Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (2): 403-411.doi: 10.12122/j.issn.1673-4254.2026.02.18
Lin YIN1,2( ), Keni ZHANG1, Tong QIAO1, Minzhu NIU2, Lixia YIN1, Xinyue LIU3, Zhijun GENG3, Jing LI1, Jianguo HU1(
), Keni ZHANG1, Tong QIAO1, Minzhu NIU2, Lixia YIN1, Xinyue LIU3, Zhijun GENG3, Jing LI1, Jianguo HU1( )
)
Received:2025-06-05
Online:2026-02-20
Published:2026-03-10
Contact:
Jianguo HU
E-mail:yljykbbmu@163.com;jghu9200@bbmu.edu.cn
Lin YIN, Keni ZHANG, Tong QIAO, Minzhu NIU, Lixia YIN, Xinyue LIU, Zhijun GENG, Jing LI, Jianguo HU. Veratric acid relieves oxidative stress and DSS-induced colitis in mice by activating the Nrf2/HO-1 signaling pathway[J]. Journal of Southern Medical University, 2026, 46(2): 403-411.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.02.18
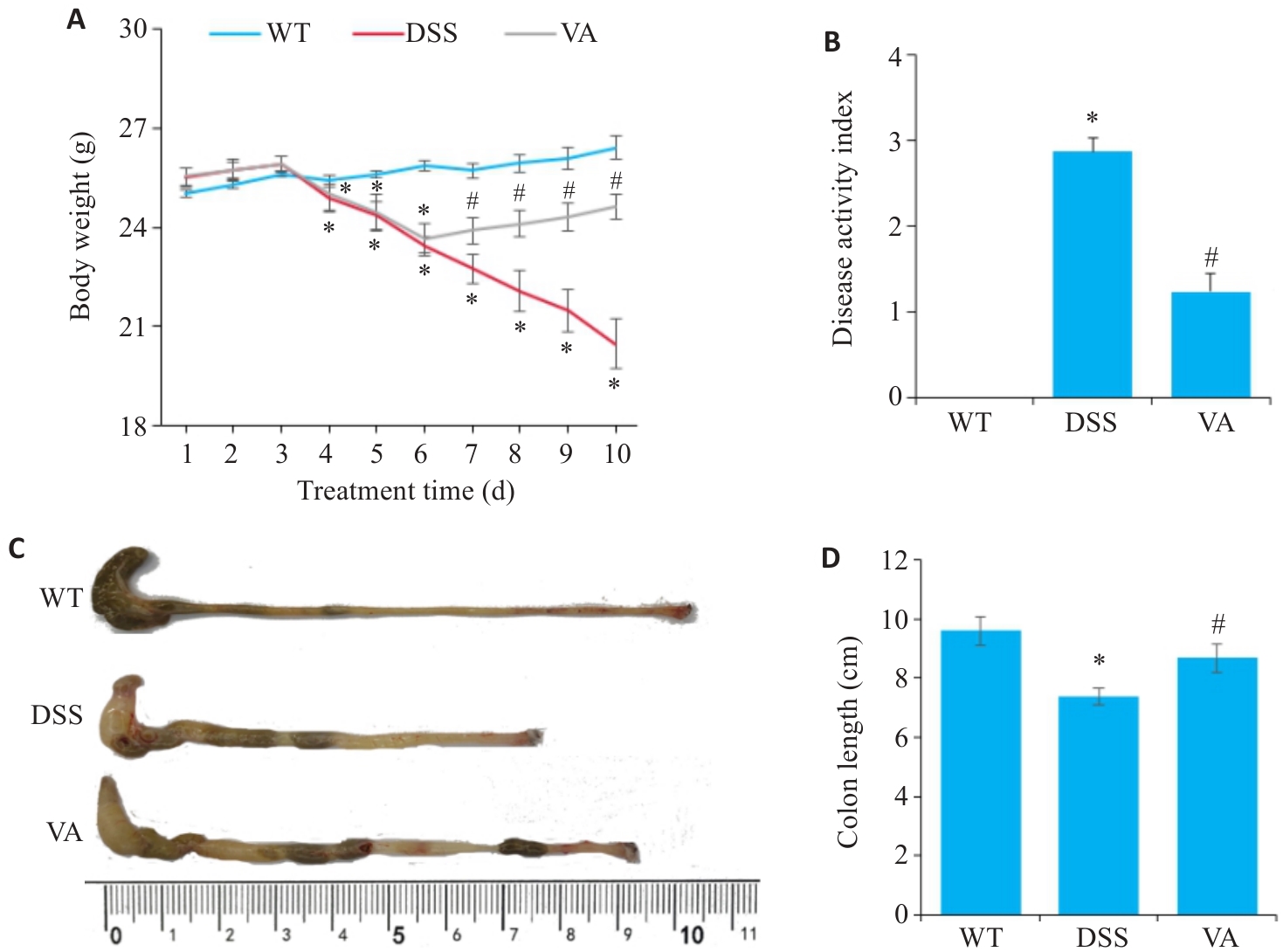
Fig.1 Effect of VA on DSS-induced colitis symptoms in mice (n=10). A: Changes of body weight. B: Changes of DAI scores. C, D: Comparison of colon length among different groups. *P<0.05 vs WT group; #P<0.05 vs DSS group.
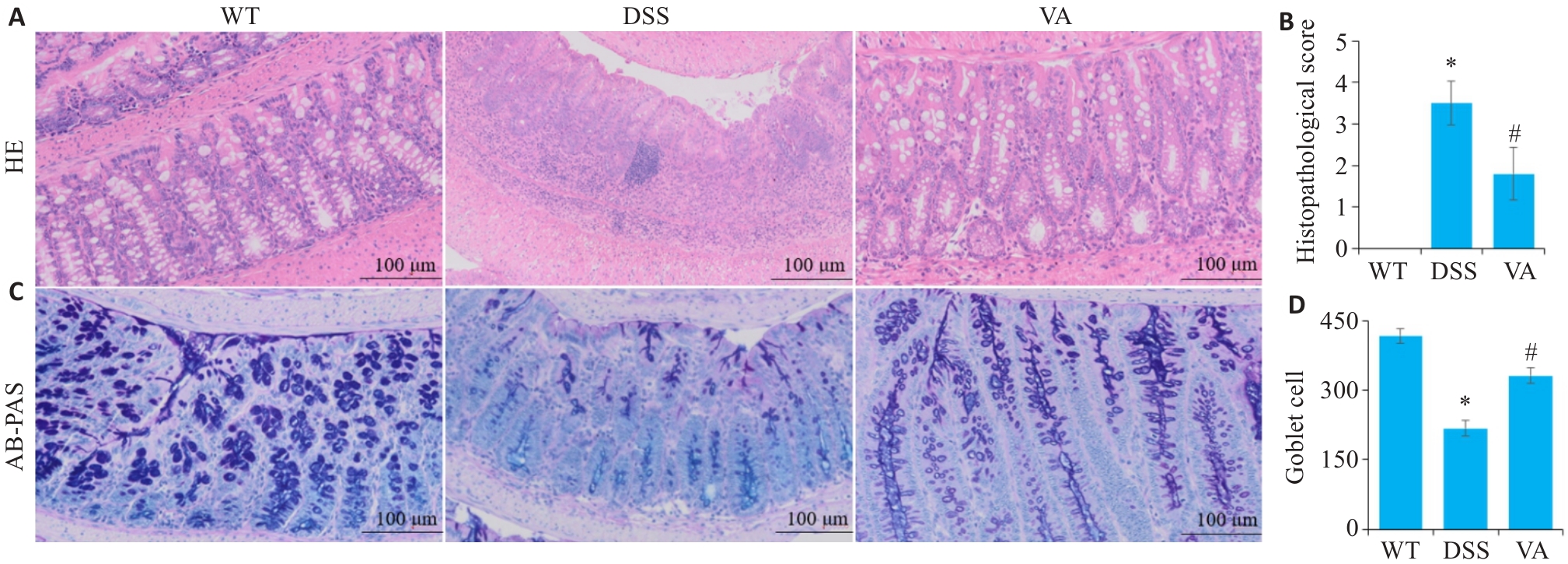
Fig.2 Effect of VA on intestinal histopathology in DSS-treated mice (n=10). A: HE staining and inflammatory scores of the intestinal tissues of mice in each group. B: Histopathological scores of mice in each group. C: AB-PAS staining of the intestinal tissues in each group. D: Comparison of goblet cell counts in each group. *P<0.05 vs WT group; #P<0.05 vs DSS group.
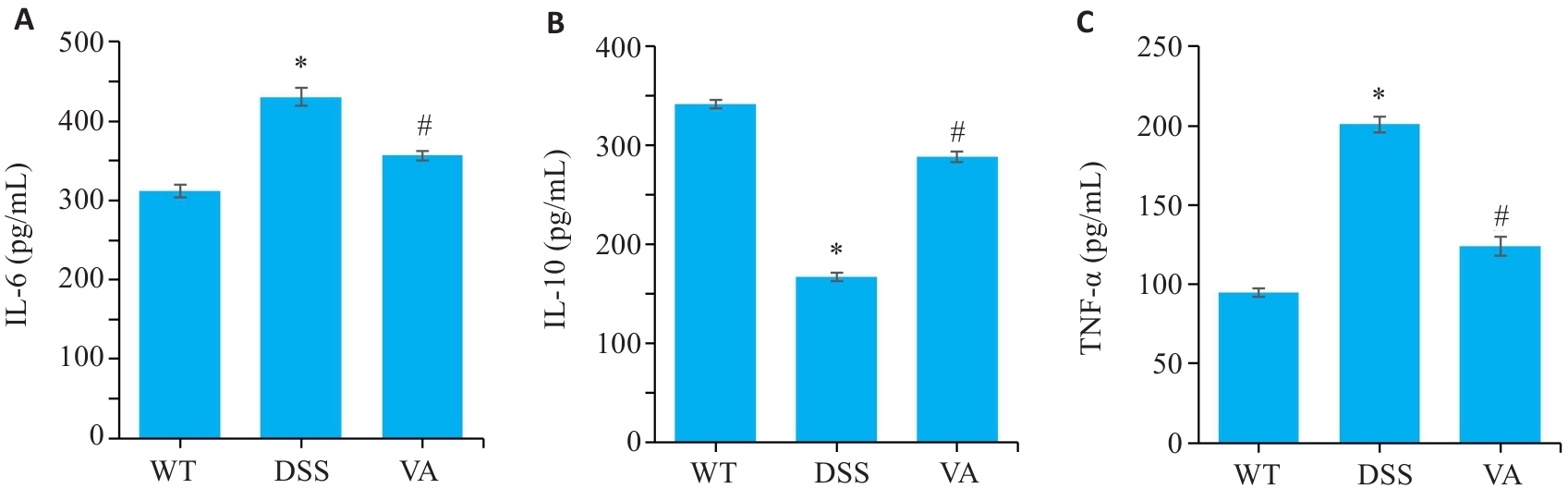
Fig.3 Effect of VA on expressions of inflammatory cytokines in the colonic tissues of DSS-treated mice (n=10). A: Levels of interleukin-6 (IL-6) in the colonic mucosa of the mice in each group detected by enzyme-linked immunosorbent assay (ELISA). B: Levels of interleukin-10 (IL-10) in the colonic mucosa of the mice in each group detected by ELISA. C: Levels of tumor necrosis factor-α (TNF-α) in the colonic mucosa of the mice in each group detected by ELISA. *P<0.05 vs WT group; #P<0.05 vs DSS group.
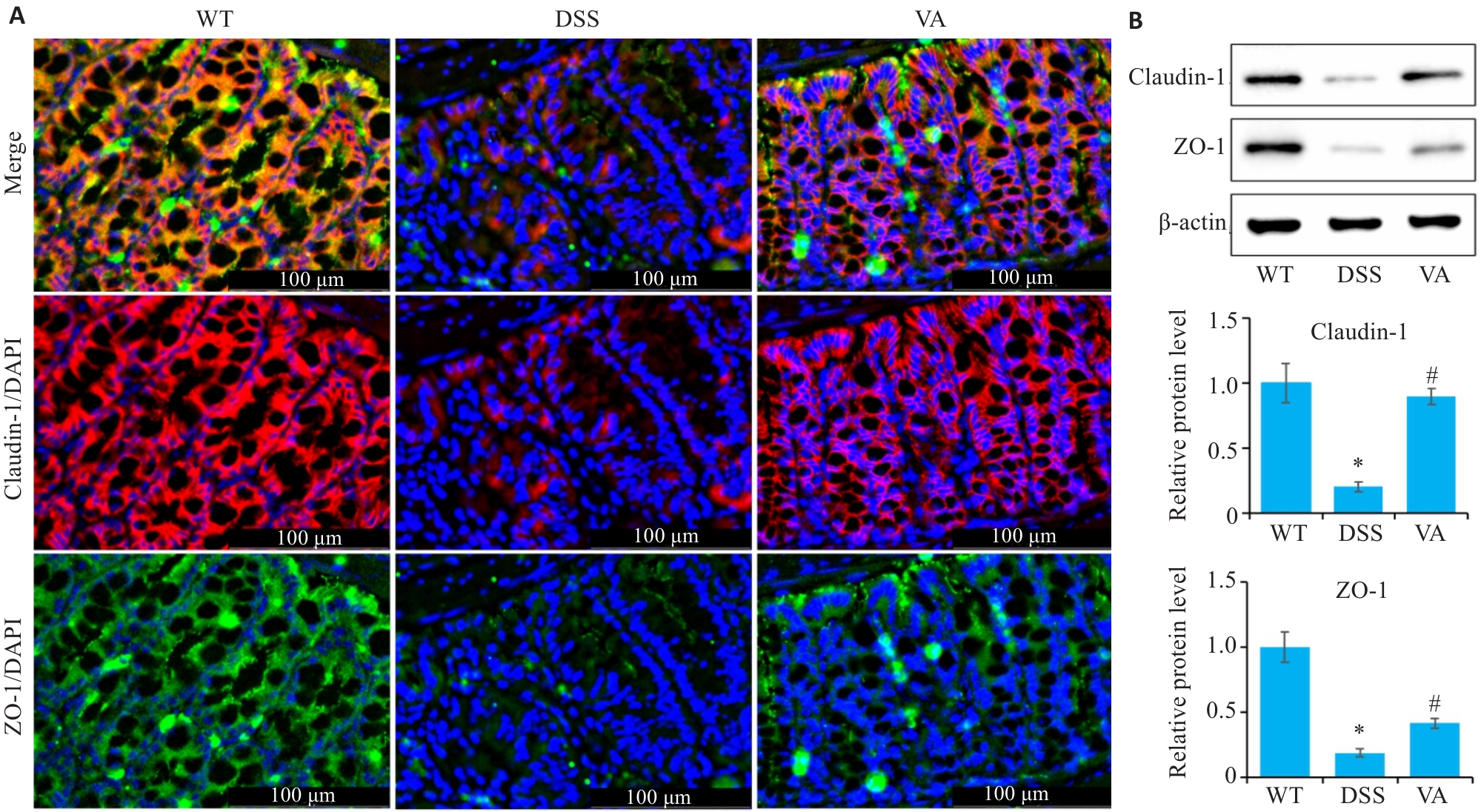
Fig.4 Effect of VA on tight junction proteins in the colonic tissues of DSS-induced mice (n=10). A: Immunofluorescence staining for claudin-1 and ZO-1 in the colons of the mice in each group. B: Western blotting for detecting expression levels of claudin-1 and ZO-1 in the colons of the mice in each group. *P<0.05 vs WT group; #P<0.05 vs DSS group.
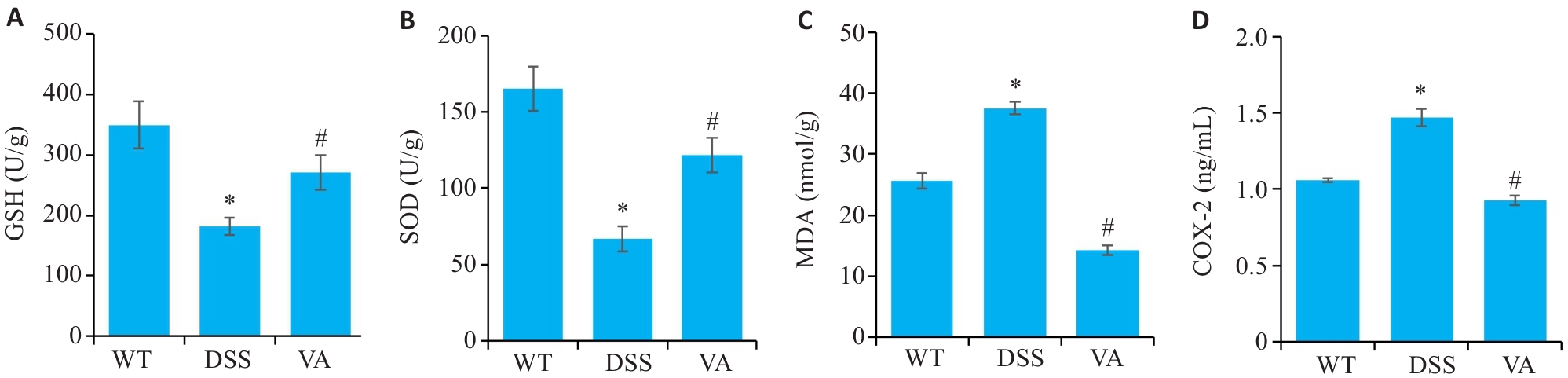
Fig.5 Regulatory effect of VA on oxidative stress in the colonic tissues of DSS-induced mice (n=10). A: Detection results of glutathione (GSH) in each group. B: levels of superoxide dismutase (SOD) in each group. C: Malondialdehyde (MDA) levels in each group. D: Cyclooxygenase-2 (COX-2) levels in each group. *P<0.05 vs WT group; #P<0.05 vs DSS group.
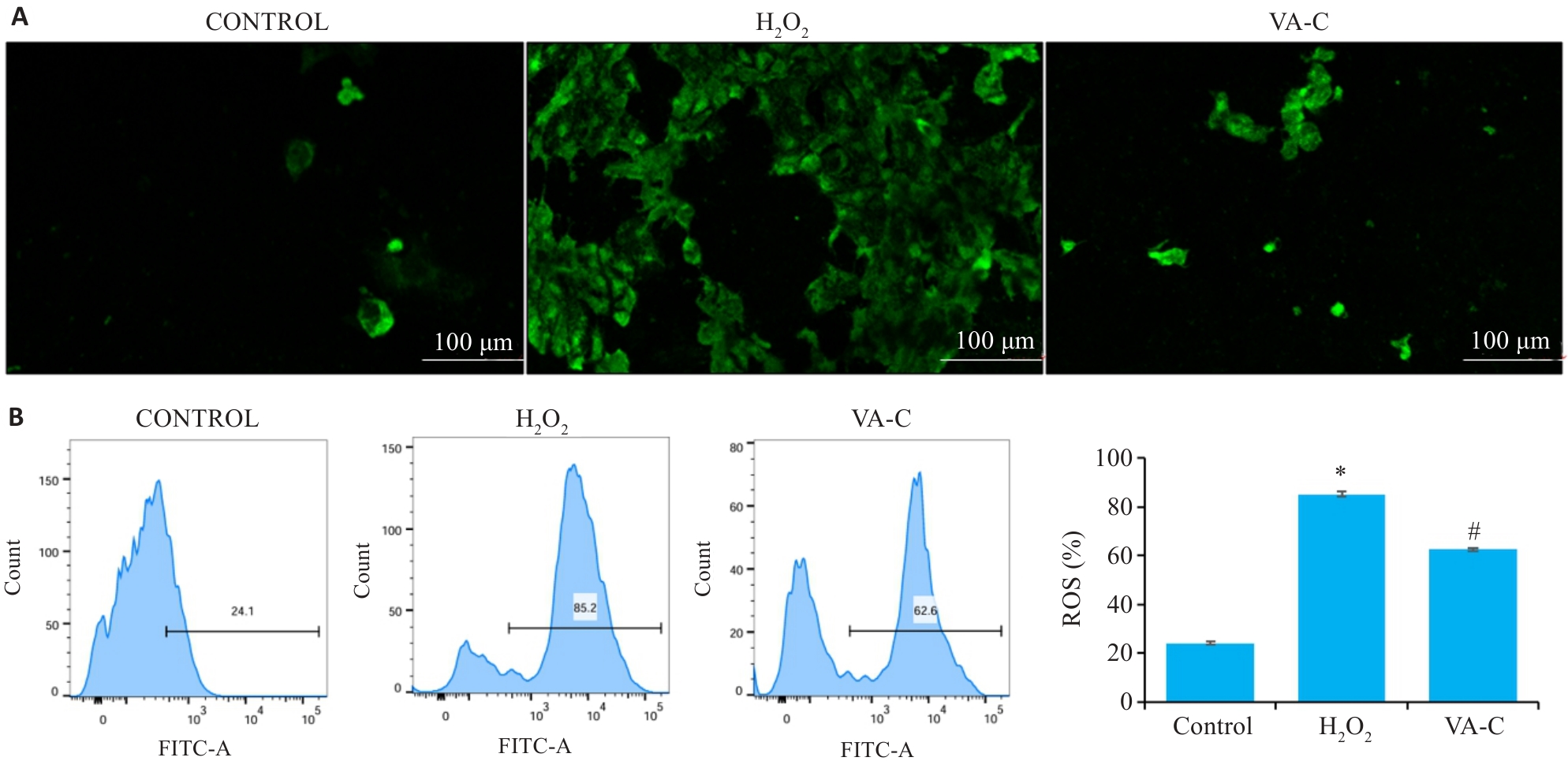
Fig.6 Effect of VA on reactive oxygen species (ROS) production induced by H₂O₂ in Caco-2 cells (n=10). A: ROS staining with the fluorescent probe (DCFH-DA) in each group. B: ROS detection by flow cytometry in each group. *P<0.05 vs Control group. #P<0.05 vs H₂O₂ group.
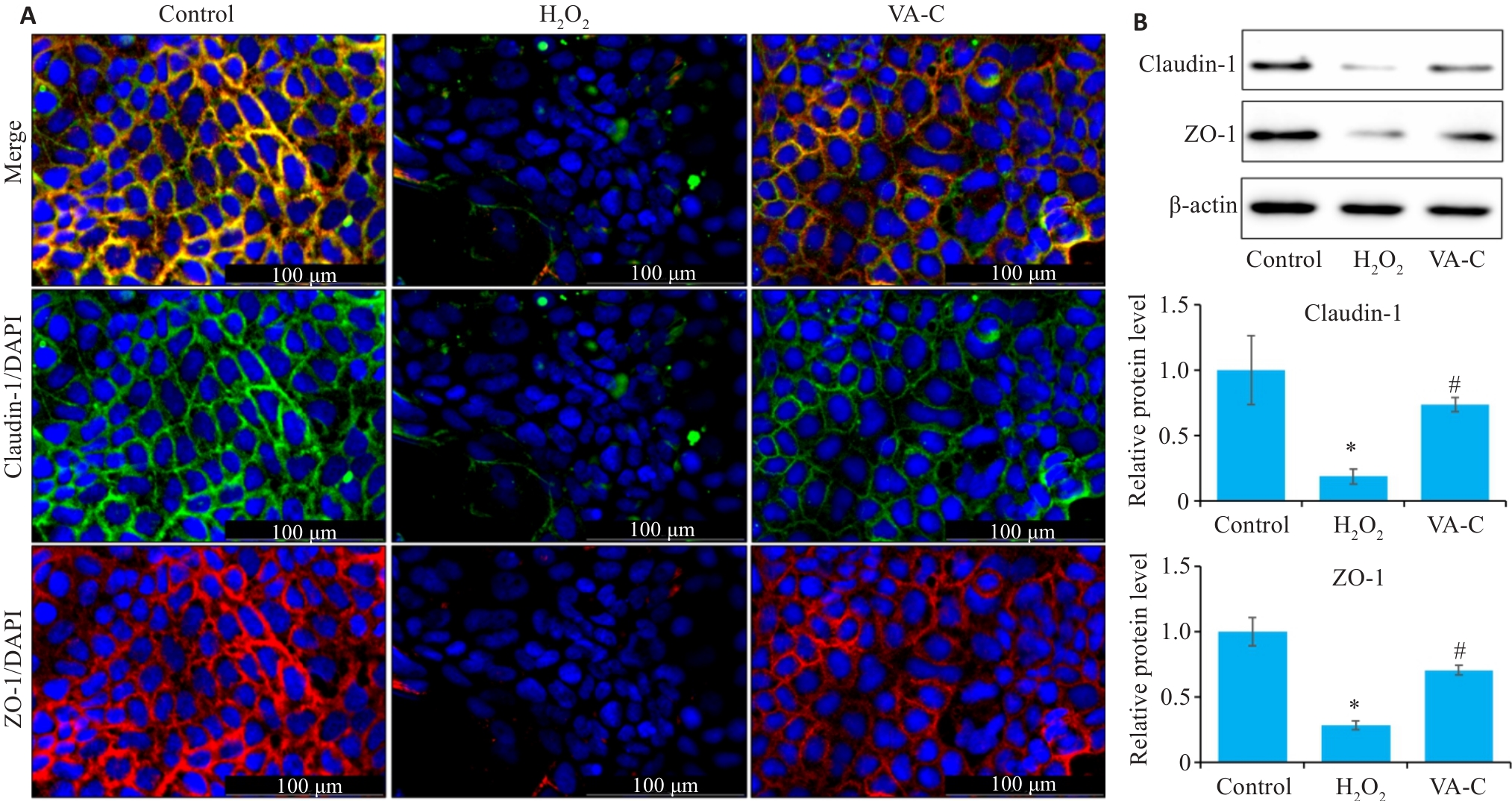
Fig.7 Effect of VA on intestinal barrier function of H₂O₂-induced Caco-2 cells (n=10). A: Immunofluorescence staining for claudin-1 and ZO-1 in each group. B: Western blotting for detecting expression levels of claudin-1 and ZO-1 in each group. *P<0.05 vs Control group; #P<0.05 vs H₂O₂ group.
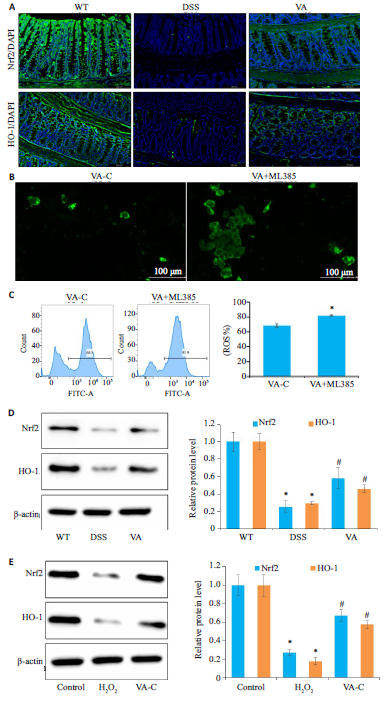
Fig.8 VA alleviates intestinal oxidativestress in colitis mice by activating the Nrf2/HO-1 pathway. A: Immunofluorescencestaining of Nrf2/HO-1 in the colons of themice in each group. B: Intracellular ROSdetection using the fluorescent probe(DCFH-DA) in VA treatment group andML385 inhibitor group. C: IntracellularROS detection by flow cytometry in VAtreatment group and ML385 inhibitorgroup. D: Western blotting for detectingcolonic expression levels of Nrf2/HO-1 ofthe mice in each group. E: Western blottingfor detecting esxpression levels of Nrf2/HO-1 in Caco-2 cells in each group. *P<0.05vs (WT/VA-C/control) group. #PP<0.05 vs(DSS/H₂O₂) group.
| [1] | Bisgaard TH, Allin KH, Keefer L, et al. Depression and anxiety in inflammatory bowel disease: epidemiology, mechanisms and treatment[J]. Nat Rev Gastroenterol Hepatol, 2022, 19(11): 717-26. doi:10.1038/s41575-022-00634-6 |
| [2] | Xu S, Li XZ, Zhang SH, et al. Oxidative stress gene expression, DNA methylation, and gut microbiota interaction trigger Crohn's disease: a multi-omics Mendelian randomization study[J]. BMC Med, 2023, 21(1): 179. doi:10.1186/s12916-023-02878-8 |
| [3] | Sahoo DK, Heilmann RM, Paital B, et al. Oxidative stress, hormones, and effects of natural antioxidants on intestinal inflammation in inflammatory bowel disease[J]. Front Endocrinol (Lausanne), 2023, 14: 1217165. doi:10.3389/fendo.2023.1217165 |
| [4] | Wang S, Dong ZX, Wan XJ. Global, regional, and national burden of inflammatory bowel disease and its associated Anemia, 1990 to 2019 and predictions to 2050: an analysis of the global burden of disease study 2019[J]. Autoimmun Rev, 2024, 23(3): 103498. doi:10.1016/j.autrev.2023.103498 |
| [5] | Chen YY, Cui WW, Li X, et al. Interaction between commensal bacteria, immune response and the intestinal barrier in inflammatory bowel disease[J]. Front Immunol, 2021, 12: 761981. doi:10.3389/fimmu.2021.761981 |
| [6] | Hsu NY, Nayar S, Gettler K, et al. NOX1 is essential for TNFα-induced intestinal epithelial ROS secretion and inhibits M cell signatures[J]. Gut, 2023, 72(4): 654-62. doi:10.1136/gutjnl-2021-326305 |
| [7] | Zheng MY, Han R, Yuan YL, et al. The role of Akkermansia muciniphila in inflammatory bowel disease: Current knowledge and perspectives[J]. Front Immunol, 2023, 13: 1089600. doi:10.3389/fimmu.2022.1089600 |
| [8] | Yu QW, Chen SY, Tang HW, et al. Veratric acid alleviates liver ischemia/reperfusion injury by activating the Nrf2 signaling pathway[J]. Int Immunopharmacol, 2021, 101(Pt B): 108294. doi:10.1016/j.intimp.2021.108294 |
| [9] | Saravanakumar M, Raja B. Veratric acid, a phenolic acid attenuates blood pressure and oxidative stress in L-NAME induced hypertensive rats[J]. Eur J Pharmacol, 2011, 671(1/2/3): 87-94. doi:10.1016/j.ejphar.2011.08.052 |
| [10] | Bian WC, Wei LL, Wang KX. Carthamin yellow alleviates dextran sodium sulfate-induced ulcerative colitis by repairing the intestinal barrier and activating the Nrf2/GPX4 axis[J]. Int Immuno-pharmacol, 2024, 141: 113020. doi:10.1016/j.intimp.2024.113020 |
| [11] | Huang LY, Qian WW, Xu YH, et al. Mesenteric adipose tissue contributes to intestinal fibrosis in Crohn’s disease through the ATX-LPA axis[J]. J Crohns Colitis, 2022, 16(7): 1124-39. doi:10.1093/ecco-jcc/jjac017 |
| [12] | Schultz M, Tonkonogy SL, Sellon RK, et al. IL-2-deficient mice raised under germfree conditions develop delayed mild focal intestinal inflammation[J]. Am J Physiol, 1999, 276(6): G1461-72. doi:10.1152/ajpgi.1999.276.6.g1461 |
| [13] | Biasi F, Leonarduzzi G, Oteiza PI, et al. Inflammatory bowel disease: mechanisms, redox considerations, and therapeutic targets[J]. Antioxid Redox Signal, 2013, 19(14): 1711-47. doi:10.1089/ars.2012.4530 |
| [14] | Farzaei MH, Bahramsoltani R, Abdolghaffari AH, et al. A mechanistic review on plant-derived natural compounds as dietary supplements for prevention of inflammatory bowel disease[J]. Expert Rev Gastroenterol Hepatol, 2016, 10(6): 745-58. doi:10.1586/17474124.2016.1145546 |
| [15] | Wang QB, Sun LY, Gong ZD, et al. Veratric acid inhibits LPS-induced IL-6 and IL-8 production in human gingival fibroblasts[J]. Inflammation, 2016, 39(1): 237-42. doi:10.1007/s10753-015-0243-9 |
| [16] | Guo XX, Xu Y, Geng RX, et al. Curcumin alleviates dextran sulfate sodium-induced colitis in mice through regulating gut microbiota[J]. Mol Nutr Food Res, 2022, 66(8): e2100943. doi:10.1002/mnfr.202100943 |
| [17] | Jayakumar T, Huang CJ, Yen TL, et al. Activation of Nrf2 by esculetin mitigates inflammatory responses through suppression of NF-κB signaling cascade in RAW 264.7 cells[J]. Molecules, 2022, 27(16): 5143. doi:10.3390/molecules27165143 |
| [18] | Gao W, Guo L, Yang Y, et al. Dissecting the crosstalk between Nrf2 and NF-κB response pathways in drug-induced toxicity[J]. Front Cell Dev Biol, 2022, 9: 809952. doi:10.3389/fcell.2021.809952 |
| [19] | Dong LJ, Xie JW, Wang YY, et al. Mannose ameliorates experimental colitis by protecting intestinal barrier integrity[J]. Nat Commun, 2022, 13(1): 4804. doi:10.1038/s41467-022-32505-8 |
| [20] | Kinchen J, Chen HH, Parikh K, et al. Structural remodeling of the human colonic mesenchyme in inflammatory bowel disease[J]. Cell, 2018, 175(2): 372-86.e17. doi:10.1016/j.cell.2018.08.067 |
| [21] | Neurath MF, Artis D, Becker C. The intestinal barrier: a pivotal role in health, inflammation, and cancer[J]. Lancet Gastroenterol Hepatol, 2025, 10(6): 573-92. doi:10.1016/s2468-1253(24)00390-x |
| [22] | Li MQ, Lv RZ, Wang CZ, et al. Tricholoma matsutake-derived peptide WFNNAGP protects against DSS-induced colitis by ameliorating oxidative stress and intestinal barrier dysfunction[J]. Food Funct, 2021, 12(23): 11883-97. doi:10.1039/d1fo02806e |
| [23] | Almousa AA, Meurens F, Krol ES, et al. Linoorbitides and enterolactone mitigate inflammation-induced oxidative stress and loss of intestinal epithelial barrier integrity[J]. Int Immuno-pharmacol, 2018, 64: 42-51. doi:10.1016/j.intimp.2018.08.012 |
| [24] | Yang XW, Mao ZM, Huang YR, et al. Reductively modified albumin attenuates DSS-Induced mouse colitis through rebalancing systemic redox state[J]. Redox Biol, 2021, 41: 101881. doi:10.1016/j.redox.2021.101881 |
| [25] | Gustafsson JK, Johansson MEV. The role of goblet cells and mucus in intestinal homeostasis[J]. Nat Rev Gastroenterol Hepatol, 2022, 19(12): 785-803. doi:10.1038/s41575-022-00675-x |
| [26] | Kim YS, Ho SB. Intestinal goblet cells and mucins in health and disease: recent insights and progress[J]. Curr Gastroenterol Rep, 2010, 12(5): 319-30. doi:10.1007/s11894-010-0131-2 |
| [27] | Zhang MM, Wu CC. The relationship between intestinal goblet cells and the immune response[J]. Biosci Rep, 2020, 40(10): BSR20201471. doi:10.1042/bsr20201471 |
| [28] | Yang X, Li GL, Lou PB, et al. Excessive nucleic acid R-loops induce mitochondria-dependent epithelial cell necroptosis and drive spontaneous intestinal inflammation[J]. Proc Natl Acad Sci USA, 2024, 121(1): e2307395120. doi:10.1073/pnas.2307395120 |
| [29] | Arda-Pirincci P, Aykol-Celik G. Galectin-1 reduces the severity of dextran sulfate sodium (DSS)-induced ulcerative colitis by suppressing inflammatory and oxidative stress response[J]. Bosn J Basic Med Sci, 2020, 20(3): 319-28. |
| [30] | Pan TL, Liu SQ, Liao QC, et al. Dietary supplement of veratric acid alleviates liver steatosis and reduces abdominal fat deposition in broilers[J]. Poult Sci, 2024, 103(12): 104406. doi:10.1016/j.psj.2024.104406 |
| [31] | Yan B, Mao XJ, Hu SS, et al. Spermidine protects intestinal mucosal barrier function in mice colitis via the AhR/Nrf2 and AhR/STAT3 signaling pathways[J]. Int Immunopharmacol, 2023, 119: 110166. doi:10.1016/j.intimp.2023.110166 |
| [32] | Ni J, Wu GD, Albenberg L, et al. Gut microbiota and IBD: causation or correlation[J]? Nat Rev Gastroenterol Hepatol, 2017, 14(10): 573-84. doi:10.1038/nrgastro.2017.88 |
| [33] | Qiu P, Ishimoto T, Fu LF, et al. The gut microbiota in inflammatory bowel disease[J]. Front Cell Infect Microbiol, 2022, 12: 733992. doi:10.3389/fcimb.2022.733992 |
| [1] | Yuce PENG, Yi JIANG, Dan MA, An HE, Dingyi LÜ, Minghao LUO, Suxin LUO. Isovitexin alleviates myocardial oxidative stress injury in diabetic mice by enhancing myocardial SIRT3 expression and reducing oxidative stress [J]. Journal of Southern Medical University, 2026, 46(2): 316-324. |
| [2] | Tong QIAO, Lin YIN, Keni ZHANG, Minzhu NIU, Ju HUANG, Zhijun Geng, Jing LI, Jianguo HU. Poricoic acid A alleviates dextran sulfate sodium-induced colitis in mice by regulating AMPK/mTOR-mediated autophagy and inhibiting intestinal epithelial cell apoptosis [J]. Journal of Southern Medical University, 2026, 46(1): 131-140. |
| [3] | Jingxian WANG, Zijing REN, Peiyang ZHOU. S1PR5 activation or overexpression enhances barrier function of mouse brain microvascular endothelial cells against OGD/R injury by modulating oxidative stress [J]. Journal of Southern Medical University, 2025, 45(7): 1451-1459. |
| [4] | Minzhu NIU, Lixia YIN, Tong QIAO, Lin YIN, Keni ZHANG, Jianguo HU, Chuanwang SONG, Zhijun GENG, Jing LI. Ecliptasaponin A ameliorates DSS-induced colitis in mice by suppressing M1 macrophage polarization via inhibiting the JAK2/STAT3 pathway [J]. Journal of Southern Medical University, 2025, 45(6): 1297-1306. |
| [5] | Anbang ZHANG, Xiuqi SUN, Bo PANG, Yuanhua WU, Jingyu SHI, Ning ZHANG, Tao YE. Electroacupuncture pretreatment alleviates cerebral ischemia-reperfusion injury in rats by inhibiting ferroptosis through the gut-brain axis and the Nrf2/HO-1 signaling pathway [J]. Journal of Southern Medical University, 2025, 45(5): 911-920. |
| [6] | Fei CHU, Xiaohua CHEN, Bowen SONG, Jingjing YANG, Lugen ZUO. Moslosooflavone ameliorates dextran sulfate sodium-induced colitis in mice by suppressing intestinal epithelium apoptosis via inhibiting the PI3K/AKT signaling pathway [J]. Journal of Southern Medical University, 2025, 45(4): 819-828. |
| [7] | Zhi GAO, Ao WU, Zhongxiang HU, Peiyang SUN. Bioinformatics analysis of oxidative stress and immune infiltration in rheumatoid arthritis [J]. Journal of Southern Medical University, 2025, 45(4): 862-870. |
| [8] | Pengwei HUANG, Jie CHEN, Jinhu ZOU, Xuefeng GAO, Hong CAO. Quercetin mitigates HIV-1 gp120-induced rat astrocyte neurotoxicity via promoting G3BP1 disassembly in stress granules [J]. Journal of Southern Medical University, 2025, 45(2): 304-312. |
| [9] | Xiaoyu LU, Zhihui LIU, Ye LIU, Tianxiao PANG, Rong BIAN, Ling GUO, Xuehong HE. Shenqi Xiezhuo Decoction alleviates renal fibrosis in rats by ameliorating oxidative stress and inflammation through the Rap1/MAPK/FoxO3a signaling pathway [J]. Journal of Southern Medical University, 2025, 45(12): 2585-2597. |
| [10] | Lu TAO, Yue CHEN, Linlin HUANG, Wang ZHENG, Xue SONG, Ping XIANG, Jianguo HU. Niranthin ameliorates Crohn's disease-like enteritis in mice by inhibiting intestinal epithelial cell apoptosis and protecting intestinal barrier via modulating p38/JNK signaling [J]. Journal of Southern Medical University, 2025, 45(11): 2483-2495. |
| [11] | Shuxian LIN, Lina GUO, Yan MA, Yao XIONG, Yingxi HE, Xinzhu XU, Wen SHENG, Suhua XU, Feng QIU. Lactobacillus plantarum ZG03 alleviates oxidative stress via its metabolites short-chain fatty acids [J]. Journal of Southern Medical University, 2025, 45(10): 2223-2230. |
| [12] | Yuming ZHANG, Shicheng XIA, Linlin ZHANG, Mengxi CHEN, Xiaojing LIU, Qin GAO, Hongwei YE. Protective effect of Lonicerae japonicae flos extract against doxorubicin-induced liver injury in mice [J]. Journal of Southern Medical University, 2024, 44(8): 1571-1581. |
| [13] | Zhijun REN, Jianxin DIAO, Yiting WANG. Xionggui Decoction alleviates heart failure in mice with myocardial infarction by inhibiting oxidative stress-induced cardiomyocyte apoptosis [J]. Journal of Southern Medical University, 2024, 44(7): 1416-1424. |
| [14] | Guoxin LIANG, Hongyue TANG, Chang GUO, Mingming ZHANG. MiR-224-5p overexpression inhibits oxidative stress by regulating the PI3K/Akt/FoxO1 axis to attenuate hypoxia/reoxygenation-induced cardiomyocyte injury [J]. Journal of Southern Medical University, 2024, 44(6): 1173-1181. |
| [15] | CHEN Guodong, LUO Suxin. Colchicine alleviates myocardial ischemia-reperfusion injury in mice by activating AMPK [J]. Journal of Southern Medical University, 2024, 44(2): 226-235. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||