南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 301-315.doi: 10.12122/j.issn.1673-4254.2026.02.08
• • 上一篇
周祺淇( ), 陈君贺, 聂城涛, 王前, 夏方博(
), 陈君贺, 聂城涛, 王前, 夏方博( ), 周宏伟(
), 周宏伟( )
)
收稿日期:2025-10-21
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
夏方博,周宏伟
E-mail:zqq19950928@126.com;xiafb08@163.com;hzhou@smu.edu.cn
作者简介:周祺淇,主管技师,在读硕士研究生,E-mail: zqq19950928@126.com
基金资助:
Qiqi ZHOU( ), Junhe CHEN, Chengtao NIE, Qian WANG, Fangbo XIA(
), Junhe CHEN, Chengtao NIE, Qian WANG, Fangbo XIA( ), Hongwei ZHOU(
), Hongwei ZHOU( )
)
Received:2025-10-21
Online:2026-02-20
Published:2026-03-10
Contact:
Fangbo XIA, Hongwei ZHOU
E-mail:zqq19950928@126.com;xiafb08@163.com;hzhou@smu.edu.cn
Supported by:摘要:
目的 建立一种人血清中类固醇激素的高灵敏度、高特异性液相色谱-质谱串联法(LC-MS/MS)分析方法。 方法 采用固相萃取(SPE)法进行样本前处理,优化LC-MS/MS确保痕量激素的准确检测。经验证方法的线性范围、正确度、精密度等指标后,将该方法应用于正常妊娠人群血清样本的检测,分析孕早、中、晚期血清中32种类固醇激素的动态变化规律。 结果 线性范围为0.001~7500 ng/mL,检出限(LOD)为0.0007 ng/mL,日内和日间变异系数(CV)均小于10%,加标回收率在为86.21%~112.67%,方法的萃取效率为85.05%~106.3%,基质效应为85.60%~113.22%。将该方法应用于妊娠期类固醇激素动态分析发现,多数激素(如11-脱氧皮质醇、11β-羟孕酮、硫酸脱氢表雄酮、雌三醇等)水平随妊娠进展显著上升(P<0.05),提示其与胎儿-胎盘功能发育密切相关;部分激素在孕晚期升高(P<0.05),可能与母体适应性调节及胎儿肾上腺功能成熟有关。 结论 本研究基于LC-MS/MS技术建立了32种类固醇激素高灵敏度检测方法,揭示了妊娠期间类固醇激素的动态变化规律,为相关生理与病理机制研究提供了基础。
周祺淇, 陈君贺, 聂城涛, 王前, 夏方博, 周宏伟. 基于液相色谱-质谱串联法技术的32种类固醇激素的检测方法建立及其在妊娠全周期变化应用[J]. 南方医科大学学报, 2026, 46(2): 301-315.
Qiqi ZHOU, Junhe CHEN, Chengtao NIE, Qian WANG, Fangbo XIA, Hongwei ZHOU. Development of an LC-MS/MS method for 32 steroid hormones and exploration of their gestational variations[J]. Journal of Southern Medical University, 2026, 46(2): 301-315.
| Elution time (min) | A (0.5 mmol/L NH4F, %) | B (Methanol, %) | Flow (mL/min) |
|---|---|---|---|
| 0.00 | 50.0 | 50.0 | 0.4 |
| 4.80 | 40.0 | 60.0 | 0.4 |
| 7.00 | 25.0 | 75.0 | 0.4 |
| 8.50 | 10.0 | 90.0 | 0.4 |
| 11.00 | 10.0 | 90.0 | 0.4 |
| 11.10 | 50.0 | 50.0 | 0.4 |
| 13.50 | 50.0 | 50.0 | 0.4 |
表1 液相洗脱程序
Tab.1 Liquid phase washing procedure
| Elution time (min) | A (0.5 mmol/L NH4F, %) | B (Methanol, %) | Flow (mL/min) |
|---|---|---|---|
| 0.00 | 50.0 | 50.0 | 0.4 |
| 4.80 | 40.0 | 60.0 | 0.4 |
| 7.00 | 25.0 | 75.0 | 0.4 |
| 8.50 | 10.0 | 90.0 | 0.4 |
| 11.00 | 10.0 | 90.0 | 0.4 |
| 11.10 | 50.0 | 50.0 | 0.4 |
| 13.50 | 50.0 | 50.0 | 0.4 |
| NO. | Analyte | Q1 Mass (m/z) | Q3 Mass (m/z) | DP (V) | CE (V) | Retention time (min) |
|---|---|---|---|---|---|---|
| 1 | Melatonin | 233.00 | 159.10 | 25 | 36 | 1.74 |
| 2 | 18-oxocortisol | 377.00 | 313.10 | 110 | 27 | 2.13 |
| 3 | 18-hydroxycortisol | 379.10 | 267.10 | 40 | 24 | 2.40 |
| 4 | Estriol | 287.10 | 171.00 | -180 | -45 | 2.72 |
| 5 | Aldosterone | 361.10 | 315.20 | 40 | 28 | 2.80 |
| 6 | Cortisone | 361.30 | 163.10 | 40 | 32 | 3.12 |
| 7 | 11β-Hydroxyandrost-4-ene-3,17-dione | 303.10 | 267.20 | 40 | 24 | 3.13 |
| 8 | 18-Hydroxycorticosterone | 363.10 | 269.20 | 60 | 28 | 3.22 |
| 9 | Adrenosterone | 301.10 | 257.30 | 75 | 32 | 3.40 |
| 10 | Cortisol | 363.22 | 121.03 | 40 | 36 | 3.57 |
| 11 | Dehydroepiandrosterone Sulfate | 367.10 | 97.00 | -280 | -60 | 3.79 |
| 12 | 11-Ketotestosterone | 303.10 | 121.20 | 40 | 36 | 3.93 |
| 13 | 21-Deoxycortisol | 347.30 | 311.20 | 40 | 24 | 4.58 |
| 14 | Dexamethasone | 393.30 | 373.40 | 25 | 16 | 4.70 |
| 15 | 11β-Hydroxytestosterone | 305.10 | 269.10 | 40 | 24 | 4.87 |
| 16 | Corticosterone | 347.10 | 329.14 | 80 | 20 | 4.96 |
| 17 | 11-Deoxycortisol | 347.10 | 97.10 | 60 | 52 | 5.21 |
| 18 | Androstenedione | 287.40 | 97.00 | 80 | 28 | 6.38 |
| 19 | Estrone | 269.10 | 145.10 | -100 | -45 | 6.55 |
| 20 | Estradiol | 271.20 | 145.10 | -40 | -45 | 6.75 |
| 21 | 21-Hydroxyprogesterone | 331.20 | 97.03 | 60 | 32 | 6.94 |
| 22 | 11β-Hydroxyprogesterone | 331.20 | 313.10 | 85 | 24 | 7.10 |
| 23 | Testosterone | 289.24 | 97.30 | 25 | 28 | 7.25 |
| 24 | 17-Hydroxyprogesterone | 331.15 | 97.10 | 40 | 28 | 7.56 |
| 25 | Dehydroepiandrosterone | 271.00 | 213.10 | 60 | 16 | 7.82 |
| 26 | 17α-Hydroxypregnenolone | 331.10 | 287.30 | -70 | -30 | 7.87 |
| 27 | Epitestosterone | 289.10 | 109.10 | 65 | 36 | 8.20 |
| 28 | Epiandrosterone | 291.00 | 273.30 | 40 | 15 | 8.32 |
| 29 | Dihydrotestosterone | 291.10 | 255.30 | 25 | 23 | 8.51 |
| 30 | Androsterone | 291.00 | 273.30 | 40 | 15 | 9.20 |
| 31 | Pregnenolone | 299.00 | 159.30 | 40 | 31 | 9.62 |
| 32 | Progesterone | 315.30 | 97.10 | 40 | 32 | 9.73 |
表2 LC-MS/MS方法的质谱参数
Tab.2 Mass spectrometry parameters of LC-MS/MS method
| NO. | Analyte | Q1 Mass (m/z) | Q3 Mass (m/z) | DP (V) | CE (V) | Retention time (min) |
|---|---|---|---|---|---|---|
| 1 | Melatonin | 233.00 | 159.10 | 25 | 36 | 1.74 |
| 2 | 18-oxocortisol | 377.00 | 313.10 | 110 | 27 | 2.13 |
| 3 | 18-hydroxycortisol | 379.10 | 267.10 | 40 | 24 | 2.40 |
| 4 | Estriol | 287.10 | 171.00 | -180 | -45 | 2.72 |
| 5 | Aldosterone | 361.10 | 315.20 | 40 | 28 | 2.80 |
| 6 | Cortisone | 361.30 | 163.10 | 40 | 32 | 3.12 |
| 7 | 11β-Hydroxyandrost-4-ene-3,17-dione | 303.10 | 267.20 | 40 | 24 | 3.13 |
| 8 | 18-Hydroxycorticosterone | 363.10 | 269.20 | 60 | 28 | 3.22 |
| 9 | Adrenosterone | 301.10 | 257.30 | 75 | 32 | 3.40 |
| 10 | Cortisol | 363.22 | 121.03 | 40 | 36 | 3.57 |
| 11 | Dehydroepiandrosterone Sulfate | 367.10 | 97.00 | -280 | -60 | 3.79 |
| 12 | 11-Ketotestosterone | 303.10 | 121.20 | 40 | 36 | 3.93 |
| 13 | 21-Deoxycortisol | 347.30 | 311.20 | 40 | 24 | 4.58 |
| 14 | Dexamethasone | 393.30 | 373.40 | 25 | 16 | 4.70 |
| 15 | 11β-Hydroxytestosterone | 305.10 | 269.10 | 40 | 24 | 4.87 |
| 16 | Corticosterone | 347.10 | 329.14 | 80 | 20 | 4.96 |
| 17 | 11-Deoxycortisol | 347.10 | 97.10 | 60 | 52 | 5.21 |
| 18 | Androstenedione | 287.40 | 97.00 | 80 | 28 | 6.38 |
| 19 | Estrone | 269.10 | 145.10 | -100 | -45 | 6.55 |
| 20 | Estradiol | 271.20 | 145.10 | -40 | -45 | 6.75 |
| 21 | 21-Hydroxyprogesterone | 331.20 | 97.03 | 60 | 32 | 6.94 |
| 22 | 11β-Hydroxyprogesterone | 331.20 | 313.10 | 85 | 24 | 7.10 |
| 23 | Testosterone | 289.24 | 97.30 | 25 | 28 | 7.25 |
| 24 | 17-Hydroxyprogesterone | 331.15 | 97.10 | 40 | 28 | 7.56 |
| 25 | Dehydroepiandrosterone | 271.00 | 213.10 | 60 | 16 | 7.82 |
| 26 | 17α-Hydroxypregnenolone | 331.10 | 287.30 | -70 | -30 | 7.87 |
| 27 | Epitestosterone | 289.10 | 109.10 | 65 | 36 | 8.20 |
| 28 | Epiandrosterone | 291.00 | 273.30 | 40 | 15 | 8.32 |
| 29 | Dihydrotestosterone | 291.10 | 255.30 | 25 | 23 | 8.51 |
| 30 | Androsterone | 291.00 | 273.30 | 40 | 15 | 9.20 |
| 31 | Pregnenolone | 299.00 | 159.30 | 40 | 31 | 9.62 |
| 32 | Progesterone | 315.30 | 97.10 | 40 | 32 | 9.73 |
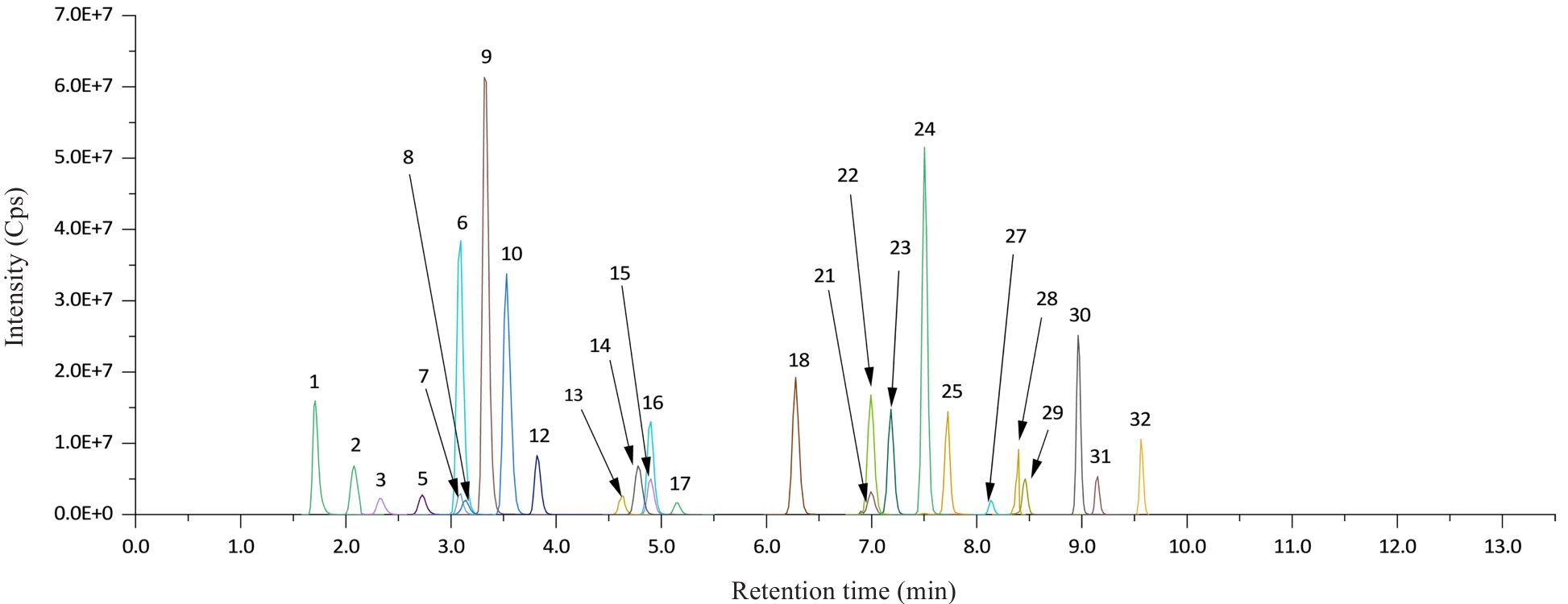
图1 正离子模式下32种类固醇激素色谱图
Fig.1 Chromatogram of 32 steroid hormones in positive ion mode. 1: Melatonin; 2: 18-oxocortisol; 3: 18-hydroxycortisol; 5: Aldosterone; 6: Cortisone; 7: 11β-Hydroxyandrost-4-ene-3,17-dione; 8: 18-Hydroxycorticosterone; 9: Adrenosterone; 10: Cortisol; 12: 11-Ketotestosterone; 13: 21-Deoxycortisol; 14: Dexamethasone; 15: 11β-Hydroxytestosterone; 16: Corticosterone; 17: 11-Deoxycortisol; 18: Androstenedione; 21: 21-Hydroxyprogesterone; 22: 11β-Hydroxyprogesterone; 23: Testosterone; 24: 17-Hydroxyprogesterone; 25: Dehydroepiandrosterone; 27: Epitestosterone; 28: Epiandrosterone; 29: Dihydrotestosterone; 30: Androsterone; 31: Pregnenolone; 32: Progesterone.
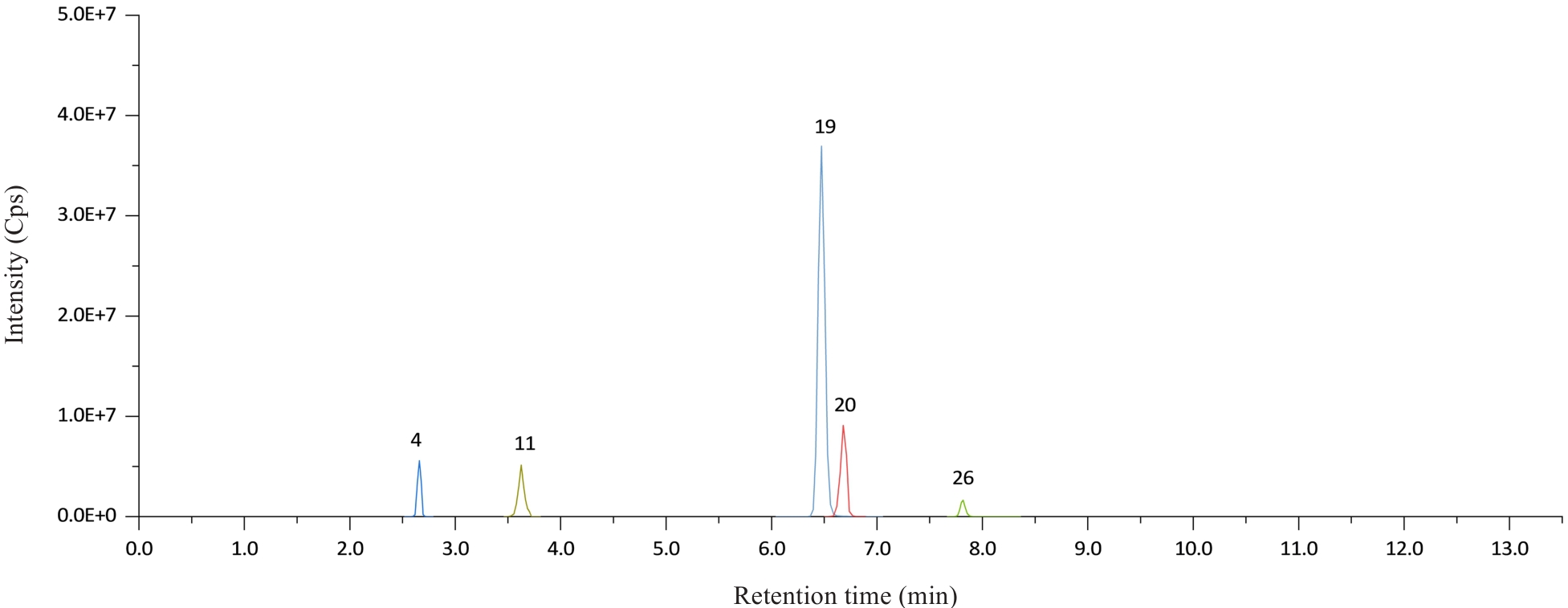
图2 负离子模式下32种类固醇激素色谱图
Fig.2 Chromatogram of 32 steroid hormones in negative ion mode. 4: Estriol; 11: Dehydroepiandrosterone Sulfate; 19: Estrone; 20: Estradiol; 26: 17α-Hydroxypregnenolone.
| NO. | Analytes | Linearity | LOQ (ng/mL) | LOD (ng/mL) | ||
|---|---|---|---|---|---|---|
| Regression | Range (ng/mL) | R2 | ||||
| 1 | Melatonin | y=202x-0.0483 | 0.005-12 | 0.994 | 0.005 | 0.002 |
| 2 | 18-Oxocortisol | y=35.6x-0.0212 | 0.01-15 | 0.993 | 0.010 | 0.007 |
| 3 | 18-Hydroxycortisol | y=5x-0.0353 | 0.03-30 | 0.997 | 0.030 | 0.020 |
| 4 | Estriol | y=2.54e+003x-107 | 0.2-300 | 0.999 | 0.200 | 0.070 |
| 5 | Aldosterone | y=41.4x-0.0763 | 0.007-20 | 0.998 | 0.007 | 0.003 |
| 6 | Cortisone | y=2.24x+0.00123 | 0.02-200 | 0.993 | 0.020 | 0.010 |
| 7 | 11β-Hydroxyandrost-4-ene-3,17-dione | y=49.7x-0.0161 | 0.005-10 | 0.996 | 0.005 | 0.003 |
| 8 | 18-Hydroxycorticosterone | y=12x+0.00666 | 0.003-15 | 0.996 | 0.003 | 0.001 |
| 9 | Adrenosterone | y=0.543x+0.00285 | 0.05-400 | 0.998 | 0.050 | 0.020 |
| 10 | Cortisol | y=3.86x+0.00437 | 0.02-400 | 0.996 | 0.020 | 0.006 |
| 11 | Dehydroepiandrosterone sulfate | y=1.49e+003x+108 | 1-7500 | 0.999 | 1.000 | 0.900 |
| 12 | 11-Ketotestosterone | y=11.5x-0.00131 | 0.001-6 | 0.993 | 0.001 | 0.0007 |
| 13 | 21-Deoxycortisol | y=4.8x-0.00272 | 0.003-15 | 0.996 | 0.003 | 0.001 |
| 14 | Dexamethasone | y=0.409x-0.00365 | 0.1-60 | 0.996 | 0.100 | 0.050 |
| 15 | 11β-Hydroxytestosterone | y=1.2x-0.00209 | 0.01-30 | 0.998 | 0.010 | 0.007 |
| 16 | Corticosterone | y=4.65x+0.00826 | 0.01-30 | 0.998 | 0.010 | 0.007 |
| 17 | 11-Deoxycortisol | y=0.358x+0.000234 | 0.01-15 | 0.997 | 0.005 | 0.001 |
| 18 | Androstenedione | y=2.14x-0.00139 | 0.01-30 | 0.997 | 0.007 | 0.003 |
| 19 | Estrone | y=1.55e+005x-676 | 0.1-900 | 0.997 | 0.100 | 0.050 |
| 20 | Estradiol | y=3.52e+004x-344 | 0.1-600 | 0.996 | 0.100 | 0.070 |
| 21 | 21-Hydroxyprogesterone | y 2.24x+ 0.0106 | 0.007-20 | 0.992 | 0.007 | 0.003 |
| 22 | 11β-Hydroxyprogesterone | y=1.21x+0.000691 | 0.02-50 | 0.996 | 0.020 | 0.010 |
| 23 | Testosterone | y=2.68x+0.0107 | 0.01-15 | 0.997 | 0.005 | 0.001 |
| 24 | 17-Hydroxyprogesterone | y=1.21x+0.00351 | 0.01-100 | 0.995 | 0.010 | 0.006 |
| 25 | Dehydroepiandrosterone | y=0.338x+0.000631 | 0.05-100 | 0.993 | 0.050 | 0.020 |
| 26 | 17α-Hydroxypregnenolone | y=5.74e+004x-846 | 0.1-100 | 0.995 | 0.100 | 0.040 |
| 27 | Epitestosterone | y=0.985x-0.00085 | 0.01-5 | 0.997 | 0.010 | 0.004 |
| 28 | Epiandrosterone | y=0.307x-0.0116 | 0.01-5 | 0.997 | 0.010 | 0.004 |
| 29 | Dihydrotestosterone | y=3.77x+0.000735 | 0.005-10 | 0.996 | 0.005 | 0.002 |
| 30 | Androsterone | y=0.938x-0.027 | 0.1-10 | 0.997 | 0.100 | 0.070 |
| 31 | Pregnenolone | y=0.581x+0.0466 | 0.1-100 | 0.995 | 0.100 | 0.040 |
| 32 | Progesterone | y=4.93x+0.00683 | 0.005-30 | 0.997 | 0.005 | 0.003 |
表3 方法的线性方程、线性范围、相关系数、LOQ和LOD
Tab.3 Linear equations, linear ranges, correlation coefficients, LOQ, and LOD of the method
| NO. | Analytes | Linearity | LOQ (ng/mL) | LOD (ng/mL) | ||
|---|---|---|---|---|---|---|
| Regression | Range (ng/mL) | R2 | ||||
| 1 | Melatonin | y=202x-0.0483 | 0.005-12 | 0.994 | 0.005 | 0.002 |
| 2 | 18-Oxocortisol | y=35.6x-0.0212 | 0.01-15 | 0.993 | 0.010 | 0.007 |
| 3 | 18-Hydroxycortisol | y=5x-0.0353 | 0.03-30 | 0.997 | 0.030 | 0.020 |
| 4 | Estriol | y=2.54e+003x-107 | 0.2-300 | 0.999 | 0.200 | 0.070 |
| 5 | Aldosterone | y=41.4x-0.0763 | 0.007-20 | 0.998 | 0.007 | 0.003 |
| 6 | Cortisone | y=2.24x+0.00123 | 0.02-200 | 0.993 | 0.020 | 0.010 |
| 7 | 11β-Hydroxyandrost-4-ene-3,17-dione | y=49.7x-0.0161 | 0.005-10 | 0.996 | 0.005 | 0.003 |
| 8 | 18-Hydroxycorticosterone | y=12x+0.00666 | 0.003-15 | 0.996 | 0.003 | 0.001 |
| 9 | Adrenosterone | y=0.543x+0.00285 | 0.05-400 | 0.998 | 0.050 | 0.020 |
| 10 | Cortisol | y=3.86x+0.00437 | 0.02-400 | 0.996 | 0.020 | 0.006 |
| 11 | Dehydroepiandrosterone sulfate | y=1.49e+003x+108 | 1-7500 | 0.999 | 1.000 | 0.900 |
| 12 | 11-Ketotestosterone | y=11.5x-0.00131 | 0.001-6 | 0.993 | 0.001 | 0.0007 |
| 13 | 21-Deoxycortisol | y=4.8x-0.00272 | 0.003-15 | 0.996 | 0.003 | 0.001 |
| 14 | Dexamethasone | y=0.409x-0.00365 | 0.1-60 | 0.996 | 0.100 | 0.050 |
| 15 | 11β-Hydroxytestosterone | y=1.2x-0.00209 | 0.01-30 | 0.998 | 0.010 | 0.007 |
| 16 | Corticosterone | y=4.65x+0.00826 | 0.01-30 | 0.998 | 0.010 | 0.007 |
| 17 | 11-Deoxycortisol | y=0.358x+0.000234 | 0.01-15 | 0.997 | 0.005 | 0.001 |
| 18 | Androstenedione | y=2.14x-0.00139 | 0.01-30 | 0.997 | 0.007 | 0.003 |
| 19 | Estrone | y=1.55e+005x-676 | 0.1-900 | 0.997 | 0.100 | 0.050 |
| 20 | Estradiol | y=3.52e+004x-344 | 0.1-600 | 0.996 | 0.100 | 0.070 |
| 21 | 21-Hydroxyprogesterone | y 2.24x+ 0.0106 | 0.007-20 | 0.992 | 0.007 | 0.003 |
| 22 | 11β-Hydroxyprogesterone | y=1.21x+0.000691 | 0.02-50 | 0.996 | 0.020 | 0.010 |
| 23 | Testosterone | y=2.68x+0.0107 | 0.01-15 | 0.997 | 0.005 | 0.001 |
| 24 | 17-Hydroxyprogesterone | y=1.21x+0.00351 | 0.01-100 | 0.995 | 0.010 | 0.006 |
| 25 | Dehydroepiandrosterone | y=0.338x+0.000631 | 0.05-100 | 0.993 | 0.050 | 0.020 |
| 26 | 17α-Hydroxypregnenolone | y=5.74e+004x-846 | 0.1-100 | 0.995 | 0.100 | 0.040 |
| 27 | Epitestosterone | y=0.985x-0.00085 | 0.01-5 | 0.997 | 0.010 | 0.004 |
| 28 | Epiandrosterone | y=0.307x-0.0116 | 0.01-5 | 0.997 | 0.010 | 0.004 |
| 29 | Dihydrotestosterone | y=3.77x+0.000735 | 0.005-10 | 0.996 | 0.005 | 0.002 |
| 30 | Androsterone | y=0.938x-0.027 | 0.1-10 | 0.997 | 0.100 | 0.070 |
| 31 | Pregnenolone | y=0.581x+0.0466 | 0.1-100 | 0.995 | 0.100 | 0.040 |
| 32 | Progesterone | y=4.93x+0.00683 | 0.005-30 | 0.997 | 0.005 | 0.003 |
| NO. | Analytes | Day 1 (n=3) | Day 2 (n=3) | Day 3 (n=3) | |||
|---|---|---|---|---|---|---|---|
| Con (ng/mL) | CV (%) | Con (ng/mL) | CV (%) | Con (ng/mL) | CV (%) | ||
| 1 | Melatonin | 12.58 | 4.83 | 11.33 | -5.58 | 12.96 | 8.0 |
| 2 | 18-Oxocortisol | 15.89 | 5.93 | 14.17 | -5.53 | 16.02 | 6.8 |
| 3 | 18-Hydroxycortisol | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
| 4 | Estriol | 312.90 | 4.30 | 287.10 | -4.30 | 324.60 | 8.20 |
| 5 | Aldosterone | 20.78 | 3.90 | 18.46 | -7.70 | 21.60 | 8.00 |
| 6 | Cortisone | 96.24 | -3.76 | 103.72 | 3.72 | 108.13 | 8.13 |
| 7 | 11β-Hydroxyandrost-4-ene-3,17-dione | 10.62 | 6.20 | 9.38 | -6.20 | 10.80 | 8.00 |
| 8 | 18-Hydroxycorticosterone | 14.18 | -5.47 | 15.73 | 4.87 | 14.48 | -3.47 |
| 9 | Adrenosterone | 417.2 | 4.30 | 383.6 | -4.10 | 432.40 | 8.10 |
| 10 | Cortisol | 423.64 | 5.91 | 382.88 | -4.28 | 414.48 | 3.62 |
| 11 | Dehydroepiandrosterone sulfate | 7875.00 | 5.00 | 7125 | -5.00 | 8100.00 | 8.00 |
| 12 | 11-Ketotestosterone | 6.36 | 6.00 | 5.58 | -7.00 | 6.54 | 9.00 |
| 13 | 21-Deoxycortisol | 15.89 | 5.93 | 14.17 | -5.53 | 16.02 | 6.80 |
| 14 | Dexamethasone | 63.60 | 6.00 | 56.52 | -5.80 | 64.80 | 80.00 |
| 15 | 11β-Hydroxytestosterone | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
| 16 | Corticosterone | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
| 17 | 11-Deoxycortisol | 14.18 | -5.47 | 15.73 | 4.87 | 14.48 | -3.47 |
| 18 | Androstenedione | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
| 19 | Estrone | 943.74 | 4.86 | 862.38 | -4.18 | 972.54 | 8.06 |
| 20 | Estradiol | 624.60 | 4.10 | 576.42 | -3.93 | 648.54 | 8.09 |
| 21 | 21-Hydroxyprogesterone | 21.83 | 9.15 | 18.38 | -8.1 | 19.14 | -4.30 |
| 22 | 11β-Hydroxyprogesterone | 53.00 | 6.00 | 46.50 | -7.00 | 54.50 | 9.00 |
| 23 | Testosterone | 14.05 | -4.33 | 14.92 | -0.53 | 15.79 | 5.27 |
| 24 | 17-Hydroxyprogesterone | 107.83 | 5.83 | 92.94 | -7.06 | 103.66 | 3.66 |
| 25 | Dehydroepiandrosterone | 94.61 | -5.39 | 105.27 | 5.27 | 107.83 | 7.83 |
| 26 | 17α-Hydroxypregnenolone | 105.38 | 5.38 | 106.13 | 6.13 | 94.11 | -5.89 |
| 27 | Epitestosterone | 5.26 | 5.20 | 4.75 | -5.00 | 5.40 | 8.00 |
| 28 | Epiandrosterone | 5.31 | 6.20 | 4.69 | -6.20 | 4.60 | -8.00 |
| 29 | Dihydrotestosterone | 9.58 | -4.20 | 10.62 | 6.20 | 9.17 | -8.3 |
| 30 | Androsterone | 9.38 | -6.20 | 10.62 | 6.20 | 10.80 | 8.00 |
| 31 | Pregnenolone | 103.72 | 3.72 | 94.18 | -5.82 | 108.13 | 8.13 |
| 32 | Progesterone | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
表5 自动进样器的稳定性验证
Tab.5 Validation of the stability of the high-performance liquid chromatography autosampler
| NO. | Analytes | Day 1 (n=3) | Day 2 (n=3) | Day 3 (n=3) | |||
|---|---|---|---|---|---|---|---|
| Con (ng/mL) | CV (%) | Con (ng/mL) | CV (%) | Con (ng/mL) | CV (%) | ||
| 1 | Melatonin | 12.58 | 4.83 | 11.33 | -5.58 | 12.96 | 8.0 |
| 2 | 18-Oxocortisol | 15.89 | 5.93 | 14.17 | -5.53 | 16.02 | 6.8 |
| 3 | 18-Hydroxycortisol | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
| 4 | Estriol | 312.90 | 4.30 | 287.10 | -4.30 | 324.60 | 8.20 |
| 5 | Aldosterone | 20.78 | 3.90 | 18.46 | -7.70 | 21.60 | 8.00 |
| 6 | Cortisone | 96.24 | -3.76 | 103.72 | 3.72 | 108.13 | 8.13 |
| 7 | 11β-Hydroxyandrost-4-ene-3,17-dione | 10.62 | 6.20 | 9.38 | -6.20 | 10.80 | 8.00 |
| 8 | 18-Hydroxycorticosterone | 14.18 | -5.47 | 15.73 | 4.87 | 14.48 | -3.47 |
| 9 | Adrenosterone | 417.2 | 4.30 | 383.6 | -4.10 | 432.40 | 8.10 |
| 10 | Cortisol | 423.64 | 5.91 | 382.88 | -4.28 | 414.48 | 3.62 |
| 11 | Dehydroepiandrosterone sulfate | 7875.00 | 5.00 | 7125 | -5.00 | 8100.00 | 8.00 |
| 12 | 11-Ketotestosterone | 6.36 | 6.00 | 5.58 | -7.00 | 6.54 | 9.00 |
| 13 | 21-Deoxycortisol | 15.89 | 5.93 | 14.17 | -5.53 | 16.02 | 6.80 |
| 14 | Dexamethasone | 63.60 | 6.00 | 56.52 | -5.80 | 64.80 | 80.00 |
| 15 | 11β-Hydroxytestosterone | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
| 16 | Corticosterone | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
| 17 | 11-Deoxycortisol | 14.18 | -5.47 | 15.73 | 4.87 | 14.48 | -3.47 |
| 18 | Androstenedione | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
| 19 | Estrone | 943.74 | 4.86 | 862.38 | -4.18 | 972.54 | 8.06 |
| 20 | Estradiol | 624.60 | 4.10 | 576.42 | -3.93 | 648.54 | 8.09 |
| 21 | 21-Hydroxyprogesterone | 21.83 | 9.15 | 18.38 | -8.1 | 19.14 | -4.30 |
| 22 | 11β-Hydroxyprogesterone | 53.00 | 6.00 | 46.50 | -7.00 | 54.50 | 9.00 |
| 23 | Testosterone | 14.05 | -4.33 | 14.92 | -0.53 | 15.79 | 5.27 |
| 24 | 17-Hydroxyprogesterone | 107.83 | 5.83 | 92.94 | -7.06 | 103.66 | 3.66 |
| 25 | Dehydroepiandrosterone | 94.61 | -5.39 | 105.27 | 5.27 | 107.83 | 7.83 |
| 26 | 17α-Hydroxypregnenolone | 105.38 | 5.38 | 106.13 | 6.13 | 94.11 | -5.89 |
| 27 | Epitestosterone | 5.26 | 5.20 | 4.75 | -5.00 | 5.40 | 8.00 |
| 28 | Epiandrosterone | 5.31 | 6.20 | 4.69 | -6.20 | 4.60 | -8.00 |
| 29 | Dihydrotestosterone | 9.58 | -4.20 | 10.62 | 6.20 | 9.17 | -8.3 |
| 30 | Androsterone | 9.38 | -6.20 | 10.62 | 6.20 | 10.80 | 8.00 |
| 31 | Pregnenolone | 103.72 | 3.72 | 94.18 | -5.82 | 108.13 | 8.13 |
| 32 | Progesterone | 28.65 | -4.50 | 31.23 | 4.10 | 27.84 | -7.20 |
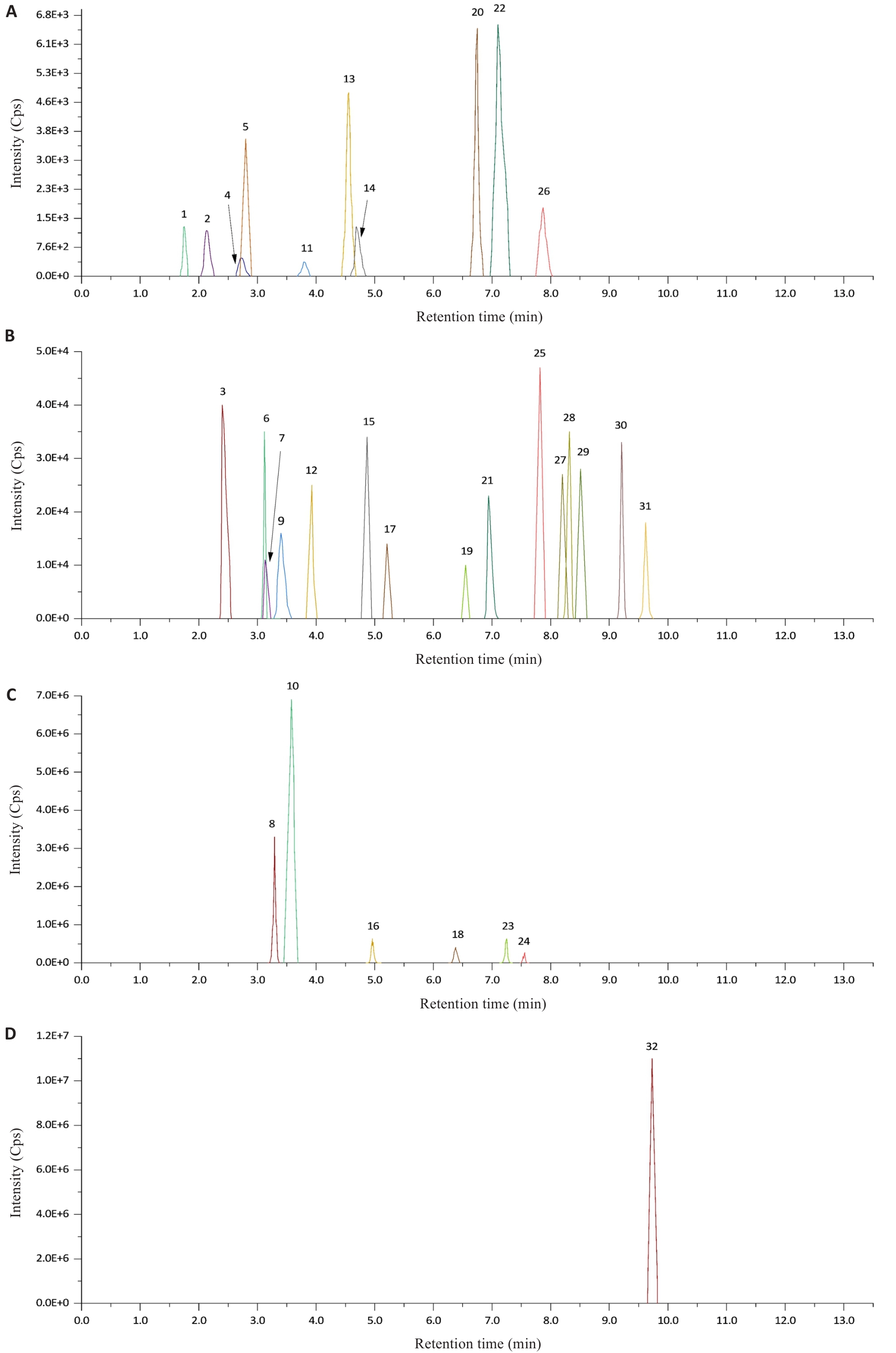
图6 孕中期血清样本中目标类固醇激素的代表性总离子流色谱图
Fig.6 Representative total ion current chromatogram of the target steroid hormones in a second-trimester serum sample. A-D: Mass spectral peak charts with different intensity ranges, showing the chromatographic separation of target compounds in low, medium-low, medium-high, and high response intervals, respectively. 1: Melatonin; 2: 18-oxocortisol; 3: 18-hydroxycortisol; 4: Estriol; 5: Aldosterone; 6: Cortisone; 7: 11β-Hydroxyandrost-4-ene-3,17-dione; 8: 18-Hydroxycorticosterone; 9: Adrenosterone; 10: Cortisol; 11: Dehydroepiandrosterone Sulfate; 12: 11-Ketotestosterone; 13: 21-Deoxycortisol; 14: Dexamethasone; 15: 11β-Hydroxytestosterone; 16: Corticosterone; 17: 11-Deoxycortisol; 18: Androstenedione; 19: Estrone; 20: Estradiol; 21: 21-Hydroxyprogesterone; 22: 11β-Hydroxyprogesterone; 23: Testosterone; 24: 17-Hydroxyprogesterone; 25: Dehydroepiandrosterone; 26: 17α-Hydroxypregnenolone; 27: Epitestosterone; 28: Epiandrosterone; 29: Dihydrotestosterone; 30: Androsterone; 31: Pregnenolone; 32: Progesterone.
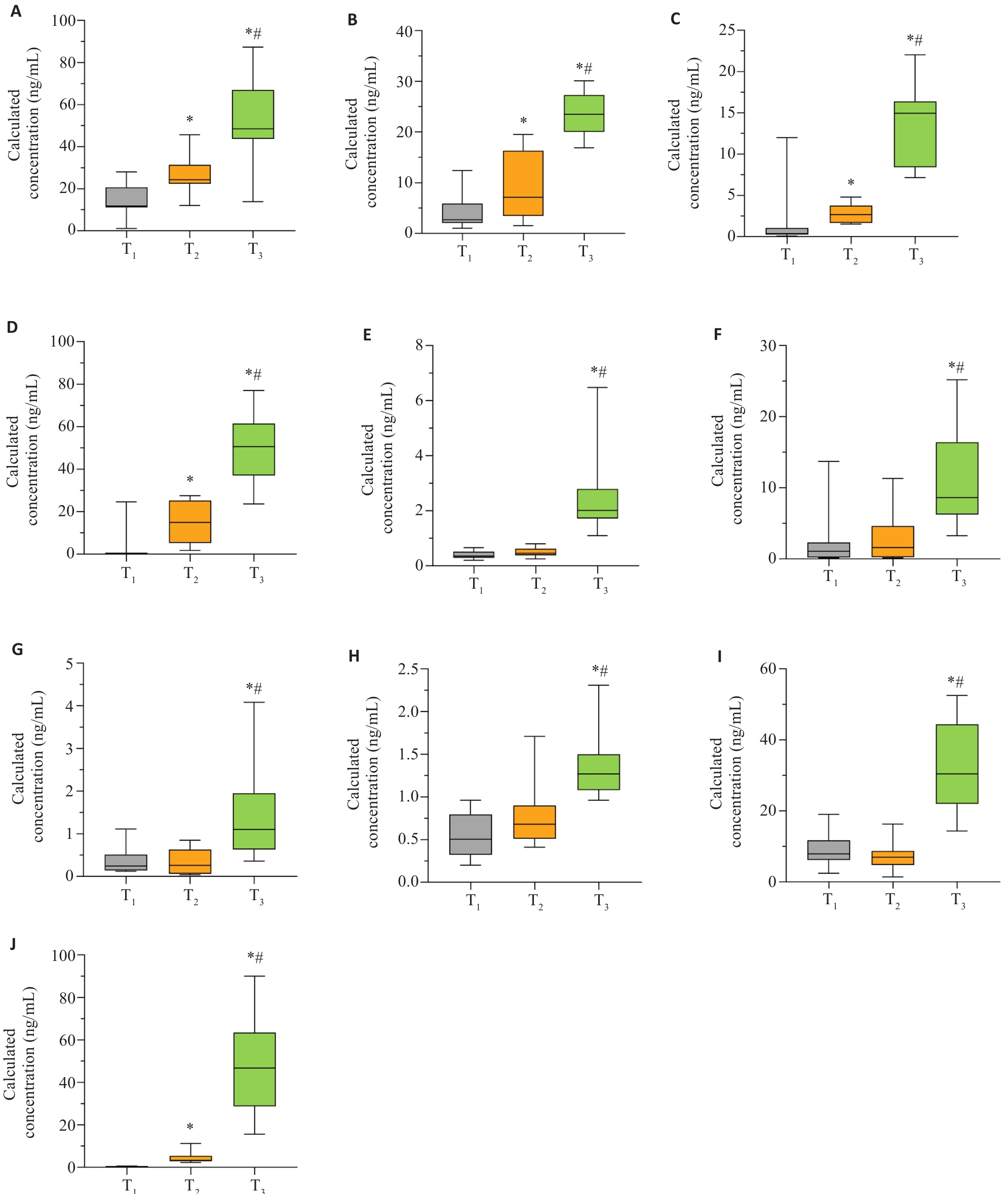
图7 孕酮、11-脱氧皮质醇、11β-羟孕酮、雌三醇、21-羟孕酮、11β-羟睾酮、表睾酮、18-羟基皮质酮、17-羟孕酮、硫酸脱氢表雄酮在妊娠全周期的变化趋势
Fig.7 Variations of progesterone, 11-deoxycortisol, 11β-hydroxyprogesterone, estriol, 21-hydroxyprogesterone, 11β-hydroxytestosterone, epitestosterone, 18-hydroxycorticosterone, 17-hydroxyprogesterone and dehydroepiandrosterone sulfate throughout pregnancy. A: Progesterone; B: 11-Deoxycortisol; C: 11β-Hydroxyprogesterone; D: Estriol; E: 21-Hydroxyprogesterone; F: 11β-Hydroxytestosterone; G: Epitestosterone; H: 18-Hydroxycorticosterone; I: 17-Hydroxyprogesterone; J: Dehydroepiandrosterone sulfate.T1:First trimester;T2: Second trimester;T3:Third trimester. *P<0.05 vs T1; #P<0.05 vs T2.
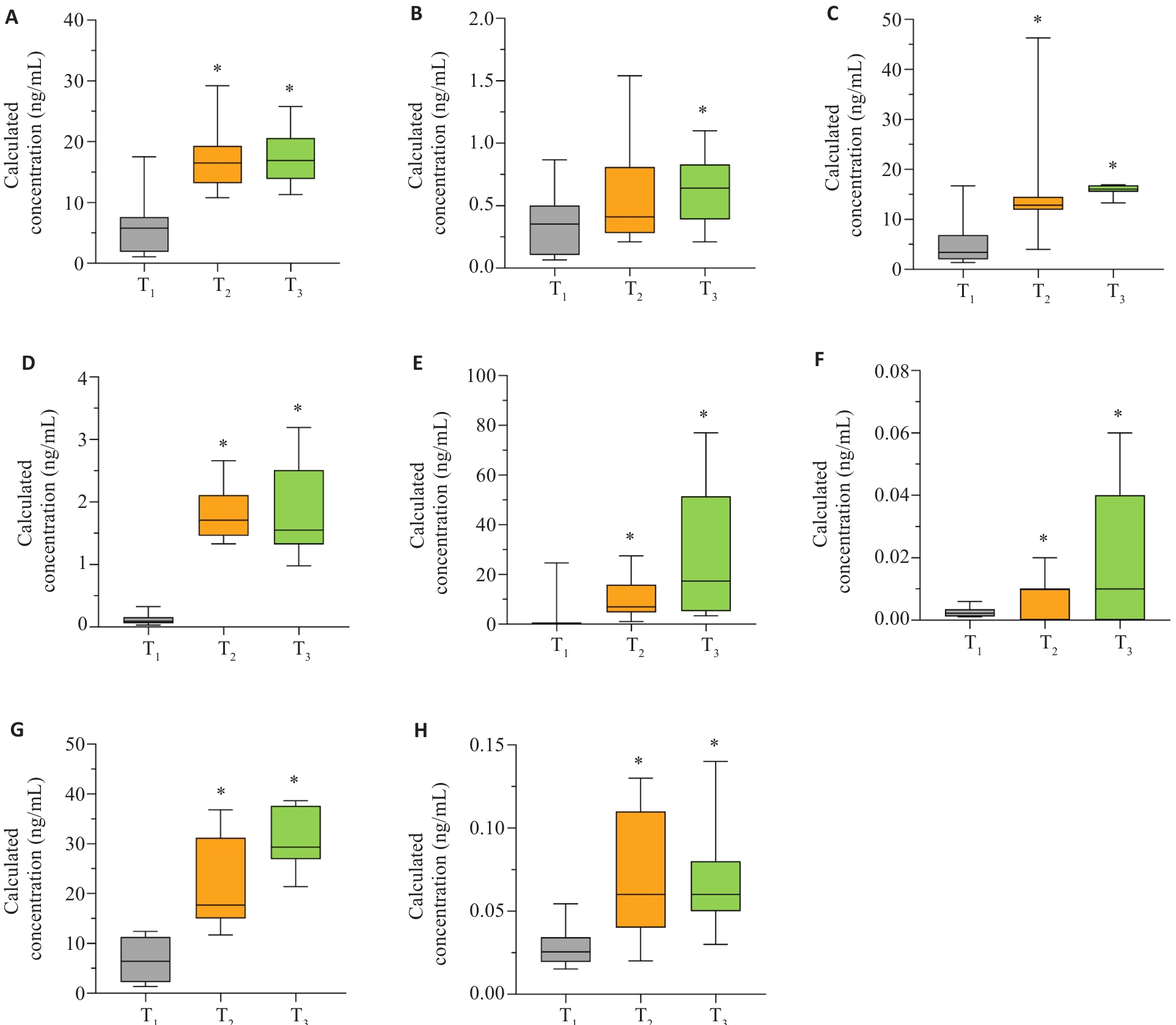
图8 雄烯二酮、皮质酮、可的松、雌酮、雌二醇、21-脱氧皮质醇、皮质醇、18-氧皮质醇在妊娠全周期的变化趋势
Fig.8 Trends of androstenedione, corticosterone, cortisone, estrone, estradiol, 21-deoxycortisol, cortisol and 18-hydro-xycortisol throughout pregnancy. A:Androstenedione. B: Corticosterone. C: Cortisone. D: Estrone. E: Estradiol. F: 21-Deoxycortisol. G: Cortisol. H: 18-Hydroxycortisol. *P<0.05 vs T1.
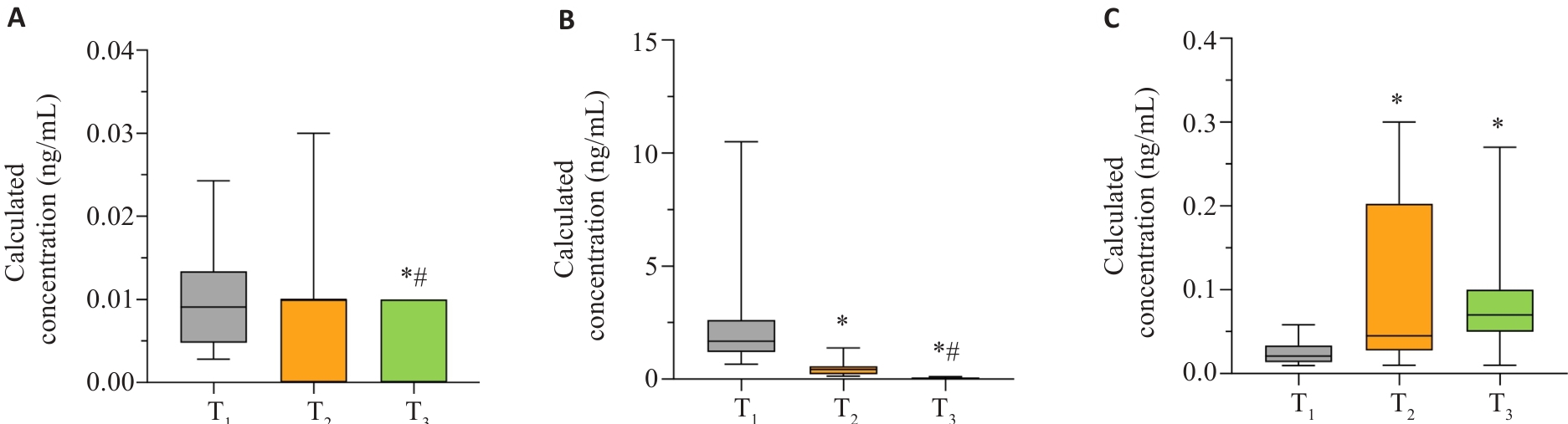
图9 11-酮-睾酮、孕烯醇酮、醛固酮在妊娠全周期的变化趋势
Fig.9 Trends of 11-ketotestosterone, pregnenolone and aldosterone throughout pregnancy. A: 11-Ketotestosterone. B: Pregnenolone. C: Aldosterone. *P<0.05 vs T1; #P<0.05 vs T2.
| [1] | Bacila IA, Elder C, Krone N. Update on adrenal steroid hormone biosynthesis and clinical implications[J]. Arch Dis Child, 2019, 104(12): 1223-8. doi:10.1136/archdischild-2017-313873 |
| [2] | Matsuyama S, DeFalco T. Steroid hormone signaling: multifaceted support of testicular function[J]. Front Cell Dev Biol, 2024, 11: 1339385. doi:10.3389/fcell.2023.1339385 |
| [3] | Bhaumik S, Lockett J, Cuffe J, et al. Glucocorticoids and their receptor isoforms: roles in female reproduction, pregnancy, and foetal development[J]. Biology (Basel), 2023, 12(8): 1104. doi:10.3390/biology12081104 |
| [4] | Márta Z, Bobály B, Fekete J, et al. Simultaneous determination of thirteen different steroid hormones using micro UHPLC-MS/MS with on-line SPE system[J]. J Pharm Biomed Anal, 2018, 150: 258-67. doi:10.1016/j.jpba.2017.12.014 |
| [5] | Lee W, Lee H, Kim YL, et al. Profiling of steroid metabolic pathways in human plasma by GC-MS/MS combined with microwave-assisted derivatization for diagnosis of gastric disorders[J]. Int J Mol Sci, 2021, 22(4): 1872. doi:10.3390/ijms22041872 |
| [6] | Yu SL, Yin YC, Zou YT, et al. A comprehensive LC-MS/MS method for the simultaneous measurement of 24 adrenal steroids: From research to clinical practice[J]. J Chromatogr B Analyt Technol Biomed Life Sci, 2024, 1232: 123941. doi:10.1016/j.jchromb.2023.123941 |
| [7] | Constantinescu G, Bidlingmaier M, Gruber M, et al. Mass spectrometry reveals misdiagnosis of primary aldosteronism with scheduling for adrenalectomy due to immunoassay interference[J]. Clin Chim Acta, 2020, 507: 98-103. doi:10.1016/j.cca.2020.04.019 |
| [8] | Athimulam S, Grebe S, Bancos I. Steroid profiling in the diagnosis of mild and overt Cushing's syndrome[J]. Best Pract Res Clin Endocrinol Metab, 2021, 35(1): 101488. doi:10.1016/j.beem.2021.101488 |
| [9] | 黄 江. 乳腺癌生物样本中内源性雌激素的代谢轮廓以及代谢组学的LC-MS/MS分析方法研究[D]. 北京: 北京协和医学院, 2011. |
| [10] | Conklin SE, Knezevic CE. Advancements in the gold standard: measuring steroid sex hormones by mass spectrometry[J]. Clin Biochem, 2020, 82: 21-32. doi:10.1016/j.clinbiochem.2020.03.008 |
| [11] | Stanczyk FZ, Jurow J, Hsing AW. Limitations of direct immunoassays for measuring circulating estradiol levels in postmenopausal women and men in epidemiologic studies[J]. Cancer Epidemiol Biomarkers Prev, 2010, 19(4): 903-6. doi:10.1158/1055-9965.epi-10-0081 |
| [12] | Ankarberg-Lindgren C, Becker C, Svala E, et al. Methodological considerations in determining sex steroids in children: comparison of conventional immunoassays with liquid chromatography-tandem mass spectrometry[J]. Clin Chem Lab Med, 2023, 62(1): 85-96. doi:10.1515/cclm-2023-0344 |
| [13] | Kumar D, Oberoi HS, Singh H, et al. Development and optimization of an in-house heterologous ELISA for detection of prednisolone drug in enzyme conjugates using spacers[J]. Front Immunol, 2023, 14: 1200328. doi:10.3389/fimmu.2023.1200328 |
| [14] | Debeljak Ž, Marković I, Pavela J, et al. Analytical bias of automated immunoassays for six serum steroid hormones assessed by LC-MS/MS[J]. Biochem Med (Zagreb), 2020, 30(3): 030701. doi:10.11613/bm.2020.030701 |
| [15] | Vierbaum L, Weiss N, Kaiser P, et al. Longitudinal analysis of external quality assessment of immunoassay-based steroid hormone measurement indicates potential for improvement in standardization[J]. Front Mol Biosci, 2024, 11: 1345356. doi:10.3389/fmolb.2024.1345356 |
| [16] | Cortez S, Arbeláez AM, Wallendorf M, et al. Peak serum Cortisol cutoffs to diagnose adrenal insufficiency across different Cortisol assays in children[J]. J Clin Res Pediatr Endocrinol, 2023, 15(4): 375-9. doi:10.4274/jcrpe.galenos.2023.2023-2-3 |
| [17] | Rosner W, Auchus RJ, Azziz R, et al. Position statement: utility, limitations, and pitfalls in measuring testosterone: an endocrine society position statement[J]. J Clin Endocrinol Metab, 2007, 92(2): 405-13. doi:10.1210/jc.2006-1864 |
| [18] | Foguet-Romero E, Samarra I, Guirro M, et al. Optimization of a GC-MS injection-port derivatization methodology to enhance metabolomics analysis throughput in biological samples[J]. J Proteome Res, 2022, 21(11): 2555-65. doi:10.1021/acs.jproteome.2c00119 |
| [19] | Souto BA, Hai BX, Asomaning J, et al. Advancing characterization of pyrolysis products: comprehensive gas chromatography methods for analytes in gas and liquid phases[J]. J Chromatogr A, 2025, 1741: 465641. doi:10.1016/j.chroma.2024.465641 |
| [20] | Thomas SN, French D, Jannetto PJ, et al. Liquid chromatography-tandem mass spectrometry for clinical diagnostics[J]. Nat Rev Methods Primers, 2022, 2(1): 96. doi:10.1038/s43586-022-00175-x |
| [21] | Xing SP, Yu HX, Liu M, et al. Recognizing contamination fragment ions in liquid chromatography-tandem mass spectrometry data[J]. J Am Soc Mass Spectrom, 2021, 32(9): 2296-305. doi:10.1021/jasms.0c00478 |
| [22] | Liu L, Zhang L, Zheng XY, et al. LC - MS/MS-based multiplex antibacterial platform for therapeutic drug monitoring in intensive care unit patients[J]. Front Pharmacol, 2023, 14: 1116071. doi:10.3389/fphar.2023.1116071 |
| [23] | Davla S, Daly E, Nedow J, et al. An LC-MS/MS method for simultaneous analysis of up to six monoamines from brain tissues[J]. J Chromatogr B, 2023, 1216: 123604. doi:10.1016/j.jchromb.2023.123604 |
| [24] | Maharjan AS, Wyness SP, Ray JA, et al. Detection and characterization of estradiol (E2) and unconjugated estriol (uE3) immunoassay interference due to anti-bovine alkaline phosphatase (ALP) antibodies[J]. Pract Lab Med, 2019, 17: e00131. doi:10.1016/j.plabm.2019.e00131 |
| [25] | Zhang J, Xu LZ, Qiao L. Falsely elevated serum estradiol in woman of reproductive age led to unnecessary intervention and delayed fertility opportunity: a case report and literature review[J]. BMC Womens Health, 2022, 22(1): 232. doi:10.1186/s12905-022-01828-5 |
| [26] | Li DN, Radulescu A, Shrestha RT, et al. Association of biotin ingestion with performance of hormone and nonhormone assays in healthy adults[J]. JAMA, 2017, 318(12): 1150-60. doi:10.1001/jama.2017.13705 |
| [27] | Li DN, Ferguson A, Cervinski MA, et al. AACC guidance document on biotin interference in laboratory tests[J]. J Appl Lab Med, 2020, 5(3): 575-87. doi:10.1093/jalm/jfz010 |
| [28] | Espiritu AI, Remalante-Rayco PPM. High-dose biotin for multiple sclerosis: a systematic review and meta-analyses of randomized controlled trials[J]. Mult Scler Relat Disord, 2021, 55: 103159. doi:10.1016/j.msard.2021.103159 |
| [29] | Nakamoto J, Fuqua JS. Laboratory assays in pediatric endo-crinology: common aspects[J]. Pediatr Endocrinol Rev, 2007, 5(): 539-54. |
| [30] | Kushnir MM, Rockwood AL, Bergquist J. Liquid chromatography-tandem mass spectrometry applications in endocrinology[J]. Mass Spectrom Rev, 2010, 29(3): 480-502. doi:10.1002/mas.20264 |
| [31] | Annesley TM. Ion suppression in mass spectrometry[J]. Clin Chem, 2003, 49(7): 1041-4. doi:10.1373/49.7.1041 |
| [32] | 张 英. 固相萃取材料评价及环境应用研究[D]. 北京: 中国地质大学(北京), 2009. |
| [33] | Tai SS, Bunk DM, White E 5th, et al. Development and evaluation of a reference measurement procedure for the determination of total 3, 3', 5-triiodothyronine in human serum using isotope-dilution liquid chromatography-tandem mass spectrometry[J]. Anal Chem, 2004, 76(17): 5092-6. doi:10.1021/ac049516h |
| [34] | Li XG, Li S, Kellermann G. Simultaneous determination of three estrogens in human saliva without derivatization or liquid-liquid extraction for routine testing via miniaturized solid phase extraction with LC-MS/MS detection[J]. Talanta, 2018, 178: 464-72. doi:10.1016/j.talanta.2017.09.062 |
| [35] | Gallagher TF, Kraychy S, Fishman J, et al. A comparison of methods for the analysis of estrone, estradiol, and estriol in extracts of human urine[J]. J Biol Chem, 1958, 233(5): 1093-6. doi:10.1016/s0021-9258(19)77345-1 |
| [36] | Jones GRD. Reevaluation of the power of error detection of Westgard multirules[J]. Clin Chem, 2004, 50(4): 762-4. doi:10.1373/clinchem.2003.025585 |
| [37] | 曹 正, 李水军, 沈 敏, 等. 液相色谱串联质谱临床检测方法的开发与验证[J]. 检验医学, 2019, 34(3): 189-96. |
| [38] | Lee KM, Han SM, Lee HJ, et al. Influence of mobile phase composition on the analytical sensitivity of LC-ESI-MS/MS for the concurrent analysis of bisphenols, parabens, chlorophenols, benzophenones, and alkylphenols[J]. Environ Res, 2023, 221: 115305. doi:10.1016/j.envres.2023.115305 |
| [39] | Di Renzo GC, Giardina I, Clerici G, et al. Progesterone in normal and pathological pregnancy[J]. Horm Mol Biol Clin Investig, 2016, 27(1): 35-48. doi:10.1515/hmbci-2016-0038 |
| [40] | Gu LQ, Zhang CN, Luo JX, et al. Efficacy and prognostic factors of combined administration of progesterone and estriol valerate tablets for preventing intrauterine adhesions in patients with early missed abortion following dilation and curettage[J]. Am J Transl Res, 2024, 16(7): 3164-70. doi:10.62347/ameb4153 |
| [41] | Basak S, Varma S, Duttaroy AK. Modulation of fetoplacental growth, development and reproductive function by endocrine disrupters[J]. Front Endocrinol (Lausanne), 2023, 14: 1215353. doi:10.3389/fendo.2023.1215353 |
| [42] | Luo L, Gao C, Fan YJ, et al. Perinatal bisphenol exposure and small-for-gestational-age neonates: the evolving effect of replacements then and now[J]. Environ Sci Technol, 2025, 59(12): 5983-93. doi:10.1021/acs.est.4c13266 |
| [43] | Karahoda R, Toit TD, Fuenzalida B, et al. Landscape of steroid dynamics in pregnancy: insights from the maternal-placental-fetal unit and placental models[J]. Mol Cell Proteomics, 2025, 24(6): 100976. doi:10.1016/j.mcpro.2025.100976 |
| [44] | 黄君婷, 张可珍, 陈蔚琳, 等. 妊娠期雄激素生理作用的研究进展[J]. 中华妇产科杂志, 2023, 58(7): 550-4. |
| [45] | Fowden AL, Vaughan OR, Murray AJ, et al. Metabolic consequences of glucocorticoid exposure before birth[J]. Nutrients, 2022, 14(11): 2304. doi:10.3390/nu14112304 |
| [46] | Zhao CX, He L, Li LJ, et al. Prenatal glucocorticoids exposure and adverse cardiovascular effects in offspring[J]. Front Endocrinol (Lausanne), 2024, 15: 1430334. doi:10.3389/fendo.2024.1430334 |
| [47] | Eberle C, Fasig T, Brüseke F, et al. Impact of maternal prenatal stress by glucocorticoids on metabolic and cardiovascular outcomes in their offspring: a systematic scoping review[J]. PLoS One, 2021, 16(1): e0245386. doi:10.1371/journal.pone.0245386 |
| [48] | Rusidzé M, Gargaros A, Fébrissy C, et al. Estrogen actions in placental vascular morphogenesis and spiral artery remodeling: a comparative view between humans and mice[J]. Cells, 2023, 12(4): 620. doi:10.3390/cells12040620 |
| [49] | Tang C, Jin MY, Ma BB, et al. RGS2 promotes estradiol biosynthesis by trophoblasts during human pregnancy[J]. Exp Mol Med, 2023, 55(1): 240-52. doi:10.1038/s12276-023-00927-z |
| [50] | Lee C, Lee SM, Byun DJ, et al. Maternal signatures of Cortisol in first trimester small-for-gestational age[J]. Reprod Sci, 2022, 29(5): 1498-505. doi:10.1007/s43032-021-00822-w |
| [51] | Yu DN, Wan HF, Tong C, et al. A multi-tissue metabolome atlas of primate pregnancy[J]. Cell, 2024, 187(3): 764-81.e14. doi:10.1016/j.cell.2023.11.043 |
| [52] | Gokulakrishnan K, Mohan V, Srikumar BN. Neurosteroid levels in pregnancy and its implications for mental health: a literature review[J]. Ann Neurosci, 2025: 09727531251351075. doi:10.1177/09727531251351075 |
| [53] | Downie E, Shanmugalingam R, Hennessy A, et al. Assessment and management of primary aldosteronism in pregnancy: a case-control study[J]. J Clin Endocrinol Metab, 2022, 107(8): e3152-8. doi:10.1210/clinem/dgac311 |
| [54] | Mistry HD, Klossner R, Kallol S, et al. Effects of aldosterone on the human placenta: insights from placental perfusion studies[J]. Placenta, 2022, 123: 32-40. doi:10.1016/j.placenta.2022.03.129 |
| [55] | Ronconi V, Turchi F, Zennaro MC, et al. Progesterone increase counteracts aldosterone action in a pregnant woman with primary aldosteronism[J]. Clin Endocrinol (Oxf), 2011, 74(2): 278-9. doi:10.1111/j.1365-2265.2010.03901.x |
| No related articles found! |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||