南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 728-741.doi: 10.12122/j.issn.1673-4254.2026.04.02
• • 上一篇
谢家润1,2( ), 罗燕玉1,2, 夏金金2, 王明1,2(
), 罗燕玉1,2, 夏金金2, 王明1,2( )
)
收稿日期:2025-10-02
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
王明
E-mail:983912829@qq.com;wming1999@163.com
作者简介:谢家润,在读博士研究生,E-mail: 983912829@qq.com
基金资助:
Jiarun XIE1,2( ), Yanyu LUO1,2, Jinjin XIA2, Ming WANG1,2(
), Yanyu LUO1,2, Jinjin XIA2, Ming WANG1,2( )
)
Received:2025-10-02
Online:2026-04-20
Published:2026-04-24
Contact:
Ming WANG
E-mail:983912829@qq.com;wming1999@163.com
Supported by:摘要:
目的 探讨加味清心莲子饮(QISD)对糖尿病肾病(DKD)小鼠肾脏损伤的干预作用及机制。 方法 从GEO数据库中下载GSE193192数据集,筛选由晚期糖基化终末产物诱导的肾小管上皮细胞(HK-2)中差异表达的免疫基因集,利用多种分析预测药理机制。动物实验方面,选用雄性ICR小鼠,先持续性高脂饮食喂养4周后,连续腹腔注射链脲佐菌素5 d,制备DKD模型,连续灌胃低(14.46 g/kg)、中(28.92 g/kg)、高剂量(57.84 g/kg)的加味清心莲子饮12周(1次/6),以达格列净作为阳性对照,8只/组。通过HE、PAS和Masson染色观察DKD小鼠肾脏病理损伤。通过RT-qPCR和Western blotting检测KDM3C、SP1、TNF-α、MCP-1等蛋白的表达水平。在体外实验中,使用脂多糖(LPS)诱导HK-2发生炎症损伤,采用小分子抑制剂干预的方法,探索加味清心莲子饮对细胞炎性损伤的影响。 结果 动物实验结果表明,加味清心莲子饮中剂量组和高剂量组能够显著降低糖尿病肾病小鼠糖化血清蛋白、肌酐和尿素氮水平(P<0.01,P<0.001),减少糖原累积,减轻肾小球肥大,降低炎性细胞浸润。此外,加味清心莲子饮中剂量组和高剂量组能够显著降低小鼠肾脏组织中KDM3C、SP1、TNF-α和MCP-1的基因和蛋白表达(P<0.05,P<0.01,P<0.001)。细胞实验结果显示KDM3C抑制剂JIB-04的应用则能抑制TNF-α、MCP-1和ICAM-1等炎性因子的表达水平(P<0.05,P<0.001)。 结论 加味清心莲子饮可通过抑制糖尿病肾病小鼠的炎症反应改善肾脏损伤,其作用机制可能与抑制KDM3C/SP1信号通路的过度激活有关。
谢家润, 罗燕玉, 夏金金, 王明. 加味清心莲子饮通过调控KDM3C/SP1信号通路改善糖尿病肾病小鼠的肾脏损伤[J]. 南方医科大学学报, 2026, 46(4): 728-741.
Jiarun XIE, Yanyu LUO, Jinjin XIA, Ming WANG. Jia Wei Qingxin Lotus Seed Drink improves diabetic kidney disease in mice by regulating the KDM3C/SP1 signaling pathway[J]. Journal of Southern Medical University, 2026, 46(4): 728-741.
| Gene name | Sequence (5ʹ-3ʹ) |
|---|---|
| KDM3C (MOUSE) | Forward Primer:CACATTCTTGGATCTGTGACCA |
| Reverse Primer:ATGCTGTCTTTGCAGTTGAGG | |
| SP1 (MOUSE) | Forward Primer:TGCAAACCAACAGATCATCCC |
| Reverse Primer:TGACAGGTAGCAAGGTGATGT | |
| MCP-1 (MOUSE) | Forward Primer:TAAAAACCTGGATCGGAACCAAA |
| Reverse Primer:GCATTAGCTTCAGATTTACGGGT | |
| TNF-α (MOUSE) | Forward Primer:CCTGTAGCCCACGTCGTAG |
| Reverse Primer:GGGAGTAGACAAGGTACAACCC | |
| β-actin (MOUSE) | Forward Primer:GTGACGTTGACATCCGTAAAGA |
| Reverse Primer:GCCGGACTCATCGTACTCC | |
| KDM3C (HUMAN) | Forward Primer:GAAGCGGAAGTCTGTTGACAC |
| Reverse Primer:TGTGGGTGGTCTGGATACAAA | |
| SP1 (HUMAN) | Forward Primer:AGTTCCAGACCGTTGATGGG |
| Reverse Primer:GTTTGCACCTGGTATGATCTGT | |
| ICAM-1 (HUMAN) | Forward Primer:TTGGGCATAGAGACCCCGTT |
| Reverse Primer:GCACATTGCTCAGTTCATACACC | |
| MCP-1 (HUMAN) | Forward Primer:CAGCCAGATGCAATCAATGCC |
| Reverse Primer:TGGAATCCTGAACCCACTTCT | |
| TNF-α (HUMAN) | Forward Primer:CCTCTCTCTAATCAGCCCTCTG |
| Reverse Primer:GAGGACCTGGGAGTAGATGAG | |
| β-actin (HUMAN) | Forward Primer:CATGTACGTTGCTATCCAGGC |
| Reverse Primer:CTCCTTAATGTCACGCACGAT |
表1 RT-qPCR 的引物序列
Tab.1 Primer sequences for RT-qPCR
| Gene name | Sequence (5ʹ-3ʹ) |
|---|---|
| KDM3C (MOUSE) | Forward Primer:CACATTCTTGGATCTGTGACCA |
| Reverse Primer:ATGCTGTCTTTGCAGTTGAGG | |
| SP1 (MOUSE) | Forward Primer:TGCAAACCAACAGATCATCCC |
| Reverse Primer:TGACAGGTAGCAAGGTGATGT | |
| MCP-1 (MOUSE) | Forward Primer:TAAAAACCTGGATCGGAACCAAA |
| Reverse Primer:GCATTAGCTTCAGATTTACGGGT | |
| TNF-α (MOUSE) | Forward Primer:CCTGTAGCCCACGTCGTAG |
| Reverse Primer:GGGAGTAGACAAGGTACAACCC | |
| β-actin (MOUSE) | Forward Primer:GTGACGTTGACATCCGTAAAGA |
| Reverse Primer:GCCGGACTCATCGTACTCC | |
| KDM3C (HUMAN) | Forward Primer:GAAGCGGAAGTCTGTTGACAC |
| Reverse Primer:TGTGGGTGGTCTGGATACAAA | |
| SP1 (HUMAN) | Forward Primer:AGTTCCAGACCGTTGATGGG |
| Reverse Primer:GTTTGCACCTGGTATGATCTGT | |
| ICAM-1 (HUMAN) | Forward Primer:TTGGGCATAGAGACCCCGTT |
| Reverse Primer:GCACATTGCTCAGTTCATACACC | |
| MCP-1 (HUMAN) | Forward Primer:CAGCCAGATGCAATCAATGCC |
| Reverse Primer:TGGAATCCTGAACCCACTTCT | |
| TNF-α (HUMAN) | Forward Primer:CCTCTCTCTAATCAGCCCTCTG |
| Reverse Primer:GAGGACCTGGGAGTAGATGAG | |
| β-actin (HUMAN) | Forward Primer:CATGTACGTTGCTATCCAGGC |
| Reverse Primer:CTCCTTAATGTCACGCACGAT |
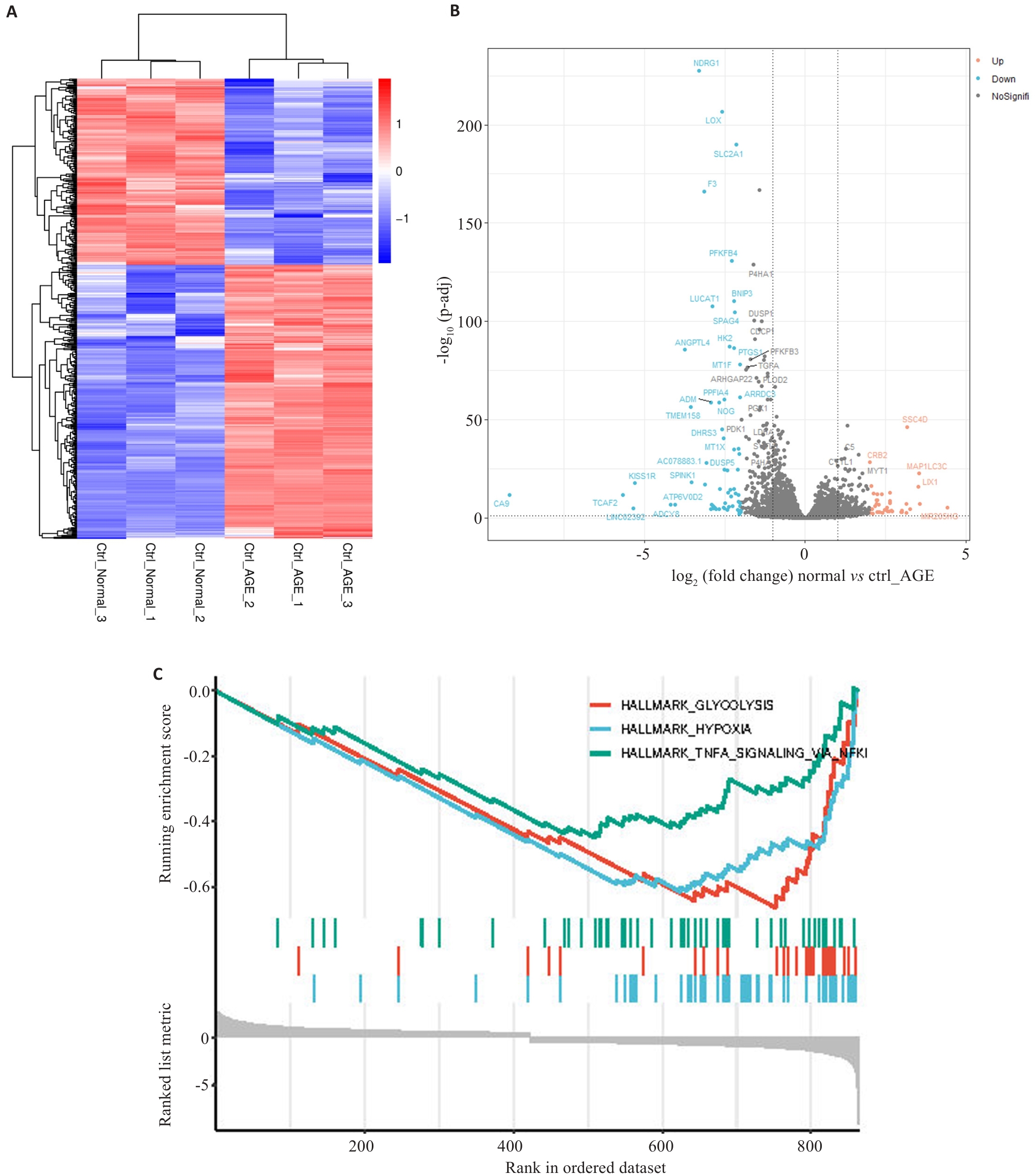
图1 AGEs诱导的肾小管上皮细胞HK-2的差异分析和基因集富集分析(GSEA)
Fig.1 Differential analysis and gene set enrichment analysis (GSEA) for HK-2 renal tubular epithelial cells induced by advanced glycation end products. A: Heat map of analysis of variance between shCtrl-Normal and shCtrl-AGEs groups. B: Volcano plots of shCtrl-Normal and shCtrl-AGEs groups in analysis of variance. Blue dots represent low expression and red dots represent high expression. C: The top 3 terms shown in GSEA.
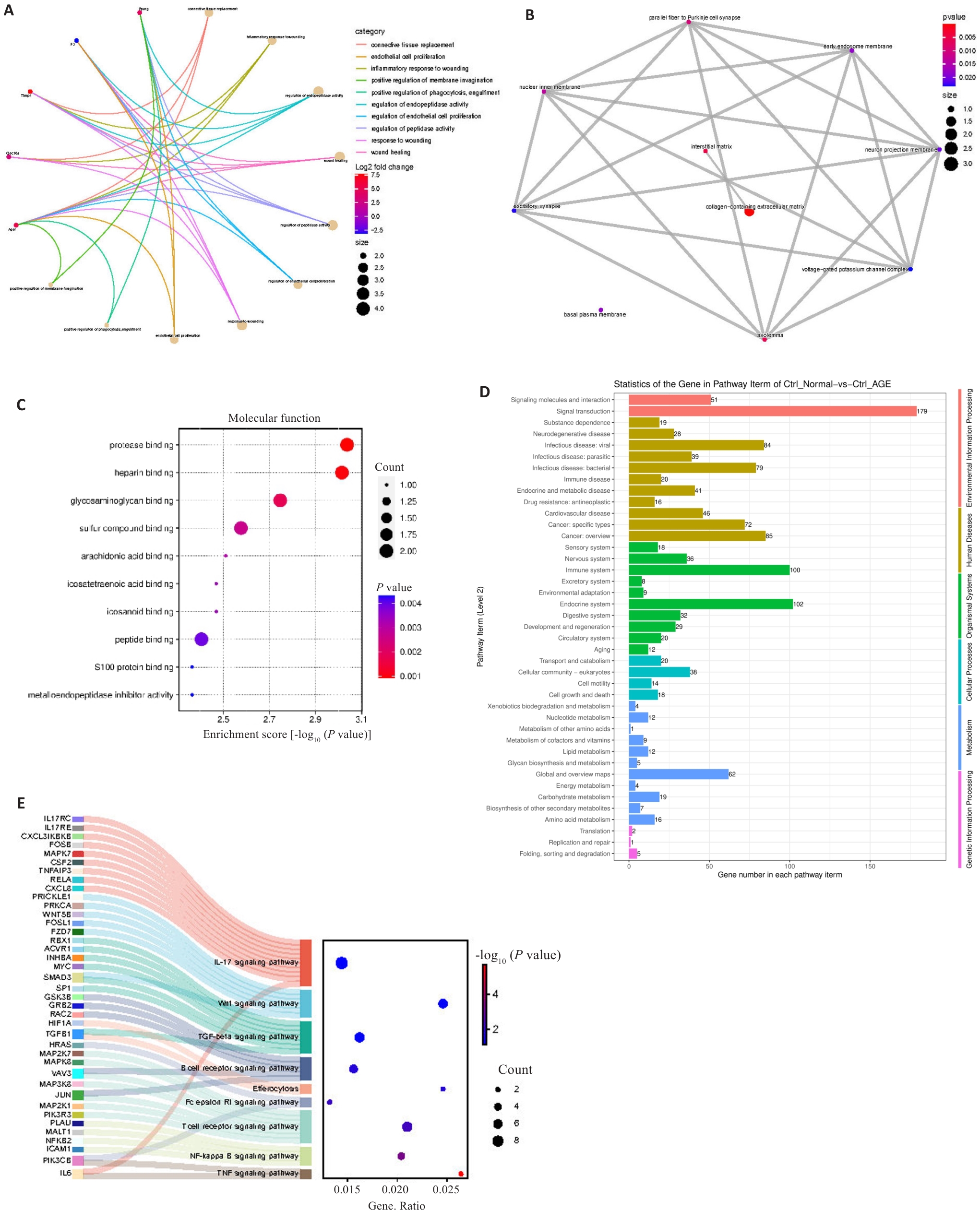
图2 基因本体论(GO)和京都基因和基因组百科全书(KEGG)
Fig.2 Gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses. A: Biological process (BP) functional enrichment in GO analysis. B: Cell component (CC) functional enrichment in GO analysis. C: Molecular function (MF) function enrichment in GO analysis. D: Functional enrichment in WEGO analysis. E: Pathway enrichment in KEGG analysis.
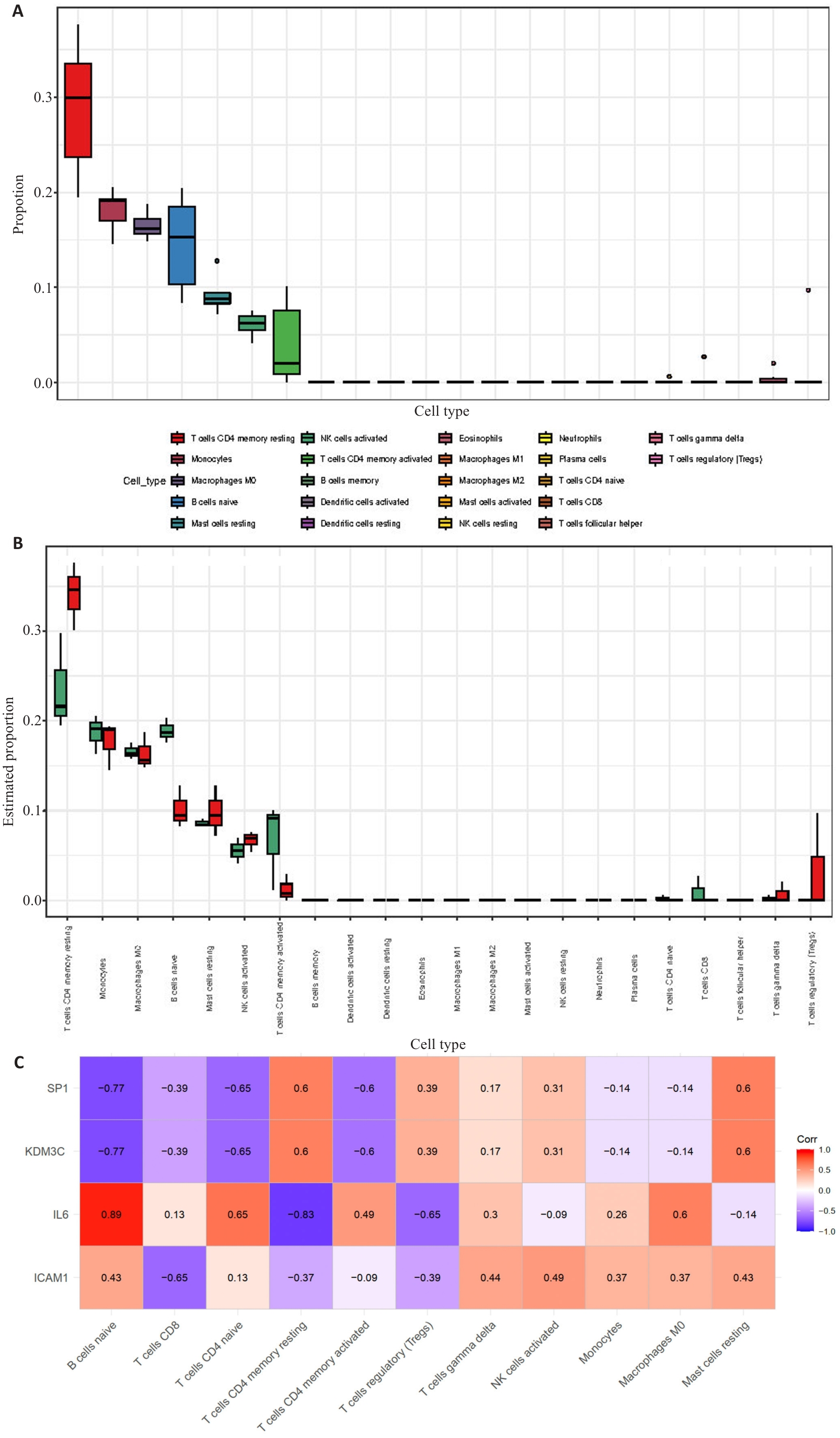
图3 免疫浸润分析
Fig.3 Immune infiltration analysis. A: Bar graph of the proportions of 22 immune cells. B: Box-and-line plot showing comparison of the proportions of 22 immune cells in DKD group versus control group. C: Correlation of immune cells with 4 key genes.
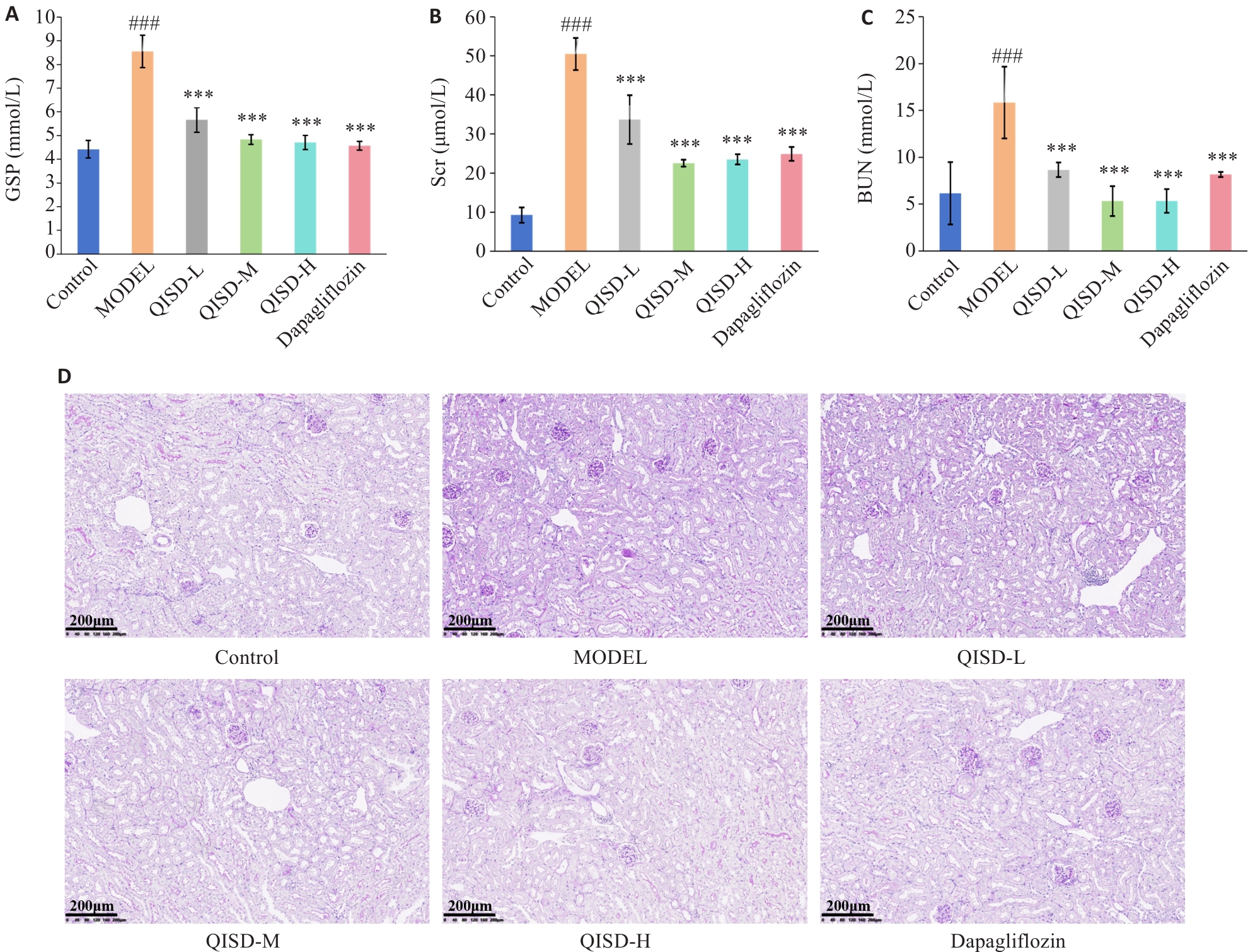
图4 各组小鼠血清GSP、Scr、BUN和肾脏组织糖原沉积比较
Fig.4 Serum GSP, Scr, and BUN levels and renal glycogen deposition in mice in each group. A: Effect of QISD on glycated serum proteins in DKD mice (n=3). B: Effect of QISD on serum creatinine in DKD mice (n=3). C: Effect of QISD on serum urea nitrogen in DKD mice (n=3). D: PAS staining of the renal tissues of the mice (Original magnification: ×200) (n=6). ###P<0.001 vs control group; ***P<0.001 vs model group.
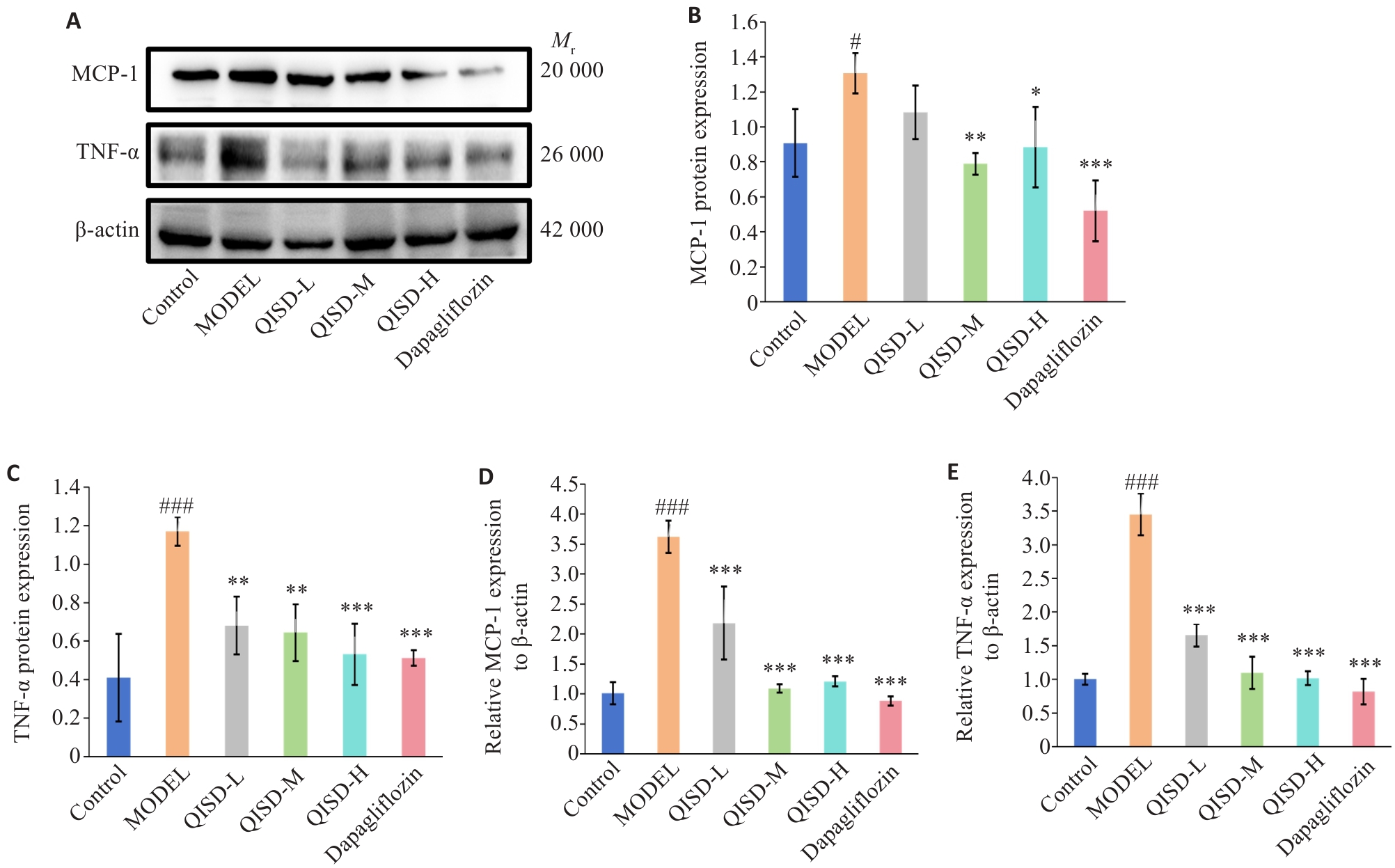
图6 各组小鼠肾脏组织炎症因子表达量的比较
Fig.6 Expression of inflammatory factors in the kidney tissues of the mice in each group. A: Western blotting of TNF-α and MCP-1 proteins in renal tissues of the mice in each group. B, C: Quantitative analysis of the expression levels of TNF-α and MCP-1 proteins (n=3). D, E: RT-qPCR for detecting TNF-α and MCP-1 mRNA expressions in the renal tissues of the mice in each group (n=3). #P<0.05, ###P<0.001 vs control group; *P<0.05, **P<0.01, ***P<0.001 vs model group.
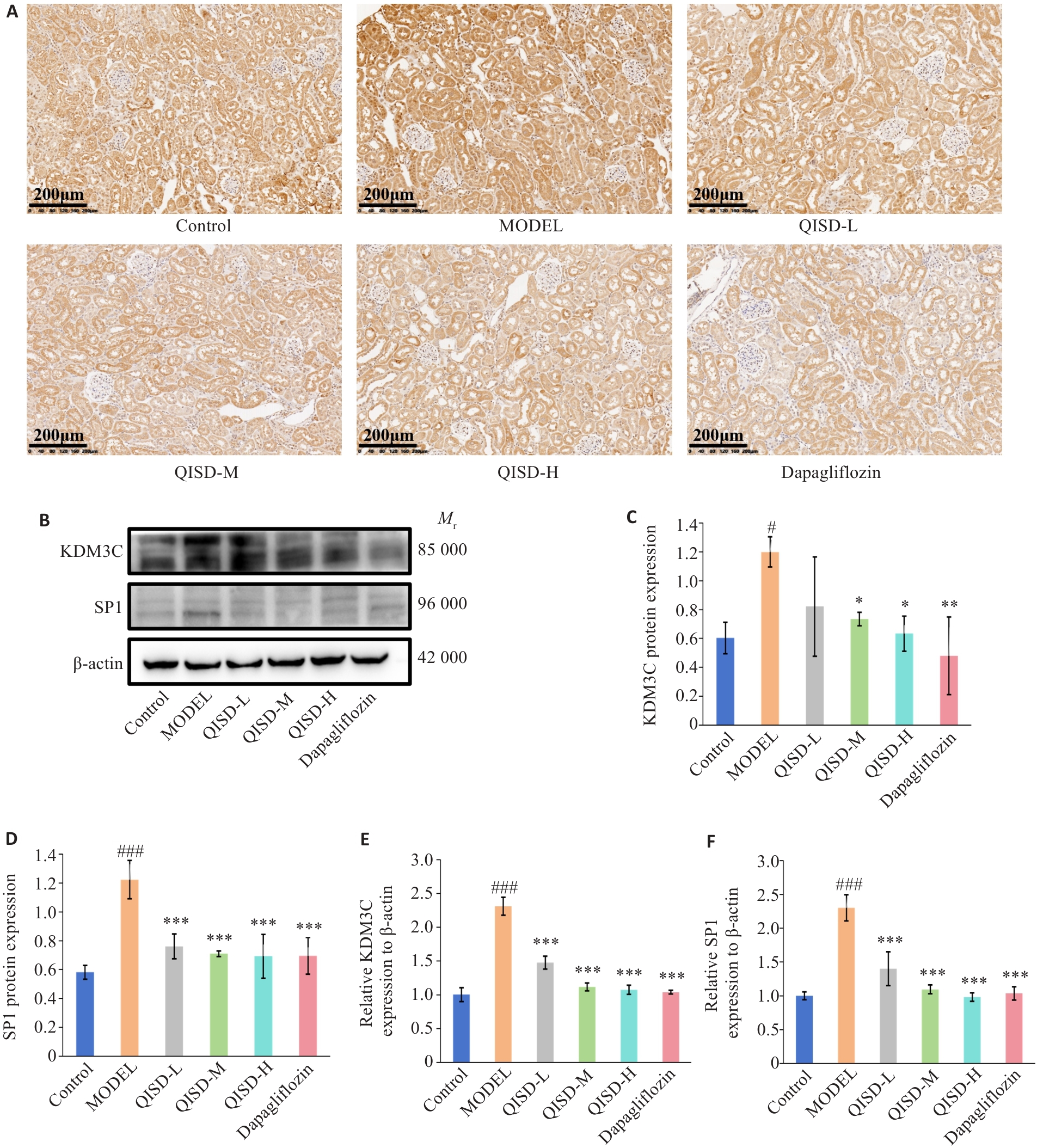
图7 各组小鼠肾脏组织KDM3C和SP1表达量的比较
Fig.7 Expression of KDM3C and SP1 in the kidney tissues of the mice in each group. A: Immunohistochemical staining for detecting KDM3C expression in the kidney tissues of the mice in each group (×200) (n=3). B: Western blotting for detecting protein expressions of KDM3C and SP1 in the kidney tissues of the mice in each group. C, D: Quantitative analysis of the expression levels of KDM3C and SP1 proteins (n=3). E, F: RT-qPCR for detecting expressions of KDM3C and SP1 mRNA in the kidney tissues of the mice in each group (n=3). #P<0.05, ###P<0.001 vs control group; *P<0.05, **P<0.01, ***P<0.001 vs model group.
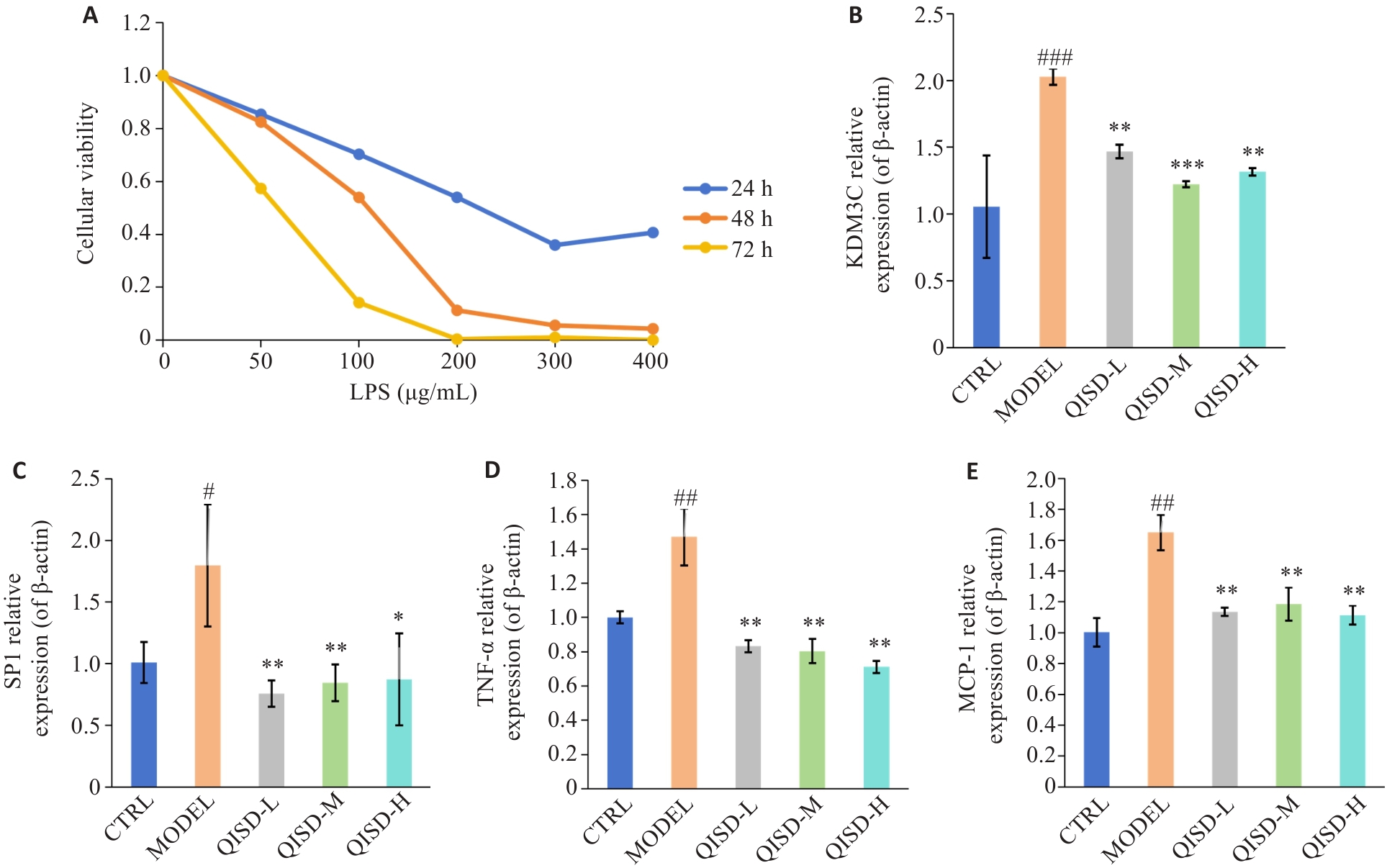
图8 QISD对LPS诱导的HK-2细胞炎症损伤的影响
Fig.8 Effect of QISD on LPS-induced inflammatory damage in HK-2 cells. A: Viability of HK-2 cells after treatment with different LPS concentrations (50, 100, 200, 300, and 400 μg/mL) for 24, 48, and 72 h (n=6) determined by CCK8 assay. B-E: RT-qPCR for detecting mRNA expressions of KDM3C, SP1, TNF-α and MCP-1 in HK-2 cells (n=3). #P<0.05, ##P<0.01, ###P<0.001 vs control group; *P<0.05, **P<0.01, ***P<0.001 vs model group.
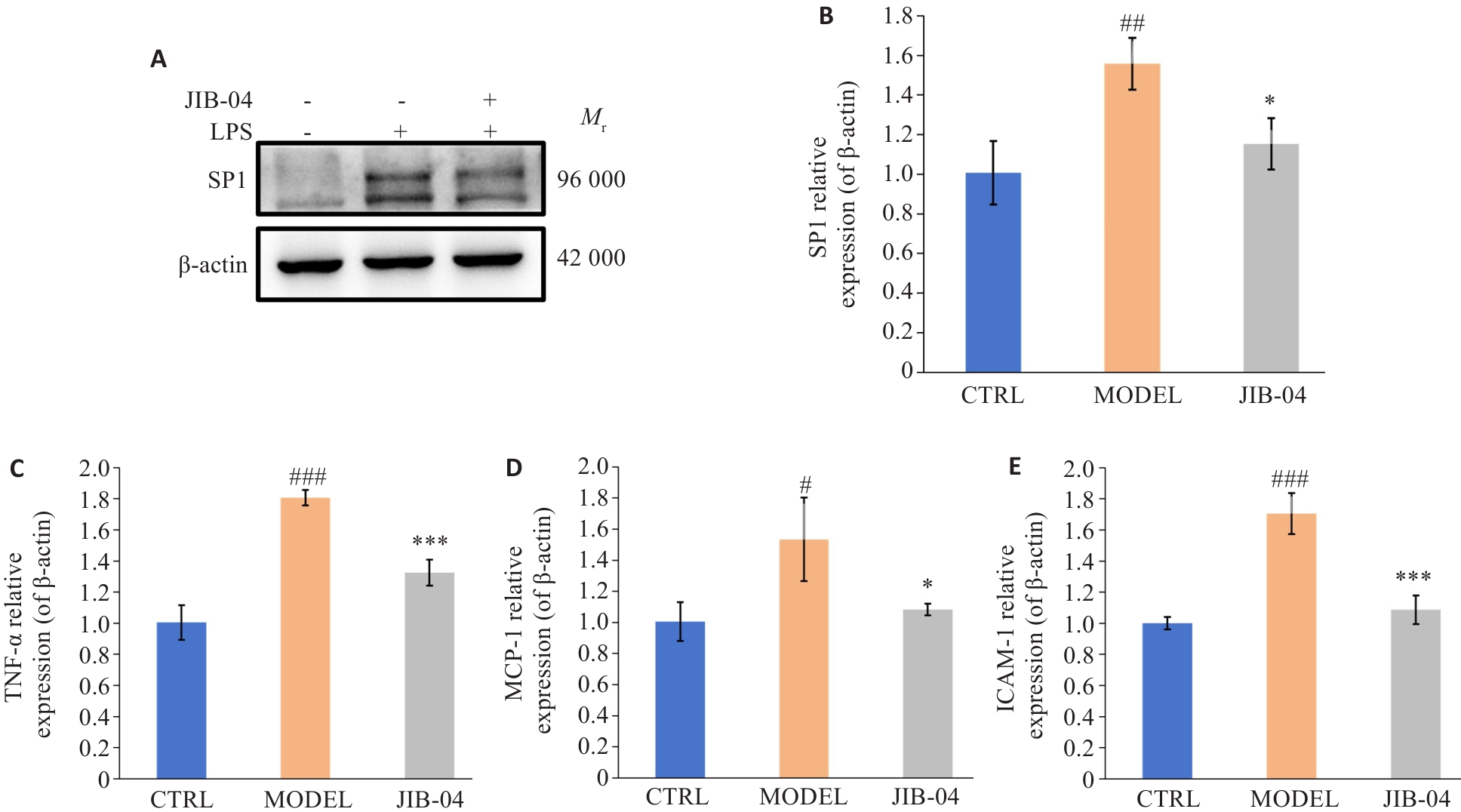
图9 在JIB-04作用下SP1、TNF-α、MCP-1和ICAM-1的表达水平变化
Fig.9 Changes in expression levels of SP1, TNF-α, MCP-1 and ICAM-1 in LPS-induced HK-2 cells after treatment with JIB-04. A: Western blotting protein bands of SP1 in HK-2 cells. B-E: RT-qPCR for detecting mRNA expressions of SP1, TNF-α, MCP-1 and ICAM-1 in HK-2 cells (n=3). #P<0.05, ##P<0.01, ###P<0.001 vs control group; *P<0.05, ***P<0.001 vs model group.
| [1] | Mishra DD, Maurya PK, Tiwari S. Reference gene panel for urinary exosome-based molecular diagnostics in patients with kidney disease[J]. World J Nephrol, 2024, 13(3): 99105. doi:10.5527/wjn.v13.i3.99105 |
| [2] | Wang H, Liu DW, Zheng B, et al. Emerging role of ferroptosis in diabetic kidney disease: molecular mechanisms and therapeutic opportunities[J]. Int J Biol Sci, 2023, 19(9): 2678-94. doi:10.7150/ijbs.81892 |
| [3] | Megur A, Daliri EB, Baltriukienė D, et al. Prebiotics as a tool for the prevention and treatment of obesity and diabetes: classification and ability to modulate the gut microbiota[J]. Int J Mol Sci, 2022, 23(11): 6097. doi:10.3390/ijms23116097 |
| [4] | Cho NH, Shaw JE, Karuranga S, et al. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045[J]. Diabetes Res Clin Pract, 2018, 138: 271-81. doi:10.1016/j.diabres.2018.02.023 |
| [5] | Li H, Chen H, Gao R, et al. Traditional Chinese medicine formulae and Chinese patent medicines for the treatment of diabetic kidney disease: efficacies and mechanisms[J]. Am J Chin Med, 2025, 53(3): 675-707. doi:10.1142/s0192415x25500260 |
| [6] | Liu J, Ren J, Zhou LL, et al. Proteomic and lipidomic analysis of the mechanism underlying astragaloside IV in mitigating ferroptosis through hypoxia-inducible factor 1α/heme oxygenase 1 pathway in renal tubular epithelial cells in diabetic kidney disease[J]. J Ethnopharmacol, 2024, 334: 118517. doi:10.1016/j.jep.2024.118517 |
| [7] | Shen Q, Fang J, Guo HJ, et al. Astragaloside IV attenuates podocyte apoptosis through ameliorating mitochondrial dysfunction by up-regulated Nrf2-ARE/TFAM signaling in diabetic kidney disease[J]. Free Radic Biol Med, 2023, 203: 45-57. doi:10.1016/j.freeradbiomed.2023.03.022 |
| [8] | Zhang XX, Chen JC, Lin RH, et al. Lactate drives epithelial-mesenchymal transition in diabetic kidney disease via the H3K14la/KLF5 pathway[J]. Redox Biol, 2024, 75: 103246. doi:10.1016/j.redox.2024.103246 |
| [9] | Chakraborty S, Nandi P, Mishra J, et al. Molecular mechanisms in regulation of autophagy and apoptosis in view of epigenetic regulation of genes and involvement of liquid-liquid phase separation[J]. Cancer Lett, 2024, 587: 216779. doi:10.1016/j.canlet.2024.216779 |
| [10] | Chao CT, Kuo FC, Lin SH. Epigenetically regulated inflammation in vascular senescence and renal progression of chronic kidney disease[J]. Semin Cell Dev Biol, 2024, 154: 305-15. doi:10.1016/j.semcdb.2022.09.012 |
| [11] | Kato M, Natarajan R. Epigenetics and epigenomics in diabetic kidney disease and metabolic memory[J]. Nat Rev Nephrol, 2019, 15(6): 327-45. doi:10.1038/s41581-019-0135-6 |
| [12] | Mimura I, Chen Z, Natarajan R. Epigenetic alterations and memory: key players in the development/progression of chronic kidney disease promoted by acute kidney injury and diabetes[J]. Kidney Int, 2025, 107(3): 434-56. doi:10.1016/j.kint.2024.10.031 |
| [13] | Zhao M, Wang ST, Zuo AN, et al. HIF-1α/JMJD1A signaling regulates inflammation and oxidative stress following hypergl-ycemia and hypoxia-induced vascular cell injury[J]. Cell Mol Biol Lett, 2021, 26(1): 40. doi:10.1186/s11658-021-00283-8 |
| [14] | 张丽香, 王 钢, 丘余良, 等. 清心莲子饮加减治疗糖尿病肾脏病Ⅲ~Ⅳ期气阴两虚证的临床观察[J]. 2024(9): 778-82. |
| [15] | 周 霄, 邹 迪, 张守琳, 等. 清心莲子饮加减在慢性肾脏病的研究进展[J].中医药学报, 2022, 50(10): 97-100. doi:10.19664/j.cnki.1002-2392.220235 |
| [16] | 李文超, 李 雪. 清心莲子饮加减治疗对早期糖尿病肾病的临床观察[J]. 2021(1): 22-3. |
| [17] | Lu J, Li XQ, Chen PP, et al. Acetyl-CoA synthetase 2 promotes diabetic renal tubular injury in mice by rewiring fatty acid metabolism through SIRT1/ChREBP pathway[J]. Acta Pharmacol Sin, 2024, 45(2): 366-77. doi:10.1038/s41401-023-01160-0 |
| [18] | Li BJ, Xia YQ, Mei SQ, et al. Histone H3K27 methyltransferase EZH2 regulates apoptotic and inflammatory responses in sepsis-induced AKI[J]. Theranostics, 13(6): 1860-75. doi:10.7150/thno.83353 |
| [19] | Jiang WJ, Xu CT, Du CL, et al. Tubular epithelial cell-to-macrophage communication forms a negative feedback loop via extracellular vesicle transfer to promote renal inflammation and apoptosis in diabetic nephropathy[J]. Theranostics, 12(1): 324-39. doi:10.7150/thno.63735 |
| [20] | Hickey FB, Martin F. Role of the immune system in diabetic kidney disease[J]. Curr Diabetes Rep, 2018, 18(4): 20. doi:10.1007/s11892-018-0984-6 |
| [21] | Zheng W, Guo J, Liu ZS. Effects of metabolic memory on inflammation and fibrosis associated with diabetic kidney disease: an epigenetic perspective[J]. Clin Epigenet, 2021, 13(1): 87. doi:10.1186/s13148-021-01079-5 |
| [22] | Wang ST, Zuo AN, Jiang WQ, et al. JMJD1A/NR4A1 signaling regulates the procession of renal tubular epithelial interstitial fibrosis induced by AGEs in HK-2[J]. Front Med, 2022, 8: 807694. doi:10.3389/fmed.2021.807694 |
| [23] | Hung PH, Hsu YC, Chen TH, et al. The histone demethylase inhibitor GSK-J4 is a therapeutic target for the kidney fibrosis of diabetic kidney disease via DKK1 modulation[J]. Int J Mol Sci, 2022, 23(16): 9407. doi:10.3390/ijms23169407 |
| [24] | Li CH, Zhou Y, Tu PF, et al. Natural carbazole alkaloid murrayafoline A displays potent anti-neuroinflammatory effect by directly targeting transcription factor Sp1 in LPS-induced microglial cells[J]. Bioorg Chem, 2022, 129: 106178. doi:10.1016/j.bioorg.2022.106178 |
| [25] | Wang R, Yang Y, Wang H, et al. miR-29c protects against inflammation and apoptosis in Parkinson's disease model in vivo and in vitro by targeting SP1[J]. Clin Exp Pharmacol Physiol, 2020, 47(3): 372-82. doi:10.1111/1440-1681.13212 |
| [26] | Medrano-Bosch M, Simón-Codina B, Jiménez W, et al. Monocyte-endothelial cell interactions in vascular and tissue remodeling[J]. Front Immunol, 2023, 14: 1196033. doi:10.3389/fimmu.2023.1196033 |
| [27] | Liu XQ, Jiang L, Li YY, et al. Wogonin protects glomerular podocytes by targeting Bcl-2-mediated autophagy and apoptosis in diabetic kidney disease[J]. Acta Pharmacol Sin, 2022, 43(1): 96-110. doi:10.1038/s41401-021-00721-5 |
| [28] | Li XH, Dong X, Zhang LY, et al. Astragaloside IV attenuates renal tubule injury in DKD rats via suppression of CD36-mediated NLRP3 inflammasome activation[J]. Front Pharmacol, 2024, 15: 1285797. doi:10.3389/fphar.2024.1285797 |
| [29] | Zhang QL, Liu XC, Sullivan MA, et al. Protective effect of yi Shen Pai du formula against diabetic kidney injury via inhibition of oxidative stress, inflammation, and epithelial-to-mesenchymal transition in db/db mice[J]. Oxid Med Cell Longev, 2021, 2021: 7958021. doi:10.1155/2021/7958021 |
| [30] | Wang JY, Shi HN, Yang Y, et al. Crosstalk between ferroptosis and innate immune in diabetic kidney disease: mechanisms and therapeutic implications[J]. Front Immunol, 2025, 16: 1505794. doi:10.3389/fimmu.2025.1505794 |
| [31] | Wang J, Fite BZ, Kare AJ, et al. Multiomic analysis for optimization of combined focal and immunotherapy protocols in murine pancreatic cancer[J]. Theranostics, 2022, 12(18): 7884-902. doi:10.7150/thno.73218 |
| [1] | 刘泽, 毛樟坤, 尤达, 王俊杰, 何咏梅, 余伊雯, 文志强, 方会龙, 何汶霞. 宽缨酮靶向抑制STAT3减轻线粒体功能障碍和炎症缓解急性肾损伤[J]. 南方医科大学学报, 2026, 46(3): 570-581. |
| [2] | 陈雪俊, 经媛, 梁惠瑜, 钟竞颖, 陈泽栋, 彭于芝, 戴娇娇, 肖雅. 不同配伍黄芪药对通过PTGS2调控脂质过氧化改善小鼠糖尿病肾病[J]. 南方医科大学学报, 2026, 46(3): 592-603. |
| [3] | 陈梅妹, 黄睿娜, 杨朝阳. 血浆代谢物介导炎症蛋白对阿尔茨海默病的因果效应:一项孟德尔随机化分析研究[J]. 南方医科大学学报, 2026, 46(2): 278-285. |
| [4] | 尹林, 张可妮, 乔通, 牛民主, 殷丽霞, 刘馨悦, 耿志军, 李静, 胡建国. 藜芦酸通过激活Nrf2/HO-1信号通路减轻氧化应激缓解葡聚糖硫酸钠诱导的小鼠实验性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 403-411. |
| [5] | 尹梦霞, 李蕊臣, 杨凯博, 焦伟杰, 刘新光. 利奈唑胺联用血必净通过抑制Hla-NLRP3通路减轻MRSA肺炎小鼠的炎症损伤[J]. 南方医科大学学报, 2026, 46(2): 412-422. |
| [6] | 黄林林, 郑旺, 胡建国, 宋雪, 陶露, 耿志军, 李静, 左芦根, 葛思堂. 马鞭草苷通过抑制PI3K-AKT通路减轻肠上皮炎症改善小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2026, 46(2): 423-433. |
| [7] | 乔通, 尹林, 张可妮, 牛民主, 黄菊, 耿志军, 李静, 胡建国. 茯苓新酸A通过调节AMPK/mTOR介导的自噬来减轻葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2026, 46(1): 131-140. |
| [8] | 徐嘉艺, 杨迪, 臧开来, 褚孟恩, 赵庆瑶, 李晴, 鲁森, 陈修丽, 李宁. EVA1A过表达通过调节脂质代谢和促进脂滴自噬改善非酒精性脂肪肝[J]. 南方医科大学学报, 2026, 46(1): 150-158. |
| [9] | 张淑芬, 黄添容, 杨灿洪, 陈家镒, 吕田明, 张嘉发. 莱菔硫烷通过抑制Aβ42寡聚体激活的U87细胞中MAPK/NF-κB信号通路降低反应性星形胶质细胞介导的SH-SY5Y凋亡[J]. 南方医科大学学报, 2026, 46(1): 191-199. |
| [10] | 沙桐, 王文研, 宣佳斌, 吴洁, 石能贤, 何劲, 胡鸿彬, 张耀元. 基于Th1/Th2细胞因子检测的脓毒症免疫状态分型及预后分析:一项回顾性研究[J]. 南方医科大学学报, 2026, 46(1): 6-22. |
| [11] | 杨勤军, 朱虹宇, 高远, 杨程, 刘桐, 张璐, 童佳兵, 李泽庚. 桑麻止咳方改善感染后咳嗽大鼠气道炎症和敏感性:基于TRPV1-SP/CGRP和细胞焦亡途径[J]. 南方医科大学学报, 2025, 45(9): 1830-1839. |
| [12] | 闫爱丽, 罗梦瑶, 常晋瑞, 李新华, 朱娟霞. 橙皮素通过调控AMPK/NLRP3通路减轻阿霉素诱导的小鼠心肌毒性[J]. 南方医科大学学报, 2025, 45(9): 1850-1858. |
| [13] | 罗善玉, 朱强, 闫玉翡, 纪宗红, 邹华杰, 张瑞霞, 巴应贵. 低氧环境下NLRP3信号通路促进非酒精性脂肪性肝炎小鼠的肝细胞焦亡[J]. 南方医科大学学报, 2025, 45(9): 2026-2033. |
| [14] | 刘辰菲, 张玮, 曾尧, 梁艳, 王梦婷, 张明芳, 李新元, 王凤超, 杨燕青. 2,6-二甲氧基-1,4-苯醌通过抑制NLRP3炎症小体活化改善葡聚糖硫酸钠诱导的小鼠溃疡性结肠炎[J]. 南方医科大学学报, 2025, 45(8): 1654-1662. |
| [15] | 尚菲菲, 师晓可, 曾尧, 陶循浅, 李天真, 梁艳, 杨燕青, 宋传旺. Avitinib抑制NLRP3炎症小体活化并改善小鼠感染性休克[J]. 南方医科大学学报, 2025, 45(8): 1697-1705. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||