南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 247-258.doi: 10.12122/j.issn.1673-4254.2026.02.02
• • 上一篇
杨佳瑶1,2( ), 何玉莲1,2, 郭延垒1, 尚芳红1,2,3, 花雷1,2, 阳勇1,2,4, 张小梅1,2,3,4(
), 何玉莲1,2, 郭延垒1, 尚芳红1,2,3, 花雷1,2, 阳勇1,2,4, 张小梅1,2,3,4( ), 魏江平1,2,3(
), 魏江平1,2,3( )
)
收稿日期:2025-08-10
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
张小梅,魏江平
E-mail:yangjiayao959@163.com;ZXM761@163.com;sichuanwjp@163.com
作者简介:杨佳瑶,在读硕士研究生,E-mail: yangjiayao959@163.com
基金资助:
Jiayao YANG1,2( ), Yulian He1,2, Yanlei GUO1, Fanghong SHANG1,2,3, Lei HUA1,2, Yong YANG1,2,4, Xiaomei ZHANG1,2,3,4(
), Yulian He1,2, Yanlei GUO1, Fanghong SHANG1,2,3, Lei HUA1,2, Yong YANG1,2,4, Xiaomei ZHANG1,2,3,4( ), Jiangping WEI1,2,3(
), Jiangping WEI1,2,3( )
)
Received:2025-08-10
Online:2026-02-20
Published:2026-03-10
Contact:
Xiaomei ZHANG, Jiangping WEI
E-mail:yangjiayao959@163.com;ZXM761@163.com;sichuanwjp@163.com
Supported by:摘要:
目的 基于UPLC-Q-TOF-MS/MS分析研究党参-茯苓配伍(CRP)治疗痴呆症的作用机制。 方法 采用于UPLC-Q-TOF-MS/MS分析单侧颈总动脉永久结扎(UCCA)模型大鼠的 CRP 的入血入脑成分,并对入血和入脑成分进行网络药理学分析。CRP灌胃UCCA 大鼠干预1个月后通过Morris水迷宫评价其学习记忆能力;苏木精-伊红染色观察海马和皮质的病理形态学变化;NeuN染色分析海马和皮层神经元数量变化;采用代谢组学分析 CRP 治疗UCCA大鼠前后脑内代谢物变化差异;酶联免疫吸附测定(ELISA)检测脑内谷氨酸和γ-氨基丁酸(GABA)的含量;免疫组化法考察雌激素受体α(ERα)的蛋白表达;Western blotting分析p-PI3K、PI3K、p-Akt、Akt的蛋白表达。 结果 在UCCA大鼠的脑组织和血液中分别鉴定出125种和126种成分,其中,脑组织中发现了89种党参成分和 36 种茯苓成分,血液中发现了85种党参成分和41种茯苓成分。网络药理学分析表明,CRP治疗痴呆主要与调节进入脑组织的成分所影响的PI3K/Akt等信号通路有关。与假手术组比较,模型大鼠的第5天的逃避潜伏期明显延长(P<0.01),平台象限活动时间、海马和皮质的神经元明显损伤且NeuN平均光密度明显下降(P<0.05);脑内谷氨酸和GABA/谷氨酸含量明显升高(P<0.05),ERα、p-PI3K/PI3K、p-Akt/Akt蛋白表达下调(P<0.05)。与模型组比较,CRP干预大鼠的第5天的逃避潜伏期明显缩短(P<0.01),穿越平台次数、平台象限活动时间均明显增加(P<0.05),海马和皮质的神经元损伤好转且NeuN平均光密度明显升高(P<0.05);代谢组学分析表明CRP治疗作用主要富集于雌激素信号通路和 GABA 能突触等信号通路。CRP组大鼠脑内的谷氨酸和GABA/谷氨酸含量明显降低(P<0.05),ERα、p-PI3K/PI3K和p-Akt/Akt蛋白表达上调(P<0.05)。 结论 CRP 能够改善UCCA痴呆大鼠的学习和记忆缺陷,这与其对脑内ERα/PI3K/Akt信号通路的调节有关。
杨佳瑶, 何玉莲, 郭延垒, 尚芳红, 花雷, 阳勇, 张小梅, 魏江平. 党参-茯苓配伍调控ERα/PI3K/Akt信号通路改善单侧颈总动脉永久结扎痴呆大鼠的认知障碍[J]. 南方医科大学学报, 2026, 46(2): 247-258.
Jiayao YANG, Yulian He, Yanlei GUO, Fanghong SHANG, Lei HUA, Yong YANG, Xiaomei ZHANG, Jiangping WEI. Radix codonopsis combined with Poria improves cognitive impairment in rats with unilateral common carotid artery ligation by regulating the ERα/PI3K/Akt signaling pathway[J]. Journal of Southern Medical University, 2026, 46(2): 247-258.
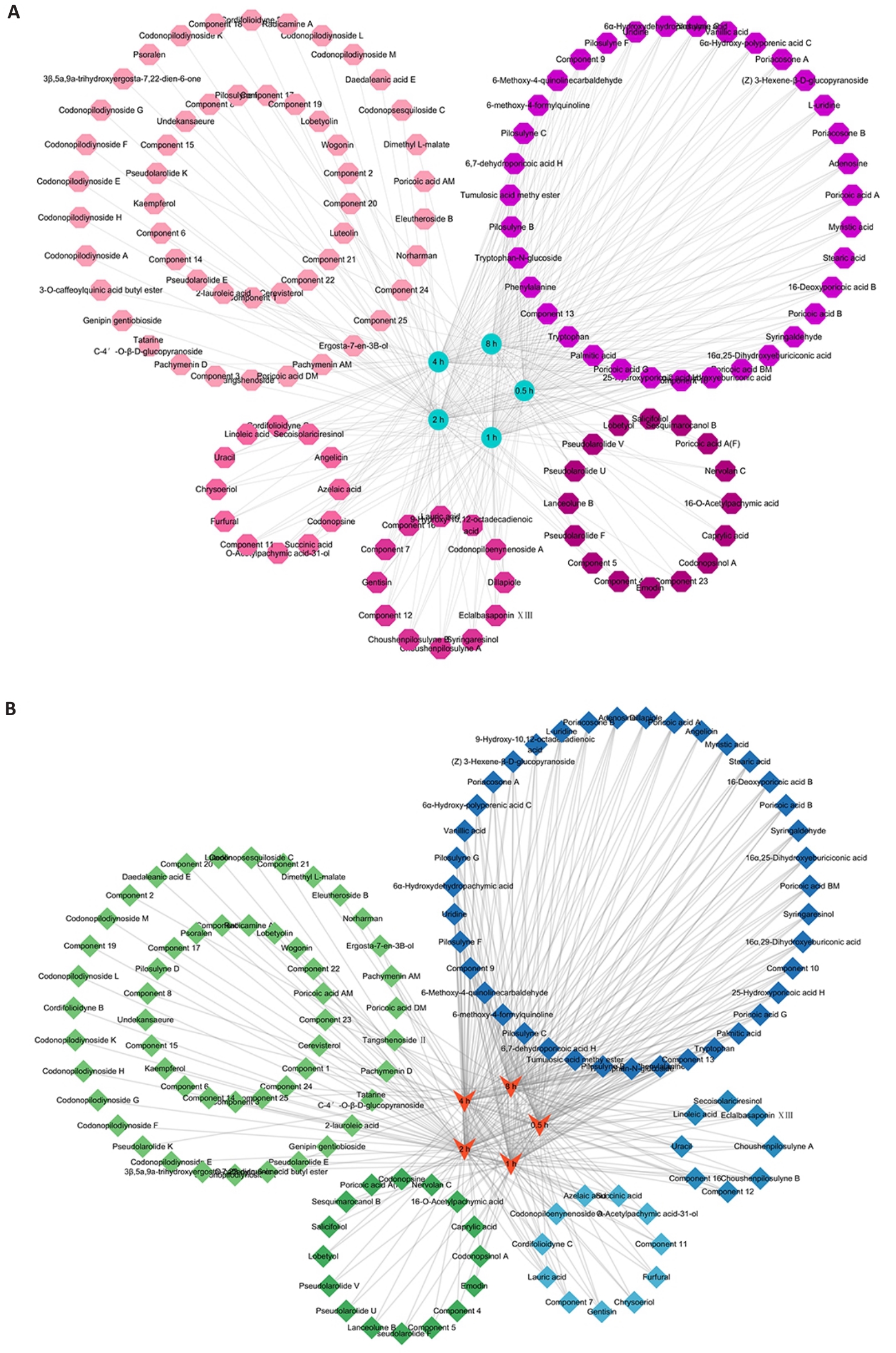
图1 CRP 在UCCA大鼠的入血成分和入脑成分
Fig.1 UPLC-Q-TOF-MS/MS for analyzing the components of Radix codonopsis combined with Poria (CRP) in blood and brain of medicated rats at different time points. A: Network diagram depicting the distribution relationships of CRP components in the blood at different time points. B: Network diagram of the distribution relationships of CRP components in the brain at different time points. The circles of the same color denote components detected at the same time point.
| NO. | Retain time (min) | Ion peak assignment | Theoretical value mass (m/z) | Measured value mass (m/z) | Deviation (ppm) | Chemical formula | Name |
|---|---|---|---|---|---|---|---|
| 1 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 6,7-dehydroporicoic acid H |
| 2 | 6.96 | [M-H]- | 466.30831 | 465.30019 | -1.8 | C30H42O4 | daedaleanic acid E |
| 3 | 7.98 | [M+H]+ | 444.32396 | 445.33071 | -1.2 | C28H44O4 | 3β,5a,9a-trihydroxyergosta-7,22-dien-6-one |
| 4 | 1.87 | [M+H]+ | 162.05282 | 163.06008 | -0.1 | C6H10O5 | Dimethyl L-malate |
| 5 | 2.78 | [M-H]- | 244.06954 | 243.06212 | -0.6 | C9H12N2O6 | L-uridine |
| 6 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | Poricoic acid A |
| 7 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poricoic acid B |
| 8 | 9.91 | [M-H]- | 200.17763 | 199.1705 | 0.7 | C12H24O2 | lauric acid |
| 9 | 8.83 | [M-H]-/[M+H]+ | 256.24023 | 255.23296 | 0 | C16H32O2 | palmitic acid |
| 10 | 19.07 | [M+H]+ | 512.35018 | 513.35749 | 0.1 | C32H48O5 | Poricoic acid AM |
| 11 | 6.68 | [M-H]- | 584.40769 | 583.40041 | 0 | C36H56O6 | Tumulosic acid methy ester |
| 12 | 5.8 | [M-H]- | 514.32944 | 513.32202 | -0.3 | C31H46O6 | Poricoic acid A(F) |
| 13 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | Poricoic acid BM |
| 14 | 6.4 | [M-H]- | 486.33453 | 485.32735 | 0.2 | C30H46O5 | Poricoic acid G |
| 15 | 5.34 | [M-H]- | 470.33961 | 469.33272 | 0.8 | C30H46O4 | 3β,16α-Dihydroxy-lanosta-7,9(11),24-trien-21-oic acid |
| 16 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 25-Hydroxyporicoic acid H |
| 17 | 6.62 | [M-H]- | 468.32396 | 467.31682 | 0.3 | C30H44O4 | 16-Deoxyporicoic acid B |
| 18 | 11.03 | [M+H]+ | 528.34509 | 529.3529 | 1 | C32H48O6 | Poricoic acid DM |
| 19 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poriacosone A |
| 20 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poriacosone B |
| 21 | 6.11 | [M-H]- | 496.31887 | 495.31138 | -0.4 | C31H44O5 | 6α-Hydroxy-polyporenic acid C |
| 22 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 16α,25-Dihydroxyeburiciconic acid |
| 23 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 16α,29-Dihydroxyeburiconic acid |
| 24 | 19.07 | [M+H]+ | 512.35018 | 513.35749 | 0.1 | C32H48O5 | Pachymenin D |
| 25 | 8.09 | [M+H]+ | 540.34509 | 541.35241 | 0.1 | C33H48O6 | 6α-Hydroxydehydropachymic acid |
| 26 | 5.28 | [M-H]-/[M+H]+ | 204.08988 | 203.08279 | 1 | C11H12N2O2 | tryptophan |
| 27 | 2.73 | [M+H]+ | 112.02728 | 113.03426 | -2.6 | C4H4N2O2 | uracil |
| 28 | 2.78 | [M-H]- | 244.06954 | 243.06212 | -0.6 | C9H12N2O6 | uridine |
| 29 | 5.92 | [M+H]+ | 187.06333 | 188.0706 | 0 | C11H9NO2 | 6-Methoxy-4-quinolinecarbaldehyde |
| 30 | 4.32 | [M+H]+ | 165.07898 | 166.08625 | 0 | C9H11NO2 | phenylalanine(L-Phenylalanine) |
| 31 | 5.38 | [M-H]- | 372.14203 | 371.13537 | 1.6 | C17H24O9 | tangshenoside Ⅱ |
| 32 | 8.14 | [M-H]- | 262.12051 | 261.11355 | 1.2 | C15H18O4 | nervolan C |
| 33 | 5.38 | [M-H]- | 372.14203 | 371.13537 | 1.6 | C17H24O9 | Eleutheroside B(syringin) |
| 34 | 7.61 | [M-H]- | 222.08921 | 221.08202 | 0.4 | C12H14O4 | dillapiole |
| 35 | 7.61 | [M-H]- | 222.08921 | 221.08202 | 0.4 | C12H14O4 | 1-allyl-2,6-dimethoxy-3,4-methylenedioxybenzene |
| 36 | 11.43 | [M+H]+ | 418.16277 | 419.16949 | -1.3 | C22H26O8 | syringaresinol |
| 37 | 10.18 | [M+H]+ | 362.17294 | 363.17942 | -2.2 | C20H26O6 | secoisolariciresinol |
| 38 | 5.88 | [M-H]- | 498.29814 | 497.29218 | 2.6 | C30H42O6 | pseudolarolide E |
| 39 | 8.68 | [M-H]- | 270.05282 | 269.04523 | -1.2 | C15H10O5 | apigenin |
| 40 | 11.9 | [M-H]-/[M+H]+ | 280.24023 | 279.23241 | -1.9 | C18H32O2 | linoleic acid |
| 41 | 11.34 | [M-H]- | 228.20893 | 227.20169 | 0.2 | C14H28O2 | myristic acid |
| 42 | 8.33 | [M-H]- | 284.27153 | 283.26384 | -1.5 | C18H36O2 | stearic acid |
| 43 | 6.52 | [M-H]- | 188.10486 | 187.09787 | 1.5 | C9H16O4 | azelaic acid |
| 44 | 8.83 | [M-H]-/[M+H]+ | 256.24023 | 255.23296 | 0 | C16H32O2 | Palmitic Acid (n-hexadecanoic acid) |
| 45 | 5.8 | [M-H]- | 182.05791 | 181.05037 | -1.5 | C9H10O4 | syringaldehyde |
| 46 | 1.9 | [M+H]+ | 96.02113 | 97.02847 | 0.7 | C5H4O2 | Furfural(2-furaldehyde) |
| 47 | 8.68 | [M-H]- | 270.05282 | 269.04523 | -1.2 | C15H10O5 | emodin |
| 48 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside F |
| 49 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside G |
| 50 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside K |
| 51 | 7.75 | [M-H]-/[M+H]+ | 250.12051 | 249.11329 | 0.2 | C14H18O4 | pilosulyne B |
| 52 | 7.46 | [M-H]-/[M+H]+ | 254.15181 | 253.14441 | -0.5 | C14H22O4 | pilosulyne F |
| 53 | 7.46 | [M-H]-/[M+H]+ | 254.15181 | 253.14441 | -0.5 | C14H22O4 | pilosulyne G |
| 54 | 5.8 | [M-H]- | 514.32944 | 513.32202 | -0.3 | C31H46O6 | pseudolarolide U |
| 55 | 1.97 | [M+H]+ | 255.11067 | 256.11797 | 0.1 | C12H17NO5 | radicamine A |
| 56 | 5.92 | [M+H]+ | 187.06333 | 188.0706 | 0 | C11H9NO2 | 6-methoxy-4-formylquinoline |
| 57 | 5.91 | [M-H]- | 486.18496 | 485.1766 | -2.2 | C21H30N2O11 | tatarine C-4′-O-β-D-glucopyranoside |
| 58 | 10.18 | [M+H]+ | 362.17294 | 363.17942 | -2.2 | C20H26O6 | 1,6-Hexanediol-3,4-di (4-hydroxy-3-methoxyphenyl) |
| 59 | 5.9 | [M-H]- | 540.23593 | 539.22752 | -2.1 | C30H36O9 | sesquimarocanol B |
表1 CRP 在UCCA大鼠的入血又入脑成分
Tab.1 Components of CRP in the blood and brain in CRP-treated UCCA rats
| NO. | Retain time (min) | Ion peak assignment | Theoretical value mass (m/z) | Measured value mass (m/z) | Deviation (ppm) | Chemical formula | Name |
|---|---|---|---|---|---|---|---|
| 1 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 6,7-dehydroporicoic acid H |
| 2 | 6.96 | [M-H]- | 466.30831 | 465.30019 | -1.8 | C30H42O4 | daedaleanic acid E |
| 3 | 7.98 | [M+H]+ | 444.32396 | 445.33071 | -1.2 | C28H44O4 | 3β,5a,9a-trihydroxyergosta-7,22-dien-6-one |
| 4 | 1.87 | [M+H]+ | 162.05282 | 163.06008 | -0.1 | C6H10O5 | Dimethyl L-malate |
| 5 | 2.78 | [M-H]- | 244.06954 | 243.06212 | -0.6 | C9H12N2O6 | L-uridine |
| 6 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | Poricoic acid A |
| 7 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poricoic acid B |
| 8 | 9.91 | [M-H]- | 200.17763 | 199.1705 | 0.7 | C12H24O2 | lauric acid |
| 9 | 8.83 | [M-H]-/[M+H]+ | 256.24023 | 255.23296 | 0 | C16H32O2 | palmitic acid |
| 10 | 19.07 | [M+H]+ | 512.35018 | 513.35749 | 0.1 | C32H48O5 | Poricoic acid AM |
| 11 | 6.68 | [M-H]- | 584.40769 | 583.40041 | 0 | C36H56O6 | Tumulosic acid methy ester |
| 12 | 5.8 | [M-H]- | 514.32944 | 513.32202 | -0.3 | C31H46O6 | Poricoic acid A(F) |
| 13 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | Poricoic acid BM |
| 14 | 6.4 | [M-H]- | 486.33453 | 485.32735 | 0.2 | C30H46O5 | Poricoic acid G |
| 15 | 5.34 | [M-H]- | 470.33961 | 469.33272 | 0.8 | C30H46O4 | 3β,16α-Dihydroxy-lanosta-7,9(11),24-trien-21-oic acid |
| 16 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 25-Hydroxyporicoic acid H |
| 17 | 6.62 | [M-H]- | 468.32396 | 467.31682 | 0.3 | C30H44O4 | 16-Deoxyporicoic acid B |
| 18 | 11.03 | [M+H]+ | 528.34509 | 529.3529 | 1 | C32H48O6 | Poricoic acid DM |
| 19 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poriacosone A |
| 20 | 5.75 | [M-H]- | 484.31887 | 483.31188 | 0.6 | C30H44O5 | Poriacosone B |
| 21 | 6.11 | [M-H]- | 496.31887 | 495.31138 | -0.4 | C31H44O5 | 6α-Hydroxy-polyporenic acid C |
| 22 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 16α,25-Dihydroxyeburiciconic acid |
| 23 | 5.83 | [M-H]- | 498.33453 | 497.32724 | 0 | C31H46O5 | 16α,29-Dihydroxyeburiconic acid |
| 24 | 19.07 | [M+H]+ | 512.35018 | 513.35749 | 0.1 | C32H48O5 | Pachymenin D |
| 25 | 8.09 | [M+H]+ | 540.34509 | 541.35241 | 0.1 | C33H48O6 | 6α-Hydroxydehydropachymic acid |
| 26 | 5.28 | [M-H]-/[M+H]+ | 204.08988 | 203.08279 | 1 | C11H12N2O2 | tryptophan |
| 27 | 2.73 | [M+H]+ | 112.02728 | 113.03426 | -2.6 | C4H4N2O2 | uracil |
| 28 | 2.78 | [M-H]- | 244.06954 | 243.06212 | -0.6 | C9H12N2O6 | uridine |
| 29 | 5.92 | [M+H]+ | 187.06333 | 188.0706 | 0 | C11H9NO2 | 6-Methoxy-4-quinolinecarbaldehyde |
| 30 | 4.32 | [M+H]+ | 165.07898 | 166.08625 | 0 | C9H11NO2 | phenylalanine(L-Phenylalanine) |
| 31 | 5.38 | [M-H]- | 372.14203 | 371.13537 | 1.6 | C17H24O9 | tangshenoside Ⅱ |
| 32 | 8.14 | [M-H]- | 262.12051 | 261.11355 | 1.2 | C15H18O4 | nervolan C |
| 33 | 5.38 | [M-H]- | 372.14203 | 371.13537 | 1.6 | C17H24O9 | Eleutheroside B(syringin) |
| 34 | 7.61 | [M-H]- | 222.08921 | 221.08202 | 0.4 | C12H14O4 | dillapiole |
| 35 | 7.61 | [M-H]- | 222.08921 | 221.08202 | 0.4 | C12H14O4 | 1-allyl-2,6-dimethoxy-3,4-methylenedioxybenzene |
| 36 | 11.43 | [M+H]+ | 418.16277 | 419.16949 | -1.3 | C22H26O8 | syringaresinol |
| 37 | 10.18 | [M+H]+ | 362.17294 | 363.17942 | -2.2 | C20H26O6 | secoisolariciresinol |
| 38 | 5.88 | [M-H]- | 498.29814 | 497.29218 | 2.6 | C30H42O6 | pseudolarolide E |
| 39 | 8.68 | [M-H]- | 270.05282 | 269.04523 | -1.2 | C15H10O5 | apigenin |
| 40 | 11.9 | [M-H]-/[M+H]+ | 280.24023 | 279.23241 | -1.9 | C18H32O2 | linoleic acid |
| 41 | 11.34 | [M-H]- | 228.20893 | 227.20169 | 0.2 | C14H28O2 | myristic acid |
| 42 | 8.33 | [M-H]- | 284.27153 | 283.26384 | -1.5 | C18H36O2 | stearic acid |
| 43 | 6.52 | [M-H]- | 188.10486 | 187.09787 | 1.5 | C9H16O4 | azelaic acid |
| 44 | 8.83 | [M-H]-/[M+H]+ | 256.24023 | 255.23296 | 0 | C16H32O2 | Palmitic Acid (n-hexadecanoic acid) |
| 45 | 5.8 | [M-H]- | 182.05791 | 181.05037 | -1.5 | C9H10O4 | syringaldehyde |
| 46 | 1.9 | [M+H]+ | 96.02113 | 97.02847 | 0.7 | C5H4O2 | Furfural(2-furaldehyde) |
| 47 | 8.68 | [M-H]- | 270.05282 | 269.04523 | -1.2 | C15H10O5 | emodin |
| 48 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside F |
| 49 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside G |
| 50 | 7.24 | [M-H]- | 542.23633 | 541.23118 | 3.9 | C26H38O12 | codonopilodiynoside K |
| 51 | 7.75 | [M-H]-/[M+H]+ | 250.12051 | 249.11329 | 0.2 | C14H18O4 | pilosulyne B |
| 52 | 7.46 | [M-H]-/[M+H]+ | 254.15181 | 253.14441 | -0.5 | C14H22O4 | pilosulyne F |
| 53 | 7.46 | [M-H]-/[M+H]+ | 254.15181 | 253.14441 | -0.5 | C14H22O4 | pilosulyne G |
| 54 | 5.8 | [M-H]- | 514.32944 | 513.32202 | -0.3 | C31H46O6 | pseudolarolide U |
| 55 | 1.97 | [M+H]+ | 255.11067 | 256.11797 | 0.1 | C12H17NO5 | radicamine A |
| 56 | 5.92 | [M+H]+ | 187.06333 | 188.0706 | 0 | C11H9NO2 | 6-methoxy-4-formylquinoline |
| 57 | 5.91 | [M-H]- | 486.18496 | 485.1766 | -2.2 | C21H30N2O11 | tatarine C-4′-O-β-D-glucopyranoside |
| 58 | 10.18 | [M+H]+ | 362.17294 | 363.17942 | -2.2 | C20H26O6 | 1,6-Hexanediol-3,4-di (4-hydroxy-3-methoxyphenyl) |
| 59 | 5.9 | [M-H]- | 540.23593 | 539.22752 | -2.1 | C30H36O9 | sesquimarocanol B |
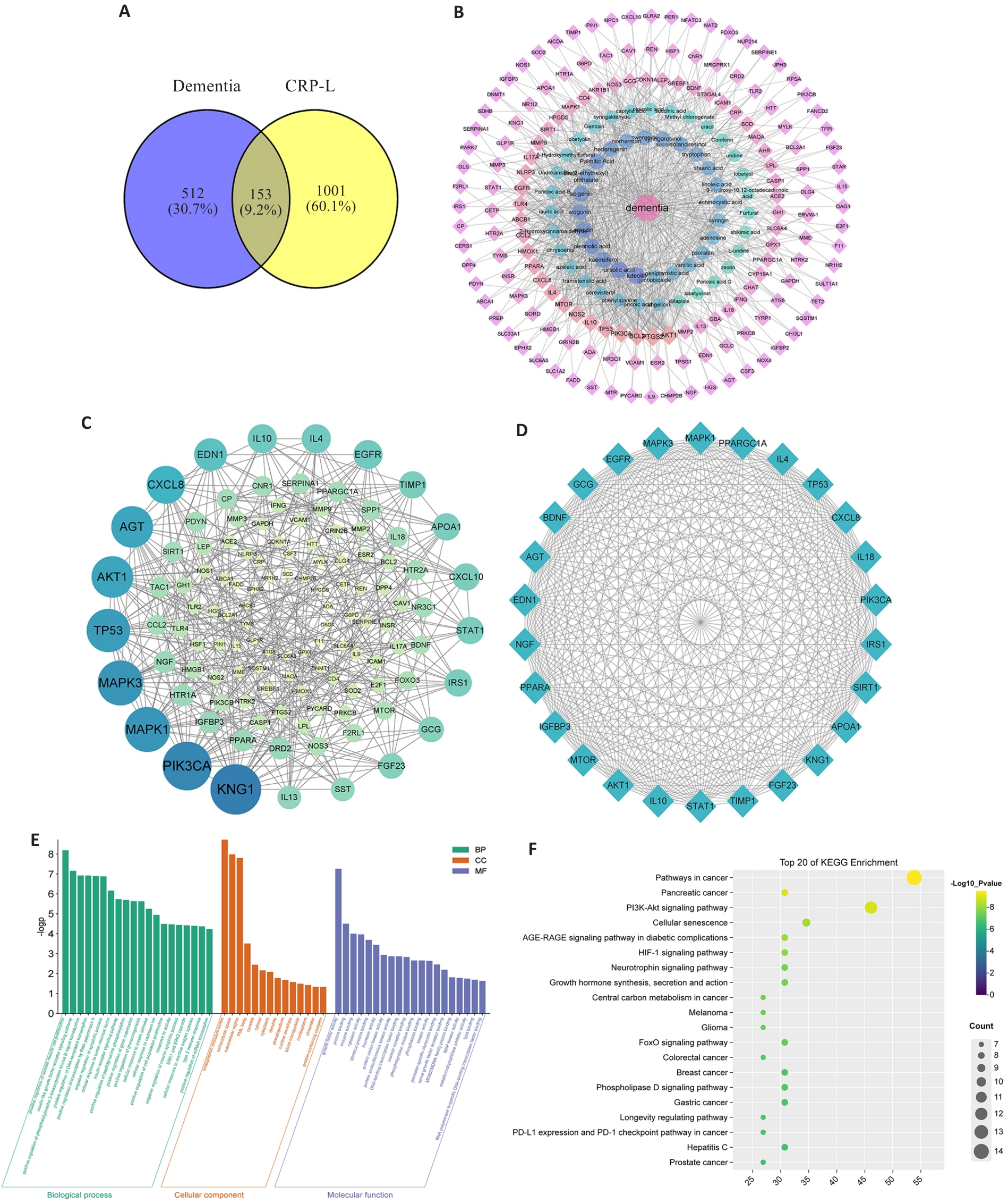
图2 CRP入血和入脑成分的网络药理学分析
Fig.2 Network pharmacology analysis of the components of CRP in the blood and brain of CRP-treated UCCA rats. A: Venn diagram of the targets of the components of CRP in rat blood and brain and the targets of dementia. B: Disease-compounds-targets network. C: PPI network of the intersection targets for treatment of dementia by CRP components in the blood and brain. D: Network diagram of the key targets. E: GO enrichment analysis of the key targets. F: KEGG enrichment analysis of the key targets.
| Group | Dose (g/kg) | First day (s) | Second day (s) | Third day (s) | Fourth day (s) | Fifth day (s) |
|---|---|---|---|---|---|---|
| Sham | - | 106.00±31.81 | 70.36±22.25 | 54.49±17.23^^ | 22.33±7.06^^^## | 16.39±5.18^^^## |
| Model | - | 111.55±35.28 | 73.46±23.23 | 57.63±18.23 | 70.05±22.15** | 57.67±18.24^^** |
| Aricept | 1.17×10-3 | 93.94±29.71 | 78.36±24.80 | 43.53±13.77^^ | 29.67±9.38^^^# | 26.05±8.24^^^# |
| CRP-L | 3.5 | 92.78±29.34 | 85.58±27.02 | 59.17±18.71 | 33.98±10.75^^^# | 23.75±7.51^^^## |
| CRP-H | 7 | 98.54±31.16 | 98.89±31.27 | 69.63±22.02 | 50.43±15.95^^^ | 22.81±7.21^^^## |
表2 CRP对UCCA大鼠隐藏平台实验逃避潜伏期的影响
Tab.2 Effects of CRP on escape latency of UCCA rats in the hidden platform (Mean±SD)
| Group | Dose (g/kg) | First day (s) | Second day (s) | Third day (s) | Fourth day (s) | Fifth day (s) |
|---|---|---|---|---|---|---|
| Sham | - | 106.00±31.81 | 70.36±22.25 | 54.49±17.23^^ | 22.33±7.06^^^## | 16.39±5.18^^^## |
| Model | - | 111.55±35.28 | 73.46±23.23 | 57.63±18.23 | 70.05±22.15** | 57.67±18.24^^** |
| Aricept | 1.17×10-3 | 93.94±29.71 | 78.36±24.80 | 43.53±13.77^^ | 29.67±9.38^^^# | 26.05±8.24^^^# |
| CRP-L | 3.5 | 92.78±29.34 | 85.58±27.02 | 59.17±18.71 | 33.98±10.75^^^# | 23.75±7.51^^^## |
| CRP-H | 7 | 98.54±31.16 | 98.89±31.27 | 69.63±22.02 | 50.43±15.95^^^ | 22.81±7.21^^^## |
| Group | Dose (g/kg) | Platform quadrant cumulative Time (s) | Times of platform crossing time (time) | Average swimming speed (cm/s) |
|---|---|---|---|---|
| Sham | - | 32.37±10.24# | 4.22±1.34 | 14.74±4.66 |
| Model | - | 21.33±6.75* | 2.50±0.79 | 12.44±3.94 |
| Aricept | 1.17×10-3 | 34.68±10.97## | 4.20±1.33 | 13.69±4.33 |
| CRP-L | 3.5 | 33.94±10.73# | 5.80±1.83## | 13.58±4.29 |
| CRP-H | 7 | 29.22±9.24 | 3.20±1.01 | 13.87±4.39 |
表3 CRP对UCCA大鼠空间探索能力的影响
Tab.3 Effects of CRP on spatial exploration of UCCA rats (Mean±SD)
| Group | Dose (g/kg) | Platform quadrant cumulative Time (s) | Times of platform crossing time (time) | Average swimming speed (cm/s) |
|---|---|---|---|---|
| Sham | - | 32.37±10.24# | 4.22±1.34 | 14.74±4.66 |
| Model | - | 21.33±6.75* | 2.50±0.79 | 12.44±3.94 |
| Aricept | 1.17×10-3 | 34.68±10.97## | 4.20±1.33 | 13.69±4.33 |
| CRP-L | 3.5 | 33.94±10.73# | 5.80±1.83## | 13.58±4.29 |
| CRP-H | 7 | 29.22±9.24 | 3.20±1.01 | 13.87±4.39 |
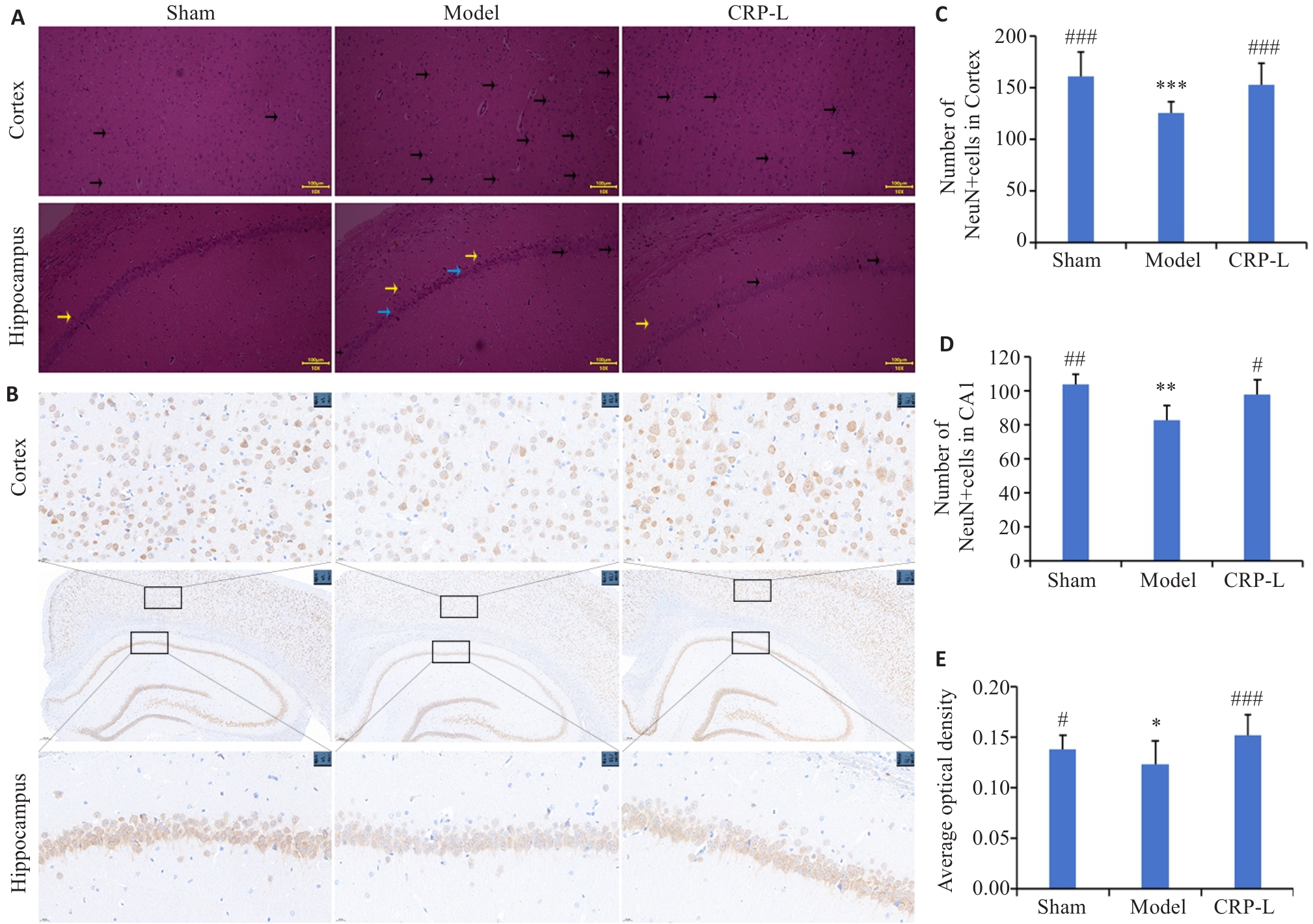
图3 CRP对UCCA大鼠海马皮层病理形态学和神经元数量的影响
Fig.3 Effects of CRP on pathological morphology and number of neurons in the hippocampus and cortex of UCCA rats. A: HE staining (Black arrow indicate the cells undergoing degeneration or death, yellow arrows indicate the nerve cells in disordered alignment, and blue arrows indicate enlargement of intercellular spaces and decreased cell layers; original magnification: ×10). B: NeuN staining (×5; ×40). C: Number of NeuN+ cells in the cortex. D: Number of NeuN+ cells in the CA1 area. E: Average optical density. Data are presented as Mean±SD (n=6). *P<0.05, **P<0.01, ***P<0.001 vs Sham; #P<0.05, ##P<0.01, ###P<0.001 vs Model.
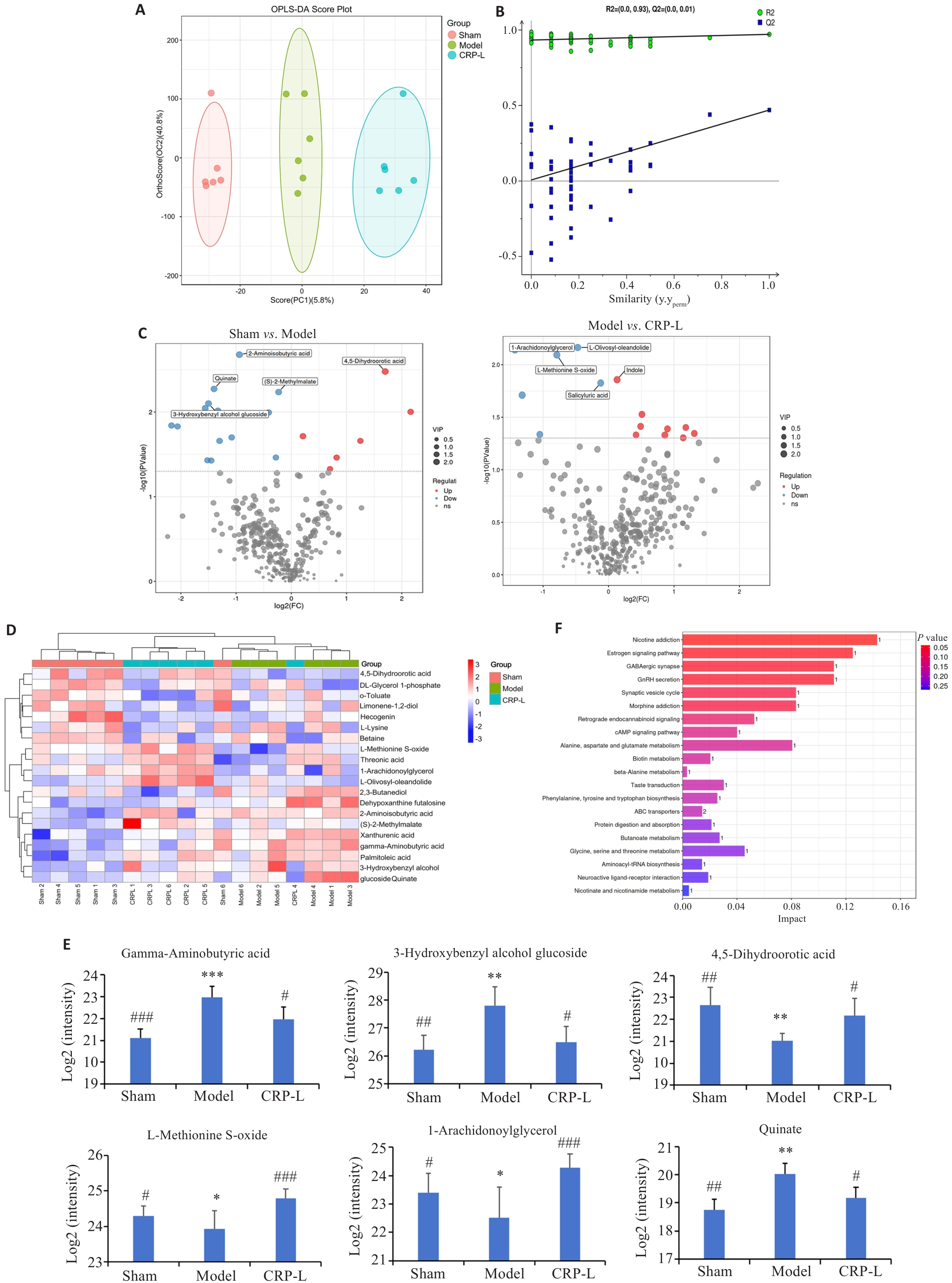
图4 CRP对UCCA大鼠脑代谢组学的影响
Fig.4 Effects of CRP on brain metabolomics in UCCA rats. A: OPLS-DA score plot. B: Permutation test. C: Volcano plot. D: Differential molecule heat map. E: Relative contents of representative differential molecules. F: KEGG enrichment analysis. Data are presented as Mean±SD (n=6). *P<0.05, **P<0.01, ***P<0.001 vs Sham, #P<0.05, ##P<0.01, ###P<0.001 vs Model.
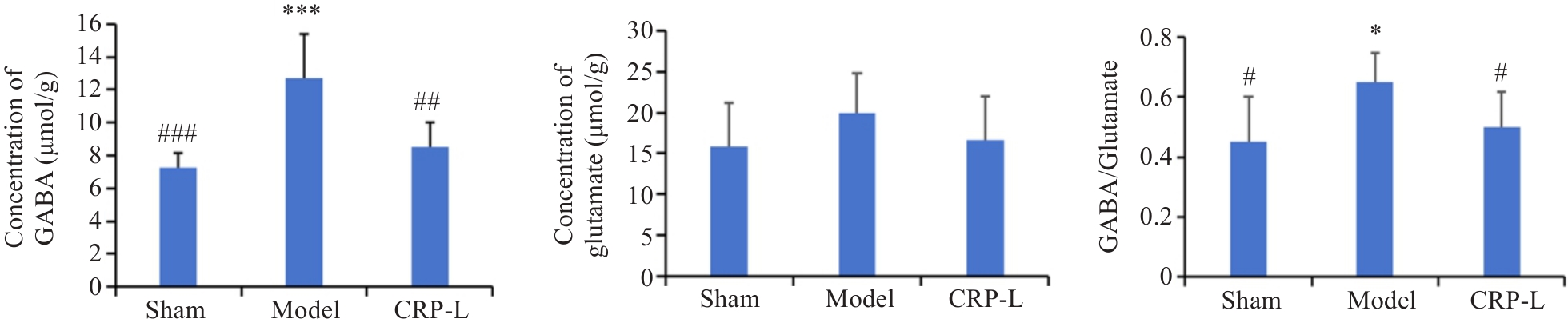
图5 CRP对UCCA大鼠脑内GABA、谷氨酸含量的影响
Fig.5 Effect of CRP on GABA and glutamate contents in the brain of UCCA rats. Data are presented as Mean±SD (n=6). *P<0.05, ***P<0.001 vs Sham; #P<0.05, ##P<0.01, ###P<0.001 vs Model.
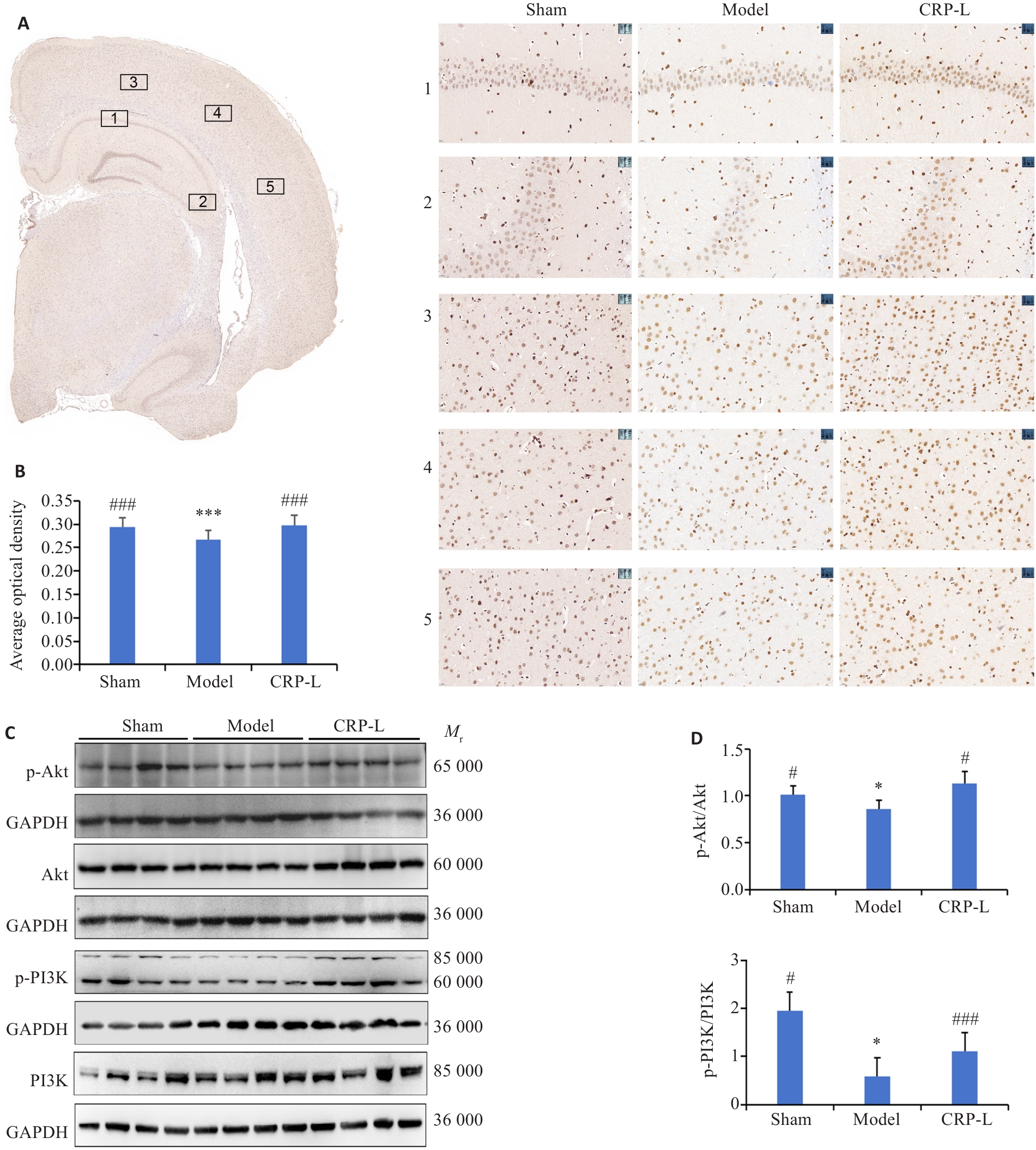
图6 CRP对UCCA大鼠脑内ERα/PI3K/Akt信号通路的影响
Fig.6 Effect of CRP on the ERα/PI3K/Akt signaling pathway in the brain of UCCA rats. A: Anatomical regions selected for ERα expression analysis and representative images from each group (×1; ×40). 1: Cornu Ammonis area 1; 2: Cornu Ammonis area 3; 3: Visual area 2, Mediolateral part; 4: Visual area 2, Lateral part; 5: Auditory Dorsal.B: Average optical density of ERα. C: Representative protein bands. D: Relative expression levels of p-Akt and p-PI3K. Data are presented as Mean±SD (n=6). *P<0.05, ***P<0.001 vs Sham; #P<0.05, ###P<0.001 vs Model.
| [1] | Zhi N, Ren RJ, Qi JL, et al. The China Alzheimer report 2025[J]. Gen Psych, 2025, 38(4): e102020. 12-26. doi:10.1136/gpsych-2024-102020 |
| [2] | 刘雨辉, 卜先乐, 马 辛, 等. 阿尔茨海默病药物治疗指南[J]. 阿尔茨海默病及相关病杂志, 2025, 8(1): 8-16. |
| [3] | Cummings JL, Zhou YD, Lee G, et al. Alzheimer's disease drug development pipeline: 2025[J]. lzheimers Dement (N Y), 2025, 11(2): e70098. doi:10.1002/trc2.70098 |
| [4] | Law BYK, Wu AG, Wang MJ, et al. Chinese medicine: a hope for neurodegenerative diseases [J]. J Alzheimers Dis, 2017, 60(s1): S151-60. doi:10.3233/jad-170374 |
| [5] | 魏江平, 赵子瑄, 曾 静, 等. 基于神经活性配体受体交互作用探讨党参-茯苓配伍改善痴呆小鼠学习记忆能力的作用机制[J]. 中药新药与临床药理, 2023, 34(11): 1514-24. |
| [6] | 徐 杨, 何芷绮, 刘晓凤, 等. 代谢组学在中药复方制剂领域的研究进展及面临的挑战[J]. 中草药, 2024, 55(4): 1354-64. |
| [7] | 刘玥欣, 徐 岩, 赵昕彤, 等. 鹿茸多肽对单侧颈总动脉结扎所致轻度认知功能障碍模型大鼠的保护作用研究[J]. 人参研究, 2019, 31(6): 42-4. |
| [8] | 曾 静, 陈 荣, 任香怡, 等. 异功散通过调控肠道微生物改善痴呆症模型大鼠的认知下降[J]. 南方医科大学学报, 2024, 44(7): 1297-305. |
| [9] | Shi ZS, Zhu LH, Li TT, et al. Neuroprotective mechanisms of Lycium barbarum polysaccharides against ischemic insults by regulating NR2B and NR2A containing NMDA receptor signaling pathways[J]. Front Cell Neurosci, 2017, 11: 288. doi:10.3389/fncel.2017.00288 |
| [10] | 王胜鹏, 胡杨洋, 陈锐娥, 等. 中药药对的系统研究(Ⅱ): 药效与药动学研究[J]. 世界科学技术: 中医药现代化, 2012, 14(2): 1322-8. |
| [11] | 牛 明, 张斯琴, 张 博, 等. 《网络药理学评价方法指南》解读[J]. 中草药, 2021, 52(14): 4119-29. |
| [12] | 武晓伟, 郝艳艳, 聂春霞, 等. 中药血清药物化学的方法学概述及应用领域研究进展[J]. 中国实验方剂学杂志, 2019, 25(3): 173-9. |
| [13] | 王一凯, 董金旭, 白玉涛, 等. 基于超高效液相色谱-四极杆飞行时间质谱和靶向网络药理学技术结合分子对接研究薤白治疗高血脂症的药效成分[J]. 分析化学, 2024, 52(4): 578-86. |
| [14] | Wang J, Chen Q, Sheng RL, et al. Integration of transdermal chemistry and network pharmacology to decipher the mechanism of ShexiangZhuifeng analgesic plaster to treat rheumatoid arthritis[J]. Phytomedicine, 2023, 108: 154507. doi:10.1016/j.phymed.2022.154507 |
| [15] | 朱星晔, 于嘉祥, 袁子越, 等. 基于LC-MS技术联合生物信息学预测半夏白术天麻汤和当归芍药散干预梅尼埃病的机制[J]. 中国实验方剂学杂志, 2025, 31(24): 50-60. |
| [16] | 杨 丽, 李孟莹, 胡文青, 等. 基于HPLC-Q-TOF-MS/MS和网络药理学的补肺丸入血成分、肺病治疗潜能及新型冠状病毒感染恢复期的应用机制研究[J]. 中国中药杂志, 2024, 49(20): 5598-609. |
| [17] | 唐志苓, 李 瑶, 支 颢, 等. 基于UPLC-Q-TOF/MS分析炙甘草汤体内成分及心脏组织分布特征[J]. 南京中医药大学学报, 2025, 41(7): 896-903. |
| [18] | Wang J, Wen W, Gong DY, et al. SZAP exerts analgesic effects on rheumatalgia in CIA rats by suppressing pain hyperalgesia and inhibiting TRPV1 and P2X3[J]. J Ethnopharmacol, 2022, 284: 114780. doi:10.1016/j.jep.2021.114780 |
| [19] | Samakashvili S, Ibáñez C, Simó C, et al. Analysis of chiral amino acids in cerebrospinal fluid samples linked to different stages of Alzheimer disease[J]. Electrophoresis, 2011, 32(19): 2757-64. doi:10.1002/elps.201100139 |
| [20] | Jo S, Yarishkin O, Hwang YJ, et al. GABA from reactive astrocytes impairs memory in mouse models of Alzheimer's disease[J]. Nat Med, 2014, 20(8): 886-96. doi:10.1038/nm.3639 |
| [21] | Conn KA, Borsom EM, Cope EK. Implications of microbe-derived ɣ-aminobutyric acid (GABA) in gut and brain barrier integrity and GABAergic signaling in Alzheimer's disease[J]. Gut Microbes, 2024, 16(1): 2371950. doi:10.1080/19490976.2024.2371950 |
| [22] | Kwakowsky A, Calvo-Flores Guzmán B, Pandya M, et al. GABAA receptor subunit expression changes in the human Alzheimer's disease hippocampus, subiculum, entorhinal cortex and superior temporal gyrus[J]. J Neurochem, 2018, 145(5): 374-92. doi:10.1111/jnc.14325 |
| [23] | Wu Z, Guo ZY, Gearing M, et al. Tonic inhibition in dentate gyrus impairs long-term potentiation and memory in an Alzheimer's [corrected] disease model[J]. Nat Commun, 2014, 5: 4159. doi:10.1038/ncomms5159 |
| [24] | Herbison AE. Estrogen regulation of GABA transmission in rat preoptic area[J]. Brain Res Bull, 1997, 44(4): 321-6. doi:10.1016/s0361-9230(97)00210-4 |
| [25] | Hara Y, Waters EM, McEwen BS, et al. Estrogen effects on cognitive and synaptic health over the lifecourse[J]. Physiol Rev, 2015, 95(3): 785-807. doi:10.1152/physrev.00036.2014 |
| [26] | Malyala A, Zhang CG, Bryant DN, et al. PI3K signaling effects in hypothalamic neurons mediated by estrogen[J]. J Comp Neurol, 2008, 506(6): 895-911. doi:10.1002/cne.21584 |
| [27] | Knowlton AA, Lee AR. Estrogen and the cardiovascular system[J]. Pharmacol Ther, 2012, 135(1): 54-70. doi:10.1016/j.pharmthera.2012.03.007 |
| [28] | Han X, Cheng XL, Xu JY, et al. Activation of TREM2 attenuates neuroinflammation via PI3K/Akt signaling pathway to improve postoperative cognitive dysfunction in mice[J]. Neuropharm-acology, 2022, 219: 109231. doi:10.1016/j.neuropharm.2022.109231 |
| [29] | Yang CZ, Wang SH, Zhang RH, et al. Neuroprotective effect of astragalin via activating PI3K/Akt-mTOR-mediated autophagy on APP/PS1 mice[J]. Cell Death Discov, 2023, 9(1): 15. doi:10.1038/s41420-023-01324-1 |
| [30] | Fu C, Wu YF, Liu SJ, et al. Rehmannioside A improves cognitive impairment and alleviates ferroptosis via activating PI3K/AKT/Nrf2 and SLC7A11/GPX4 signaling pathway after ischemia[J]. J Ethnopharmacol, 2022, 289: 115021. doi:10.1016/j.jep.2022.115021 |
| [31] | 刘志华, 孙晓波. 网络药理学: 中医药现代化的新机遇[J]. 药学学报, 2012, 47(6): 696-703. |
| [32] | Taleb O, Maammar M, Klein C, et al. A role for xanthurenic acid in the control of brain dopaminergic activity[J]. Int J Mol Sci, 2021, 22(13): 6974. doi:10.3390/ijms22136974 |
| [33] | Yang ZJ, Huang SY, Zhong KY, et al. Betaine alleviates cognitive impairment induced by homocysteine through attenuating NLRP3-mediated microglial pyroptosis in an m6A-YTHDF2-dependent manner[J]. Redox Biol, 2024, 69: 103026. doi:10.1016/j.redox.2024.103026 |
| [34] | Shi MM, Xu XF, Sun QM, et al. Betaine prevents cognitive dysfunction by suppressing hippocampal microglial activation in chronic social isolated male mice[J]. Phytother Res, 2023, 37(10): 4755-70. doi:10.1002/ptr.7944 |
| [1] | 许嘉蓉, 黄傲, 丁祉恺, 鲍宇, 赵仓焕, 蔡文智. 子午流注针法对失眠大鼠Glu/GABA-Gln代谢环路异常的调节作用[J]. 南方医科大学学报, 2025, 45(8): 1616-1624. |
| [2] | 曾静, 花雷, 阳勇, 张小梅, 魏江平, 李利生. 异功散通过调控脑水液代谢改善APP/PS1转基因小鼠的学习记忆能力[J]. 南方医科大学学报, 2024, 44(10): 2015-2023. |
| [3] | 曹福羊, 郭永馨, 郭舒婷, 周志康, 曹江北, 仝 黎, 米卫东. 激活小鼠ZI 区GABA 能神经元可促进七氟醚和丙泊酚的麻醉诱导而对麻醉维持及觉醒无影响[J]. 南方医科大学学报, 2023, 43(5): 718-726. |
| [4] | 杨鑫宇, 朱苏月, 靳 娜, 李 妍, 甄 骋, 张环环, 徐爱萍, 汪萌芽, 郑 超. Orexin-A通过激活OX1R、OX2R和非Ca2+依赖的PKC抑制新生大鼠脊髓腹角神经元的γ-氨基丁酸电流[J]. 南方医科大学学报, 2021, 41(5): 694-701. |
| [5] | 李 洋,李秀娟,谢明丹,程 莉,陈恒胜,孙 红,蒋 莉. 邻苯二甲酸二丁酯对原代培养海马神经元的毒性及机制[J]. 南方医科大学学报, 2020, 40(02): 225-232. |
| [6] | 郑超,黄艳,张环环,查盈盈,汪萌芽. β2-nAChR促进小鼠海马CA1和CA3锥体神经元GABAA受体的功能成熟[J]. 南方医科大学学报, 2018, 38(09): 1045-. |
| [7] | 林芳波,刘鑫,谢婧雯,罗静,奉夏露,侯德仁. SORL1基因敲除小鼠可作为散发性阿尔茨海默病模型[J]. 南方医科大学学报, 2018, 38(03): 289-. |
| [8] | 第五永长,田金洲,时晶. 洗心汤对SAD大鼠脑内tau蛋白O-GlcNAc 糖基化修饰相关酶的影响[J]. 南方医科大学学报, 2013, 33(10): 1442-. |
| [9] | 汪燕,林春水,古妙宁,郭高锋,周枝凤,陈莺. 脑摄取动态平衡时不同麻醉深度丙泊酚对犬脑不同区域γ-氨基丁酸的影响[J]. 南方医科大学学报, 2012, 32(03): 427-. |
| [10] | 张雪,黄仕营,张燊,丁秀芳,陈宝田. 安神方的抗焦虑作用[J]. 南方医科大学学报, 2012, 32(01): 80-. |
| [11] | 谢炜; 李长征; 鲍勇; 于礼建;. 戊四氮慢性点燃大鼠海马谷氨酸、γ-氨基丁酸阳性细胞变化及柴胡总皂甙的干预作用[J]. 南方医科大学学报, 2006, 26(08): 1132-1135. |
| [12] | 陈静; 黄泳; 王升旭; 李求实; 梁永就; 郭勇宁;. 针刺百会水沟神门影响血管性痴呆患者18FDGPET脑功能成像10例临床分析[J]. 南方医科大学学报, 2006, 26(05): 610-612. |
| [13] | 黄泳1, 陈静1, 赖新生2, 唐安戊3, 李东江3. 针刺百会、水沟、神门穴对血管性痴呆患者豆状核葡萄糖代谢的影响[J]. 南方医科大学学报, 2005, 25(11): 1405-1407. |
| [14] | 李玉明, 王鲁妮. 路易体痴呆误诊1例报道[J]. 南方医科大学学报, 2005, 25(11): 1364-1364. |
| [15] | 欧阳石, 孙莉莎, 郭胜蓝, 刘旭, 徐江平. 知母皂苷元对痴呆模型大鼠的影响[J]. 南方医科大学学报, 2005, 25(02): 121-126. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||