Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 655-665.doi: 10.12122/j.issn.1673-4254.2026.03.19
Hexin WEN1( ), Jie LIN2, Lugen ZUO1(
), Jie LIN2, Lugen ZUO1( ), Mulin LIU1(
), Mulin LIU1( )
)
Received:2025-08-03
Online:2026-03-20
Published:2026-03-26
Contact:
Lugen ZUO, Mulin LIU
E-mail:wenhexin66@126.com;zuolugen@126.com;liumulin66@aliyun.com
Hexin WEN, Jie LIN, Lugen ZUO, Mulin LIU. Niranthin ameliorates Crohn's disease-like colitis in mice via antagonizing intestinal epithelial cell apoptosis and regulating intestinal Th1/Th2 immune homeostasis[J]. Journal of Southern Medical University, 2026, 46(3): 655-665.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.19
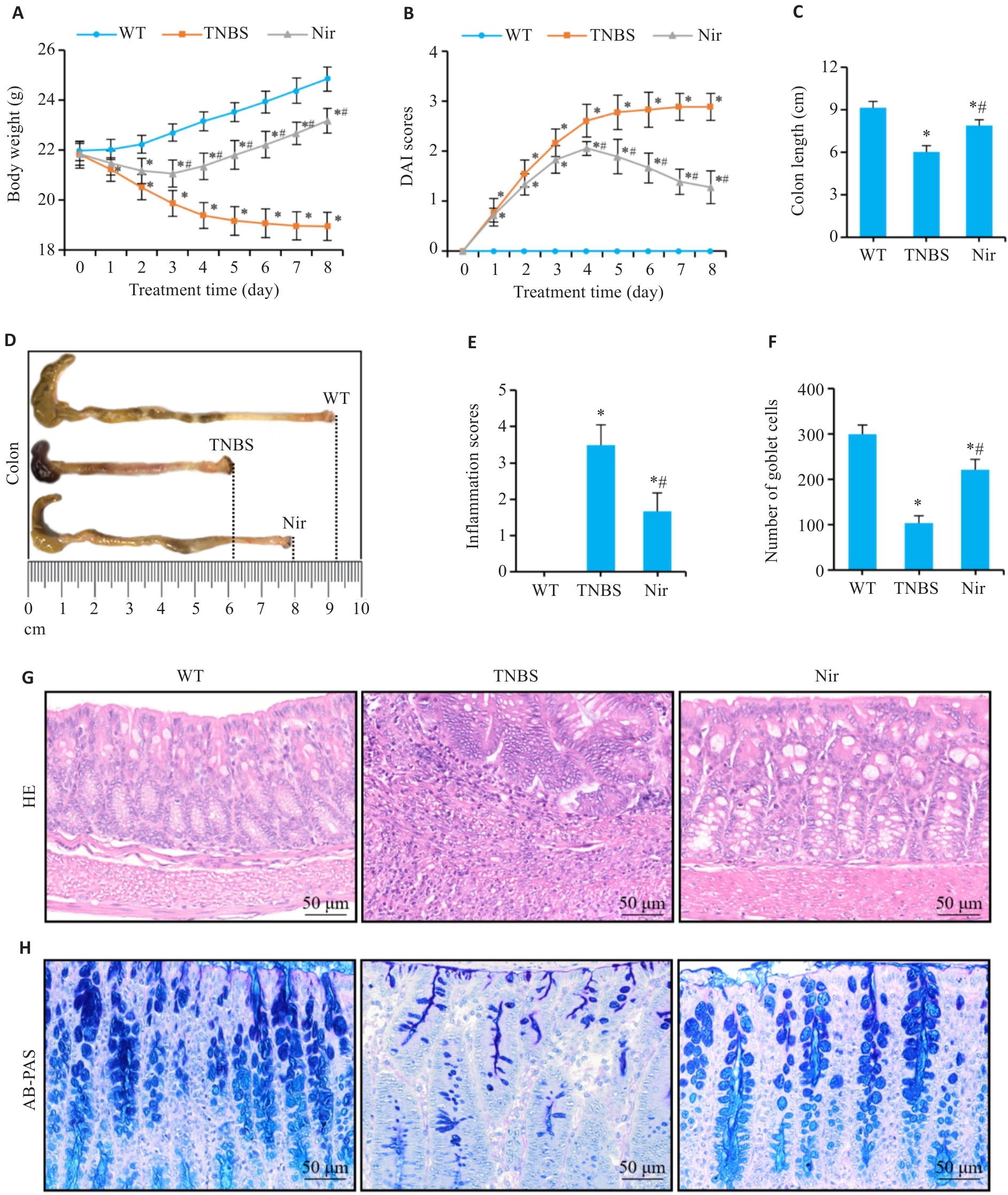
Fig.1 Effects of niranthin (Nir) intervention in mice with TNBS-induced colitis. A: Body weight changes. B: DAI scores. C, D: Colon length. E, G: Inflammation scores and HE staining of mouse colon tissues. F, H: Number of goblet cells and AB-PAS staining. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
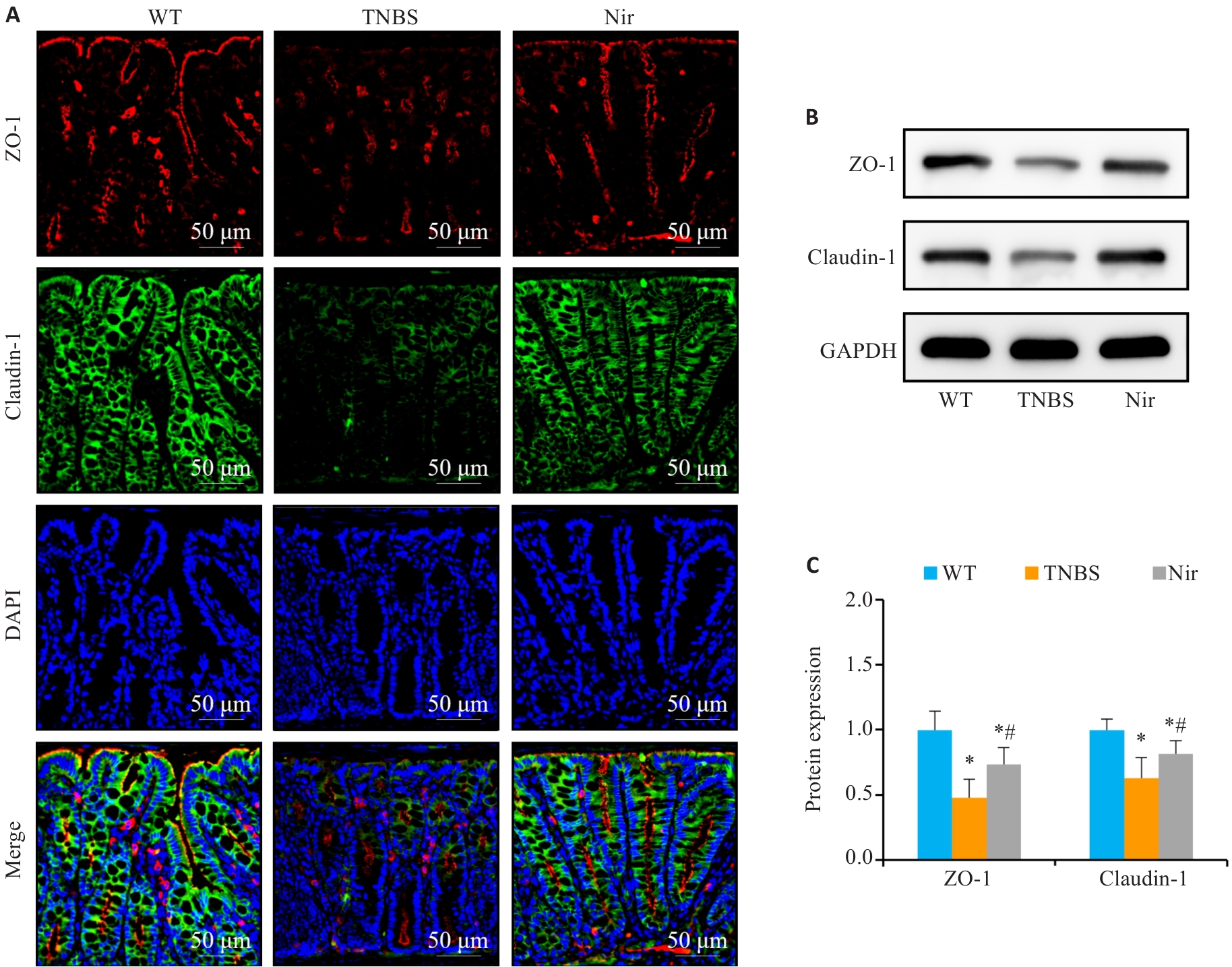
Fig.2 Effects of Nir intervention on intestinal barrier function in TNBS-induced colitis mice. A: Immunofluorescence assay of ZO-1 and claudin-1 expression. B, C: Western blotting for claudin-1 and ZO-1 expressions. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
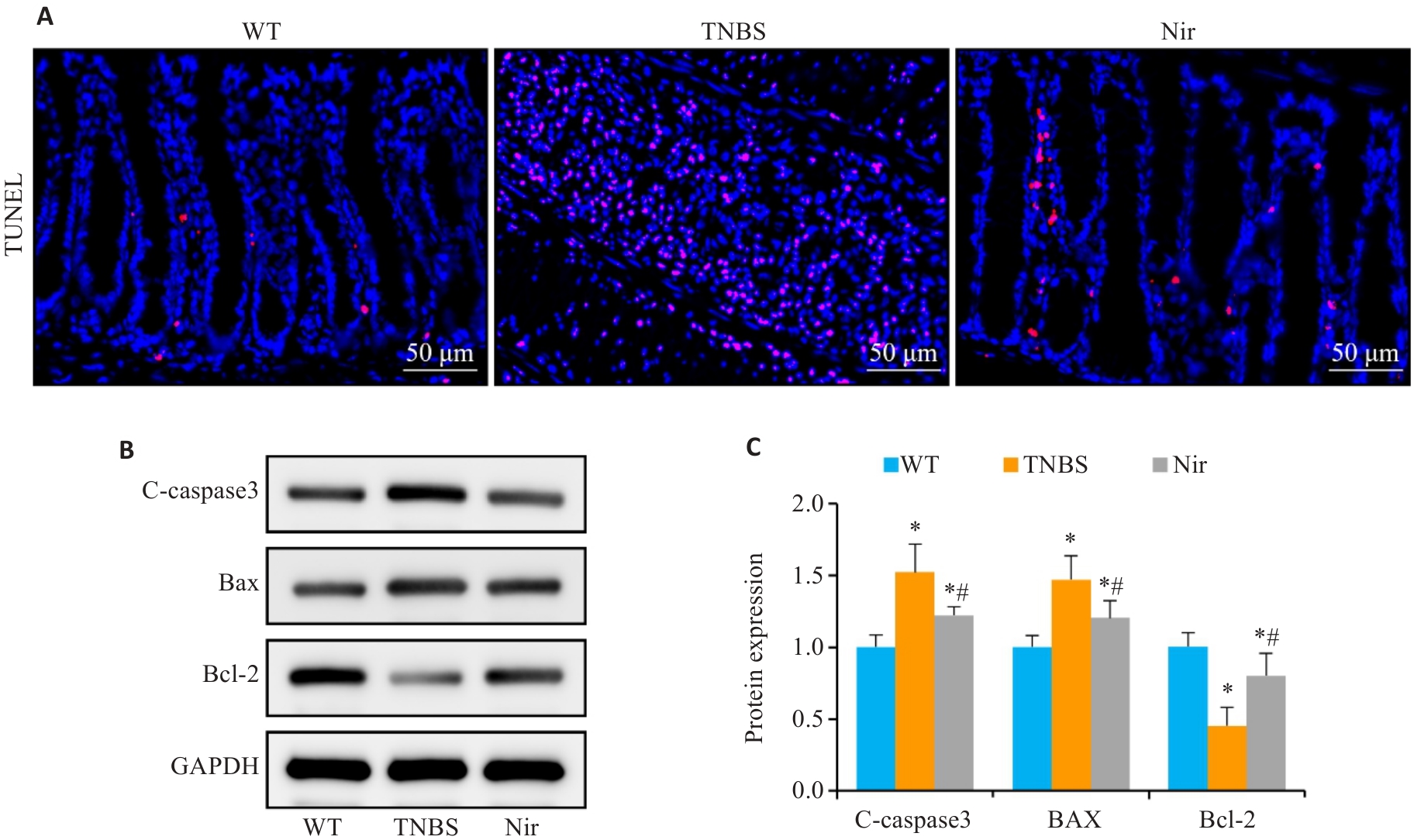
Fig.3 The effect of Nir intervention on colonic cell apoptosis in TNBS-induced colitis mice. A: TUNEL staining. B, C: Western blotting for C-caspase-3, Bax and Bcl-2 expressions. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
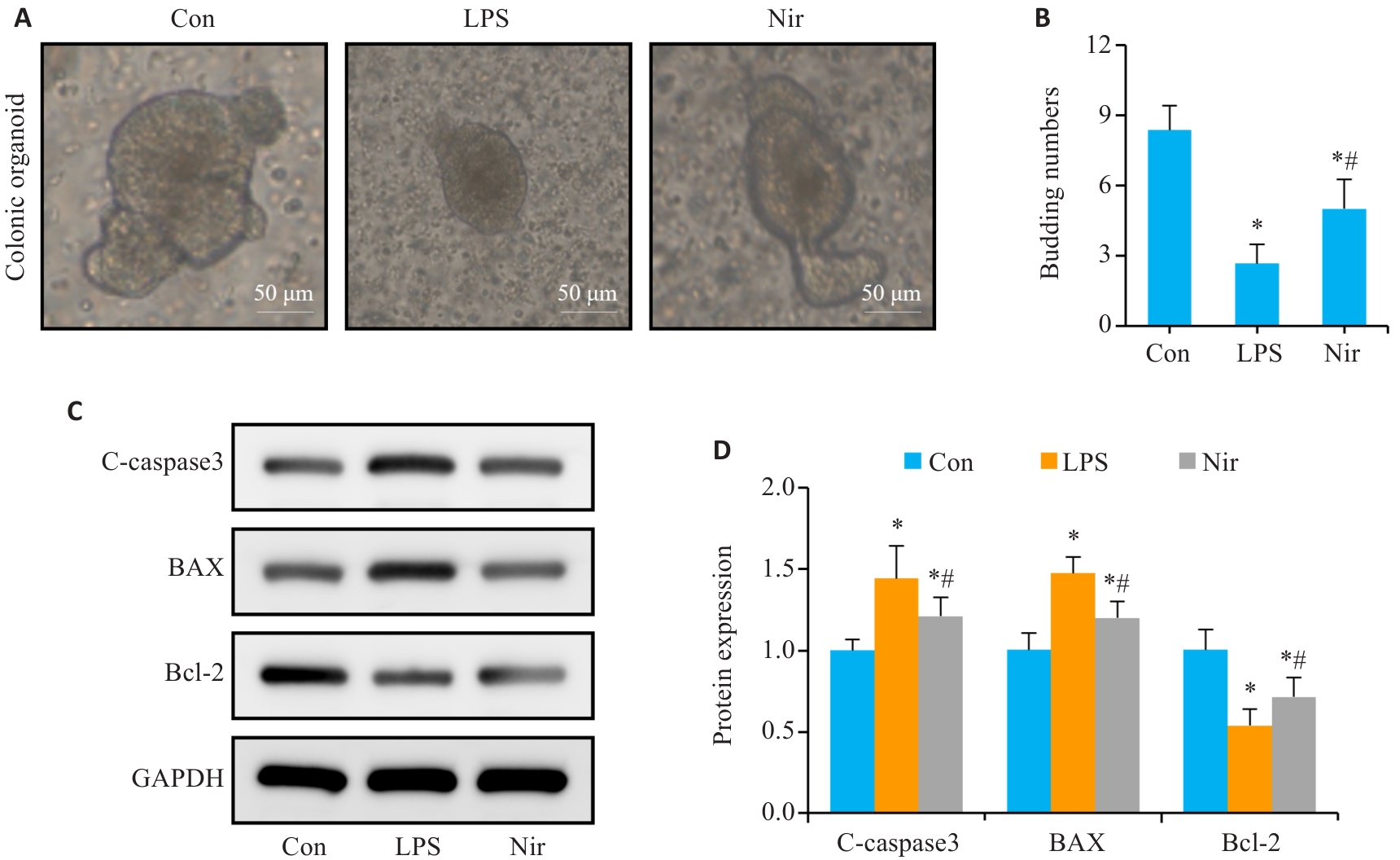
Fig.4 Effect of Nir on LPS-induced intestinal epithelial cell apoptosis in mouse colon organoids. A: Representative images of colon organoids. B: Number of organoids budding. C, D: Western blotting and bar chart for C-caspase-3, Bax and Bcl-2 expressions. n=6, *P<0.05 vs Con; #P<0.05 vs LPS.
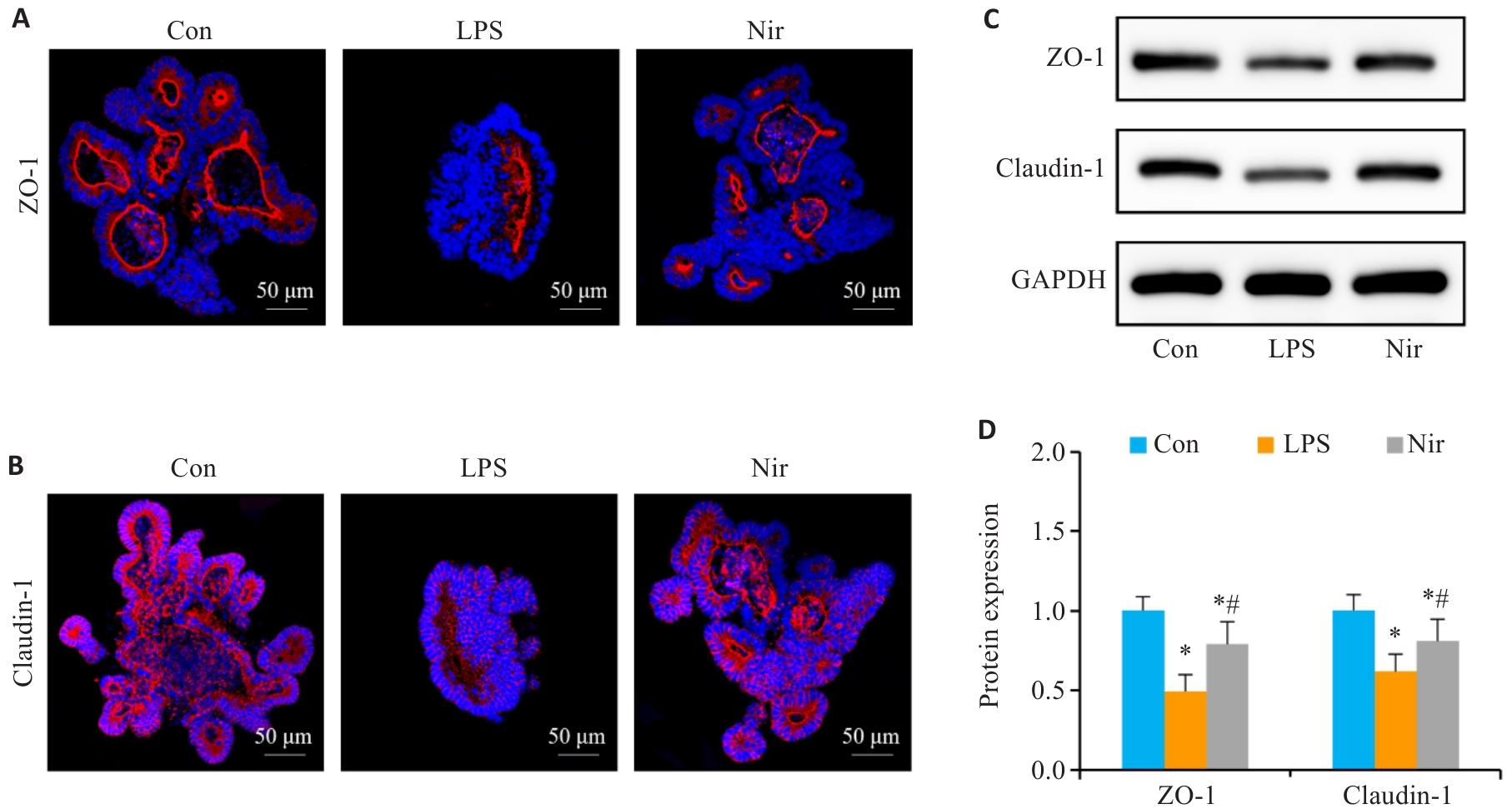
Fig.5 Effects of Nir intervention on intestinal barrier function in LPS-induced colonic organoids. A, B: Immunofluorescence assay of ZO-1 and claudin-1 expression. C, D: Western blotting for claudin-1 and ZO-1 expressions. n=6, *P<0.05 vs Con; #P<0.05 vs LPS.
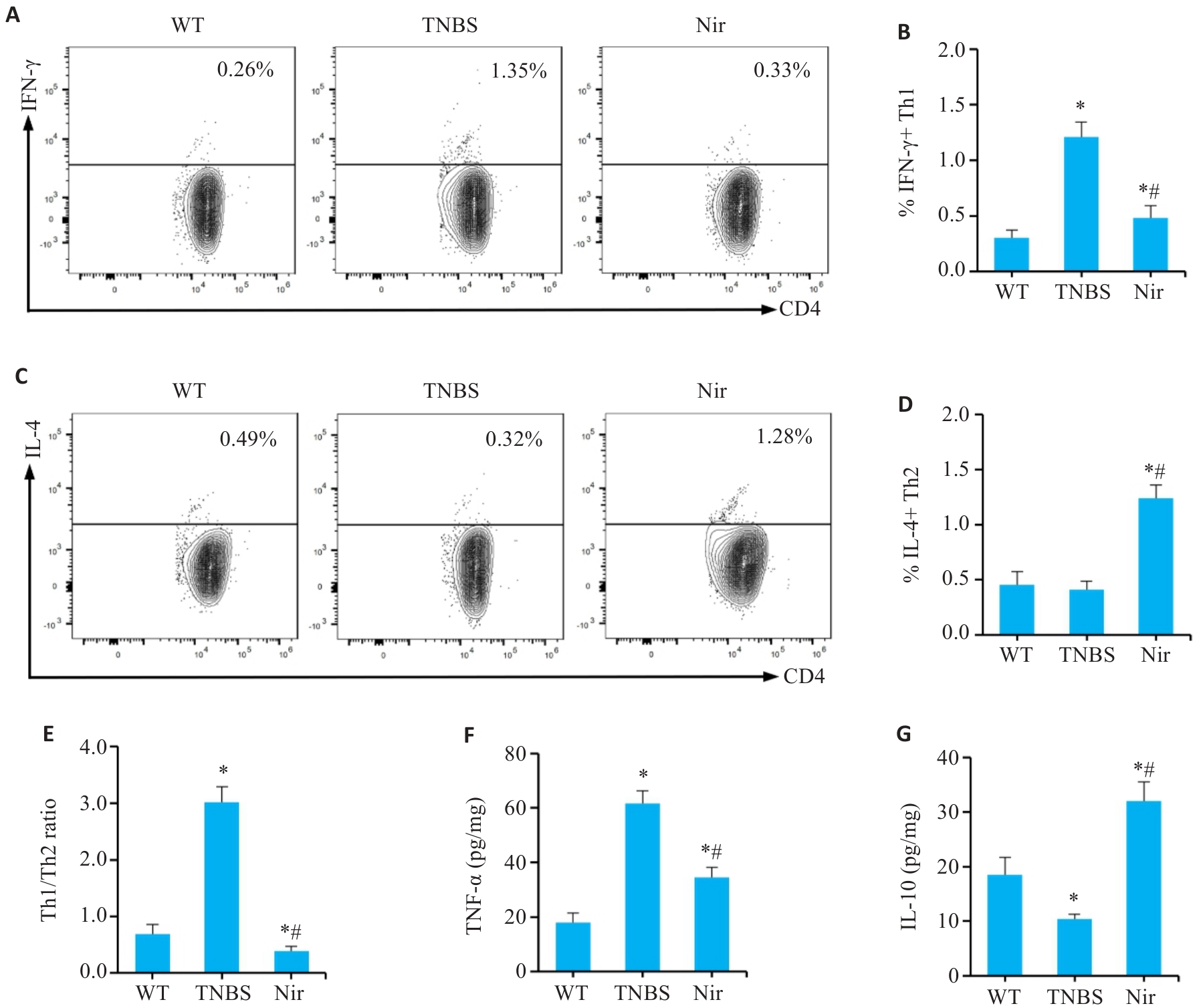
Fig.6 Effects of Nir intervention on Th1/Th2 balance in mesenteric lymph nodes and inflammatory factor levels in colonic tissues of TNBS-induced colitis mice. A, B: Proportion of Th1 cells in the mesenteric lymph nodes. C, D: Proportion of Th2 cells in the mesenteric lymph nodes. E: Th1/Th2 ratio. F, G: Expression levels of intestinal mucosal inflammatory mediators. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
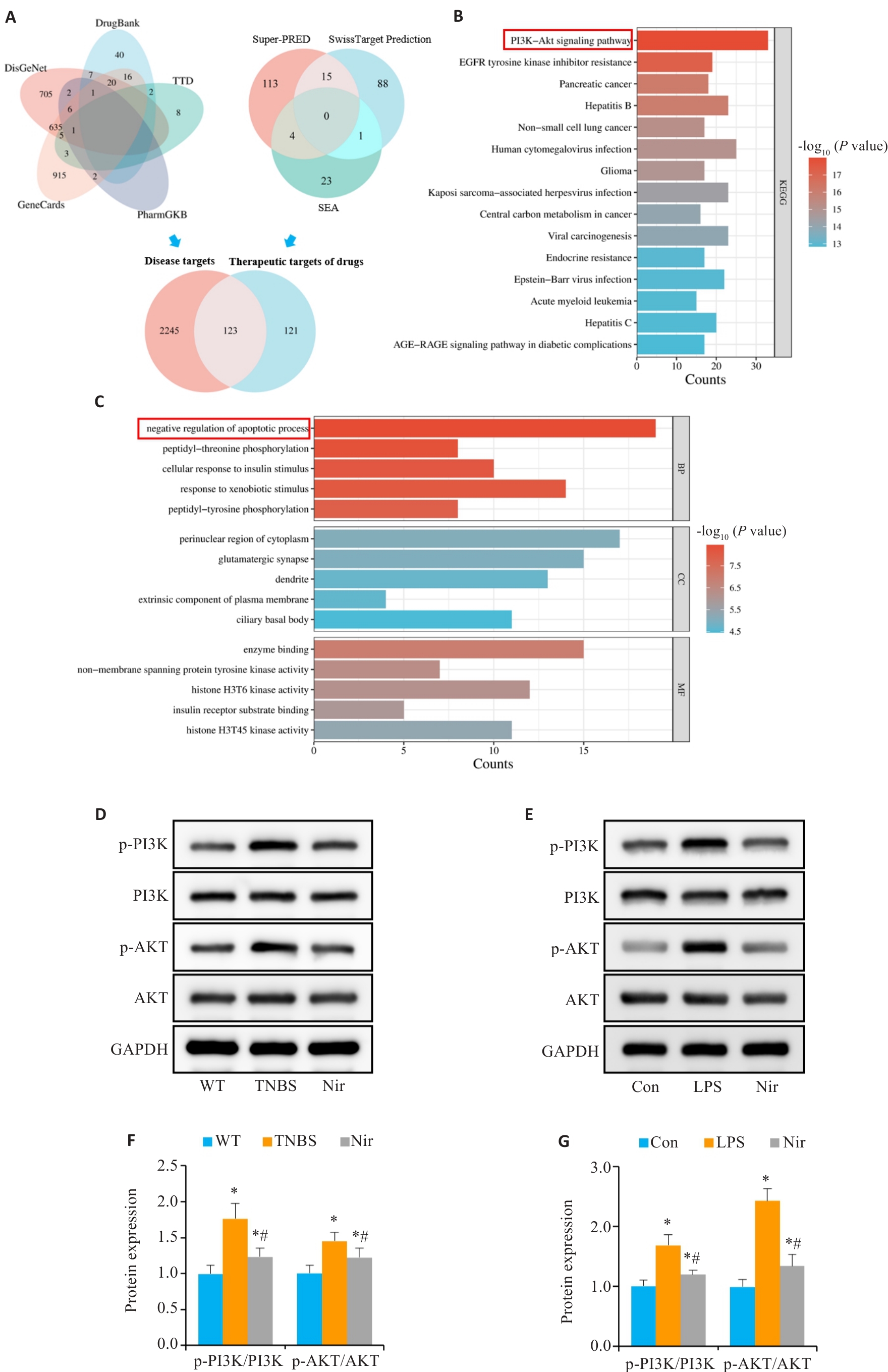
Fig.7 Ameliorative effect of Nir on colitis is possibly associated with the PI3K/AKT pathway. A: Venn diagram. B: KEGG enrichment analysis. C: GO enrichment analysis. D, F: Western blotting of p-PI3K, PI3K, p-AKT and AKT expressions in the colon tissue. E, G: Western blotting of p-PI3K, PI3K, p-AKT and AKT expressions in the colonic organoids. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
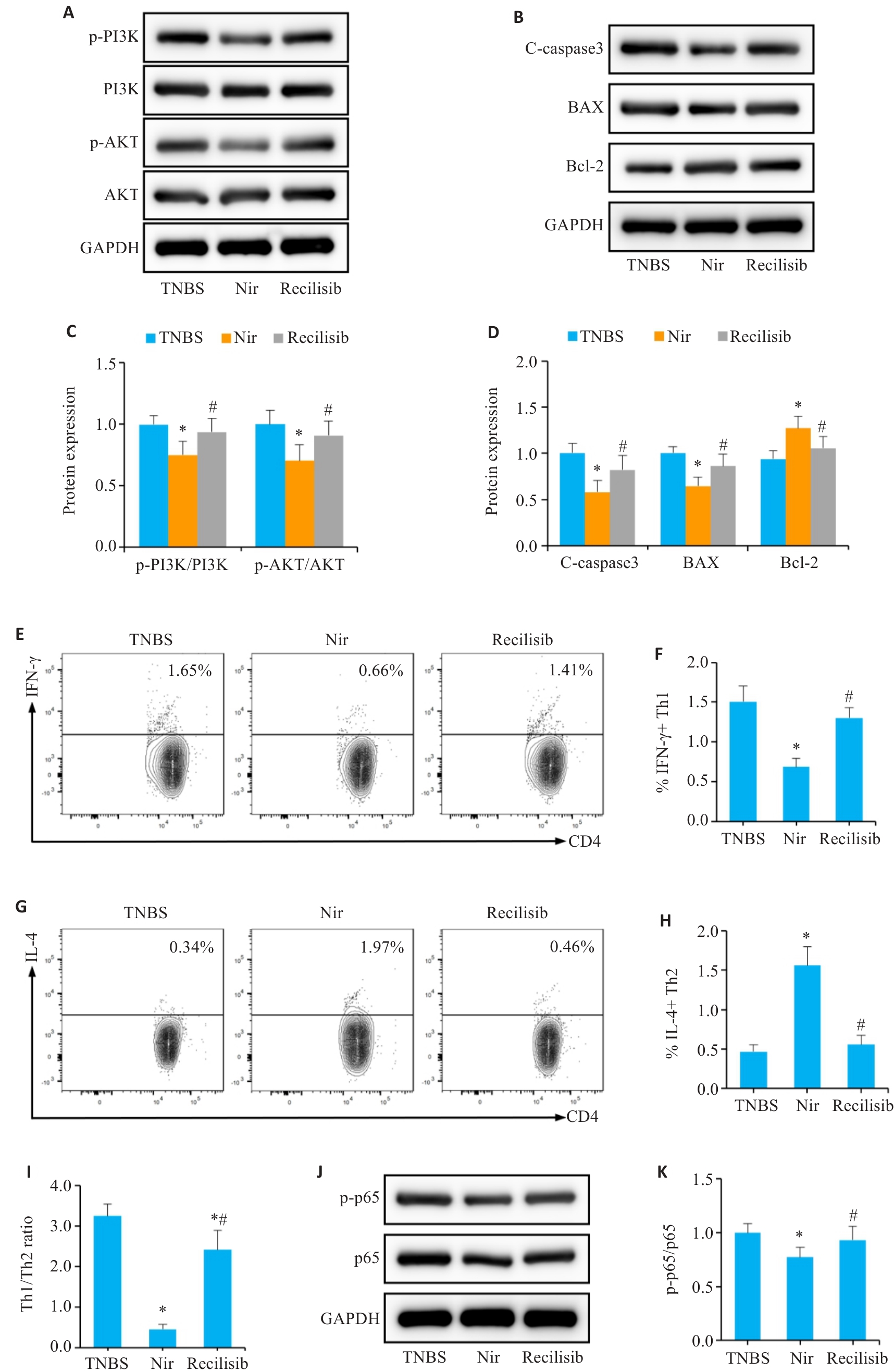
Fig.8 Nir can restore immune homeostasis and barrier integrity by negatively regulating the PI3K/AKT pathway. A, C: Western blotting of p-PI3K, PI3K, p-AKT and AKT expressions. B, D: Western blotting for C-caspase-3, Bax and Bcl-2 expressions. E, F: Proportion of Th1 cells in the mesenteric lymph nodes. G, H: Proportion of Th2 cells in the mesenteric lymph nodes. I: Th1/Th2 ratio. J, K: Western blotting for p-p65 and p65 expressions. n=6, *P<0.05 vs TNBS; #P<0.05 vs Nir.
| [1] | Torres J, Mehandru S, Colombel JF, et al. Crohn's disease[J]. Lancet, 2017, 389(10080): 1741-55. doi:10.1016/s0140-6736(16)31711-1 |
| [2] | 郭亚慧, 牛巍巍, 张晓岚. 炎症性肠病诊断与治疗的共识意见(2018年, 北京): 克罗恩病部分解读[J]. 临床荟萃, 2018, 33(12): 1077-9, 1082. doi:10.3969/j.issn.1004-583X.2018.12.016 |
| [3] | Veauthier B, Hornecker JR. Crohn's disease: diagnosis and management[J]. Am Fam Physician, 2018, 98(11): 661-9. |
| [4] | Edogawa S, Edwinson AL, Peters SA, et al. Serine proteases as luminal mediators of intestinal barrier dysfunction and symptom severity in IBS[J]. Gut, 2020, 69(1): 62-73. doi:10.1136/gutjnl-2018-317416 |
| [5] | 陈鑫明, 刘 洋, 何运胜, 等. 克罗恩病发病影响因素的研究进展[J]. 现代消化及介入诊疗, 2023, 28(7): 912-8. |
| [6] | Xavier RJ, Podolsky DK. Unravelling the pathogenesis of inflammatory bowel disease[J]. Nature, 2007, 448(7152): 427-34. doi:10.1038/nature06005 |
| [7] | Di Tommaso N, Gasbarrini A, Ponziani FR. Intestinal barrier in human health and disease[J]. Int J Environ Res Public Health, 2021, 18(23): 12836. doi:10.3390/ijerph182312836 |
| [8] | Dunleavy KA, Raffals LE, Camilleri M. Intestinal barrier dysfunction in inflammatory bowel disease: underpinning pathogenesis and therapeutics[J]. Dig Dis Sci, 2023, 68(12): 4306-20. doi:10.1007/s10620-023-08122-w |
| [9] | Farzaei MH, Bahramsoltani R, Abdolghaffari AH, et al. A mechanistic review on plant-derived natural compounds as dietary supplements for prevention of inflammatory bowel disease[J]. Expert Rev Gastroenterol Hepatol, 2016, 10(6): 745-58. doi:10.1586/17474124.2016.1145546 |
| [10] | 李晓花, 杨文玉, 王 剑. 傣药“芽害巴” (珠子草)化学成分研究[J]. 亚太传统医药, 2019, 15(2): 59-63. doi:10.11954/ytctyy.201902017 |
| [11] | Kassuya CAL, Silvestre A, Menezes-de-Lima O, et al. Antiin-flammatory and antiallodynic actions of the lignan niranthin isolated from Phyllanthus amarus Evidence for interaction with platelet activating factor receptor[J]. Eur J Pharmacol, 2006, 546(1/2/3): 182-8. doi:10.1016/j.ejphar.2006.07.025 |
| [12] | Kassuya CAL, Leite DFP, de Melo LV, et al. Anti-inflammatory properties of extracts, fractions and lignans isolated from Phyllanthus amarus[J]. Planta Med, 2005, 71(8): 721-6. doi:10.1055/s-2005-871258 |
| [13] | Ismail EN, Jantan I, Vidyadaran S, et al. Phyllanthus amarus prevents LPS-mediated BV2 microglial activation via MyD88 and NF-κB signaling pathways[J]. BMC Complement Med Ther, 2020, 20(1): 202. doi:10.1186/s12906-020-02961-0 |
| [14] | Alagan A, Jantan I, Kumolosasi E, et al. Protective effects of Phyllanthus amarus against lipopolysaccharide-induced neuroin-flammation and cognitive impairment in rats[J]. Front Pharmacol, 2019, 10: 632. doi:10.3389/fphar.2019.00632 |
| [15] | Harikrishnan H, Jantan I, Haque MA, et al. Anti-inflammatory effects of Phyllanthus amarus Schum. & Thonn. through inhibition of NF‑κB, MAPK, and PI3K-Akt signaling pathways in LPS-induced human macrophages[J]. BMC Complement Altern Med, 2018, 18(1): 224. doi:10.1186/s12906-018-2289-3 |
| [16] | Harikrishnan H, Jantan I, Alagan A, et al. Modulation of cell signaling pathways by Phyllanthus amarus and its major constituents: potential role in the prevention and treatment of inflammation and cancer[J]. Inflammopharmacology, 2020, 28(1): 1-18. doi:10.1007/s10787-019-00671-9 |
| [17] | Harikrishnan H, Jantan I, Haque MA, et al. Anti-inflammatory effects of hypophyllanthin and niranthin through downregulation of NF-κB/MAPKs/PI3K-Akt signaling pathways[J]. Inflammation, 2018, 41(3): 984-95. doi:10.1007/s10753-018-0752-4 |
| [18] | Chowdhury S, Mukherjee T, Mukhopadhyay R, et al. The lignan niranthin poisons Leishmania donovani topoisomerase IB and favours a Th1 immune response in mice[J]. EMBO Mol Med, 2012, 4(10): 1126-43. doi:10.1002/emmm.201201316 |
| [19] | Wirtz S, Popp V, Kindermann M, et al. Chemically induced mouse models of acute and chronic intestinal inflammation[J]. Nat Protoc, 2017, 12(7): 1295-309. doi:10.1038/nprot.2017.044 |
| [20] | Gong ZW, Lao DY, Wu Y, et al. Inhibiting PI3K/Akt-signaling pathway improves neurobehavior changes in anti-NMDAR encephalitis mice by ameliorating blood-brain barrier disruption and neuronal damage[J]. Cell Mol Neurobiol, 2023, 43(7): 3623-37. doi:10.1007/s10571-023-01371-3 |
| [21] | Zuo LG, Geng ZJ, Song X, et al. Browning of mesenteric white adipose tissue in Crohn's disease: a new pathological change and therapeutic target[J]. J Crohns Colitis, 2023, 17(8): 1179-92. doi:10.1093/ecco-jcc/jjad046 |
| [22] | Zuo LG, Li J, Zhang XF, et al. Aberrant mesenteric adipose extracellular matrix remodelling is involved in adipocyte dysfunction in Crohn's disease: the role of TLR-4-mediated macrophages[J]. J Crohns Colitis, 2022, 16(11): 1762-76. doi:10.1093/ecco-jcc/jjac087 |
| [23] | Paone P, Cani PD. Mucus barrier, mucins and gut microbiota: the expected slimy partners?[J]. Gut, 2020, 69(12): 2232-43. doi:10.1136/gutjnl-2020-322260 |
| [24] | Wang ZC, Shen J. The role of goblet cells in Crohn's disease[J]. Cell Biosci, 2024, 14(1): 43. doi:10.1186/s13578-024-01220-w |
| [25] | Kaminsky LW, Al-Sadi R, Ma TY. IL-1β and the intestinal epithelial tight junction barrier[J]. Front Immunol, 2021, 12: 767456. doi:10.3389/fimmu.2021.767456 |
| [26] | Schlegel N, Boerner K, Waschke J. Targeting desmosomal adhesion and signalling for intestinal barrier stabilization in inflammatory bowel diseases-Lessons from experimental models and patients[J]. Acta Physiol (Oxf), 2021, 231(1): e13492. doi:10.1111/apha.13492 |
| [27] | Saez A, Gomez-Bris R, Herrero-Fernandez B, et al. Innate lymphoid cells in intestinal homeostasis and inflammatory bowel disease[J]. Int J Mol Sci, 2021, 22(14): 7618. doi:10.3390/ijms22147618 |
| [28] | Gomez-Bris R, Saez A, Herrero-Fernandez B, et al. CD4 T-cell subsets and the pathophysiology of inflammatory bowel disease[J]. Int J Mol Sci, 2023, 24(3): 2696. doi:10.3390/ijms24032696 |
| [29] | Foerster EG, Mukherjee T, Cabral-Fernandes L, et al. How autophagy controls the intestinal epithelial barrier[J]. Autophagy, 2022, 18(1): 86-103. doi:10.1080/15548627.2021.1909406 |
| [30] | Guo NK, She H, Tan L, et al. Nano parthenolide improves intestinal barrier function of sepsis by inhibiting apoptosis and ROS via 5-HTR2A[J]. Int J Nanomedicine, 2023, 18: 693-709. doi:10.2147/ijn.s394544 |
| [31] | Woznicki JA, Saini NS, Flood P, et al. TNF-α synergises with IFN-γ to induce caspase-8-JAK1/2-STAT1-dependent death of intestinal epithelial cells[J]. Cell Death Dis, 2021, 12(10): 864. doi:10.1038/s41419-021-04151-3 |
| [32] | Zhang Y, Yang X, Ge XH, et al. Puerarin attenuates neurological deficits via Bcl-2/Bax/cleaved caspase-3 and Sirt3/SOD2 apoptotic pathways in subarachnoid hemorrhage mice[J]. Biomed Pharmacother, 2019, 109: 726-33. doi:10.1016/j.biopha.2018.10.161 |
| [33] | Sha JY, Chen KC, Liu ZB, et al. Ginseng-DF ameliorates intestinal mucosal barrier injury and enhances immunity in immuno-suppressed mice by regulating MAPK/NF‑κB signaling pathways[J]. Eur J Nutr, 2024, 63(5): 1487-500. doi:10.1007/s00394-024-03378-y |
| [34] | Mei XP, Zhou HK, Song ZW, et al. PCSK6 mediates Th1 differentiation and promotes chronic colitis progression and mucosal barrier injury via STAT1[J]. Aging (Albany NY), 2023, 15(10): 4363-73. doi:10.18632/aging.204739 |
| [35] | Ma BW, Athari SS, Mehrabi Nasab E, et al. PI3K/AKT/mTOR and TLR4/MyD88/NF‑κB signaling inhibitors attenuate pathological mechanisms of allergic asthma[J]. Inflammation, 2021, 44(5): 1895-907. doi:10.1007/s10753-021-01466-3 |
| [36] | Li ZH, Sun QM, Liu QY, et al. Compound 511 ameliorates MRSA-induced lung injury by attenuating morphine-induced immuno-suppression in mice via PI3K/AKT/mTOR pathway[J]. Phytomedicine, 2023, 108: 154475. doi:10.1016/j.phymed.2022.154475 |
| [37] | Wang JC, Hu KL, Cai XY, et al. Targeting PI3K/AKT signaling for treatment of idiopathic pulmonary fibrosis[J]. Acta Pharm Sin B, 2022, 12(1): 18-32. doi:10.1016/j.apsb.2021.07.023 |
| [38] | Lin CY, Tsai PH, Kandaswami CC, et al. Role of tissue transglutaminase 2 in the acquisition of a mesenchymal-like phenotype in highly invasive A431 tumor cells[J]. Mol Cancer, 2011, 10: 87. doi:10.1186/1476-4598-10-87 |
| [1] | Qile SONG, Yikai MIAO, Xiaotong FENG, Yifan WANG, Wei LIU, Qi WEI, Xinru YU, Wenwen CHEN, Xiaoyan FU. Selenocystine inhibits colon cancer cell growth by promoting reactive oxygen species generation to trigger oxidative damage [J]. Journal of Southern Medical University, 2026, 46(3): 532-540. |
| [2] | Jiayao YANG, Yulian He, Yanlei GUO, Fanghong SHANG, Lei HUA, Yong YANG, Xiaomei ZHANG, Jiangping WEI. Radix codonopsis combined with Poria improves cognitive impairment in rats with unilateral common carotid artery ligation by regulating the ERα/PI3K/Akt signaling pathway [J]. Journal of Southern Medical University, 2026, 46(2): 247-258. |
| [3] | Jiahui QIU, Meng CHEN, Ru MAN, Xin CHEN, Dongrui QIU, Qitong CHANG, Hongyu MA. Huayu Tongbian Decoction promotes efferocytosis of interstitial Cajal cells in rats with slow transit constipation by inhibiting the PI3K/Akt signaling pathway [J]. Journal of Southern Medical University, 2026, 46(2): 293-300. |
| [4] | Binjie LI, Xiaofang ZHOU, Xiaomeng LANG, Xin KANG, Jianping LIU. Xiezhuo Jiedu Recipe improves ulcerative colitis in rats by regulating Th17/Treg immune balance [J]. Journal of Southern Medical University, 2026, 46(2): 335-344. |
| [5] | Lin YIN, Keni ZHANG, Tong QIAO, Minzhu NIU, Lixia YIN, Xinyue LIU, Zhijun GENG, Jing LI, Jianguo HU. Veratric acid relieves oxidative stress and DSS-induced colitis in mice by activating the Nrf2/HO-1 signaling pathway [J]. Journal of Southern Medical University, 2026, 46(2): 403-411. |
| [6] | Linlin HUANG, Wang ZHENG, Jianguo HU, Xue SONG, Lu TAO, Zhijun GENG, Jing LI, Lugen ZUO, Sitang GE. Verbenalin ameliorates intestinal inflammation and colitis in a mouse model of Crohn's disease by inhibiting the PI3K-AKT pathway [J]. Journal of Southern Medical University, 2026, 46(2): 423-433. |
| [7] | Tong QIAO, Lin YIN, Keni ZHANG, Minzhu NIU, Ju HUANG, Zhijun Geng, Jing LI, Jianguo HU. Poricoic acid A alleviates dextran sulfate sodium-induced colitis in mice by regulating AMPK/mTOR-mediated autophagy and inhibiting intestinal epithelial cell apoptosis [J]. Journal of Southern Medical University, 2026, 46(1): 131-140. |
| [8] | Shufen ZHANG, Tianrong HUANG, Canhong YANG, Jiayi CHEN, Tianming LÜ, Jiafa ZHANG. Sulforaphane reduces reactive astrocyte-mediated neuron apoptosis in vitro by inhibiting the MAPK/NF-κB signaling pathway in Aβ42 oligomer-activated astrocytes [J]. Journal of Southern Medical University, 2026, 46(1): 191-199. |
| [9] | Tong SHA, Wenyan WANG, Jiabin XUAN, Jie WU, Nengxian SHI, Jin HE, Hongbin HU, Yaoyuan ZHANG. Identification of immune status subtypes and prognostic analysis of septic patients based on Th1/Th2 cytokine assays [J]. Journal of Southern Medical University, 2026, 46(1): 6-22. |
| [10] | Jinyan ZHAO, Jiao PENG, Minghe LIN, Xiaoqin ZHU, Bin HUANG, Jiumao LIN. Qingjie Fuzheng Granules alleviates 5-fluorouracil-induced skeletal muscle injury in tumor-bearing mice by inhibiting mitochondria-dependent apoptosis and activating the AMPK-PGC-1α pathway [J]. Journal of Southern Medical University, 2026, 46(1): 94-103. |
| [11] | Lu RAO, Jiahe DING, Jiangping WEI, Yong YANG, Xiaomei ZHANG, Jirui WANG. Flos Sophorae improves psoriasis in mice by inhibiting the PI3K/AKT pathway [J]. Journal of Southern Medical University, 2025, 45(9): 1989-1996. |
| [12] | Xiaoyu CHANG, Hanwen ZHANG, Hongting CAO, Ling HOU, Xin MENG, Hong TAO, Yan LUO, Guanghua LI. Heat stress affects expression levels of circadian clock gene Bmal1 and cyclins in rat thoracic aortic endothelial cells [J]. Journal of Southern Medical University, 2025, 45(7): 1353-1362. |
| [13] | Xuan WU, Jiamin FANG, Weiwei HAN, Lin CHEN, Jing SUN, Qili JIN. High PRELID1 expression promotes epithelial-mesenchymal transition in gastric cancer cells and is associated with poor prognosis [J]. Journal of Southern Medical University, 2025, 45(7): 1535-1542. |
| [14] | Yujia YANG, Lifang YANG, Yaling WU, Zhaoda DUAN, Chunze YU, Chunyun WU, Jianyun YU, Li YANG. Cannabidiol inhibits neuronal endoplasmic reticulum stress and apoptosis in rats with multiple concussions by regulating the PERK-eIF2α-ATF4-CHOP pathway [J]. Journal of Southern Medical University, 2025, 45(6): 1240-1250. |
| [15] | Minzhu NIU, Lixia YIN, Tong QIAO, Lin YIN, Keni ZHANG, Jianguo HU, Chuanwang SONG, Zhijun GENG, Jing LI. Ecliptasaponin A ameliorates DSS-induced colitis in mice by suppressing M1 macrophage polarization via inhibiting the JAK2/STAT3 pathway [J]. Journal of Southern Medical University, 2025, 45(6): 1297-1306. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||