南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 316-324.doi: 10.12122/j.issn.1673-4254.2026.02.09
• • 上一篇
彭煜策( ), 姜毅, 马丹, 何岸, 吕鼎一(
), 姜毅, 马丹, 何岸, 吕鼎一( ), 罗明昊(
), 罗明昊( ), 罗素新(
), 罗素新( )
)
收稿日期:2025-07-08
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
吕鼎一,罗明昊,罗素新
E-mail:Yuce1999@163.com;dingyi.lyu@hospital.cqmu.edu.cn;luominghao001@foxmail.com;luosuxin0204@163.com
作者简介:彭煜策,在读博士研究生,E-mail: Yuce1999@163.com
基金资助:
Yuce PENG( ), Yi JIANG, Dan MA, An HE, Dingyi LÜ(
), Yi JIANG, Dan MA, An HE, Dingyi LÜ( ), Minghao LUO(
), Minghao LUO( ), Suxin LUO(
), Suxin LUO( )
)
Received:2025-07-08
Online:2026-02-20
Published:2026-03-10
Contact:
Dingyi Lü, Minghao LUO, Suxin LUO
E-mail:Yuce1999@163.com;dingyi.lyu@hospital.cqmu.edu.cn;luominghao001@foxmail.com;luosuxin0204@163.com
Supported by:摘要:
目的 研究异牡荆素(ISO)对糖尿病小鼠心肌的保护作用及机制。 方法 选用成年雄性C57小鼠18只,随机分为对照组、糖尿病组(DM)和DM+ISO组(n=6)。分离新生小鼠原代心肌细胞(NMCMs),分为对照组、高糖组(HG,33 mmol/L葡萄糖)、HG+ISO组及HG+ISO+3-TYP组(SIRT3抑制剂)。HE染色评估心肌损伤;ELISA检测心肌丙二醛(MDA)、超氧化物歧化酶(SOD)、谷胱甘肽过氧化物酶(GSH-Px)水平;免疫组化和荧光检测iNOS、NOX2、8-OHdG表达;Western blotting分析NRF2、NOX2、NQO1、SIRT3等通路蛋白;流式细胞术定量活性氧水平。 结果 与对照组相比,DM组心肌细胞炎症浸润增加,白细胞介素-1β(IL-1β),白细胞介素-6(IL-6),α-肿瘤坏死因子(TNF-α)水平升高(P<0.01),NQO1,NRF2,SIRT3蛋白水平下降(P<0.01),NOX2,AC-SOD2蛋白水平增加(P<0.01),与DM组相比,DM+ISO组心肌细胞炎症浸润减少,IL-1β,IL-6,TNF-α水平下降(P<0.01),NQO1,NRF2,SIRT3蛋白水平恢复(P<0.01),NOX2,AC-SOD2蛋白水平减少(P<0.01)。 结论 ISO可通过促进SIRT3表达,有效减轻氧化应激来减缓糖尿病心肌损伤,从而显著改善糖尿病心肌病。
彭煜策, 姜毅, 马丹, 何岸, 吕鼎一, 罗明昊, 罗素新. 异牡荆素通过促进SIRT3表达减轻糖尿病小鼠的心肌氧化应激损伤[J]. 南方医科大学学报, 2026, 46(2): 316-324.
Yuce PENG, Yi JIANG, Dan MA, An HE, Dingyi LÜ, Minghao LUO, Suxin LUO. Isovitexin alleviates myocardial oxidative stress injury in diabetic mice by enhancing myocardial SIRT3 expression and reducing oxidative stress[J]. Journal of Southern Medical University, 2026, 46(2): 316-324.
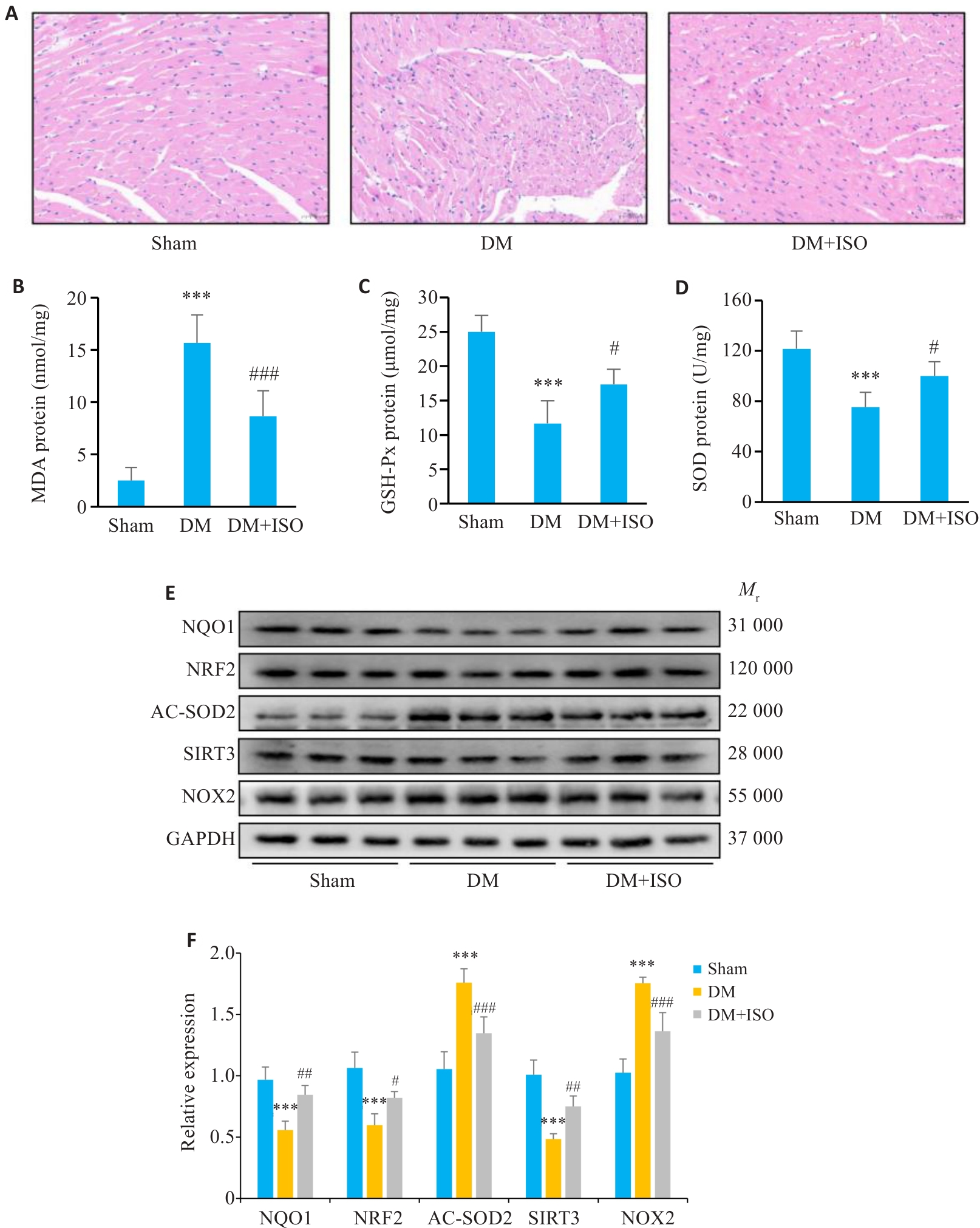
图1 ISO减轻糖尿病小鼠心肌损伤
Fig.1 Isovitexin (ISO) alleviates myocardial injury in diabetic mice. A: HE staining of the myocardial tissues of mice (Scale bar=50 μm). B: MDA content in mouse myocardial tissue (n=6). C: GSH-Px content in mouse myocardial tissue (n=6). D: SOD content in mouse myocardial tissue (n=6). E, F: Expression levels of NQO1, NRF2, AC-SOD2, SIRT3, and NOX2 proteins detected by Western blotting (n=4). DM: Diabetes mellitus. ***P<0.001 vs Sham group; #P<0.05, ##P<0.01, ###P<0.001 vs DM group.
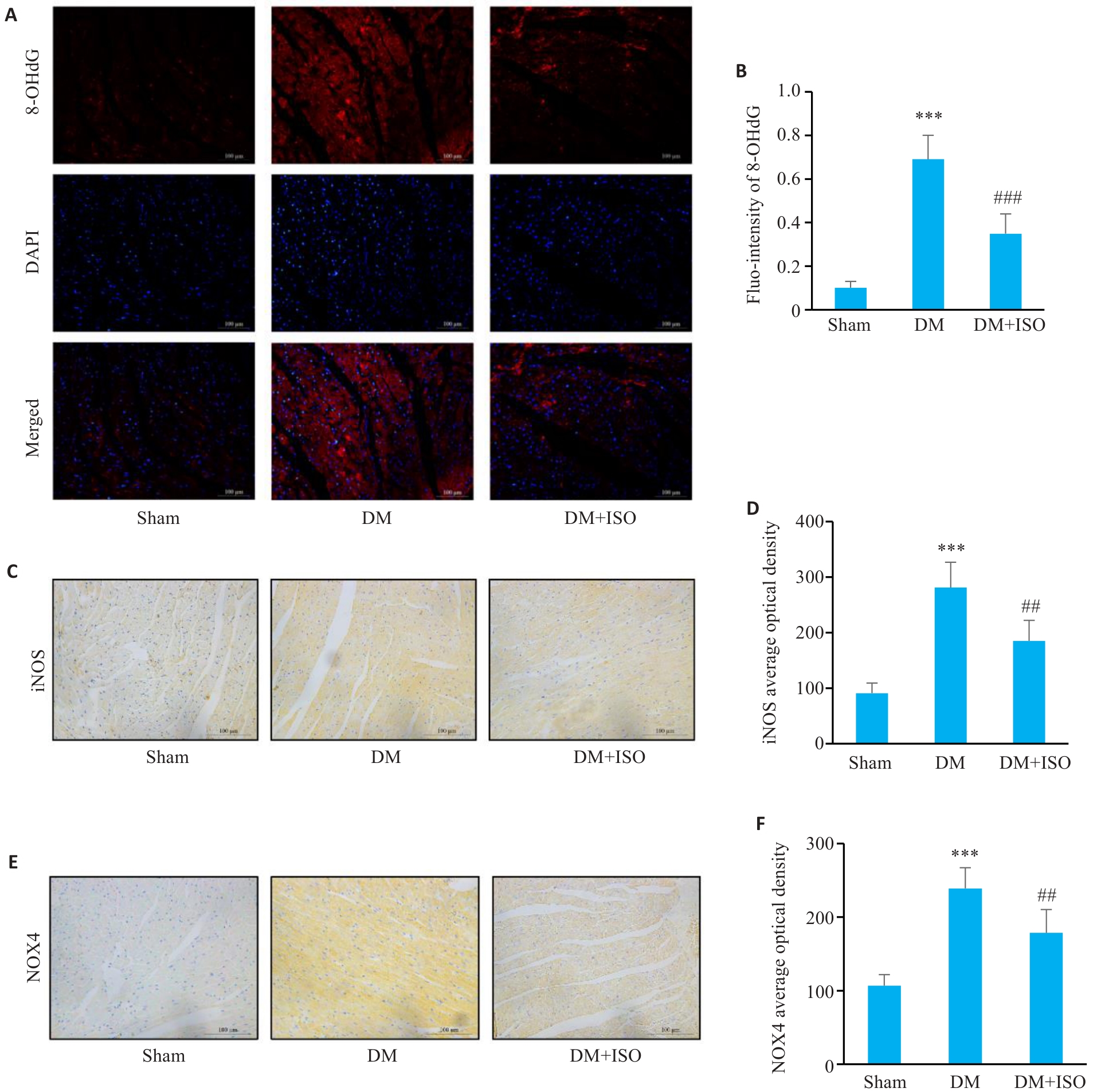
图2 ISO减轻糖尿病小鼠心肌氧化应激
Fig.2 ISO alleviates myocardial oxidative stress in diabetic mice. A: Expression and location of 8-OHdG determined by immunofluorescence staining (Scale bar=100 μm). B: Statistical analysis of fluorescence intensity (n=6). C: Expression and location of iNOS determined by immunohistochemistry (Scale bar=100 μm). D: Statistical analysis of iNOS-positive area (n=6). E: Expression and location of NOX4 determined by immunohistochemistry (Scale bar=100 μm). F: Statistical analysis of NOX4-positive area (n=6). ***P<0.001 vs Sham group; ##P<0.01, ###P<0.001 vs DM group.
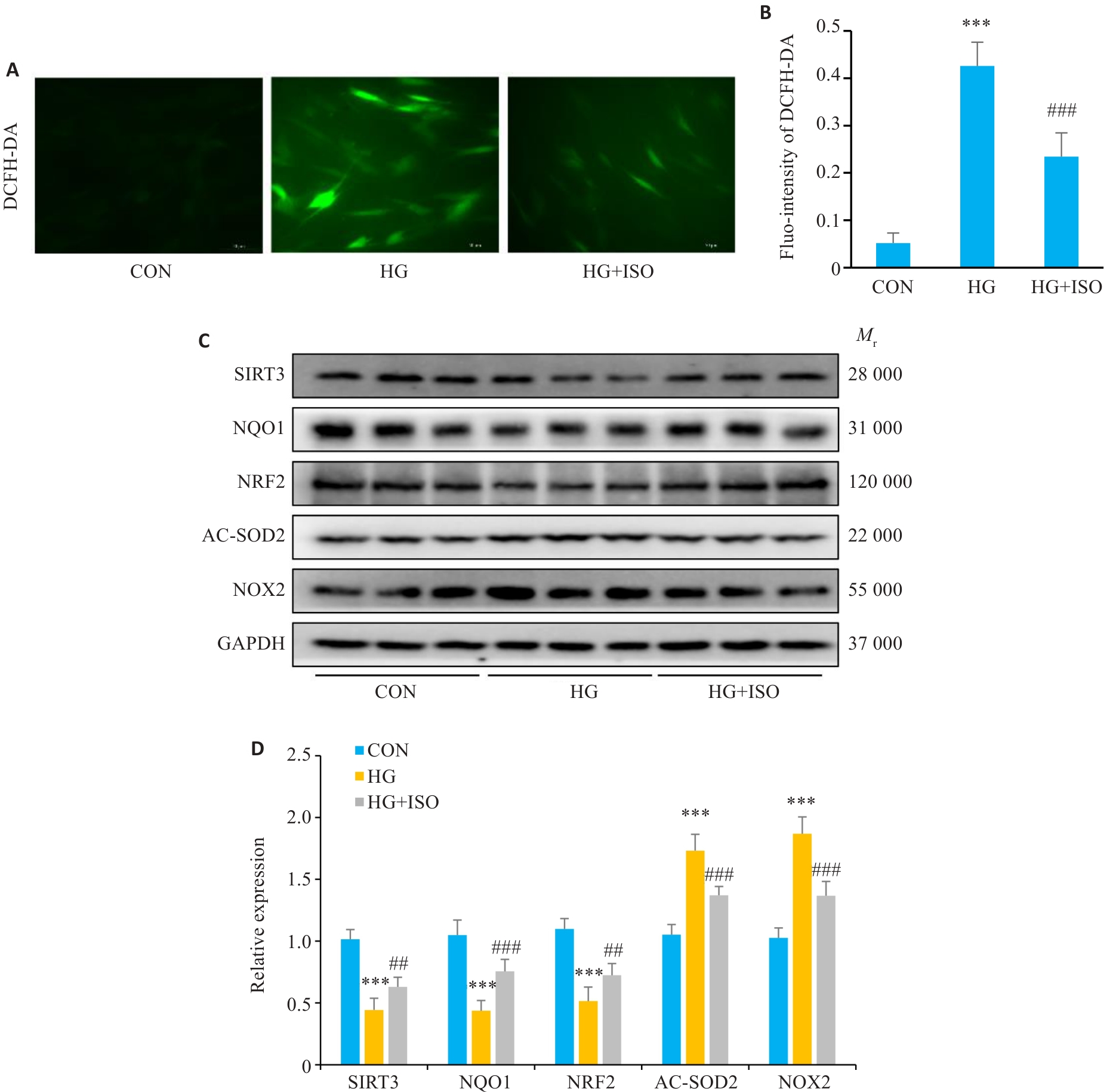
图3 ISO减轻新生小鼠心肌细胞氧化应激
Fig.3 ISO alleviates oxidative stress in primary cultures of neonatal mouse cardiomyocytes (NMCMs). A: Intracellular ROS levels determined by immunofluorescence staining (Scale bar=50 μm). B: Statistical analysis of fluorescence intensity (n=6). C, D: Expression levels of SIRT3, NQO1, AC-SOD2, NRF2, and NOX2 proteins detected by Western blotting (n=6). ***P<0.001 vs CON group; ##P<0.01, ###P<0.001 vs HG group.
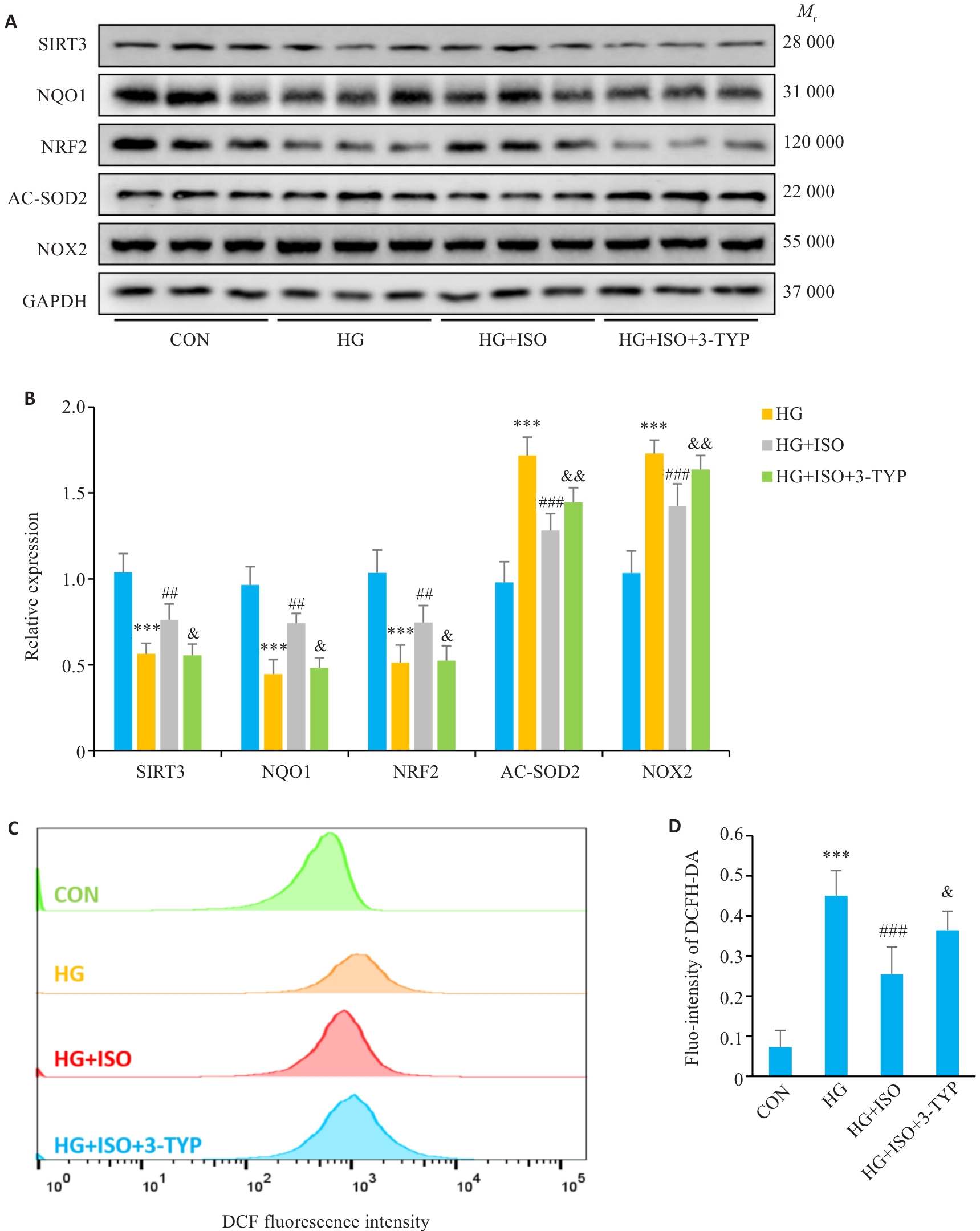
图4 ISO通过激活SIRT3在体外发挥保护作用
Fig.4 ISO protects NMCMs against high glucose-induced injury by activating SIRT3. A, B: Expression levels of SIRT3, NQO1, AC-SOD2, NRF2, and NOX2 detected by Western blotting (n=6). C, D: Flow cytometry for quantitative analysis of cellular oxidative stress damage (n=6). ***P<0.001 vs CON group; ##P<0.01, ###P<0.001 vs HG group; &P<0.05, &&P<0.01 vs HG+ISO group.
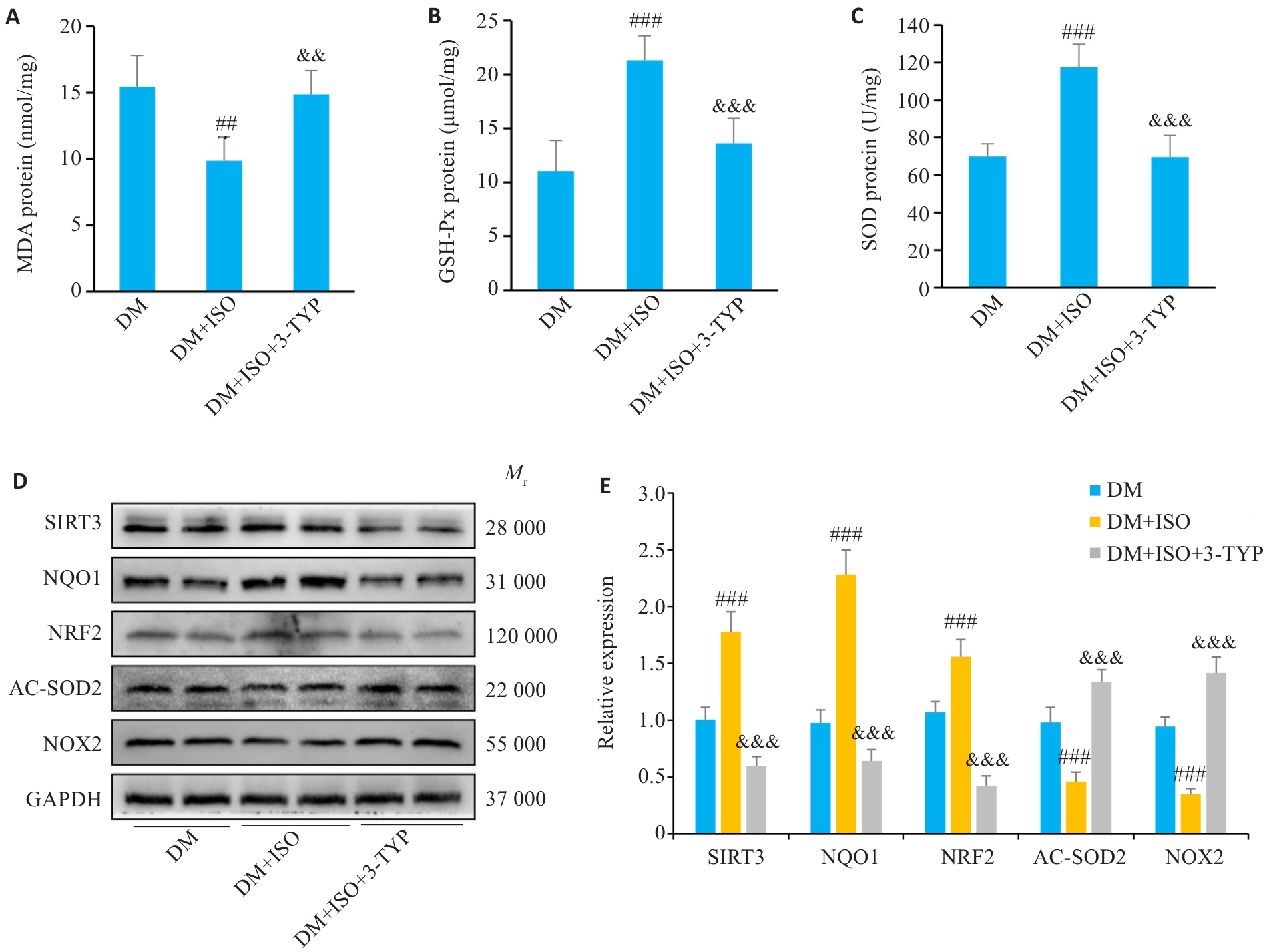
图5 ISO通过激活SIRT3在体内发挥保护作用
Fig5 ISO protects myocardial tissue of diabetic mice against oxidative stress by activating SIRT3. A: MDA content in mouse myocardial tissues (n=6). B: GSH-Px content in mouse myocardial tissues (n=6). C: SOD content in mouse myocardial tissue (n=6). D: Expression levels of NQO1, NRF2, AC-SOD2, SIRT3, and NOX2 proteins in mouse myocardial tissues detected using Western blotting (n=6). Comparisons were performed using one-way ANOVA for multiple groups. ##P<0.01, ###P<0.001 vs DM group; &&P<0.01, &&&P<0.001 vs DM+ISO group.
| [1] | Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9(th) edition [J]. Diabetes Res Clin Pract, 2019, 157: 107843. doi:10.1016/j.diabres.2019.107843 |
| [2] | Qin FZ, Siwik DA, Luptak I, et al. The polyphenols resveratrol and S17834 prevent the structural and functional sequelae of diet-induced metabolic heart disease in mice[J]. Circulation, 2012, 125(14): 1757-64, S1-6. doi:10.1161/circulationaha.111.067801 |
| [3] | Kang QZ, Yang CX. Oxidative stress and diabetic retinopathy: molecular mechanisms, pathogenetic role and therapeutic implications[J]. Redox Biol, 2020, 37: 101799. doi:10.1016/j.redox.2020.101799 |
| [4] | Zhang GY, Wang XD, Li C, et al. Integrated stress response couples mitochondrial protein translation with oxidative stress control[J]. Circulation, 2021, 144(18): 1500-15. doi:10.1161/circulationaha.120.053125 |
| [5] | Heinke L. Mitochondrial ROS drive cell cycle progression[J]. Nat Rev Mol Cell Biol, 2022, 23(9): 581. doi:10.1038/s41580-022-00523-5 |
| [6] | Jin LG, Geng LL, Ying L, et al. FGF21-sirtuin 3 axis confers the protective effects of exercise against diabetic cardiomyopathy by governing mitochondrial integrity[J]. Circulation, 2022, 146(20): 1537-57. doi:10.1161/circulationaha.122.059631 |
| [7] | Lv HM, Yu ZX, Zheng YW, et al. Isovitexin exerts anti-inflammatory and anti-oxidant activities on lipopolysaccharide-induced acute lung injury by inhibiting MAPK and NF‑κB and activating HO-1/Nrf2 pathways[J]. Int J Biol Sci, 2016, 12(1): 72-86. doi:10.7150/ijbs.13188 |
| [8] | Lin JQ, Ran H, Feng QQ, et al. Unveiling the differences between vitexin and isovitexin: from the perspective of sources, green advanced extraction technologies, biological activities, and safety[J]. Food Chem, 2025, 485: 144600. doi:10.1016/j.foodchem.2025.144600 |
| [9] | He M, Min JW, Kong WL, et al. A review on the pharmacological effects of vitexin and isovitexin[J]. Fitoterapia, 2016, 115: 74-85. doi:10.1016/j.fitote.2016.09.011 |
| [10] | Peng F, Liao MR, Jin WK, et al. 2-APQC, a small-molecule activator of Sirtuin-3 (SIRT3), alleviates myocardial hypertrophy and fibrosis by regulating mitochondrial homeostasis[J]. Signal Transduct Target Ther, 2024, 9(1): 133. doi:10.1038/s41392-024-01816-1 |
| [11] | Zhang J, Ye J, Zhu SO, et al. Context-dependent role of SIRT3 in cancer[J]. Trends Pharmacol Sci, 2024, 45(2): 173-90. doi:10.1016/j.tips.2023.12.005 |
| [12] | Palomer X, Román-Azcona MS, Pizarro-Delgado J, et al. SIRT3-mediated inhibition of FOS through histone H3 deacetylation prevents cardiac fibrosis and inflammation[J]. Signal Transduct Target Ther, 2020, 5(1): 14. doi:10.1038/s41392-020-0114-1 |
| [13] | Gao P, You M, Li L, et al. Salt-induced hepatic inflammatory memory contributes to cardiovascular damage through epigenetic modulation of SIRT3[J]. Circulation, 2022, 145(5): 375-91. doi:10.1161/circulationaha.121.055600 |
| [14] | Jheng JR, Bai Y, Noda K, et al. Skeletal muscle SIRT3 deficiency contributes to pulmonary vascular remodeling in pulmonary hypertension due to heart failure with preserved ejection fraction[J]. Circulation, 2024, 150(11): 867-83. doi:10.1161/circulationaha.124.068624 |
| [15] | Xie SY, Liu SQ, Zhang T, et al. USP28 serves as a key suppressor of mitochondrial morphofunctional defects and cardiac dysfunction in the diabetic heart[J]. Circulation, 2024, 149(9): 684-706. doi:10.1161/circulationaha.123.065603 |
| [16] | Fan YN, Chen ZW, Wang HX, et al. Isovitexin targets SIRT3 to prevent steroid-induced osteonecrosis of the femoral head by modulating mitophagy-mediated ferroptosis[J]. Bone Res, 2025, 13(1): 18. doi:10.1038/s41413-024-00390-0 |
| [17] | Peng YC, Guo MY, Luo MH, et al. Dapagliflozin ameliorates myocardial infarction injury through AMPKα-dependent regulation of oxidative stress and apoptosis[J]. Heliyon, 2024, 10(7): e29160. doi:10.1016/j.heliyon.2024.e29160 |
| [18] | Rubler S, Dlugash J, Yuceoglu YZ, et al. New type of cardiomyopathy associated with diabetic glomerulosclerosis[J]. Am J Cardiol, 1972, 30(6): 595-602. doi:10.1016/0002-9149(72)90595-4 |
| [19] | Kautzky-Willer A, Leutner M, Harreiter J. Sex differences in type 2 diabetes[J]. Diabetologia, 2023, 66(6): 986-1002. doi:10.1007/s00125-023-05891-x |
| [20] | Tan Y, Zhang ZG, Zheng C, et al. Mechanisms of diabetic cardiomyopathy and potential therapeutic strategies: preclinical and clinical evidence[J]. Nat Rev Cardiol, 2020, 17(9): 585-607. doi:10.1038/s41569-020-0339-2 |
| [21] | Wang DZ, Yin Y, Wang SY, et al. FGF1ΔHBS prevents diabetic cardiomyopathy by maintaining mitochondrial homeostasis and reducing oxidative stress via AMPK/Nur77 suppression[J]. Signal Transduct Target Ther, 2021, 6(1): 133. doi:10.1038/s41392-021-00542-2 |
| [22] | Kyriazis ID, Hoffman M, Gaignebet L, et al. KLF5 is induced by FOXO1 and causes oxidative stress and diabetic cardiomyopathy[J]. Circ Res, 2021, 128(3): 335-57. doi:10.1161/circresaha.120.316738 |
| [23] | Wang MY, Zhang SW, Tian JW, et al. Impaired iron-sulfur cluster synthesis induces mitochondrial PARthanatos in diabetic cardiomyopathy[J]. Adv Sci (Weinh), 2025, 12(1): e2406695. doi:10.1002/advs.202406695 |
| [24] | Huang YS, Luo W, Chen SY, et al. Isovitexin alleviates hepatic fibrosis by regulating miR-21-mediated PI3K/Akt signaling and glutathione metabolic pathway: based on transcriptomics and metabolomics[J]. Phytomedicine, 2023, 121: 155117. doi:10.1016/j.phymed.2023.155117 |
| [25] | Hao RL, Li MQ, Li F, et al. Protective effects of the phenolic compounds from mung bean hull against H2O2-induced skin aging through alleviating oxidative injury and autophagy in HaCaT cells and HSF cells[J]. Sci Total Environ, 2022, 841: 156669. doi:10.1016/j.scitotenv.2022.156669 |
| [26] | Pal S, Singh M, Porwal K, et al. Adiponectin receptors by increasing mitochondrial biogenesis and respiration promote osteoblast differentiation: Discovery of isovitexin as a new class of small molecule adiponectin receptor modulator with potential osteoanabolic function[J]. Eur J Pharmacol, 2021, 913: 174634. doi:10.1016/j.ejphar.2021.174634 |
| [27] | Zhu J, Sun RP, Sun KQ, et al. The deubiquitinase USP11 ameliorates intervertebral disc degeneration by regulating oxidative stress-induced ferroptosis via deubiquitinating and stabilizing Sirt3[J]. Redox Biol, 2023, 62: 102707. doi:10.1016/j.redox.2023.102707 |
| [28] | Ning Y, Dou XY, Wang ZC, et al. SIRT3: a potential therapeutic target for liver fibrosis[J]. Pharmacol Ther, 2024, 257: 108639. doi:10.1016/j.pharmthera.2024.108639 |
| [29] | D'Onofrio N, Prattichizzo F, Marfella R, et al. SIRT3 mediates the effects of PCSK9 inhibitors on inflammation, autophagy, and oxidative stress in endothelial cells[J]. Theranostics, 2023, 13(2): 531-42. doi:10.7150/thno.80289 |
| [30] | Mihanfar A, Nejabati HR, Fattahi A, et al. SIRT3-mediated cardiac remodeling/repair following myocardial infarction[J]. Biomed Pharmacother, 2018, 108: 367-73. doi:10.1016/j.biopha.2018.09.079 |
| [31] | Su H, Cantrell AC, Chen JX, et al. SIRT3 deficiency enhances ferroptosis and promotes cardiac fibrosis via p53 acetylation[J]. Cells, 2023, 12(10): 1428. doi:10.3390/cells12101428 |
| [1] | 林心君, 何昱霖, 施红, 刘佳绣, 胡海霞. 石斛合剂通过调控Sirt3介导的线粒体自噬通路缓解大鼠糖尿病心肌病[J]. 南方医科大学学报, 2026, 46(1): 47-54. |
| [2] | 刘信邦, 常柏. 糖尿病胫前色素斑的病理改变揭示血管-免疫异常[J]. 南方医科大学学报, 2026, 46(1): 66-73. |
| [3] | 李钊泳, 周凤华, 孙晓敏, 赵华杉, 金瑶, 何培坤, 贾钰华. 降脂祛斑方多成分协同调控炎症-代谢网络改善2型糖尿病合并高脂血症:网络药理学与临床验证[J]. 南方医科大学学报, 2026, 46(1): 83-93. |
| [4] | 马倩倩, 牛钰琪, 左铭钰, 李鑫, 符竣轲, 王瑾瑾. 鬼箭羽通过抑制AGEs-RAGE信号转导通路改善晚期糖基化终末产物诱导的小鼠肾足细胞损伤[J]. 南方医科大学学报, 2025, 45(9): 1938-1945. |
| [5] | 王静娴, 任自敬, 周佩洋. S1PR5激动与过表达通过调控氧化应激增强脑微血管内皮细胞屏障功能抵抗氧糖剥夺/复氧复糖损伤[J]. 南方医科大学学报, 2025, 45(7): 1451-1459. |
| [6] | 韩瑞敏, 赵曼可, 袁俊芳, 史振红, 王珍, 王德峰. 枯草杆菌二联活菌肠溶胶囊调节2型糖尿病合并昼夜节律紊乱小鼠糖脂代谢的作用机制[J]. 南方医科大学学报, 2025, 45(7): 1490-1497. |
| [7] | 张安邦, 孙秀颀, 庞博, 吴远华, 时靖宇, 张宁, 叶涛. 电针预处理通过调节肠道-大脑轴及Nrf2/HO-1信号通路抑制铁死亡减轻大鼠脑缺血再灌注损伤[J]. 南方医科大学学报, 2025, 45(5): 911-920. |
| [8] | 高志, 吴傲, 胡仲翔, 孙培养. 类风湿性关节炎中氧化应激与免疫浸润的生物信息学分析[J]. 南方医科大学学报, 2025, 45(4): 862-870. |
| [9] | 董妍妍, 张可敬, 储俊, 储全根. 抵当汤含药血清通过PI3K/Akt/mTOR信号通路增强高糖诱导的大鼠肾小球内皮细胞自噬[J]. 南方医科大学学报, 2025, 45(3): 461-469. |
| [10] | 孙红燕, 卢国庆, 付程文, 徐梦文, 朱小翌, 邢国权, 刘乐强, 柯雨菲, 崔乐妹, 陈睿旸, 王磊, 康品方, 唐碧. 槲皮素通过调控L型钙通道改善糖尿病大鼠心肌损伤[J]. 南方医科大学学报, 2025, 45(3): 531-541. |
| [11] | 黄鹏伟, 陈洁, 邹金虎, 高雪锋, 曹虹. 槲皮素促进应激颗粒G3BP1解聚改善HIV-1 gp120诱导的星形胶质细胞神经毒性[J]. 南方医科大学学报, 2025, 45(2): 304-312. |
| [12] | 卢晓宇, 刘智慧, 刘烨, 庞天霄, 卞蓉, 郭玲, 何学红. 参芪泄浊饮通过调控Rap1/MAPK/FoxO3a信号通路改善氧化应激及炎症反应延缓大鼠肾纤维化[J]. 南方医科大学学报, 2025, 45(12): 2585-2597. |
| [13] | 刘新新, 徐迎芮, 盛红娜, 刘昊. 人源脐带间充质干细胞移植通过Chi3l1抑制M1型巨噬细胞极化减轻1型糖尿病小鼠的炎症反应[J]. 南方医科大学学报, 2025, 45(12): 2738-2746. |
| [14] | 邱晓慧, 王檬, 唐江解, 周建大, 金晨. 负载人脐带间充质干细胞外泌体的壳聚糖水凝胶促进慢性糖尿病大鼠的伤口愈合[J]. 南方医科大学学报, 2025, 45(10): 2082-2091. |
| [15] | 林淑娴, 郭丽娜, 马燕, 熊尧, 何盈犀, 许欣筑, 盛雯, 许素哗, 邱峰. 植物乳植杆菌ZG03通过其代谢物短链脂肪酸缓解斑马鱼的氧化应激[J]. 南方医科大学学报, 2025, 45(10): 2223-2230. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||