1 |
| [1] | Kang WANG, Haibin LI, Jing YU, Yuan MENG, Hongli ZHANG. High expression of ELFN1 is a prognostic biomarker and promotes proliferation and metastasis of colorectal cancer cells [J]. Journal of Southern Medical University, 2025, 45(7): 1543-1553. |
| [2] | Nuozhou WENG, Bin TAN, Wentao ZENG, Jiayu GU, Lianji WENG, Kehong ZHENG. RGL1 overexpression promotes metastasis of colorectal cancer by upregulating motile focal adhesion assembly via activating the CDC42/RAC1 complex [J]. Journal of Southern Medical University, 2025, 45(5): 1031-1038. |
| [3] | Zhennan MA, Fuquan LIU, Xuefeng ZHAO, Xiaowei ZHANG. High expression of DTX2 promotes proliferation, invasion and epithelial-mesenchymal transition of oxaliplatin-resistant colorectal cancer cells [J]. Journal of Southern Medical University, 2025, 45(4): 829-836. |
| [4] | Xiawei ZHANG, Jingjing YANG, Yanan WEN, Qingyang LIU, Liping DOU, Chunji GAO. METTL3-mediated m6A modification promotes FOXO3 expression and anthracycline resistance in acute myeloid leukemia cells through autophagy regulation [J]. Journal of Southern Medical University, 2025, 45(3): 470-478. |
| [5] | Shunjie QING, Zhiyong SHEN. High expression of hexokinase 2 promotes proliferation, migration and invasion of colorectal cancer cells by activating the JAK/STAT pathway and regulating tumor immune microenvironment [J]. Journal of Southern Medical University, 2025, 45(3): 542-553. |
| [6] | Zhenni YU, Jingzhe GAO, Hui SUN, Qin Feng, Xiaoqi NA, Ning ZHANG, Kungshuang SHEN, Yuanyuan WANG, Xijun WANG. Causal relationship between gut microbiota and T cell subsets in the development of colorectal cancer: a Mendelian randomization analysis [J]. Journal of Southern Medical University, 2025, 45(12): 2756-2766. |
| [7] | Yue DU, Xiusen ZHANG, Kexu ZHOU, Xing JIN, Xiang YUAN, Shegan GAO. RgpB contributes to chemoresistance in esophageal squamous cell carcinoma by preventing Cx43 degradation via inhibiting autophagosome-lysosome fusion [J]. Journal of Southern Medical University, 2024, 44(9): 1670-1676. |
| [8] | Kai JI, Guanyu YU, Leqi ZHOU, Tianshuai ZHANG, Qianlong LING, Wenjiang MAN, Bing ZHU, Wei ZHANG. HNRNPA1 gene is highly expressed in colorectal cancer: its prognostic implications and potential as a therapeutic target [J]. Journal of Southern Medical University, 2024, 44(9): 1685-1695. |
| [9] | Yinliang ZHANG, Zetan LUO, Rui ZHAO, Na ZHAO, Zhidong XU, Di AO, Guyi CONG, Xinyu LIU, Hailun ZHENG. Sanguinarine induces ferroptosis of colorectal cancer cells by upregulating STUB1 and downregulating GPX4 [J]. Journal of Southern Medical University, 2024, 44(8): 1537-1544. |
| [10] | Heping LI, Gaohua LI, Xuehua ZHANG, Yanan WANG. Genetic drivers for inflammatory protein markers in colorectal cancer: a Mendelian randomization approach to clinical prognosis study [J]. Journal of Southern Medical University, 2024, 44(7): 1361-1370. |
| [11] | Nan WANG, Bin SHI, Xiaolan MAN, Weichao WU, Jia CAO. High expression of fragile X mental retardation protein inhibits ferroptosis of colorectal tumor cells by activating the RAS/MAPK signaling pathway [J]. Journal of Southern Medical University, 2024, 44(5): 885-893. |
| [12] | Xueyan XI, Ting DENG, Boyu DU. Colorectal fibroblasts promote malignant phenotype of colorectal cancer cells by activating the ERK signaling pathway [J]. Journal of Southern Medical University, 2024, 44(10): 1866-1873. |
| [13] | YAN Chang, LIU Shuang, SONG Qingzhi, HU Yibing. Metformin inhibits self-renewal of colorectal cancer stem cells by inhibiting mitochondrial oxidative phosphorylation [J]. Journal of Southern Medical University, 2023, 43(8): 1279-1286. |
| [14] | WEI Ke, SHI Jiwen, XIAO Yuhan, WANG Wenrui, YANG Qingling, CHEN Changjie. MiR-30e-5p overexpression promotes proliferation and migration of colorectal cancer cells by activating the CXCL12 axis via downregulating PTEN [J]. Journal of Southern Medical University, 2023, 43(7): 1081-1092. |
| [15] | ZHANG Xuefang, CHEN Yanhua, LI Zongheng, SHANG Jing, YUAN Zeting, DENG Wanli, LUO Ying, HAN Na, YIN Peihao, YIN Jun. Analysis of therapeutic mechanism of Liushen Wan against colitis-associated colorectal cancer based on network pharmacology and validation in mice [J]. Journal of Southern Medical University, 2023, 43(7): 1051-1062. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||