Journal of Southern Medical University ›› 2025, Vol. 45 ›› Issue (11): 2483-2495.doi: 10.12122/j.issn.1673-4254.2025.11.21
Lu TAO1,2( ), Yue CHEN2, Linlin HUANG2, Wang ZHENG2, Xue SONG1,2, Ping XIANG1,2, Jianguo HU2(
), Yue CHEN2, Linlin HUANG2, Wang ZHENG2, Xue SONG1,2, Ping XIANG1,2, Jianguo HU2( )
)
Received:2025-04-28
Online:2025-11-20
Published:2025-11-28
Contact:
Jianguo HU
E-mail:bytaolu@bbmu.edu.cn;jghu9200@bbmu.edu.cn
Lu TAO, Yue CHEN, Linlin HUANG, Wang ZHENG, Xue SONG, Ping XIANG, Jianguo HU. Niranthin ameliorates Crohn's disease-like enteritis in mice by inhibiting intestinal epithelial cell apoptosis and protecting intestinal barrier via modulating p38/JNK signaling[J]. Journal of Southern Medical University, 2025, 45(11): 2483-2495.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2025.11.21
| Gene | Primer sequences (5'-3') |
|---|---|
| TNF-α | F: CACGCTCTTCTGTCTACTGAACTTC |
| R: CTTGGTGGTTTGTGAGTGTGAGG | |
| IL-1β | F: AATCTCGCAGCAGCACATCAAC |
| R: AGGTCCACGGGAAAGACACAG | |
| IL-6 | F: GAGAGGAGACTTCACAGAGGATACC |
| R: TCATTTCCACGATTTCCCAGAGAAC | |
| IL-10 | F: GGACAACATACTGCTAACCGACTC |
| R: GGGCATCACTTCTACCAGGTAAAAC | |
| IL-17A | F: TGGCGGCTACAGTGAAGGC |
| R: AGGGAGTTAAAGACTTTGAGGTTGAC | |
| GAPDH | F: AACTCCCACTCTTCCACCTTCG |
| R: TCCACCACCCTGTTGCTGTAG |
Tab.1 Primer sequences for qRT-PCR in this study
| Gene | Primer sequences (5'-3') |
|---|---|
| TNF-α | F: CACGCTCTTCTGTCTACTGAACTTC |
| R: CTTGGTGGTTTGTGAGTGTGAGG | |
| IL-1β | F: AATCTCGCAGCAGCACATCAAC |
| R: AGGTCCACGGGAAAGACACAG | |
| IL-6 | F: GAGAGGAGACTTCACAGAGGATACC |
| R: TCATTTCCACGATTTCCCAGAGAAC | |
| IL-10 | F: GGACAACATACTGCTAACCGACTC |
| R: GGGCATCACTTCTACCAGGTAAAAC | |
| IL-17A | F: TGGCGGCTACAGTGAAGGC |
| R: AGGGAGTTAAAGACTTTGAGGTTGAC | |
| GAPDH | F: AACTCCCACTCTTCCACCTTCG |
| R: TCCACCACCCTGTTGCTGTAG |
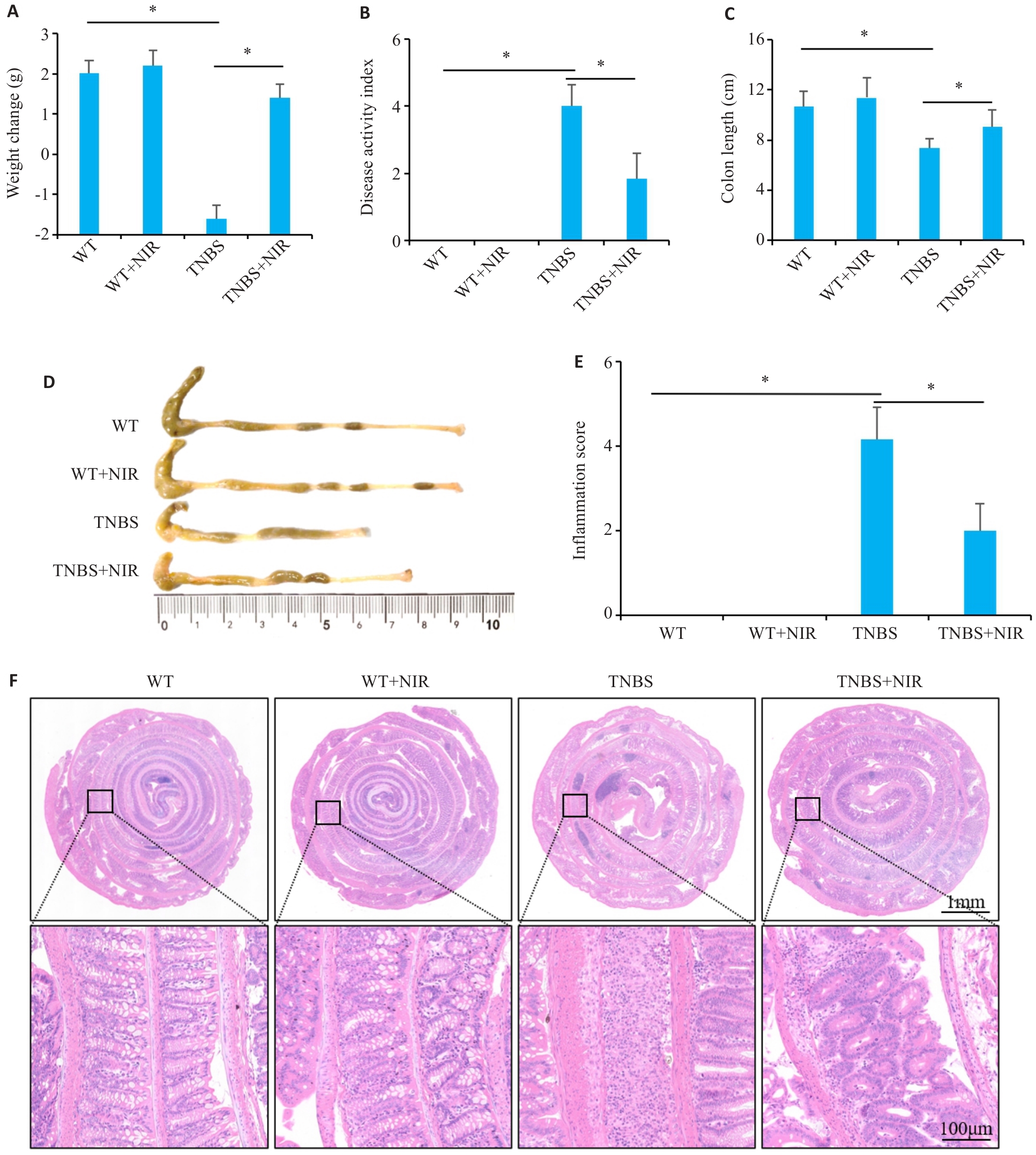
Fig.1 Effect of niranthin (NIR) treatment on symptoms of TNBS-induced colitis in mice. A: Body weight changes of the mice. B: DAI score. C: Colon length. D: Comparison of colon length of the mice among the 4 groups. E: Colon inflammation score of the mice. F: HE staining of the colon tissues of the mice in the 4 groups.*P<0.05.
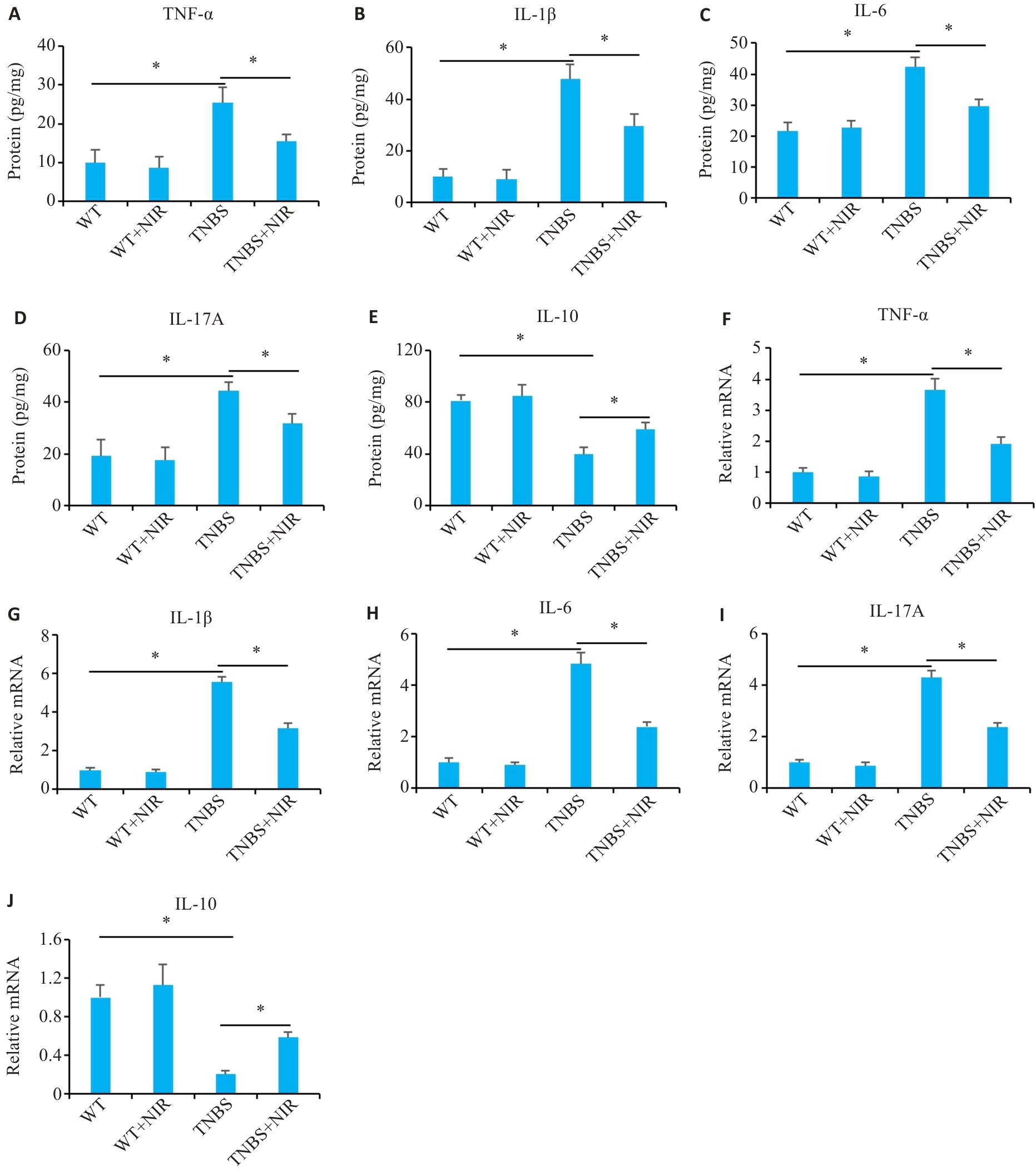
Fig.2 Effect of NIR treatment on inflammatory factor levels in mice with TNBS-induced colitis. A-E: Results of ELISA for detecting the levels of IL-6, IL-1β, IL-17A, TNF-α and IL-10 in the colon tissue of TNBS-induced mice. F-J: qRT-PCR for detecting IL-6, IL-1β, IL-17A, TNF‑α and IL-10 mRNA expressions in the colon tissues of TNBS-induced mice. *P<0.05.
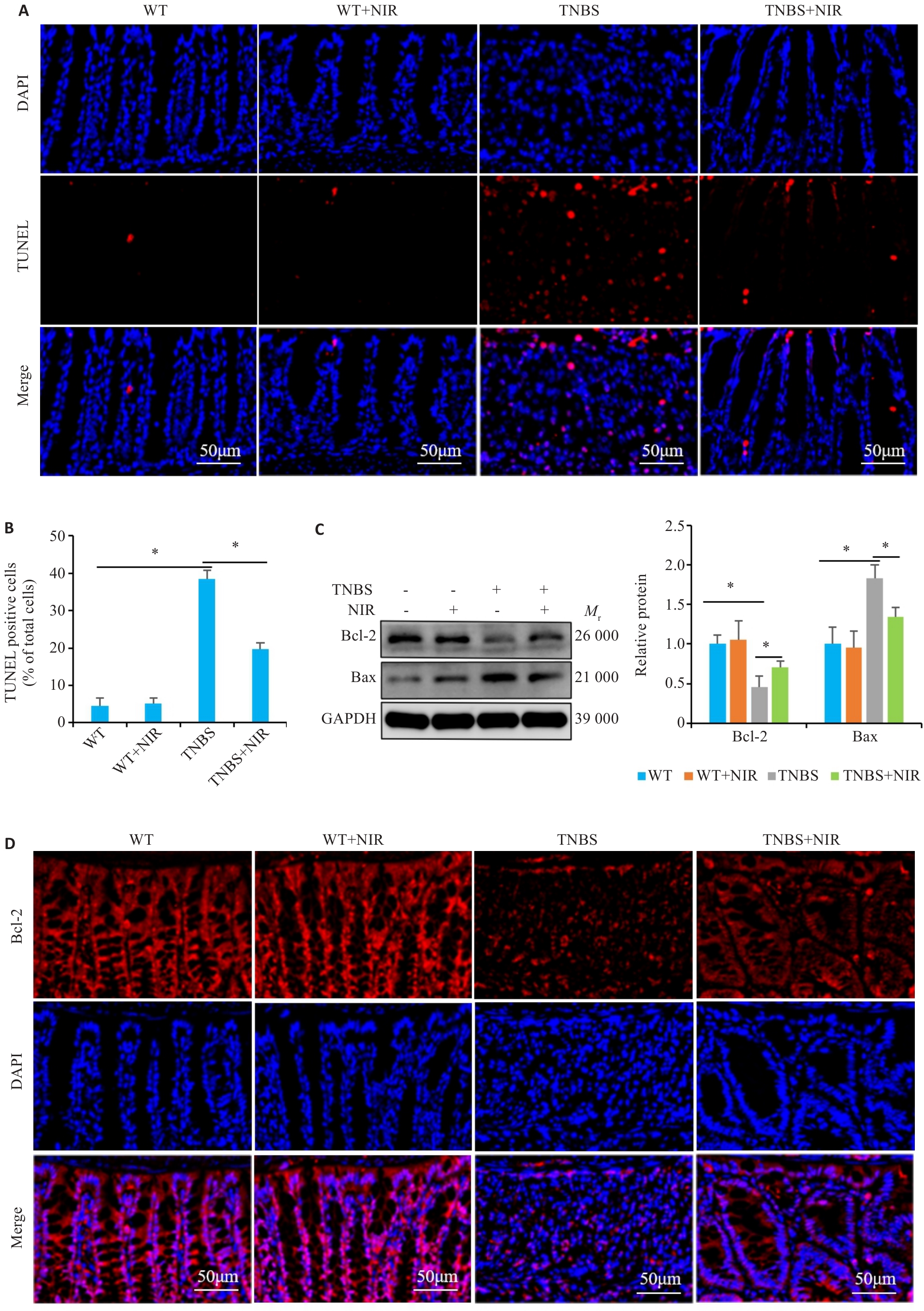
Fig.3 Effect of NIR treatment on TNBS-induced apoptosis of intestinal epithelial cells in the mouse models of colitis. A, B: TUNEL staining for detecting apoptosis in TNBS-induced mice with NIR treatment. C: Western blotting for detecting the expression of Bcl-2 and Bax proteins in TNBS-induced mice with NIR treatment. D: Immunofluorescence staining for detecting Bcl-2 expression in TNBS-induced mice with NIR treatment. *P<0.05.

Fig.4 Effect of NIR treatment on intestinal barrier function in mice with TNBS-induced colitis. A: Immunofluorescence staining for detecting ZO-1 and Claudin-1 expression in TNBS-induced mice with NIR treatment. B: Western blotting for detecting ZO-1 and claudin-1 protein expressions in TNBS-induced mice with NIR treatment. *P<0.05.
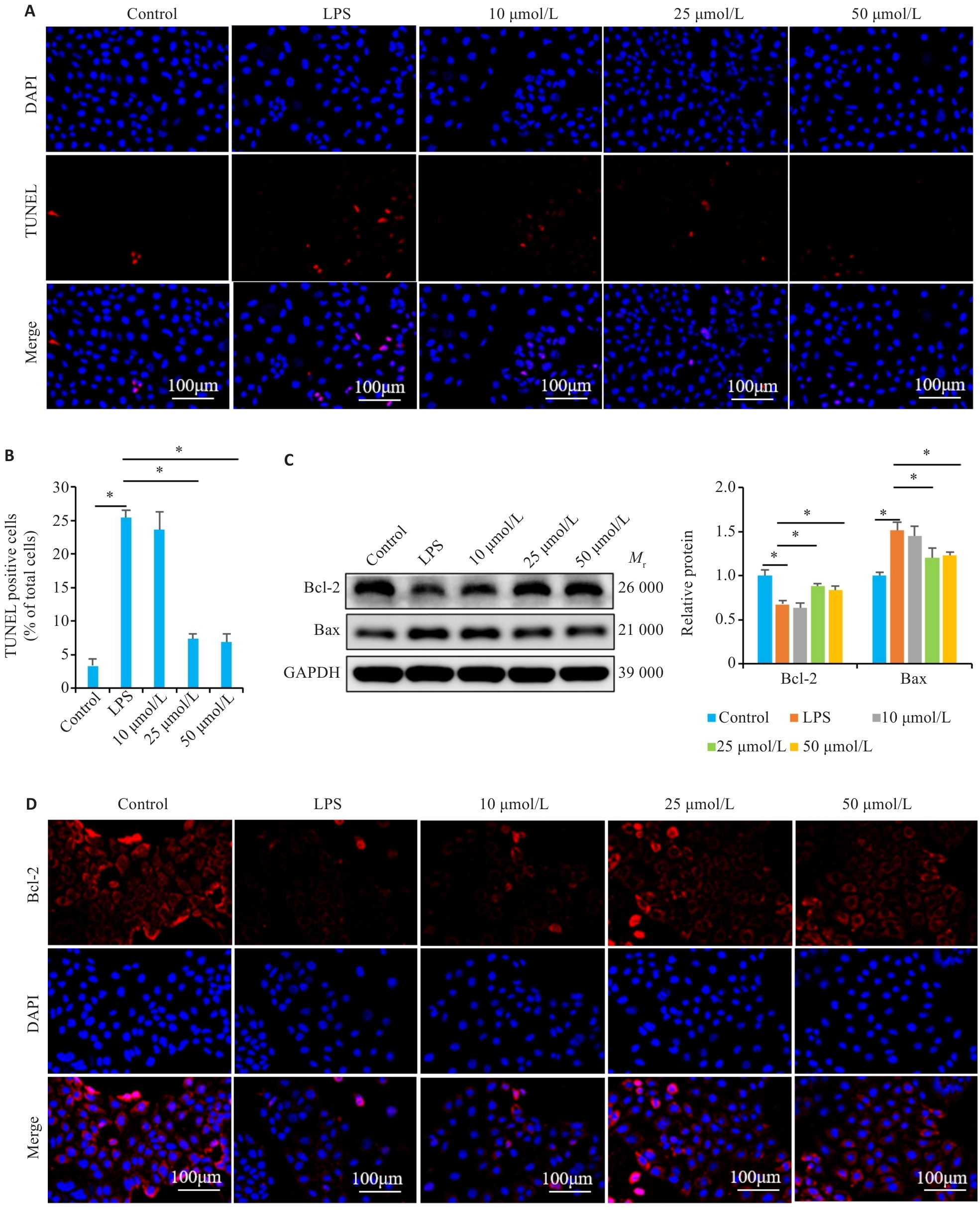
Fig.5 Effect of NIR treatment on LPS-induced apoptosis in Caco-2 cells. A, B: TUNEL staining for detecting LPS-induced apoptosis of Caco-2 cells with NIR treatment. C: Western blotting for detecting the expression of Bcl-2 and Bax proteins in LPS-induced Caco-2 cells with NIR treatment. D: Immunofluorescence staining for detecting Bcl-2 expression in LPS-induced Caco-2 cells with NIR treatment. *P<0.05.
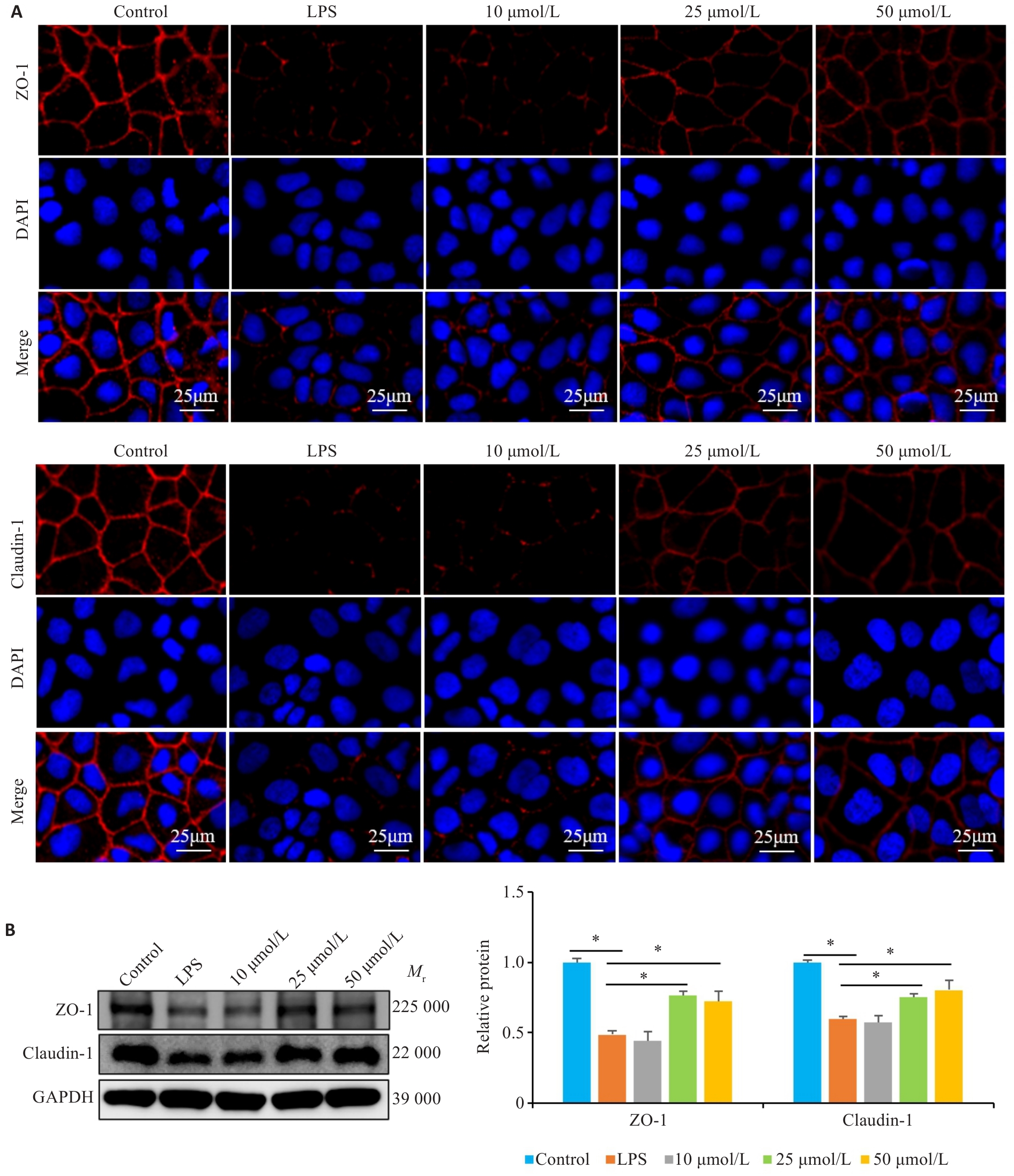
Fig. 6 Effect of NIR treatment on expressions of tight junction proteins in LPS-induced Caco-2 cells. A: Immunofluorescence staining showing ZO-1 and claudin-1 expressions in LPS-induced Caco-2 cells with NIR treatment. B: Western blotting for detecting ZO-1 and claudin-1 protein expressions in LPS-induced Caco-2 cells with NIR treatment. *P<0.05.
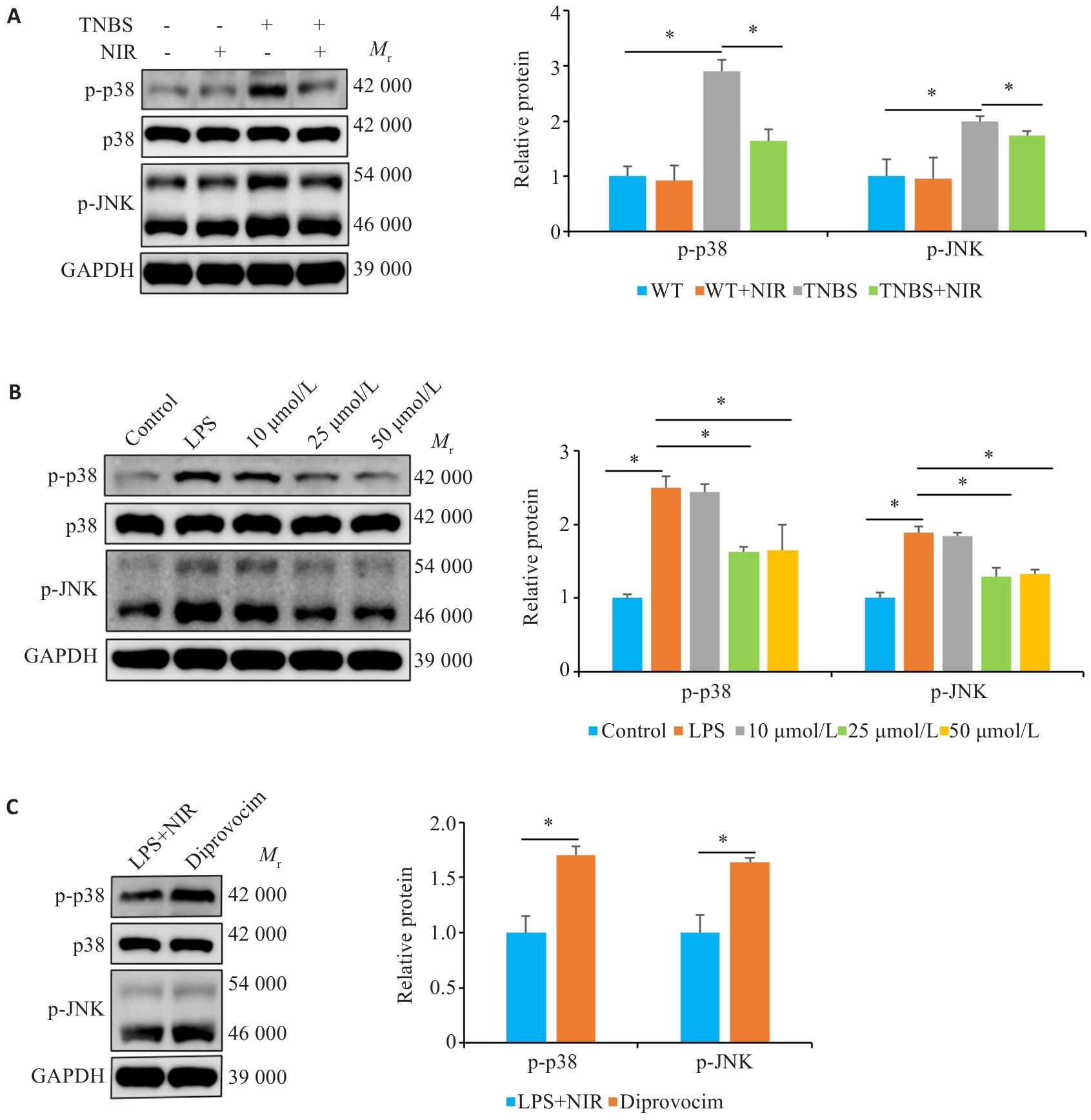
Fig. 7 NIR treatment modulates p38/JNK signaling. A: Western blotting for detecting p-p38 and p-JNK protein expressions in TNBS-induced mice with NIR treatment. B: Western blotting for detecting p-p38 and p-JNK protein expressions in LPS-induced Caco-2 cells with NIR treatment. C: Western blotting for detecting p-p38 and p-JNK protein expressions in the colon tissues of the mice with NIR and diprovocim treatment. *P<0.05.
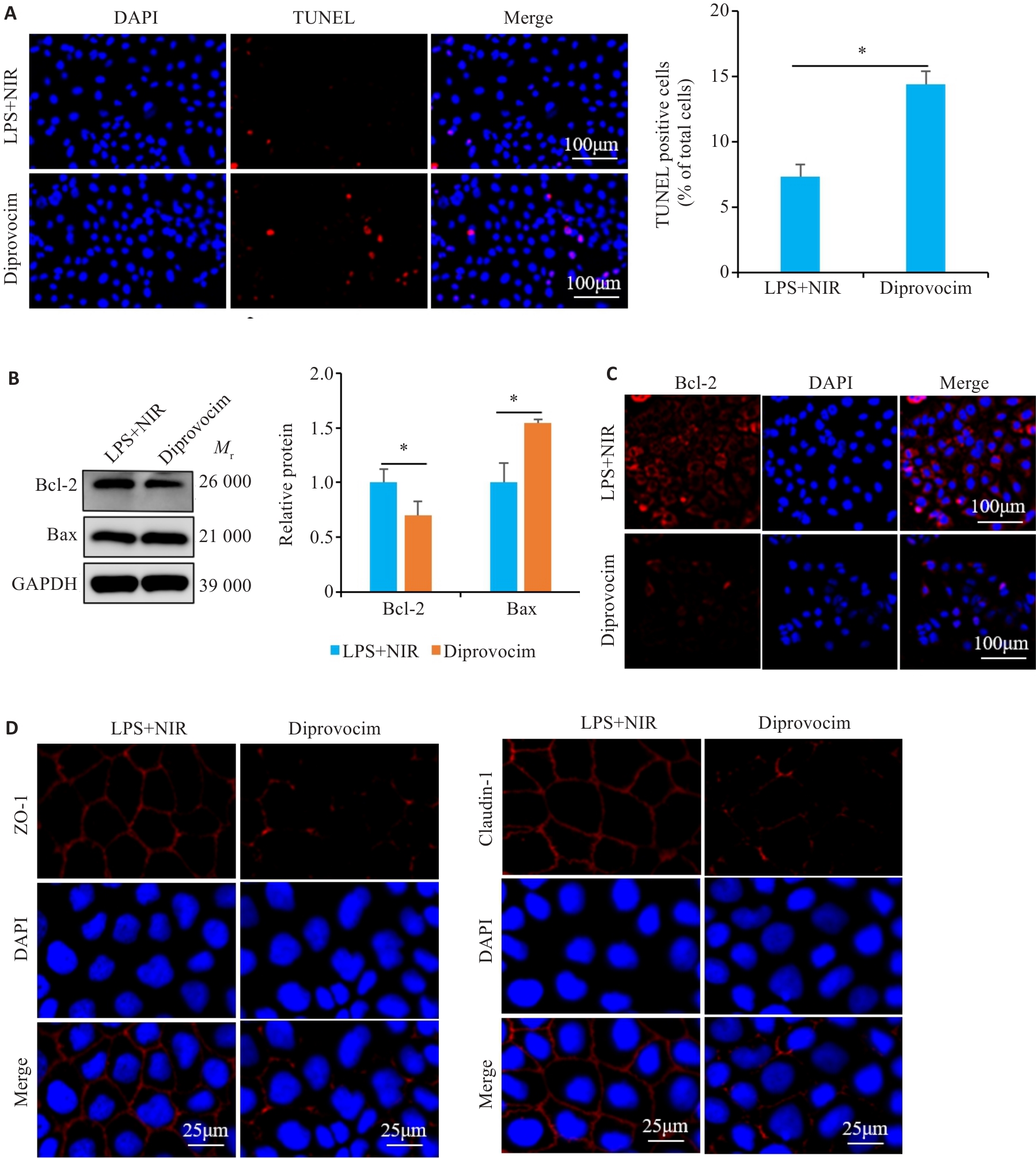
Fig. 8 Effects of diprovocim intervention on apoptosis and expressions of tight junction proteins in NIR-treated Caco-2 cells. A: TUNEL staining for detecting apoptosis in NIR-treated Caco-2 cells by Diprovocim treatment. B: Western blotting for detecting the expression of Bcl-2 and Bax proteins in NIR-treated Caco-2 cells with diprovocim treatment. C, D: Immunofluorescence staining for detecting Bcl-2, ZO-1 and Claudin-1 expression in NIR-treated Caco-2 cells with diprovocim treatment. *P<0.05.
| [1] | Dolinger M, Torres J, Vermeire S. Crohn’s disease[J]. Lancet, 2024, 403(10432): 1177-91. doi:10.1016/s0140-6736(23)02586-2 |
| [2] | Loftus EV Jr, Panés J, Lacerda AP, et al. Upadacitinib induction and maintenance therapy for Crohn’s disease[J]. N Engl J Med, 2023, 388(21): 1966-80. doi:10.1056/nejmoa2212728 |
| [3] | Honap S, Jairath V, Danese S, et al. Navigating the complexities of drug development for inflammatory bowel disease[J]. Nat Rev Drug Discov, 2024, 23(7): 546-62. doi:10.1038/s41573-024-00953-0 |
| [4] | Wang L, Song X, Zhou YQ, et al. Sclareol protected against intestinal barrier dysfunction ameliorating Crohn’s disease-like colitis via Nrf2/NF-B/MLCK signalling[J]. Int Immunopharmacol, 2024, 133: 112140. doi:10.1016/j.intimp.2024.112140 |
| [5] | Zhou L, Zhu L, Wu X, et al. Decreased TMIGD1 aggravates colitis and intestinal barrier dysfunction via the BANF1-NF-κB pathway in Crohn’s disease[J]. BMC Med, 2023, 21(1): 287. doi:10.1186/s12916-023-02989-2 |
| [6] | Xu S, Peng YF, Yang K, et al. PROTAC based STING degrader attenuates acute colitis by inhibiting macrophage M1 polarization and intestinal epithelial cells pyroptosis mediated by STING-NLRP3 axis[J]. Int Immunopharmacol, 2024, 141: 112990. doi:10.1016/j.intimp.2024.112990 |
| [7] | Tan G, Huang C, Chen J, et al. HMGB1 released from GSDME-mediated pyroptotic epithelial cells participates in the tumorigenesis of colitis-associated colorectal cancer through the ERK1/2 pathway[J]. J Hematol Oncol, 2020, 13(1): 149. doi:10.1186/s13045-020-00985-0 |
| [8] | Liu J, Di B, Xu LL. Recent advances in the treatment of IBD: targets, mechanisms and related therapies[J]. Cytokine Growth Factor Rev, 2023, 71: 1-12. doi:10.1016/j.cytogfr.2023.07.001 |
| [9] | Veyrard P, Nancey S, Roblin X. Editorial: 5-ASA in IBD patients on biologics-' stop or continue'[J]? Aliment Pharmacol Ther, 2021, 54(6): 843-4. doi:10.1111/apt.16541 |
| [10] | Sridhar A, Bakke I, Gopalakrishnan S, et al. Tofacitinib and budesonide treatment affect stemness and chemokine release in IBD patient-derived colonoids[J]. Sci Rep, 2025, 15(1): 3753. doi:10.1038/s41598-025-86314-2 |
| [11] | Biologic therapy for inflammatory bowel disease: real-world comparative effectiveness and impact of drug sequencing in 13 222 patients within the UK IBD BioResource[J]. J Crohns Colitis, 2024, 18(6): 790-800. doi:10.1093/ecco-jcc/jjad212.1168 |
| [12] | Ota R, Karasawa D, Oshima M, et al. Asymmetric total synthesis of four bioactive lignans using donor–acceptor cyclopropanes and bioassay of (-)- and (+)-niranthin against hepatitis B and influenza viruses[J]. RSC Adv, 2022, 12(8): 4635-9. doi:10.1039/d2ra00499b |
| [13] | Harikrishnan H, Jantan I, Alagan A, et al. Modulation of cell signaling pathways by Phyllanthus amarus and its major constituents: potential role in the prevention and treatment of inflammation and cancer[J]. Inflammopharmacology, 2020, 28(1): 1-18. doi:10.1007/s10787-019-00671-9 |
| [14] | Chowdhury S, Mukherjee T, Mukhopadhyay R, et al. The lignan niranthin poisons Leishmania donovani topoisomerase IB and favours a Th1 immune response in mice[J]. EMBO Mol Med, 2012, 4(10): 1126-43. doi:10.1002/emmm.201201316 |
| [15] | Harikrishnan H, Jantan I, Haque MA, et al. Anti-inflammatory effects of hypophyllanthin and niranthin through downregulation of NF-κB/MAPKs/PI3K-Akt signaling pathways[J]. Inflammation, 2018, 41(3): 984-95. doi:10.1007/s10753-018-0752-4 |
| [16] | Tan G, Huang CY, Chen JY, et al. Gasdermin-E-mediated pyroptosis participates in the pathogenesis of Crohn’s disease by promoting intestinal inflammation[J]. Cell Rep, 2021, 35(11): 109265. doi:10.1016/j.celrep.2021.109265 |
| [17] | Spencer DM, Veldman GM, Banerjee S, et al. Distinct inflammatory mechanisms mediate early versus late colitis in mice[J]. Gastroenterology, 2002, 122(1): 94-105. doi:10.1053/gast.2002.30308 |
| [18] | Rath HC, Herfarth HH, Ikeda JS, et al. Normal luminal bacteria, especially Bacteroides species, mediate chronic colitis, gastritis, and arthritis in HLA-B27/human beta2 microglobulin transgenic rats[J]. J Clin Invest, 1996, 98(4): 945-53. doi:10.1172/jci118878 |
| [19] | Morin MD, Wang Y, Jones BT, et al. Diprovocims: A New and Exceptionally Potent Class of Toll-like Receptor Agonists[J]. J Am Chem Soc, 2018, 140(43):14440-54. doi:10.1021/jacs.8b09223 |
| [20] | Wei ZY, Ni X, Cui H, et al. Engeletin attenuates the inflammatory response via inhibiting TLR4-NFκB signaling pathway in Crohn’s disease-like colitis[J]. J Ethnopharmacol, 2025, 336: 118733. doi:10.1016/j.jep.2024.118733 |
| [21] | Mehandru S, Colombel JF. The intestinal barrier, an arbitrator turned provocateur in IBD[J]. Nat Rev Gastroenterol Hepatol, 2021, 18(2): 83-4. doi:10.1038/s41575-020-00399-w |
| [22] | Odenwald MA, Turner JR. The intestinal epithelial barrier: a therapeutic target[J]? Nat Rev Gastroenterol Hepatol, 2017, 14(1): 9-21. doi:10.1038/nrgastro.2016.169 |
| [23] | Huang S, Xie Z, Han J, et al. Protocadherin 20 maintains intestinal barrier function to protect against Crohn’s disease by targeting ATF6[J]. Genome Biol, 2023, 24(1): 159. doi:10.1186/s13059-023-02991-0 |
| [24] | Safari F, Sharifi M, Talebi A, et al. Alleviation of cholestatic liver injury and intestinal permeability by lubiprostone treatment in bile duct ligated rats: role of intestinal FXR and tight junction proteins claudin-1, claudin-2, and occludin[J]. Naunyn Schmiedeberg’s Arch Pharmacol, 2023, 396(9): 2009-22. doi:10.1007/s00210-023-02455-z |
| [25] | Pan Z, Huang J, Hu T, et al. Protective effects of selenium nanoparticles against bisphenol A-induced toxicity in porcine intestinal epithelial cells[J]. Int J Mol Sci, 2023, 24(8): 7242. doi:10.3390/ijms24087242 |
| [26] | Yin M, Shen Z, Yang L, et al. Protective effects of CXCR3/HO-1 gene-modified BMMSCs on damaged intestinal epithelial cells: Role of the p38-MAPK signaling pathway[J]. Int J Mol Med, 2019, 43(5): 2086-102. |
| [27] | Zeng ZW, Shi YM, Cai YH, et al. PHLDA1 protects intestinal barrier function via restricting intestinal epithelial cells apoptosis in inflammatory bowel disease[J]. Exp Cell Res, 2024, 443(1): 114322. doi:10.1016/j.yexcr.2024.114322 |
| [28] | Geng Z, Zuo L, Li J, et al. Ginkgetin improved experimental colitis by inhibiting intestinal epithelial cell apoptosis through EGFR/PI3K/AKT signaling[J]. FASEB J, 2024, 38(14): e23817. doi:10.1096/fj.202400211rr |
| [29] | Xu Q, Liu M, Chao X, et al. Stevioside improves antioxidant capacity and intestinal barrier function while attenuating inflammation and apoptosis by regulating the NF-κB/MAPK pathways in diquat-induced oxidative stress of IPEC-J2 cells[J]. Antioxidants: Basel, 2023, 12(5): 1070. doi:10.3390/antiox12051070 |
| [30] | Xing Z, Li X, He J, et al. OLFM4 modulates intestinal inflammation by promoting IL-22+ILC3 in the gut[J]. Commun Biol, 2024, 7(1): 914. doi:10.1038/s42003-024-06601-y |
| [31] | Xiao L, Zhang WH, Huang Y, et al. Intestinal ischemia-reperfusion induces the release of IL-17A to regulate cell inflammation, apoptosis and barrier damage[J]. Exp Ther Med, 2022, 23(2): 158. doi:10.3892/etm.2021.11081 |
| [32] | Chu C, Ru H, Chen Y, et al. Gallic acid attenuates LPS-induced inflammation in Caco-2 cells by suppressing the activation of the NF-κB/MAPK signaling pathway[J]. Acta Biochim Biophys Sin: Shanghai, 2024, 56(6): 905-15. |
| [33] | Tao H, Bao Z, Fu Z, et al. Chlorothalonil induces the intestinal epithelial barrier dysfunction in Caco-2 cell-based in vitro monolayer model by activating MAPK pathway[J]. Acta Biochim Biophys Sin: Shanghai, 2021, 53(11): 1459-68. doi:10.1093/abbs/gmab125 |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||