南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 374-384.doi: 10.12122/j.issn.1673-4254.2026.02.15
• • 上一篇
王煜煌1( ), 王文锐2(
), 王文锐2( ), 程淑洁1, 冯云龙1, 张卓1, 施其英1, 李雨佳1, 胡茜文1, 吴明彩1(
), 程淑洁1, 冯云龙1, 张卓1, 施其英1, 李雨佳1, 胡茜文1, 吴明彩1( )
)
收稿日期:2025-04-08
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
吴明彩
E-mail:1723221321@qq.com;wenrui-wang1983@163.com;williyia@wnmc.edu.cn
作者简介:王煜煌,在读硕士研究生,E-mail: 1723221321@qq.com基金资助:
Yuhuang WANG1( ), Wenrui WANG2(
), Wenrui WANG2( ), Shujie CHENG1, Yunlong FENG1, Zhuo ZHANG1, Qiying SHI1, Yujia LI1, Qianwen HU1, Mingcai WU1(
), Shujie CHENG1, Yunlong FENG1, Zhuo ZHANG1, Qiying SHI1, Yujia LI1, Qianwen HU1, Mingcai WU1( )
)
Received:2025-04-08
Online:2026-02-20
Published:2026-03-10
Contact:
Mingcai WU
E-mail:1723221321@qq.com;wenrui-wang1983@163.com;williyia@wnmc.edu.cn
摘要:
目的 探索槲皮素调控胃癌细胞增殖、迁移与凋亡的分子机制。 方法 生物信息学分析Hippo通路蛋白YAP在胃癌和胃癌旁正常组织的表达差异和生存率。Western blotting检测各样本中YAP和磷酸化YAP(p-YAP)的表达水平。槲皮素处理胃癌细胞,CCK8检测细胞活力;敲低YAP基因和槲皮素处理胃癌细胞进行分析比较,EdU、集落形成实验检测细胞增殖,Transwell检测细胞迁移;流式细胞术检测细胞凋亡;Western blotting 和RT-qPCR检测相关基因和蛋白表达水平;免疫荧光检测蛋白定位。过表达YAP基因进行回复实验验证。 结果 YAP在胃癌组织的表达显著高于癌旁正常组织(P<0.05);YAP的表达与胃癌患者存活率呈负相关(P<0.01);槲皮素可明显降低胃癌细胞的存活率(P<0.01),促进MST1和LATS1蛋白表达,激活Hippo信号通路,导致YAP的磷酸化失活,核转位减少(P<0.05);敲降YAP表达和槲皮素处理均能够明显抑制胃癌细胞的增殖、迁移能力,促进细胞的凋亡(P<0.05);过表达YAP能够明显降低槲皮素对胃癌细胞增殖、迁移能力的抑制作用和凋亡的促进作用(P<0.05)。 结论 槲皮素可促进YAP磷酸化降解抑制胃癌细胞的增殖和迁移,促进细胞凋亡。
王煜煌, 王文锐, 程淑洁, 冯云龙, 张卓, 施其英, 李雨佳, 胡茜文, 吴明彩. 槲皮素通过调控Hippo通路蛋白YAP表达抑制胃癌细胞增殖与迁移并促进其凋亡[J]. 南方医科大学学报, 2026, 46(2): 374-384.
Yuhuang WANG, Wenrui WANG, Shujie CHENG, Yunlong FENG, Zhuo ZHANG, Qiying SHI, Yujia LI, Qianwen HU, Mingcai WU. Quercetin inhibits proliferation and migration and promotes apoptosis of gastric cancer cells by promoting phosphorylation-mediated YAP inactivation[J]. Journal of Southern Medical University, 2026, 46(2): 374-384.
| Primer | Forward (5'-3') | Reverse (5'-3') |
|---|---|---|
| LATS1 | GTCGACGAGAGCAGATGGC | CACTTTCTCCTAGTGGCGGG |
| YAP | TGACCCTCGTTTTGCCATGA | GTTGCTGCGGTTGGAGTTG |
| CYR61 | CAGGACTGTGAAGATGCGT | AGCCTGTAGAAGGGAAACGC |
| CTGF | GTCGAGTATGTACCGACGCG | GCAGGCACAGGTCTTGATGA |
| GAPDH | CGAGCCACATCGCTCAGACA | GTGGTGAAGACGCCAGTGGA |
表1 LATS1、YAP、CYR61、CTGF和GAPDH引物序列
Tab.1 Primer sequences for RT-qPCR
| Primer | Forward (5'-3') | Reverse (5'-3') |
|---|---|---|
| LATS1 | GTCGACGAGAGCAGATGGC | CACTTTCTCCTAGTGGCGGG |
| YAP | TGACCCTCGTTTTGCCATGA | GTTGCTGCGGTTGGAGTTG |
| CYR61 | CAGGACTGTGAAGATGCGT | AGCCTGTAGAAGGGAAACGC |
| CTGF | GTCGAGTATGTACCGACGCG | GCAGGCACAGGTCTTGATGA |
| GAPDH | CGAGCCACATCGCTCAGACA | GTGGTGAAGACGCCAGTGGA |
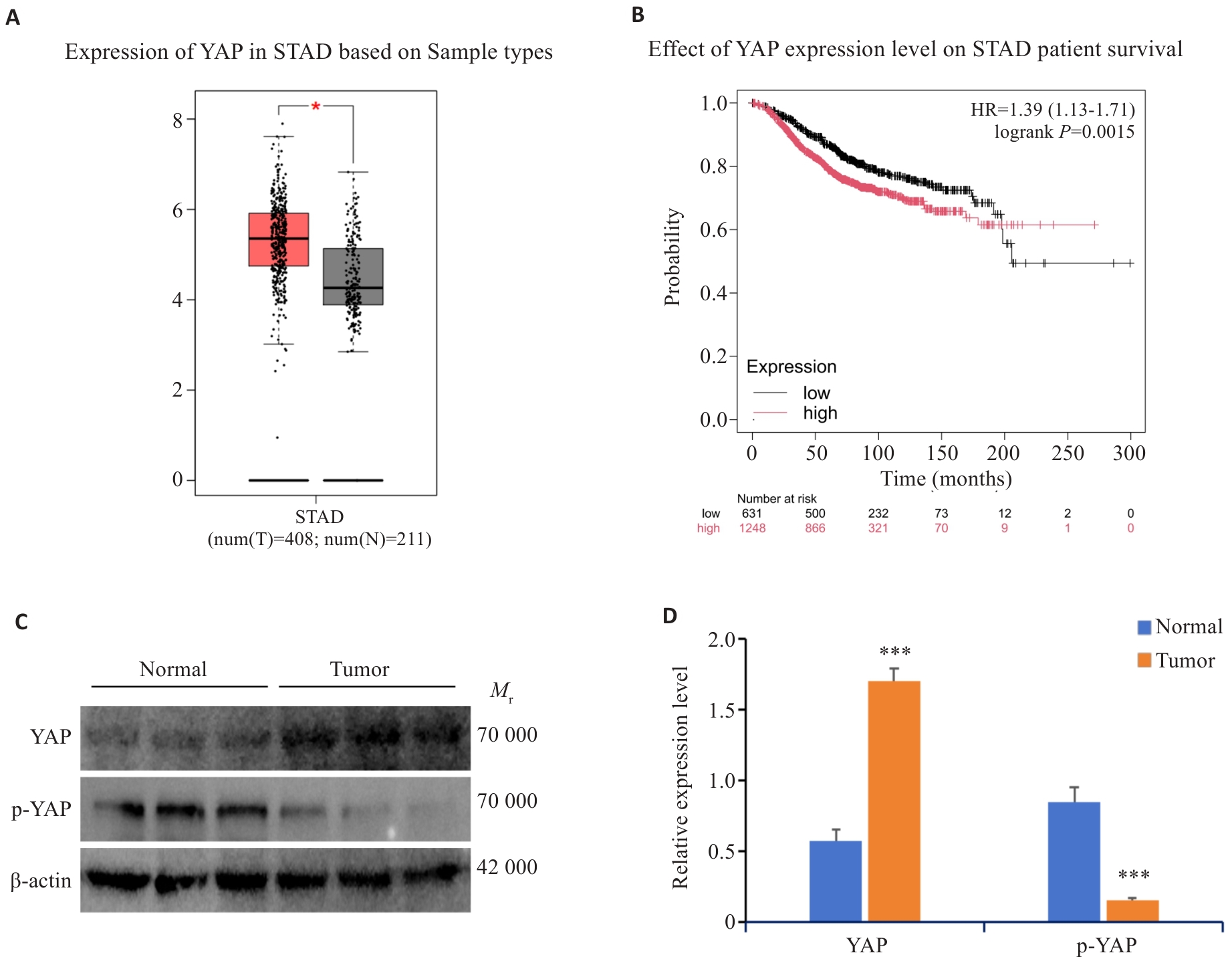
图1 YAP在胃癌组织的表达水平及YAP表达在胃癌患者存活率的关系
Fig.1 Expression levels of YAP and p-YAP in gastric cancer (GC) tissues and the correlation of YAP expression level with patient survival. A: Expression of YAP in GC (T) tissues and normal tissues (N) analyzed using the TCGA database. B: Correlation of YAP expression level with patient survival. C, D: Expression of YAP and p-YAP protein in normal gastric tissues and GC tissues. *P<0.05, ***P<0.001 vs Normal tissues.
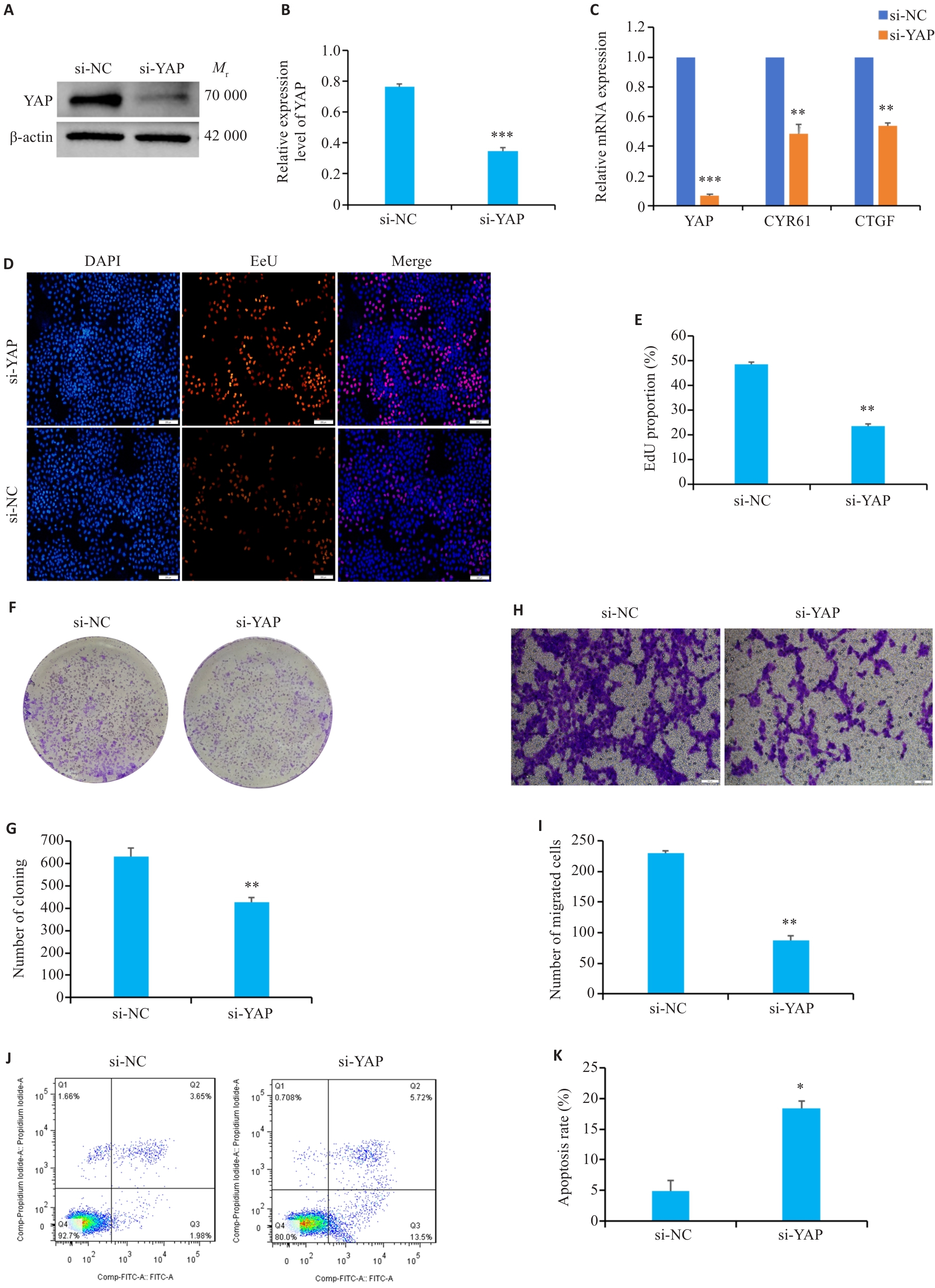
图2 敲降YAP表达对细胞增殖、迁移和凋亡的影响
Fig.2 Effects of YAP knockdown on cell proliferation, migration and apoptosis. A, B: YAP expression levels in HGC-27 cells with YAP knockdown. C: mRNA expression levels of YAP, CYR61 and CTGF in HGC-27 cells with YAP knockdown. D, E: EdU assays of HGC-27 cells with YAP knockdown (Scale bar=200 μm). F, G: Colony formation assays of HGC-27 cells with YAP knockdown. H, I: Transwell migration assays of HGC-27 cells with YAP knockdown (Scale bar=100 μm). J, K: Flow cytometric analysis of apoptosis of HGC-27 cells with YAP knockdown. *P<0.05, **P<0.01, ***P<0.001 vs si-NC group.
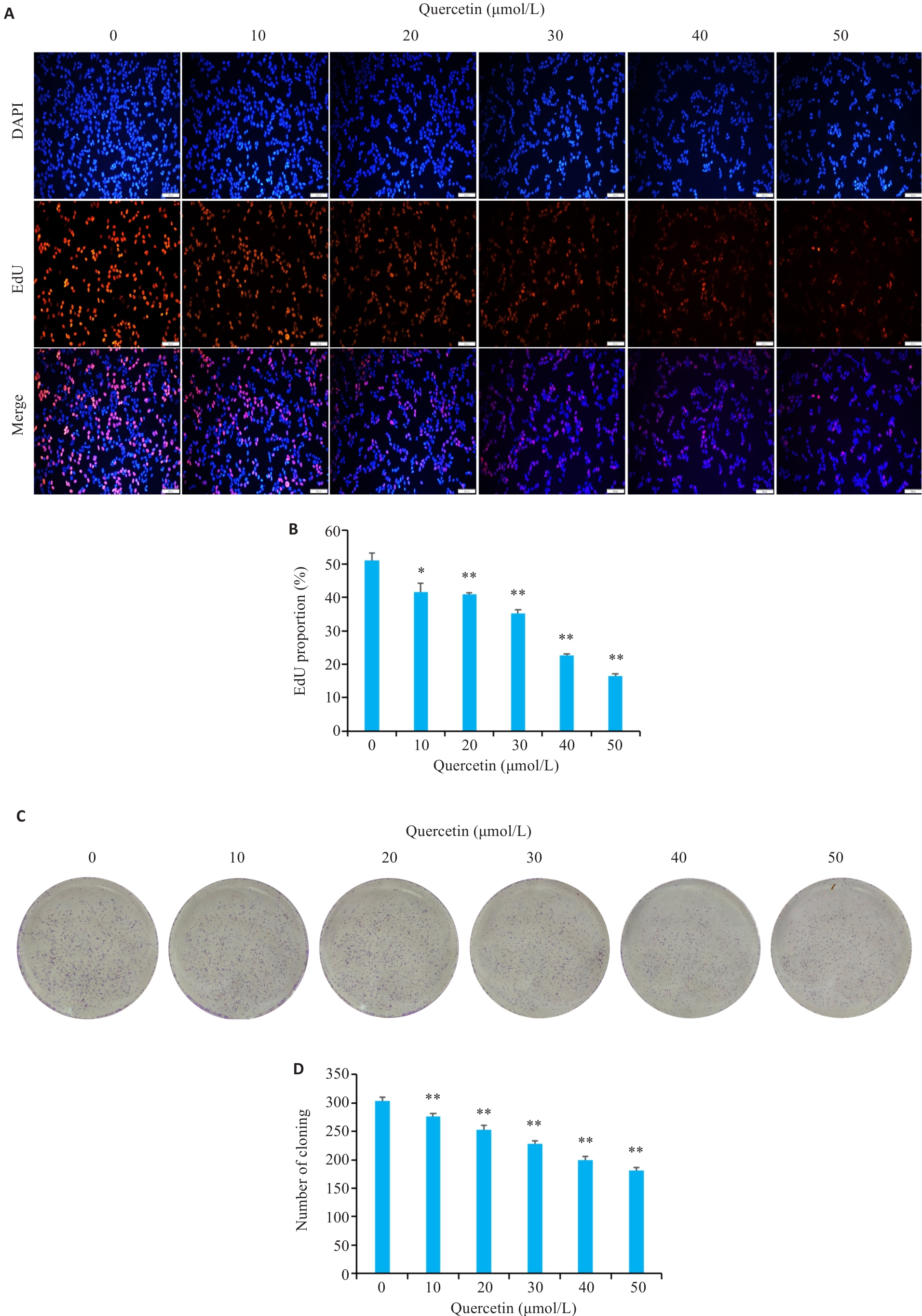
图4 槲皮素对胃癌细胞的增殖的影响
Fig.4 Effects of quercetin on proliferation of HGC-27 cells. A, B:EdU assays for assessing proliferation of quercetin-treated HGC-27 cells (Scale bar=200 μm). C, D: Colony formation assays assessing proliferation of quercetin-treated HGC-27 cells. *P<0.05, **P<0.01 vs 0 μmol/L group.
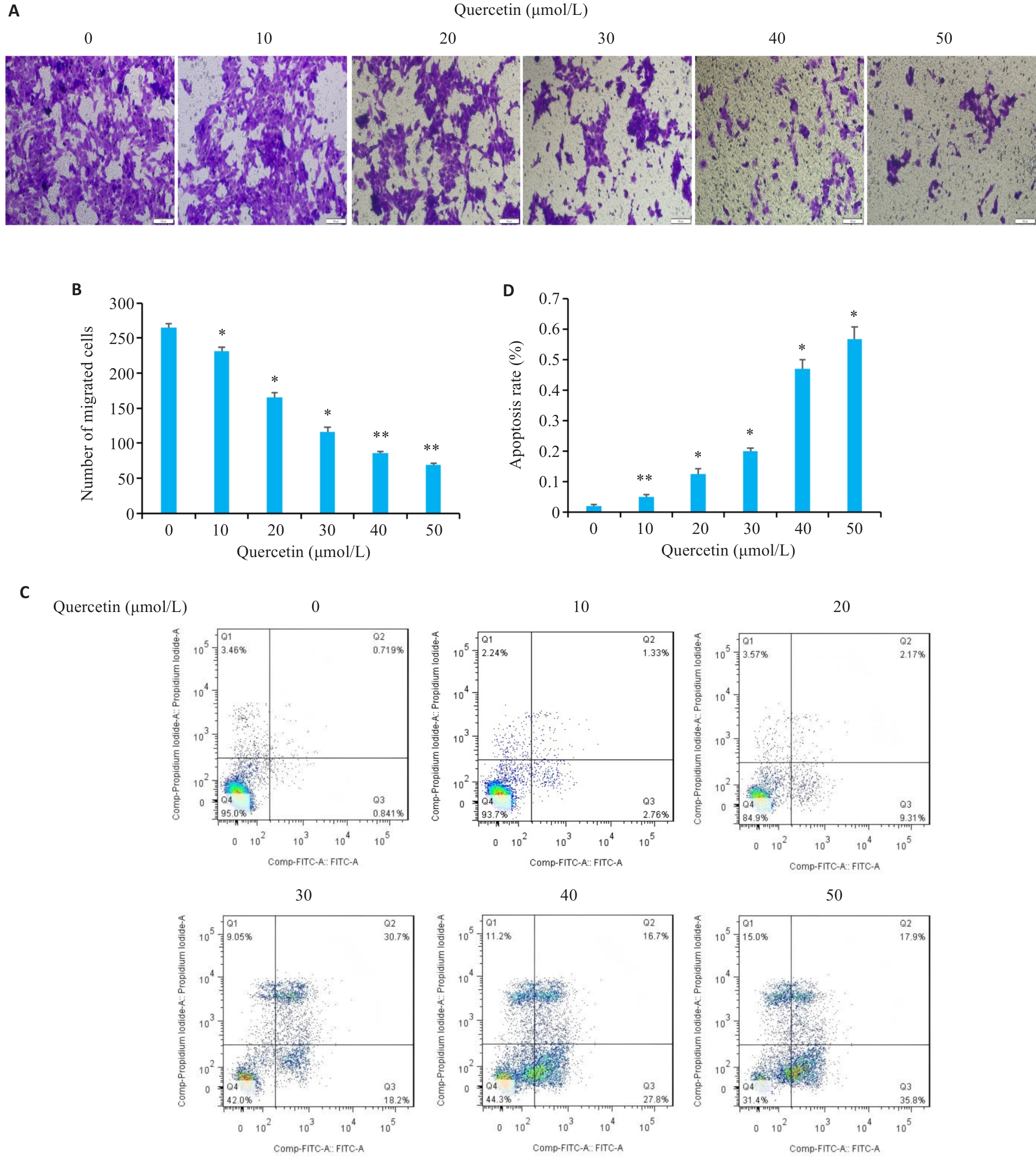
图5 槲皮素对胃癌细胞迁移和凋亡的影响
Fig.5 Effects of quercetin on migration and apoptosis of HGC-27 cells. A, B: Transwell migration assays for assessing migratory ability of quercetin-treated HGC-27 cells (Scale bar=100 μm). C, D: Flow cytometry analysis of apoptosis of quercetin on quercetin-treated HGC-27 cells. *P<0.05, **P<0.01 vs 0 μmol/L group.
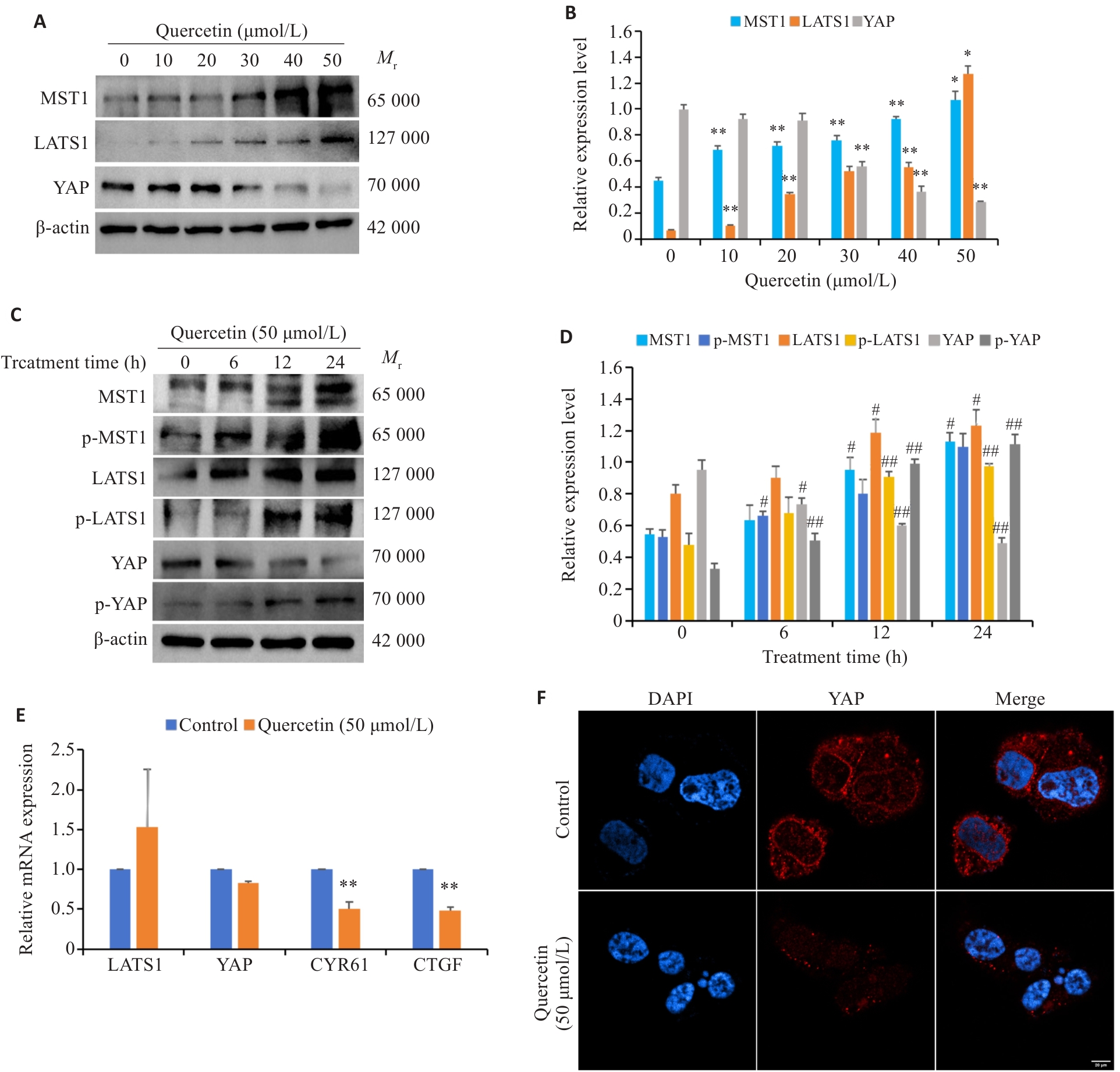
图6 槲皮素对Hippo信号通路蛋白的表达影响
Fig.6 Effect of quercetin on Hippo signaling pathway protein expression in HGC-27 cells. A, B: Effect of 0-50 μmol/L quercetin on MST1, LATS1 and YAP protein expressions in HGC-27 cells. C, D: Effects of 50 μmol/L quercetin treatment for 24 h on MST1, p-MST1 LATS1, p-LATS1, YAP and p-YAP expression. E: Effect of 50 μmol/L quercetin on mRNA expression of LATS1, YAP and its downstream target genes CYR61 and CTGF in HGC-27 cells. F: Immunofluorescence staining for analyzing YAP nuclear and cytoplasmic expression in 50 μmol/L quercetin-treated HGC-27 cells (Scale bar=20 μm).*P<0.05, **P<0.01 vs 0 μmol/L group. #P<0.05, ##P<0.01 vs 0 h group.
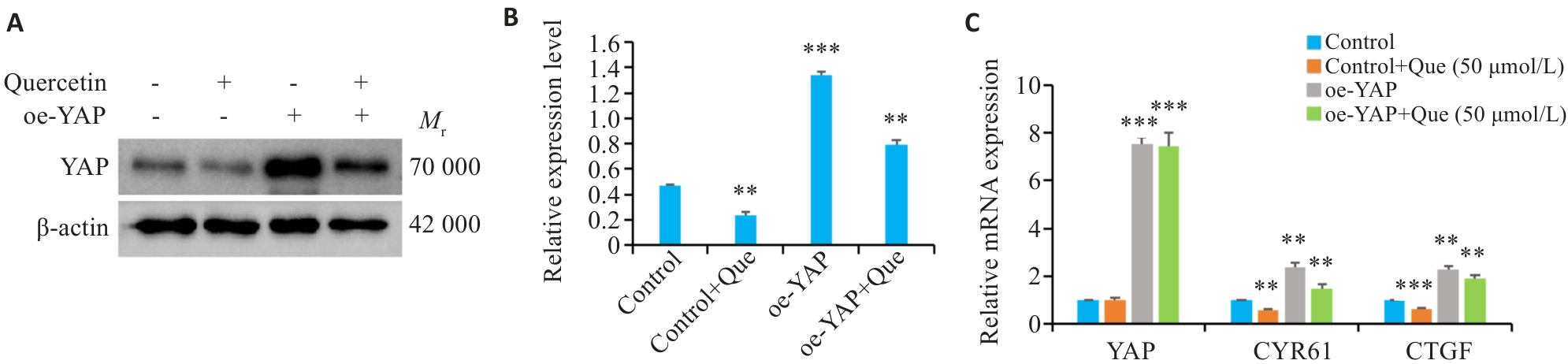
图7 过表达YAP对槲皮素作用胃癌细胞下YAP下游基因表达的影响
Fig.7 Effect of YAP overexpression on YAP downstream genes in quercetin-treated gastric cancer cells. A, B: Western blotting for analyzing the inhibitory effects of quercetin on YAP in YAP-overexpressing HGC-27 cells. C: mRNA expression of YAP and its downstream target genes CYR61 and CTGF in HGC-27 cells in Control, Control+Que (50 μmol/L), oe-YAP and oe-YAP+Que (50 μmol/L) groups. **P<0.01, ***P<0.001 vs Control group.
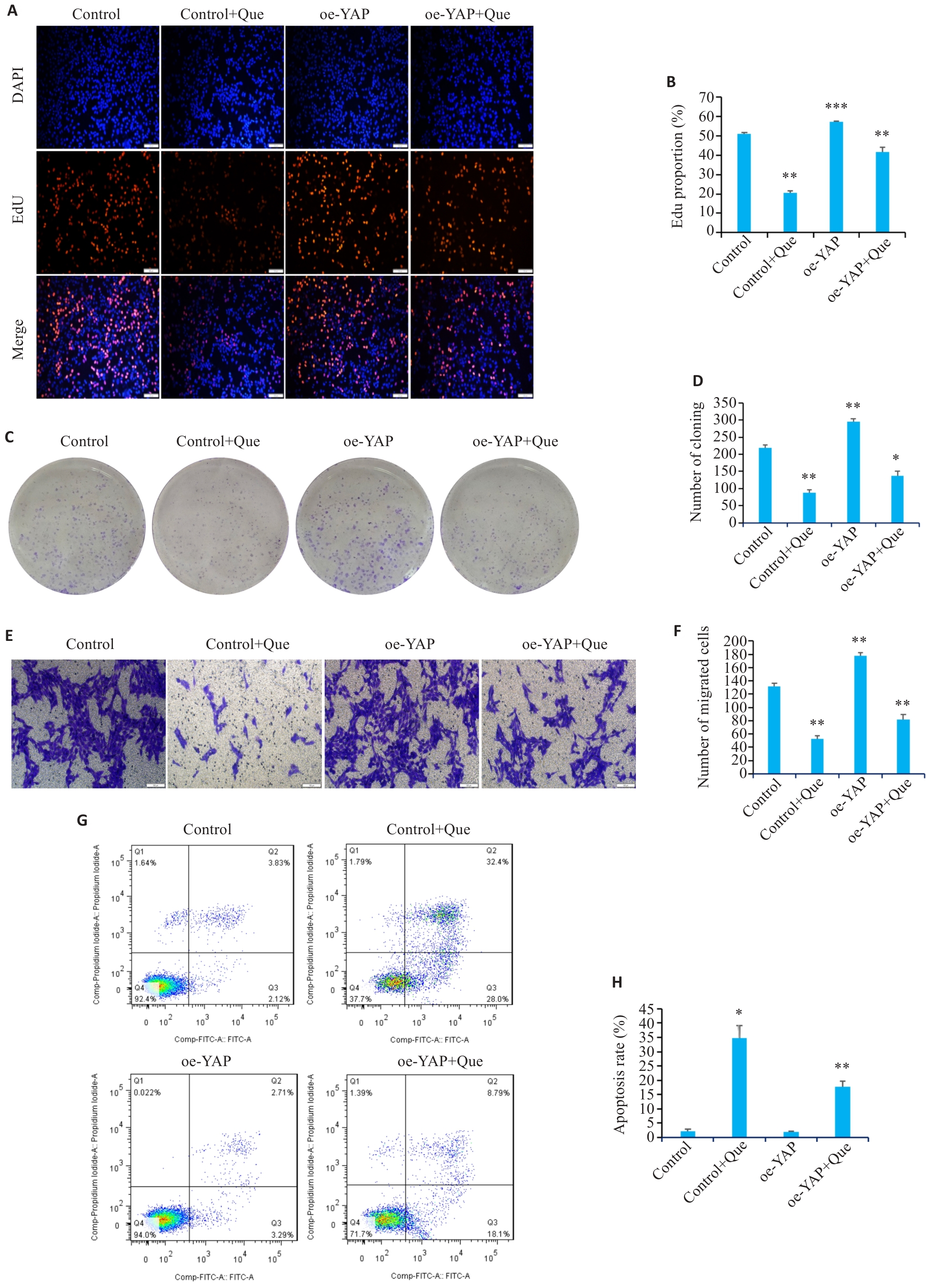
图8 过表达YAP对槲皮素作用胃癌细胞增殖、迁移和凋亡的影响
Fig.8 Effect of YAP overexpression on proliferation, migration, and apoptosis of quercetin-treated gastric cancer cells. A, B: EdU assays for assessing proliferation of quercetin-treated HGC-27 cells overexpressing YAP. C, D: Colony formation assays for assessing proliferation of quercetin-treated HGC-27 cells overexpressing YAP. E, F: Transwell migration assays of quercetin-treated HGC-27 cells overexpressing YAP (Scale bar=100 μm). G, H: Flow cytometry for assessing apoptosis of quercetin-treated HGC-27 cells overexpressing YAP. *P<0.05, **P<0.01, ***P<0.001 vs Control group.
| [1] | Zhang X, Yang L, Liu S, et al. Interpretation on the report of global cancer statistics 2022[J]. Zhonghua Zhong Liu Za Zhi Chin J Oncol, 2024, 46(7): 710-21. |
| [2] | Zheng RS, Chen R, Han BF, et al. [Cancer incidence and mortality in China, 2022]. Zhonghua Zhong Liu Za Zhi. 2024, 46(3): 221-31. |
| [3] | Christodoulidis G, Koumarelas KE, Kouliou MN, et al. Gastric cancer in the era of epigenetics[J]. Int J Mol Sci, 2024, 25(6): 3381. doi:10.3390/ijms25063381 |
| [4] | Saito M, Kono K. Landscape of EBV-positive gastric cancer[J]. Gastric Cancer, 2021, 24(5): 983-9. doi:10.1007/s10120-021-01215-3 |
| [5] | Cao ZF, An LW, Han Y, et al. The Hippo signaling pathway in gastric cancer[J]. Acta Biochim Biophys Sin (Shanghai), 2023, 55(6): 893-903. |
| [6] | Ma YL, Yang YZ, Wang F, et al. Hippo-YAP signaling pathway: a new paradigm for cancer therapy[J]. Int J Cancer, 2015, 137(10): 2275-86. doi:10.1002/ijc.29073 |
| [7] | Zhu J, Wu TT, Lin Q. Non-hippo kinases: indispensable roles in YAP/TAZ signaling and implications in cancer therapy[J]. Mol Biol Rep, 2023, 50(5): 4565-78. doi:10.1007/s11033-023-08329-0 |
| [8] | Leask A, Nguyen J, Naik A, et al. The role of yes activated protein (YAP) in melanoma metastasis[J]. iScience, 2024, 27(6): 109864. doi:10.1016/j.isci.2024.109864 |
| [9] | Fu MY, Hu Y, Lan TX, et al. The Hippo signalling pathway and its implications in human health and diseases[J]. Signal Transduct Target Ther, 2022, 7(1): 376. doi:10.1038/s41392-022-01191-9 |
| [10] | Akrida I, Bravou V, Papadaki H. The deadly cross-talk between Hippo pathway and epithelial–mesenchymal transition (EMT) in cancer[J]. Mol Biol Rep, 2022, 49(10): 10065-76. doi:10.1007/s11033-022-07590-z |
| [11] | Bustamante A, Baritaki S, Zaravinos A, et al. Relationship of signaling pathways between RKIP expression and the inhibition of EMT-inducing transcription factors SNAIL1/2, TWIST1/2 and ZEB1/2[J]. Cancers (Basel), 2024, 16(18): 3180. doi:10.3390/cancers16183180 |
| [12] | Zhao AY, Dai YJ, Lian JF, et al. YAP regulates ALDH1A1 expression and stem cell property of bladder cancer cells[J]. Onco Targets Ther, 2018, 11: 6657-63. doi:10.2147/ott.s170858 |
| [13] | Cai J, Shi SY, Cheng F, et al. Polyacetylene isomers isolated from Bidens pilosa L. suppress the metastasis of gastric cancer cells by inhibiting Wnt/β‑catenin and hippo/YAP signaling pathways[J]. Molecules, 2023, 28(4): 1837. doi:10.3390/molecules28041837 |
| [14] | Driskill JH, Pan DJ. The hippo pathway in liver homeostasis and pathophysiology[J]. Annu Rev Pathol, 2021, 16: 299-322. doi:10.1146/annurev-pathol-030420-105050 |
| [15] | Andres S, Pevny S, Ziegenhagen R, et al. Safety aspects of the use of quercetin as a dietary supplement[J]. Mol Nutr Food Res, 2018, 62(1). DOI: 10.1002/mnfr.201700447 . |
| [16] | Özsoy Gökbilen S, Becer E, Vatansever HS. Senescence-mediated anticancer effects of quercetin[J]. Nutr Res, 2022, 104: 82-90. doi:10.1016/j.nutres.2022.04.007 |
| [17] | Goyal A, Agrawal N. Quercetin: a potential candidate for the treatment of arthritis[J]. Curr Mol Med, 2022, 22(4): 325-35. doi:10.2174/1566524021666210315125330 |
| [18] | Popiolek-Kalisz J, Fornal E. The effects of quercetin supplem-entation on blood pressure-meta-analysis[J]. Curr Probl Cardiol, 2022, 47(11): 101350. doi:10.1016/j.cpcardiol.2022.101350 |
| [19] | Li T, Li YJ. Quercetin acts as a novel anti-cancer drug to suppress cancer aggressiveness and cisplatin-resistance in nasopharyngeal carcinoma (NPC) through regulating the yes-associated protein/Hippo signaling pathway[J]. Immunobiology, 2023, 228(2): 152324. doi:10.1016/j.imbio.2022.152324 |
| [20] | López MJ, Carbajal J, Alfaro AL, et al. Characteristics of gastric cancer around the world[J]. Crit Rev Oncol Hematol, 2023, 181: 103841. doi:10.1016/j.critrevonc.2022.103841 |
| [21] | Li X, Zhuo S, Cho YS, et al. YAP antagonizes TEAD-mediated AR signaling and prostate cancer growth[J]. EMBO J, 2023, 42(4): e112184. doi:10.15252/embj.2022112184 |
| [22] | Yan C, Yang HJ, Su P, et al. OTUB1 suppresses Hippo signaling via modulating YAP protein in gastric cancer[J]. Oncogene, 2022, 41(48): 5186-98. doi:10.1038/s41388-022-02507-3 |
| [23] | Yong JX, Li Y, Lin SH, et al. Inhibitors targeting YAP in gastric cancer: current status and future perspectives[J]. Drug Des Devel Ther, 2021, 15: 2445-56. doi:10.2147/dddt.s308377 |
| [24] | Udaya RR, Sangeetha D. Therapeutic potentials and targeting strategies of quercetin on cancer cells: Challenges and future prospects[J]. Phytomedicine, 2024, 133: 155902. doi:10.1016/j.phymed.2024.155902 |
| [25] | Ding LX, Dang SW, Sun MJ, et al. Quercetin induces ferroptosis in gastric cancer cells by targeting SLC1A5 and regulating the p-Camk2/p-DRP1 and NRF2/GPX4 Axes[J]. Free Radic Biol Med, 2024, 213: 150-63. doi:10.1016/j.freeradbiomed.2024.01.002 |
| [26] | Wu HY, Pan LL, Gao CX, et al. Quercetin inhibits the proliferation of glycolysis-addicted HCC cells by reducing hexokinase 2 and Akt-mTOR pathway[J]. Molecules, 2019, 24(10): 1993. doi:10.3390/molecules24101993 |
| [27] | 马科文, 赵 强, 代春燕, 等. 中药抑制胃癌血管生成的作用机制研究进展[J]. 世界中医药, 2025, 20(7): 1233-9. |
| [28] | Abdel-Rahman O. Targeting vascular endothelial growth factor (VEGF) pathway in gastric cancer: preclinical and clinical aspects[J]. Crit Rev Oncol Hematol, 2015, 93(1): 18-27. doi:10.1016/j.critrevonc.2014.05.012 |
| [29] | Li FT, Bai YJ, Zhao M, et al. Quercetin inhibits vascular endothelial growth factor-induced choroidal and retinal angiogenesis in vitro [J]. Ophthalmic Res, 2015, 53(3): 109-16. doi:10.1159/000369824 |
| [30] | Ruan HR, Zhu TT, Wang TT, et al. Quercetin modulates ferroptosis via the SIRT1/nrf-2/HO-1 pathway and attenuates cartilage destruction in an osteoarthritis rat model[J]. Int J Mol Sci, 2024, 25(13): 7461. doi:10.3390/ijms25137461 |
| [31] | 潘小娟. 槲皮素通过Hippo-Yap信号通路对乳腺癌细胞的影响及机制研究[D]. 北京: 北京中医药大学, 2023. |
| [32] | Chestnut C, Subramaniam D, Dandawate P, et al. Targeting major signaling pathways of bladder cancer with phytochemicals: a review[J]. Nutr Cancer, 2021, 73(11/12): 2249-71. doi:10.1080/01635581.2020.1856895 |
| [1] | 李艳宇, 李晨, 戴传君, 郭润之, 韩浩宇, 卢林明, 周芳芳, 支慧. 皖南蝮蛇抑瘤组分-Ι通过调控RAI14抑制顺铂耐药胃癌细胞的增殖与侵袭[J]. 南方医科大学学报, 2026, 46(1): 113-121. |
| [2] | 骆碧云, 易欣, 蔡怡静, 张世卿, 王鹏, 李彤, 翁建霖, 周平正. 清心牛黄丸通过改善脂质代谢紊乱缓解小鼠非酒精性脂肪性肝病[J]. 南方医科大学学报, 2025, 45(9): 1840-1849. |
| [3] | 王子良, 陈孝华, 杨晶晶, 严晨, 张志郅, 黄炳轶, 赵萌, 刘嵩, 葛思堂, 左芦根, 陈德利. 高表达SURF4通过抑制紧密连接蛋白表达促进胃癌细胞的恶性生物学行为[J]. 南方医科大学学报, 2025, 45(8): 1732-1742. |
| [4] | 陈鑫源, 吴成挺, 李瑞迪, 潘雪芹, 张耀丹, 陶俊宇, 林才志. 双术汤通过P53/SLC7A11/GPX4通路诱导胃癌细胞铁死亡[J]. 南方医科大学学报, 2025, 45(7): 1363-1371. |
| [5] | 吴璇, 方家敏, 韩玮玮, 陈琳, 孙菁, 金齐力. 高表达PRELID1促进胃癌细胞上皮间质转化并与不良预后相关[J]. 南方医科大学学报, 2025, 45(7): 1535-1542. |
| [6] | 侯鑫睿, 张振东, 曹明远, 杜予心, 王小平. 红景天苷靶向miR-1343-3p-OGDHL/PDHB糖代谢轴抑制胃癌细胞的体内外增殖[J]. 南方医科大学学报, 2025, 45(6): 1226-1239. |
| [7] | 张毅, 沈昱, 万志强, 陶嵩, 柳亚魁, 王栓虎. CDKN3高表达促进胃癌细胞的迁移和侵袭:基于调控p53/NF-κB信号通路和抑制胃癌细胞凋亡[J]. 南方医科大学学报, 2025, 45(4): 853-861. |
| [8] | 孙红燕, 卢国庆, 付程文, 徐梦文, 朱小翌, 邢国权, 刘乐强, 柯雨菲, 崔乐妹, 陈睿旸, 王磊, 康品方, 唐碧. 槲皮素通过调控L型钙通道改善糖尿病大鼠心肌损伤[J]. 南方医科大学学报, 2025, 45(3): 531-541. |
| [9] | 黄晴晴, 张文静, 张小凤, 王炼, 宋雪, 耿志军, 左芦根, 王月月, 李静, 胡建国. 高表达MYO1B促进胃癌细胞增殖、迁移和侵袭并与患者的不良预后有关[J]. 南方医科大学学报, 2025, 45(3): 622-631. |
| [10] | 宋雪, 陈悦, 张敏, 张诺, 左芦根, 李静, 耿志军, 张小凤, 王月月, 王炼, 胡建国. GPSM2在胃癌组织中高表达并通过促进肿瘤细胞的增殖影响患者预后[J]. 南方医科大学学报, 2025, 45(2): 229-238. |
| [11] | 黄鹏伟, 陈洁, 邹金虎, 高雪锋, 曹虹. 槲皮素促进应激颗粒G3BP1解聚改善HIV-1 gp120诱导的星形胶质细胞神经毒性[J]. 南方医科大学学报, 2025, 45(2): 304-312. |
| [12] | 高俊杰, 叶开, 吴竞. 槲皮素通过调控TP53基因抑制肾透明细胞癌的增殖和迁移[J]. 南方医科大学学报, 2025, 45(2): 313-321. |
| [13] | 周仁杰, 杨晶晶, 宋博文, 陈孝华, 王炼, 王月月, 左芦根, 朱冰. 高表达PSMD11促进胃癌细胞上皮-间质转化进程并影响患者预后[J]. 南方医科大学学报, 2025, 45(12): 2747-2755. |
| [14] | 宋博文, 周仁杰, 徐盈, 施金冉, 张志郅, 李静, 耿志军, 宋雪, 王炼, 王月月, 左芦根. TMCO1在胃癌中高表达与患者不良预后相关并通过抑制调亡促进肿瘤恶性进展[J]. 南方医科大学学报, 2025, 45(11): 2385-2393. |
| [15] | 姜雪凝, 黄晴晴, 徐盈, 王舜印, 张小凤, 王炼, 王月月, 左芦根. 高表达YEATS2通过激活Wnt/β-catenin通路促进胃癌细胞上皮-间质转化进程[J]. 南方医科大学学报, 2025, 45(11): 2416-2426. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||