Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 532-540.doi: 10.12122/j.issn.1673-4254.2026.03.07
Qile SONG1( ), Yikai MIAO2, Xiaotong FENG1, Yifan WANG1, Wei LIU1, Qi WEI1, Xinru YU1, Wenwen CHEN3(
), Yikai MIAO2, Xiaotong FENG1, Yifan WANG1, Wei LIU1, Qi WEI1, Xinru YU1, Wenwen CHEN3( ), Xiaoyan FU4(
), Xiaoyan FU4( )
)
Received:2025-09-23
Online:2026-03-20
Published:2026-03-26
Contact:
Wenwen CHEN, Xiaoyan FU
E-mail:sqlbangong@163.com;wen-860521@163.com;txyfu66@163.com
Supported by:Qile SONG, Yikai MIAO, Xiaotong FENG, Yifan WANG, Wei LIU, Qi WEI, Xinru YU, Wenwen CHEN, Xiaoyan FU. Selenocystine inhibits colon cancer cell growth by promoting reactive oxygen species generation to trigger oxidative damage[J]. Journal of Southern Medical University, 2026, 46(3): 532-540.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.07
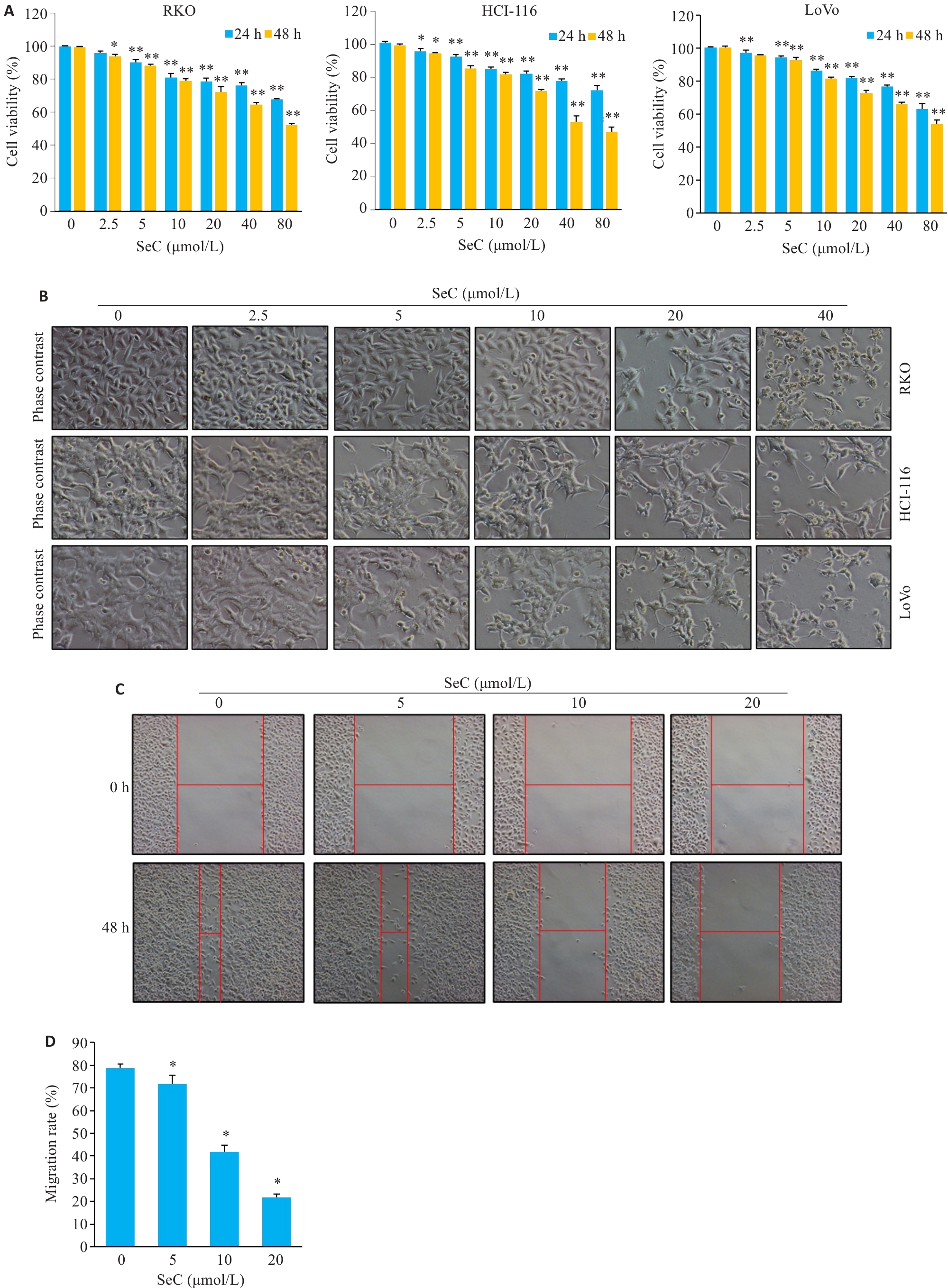
Fig.1 SeC inhibits growth of colon cancer cells in a dose-dependent manner. A: SeC inhibits viability of RKO, HCT-116 and LoVo cells detected by MTT assay. B: Changes in cell morphology (Original magnification: ×200). C: SeC inhibits the migration of RKO cells detected by wound healing assay. (×100). D: Quantitative analysis of cell migration. All experiments were repeated 3 times (n=3). All data were presented as Mean±SD. *P<0.05, **P<0.01 vs SeC 0 μmol/L.
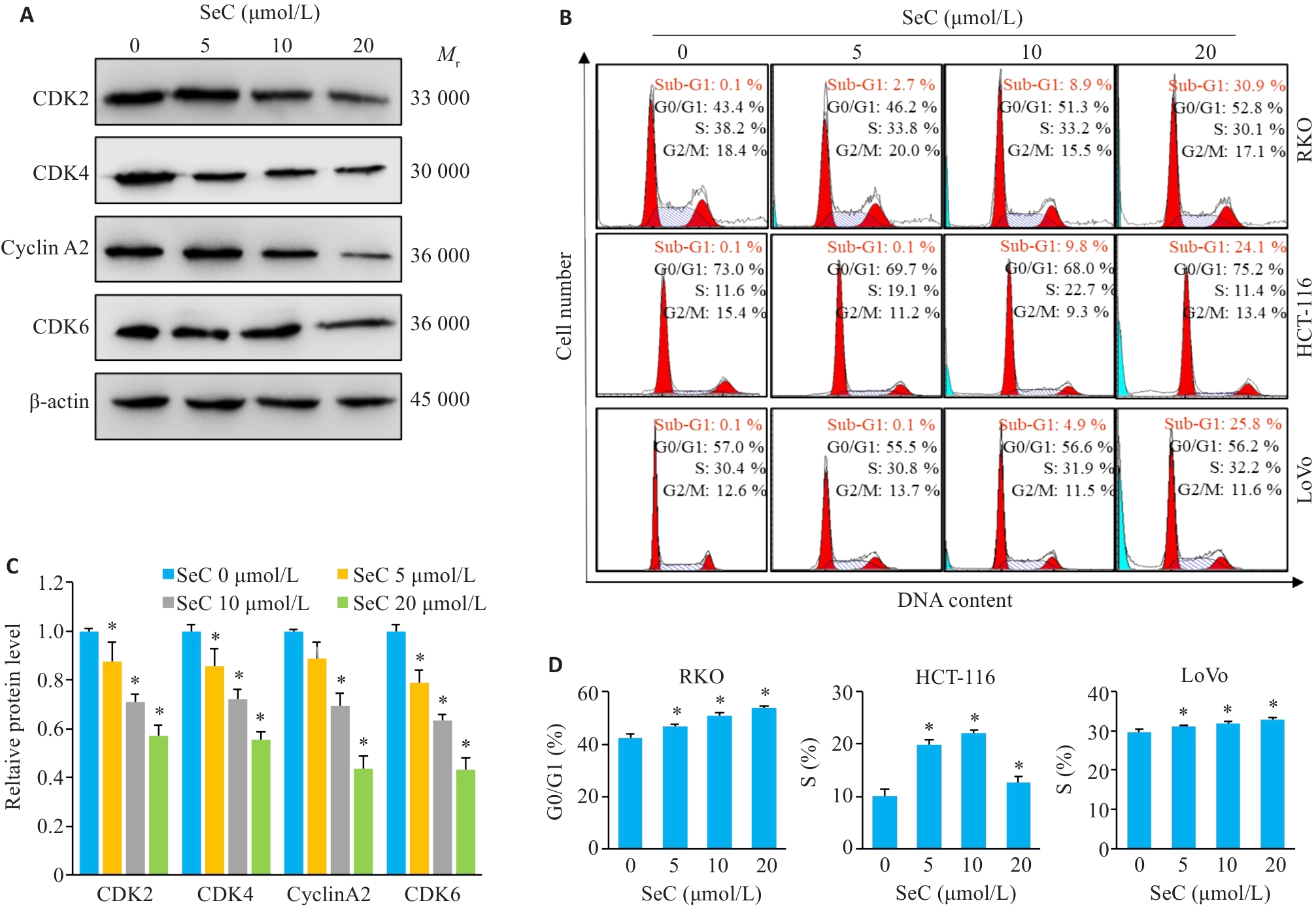
Fig.2 SeC induces cell cycle arrest and apoptosis in colon cancer cells. A: SeC inhibits expressions of RKO cell cycle-related proteins detected by Western blotting. B: SeC-induced cell cycle arrest and apoptosis in RKO, HCT-116, and LoVo cells detected by flow cytometry with PI staining. C: Quantitative analysis of cell cycle proteins. D: Quantitative analysis of cell cycle arrest in RKO, HCT-116, and LoVo cells. All experiments were repeated three times (n=3). All data are presented as Mean±SD. *P<0.05 vs SeC 0 μmol/L.
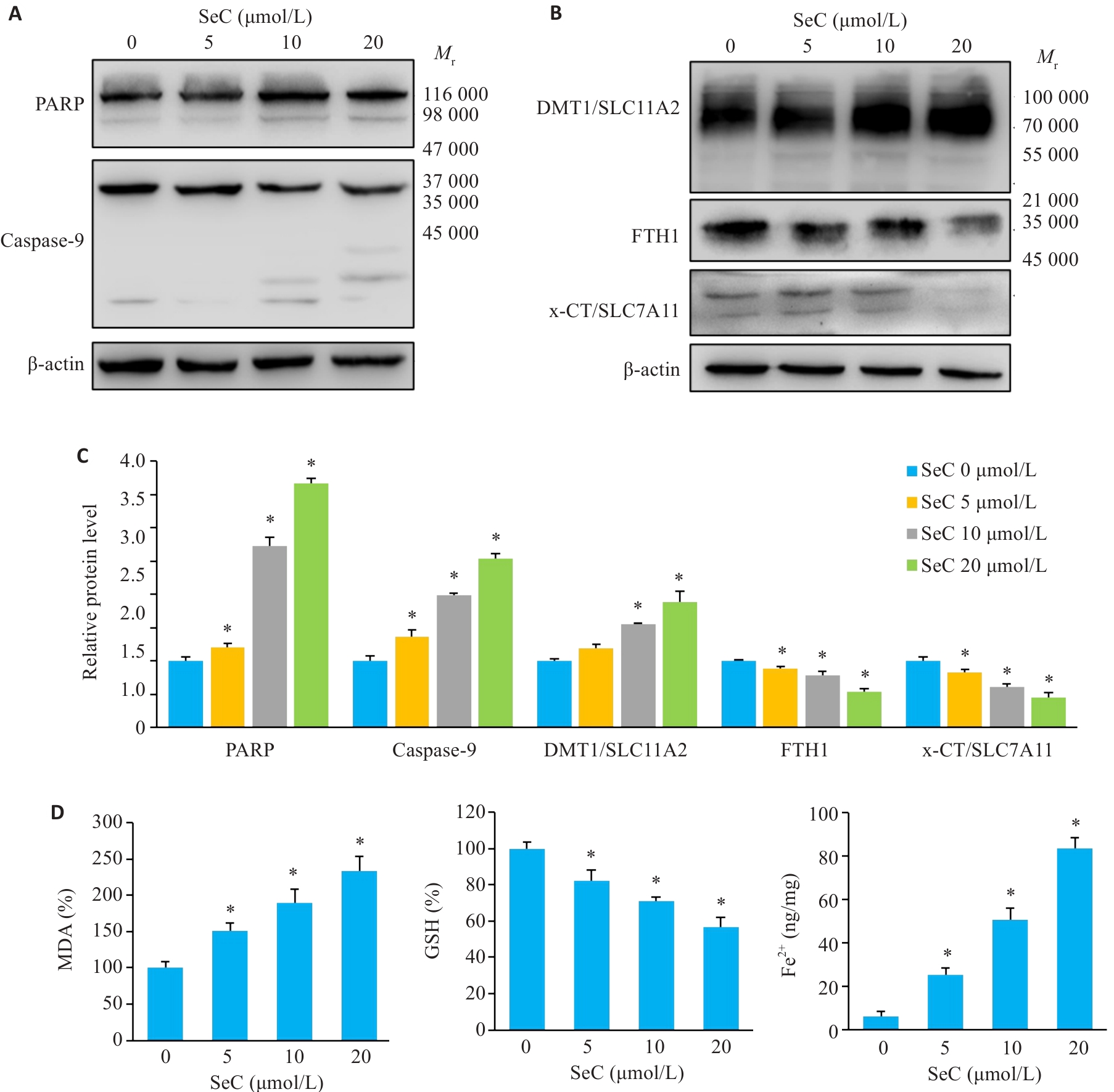
Fig.3 SeC induces apoptosis and ferroptosis in colon cancer cells. A: PARP cleavage and caspase-9 activation. B: Effect of SeC on ferroptosis-related proteins in RKO cells detected by Western blotting. C: Quantitative analysis of apoptotic and ferroptosis-related proteins. D: Measurement of MDA, GSH and Fe2+ in RKO cells after SeC treatment. All experiments were repeated three times (n=3). All data are presented as Mean±SD. *P<0.05 vs SeC 0 μmol/L.
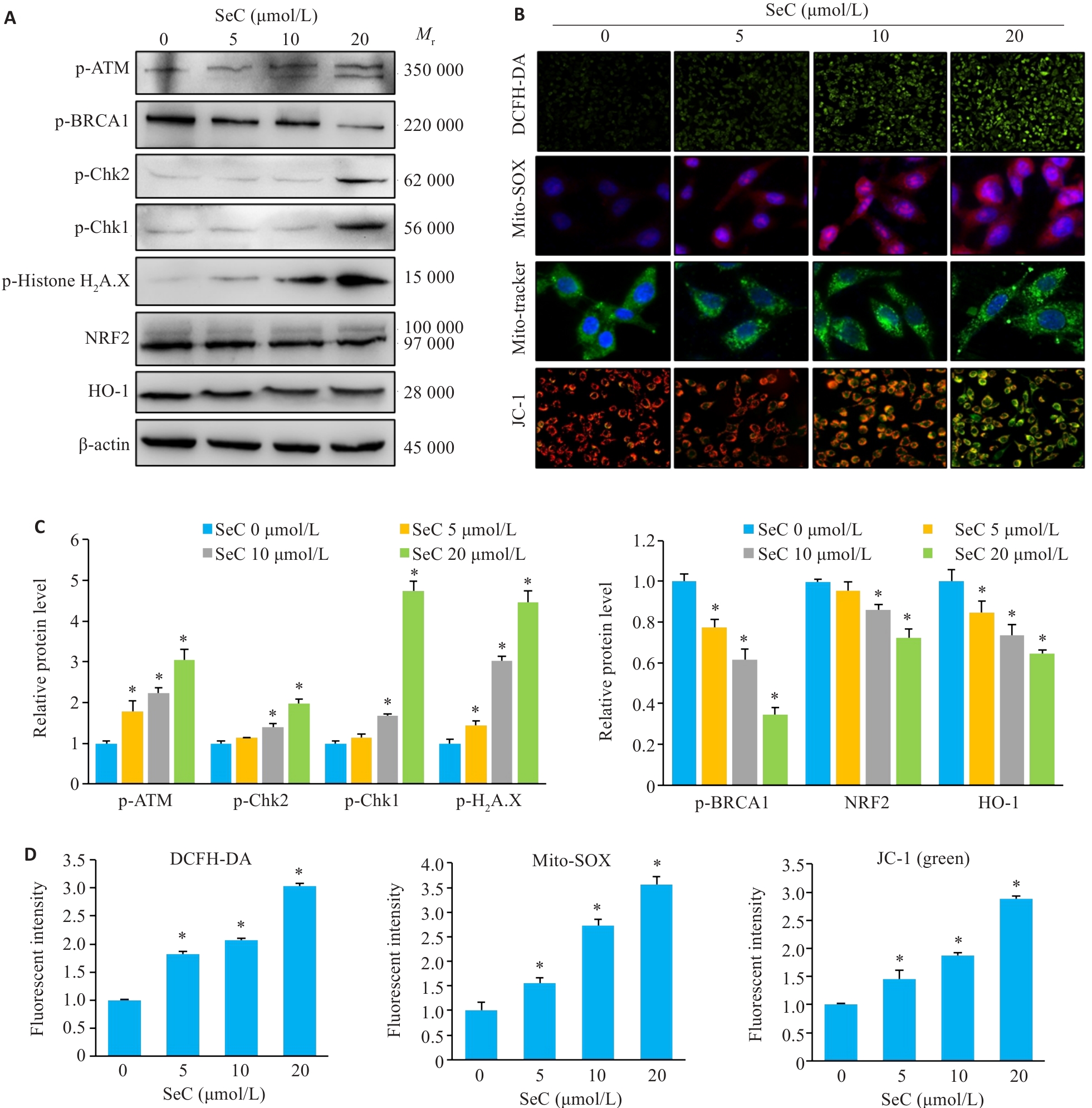
Fig.4 SeC induces mitochondrial dysfunction and oxidative stress damage in colon cancer cells. A: Effects of SeC on oxidative stress-related proteins in RKO cells detected by Western blotting. B: SeC induces ROS accumulation and mitochondrial dysfunction in RKO cells. DCFH-DA fluorescent probe was used to detect ROS production (×100). Mito-SOX fluorescent probe was used to detect mitochondrial superoxide production (×400). Mito-Tracker fluorescent probe was used to detect mitochondrial structural damage (×400). JC-1 fluorescent probe was used to detect mitochondrial membrane potential changes (×200). C: Quantitative analysis of oxidative stress-related protein and DNA damage-related protein results. D: Quantitative analysis of ROS, Mito-SOX and JC-1 fluorescence results. All experiments were repeated 3 times (n=3). All data are presented as Mean±SD. *P<0.05 vs SeC 0 μmol/L.
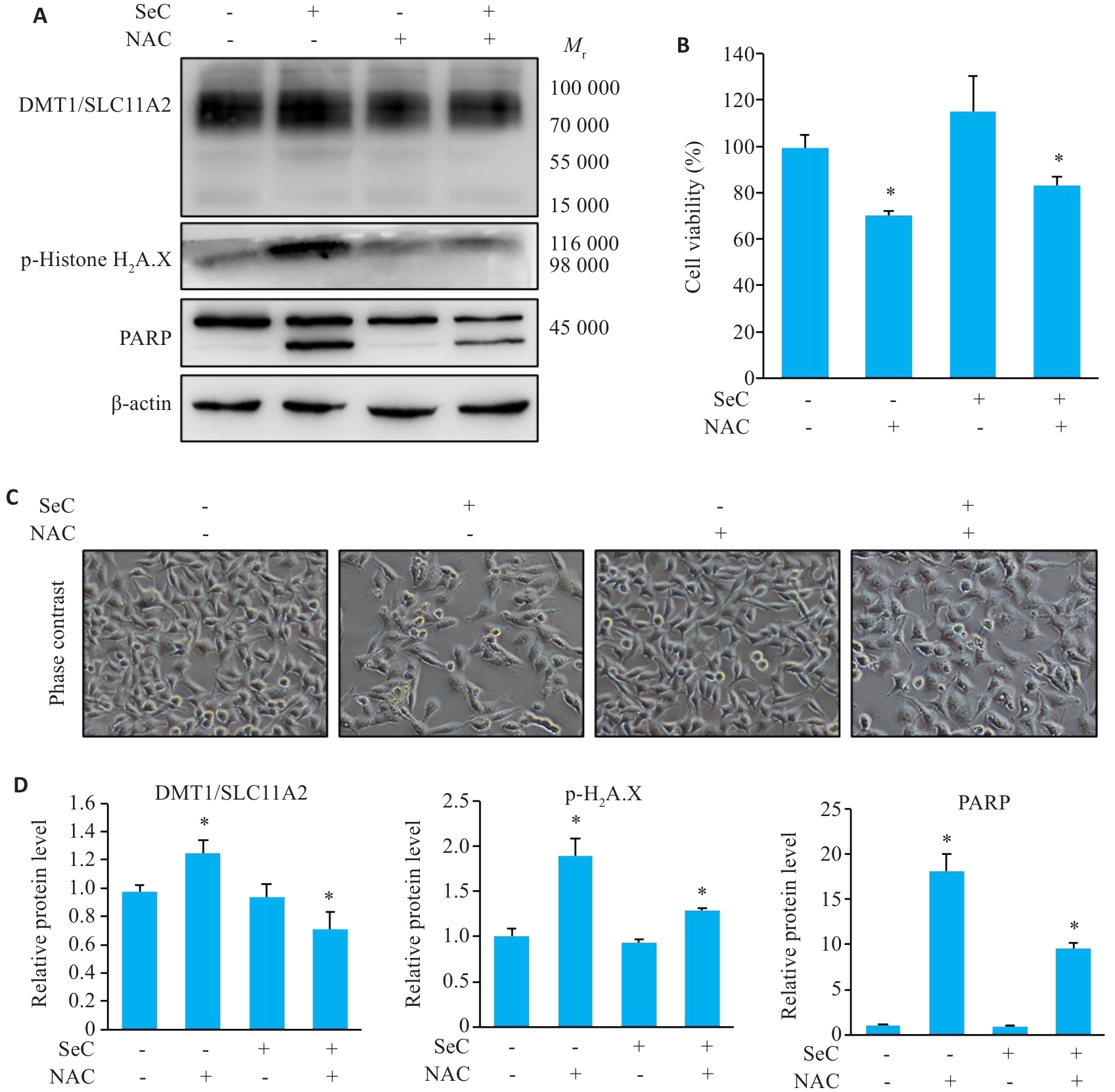
Fig.5 ROS elimination attenuates SeC-induced apoptosis and ferroptosis. A: NAC pretreatment inhibits SeC-induced DNA damage, apoptosis, and ferroptosis detected by Western blotting. B: NAC pretreatment reverses the effects of SeC on colon cancer cells. C: Changes in cell morphology (×200). D: Quantitative analysis of apoptosis-related proteins, ferroptosis-related proteins, and DNA damage-related proteins. All experiments were repeated 3 times (n=3). All data are presented as Mean±SD. *P<0.05 vs SeC 0 μmol/L.
| [1] | Malki A, ElRuz RA, Gupta I, et al. Molecular Mechanisms of Colon Cancer Progression and Metastasis: Recent Insights and Advancements[J]. Int J Mol Sci, 2020, 22(1): 130. doi:10.3390/ijms22010130 |
| [2] | Peters KM, Carlson BA, Gladyshev VN, et al. Selenoproteins in colon cancer[J]. Free Radic Biol Med, 2018, 127: 14-25. doi:10.1016/j.freeradbiomed.2018.05.075 |
| [3] | Audisio A, Fazio R, Dapra V, et al. Neoadjuvant chemotherapy for early-stage colon cancer[J]. Cancer Treat Rev, 2024, 123:102676. doi:10.1016/j.ctrv.2023.102676 |
| [4] | Benarba B, Pandiella A. Colorectal cancer and medicinal plants: Principle findings from recent studies[J]. Biomed Pharmacother, 2018, 107: 408-23. doi:10.1016/j.biopha.2018.08.006 |
| [5] | Aiello P, Sharghi M, Mansourkhani SM, et al. Medicinal Plants in the Prevention and Treatment of Colon Cancer[J]. Oxid Med Cell Longev, 2019, 2019: 2075614. doi:10.1155/2019/2075614 |
| [6] | Thiengsusuk A, Boonprasert K, Na-Bangchang K. A Systematic Review of Drug Metabolism Studies of Plants With Anticancer Properties: Approaches Applied and Limitations[J]. Eur J Drug Metab Pharmacokinet, 2020, 45(2): 173-225. doi:10.1007/s13318-019-00582-8 |
| [7] | Winther KH, Rayman MP, Bonnema SJ, et al. Selenium in thyroid disorders-essential knowledge for clinicians[J]. Nat Rev Endocrinol, 2020, 16(3): 165-76. doi:10.1038/s41574-019-0311-6 |
| [8] | Wichman J, Winther KH, Bonnema SJ, et al. Selenium Supplementation Significantly Reduces Thyroid Autoantibody Levels in Patients with Chronic Autoimmune Thyroiditis: A Systematic Review and Meta-Analysis[J]. Thyroid, 2016, 26(12):1681-92. doi:10.1089/thy.2016.0256 |
| [9] | Mojadadi A, Au A, Salah W, et al. Role for Selenium in Metabolic Homeostasis and Human Reproduction[J]. Nutrients, 2021, 13(9):3256. doi:10.3390/nu13093256 |
| [10] | Hsu WL, Wang CM, Yao CL, et al. Blockage of Nrf2 and autophagy by L-selenocystine induces selective death in Nrf2-addicted colorectal cancer cells through p62-Keap-1-Nrf2 axis[J]. Cell Death Dis, 2022, 13(12): 1060. doi:10.1038/s41419-022-05512-2 |
| [11] | Long M, Wu J, Hao J, et al. Selenocystine-induced cell apoptosis and S-phase arrest inhibit human triple-negative breast cancer cell proliferation[J]. In Vitro Cell Dev Biol Anim, 2015, 51(10):1077-84. doi:10.1007/s11626-015-9937-4 |
| [12] | Bartolini D, Sancineto L, Fabro De Bem A, et al. Selenocompounds in Cancer Therapy: An Overview[J]. Adv Cancer Res, 2017, 136:259-302. doi:10.1016/bs.acr.2017.07.007 |
| [13] | Wahyuni EA, Yii CY, Liang HL, et al. Selenocystine induces oxidative-mediated DNA damage via impairing homologous recombination repair of DNA double-strand breaks in human hepatoma cells[J]. Chem Biol Interact, 2022, 365: 110046. doi:10.1016/j.cbi.2022.110046 |
| [14] | Chen T, Wong YS. Selenocystine induces reactive oxygen species-mediated apoptosis in human cancer cells[J]. Biomed Pharmacother, 2009, 63(2): 105-13. doi:10.1016/j.biopha.2008.03.009 |
| [15] | LeFort KR, Rungratanawanich W, Song BJ. Contributing roles of mitochondrial dysfunction and hepatocyte apoptosis in liver diseases through oxidative stress, post-translational modifications, inflamm-ation, and intestinal barrier dysfunction[J]. Cell Mol Life Sci, 2024, 81(1): 34. doi:10.1007/s00018-023-05061-7 |
| [16] | Yu W, Liu T, Yuan Z, et al. Polymer-engineered liposome-delivered Ebselen against tumor through GSH/H2O2-responsive disruption of redox homeostasis and direct p53 activation[J]. Biomaterials, 2026, 326: 123698. doi:10.1016/j.biomaterials.2025.123698 |
| [17] | Dai C, He Y, Lu H, et al. Nanoquadruplex-driven hydrogen therapy: NIR-controlled release for targeted cancer ferroptosis[J]. Biomaterials, 2026, 326: 123635. doi:10.1016/j.biomaterials.2025.123635 |
| [18] | Riedl SJ, Shi Y. Molecular mechanisms of caspase regulation during apoptosis[J]. Nat Rev Mol Cell Biol, 2004, 5(11): 897-907. doi:10.1038/nrm1496 |
| [19] | Bock FJ, Tait SWG. Mitochondria as multifaceted regulators of cell death[J]. Nat Rev Mol Cell Biol, 2020, 21(2): 85-100. doi:10.1038/s41580-019-0173-8 |
| [20] | Bou-Teen D, Kaludercic N, Weissman D, et al. Mitochondrial ROS and mitochondria-targeted antioxidants in the aged heart[J]. Free Radic Biol Med, 2021, 167: 109-24. doi:10.1016/j.freeradbiomed.2021.02.043 |
| [21] | Sun CY, Cong KP, Zhao DD, et al. 8-Nitrotryptanthrin inhibits colorectal cancer progression via TGF-beta/SMAD and PI3K/AKT/mTOR pathways[J]. Front Pharmacol, 2025, 16: 1576673. doi:10.3389/fphar.2025.1576673 |
| [22] | Wang R, Ha KY, Dhandapani S, et al. Biologically synthesized black ginger-selenium nanoparticle induces apoptosis and autophagy of AGS gastric cancer cells by suppressing the PI3K/Akt/mTOR signaling pathway[J]. J Nanobiotechnology, 2022, 20(1): 441. doi:10.1186/s12951-022-01576-6 |
| [23] | Wang M, Xu H, Xiong X, et al. Antiproliferative activity of selenium-enriched coumarin derivatives on the SK-N-SH neuroblastoma cell line: Mechanistic insights[J]. Eur J Med Chem, 2025, 286: 117322. doi:10.1016/j.ejmech.2025.117322 |
| [24] | Dixon SJ, Lemberg KM, Lamprecht MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death[J]. Cell, 2012, 149(5):1060-72. doi:10.1016/j.cell.2012.03.042 |
| [25] | Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease[J]. Nat Rev Mol Cell Biol, 2021, 22(4):266-82. doi:10.1038/s41580-020-00324-8 |
| [26] | Chen J, Wang Y, Wu J, et al. The Potential Value of Targeting Ferroptosis in Early Brain Injury After Acute CNS Disease[J]. Front Mol Neurosci, 2020, 13: 110. doi:10.3389/fnmol.2020.00110 |
| [27] | Sun Y, Li Q, Guo H, et al. Ferroptosis and Iron Metabolism after Intracerebral Hemorrhage[J]. Cells, 2022, 12(1): 90. doi:10.3390/cells12010090 |
| [28] | Choi JA, Lee EH, Cho H, et al. High-Dose Selenium Induces Ferroptotic Cell Death in Ovarian Cancer[J]. Int J Mol Sci, 2023, 24(3): 1918. doi:10.3390/ijms24031918 |
| [29] | Chen Y, Liu X, Chen B, et al. Selenium nanoparticles decorated by fucoidan induce ferroptosis in HepG2 cells[J]. Int J Biol Macromol, 2025, 289: 138841. doi:10.1016/j.ijbiomac.2024.138841 |
| [30] | Wang B, Wang Y, Zhang J, et al. ROS-induced lipid peroxidation modulates cell death outcome: mechanisms behind apoptosis, autophagy, and ferroptosis[J]. Arch Toxicol, 2023, 97(6): 1439-51. doi:10.1007/s00204-023-03476-6 |
| [31] | Tang Q, Chen H, Mai Z, et al. Bim- and Bax-mediated mitochondrial pathway dominates abivertinib-induced apoptosis and ferroptosis[J]. Free Radic Biol Med, 2022, 180: 198-209. doi:10.1016/j.freeradbiomed.2022.01.013 |
| [32] | Qiao R, Yuan Z, Yang M, et al. Selenium-Doped Nanohetero-junctions for Highly Efficient Cancer Radiosensitization[J]. Adv Sci (Weinh), 2024, 11(29): e2402039. doi:10.1002/advs.202402039 |
| [33] | Ren M, Liang S, Lin S, et al. Design, synthesis and biological evaluation of artesunate-Se derivatives as anticancer agents by inducing GPX4-mediated ferroptosis[J]. Bioorg Chem, 2024, 152:107733. doi:10.1016/j.bioorg.2024.107733 |
| [34] | Chen QM. Nrf2 for protection against oxidant generation and mitochondrial damage in cardiac injury[J]. Free Radic Biol Med, 2022, 179: 133-43. doi:10.1016/j.freeradbiomed.2021.12.001 |
| [35] | Qin Q, Zhang H, Lai M, et al. Sulfasalazine induces ferroptosis in osteosarcomas by regulating Nrf2/SLC7A11/GPX4 signaling axis[J]. Sci Rep, 2025, 15(1): 30197. doi:10.1038/s41598-025-13324-5 |
| [1] | Yuce PENG, Yi JIANG, Dan MA, An HE, Dingyi LÜ, Minghao LUO, Suxin LUO. Isovitexin alleviates myocardial oxidative stress injury in diabetic mice by enhancing myocardial SIRT3 expression and reducing oxidative stress [J]. Journal of Southern Medical University, 2026, 46(2): 316-324. |
| [2] | Lin YIN, Keni ZHANG, Tong QIAO, Minzhu NIU, Lixia YIN, Xinyue LIU, Zhijun GENG, Jing LI, Jianguo HU. Veratric acid relieves oxidative stress and DSS-induced colitis in mice by activating the Nrf2/HO-1 signaling pathway [J]. Journal of Southern Medical University, 2026, 46(2): 403-411. |
| [3] | Tong QIAO, Lin YIN, Keni ZHANG, Minzhu NIU, Ju HUANG, Zhijun Geng, Jing LI, Jianguo HU. Poricoic acid A alleviates dextran sulfate sodium-induced colitis in mice by regulating AMPK/mTOR-mediated autophagy and inhibiting intestinal epithelial cell apoptosis [J]. Journal of Southern Medical University, 2026, 46(1): 131-140. |
| [4] | Shufen ZHANG, Tianrong HUANG, Canhong YANG, Jiayi CHEN, Tianming LÜ, Jiafa ZHANG. Sulforaphane reduces reactive astrocyte-mediated neuron apoptosis in vitro by inhibiting the MAPK/NF-κB signaling pathway in Aβ42 oligomer-activated astrocytes [J]. Journal of Southern Medical University, 2026, 46(1): 191-199. |
| [5] | Jinyan ZHAO, Jiao PENG, Minghe LIN, Xiaoqin ZHU, Bin HUANG, Jiumao LIN. Qingjie Fuzheng Granules alleviates 5-fluorouracil-induced skeletal muscle injury in tumor-bearing mice by inhibiting mitochondria-dependent apoptosis and activating the AMPK-PGC-1α pathway [J]. Journal of Southern Medical University, 2026, 46(1): 94-103. |
| [6] | Rongmao HE, Zeyang FANG, Yunyun ZHANG, Youliang WU, Shixiu LIANG, Tao JI, Kequan CHEN, Siqi WANG. Diagnostic and predictive value of ferroptosis-related genes in patients with ulcerative colitis [J]. Journal of Southern Medical University, 2025, 45(9): 1927-1937. |
| [7] | Qi YUN, Ruoli DU, Yuying HE, Yixin ZHANG, Jiahui WANG, Hongwei YE, Zhenghong LI, Qin GAO. Cinnamic acid ameliorates doxorubicin-induced myocardial injury in mice by attenuating cardiomyocyte ferroptosis via inhibiting TLR4 [J]. Journal of Southern Medical University, 2025, 45(9): 1946-1958. |
| [8] | Zejin OU, Ying LI, Shi CHEN, Ziyi WANG, Meiyi HE, Zhicheng CHEN, Shihao TANG, Xiaojing MENG, Zhi WANG. Inhibition of ferroptosis alleviates acute kidney injury caused by diquat in zebrafish [J]. Journal of Southern Medical University, 2025, 45(8): 1743-1750. |
| [9] | Junyi LI, Siyuan CHEN, Liyao XIE, Jin WANG, Ao CHENG, Shaowei ZHANG, Jiyu LIN, Zhihan FANG, Yirui PAN, Chonghe CUI, Gengxin CHEN, Chao ZHANG, Li LI. β-sitosterol, an important component in the fruits of Alpinia oxyphylla Miq., prolongs lifespan of Caenorhabditis elegans by suppressing the ferroptosis pathway [J]. Journal of Southern Medical University, 2025, 45(8): 1751-1757. |
| [10] | Xiaoyu CHANG, Hanwen ZHANG, Hongting CAO, Ling HOU, Xin MENG, Hong TAO, Yan LUO, Guanghua LI. Heat stress affects expression levels of circadian clock gene Bmal1 and cyclins in rat thoracic aortic endothelial cells [J]. Journal of Southern Medical University, 2025, 45(7): 1353-1362. |
| [11] | Xinyuan CHEN, Chengting WU, Ruidi LI, Xueqin PAN, Yaodan ZHANG, Junyu TAO, Caizhi LIN. Shuangshu Decoction inhibits growth of gastric cancer cell xenografts by promoting cell ferroptosis via the P53/SLC7A11/GPX4 axis [J]. Journal of Southern Medical University, 2025, 45(7): 1363-1371. |
| [12] | Jingxian WANG, Zijing REN, Peiyang ZHOU. S1PR5 activation or overexpression enhances barrier function of mouse brain microvascular endothelial cells against OGD/R injury by modulating oxidative stress [J]. Journal of Southern Medical University, 2025, 45(7): 1451-1459. |
| [13] | Mengying ZHANG, Chenling ZHAO, Liwei TIAN, Guofang YU, Wenming YANG, Ting DONG. Gandou Fumu Decoction improves liver steatosis by inhibiting hepatocyte ferroptosis in mice with Wilson's disease through the GPX4/ACSL4/ALOX15 signaling pathway [J]. Journal of Southern Medical University, 2025, 45(7): 1471-1478. |
| [14] | Jiahao LI, Ruiting XIAN, Rong LI. Down-regulation of ACADM-mediated lipotoxicity inhibits invasion and metastasis of estrogen receptor-positive breast cancer cells [J]. Journal of Southern Medical University, 2025, 45(6): 1163-1173. |
| [15] | Yujia YANG, Lifang YANG, Yaling WU, Zhaoda DUAN, Chunze YU, Chunyun WU, Jianyun YU, Li YANG. Cannabidiol inhibits neuronal endoplasmic reticulum stress and apoptosis in rats with multiple concussions by regulating the PERK-eIF2α-ATF4-CHOP pathway [J]. Journal of Southern Medical University, 2025, 45(6): 1240-1250. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||