南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 278-285.doi: 10.12122/j.issn.1673-4254.2026.02.05
• • 上一篇
收稿日期:2025-07-09
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
杨朝阳
E-mail:chenmeimei1984@163.com;yzy813@126.com
作者简介:陈梅妹,博士,副研究员,硕士生导师,E-mail: chenmeimei1984@163.com
基金资助:
Meimei CHEN1,2( ), Ruina HUANG1,2, Zhaoyang YANG1,2(
), Ruina HUANG1,2, Zhaoyang YANG1,2( )
)
Received:2025-07-09
Online:2026-02-20
Published:2026-03-10
Contact:
Zhaoyang YANG
E-mail:chenmeimei1984@163.com;yzy813@126.com
摘要:
目的 探讨炎症蛋白与阿尔茨海默病(AD)之间的因果关系,并评估血浆代谢物在该关系中的中介作用。 方法 采用孟德尔随机化(MR)方法进行分析。研究利用公开可获得的全基因组关联研究(GWAS)数据,选取91种炎症蛋白强相关且无反向因果关系的单核苷酸多态性(snp)作为暴露,以AD为结局,进行双向双样本MR分析。通过逆方差加权法(IVW)筛选出与AD存在因果关系的炎症蛋白,进一步基于中介MR分析,以1400种血浆代谢物作为中介变量,评估代谢物在炎症蛋白对AD因果效应中的中介作用。 结果 初步双向MR分析识别出3种与AD存在潜在正向因果关联且无反向因果关联的炎症蛋白,分别是Axin-1、C-X-C基序趋化因子11(CXCL11)及白细胞介素-12β(IL-12β)。其中,Axin-1水平升高与AD风险呈潜在正向因果效应(OR=1.082,95% CI:1.009~1.159,P=0.026),而CXCL11(OR=0.951,95% CI:0.914~0.990,P=0.026)和IL-12β(OR=0.959,95% CI:0.926~0.994,P=0.026)与AD风险则呈潜在负向因果效应。敏感性分析表明,这些因果关联均无异质性和多效性。在中介分析中,筛选出18种与AD具有潜在因果关联的血浆代谢物,其中3种血浆代谢物在炎症蛋白-AD因果中具有中介效应。甲基-4-羟基苯甲酸硫酸盐在Axin-1与AD之间起到部分中介作用(占比为20.10%)。孕烯三醇硫酸盐在CXCL11与AD之间起到部分中介作用(占比为18.20%)。亚精胺/鸟氨酸比值在IL-12β与AD之间起到重要中介作用,中介效应占比为43.40%。 结论 本研究首次揭示了特定炎症蛋白通过血浆代谢物影响AD风险的潜在机制,为AD的炎症-代谢交互机制提供了遗传学证据,为AD的早期检测和干预提供了潜在的生物标志物和靶点。
陈梅妹, 黄睿娜, 杨朝阳. 血浆代谢物介导炎症蛋白对阿尔茨海默病的因果效应:一项孟德尔随机化分析研究[J]. 南方医科大学学报, 2026, 46(2): 278-285.
Meimei CHEN, Ruina HUANG, Zhaoyang YANG. Plasma metabolites mediates the causal effect of inflammatory proteins on Alzheimer's disease: a Mendelian randomization study[J]. Journal of Southern Medical University, 2026, 46(2): 278-285.
| Type | Exposure | Outcome | Nsnp | Beta | SE | P | OR (95% CI) |
|---|---|---|---|---|---|---|---|
| Inflammation proteins | Axin-1 | AD | 13 | 0.079 | 0.035 | 0.026 | 1.082 (1.009-1.159) |
| C-X-C motif chemokine 11 | AD | 39 | -0.050 | 0.020 | 0.026 | 0.951 (0.914-0.990) | |
| Interleukin-12 subunit beta | AD | 31 | -0.041 | 0.018 | 0.026 | 0.959 (0.926-0.994) | |
| Blood metabolites | Methyl-4-hydroxybenzoate sulfate | AD | 21 | 0.083 | 0.025 | 0.003 | 1.086 (1.034-1.141) |
| Epiandrosterone sulfate | AD | 24 | -0.038 | 0.014 | 0.006 | 0.963 (0.938-0.989) | |
| 11beta-hydroxyandrosterone glucuronide | AD | 29 | -0.062 | 0.019 | 0.003 | 0.940 (0.906-0.976) | |
| Pregnenetriol sulfate | AD | 8 | 0.092 | 0.033 | 0.006 | 1.096 (1.027-1.169) | |
| Palmitoleoylcarnitine (C16:1) | AD | 19 | 0.083 | 0.026 | 0.003 | 1.087 (1.034-1.143) | |
| Spermidine to ornithine ratio | AD | 4 | -0.168 | 0.057 | 0.006 | 0.845 (0.756-0.945) | |
| Phosphate to threonine ratio | AD | 7 | 0.141 | 0.050 | 0.006 | 1.151 (1.043-1.271) | |
| Citrulline to phosphate ratio | AD | 33 | -0.053 | 0.021 | 0.010 | 0.948 (0.911-0.987) |
表1 炎症蛋白、血浆代谢物对AD影响的MR分析结果
Tab.1 MR analysis results of the effects of inflammatory proteins and plasma metabolites on Alzheimer's disease (AD)
| Type | Exposure | Outcome | Nsnp | Beta | SE | P | OR (95% CI) |
|---|---|---|---|---|---|---|---|
| Inflammation proteins | Axin-1 | AD | 13 | 0.079 | 0.035 | 0.026 | 1.082 (1.009-1.159) |
| C-X-C motif chemokine 11 | AD | 39 | -0.050 | 0.020 | 0.026 | 0.951 (0.914-0.990) | |
| Interleukin-12 subunit beta | AD | 31 | -0.041 | 0.018 | 0.026 | 0.959 (0.926-0.994) | |
| Blood metabolites | Methyl-4-hydroxybenzoate sulfate | AD | 21 | 0.083 | 0.025 | 0.003 | 1.086 (1.034-1.141) |
| Epiandrosterone sulfate | AD | 24 | -0.038 | 0.014 | 0.006 | 0.963 (0.938-0.989) | |
| 11beta-hydroxyandrosterone glucuronide | AD | 29 | -0.062 | 0.019 | 0.003 | 0.940 (0.906-0.976) | |
| Pregnenetriol sulfate | AD | 8 | 0.092 | 0.033 | 0.006 | 1.096 (1.027-1.169) | |
| Palmitoleoylcarnitine (C16:1) | AD | 19 | 0.083 | 0.026 | 0.003 | 1.087 (1.034-1.143) | |
| Spermidine to ornithine ratio | AD | 4 | -0.168 | 0.057 | 0.006 | 0.845 (0.756-0.945) | |
| Phosphate to threonine ratio | AD | 7 | 0.141 | 0.050 | 0.006 | 1.151 (1.043-1.271) | |
| Citrulline to phosphate ratio | AD | 33 | -0.053 | 0.021 | 0.010 | 0.948 (0.911-0.987) |
| Outcome | Exposure | MR Egger | Inverse variance weighted | ||
|---|---|---|---|---|---|
| Q | Q_P | Q | Q_P | ||
| AD | Axin-1 | 11.388 | 0.411 | 11.440 | 0.492 |
| AD | C-X-C motif chemokine 11 | 40.637 | 0.313 | 41.066 | 0.338 |
| AD | Interleukin-12 subunit beta | 30.999 | 0.365 | 31.852 | 0.374 |
表2 炎症蛋白与AD因果关系的异质性分析结果
Tab.2 Analysis of heterogeneity of causal relationship between inflammatory proteins and AD
| Outcome | Exposure | MR Egger | Inverse variance weighted | ||
|---|---|---|---|---|---|
| Q | Q_P | Q | Q_P | ||
| AD | Axin-1 | 11.388 | 0.411 | 11.440 | 0.492 |
| AD | C-X-C motif chemokine 11 | 40.637 | 0.313 | 41.066 | 0.338 |
| AD | Interleukin-12 subunit beta | 30.999 | 0.365 | 31.852 | 0.374 |
| Outcome | Exposure | MR-Egger intercept | SE | P |
|---|---|---|---|---|
| AD | Axin-1 | -0.002 | 0.009 | 0.826 |
| AD | C-X-C motif chemokine 11 | -0.004 | 0.006 | 0.536 |
| AD | Interleukin-12 subunit beta | -0.004 | 0.004 | 0.379 |
表3 炎症蛋白与AD因果关系的多效性检验结果
Tab.3 Results of multivariate analysis of the causality between inflammatory proteins and AD
| Outcome | Exposure | MR-Egger intercept | SE | P |
|---|---|---|---|---|
| AD | Axin-1 | -0.002 | 0.009 | 0.826 |
| AD | C-X-C motif chemokine 11 | -0.004 | 0.006 | 0.536 |
| AD | Interleukin-12 subunit beta | -0.004 | 0.004 | 0.379 |
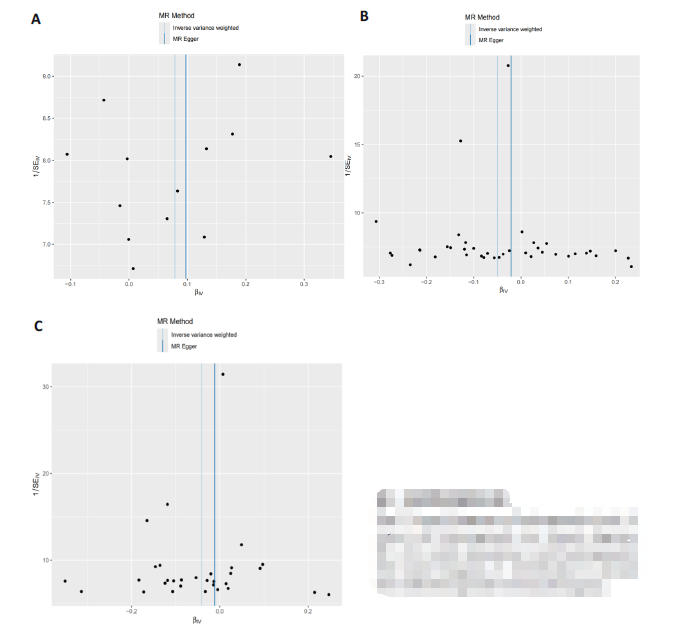
图3 异质性检验漏斗图
Fig.3 Funnel diagrams of heterogeneity tests. A:Axin-1-AD heterogeneity test funnel diagram;B: C-X-C motif chemokine 11-AD heterogeneitytest funnel diagram; C: Interleukin-12 subunitbeta-AD heterogeneity test funnel diagram.
| Outcome | Exposure | Nsnp | Beta | SE | P | OR (95% CI) |
|---|---|---|---|---|---|---|
| Axin-1 | AD | 59 | -0.043 | 0.022 | 0.053 | 0.957 (0.916-1.001 ) |
| C-X-C motif chemokine 11 | AD | 59 | 0.010 | 0.020 | 0.634 | 1.010 (0.970-1.050) |
| Interleukin-12 subunit beta | AD | 58 | 0.012 | 0.023 | 0.614 | 1.012 (0.967-1.058 ) |
表4 炎症蛋白与AD反向MR分析结果
Tab.4 Results of reverse MR analysis of inflammatory proteins and AD
| Outcome | Exposure | Nsnp | Beta | SE | P | OR (95% CI) |
|---|---|---|---|---|---|---|
| Axin-1 | AD | 59 | -0.043 | 0.022 | 0.053 | 0.957 (0.916-1.001 ) |
| C-X-C motif chemokine 11 | AD | 59 | 0.010 | 0.020 | 0.634 | 1.010 (0.970-1.050) |
| Interleukin-12 subunit beta | AD | 58 | 0.012 | 0.023 | 0.614 | 1.012 (0.967-1.058 ) |
| Exposure | Metabolite | Outcome | Beta-all | Beta1 | Beta2 | Mediated effect | Mediated proportion | P |
|---|---|---|---|---|---|---|---|---|
| Axin-1 | Methyl-4-hydroxybenzoate sulfate | AD | 0.079 | 0.191 | 0.083 | 0.016 | 20.10% | 0.001 |
C-X-C motif chemokine 11 | Epiandrosterone sulfate | AD | -0.050 | -0.094 | -0.038 | 0.004 | NA | NA |
| 11beta-hydroxyandrosterone glucuronide | AD | -0.050 | -0.078 | -0.062 | 0.005 | NA | NA | |
| Pregnenetriol sulfate | AD | -0.050 | -0.099 | 0.092 | -0.009 | 18.20% | 0.038 | |
| Interleukin-12 subunit beta | Palmitoleoylcarnitine (C16:1) | AD | -0.041 | 0.075 | 0.083 | 0.006 | NA | NA |
| Spermidine to ornithine ratio | AD | -0.041 | 0.107 | -0.168 | -0.018 | 43.40% | 0.0004 | |
| Phosphate to threonine ratio | AD | -0.041 | 0.083 | 0.141 | 0.012 | NA | NA | |
| Citrulline to phosphate ratio | AD | -0.041 | -0.083 | -0.053 | 0.004 | NA | NA |
表5 两步孟德尔随机化分析炎症蛋白、血浆代谢物和AD之间的因果关系
Tab.5 Two-step MR analysis of the causality between inflammatory proteins, plasma metabolites and AD
| Exposure | Metabolite | Outcome | Beta-all | Beta1 | Beta2 | Mediated effect | Mediated proportion | P |
|---|---|---|---|---|---|---|---|---|
| Axin-1 | Methyl-4-hydroxybenzoate sulfate | AD | 0.079 | 0.191 | 0.083 | 0.016 | 20.10% | 0.001 |
C-X-C motif chemokine 11 | Epiandrosterone sulfate | AD | -0.050 | -0.094 | -0.038 | 0.004 | NA | NA |
| 11beta-hydroxyandrosterone glucuronide | AD | -0.050 | -0.078 | -0.062 | 0.005 | NA | NA | |
| Pregnenetriol sulfate | AD | -0.050 | -0.099 | 0.092 | -0.009 | 18.20% | 0.038 | |
| Interleukin-12 subunit beta | Palmitoleoylcarnitine (C16:1) | AD | -0.041 | 0.075 | 0.083 | 0.006 | NA | NA |
| Spermidine to ornithine ratio | AD | -0.041 | 0.107 | -0.168 | -0.018 | 43.40% | 0.0004 | |
| Phosphate to threonine ratio | AD | -0.041 | 0.083 | 0.141 | 0.012 | NA | NA | |
| Citrulline to phosphate ratio | AD | -0.041 | -0.083 | -0.053 | 0.004 | NA | NA |
| [1] | Scheltens P, De Strooper B, Kivipelto M, et al. Alzheimer’s disease[J]. Lancet, 2021, 397(10284): 1577-90. doi:10.1016/s0140-6736(20)32205-4 |
| [2] | GBD 2019 Dementia Forecasting Collaborators. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019[J]. Lancet Public Health, 2022, 7(2): e105-25. |
| [3] | Leng FD, Edison P. Neuroinflammation and microglial activation in Alzheimer disease: where do we go from here[J]? Nat Rev Neurol, 2021, 17(3): 157-72. doi:10.1038/s41582-020-00435-y |
| [4] | Arnold SE, Arvanitakis Z, Macauley-Rambach SL, et al. Brain insulin resistance in type 2 diabetes and Alzheimer disease: concepts and conundrums[J]. Nat Rev Neurol, 2018, 14(3): 168-81. doi:10.1038/nrneurol.2017.185 |
| [5] | Brandl S, Reindl M. Blood-brain barrier breakdown in neuroinflammation: current in vitro models[J]. Int J Mol Sci, 2023, 24(16): 12699. doi:10.3390/ijms241612699 |
| [6] | Ponce-Lopez T. Peripheral inflammation and insulin resistance: their impact on blood-brain barrier integrity and Glia activation in Alzheimer's disease[J]. Int J Mol Sci, 2025, 26(9): 4209. doi:10.3390/ijms26094209 |
| [7] | Amelimojarad M, Amelimojarad M, Cui XN. The emerging role of brain neuroinflammatory responses in Alzheimer's disease[J]. Front Aging Neurosci, 2024, 16: 1391517. doi:10.3389/fnagi.2024.1391517 |
| [8] | Hegazy SH, Thomassen JQ, Rasmussen IJ, et al. C-reactive protein levels and risk of dementia-Observational and genetic studies of 111, 242 individuals from the general population[J]. Alzheimers Dement, 2022, 18(11): 2262-71. doi:10.1002/alz.12568 |
| [9] | Shao YP, Ouyang Y, Li TB, et al. Alteration of metabolic profile and potential biomarkers in the plasma of Alzheimer's disease[J]. Aging Dis, 2020, 11(6): 1459-70. doi:10.14336/ad.2020.0217 |
| [10] | Borkowski K, Seyfried NT, Arnold M, et al. Integration of plasma and CSF metabolomics with CSF proteomic reveals novel associations between lipid mediators and central nervous system vascular and energy metabolism[J]. Sci Rep, 2023, 13(1): 13752. doi:10.1038/s41598-023-39737-8 |
| [11] | Shen M, Zhang LL, Chen C, et al. Investigating the causal relationship between immune cell and Alzheimer's disease: a mendelian randomization analysis[J]. BMC Neurol, 2024, 24(1): 98. doi:10.1186/s12883-024-03599-y |
| [12] | Li YC, Yao YL, Wu Y. Causal relationships between plasma proteins and Alzheimer's disease using bidirectional Mendelian rando-mization[J]. J Alzheimers Dis, 2025, 106(2): 765-73. doi:10.1177/13872877251345151 |
| [13] | Zhao JH, Stacey D, Eriksson N, et al. Genetics of circulating inflammatory proteins identifies drivers of immune-mediated disease risk and therapeutic targets[J]. Nat Immunol, 2023, 24(9): 1540-51. doi:10.1038/s41590-023-01588-w |
| [14] | Chen YH, Lu TY, Pettersson-Kymmer U, et al. Genomic atlas of the plasma metabolome prioritizes metabolites implicated in human diseases[J]. Nat Genet, 2023, 55(1): 44-53. doi:10.1038/s41588-022-01270-1 |
| [15] | Papadimitriou N, Dimou N, Tsilidis KK, et al. Physical activity and risks of breast and colorectal cancer: a Mendelian randomisation analysis[J]. Nat Commun, 2020, 11(1): 597. |
| [16] | 李文婕, 洪耀南, 黄 蕊, 等. 自身免疫性疾病是再生障碍性贫血的危险因素: 一项孟德尔随机化分析[J]. 南方医科大学学报, 2025, 45(4): 871-9. |
| [17] | Carter AR, Sanderson E, Hammerton G, et al. Mendelian randomisation for mediation analysis: current methods and challenges for implementation[J]. Eur J Epidemiol, 2021, 36(5): 465-78. doi:10.1007/s10654-021-00757-1 |
| [18] | Qiu L, Sun YX, Ning HM, et al. The scaffold protein AXIN1 gene ontology, signal network, and physiological function[J]. Cell Commun Signal, 2024, 22(1): 77. doi:10.1186/s12964-024-01482-4 |
| [19] | Tapia-Rojas C, Inestrosa NC. Wnt signaling loss accelerates the appearance of neuropathological hallmarks of Alzheimer’s disease in J20-APP transgenic and wild-type mice[J]. J Neurochem, 2018, 144(4): 443-65. doi:10.1111/jnc.14278 |
| [20] | Tapia-Rojas C, Burgos PV, Inestrosa NC. Inhibition of Wnt signaling induces amyloidogenic processing of amyloid precursor protein and the production and aggregation of Amyloid-β (Aβ)42 peptides[J]. J Neurochem, 2016, 139(6): 1175-91. doi:10.1111/jnc.13873 |
| [21] | Koper OM, Kamińska J, Sawicki K, et al. CXCL9, CXCL10, CXCL11, and their receptor (CXCR3) in neuroinflammation and neurodegeneration[J]. Adv Clin Exp Med, 2018, 27(6): 849-56. doi:10.17219/acem/68846 |
| [22] | Wojcieszak J, Kuczyńska K, Zawilska JB. Role of chemokines in the development and progression of Alzheimer's disease[J]. J Mol Neurosci, 2022, 72(9): 1929-51. doi:10.1007/s12031-022-02047-1 |
| [23] | Wang XM, Zhang YY, Wang S, et al. The role of CXCR3 and its ligands in cancer[J]. Front Oncol, 2022, 12: 1022688. doi:10.3389/fonc.2022.1022688 |
| [24] | Moore TC, Hasenkrug KJ. B-cell control of regulatory T cells in friend virus infection[J]. J Mol Biol, 2021, 433(1): 166583. doi:10.1016/j.jmb.2020.06.022 |
| [25] | Ciccocioppo F, Lanuti P, Pierdomenico L, et al. The characterization of regulatory T-cell profiles in Alzheimer's disease and multiple sclerosis[J]. Sci Rep, 2019, 9: 8788. doi:10.1038/s41598-019-45433-3 |
| [26] | Machhi J, Yeapuri P, Lu YM, et al. CD4+ effector T cells accelerate Alzheimer's disease in mice[J]. J Neuroinflammation, 2021, 18(1): 272. doi:10.1186/s12974-021-02308-7 |
| [27] | Rentzos M, Paraskevas GP, Kapaki E, et al. Interleukin-12 is reduced in cerebrospinal fluid of patients with Alzheimer's disease and frontotemporal dementia[J]. J Neurol Sci, 2006, 249(2): 110-4. doi:10.1016/j.jns.2006.05.063 |
| [28] | Yang HS, Zhang C, Carlyle BC, et al. Plasma IL-12/IFN‑γ axis predicts cognitive trajectories in cognitively unimpaired older adults[J]. Alzheimers Dement, 2022, 18(4): 645-53. doi:10.1002/alz.12399 |
| [29] | Zhu XC, Tan L, Jiang T, et al. Association of IL-12A and IL-12B polymorphisms with Alzheimer's disease susceptibility in a Han Chinese population[J]. J Neuroimmunol, 2014, 274(1/2): 180-4. doi:10.1016/j.jneuroim.2014.06.026 |
| [30] | Breitkreuz-Korff O, Tscheik C, Del Vecchio G, et al. M01 as a novel drug enhancer for specifically targeting the blood-brain barrier[J]. J Control Release, 2021, 338: 137-48. doi:10.1016/j.jconrel.2021.08.014 |
| [31] | Wei DF, Xu M, Cui WT, et al. Research progress on the role of Claudin family proteins in mediating blood-brain barrier selective permeability in tumor metastasis[J]. Am J Transl Res, 2025, 17(4): 2411-21. doi:10.62347/gggx3909 |
| [32] | Vallée M, Mayo W, Le Moal M. Role of pregnenolone, dehydroepiandrosterone and their sulfate esters on learning and memory in cognitive aging[J]. Brain Res Brain Res Rev, 2001, 37(1/2/3): 301-12. doi:10.1016/s0165-0173(01)00135-7 |
| [33] | 王 丹, 鲁映青, 于 榕. 神经甾体的合成和代谢及其对神经系统作用[J]. 中国临床药理学与治疗学, 2004, 9(7): 725-9. |
| [34] | Ghosh I, Sankhe R, Mudgal J, et al. Spermidine, an autophagy inducer, as a therapeutic strategy in neurological disorders[J]. Neuropeptides, 2020, 83: 102083. doi:10.1016/j.npep.2020.102083 |
| [35] | Ju YH, Bhalla M, Hyeon SJ, et al. Astrocytic urea cycle detoxifies Aβ-derived ammonia while impairing memory in Alzheimer's disease[J]. Cell Metab, 2022, 34(8): 1104-20.e8. doi:10.1016/j.cmet.2022.05.011 |
| [36] | Mein H, Jing Y, Ahmad F, et al. Altered brain arginine metabolism and polyamine system in a P301S tauopathy mouse model: a time-course study[J]. Int J Mol Sci, 2022, 23(11): 6039. doi:10.3390/ijms23116039 |
| [1] | 梁笑笑, 栾慧林, 马鹏珍, 邓文珠, 金佳欣, 全晨阳, 陈婷婷, 杨友财, 谢颖桢. 基于生命八要素的心血管健康评分与缺血性脑卒中预后相关性及临床检验指标的中介效应:一项多中心前瞻性队列研究[J]. 南方医科大学学报, 2026, 46(2): 239-246. |
| [2] | 冉念东, 刘杰, 徐剑, 张永萍, 郭江涛. 黑骨藤正丁醇萃取成分治疗大鼠阿尔茨海默病的药效学及作用机制[J]. 南方医科大学学报, 2025, 45(4): 785-798. |
| [3] | 李文婕, 洪耀南, 黄蕊, 李煜宸, 张莹, 张蕴, 吴迪炯. 自身免疫性疾病是再生障碍性贫血的危险因素:一项孟德尔随机化分析[J]. 南方医科大学学报, 2025, 45(4): 871-879. |
| [4] | 高俊杰, 叶开, 吴竞. 槲皮素通过调控TP53基因抑制肾透明细胞癌的增殖和迁移[J]. 南方医科大学学报, 2025, 45(2): 313-321. |
| [5] | 喻珍妮, 高竟哲, 孙惠, 冯芹, 那效旗, 张宁, 沈昆双, 王媛媛, 王喜军. 肠道菌群、T细胞在结直肠癌发病中的因果关联:孟德尔随机化分析[J]. 南方医科大学学报, 2025, 45(12): 2756-2766. |
| [6] | 李孝媛, 张逸悦, 顾雨铖, 陈霓红, 钱鑫宇, 张朋俊, 郝佳欣, 王峰. Tau蛋白沉积与脑代谢物的关联性:N-乙酰天门冬氨酸与肌酸作为进展期阿尔茨海默病的潜在生物标志物[J]. 南方医科大学学报, 2025, 45(11): 2350-2357. |
| [7] | 潘兴旭, 张秉祺, 张智华, 曹秋实. 戈登杆菌属丰度降低与肾结石风险增加相关:一项孟德尔随机化分析与动物实验研究[J]. 南方医科大学学报, 2025, 45(11): 2405-2415. |
| [8] | 马会华, 闫奎坡, 刘刚, 徐亚洲, 张磊, 李一卓. 1990~2021年心房颤动/扑动流行病学及其危险因素分析:基于2021年中国全球疾病负担研究与孟德尔随机化研究的系统分析[J]. 南方医科大学学报, 2025, 45(10): 2182-2190. |
| [9] | 姚辰, 李文佳, 庞瑞明, 周继红. 臀肌腱炎、原发性髋关节病可能导致髂胫束综合征—一项孟德尔随机化研究[J]. 南方医科大学学报, 2024, 44(9): 1821-1830. |
| [10] | 钟帷韬, 李伟松, 李泽霖, 王强, 张旺明. 睡眠性状与特发性正常压力脑积水的因果关联:一项两样本双向孟德尔随机化研究[J]. 南方医科大学学报, 2024, 44(8): 1612-1619. |
| [11] | 刘佳进, 缪长宏, 徐健康, 余伟杰, 陈继鑫, 唐好知, 刘爱峰. 肠道菌群与色素沉着绒毛结节性滑膜炎之间的因果关系:基于孟德尔随机化分析[J]. 南方医科大学学报, 2024, 44(7): 1397-1406. |
| [12] | 李和平, 李高桦, 张学华, 王亚楠. 直肠癌炎症蛋白因子的遗传驱动:孟德尔随机化方法在临床预后研究中的应用[J]. 南方医科大学学报, 2024, 44(7): 1361-1370. |
| [13] | 王妍, 阮毓卿, 崔璨, 王秀. 交泰丸通过激活PI3K/AKT信号通路改善阿尔茨海默病模型小鼠大脑的葡萄糖代谢[J]. 南方医科大学学报, 2024, 44(5): 894-903. |
| [14] | 占雯婕, 赵 玲. 1型糖尿病可能增加甲状腺毒症的风险:基于两样本孟德尔随机化方法[J]. 南方医科大学学报, 2023, 43(8): 1396-1401. |
| [15] | 苏 超, 田彧潇, 张 清, 万田豪, 夏 迪. 肌肉质量增加可能导致椎间盘退变:一项两样本孟德尔随机化研究[J]. 南方医科大学学报, 2023, 43(12): 2029-2034. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||