南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 592-603.doi: 10.12122/j.issn.1673-4254.2026.03.13
陈雪俊1( ), 经媛2, 梁惠瑜1, 钟竞颖1, 陈泽栋1, 彭于芝1, 戴娇娇2(
), 经媛2, 梁惠瑜1, 钟竞颖1, 陈泽栋1, 彭于芝1, 戴娇娇2( ), 肖雅1(
), 肖雅1( )
)
收稿日期:2025-09-17
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
戴娇娇,肖雅
E-mail:1962889575@qq.com;djj52092@smu.edu.cn;xiaoya0527@126.com
作者简介:陈雪俊,在读硕士研究生,E-mail: 1962889575@qq.com
基金资助:
Xuejun CHEN1( ), Yuan JING2, Huiyu LIANG1, Jingying ZHONG1, Zedong CHEN1, Yuzhi PENG1, Jiaojiao DAI2(
), Yuan JING2, Huiyu LIANG1, Jingying ZHONG1, Zedong CHEN1, Yuzhi PENG1, Jiaojiao DAI2( ), Ya XIAO1(
), Ya XIAO1( )
)
Received:2025-09-17
Online:2026-03-20
Published:2026-03-26
Contact:
Jiaojiao DAI, Ya XIAO
E-mail:1962889575@qq.com;djj52092@smu.edu.cn;xiaoya0527@126.com
Supported by:摘要:
目的 探究芪丹地黄颗粒中不同黄芪药对(黄芪丹参药对、黄芪地黄药对、黄芪山药药对)调节糖尿病肾病(DKD)小鼠PTGS2介导的脂质过氧化的不同影响。 方法 通过网络药理学方法筛选芪丹地黄颗粒中黄芪、丹参、生地黄和山药的活性成分及其作用靶点,构建药对-有效成分-靶点网络,并与DKD相关靶点进行交集分析,构建PPI蛋白互作网络和富集分析。利用分子对接技术验证关键活性成分与PTGS2的结合能力。通过动物实验评估不同黄芪药对对DKD小鼠的影响。30只小鼠随机分为6组(n=5),包括正常对照组、模型组、厄贝沙坦组(50 mg/kg/d)及3个黄芪药对组(黄芪丹参、黄芪地黄、黄芪山药,给药剂量均为6 g/kg/d)。除正常组外,其余各组采用链脲佐菌素(50 mg/kg)诱导DKD模型,造模成功后给予相应药物干预8周。 结果 网络药理学结果显示芪丹地黄颗粒中各药对的活性成分与DKD相关靶点存在广泛交集,PTGS2为关键靶点。主要活性成分与PTGS2具有良好的结合亲和力。与模型组相比,黄芪-丹参组和黄芪-地黄组小鼠的体质量、空腹血糖、血清肌酐、尿素氮和24 h尿白蛋白水平改善(P<0.05),其中黄芪-丹参组改善最显著;而黄芪-山药组仅在血糖和肌酐水平方面有轻微改善(P<0.05),对尿素氮和尿白蛋白无统计学差异(P>0.05)。PTGS2蛋白表达在模型组升高(P<0.01),黄芪-丹参组显著下调其表达(P<0.01)。黄芪-丹参组显著提高SOD活性(P<0.01),降低MDA含量(P<0.01)并上调GPX4表达(P<0.01);黄芪-地黄组改善作用相对较弱,黄芪-山药组效果最差。 结论 芪丹地黄颗粒中3个黄芪药对可在一定程度上通过PTGS2介导的脂质过氧化改善糖尿病肾病,其中黄芪丹参药对效果最为显著。
陈雪俊, 经媛, 梁惠瑜, 钟竞颖, 陈泽栋, 彭于芝, 戴娇娇, 肖雅. 不同配伍黄芪药对通过PTGS2调控脂质过氧化改善小鼠糖尿病肾病[J]. 南方医科大学学报, 2026, 46(3): 592-603.
Xuejun CHEN, Yuan JING, Huiyu LIANG, Jingying ZHONG, Zedong CHEN, Yuzhi PENG, Jiaojiao DAI, Ya XIAO. Different Astragalus medicinal pairs improve diabetic nephropathy in mice by regulating lipid peroxidation through PTGS2[J]. Journal of Southern Medical University, 2026, 46(3): 592-603.
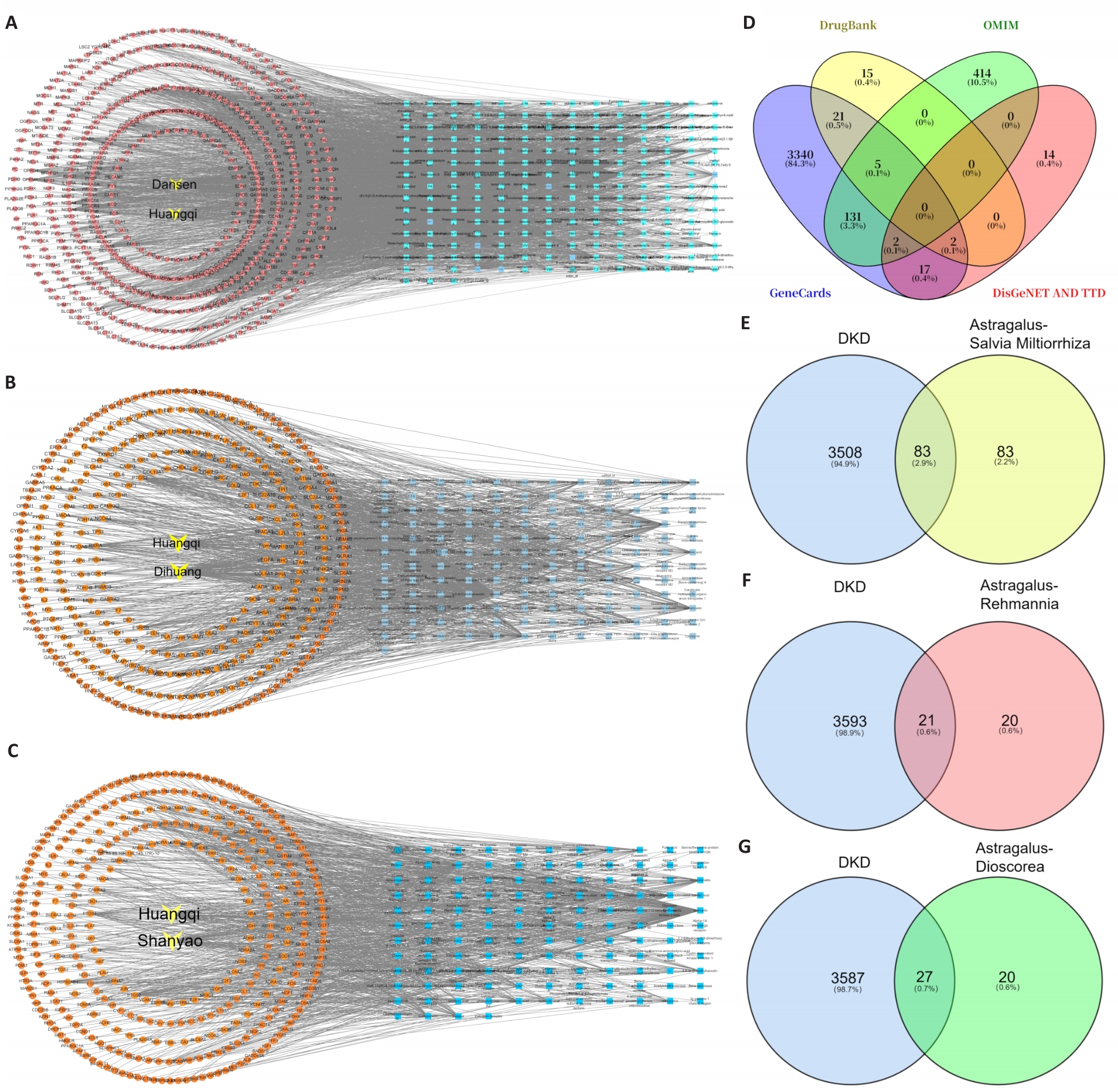
图1 筛选各黄芪药对治疗糖尿病肾病的有效成分和靶点
Fig.1 Identification of the effective components and targets of each Astragalus prescription for treatment of diabetic kidney disease (DKD). A-C: Bioactive components of each Astragalus herb pair and their targets. D: Disease-related targets of DKD screened from multiple databases. E-G: Intersection gene targets between each Astragalus herb pair and DKD.
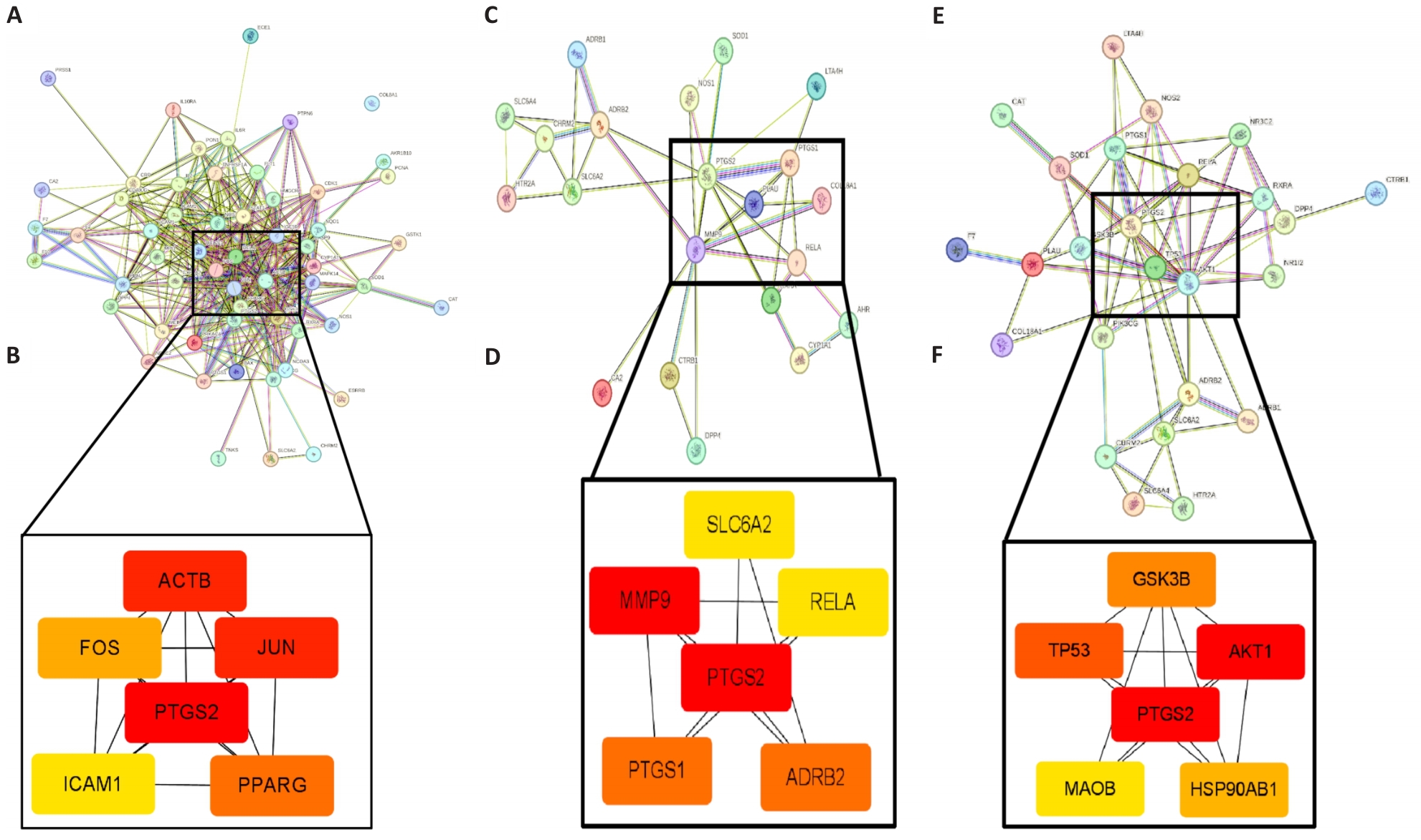
图2 筛选各黄芪药对治疗糖尿病肾病的核心靶点
Fig.2 Identification of the core targets of different Astragalus prescriptions for treatment of DKD. A, C, E: Protein-protein interaction (PPI) networks of shared genes between DKD and the herb pairs Astragalus-Salvia miltiorrhiza, Astragalus-Rehmannia, and Astragalus-Dioscorea, respectively. B, D, F: Core therapeutic targets of each herb pair against DKD.
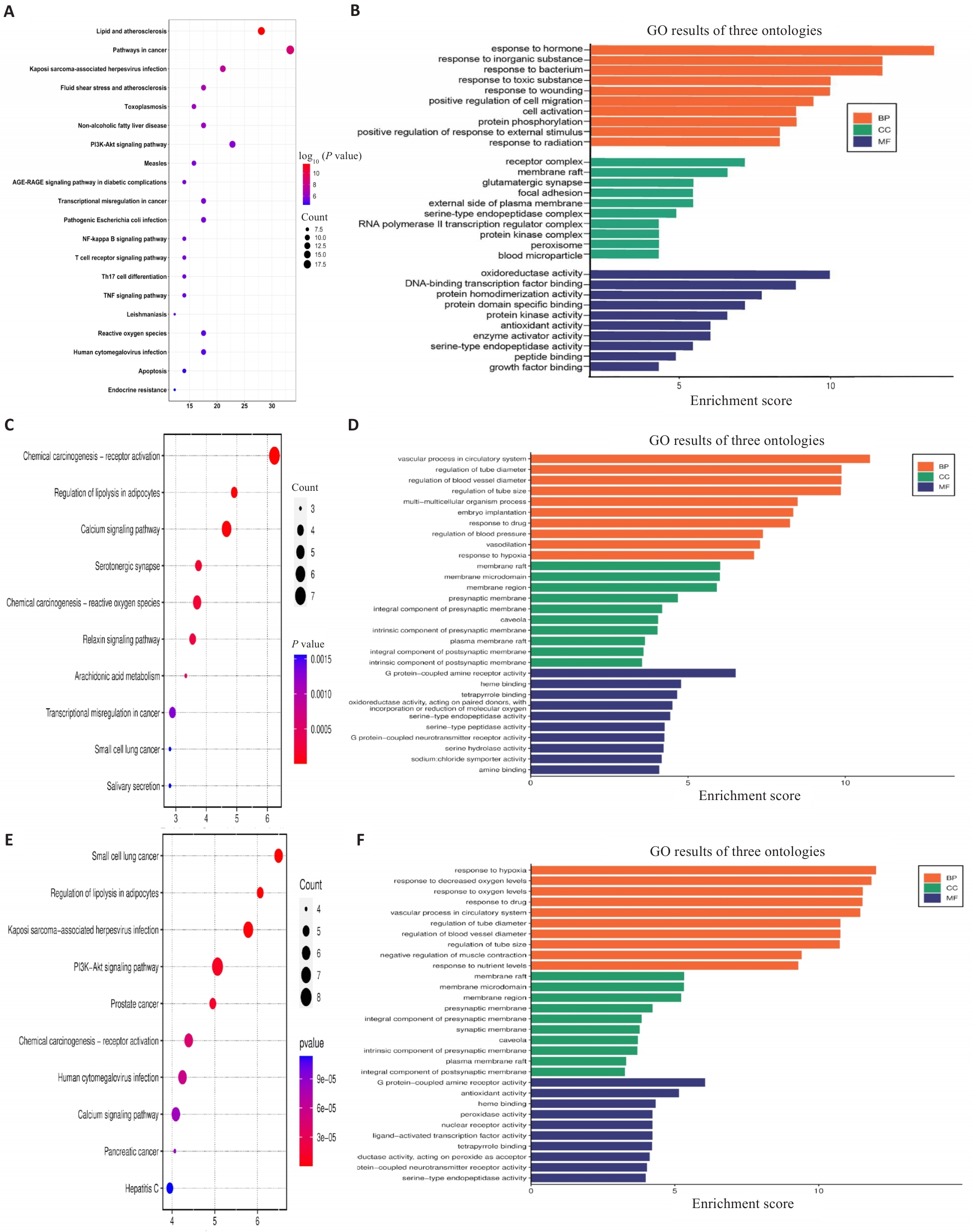
图3 治疗糖尿病肾病的潜在信号通路分析
Fig.3 Analysis of potential signaling pathways for treatment of DKD. A, C, E: Bubble plots of KEGG pathway enrichment analysis for shared genes between DKD and the herb pairs Astragalus-Salvia miltiorrhiza, Astragalus-Rehmannia, and Astragalus-Dioscorea, respectively. B, D, F: Gene Ontology (GO) analysis of shared genes between each herb pair and DKD, categorized into biological process (BP), molecular function (MF), and cellular component (CC).
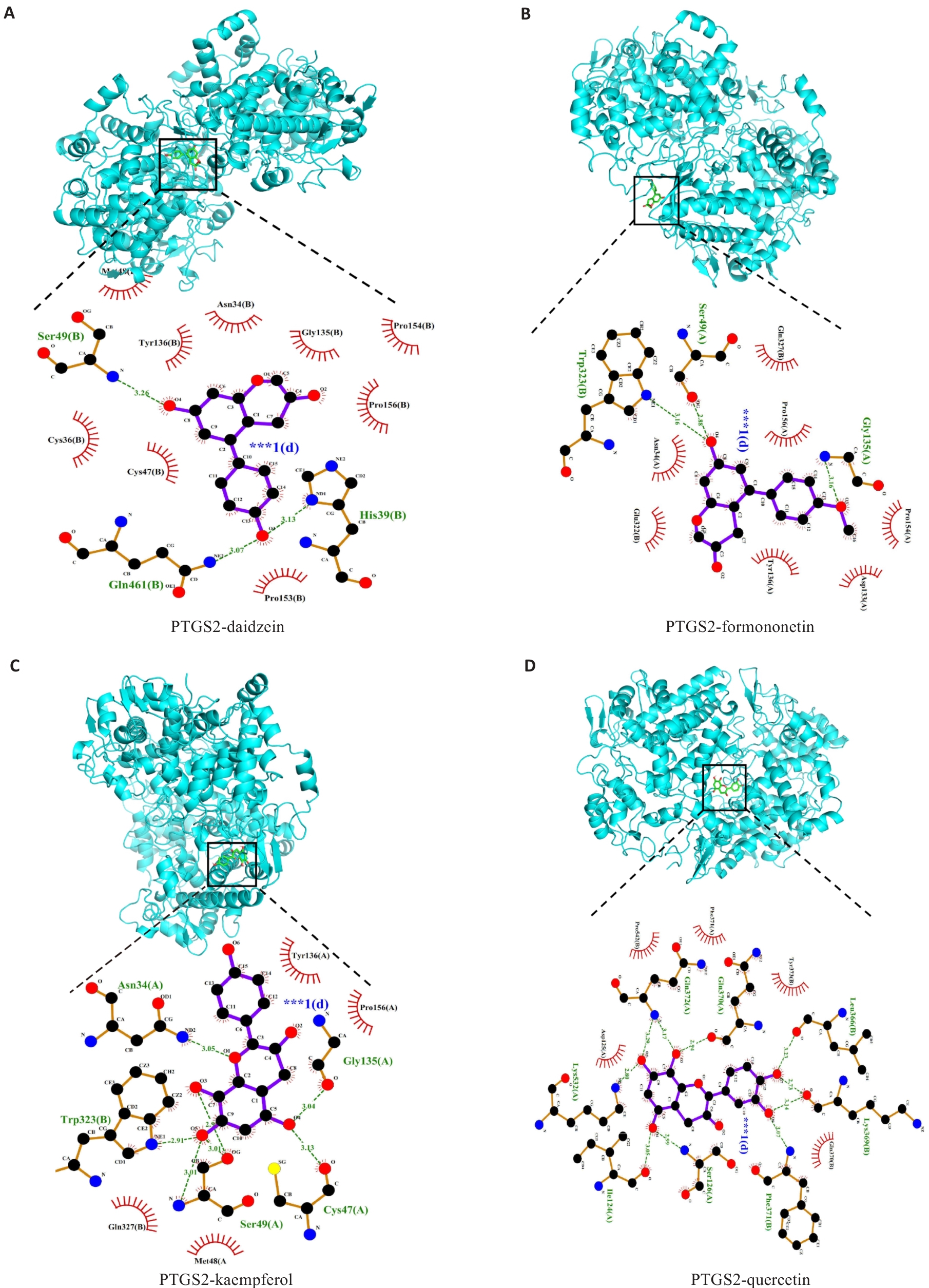
图4 有效成分与关键核心靶点PTGS2的分子对接
Fig.4 Molecular docking analysis of bioactive compounds with the key core target PTGS2. A: PTGS2 and Daidzein. B: PTGS2 and formononetin. C: PTGS2 and kaempferol. D: PTGS2 and quercetin.
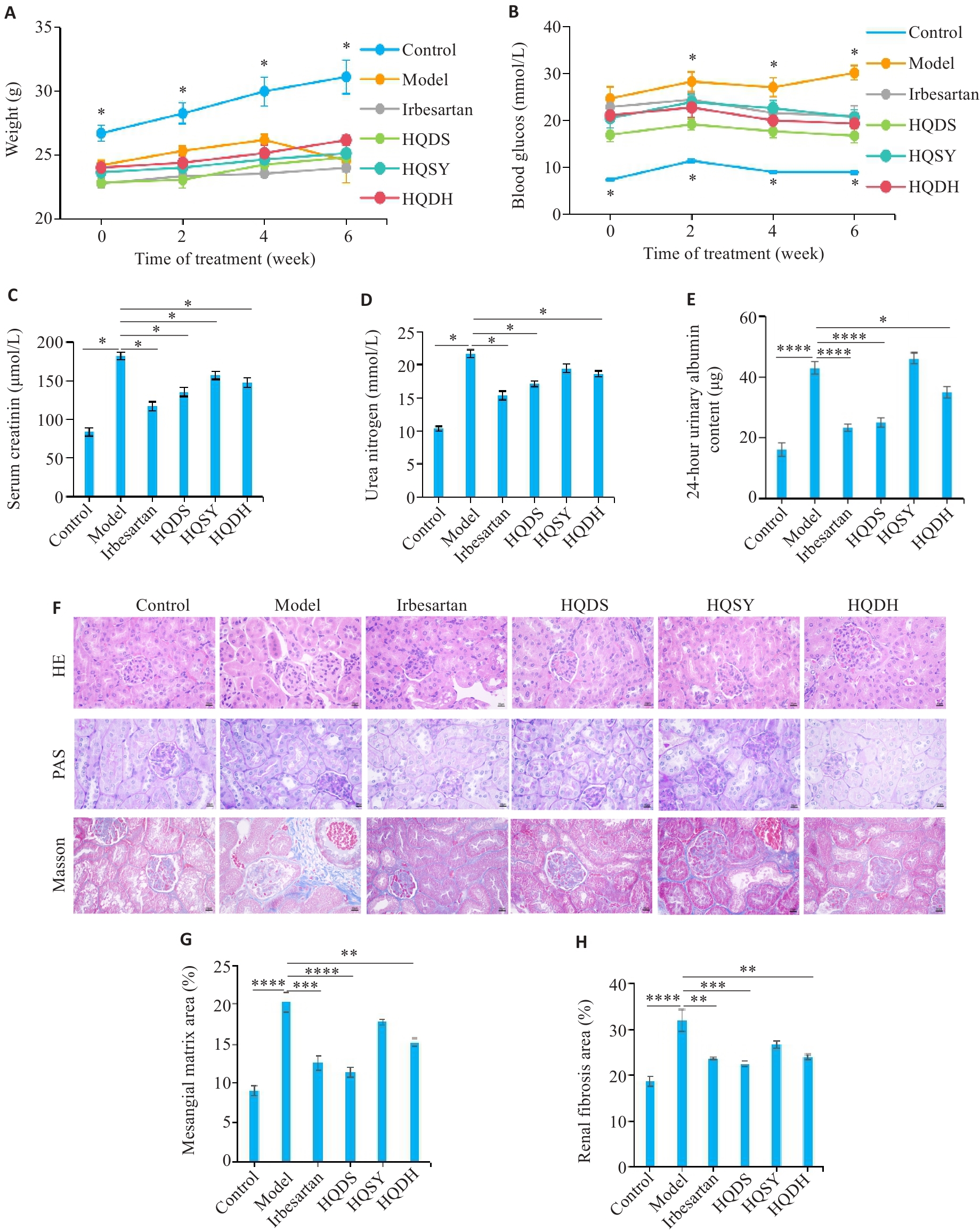
图5 不同黄芪药对对糖尿病肾病小鼠肾脏损伤的改善作用
Fig.5 Therapeutic effects of the Astragalus combinations on kidney damage in DKD mice.A: Body weight in Control group, Model group, Irbesartan group, Astragalus and Danshen group (HQDS), Astragalus and Dioscorea opposita group (HQSY), and Astragalus and Rehmannia glutinosa group (HQDH) (n=5). B: Fasting blood glucose levels in each group (n=5). C: Serum creatinine levels in each group (n=5). D: Urea nitrogen levels in each group (n=5). E: 24-hour urinary albumin content in each group (n=5). F: Results of HE, periodic acid-Schiff (PAS), and Masson staining of mouse kidney tissue (Original magnification: ×400). G: Quantifications of PAS trichrome staining (n=3). H: Quantitative analysis of Masson staining results (n=3). Data are presented as Mean±SE. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
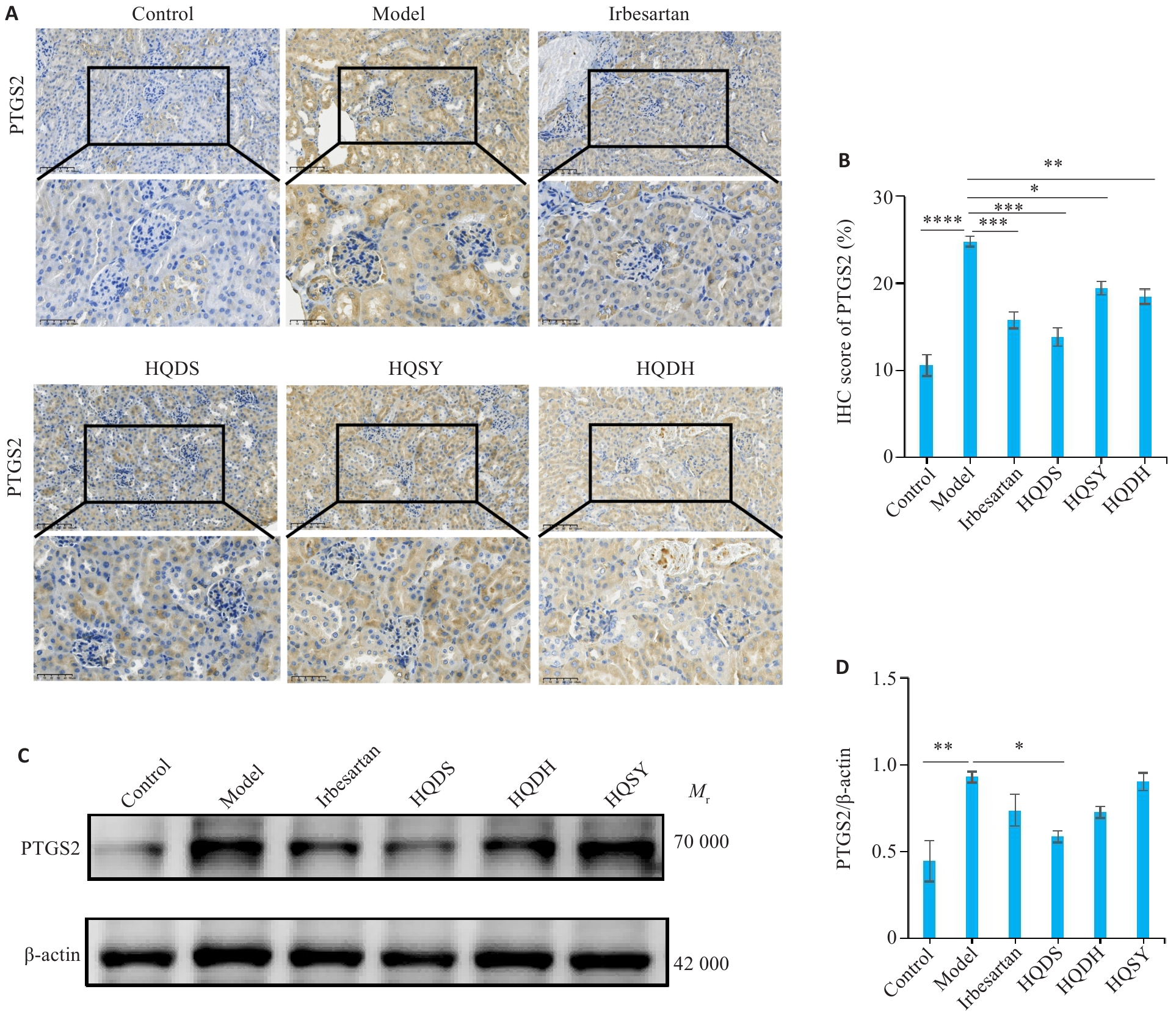
图6 不同黄芪药对调控DKD小鼠肾脏组织PTGS2的表达
Fig.6 Expression and localization of PTGS2 in renal tissues of DKD mice. A: Immunohistochemical staining showing localization and expression level of PTGS2 protein in the renal tissues of each group (×400). B: Quantitative analysis of PTGS2 immunohistochemical staining intensity. C: Western blotting of PTGS2. D: Quantitative analysis of PTGS2 protein expression levels. Data are presented as Mean±SE. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
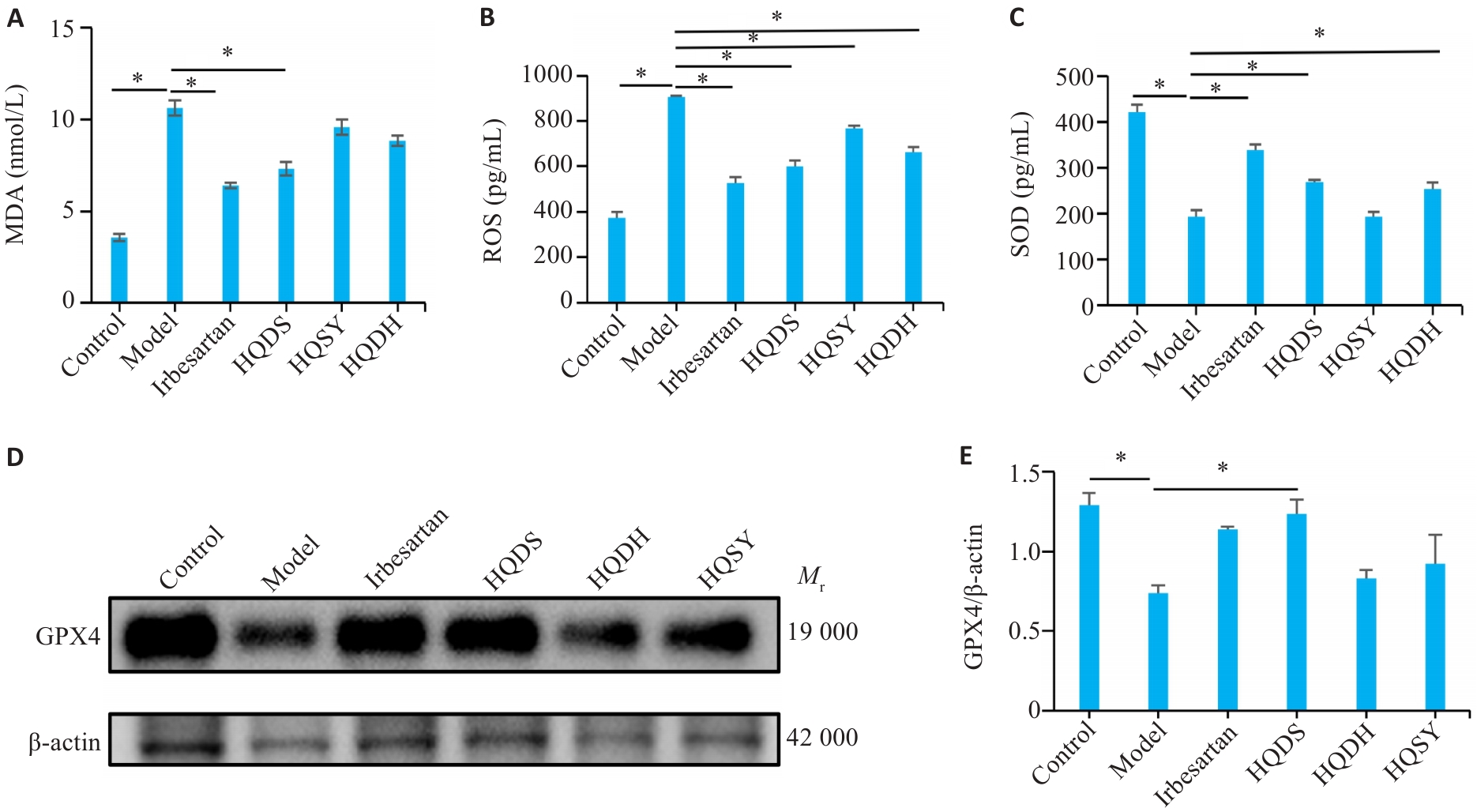
图7 不同黄芪药对调节DKD小鼠脂质过氧化水平
Fig.7 Differential regulatory effects of the 3 Astragali herb pairs on lipid peroxidation in DKD mice. A: Malondialdehyde (MDA) levels in each group. B: Reactive oxygen species (ROS) levels in each group. C: Superoxide dismutase (SOD) levels in each group. D: Western blotting results of GPX4. E: Quantitative analysis of GPX4 protein expression levels. Data are presented as Mean±SE. *P<0.05.
| [1] | Harreiter J, Roden M. Diabetes mellitus: definition, classification, diagnosis, screening and prevention (Update 2023)[J]. Wien Klin Wochenschr, 2023, 135(): 7-17. doi:10.1007/s00508-022-02122-y |
| [2] | 黄为钧, 赵进喜. 基于“新三消论”辨证论治糖尿病及其微血管并发症[J]. 中国中医基础医志, 2025, 31(2): 326-8. |
| [3] | Chaudhuri A, Ghanim H, Arora P. Improving the residual risk of renal and cardiovascular outcomes in diabetic kidney disease: a review of pathophysiology, mechanisms, and evidence from recent trials[J]. Diabetes Obes Metab, 2022, 24(3): 365-76. doi:10.1111/dom.14601 |
| [4] | Shafi T, Sozio SM, Plantinga LC, et al. Serum fructosamine and glycated albumin and risk of mortality and clinical outcomes in hemodialysis patients[J]. Diabetes Care, 2013, 36(6): 1522-33. doi:10.2337/dc12-1896 |
| [5] | Wang Y, He WC. Improving the dysregulation of FoxO1 activity is a potential therapy for alleviating diabetic kidney disease[J]. Front Pharmacol, 2021, 12: 630617. doi:10.3389/fphar.2021.630617 |
| [6] | 中华医学会肾脏病学分会专家组, 陈江华, 孙 林, 等. 糖尿病肾脏疾病临床诊疗中国指南[J]. 中华肾脏病杂志, 2021, 37(3): 255-304. doi:10.3760/cma.j.cn441217-20201125-00041 |
| [7] | Tamborlane WV, Laffel LM, Shehadeh N, et al. Efficacy and safety of dapagliflozin in children and young adults with type 2 diabetes: a prospective, multicentre, randomised, parallel group, phase 3 study[J]. Lancet Diabetes Endocrinol, 2022, 10(5): 341-50. doi:10.1016/s2213-8587(22)00052-3 |
| [8] | Fu ZM, Su XJ, Zhou Q, et al. Protective effects and possible mechanisms of catalpol against diabetic nephropathy in animal models: a systematic review and meta-analysis[J]. Front Pharmacol, 2023, 14: 1192694. doi:10.3389/fphar.2023.1192694 |
| [9] | Gupta S, Dominguez M, Golestaneh L. Diabetic kidney disease: an update[J]. Med Clin North Am, 2023, 107(4): 689-705. doi:10.1016/j.mcna.2023.03.004 |
| [10] | A/L B Vasanth Rao VR, Tan SH, Candasamy M, et al. Diabetic nephropathy: an update on pathogenesis and drug development[J]. Diabetes Metab Syndr, 2019, 13(1): 754-62. doi:10.1016/j.dsx.2018.11.054 |
| [11] | Shi CR, Kwong DL, Li X, et al. MAEL augments cancer stemness properties and resistance to sorafenib in hepatocellular carcinoma through the PTGS2/AKT/STAT3 axis[J]. Cancers (Basel), 2022, 14(12): 2880. doi:10.3390/cancers14122880 |
| [12] | Wang S, Qin S, Cai B, et al. Promising therapeutic mechanism for Chinese herbal medicine in ameliorating renal fibrosis in diabetic nephropathy[J]. Front Endocrinol (Lausanne), 2023, 14:932649. doi:10.3389/fendo.2023.932649 |
| [13] | 李雨婷. 糖尿病肾病的中医治疗[J]. 2025(2): 77-8. |
| [14] | 胡筱娟, 秦 艳, 刘恬园. 基于肾络瘀阻病机与糖尿病肾病相关性临床研究[J]. 陕西中医, 2015, 36(12): 1614-6. doi:10.3969/j.issn.1000-7369.2015.12.022 |
| [15] | 蔡 寸, 马红岩. 杨霓芝教授运用益气活血法治疗糖尿病肾病经验[J]. 河北中医, 2018, 40(6): 805-8. doi:10.3969/j.issn.1002-2619.2018.06.001 |
| [16] | 陈洪宇. 王永钧教授诊治2型糖尿病肾损害的临证经验[J]. 中国中西医结合肾病杂志, 2008, 9(9): 756-9. doi:10.3969/j.issn.1009-587X.2008.09.002 |
| [17] | 魏 敏, 赵晓山, 戴红芳, 等. 罗仁教授治疗糖尿病肾病的经验[C]//中华中医药学会第二十一届全国中医肾病学术会议论文汇编(上). 北京, 2008: 69-71. doi:ConferenceArticle/5aa07878c095d72220743076 |
| [18] | Liang QE, Bai ZY, Xie T, et al. Deciphering the pharmacological mechanisms of qidan Dihuang decoction in ameliorating renal fibrosis in diabetic nephropathy through experimental validation in vitro and in vivo[J]. Evid Based Complement Alternat Med, 2022, 2022: 4137578. doi:10.1155/2022/4137578 |
| [19] | 谢 伟, 沈小勇, 王 硕, 等. 知识史视野下黄芪性-效-用-忌的古今源流考证[J]. 中成药, 2024, 46(7): 2470-4. doi:10.3969/j.issn.1001-1528.2024.07.057 |
| [20] | 王小平, 王振国. 基于本草考证的丹参功效研究[J]. 中医药信息, 2023, 40(7): 58-61. doi:10.19656/j.cnki.1002-2406.20230709 |
| [21] | Xiang L, Jiang PP, Zhou L, et al. Additive effect of qidan Dihuang grain, a traditional Chinese medicine, and angiotensin receptor blockers on albuminuria levels in patients with diabetic nephropathy: a randomized, parallel-controlled trial[J]. Evid Based Complement Alternat Med, 2016, 2016: 1064924. doi:10.1155/2016/1064924 |
| [22] | Xiang L, Cai XS, Zhao XS, et al. Uncovering the mechanism of Qidan Dihuang Granule in the treatment of diabetic kidney disease combined network pharmacology, UHPLC-MS/MS with experimental validation[J]. Heliyon, 2023, 9(11): e21714. doi:10.1016/j.heliyon.2023.e21714 |
| [23] | 韩 明珠, 李晓华. 中医药治疗早期糖尿病肾病的研究进展[J].中国医药导报, 2018, 15(29): 42-5. |
| [24] | 吴 颖, 苗春平, 金海杰, 等. 张银霞运用中药治疗糖尿病经验探讨分析[J]. 中国保健营养, 2019, 29(29): 3-4. |
| [25] | Tian Y, Zhou CX, Yan Q, et al. Dapagliflozin improves diabetic kidney disease by inhibiting ferroptosis through β-hydroxybutyrate production[J]. Ren Fail, 2025, 47(1): 2438857. doi:10.1080/0886022x.2024.2438857 |
| [26] | Lin ZW, Xu YF, Guan LL, et al. Seven ferroptosis-specific expressed genes are considered as potential biomarkers for the diagnosis and treatment of cigarette smoke-induced chronic obstructive pulmonary disease[J]. Ann Transl Med, 2022, 10(6): 331. doi:10.21037/atm-22-1009 |
| [27] | Machado-Junior PAB, Lass A, de Bortolo J, et al. Sodium-glucose cotransporter-2 inhibitor improves renal injury by regulating the redox profile, inflammatory parameters, and pyroptosis in an experimental model of diabetic kidney disease[J]. ACS Pharmacol Transl Sci, 2025, 8(5): 1270-81. doi:10.1021/acsptsci.4c00552 |
| [28] | Kajal A, Singh R. Coriandrum sativum seeds extract mitigate progression of diabetic nephropathy in experimental rats via AGEs inhibition[J]. PLoS One, 2019, 14(3): e0213147. doi:10.1371/journal.pone.0213147 |
| [29] | Zou PJ, He QM, Xia HM, et al. Ferroptosis and its impact on common diseases[J]. PeerJ, 2024, 12: e18708. doi:10.7717/peerj.18708 |
| [30] | Geng JN, Yu XY, Liu CY, et al. Herba artemisiae capillaris extract prevents the development of streptozotocin-induced diabetic nephropathy of rat[J]. Evid Based Complement Alternat Med, 2018: 5180165. doi:10.1155/2018/5180165 |
| [31] | Hartal-Benishay LH, Tal S, Elkader AA, et al. Activity-dependent COX-2 proteolysis modulates aerobic respiration and proliferation in a prostaglandin-independent manner[J]. iScience, 2024, 27(12): 111403. doi:10.1016/j.isci.2024.111403 |
| [32] | Zou C, Xu FF, Shen JC, et al. Identification of a ferroptosis-related prognostic gene PTGS2 based on risk modeling and immune microenvironment of early-stage cervical cancer[J]. J Oncol, 2022: 3997562. doi:10.1155/2022/3997562 |
| [33] | Gao Q, Gao S, Li HY, et al. Multi-omics exploration of the role of PTGS2 as a hub gene in ferroptosis within the artery of takayasu arteritis[J]. J Inflamm Res, 2024, 17: 9135-46. doi:10.2147/jir.s478413 |
| [34] | Yang WS, SriRamaratnam R, Welsch ME, et al. Regulation of ferroptotic cancer cell death by GPX4[J]. Cell, 2014, 156(1/2): 317-31. doi:10.1016/j.cell.2013.12.010 |
| [35] | Xu JL, Weng CY, Zhang YQ, et al. GPX4 knockdown suppresses M2 macrophage polarization in gastric cancer by modulating kynurenine metabolism[J]. Theranostics, 2025, 15(12): 5826-45. doi:10.7150/thno.108817 |
| [36] | Sacco A, Battaglia AM, Botta C, et al. Iron metabolism in the tumor microenvironment-implications for anti-cancer immune response[J]. Cells, 2021, 10(2): 303. doi:10.3390/cells10020303 |
| [37] | Forcina GC, Dixon SJ. GPX4 at the crossroads of lipid homeostasis and ferroptosis[J]. Proteomics, 2019, 19(18): e1800311. doi:10.1002/pmic.201800311 |
| [38] | Xu JY, Pi JK, Zhang YJ, et al. Effects of ferroptosis on cardiovascular diseases[J]. Mediators Inflamm, 2023: 6653202. doi:10.1155/2023/6653202 |
| [1] | 董妍妍, 张可敬, 储俊, 储全根. 抵当汤含药血清通过PI3K/Akt/mTOR信号通路增强高糖诱导的大鼠肾小球内皮细胞自噬[J]. 南方医科大学学报, 2025, 45(3): 461-469. |
| [2] | 朱思佳, 马竞成, 郑玉娇, 吴传云, 赵建根, 李玲秀, 汪莉, 周雪梅. 丹蛭降糖胶囊通过下调Notch1/NICD/MAML1信号通路改善糖尿病肾病大鼠肾脏血管内皮功能[J]. 南方医科大学学报, 2025, 45(10): 2250-2257. |
| [3] | 郭克磊, 李颖利, 宣晨光, 侯紫君, 叶松山, 李林运, 陈丽平, 韩立, 卞华. 益气养阴化浊通络方通过调控miR-21a-5p/FoxO1/PINK1介导的线粒体自噬减轻糖尿病肾病小鼠的足细胞损伤[J]. 南方医科大学学报, 2025, 45(1): 27-34. |
| [4] | 姜一凡, 李小荣, 耿嘉逸, 陈永锋, 唐碧, 康品方. 槲皮素通过抑制HMGB1/RAGE/NF-κB信号通路减轻糖尿病引起的大鼠肾脏损伤[J]. 南方医科大学学报, 2024, 44(9): 1769-1775. |
| [5] | 王瑾瑾, 崔文飞, 窦雪伟, 尹冰磊, 牛钰琪, 牛羚, 闫国立. 鬼箭羽通过调节EGFR酪氨酸激酶抑制剂耐药信号通路延缓糖尿病肾病的进展[J]. 南方医科大学学报, 2024, 44(7): 1243-1255. |
| [6] | 李 莹, 王 倩, 陈小鸟, 席 悦, 杨 建, 刘晓敏, 王远大, 张 利, 蔡广研, 陈香美, 董哲毅. 基于糖尿病视网膜病变的诊断模型对糖尿病肾病有较好诊断效能[J]. 南方医科大学学报, 2023, 43(9): 1585-1590. |
| [7] | 任 毅, 卢金莹, 于 露, 李宗泽, 王 高, 杨 菁. 肌肽对糖尿病肾病大鼠肾组织AKT/mTOR通路及自噬的影响[J]. 南方医科大学学报, 2023, 43(11): 1965-1970. |
| [8] | 王 影, 周明俊, 朱倩文, 张 翠, 王 林, 李 曙, 胡泽波. HIF-1α活化介导的胆固醇稳态失调加速大鼠的糖尿病肾病进展[J]. 南方医科大学学报, 2023, 43(10): 1782-1788. |
| [9] | 王欢岚, 刘 红, 张燕敏, 陈伟栋. miR-34a通过靶向抑制Notch信号通路减轻糖尿病肾病小鼠的足细胞损伤[J]. 南方医科大学学报, 2022, 42(12): 1839-1845. |
| [10] | 沈炳香, 王法财, 周 艺, 李 婷, 何春远, 赵为陈. 人参皂苷Rh2调控盘状结构域受体1对糖尿病大鼠肾纤维化和细胞凋亡的影响[J]. 南方医科大学学报, 2021, 41(7): 1107-1113. |
| [11] | 李志阳, 梁 丹, 肖雅文, 代云莉, 艾福军, 丁 菁, 石明隽, 肖 瑛, 郭 兵. 氧化苦参碱通过抑制CHK1/2磷酸化改善糖尿病大鼠肾组织的纤维化和炎症[J]. 南方医科大学学报, 2021, 41(10): 1519-1526. |
| [12] | 赵为陈, 何春远, 王法财. 以caspase-1介导细胞焦亡筛选治疗糖尿病肾病潜在的中药及单体成分[J]. 南方医科大学学报, 2020, 40(09): 1280-1287. |
| [13] | 储全根,蔡正银,储 俊,轩 云,程 婕,王 盼. 痰瘀同治法对糖尿病大鼠肾脏TGF-β1/Smad3信号通路的影响[J]. 南方医科大学学报, 2020, 40(05): 708-712. |
| [14] | 周雪梅,徐从书,王凯,储全根,董昌武,吴传云,赵建根,李玲秀,汪莉. 益气养阴活血中药对糖尿病肾病大鼠Notch/Hes1 通路与血管内皮CD34、CD144 的影响[J]. 南方医科大学学报, 2019, 39(07): 855-. |
| [15] | 吴凡,陈韵颖,肖花,邹子良,宁靖,陈海珊,邹和群. 尿液细胞外囊泡中的podocalyxin在糖尿病肾病诊断中的价值[J]. 南方医科大学学报, 2018, 38(09): 1126-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||