南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 629-637.doi: 10.12122/j.issn.1673-4254.2026.03.16
• 基础研究 • 上一篇
王泽安1( ), 刘丽丽2, 施美2, 侯勇2, 董莉莉3, 张国梁2(
), 刘丽丽2, 施美2, 侯勇2, 董莉莉3, 张国梁2( )
)
收稿日期:2025-08-05
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
张国梁
E-mail:zf_wangzean@126.com;zhangguoliang61@sina.com
作者简介:王泽安,在读硕士研究生,E-mail: zf_wangzean@126.com
基金资助:
Ze'an WANG1( ), Lili LIU2, Mei SHI2, Yong HOU2, Lili DONG3, Guoliang ZHANG2(
), Lili LIU2, Mei SHI2, Yong HOU2, Lili DONG3, Guoliang ZHANG2( )
)
Received:2025-08-05
Online:2026-03-20
Published:2026-03-26
Contact:
Guoliang ZHANG
E-mail:zf_wangzean@126.com;zhangguoliang61@sina.com
摘要:
目的 探究益气解毒方对Hep3B肝癌小鼠模型中免疫微环境的调控机制。 方法 选取40只SPF级雄性小鼠,小鼠腋下注射Hep3B肿瘤细胞,建立异位小鼠模型,将小鼠随机分为模型组(生理盐水0.2 g/kg),华蟾素片组(0.22 g/kg),益气解毒方低(0.5 g/kg)、中(1 g/kg)、高(2 g/kg)剂量组,8只/组。连续灌胃给药28 d,每7 d记录各组小鼠瘤重、体积。给药结束后计算抑瘤率,通过HE染色观察小鼠肿瘤组织病理变化,流式细胞术检测肿瘤组织中M1/M2型巨噬细胞极化比例,免疫荧光术检测CD86、CD206蛋白表达,Western blotting检测小鼠瘤体组织中iNOS、Arg-1、p65、p50蛋白表达,q-PCR检测iNOS、Arg-1、p65、p50 mRNA表达,ELISA法检测血清IL-4、IL-6、IL-10、INF-γ、TNF-α、TGF-β1水平。 结果 与模型组比较,益气解毒方各组小鼠肿瘤质量、体积减小;肿瘤细胞数量减少,细胞核缩小,坏死区域增大;M1型巨噬细胞比例升高,M2型巨噬细胞比例降低;肿瘤组织中CD86表达上升,CD206表达下降;iNOS、Arg-1、p65、p50蛋白及mRNA表达降低;血清IL-4、IL-10、TNF-α、TGF-β1水平降低,IL-6、INF-γ水平升高(P<0.05)。 结论 益气解毒方能有效抑制Hep3B肝癌荷瘤小鼠肿瘤生长,逆转肿瘤相关巨噬细胞的极化状态,将M1/M2比例从M2主导调整至M1主导,从而改善肿瘤免疫抑制微环境,增强抗肿瘤效应,其作用机制可能与抑制NF-κB信号通路的过度激活有关。
王泽安, 刘丽丽, 施美, 侯勇, 董莉莉, 张国梁. 益气解毒方通过抑制NF-κB通路的过度激活改善小鼠肝癌免疫微环境[J]. 南方医科大学学报, 2026, 46(3): 629-637.
Ze'an WANG, Lili LIU, Mei SHI, Yong HOU, Lili DONG, Guoliang ZHANG. Yiqi Jiedu Formula regulates immune microenvironment of liver cancer in mice by inhibiting overactivation of NF-κB signaling pathway[J]. Journal of Southern Medical University, 2026, 46(3): 629-637.
| Primer | Sequence | Primer length (bp) | Product size (bp) |
|---|---|---|---|
mouse-β-actin F mouse-β-actin R mouse-iNOS F mouse-iNOS R mouse-Arg-1 F mouse-Arg-1 R mouse-P65 F mouse-P65 R mouse-p50 F mouse-p50 R | CAGCCTTCCTTCTTGGGTATG GGCATAGAGGTCTTTACGGATG GGAGTGACGGCAAACATGACT TCGATGCACAACTGGGTGAAC TGTCCCTAATGACAGCTCCTT GCATCCACCCAAATGACACAT ACTGCCGGGATGGCTACTAT TCTGGATTCGCTGGCTAATGG ATGGCAGACGATGATCCCTAC TGTTGACAGTGGTATTTCTGGTG | 21 22 21 21 21 21 20 21 21 23 | 99 127 204 126 111 |
表1 各基因引物序列
Tab.1 Primer sequences for q-PCR
| Primer | Sequence | Primer length (bp) | Product size (bp) |
|---|---|---|---|
mouse-β-actin F mouse-β-actin R mouse-iNOS F mouse-iNOS R mouse-Arg-1 F mouse-Arg-1 R mouse-P65 F mouse-P65 R mouse-p50 F mouse-p50 R | CAGCCTTCCTTCTTGGGTATG GGCATAGAGGTCTTTACGGATG GGAGTGACGGCAAACATGACT TCGATGCACAACTGGGTGAAC TGTCCCTAATGACAGCTCCTT GCATCCACCCAAATGACACAT ACTGCCGGGATGGCTACTAT TCTGGATTCGCTGGCTAATGG ATGGCAGACGATGATCCCTAC TGTTGACAGTGGTATTTCTGGTG | 21 22 21 21 21 21 20 21 21 23 | 99 127 204 126 111 |
| Group | Tumor weight (g) | P | Tumor volume (mm3) | P | Tumor inhibition rate (%) |
|---|---|---|---|---|---|
| Model | 0.63±0.18 | - | 682.901±214.39 | - | - |
| Low dose group of Yiqi Jiedu Formula | 0.41±0.19 | 0.028 | 410.06±184.38 | 0.018 | 40.95 |
| Medium dose group of Yiqi Jiedu Formula | 0.35±0.15 | 0.020 | 375.16±128.08 | 0.009 | 43.99 |
| High dose group of Yiqi Jiedu Formula | 0.33±0.13 | 0.002 | 200.66±120.57 | 0.00 | 60.13 |
| Cinobufagin Tablets | 0.24±0.09 | 0.002 | 174.00±63.65 | 0.00 | 61.34 |
表2 裸鼠体质量、瘤质量、肿瘤体积与抑瘤率
Tab.2 Tumor weight, tumor volume, and tumor inhibition rate in the nude mice (Mean±SD, n=5)
| Group | Tumor weight (g) | P | Tumor volume (mm3) | P | Tumor inhibition rate (%) |
|---|---|---|---|---|---|
| Model | 0.63±0.18 | - | 682.901±214.39 | - | - |
| Low dose group of Yiqi Jiedu Formula | 0.41±0.19 | 0.028 | 410.06±184.38 | 0.018 | 40.95 |
| Medium dose group of Yiqi Jiedu Formula | 0.35±0.15 | 0.020 | 375.16±128.08 | 0.009 | 43.99 |
| High dose group of Yiqi Jiedu Formula | 0.33±0.13 | 0.002 | 200.66±120.57 | 0.00 | 60.13 |
| Cinobufagin Tablets | 0.24±0.09 | 0.002 | 174.00±63.65 | 0.00 | 61.34 |
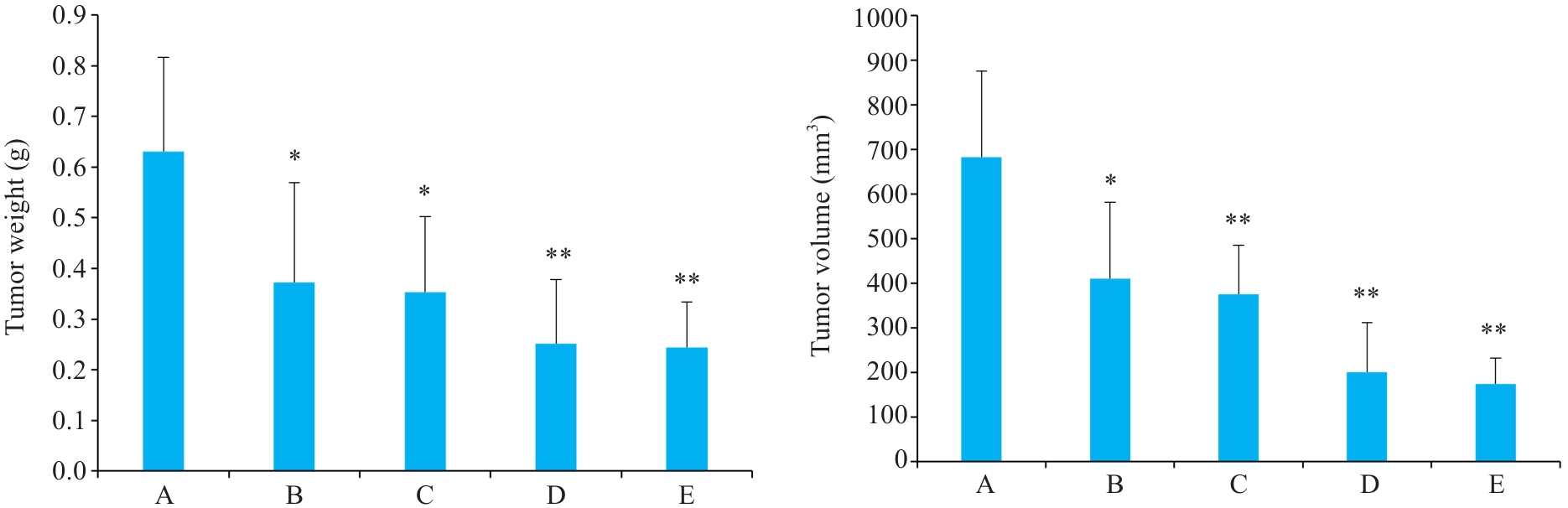
图1 益气解毒方对各组Hep3B肝癌小鼠肿瘤质量、体积的影响
Fig.1 Effects of Yiqi Jiedu Formula on tumor weight and volume in Hep3B hepatoma-bearing mice. A: Model group. B: Low-dose Yiqi Jiedu Formula group. C: Medium-dose Yiqi Jiedu Formula group. D: High-dose Yiqi Jiedu Formula group. E: Cinobufagin Tablets. *P<0.05, **P<0.01 vs model group (n=5).
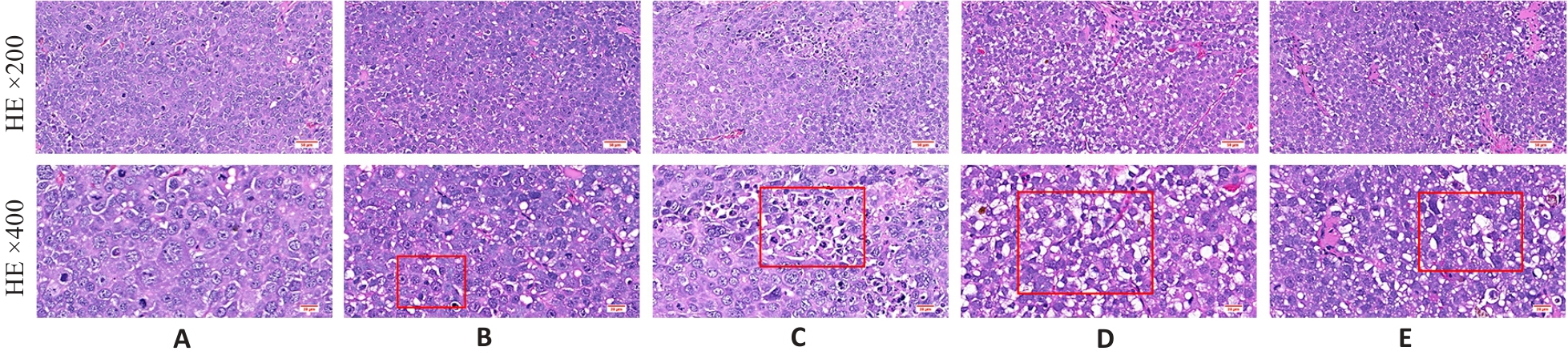
图2 益气解毒方对各组Hep3B肝癌小鼠肿瘤组织病理的影响
Fig.2 Effect of Yiqi Jiedu Formula on histopathology of tumor tissues in Hep3B hepatoma-bearing mice. A: Model group. B: Low-dose Yiqi Jiedu Formula group. C: Medium-dose Yiqi Jiedu Formula group. D: High-dose Yiqi Jiedu Formula group. E: Cinobufagin Tablets.
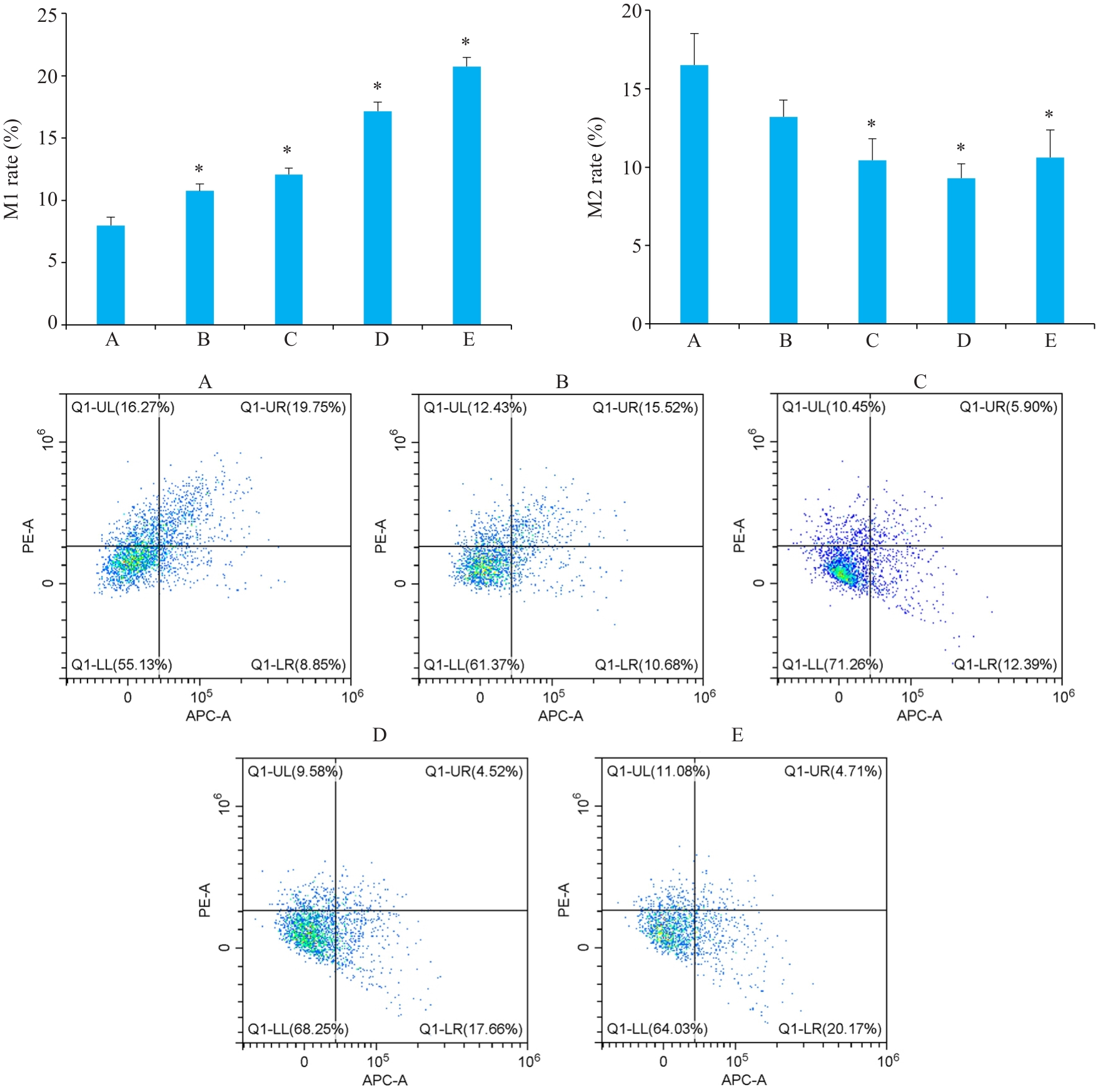
图3 流式检测各组Hep3B肝癌小鼠中巨噬细胞极化水平
Fig.3 Flow cytometric analysis of macrophage polarization levels in the tumor tissues of Hep3B hepatoma-bearing mice. A: Model group. B: Low-dose Yiqi Jiedu Formula group. C: Medium-dose Yiqi Jiedu Formula group. D: High-dose Yiqi Jiedu Formula group. E: Cinobufagin Tablets. *P<0.01 vs model group.
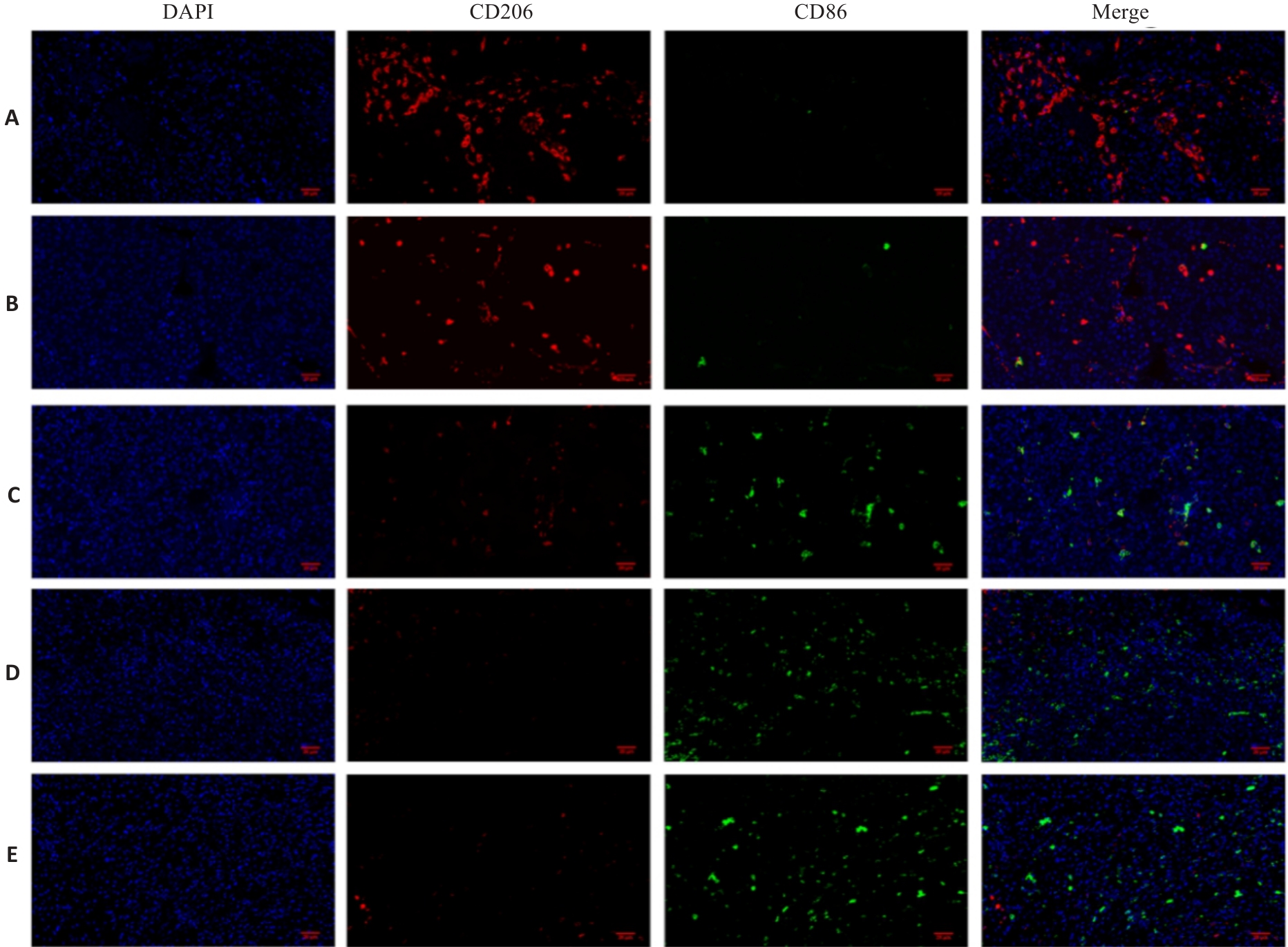
图4 益气解毒方对各组Hep3B肝癌小鼠组织免疫荧光染色观察
Fig.4 Effect of Yiqi Jiedu Formula on tumor tissue assessed by immunofluorescence staining in Hep3B hepatoma-bearing mice (Original magnification: ×400). A: Model group. B: Low-dose Yiqi Jiedu Formula group. C: Medium-dose Yiqi Jiedu Formula group. D: High-dose Yiqi Jiedu Formula group. E: Cinobufagin Tablets.
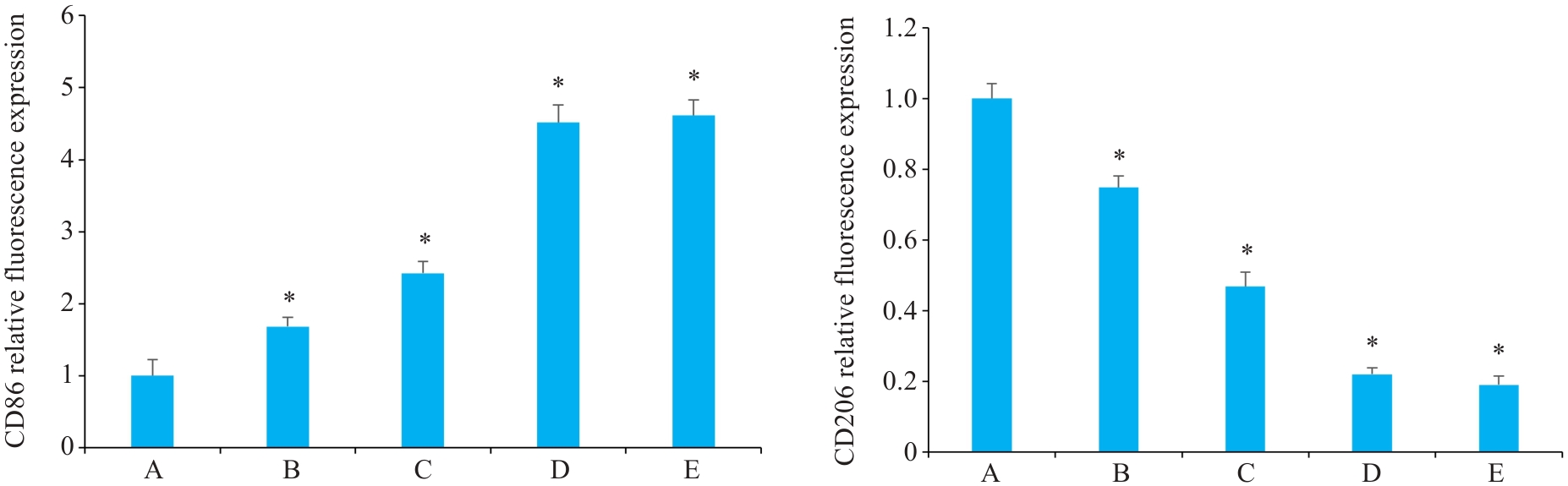
图5 益气解毒方对各组Hep3B肝癌小鼠组织免疫荧光相对表达
Fig.5 Effect of Yiqi Jiedu Formula on immunofluorescence intensity of CD 86 and CD206 in the tumor tissues of Hep3B hepatoma-bearing mice. A: Model group. B: Low-dose Yiqi Jiedu Formula group. C: Medium-dose Yiqi Jiedu Formula group. D: High-dose Yiqi Jiedu Formula group. E: Cinobufagin Tablets. *P<0.01 vs model group (n=5).
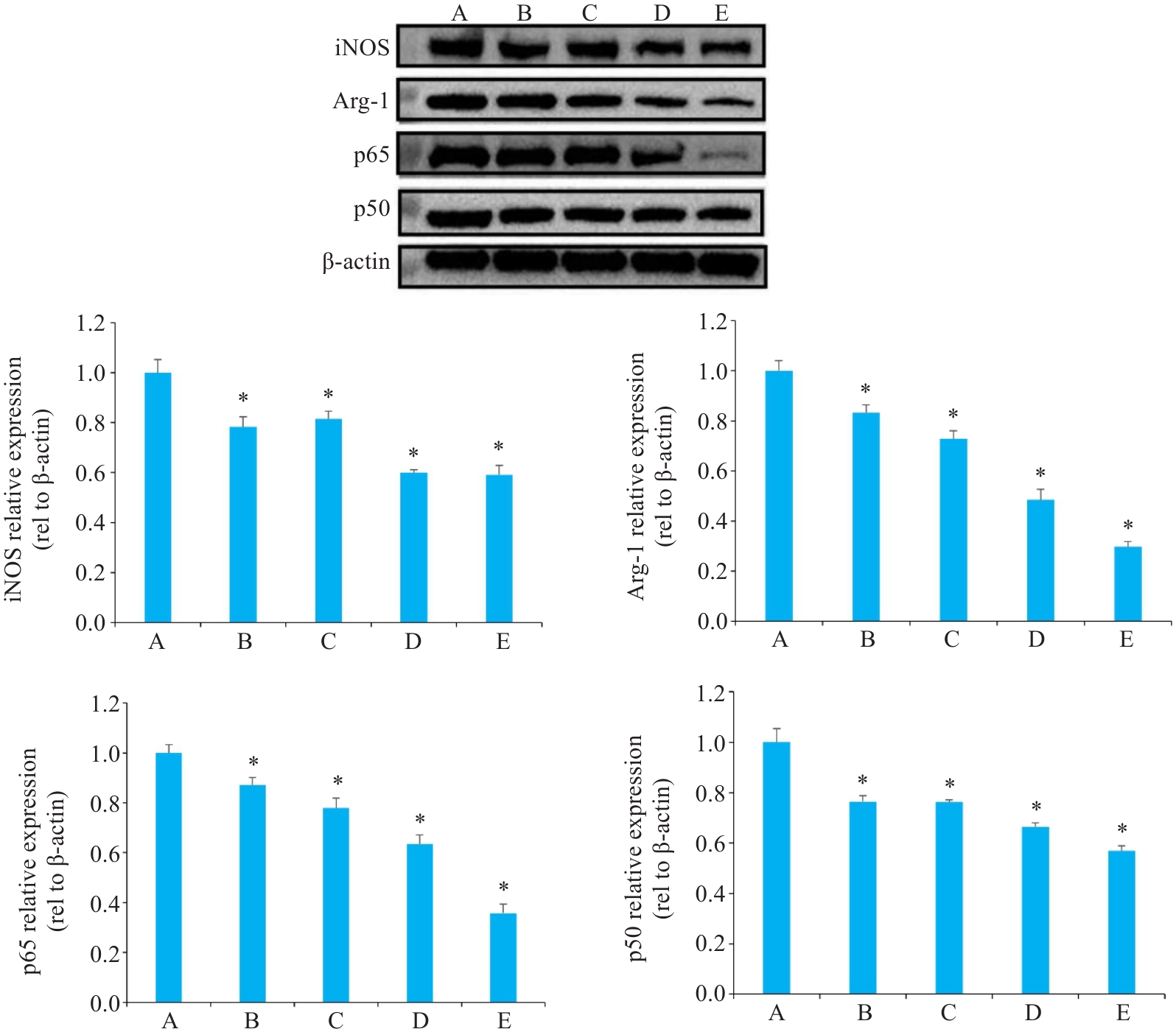
图6 各组Hep3B肝癌小鼠组织中目标蛋白免疫印迹分析及相对蛋白表达量
Fig.6 Western blotting of the target proteins and their relative expression levels in tumor tissues from Hep3B hepatoma-bearing mice. A: Model group. B: Low-dose Yiqi Jiedu Formula group. C: Medium-dose Yiqi Jiedu Formula group. D: High-dose Yiqi Jiedu Formula group. E: Cinobufagin Tablets. *P<0.01 vs model group.
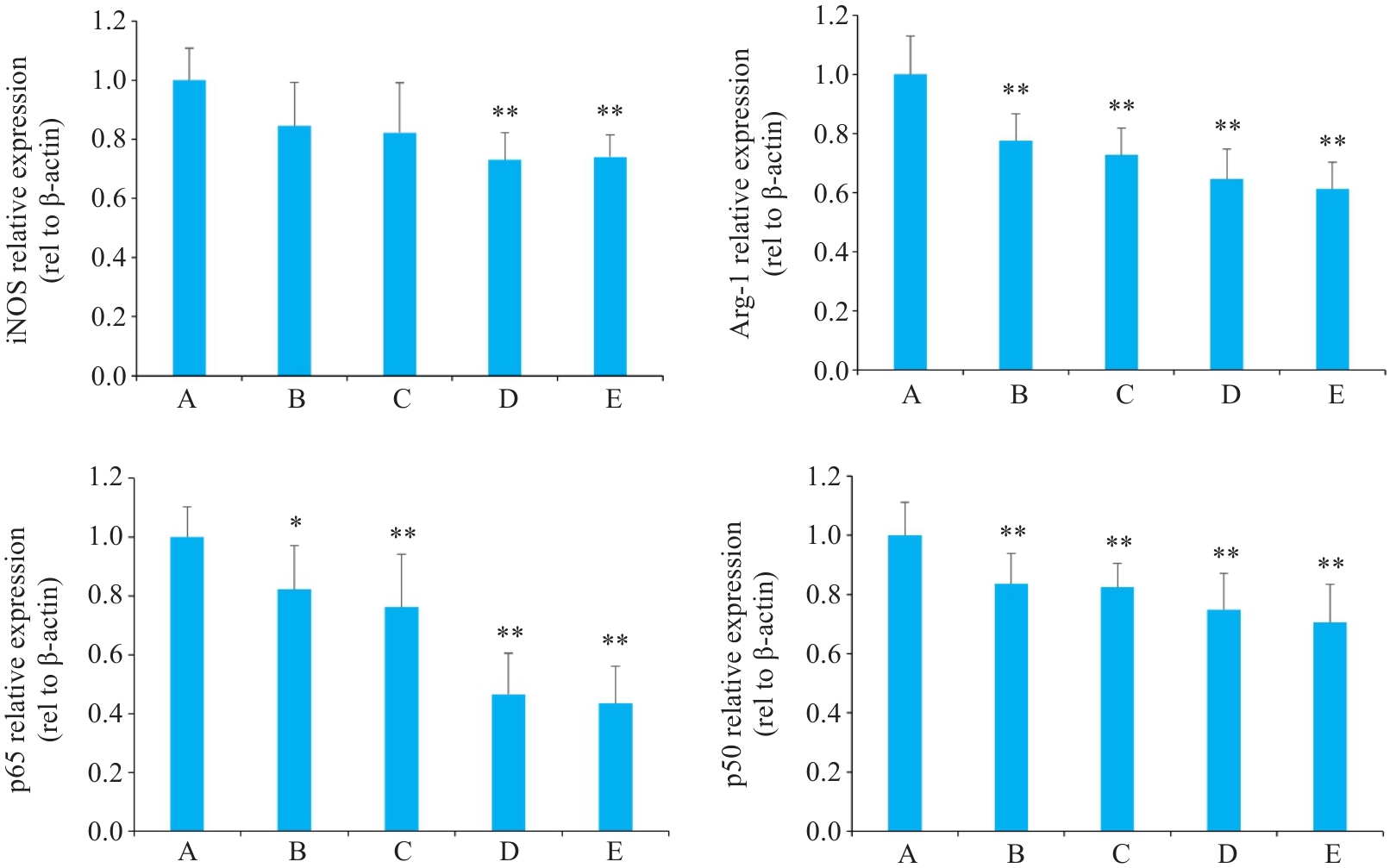
图7 各组Hep3B肝癌小鼠组织中相关mRNA的影响
Fig.7 Effect of Yiqi Jiedu Formula on mRNA expression levels in tumor tissues of Hep3B hepatoma-bearing mice. A: Model group. B: Low-dose Yiqi Jiedu Formula group. C: Medium-dose Yiqi Jiedu Formula group. D: High-dose Yiqi Jiedu Formula group. E: Cinobufagin Tablets. *P<0.05, **P<0.01 vs model group.
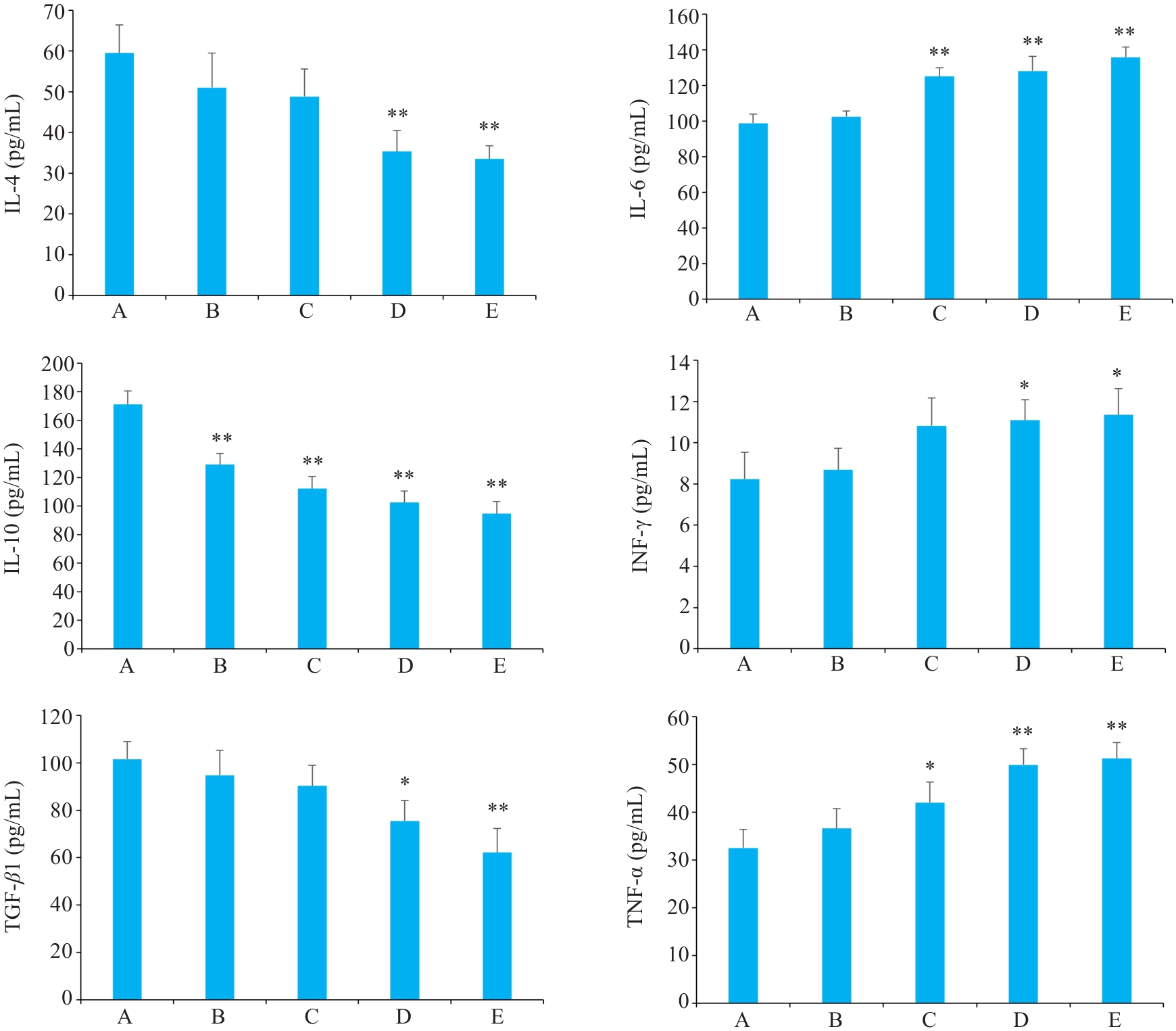
图8 各组Hep3B肝癌小鼠血清IL-4, IL-6, IL-10, INF-γ, TNF-α, TGF-β1水平的影响
Fig.8 Effect of Yiqi Jiedu Formula on serum levels of IL-4, IL-6, IFN-γ, TNF-α, and TGF-β1 in Hep3B hepatoma-bearing mice. A: Model group. B: Low-dose Yiqi Jiedu Formula group. C: Medium-dose Yiqi Jiedu Formula group. D: High-dose Yiqi Jiedu Formula group. E: Cinobufagin Tablets. *P<0.05, **P<0.01 vs model group.
| [1] | Feng M, Pan YS, Kong RR, et al. Therapy of primary liver cancer[J]. Innov, 2020, 1(2): 100032. doi:10.1016/j.xinn.2020.100032 |
| [2] | 中华医学会肝病学分会. 原发性肝癌二级预防共识(2021年版)[J]. 中华肝脏病杂志, 2021, 29(3): 216-26. doi:10.3969/j.issn.1001-5256.2021.03.008 |
| [3] | 陈杨柳, 李 媛, 官志杰. 中医药治疗原发性肝癌的研究进展[J].基层中医药, 2024, 3(12): 112-6. |
| [4] | Yang YM, Kim SY, Seki E. Inflammation and liver cancer: molecular mechanisms and therapeutic targets[J]. Semin Liver Dis, 2019, 39(1): 26-42. doi:10.1055/s-0038-1676806 |
| [5] | Nault JC, Ningarhari M, Rebouissou S, et al. The role of telomeres and telomerase in cirrhosis and liver cancer[J]. Nat Rev Gastroenterol Hepatol, 2019, 16(9): 544-58. doi:10.1038/s41575-019-0165-3 |
| [6] | Boutilier AJ, Elsawa SF. Macrophage polarization states in the tumor microenvironment[J]. Int J Mol Sci, 2021, 22(13): 6995. doi:10.3390/ijms22136995 |
| [7] | Wang C, Ma C, Gong LH, et al. Macrophage polarization and its role in liver disease[J]. Front Immunol, 2021, 12: 803037. doi:10.3389/fimmu.2021.803037 |
| [8] | Mantovani A, Marchesi F, Malesci A, et al. Tumour-associated macrophages as treatment targets in oncology[J]. Nat Rev Clin Oncol, 2017, 14(7): 399-416. doi:10.1038/nrclinonc.2016.217 |
| [9] | Pittet MJ, Michielin O, Migliorini D. Clinical relevance of tumour-associated macrophages[J]. Nat Rev Clin Oncol, 2022, 19(6): 402-21. doi:10.1038/s41571-022-00620-6 |
| [10] | 中华人民共和国国家卫生健康委员会医政司. 原发性肝癌诊疗指南(2024年版)[J].协和医学杂志, 2024, 15(3): 532-59. doi:10.3877/cma.j.issn.2095-3232.2024.04.001 |
| [11] | 韩 雅, 施 美, 董莉莉, 等. 益气解毒方对HepG2小鼠模型免疫作用机制调控的实验研究[J]. 环球中医药, 2025, 18(1): 20-6. doi:10.3969/j.issn.1674-1749.2025.01.004 |
| [12] | 徐 朦, 张 鹏, 张国梁. 益气解毒方治疗原发性肝癌的作用机制:基于网络药理学及分子对接方法[J]. 南方医科大学学报, 2022, 42(6):805-14. |
| [13] | 徐 朦, 陈丽娜, 吴金玉, 等. “白花蛇舌草-半枝莲”治疗原发性肝癌的机制 研究: 基于网络药理学、分子对接及体外实验验证[J]. 南方医科大学学报, 2025, 45(1): 80-9. |
| [14] | Cheng K, Cai N, Zhu J, et al. Tumor-associated macrophages in liver cancer: From mechanisms to therapy[J]. Cancer Commun: Lond, 2022, 42(11): 1112-40. doi:10.1002/cac2.12345 |
| [15] | He R, He Y, Du R, et al. Revisiting of TAMs in tumor immune microenvironment: Insight from NF‑κB signaling pathway[J]. Biomed Pharmacother, 2023, 165: 115090. doi:10.1016/j.biopha.2023.115090 |
| [16] | Dolcet X, Llobet D, Pallares J, et al. NF-kB in development and progression of human cancer[J]. Virchows Arch, 2005, 446(5): 475-82. doi:10.1007/s00428-005-1264-9 |
| [17] | Galasso L, Cerrito L, Maccauro V, et al. Inflammatory response in the pathogenesis and treatment of hepatocellular carcinoma: a double-edged weapon[J]. Int J Mol Sci, 2024, 25(13): 7191. doi:10.3390/ijms25137191 |
| [18] | Lawrence T. The nuclear factor NF-kappaB pathway in inflammation[J]. Cold Spring Harb Perspect Biol, 2009, 1(6): a001651. doi:10.1101/cshperspect.a001651 |
| [19] | Farhood B, Najafi M, Mortezaee K. CD8+ cytotoxic T lymphocytes in cancer immunotherapy: a review[J]. J Cell Physiol, 2019, 234(6): 8509-21. doi:10.1002/jcp.27782 |
| [20] | Shapouri-Moghaddam A, Mohammadian S, Vazini H, et al. Macrophage plasticity, polarization, and function in health and disease[J]. J Cell Physiol, 2018, 233(9): 6425-40. doi:10.1002/jcp.26429 |
| [21] | Kashfi K, Kannikal J, Nath N. Macrophage reprogramming and cancer therapeutics: role of iNOS-derived NO[J]. Cells, 2021, 10(11): 3194. doi:10.3390/cells10113194 |
| [22] | Bejarano L, Jordāo MJC, Joyce JA. Therapeutic targeting of the tumor microenvironment[J]. Cancer Discov, 2021, 11(4): 933-59. doi:10.1158/2159-8290.cd-20-1808 |
| [23] | Zhang X, Lao M, Sun K, et al. Sphingolipid synthesis in tumor-associated macrophages confers immunotherapy resistance in hepatocellular carcinoma[J]. Sci Adv, 2025, 11(21): eadv0558. doi:10.1126/sciadv.adv0558 |
| [24] | Zhou WL, Yang FT, Zhang XZ. Roles of M1 macrophages and their extracellular vesicles in cancer therapy[J]. Cells, 2024, 13(17): 1428. doi:10.3390/cells13171428 |
| [25] | Shao N, Qiu H, Liu J, et al. Targeting lipid metabolism of macrophages: a new strategy for tumor therapy[J]. J Adv Res, 2025, 68: 99-114. doi:10.1016/j.jare.2024.02.009 |
| [26] | Cornice J, Verzella D, Arboretto P, et al. NF‑κB: Governing macrophages in cancer[J].. Genes (Basel), 2024, 15(2):197. doi:10.3390/genes15020197 |
| [27] | Giridharan S, Srinivasan M. Mechanisms of NF‑κB p65 and strategies for therapeutic manipulation[J]. J Inflamm Res, 2018, 11: 407-19. doi:10.2147/jir.s140188 |
| [28] | Napetschnig J, Wu H. Molecular basis of NF-κB signaling[J]. Annu Rev Biophys, 2013, 42: 443-68. doi:10.1146/annurev-biophys-083012-130338 |
| [29] | Yu Z, Li Y, Li Y, et al. Bufalin stimulates antitumor immune response by driving tumor-infiltrating macrophage toward M1 phenotype in hepatocellular carcinoma[J]. J Immunother Cancer, 2022, 10(5): e004297. doi:10.1136/jitc-2021-004297 |
| [30] | Porta C, Rimoldi M, Raes G, et al. Tolerance and M2 (alternative) macrophage polarization are related processes orchestrated by p50 nuclear factor kappaB[J]. Proc Natl Acad Sci USA, 2009, 106(35): 14978-83. doi:10.1073/pnas.0809784106 |
| [1] | 梁芷晴, 潘富珍, 邓利强, 麦哲芬, 马云, 施传坚, 付卫明. 生脉散通过调控乳酸/Wnt/β-catenin/LDHA通路改善非小细胞肺癌奥希替尼耐药的作用机制[J]. 南方医科大学学报, 2026, 46(3): 523-531. |
| [2] | 杨佳瑶, 何玉莲, 郭延垒, 尚芳红, 花雷, 阳勇, 张小梅, 魏江平. 党参-茯苓配伍调控ERα/PI3K/Akt信号通路改善单侧颈总动脉永久结扎痴呆大鼠的认知障碍[J]. 南方医科大学学报, 2026, 46(2): 247-258. |
| [3] | 许红淼, 何兰, 熊雨, 邹芳, 蔺婷, 江志超, 唐乐, 何迎春, 周芳亮. 益气解毒方通过调控AKT1/GLUT1信号通路抑制鼻咽癌的恶性进展[J]. 南方医科大学学报, 2026, 46(2): 259-270. |
| [4] | 邱佳惠, 陈萌, 满如, 陈鑫, 邱东瑞, 常启同, 马洪玉. 化瘀通便汤通过抑制PI3K/Akt信号通路促进慢传输型便秘大鼠肠道Cajal间质细胞的胞葬作用[J]. 南方医科大学学报, 2026, 46(2): 293-300. |
| [5] | 王煜煌, 王文锐, 程淑洁, 冯云龙, 张卓, 施其英, 李雨佳, 胡茜文, 吴明彩. 槲皮素通过调控Hippo通路蛋白YAP表达抑制胃癌细胞增殖与迁移并促进其凋亡[J]. 南方医科大学学报, 2026, 46(2): 374-384. |
| [6] | 尹林, 张可妮, 乔通, 牛民主, 殷丽霞, 刘馨悦, 耿志军, 李静, 胡建国. 藜芦酸通过激活Nrf2/HO-1信号通路减轻氧化应激缓解葡聚糖硫酸钠诱导的小鼠实验性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 403-411. |
| [7] | 王喆, 孔柯瑜, 金明昊, 伍信儒, 范文轩, 翟赞京, 胡子豪, 牛琳, 齐岩松, 徐永胜. 叶酸预处理髌下脂肪垫来源间充质干细胞来源的外泌体调控巨噬细胞极化[J]. 南方医科大学学报, 2026, 46(1): 166-174. |
| [8] | 赵铖, 李稳, 郑宝寿, 王光明, 肖芝松, 李云鹏. lncRNA SNHG12与ELAVL1相互作用激活PI3K/AKT信号通路促进前列腺癌细胞多西他赛的耐药机制[J]. 南方医科大学学报, 2026, 46(1): 183-190. |
| [9] | 李楠, 张亮, 郭俏凤, 周越, 刘长江. 有氧运动通过调控miR-221-3p介导的脂肪组织巨噬细胞极化改善小鼠胰岛素抵抗[J]. 南方医科大学学报, 2026, 46(1): 74-82. |
| [10] | 马倩倩, 牛钰琪, 左铭钰, 李鑫, 符竣轲, 王瑾瑾. 鬼箭羽通过抑制AGEs-RAGE信号转导通路改善晚期糖基化终末产物诱导的小鼠肾足细胞损伤[J]. 南方医科大学学报, 2025, 45(9): 1938-1945. |
| [11] | 张玉, 胡音琦, 李佩佩, 时潇, 徐伟, 胡建鹏. 脑络欣通通过激活HIF-1α/VEGF信号通路促进氧糖剥夺/复氧复糖损伤后大鼠的脑微血管内皮细胞增殖[J]. 南方医科大学学报, 2025, 45(9): 1980-1988. |
| [12] | 饶璐, 丁家和, 魏江平, 阳勇, 张小梅, 王计瑞. 槐花通过抑制PI3K/AKT通路减轻炎症反应治疗银屑病[J]. 南方医科大学学报, 2025, 45(9): 1989-1996. |
| [13] | 王庆阁, 赵晓慧, 何宇轩, 刘飞祥, 张运克. 芪芎左归颗粒通过上调BDNF/TrkB通路提高衰老大鼠突触可塑性[J]. 南方医科大学学报, 2025, 45(8): 1589-1598. |
| [14] | 张兆君, 吴琼, 谢苗苗, 叶洳吟, 耿晨晨, 石纪雯, 杨清玲, 王文锐, 石玉荣. 层状双氢氧化物负载si-NEAT1通过miR-133b/PD-L1轴调控乳腺癌紫杉醇耐药及巨噬细胞极化[J]. 南方医科大学学报, 2025, 45(8): 1718-1731. |
| [15] | 范正媛, 沈子涵, 李亚, 沈婷婷, 李高峰, 李素云. 补肺益肾方对香烟烟雾提取物诱导的人支气管上皮细胞损伤的保护作用及其机制[J]. 南方医科大学学报, 2025, 45(7): 1372-1379. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||