南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (6): 1131-1142.doi: 10.12122/j.issn.1673-4254.2025.06.03
夏冰1( ), 彭进1,2, 丁九阳1, 王杰1, 唐国伟3, 刘国杰4, 王沄4, 万昌武1(
), 彭进1,2, 丁九阳1, 王杰1, 唐国伟3, 刘国杰4, 王沄4, 万昌武1( ), 乐翠云1(
), 乐翠云1( )
)
收稿日期:2025-01-05
出版日期:2025-06-20
发布日期:2025-06-27
通讯作者:
万昌武,乐翠云
E-mail:372732293@qq.com;wcw005@sina.com;675493755@qq.com
作者简介:夏 冰,主任法医师,E-mail: 372732293@qq.com
基金资助:
Bing XIA1( ), Jin PENG1,2, Jiuyang DING1, Jie WANG1, Guowei TANG3, Guojie LIU4, Yun WANG4, Changwu WAN1(
), Jin PENG1,2, Jiuyang DING1, Jie WANG1, Guowei TANG3, Guojie LIU4, Yun WANG4, Changwu WAN1( ), Cuiyun LE1(
), Cuiyun LE1( )
)
Received:2025-01-05
Online:2025-06-20
Published:2025-06-27
Contact:
Changwu WAN, Cuiyun LE
E-mail:372732293@qq.com;wcw005@sina.com;675493755@qq.com
Supported by:摘要:
目的 探讨转录激活因子3(ATF3)在动脉粥样硬化斑块内的表达及其调控炎症反应参与动脉粥样硬化(AS)进程的机制。 方法 收集尸检案例中的人冠状动脉标本,免疫荧光和Western blotting检测ATF3蛋白表达及分布;高脂饮食12周后的载脂蛋白E基因敲除(ApoE-/-)小鼠尾静脉注射9型腺相关病毒(AAV9)敲低ATF3的表达,继续高脂喂养5周构建ATF3基因敲除的动脉粥样硬化ApoE-/-小鼠模型。麻醉处死后观察主动脉斑块结构改变,检测斑块内ATF3、炎症相关因子及NF-κB信号通路蛋白表达改变,并在体外构建ATF3的过表达质粒及siRNA干预THP-1诱导的泡沫细胞模型验证ATF3与NF-κB信号通路间关系。 结果 在人冠状动脉粥样硬化斑块内,ATF3的表达增高(P<0.05),且与CD68呈部分共表达;在ATF3基因敲除后,小鼠的主动脉斑块体积增大(P<0.05),斑块内炎症相关因子(CD45、CD68、IL-1β、TNF-α)表达增强(P<0.05),NF-κB信号通路相关蛋白(P-IKKα/β、P-NF-κB p65)表达增高(P<0.05),VCAM1、MMP9及MMP2表达增强(P<0.05);在离体THP-1细胞实验中验证了沉默ATF3后NF-κB信号通路进一步激活,而过表达ATF3后NF-κB信号受到抑制。 结论 动脉粥样硬化诱导ATF3的表达,ATF3的缺乏会促进AS的发生,增强斑块内炎症反应,其机制可能是通过进一步激活NF-κB信号通路导致,ATF3可能是AS潜在的治疗靶点。
夏冰, 彭进, 丁九阳, 王杰, 唐国伟, 刘国杰, 王沄, 万昌武, 乐翠云. ATF3通过NF-κB信号通路调控动脉粥样硬化斑块内的炎症反应[J]. 南方医科大学学报, 2025, 45(6): 1131-1142.
Bing XIA, Jin PENG, Jiuyang DING, Jie WANG, Guowei TANG, Guojie LIU, Yun WANG, Changwu WAN, Cuiyun LE. ATF3 regulates inflammatory response in atherosclerotic plaques in mice through the NF-κB signaling pathway[J]. Journal of Southern Medical University, 2025, 45(6): 1131-1142.
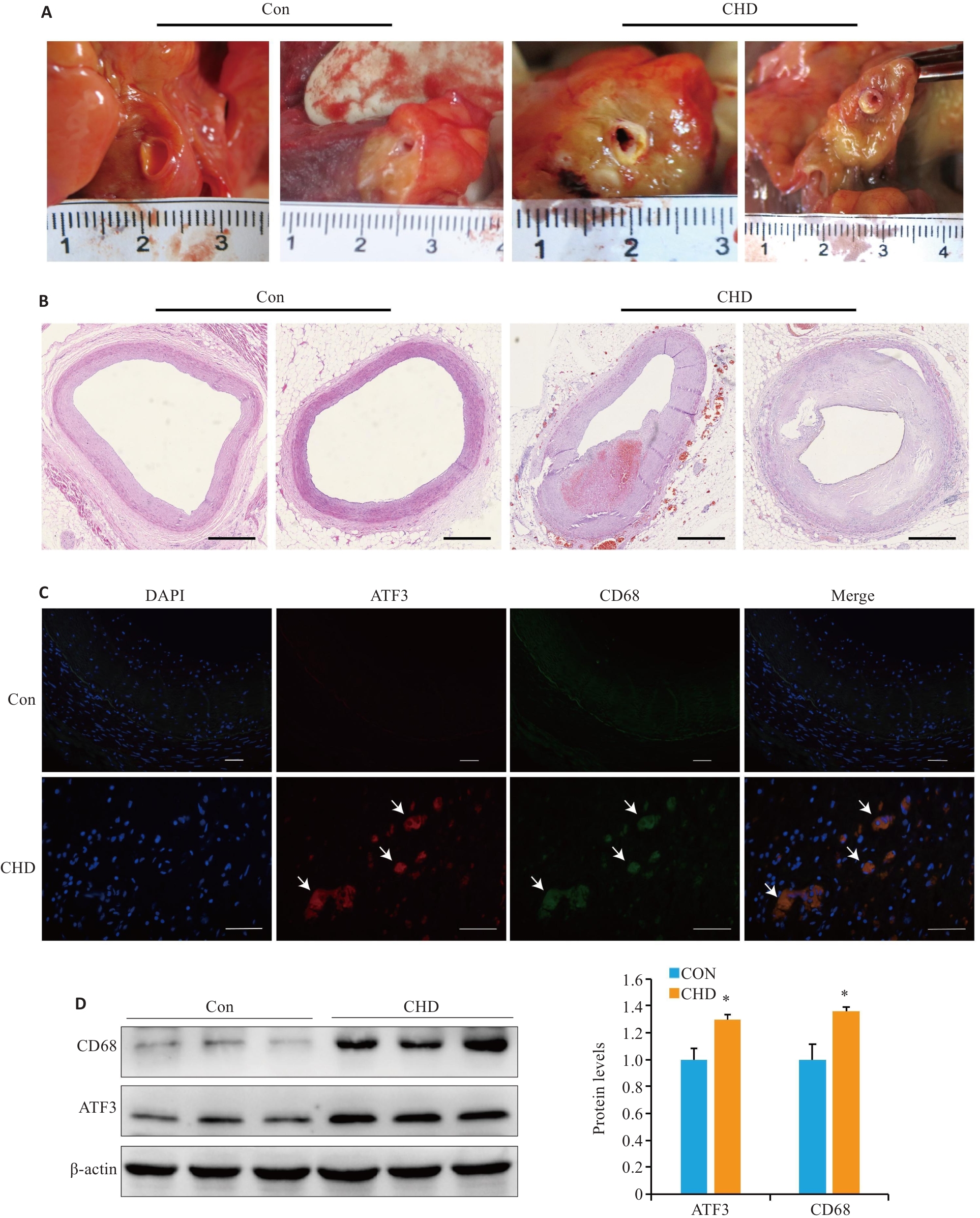
图1 人冠状动脉实验
Fig.1 Examinations of human coronary arteries. A: Human coronary artery tissue specimens. B: Microscopic observation of coronary atherosclerosis (AS; HE staining, scale bar=1 mm). C: Partial co-localization of ATF3 (red) and CD68 (green) in the plaques (arrow,scale bar=50 μm). D: Western blotting for detecting ATF3 and CD68 expressions in the plaques. All experiments were repeated at least 3 times, and data are presented as Mean±SD (n=20). *P<0.05 vs Con group.
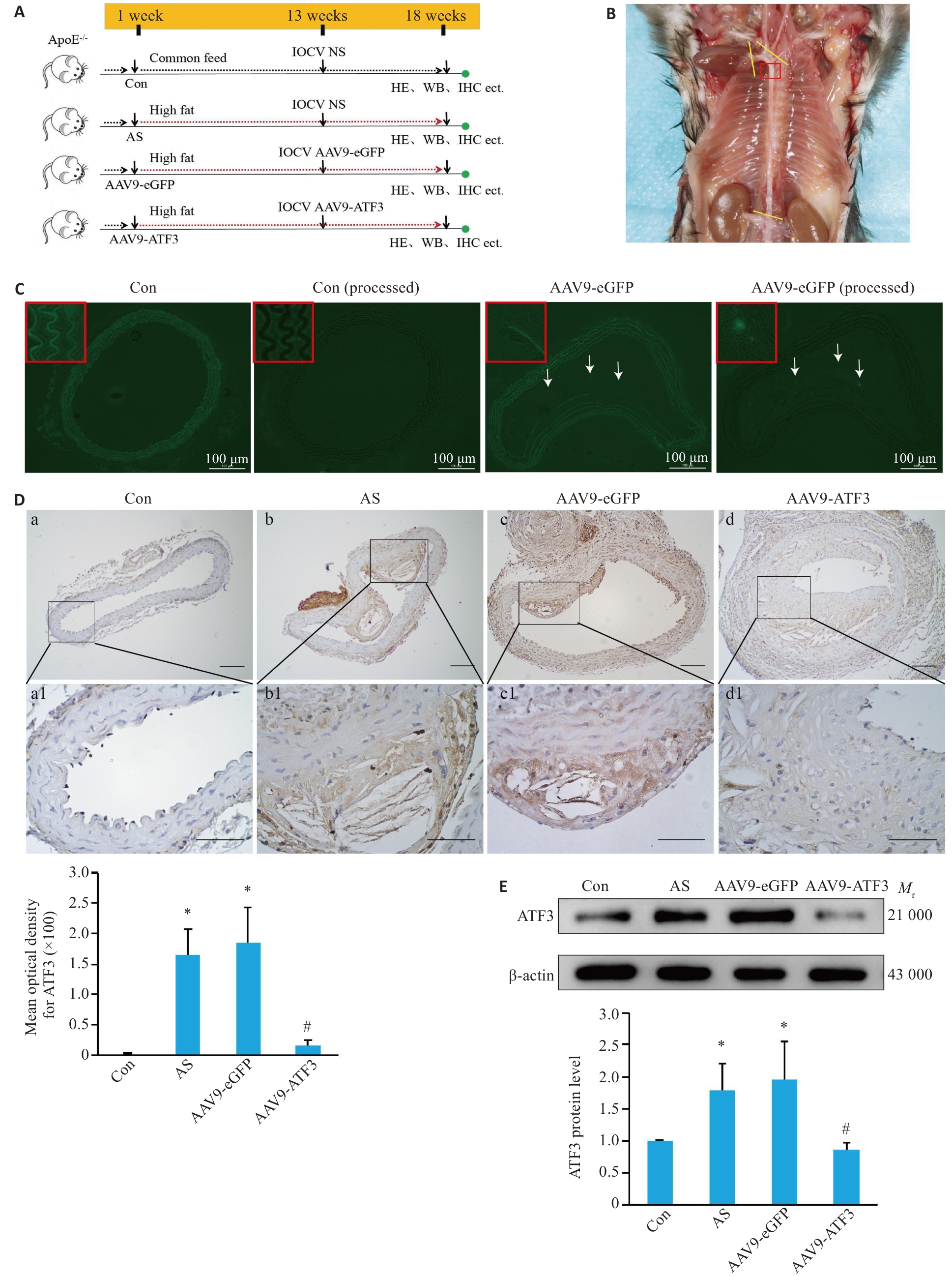
图2 ApoE-/-小鼠ATF3基因敲除模型鉴定
Fig.2 Verification of ATF3 gene knockout in ApoE-/- mice. A: Schematic diagram of mouse grouping and treatment. B: Observation of a mouse aorta. The yellow lines indicate the sampling site, and the red box indicates the tissues used for preparing frozen sections or paraffin sections. C: Expression of eGFP in the aortic vessels. After masking autofluorescence in the mouse aortas with pontamine sky blue, no fluorescence signals were detected in the vessels in the control group, but eGFP expression (arrows) was observed in the aortic plaques in AAV9-eGFP group. D: Immunohistochemical staining for detecting distribution of ATF3 expression in the aorta (a1-d1 are enlarged images of the boxed areas in a-d. Scale bar=50 μm, n=6). E: Western blotting for detecting aortic ATF3 protein content (n=6). Data are presented as Mean±SD. *P<0.05 vs Con group; #P<0.05 vs AS or AAV9-eGFP group.
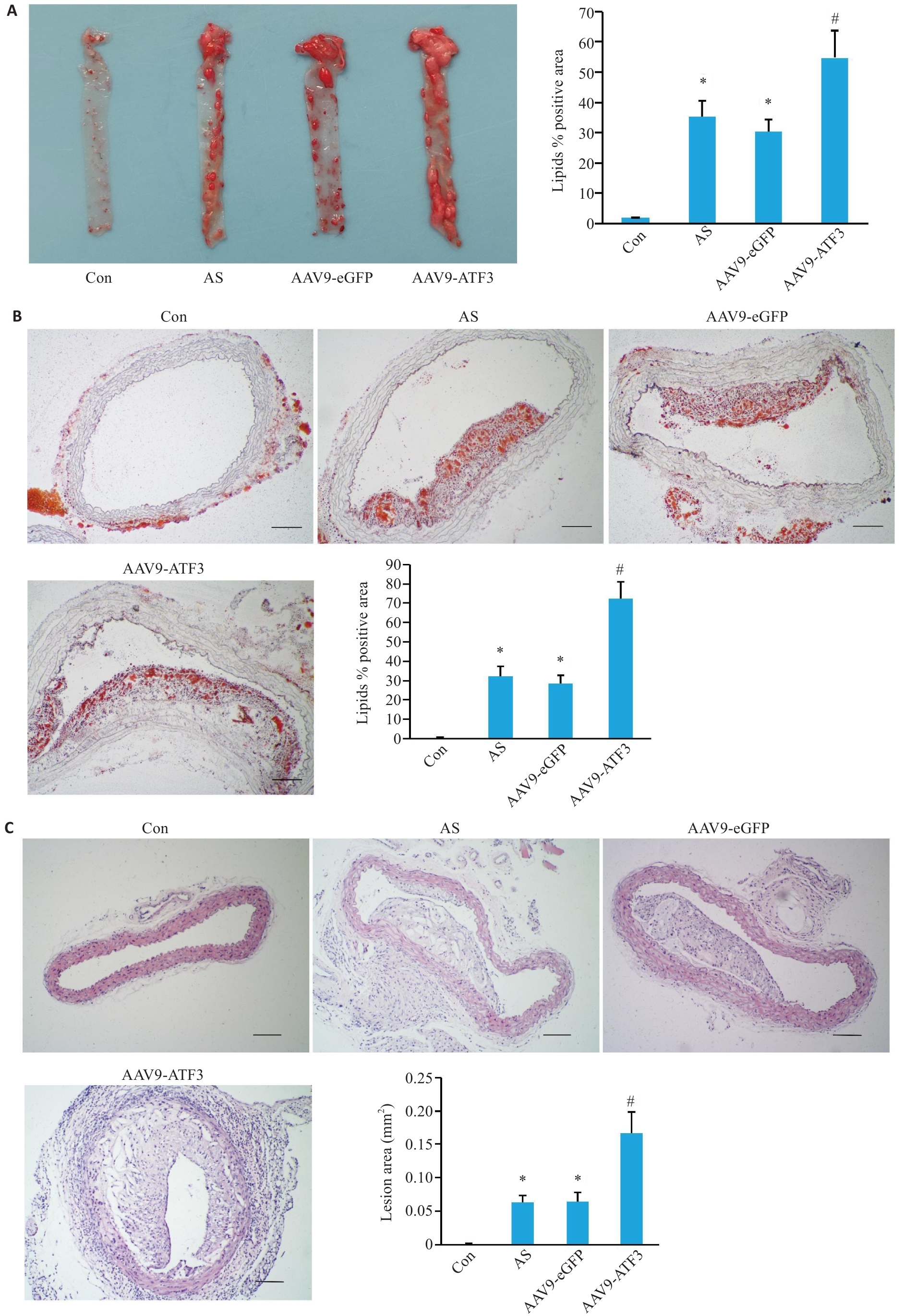
图3 ATF3基因敲除后ApoE-/-小鼠主动脉斑块改变
Fig.3 Changes in mouse aortic plaques after ATF3 gene knockout. A: Gross oil red O staining of the aorta showing lipid deposition in the total vascular area (n=6). B: Oil red O staining of cross-sections within the plaque area showing lipid deposition in the aortic lumen (n=3). C: HE staining of the aortic plaque area (n=6). Scale bar=50 μm. Data are presented as Mean±SD. *P<0.05 vs Con group, #P<0.05 vs AS or AAV9-eGFP group.
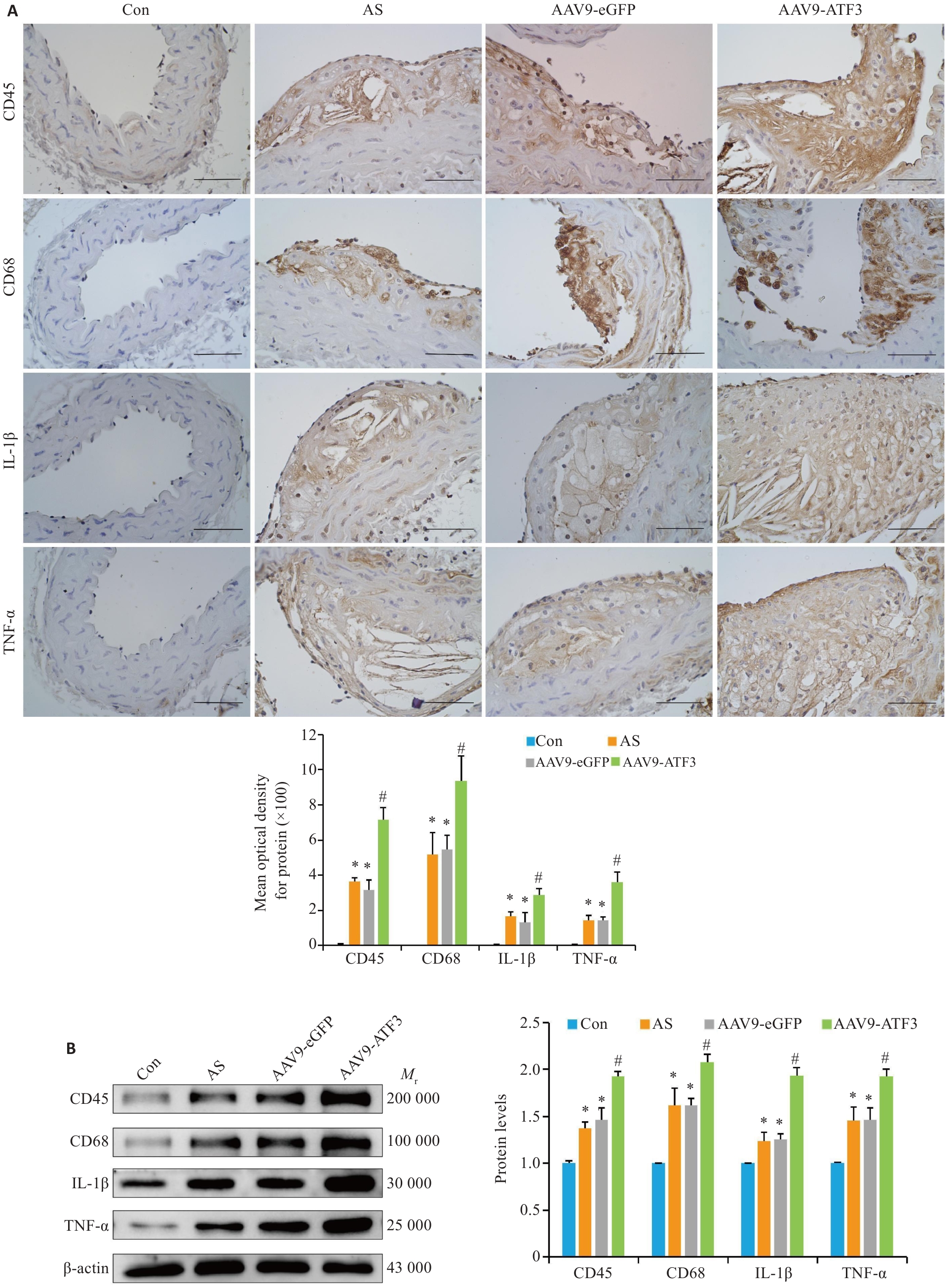
图4 ATF3基因敲除后斑块内炎症因子表达增强
Fig.4 ATF3 gene knockout increases expressions of inflammatory factors in aortic plaques of the mice. A: Immunohistochemical staining for detecting aortic CD45, CD68, IL-1β and TNF-α protein expressions and MOD value statistics (scale bar=50 μm, n=6). B: Western blotting of aortic CD45, CD68, IL-1β and TNF-α protein expressions (n=6). Data are presented as Mean±SD. *P<0.05 vs Con group, #P<0.05 vs Con, AS or AAV9-eGFP group.
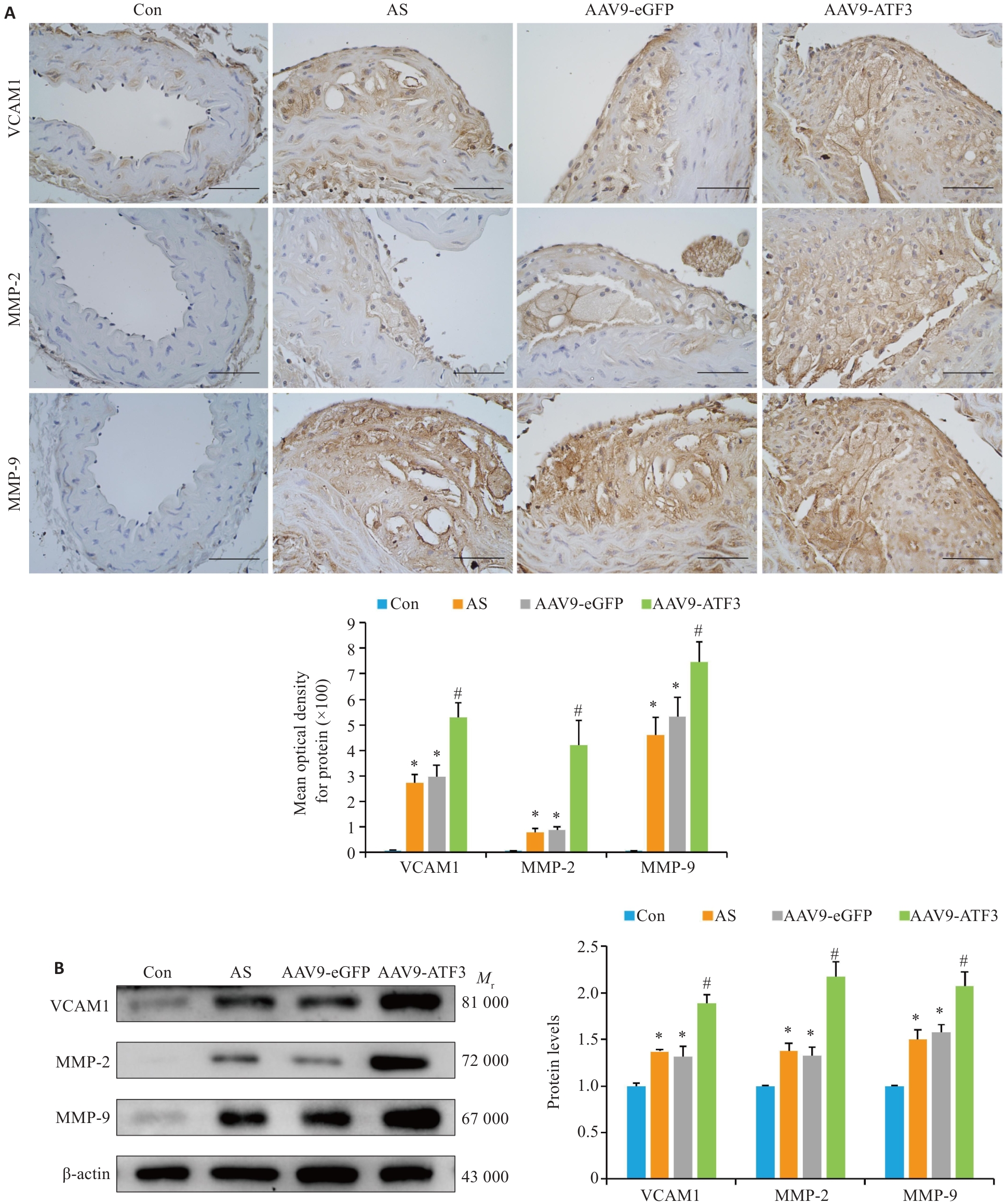
图5 ATF3基因敲除后斑块内VCAM1、MMP-2和MMP-9蛋白增强
Fig. 5 ATF3 knockout increases expressions of VCAM1, MMP-2 and MMP-9 in aortic plaques of the mice. A: Immunohistochemical staining for detecting aortic VCAM1, MMP-2 and MMP-9 protein expressions and MOD value statistics (scale bar=50 μm, n=6). B: Western blotting for detecting aortic VCAM1, MMP-2 and MMP-9 protein expressions (n=6). Data are presented as Mean±SD. *P<0.05 vs Con group, #P<0.05 vs AS or AAV9-eGFP group.
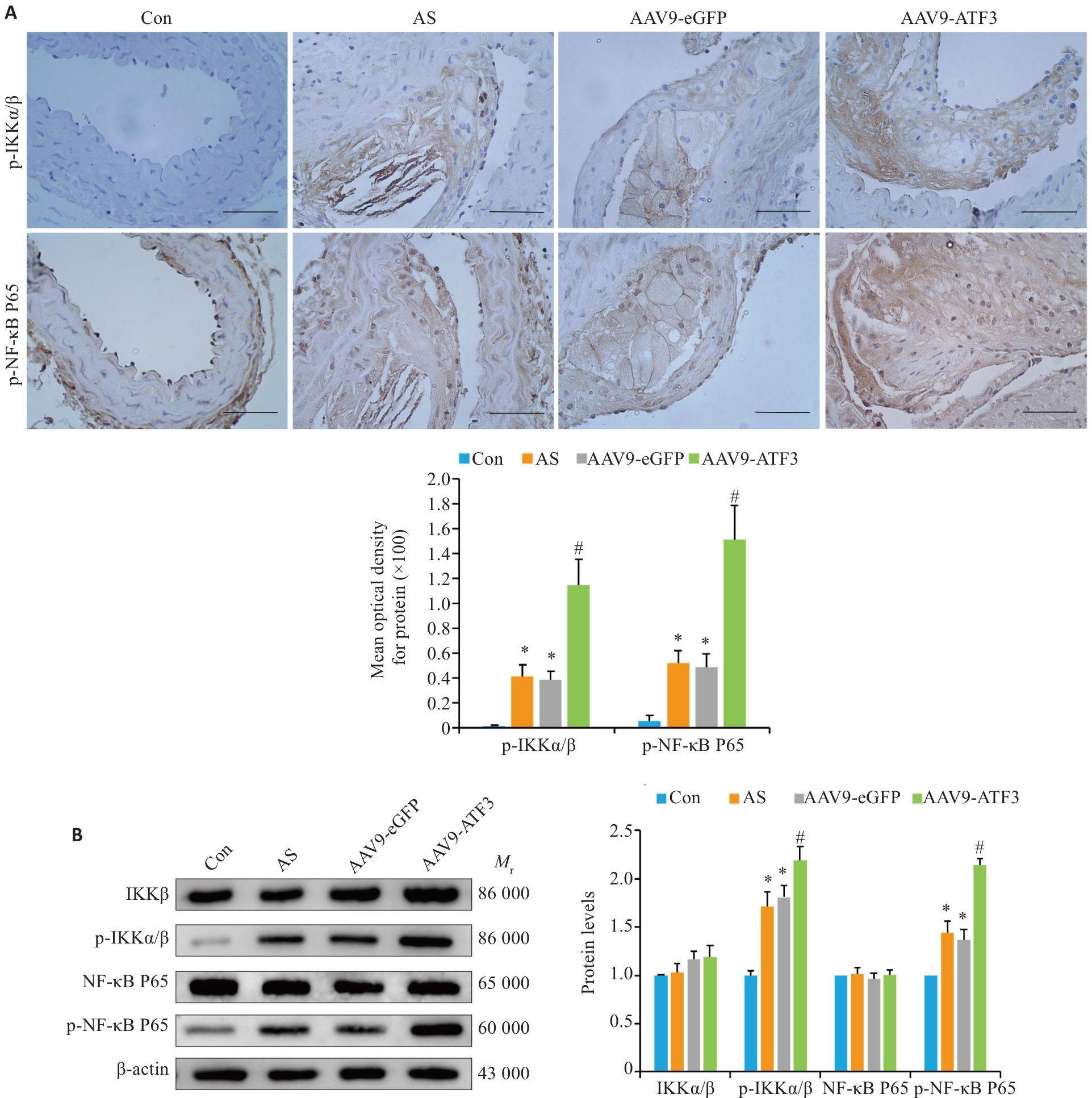
图6 ATF3基因敲除后NF-κB信号通路激活
Fig.6 ATF3 knockout activates the NF-κB signaling pathway in aortic plaques of the mice. A: Immunohistochemical staining for detecting aortic p-IKKα/β and p-NF-κB P65 protein expressions and MOD value statistics (scale bar=50 μm, n=6). B: Western blotting for detecting aortic IKKα/β, p-IKKα/β, NF-κB P65 and p-NF-κB P65 expressions (n=6). Data are presented as Mean±SD. *P<0.05 vs Con group, #P<0.05 vs AS or AAV9-eGFP group.

图7 细胞实验结果
Fig.7 Cell experiment results. A: THP-1 cells, macrophages and foam cells stained with oil red O to identify lipid deposition (scale bar=50 μm). B: Western blotting for detecting ATF3 silencing efficiency and IKKα/β, p-IKKα/β, NF-κB P65, and p-NF-κB P65 protein expressions. C: Western blotting for detecting ATF3 overexpression efficiency and IKKα/β, p-IKKα/β, NF-κB P65, and p-NF-κB P65 protein expressions. Data are presented as Mean±SD. *P<0.05 vs PMA group, #P<0.05 vs PMA+Ox-LDL, PMA+Ox-LDL+Lip3000 or PMA+Ox-LDL+OE-vector group.
| 1 | Grandhi GR, Mszar R, Vahidy F, et al. Sociodemographic disparities in influenza vaccination among adults with atherosclerotic cardiovascular disease in the United States[J]. JAMA Cardiol, 2021, 6(1): 87-91. |
| 2 | Döring Y, van der Vorst EPC, Weber C. Targeting immune cell recruitment in atherosclerosis[J]. Nat Rev Cardiol, 2024, 21(11): 824-40. doi:10.1038/s41569-024-01023-z |
| 3 | Akazawa H. Mechanisms of cardiovascular homeostasis and pathophysiology: from gene expression, signal transduction to cellular communication[J]. Circ J, 2015, 79(12): 2529-36. doi:10.1253/circj.cj-15-0818 |
| 4 | Ghigo A, Laffargue M, Li MC, et al. PI3K and calcium signaling in cardiovascular disease[J]. Circ Res, 2017, 121(3): 282-92. doi:10.1161/circresaha.117.310183 |
| 5 | Wang XW, Yan KY, Wen CF, et al. Simvastatin combined with resistance training improves outcomes in patients with chronic heart failure by modulating mitochondrial membrane potential and the Janus kinase/signal transducer and activator of transcription 3 signaling pathways[J]. Cardiovasc Ther, 2022, 2022: 8430733. doi:10.1155/2022/8430733 |
| 6 | Bauer AJ, Martin KA. Coordinating regulation of gene expression in cardiovascular disease: interactions between chromatin modifiers and transcription factors[J]. Front Cardiovasc Med, 2017, 4: 19. doi:10.3389/fcvm.2017.00019 |
| 7 | Hai T, Wolfgang CD, Marsee DK, et al. ATF3 and stress responses[J]. Gene Expr, 1999, 7(4-6): 321–35. |
| 8 | Thompson MR, Xu DK, Williams BRG. ATF3 transcription factor and its emerging roles in immunity and cancer[J]. J Mol Med (Berl), 2009, 87(11): 1053-60. doi:10.1007/s00109-009-0520-x |
| 9 | Zhou H, Li N, Yuan Y, et al. Activating transcription factor 3 in cardiovascular diseases: a potential therapeutic target[J]. Basic Res Cardiol, 2018, 113(5): 37. doi:10.1007/s00395-018-0698-6 |
| 10 | Peng J, Le CY, Xia B, et al. Research on the correlation between activating transcription factor 3 expression in the human coronary artery and atherosclerotic plaque stability[J]. BMC Cardiovasc Disord, 2021, 21(1): 356. doi:10.21203/rs.3.rs-155718/v1 |
| 11 | Kwon JW, Kwon HK, Shin HJ, et al. Activating transcription factor 3 represses inflammatory responses by binding to the p65 subunit of NF-κB[J]. Sci Rep, 2015, 5: 14470. doi:10.1038/srep14470 |
| 12 | Mussbacher M, Salzmann M, Brostjan C, et al. Cell type-specific roles of NF-κB linking inflammation and thrombosis[J]. Front Immunol, 2019, 10: 85. doi:10.3389/fimmu.2019.00085 |
| 13 | Zhao YL, Shao CY, Zhou HF, et al. Salvianolic acid B inhibits atherosclerosis and TNF-α-induced inflammation by regulating NF-κB/NLRP3 signaling pathway[J]. Phytomedicine, 2023, 119: 155002. doi:10.1016/j.phymed.2023.155002 |
| 14 | Sun Y, Guan J, Hou YF, et al. Silencing of junctional adhesion molecule-like protein attenuates atherogenesis and enhances plaque stability in ApoE-/- mice[J]. Clin Sci (Lond), 2019, 133(11): 1215-28. doi:10.1042/cs20180561 |
| 15 | Chen QJ, Zhai H, Li XM, et al. Recombinant adeno-associated virus serotype 9 in a mouse model of atherosclerosis: Determination of the optimal expression time in vivo [J]. Mol Med Rep, 2017, 15(4): 2090-6. doi:10.3892/mmr.2017.6235 |
| 16 | Wen YC, Ahmad F, Mohri Z, et al. Cysteamine inhibits lysosomal oxidation of low density lipoprotein in human macrophages and reduces atherosclerosis in mice[J]. Atherosclerosis, 2019, 291: 9-18. doi:10.1016/j.atherosclerosis.2019.09.019 |
| 17 | Li HY, Sun K, Zhao RP, et al. Inflammatory biomarkers of coronary heart disease[J]. Front Biosci (Schol Ed), 2018, 10(1): 185-96. doi:10.2741/s508 |
| 18 | Ma CY, Xu ZY, Wang SP, et al. Change of inflammatory factors in patients with acute coronary syndrome[J]. Chin Med J (Engl), 2018, 131(12): 1444-9. doi:10.4103/0366-6999.233953 |
| 19 | Hou PB, Fang JK, Liu ZH, et al. Macrophage polarization and metabolism in atherosclerosis[J]. Cell Death Dis, 2023, 14(10): 691. doi:10.1038/s41419-023-06206-z |
| 20 | Radecke CE, Warrick AE, Singh GD, et al. Coronary artery endo-thelial cells and microparticles increase expression of VCAM-1 in myocardial infarction[J]. Thromb Haemost, 2015, 113(3): 605-16. doi:10.1160/th14-02-0151 |
| 21 | Woudstra L, Biesbroek PS, Emmens RW, et al. CD45 is a more sensitive marker than CD3 to diagnose lymphocytic myocarditis in the endomyocardium[J]. Hum Pathol, 2017, 62: 83-90. doi:10.1016/j.humpath.2016.11.006 |
| 22 | Libby P. Interleukin-1 beta as a target for atherosclerosis therapy: biological basis of CANTOS and beyond[J]. J Am Coll Cardiol, 2017, 70(18): 2278-89. doi:10.1016/j.jacc.2017.09.028 |
| 23 | Williams JW, Huang LH, Randolph GJ. Cytokine circuits in cardiovascular disease[J]. Immunity, 2019, 50(4): 941-54. doi:10.1016/j.immuni.2019.03.007 |
| 24 | Xia F, Zeng QR, Chen J. Circulating brain-derived neurotrophic factor dysregulation and its linkage with lipid level, stenosis degree, and inflammatory cytokines in coronary heart disease[J]. J Clin Lab Anal, 2022, 36(7): e24546. doi:10.1002/jcla.24546 |
| 25 | Wågsäter D, Zhu CY, Björkegren J, et al. MMP-2 and MMP-9 are prominent matrix metalloproteinases during atherosclerosis development in the Ldlr (-/-) Apob(100/100) mouse[J]. Int J Mol Med, 2011, 28(2): 247-53. doi:10.3892/ijmm.2011.693 |
| 26 | Volkov AM, Murashov IS, Polonskaya YV, et al. Changes of content of matrix metalloproteinases and their tissue expression in various types of atherosclerotic plaques[J]. Kardiologiia, 2018(10): 12-8. doi:10.18087/cardio.2018.10.10180 |
| 27 | Hai T, Wolford CC, Chang YS. ATF3, a hub of the cellular adaptive-response network, in the pathogenesis of diseases: is modulation of inflammation a unifying component[J]? Gene Expr, 2010, 15(1): 1-11. doi:10.3727/105221610x12819686555015 |
| 28 | Udayakumar TS, Stoyanova R, Shareef MM, et al. Edelfosine promotes apoptosis in androgen-deprived prostate tumors by increasing ATF3 and inhibiting androgen receptor activity[J]. Mol Cancer Ther, 2016, 15(6): 1353-63. doi:10.1158/1535-7163.mct-15-0332 |
| 29 | Zhao J, Li XY, Guo MX, et al. The common stress responsive transcription factor ATF3 binds genomic sites enriched with p300 and H3K27ac for transcriptional regulation[J]. BMC Genomics, 2016, 17: 335. doi:10.1186/s12864-016-2664-8 |
| 30 | Jeong BC, Kim JH, Kim K, et al. ATF3 modulates calcium signaling in osteoclast differentiation and activity by associating with c-Fos and NFATc1 proteins[J]. Bone, 2017, 95: 33-40. doi:10.1016/j.bone.2016.11.005 |
| 31 | Qin WW, Yang HY, Liu GZ, et al. Activating transcription factor 3 is a potential target and a new biomarker for the prognosis of atherosclerosis[J]. Hum Cell, 2021, 34(1): 49-59. doi:10.1007/s13577-020-00432-9 |
| 32 | Gold ES, Ramsey SA, Sartain MJ, et al. ATF3 protects against atherosclerosis by suppressing 25-hydroxycholesterol-induced lipid body formation[J]. J Exp Med, 2012, 209(4): 807-17. doi:10.1084/jem.20111202 |
| 33 | Clément M, Basatemur G, Masters L, et al. Necrotic cell sensor Clec4e promotes a proatherogenic macrophage phenotype through activation of the unfolded protein response[J]. Circulation, 2016, 134(14): 1039-51. doi:10.1161/circulationaha.116.022668 |
| 34 | Ren JC, Han YS, Ren TM, et al. AEBP1 promotes the occurrence and development of abdominal aortic aneurysm by modulating inflammation via the NF-κB pathway[J]. J Atheroscler Thromb, 2020, 27(3): 255-70. doi:10.5551/jat.49106 |
| 35 | Gilchrist M, Thorsson V, Li B, et al. Systems biology approaches identify ATF3 as a negative regulator of Toll-like receptor 4[J]. Nature, 2006, 441(7090): 173-8. doi:10.1038/nature04768 |
| 36 | Li HF, Cheng CF, Liao WJ, et al. ATF3-mediated epigenetic regulation protects against acute kidney injury[J]. J Am Soc Nephrol, 2010, 21(6): 1003-13. doi:10.1681/asn.2009070690 |
| 37 | Wang L, Deng S, Lu Y, et al. Increased inflammation and brain injury after transient focal cerebral ischemia in activating tran-scription factor 3 knockout mice[J]. Neuroscience, 2012, 220: 100-8. doi:10.1016/j.neuroscience.2012.06.010 |
| [1] | 范正媛, 沈子涵, 李亚, 沈婷婷, 李高峰, 李素云. 补肺益肾方对香烟烟雾提取物诱导的人支气管上皮细胞损伤的保护作用及其机制[J]. 南方医科大学学报, 2025, 45(7): 1372-1379. |
| [2] | 梁晓涛, 熊一凡, 刘雪琪, 梁小珊, 朱晓煜, 谢炜. 活血疏风颗粒通过抑制TLR4/NF-κB通路改善慢性偏头痛小鼠的中枢敏化[J]. 南方医科大学学报, 2025, 45(5): 986-994. |
| [3] | 张毅, 沈昱, 万志强, 陶嵩, 柳亚魁, 王栓虎. CDKN3高表达促进胃癌细胞的迁移和侵袭:基于调控p53/NF-κB信号通路和抑制胃癌细胞凋亡[J]. 南方医科大学学报, 2025, 45(4): 853-861. |
| [4] | 钟娜, 王会杰, 赵文英, 孙珍贵, 耿彪. 高表达RNF7增强非小细胞肺癌细胞的PD-1耐药:基于活化NF-kB通路促进CXCL1表达和髓源性抑制细胞的募集[J]. 南方医科大学学报, 2024, 44(9): 1704-1711. |
| [5] | 左涵珺, 段兆达, 王朝, 郭涛, 石金沙, 石浩龙, 李娟娟. 天麻素经PI3K/AKT通路改善新生大鼠缺氧缺血性脑损伤后小胶质细胞介导的炎症反应[J]. 南方医科大学学报, 2024, 44(9): 1712-1719. |
| [6] | 姜一凡, 李小荣, 耿嘉逸, 陈永锋, 唐碧, 康品方. 槲皮素通过抑制HMGB1/RAGE/NF-κB信号通路减轻糖尿病引起的大鼠肾脏损伤[J]. 南方医科大学学报, 2024, 44(9): 1769-1775. |
| [7] | 赵娜, 沈梦迪, 赵睿, 奥迪, 骆泽谭, 张银亮, 徐志东, 范方田, 郑海伦. 血根碱通过调控Nrf2/NF-κB通路缓解小鼠溃疡性结肠炎[J]. 南方医科大学学报, 2024, 44(8): 1467-1475. |
| [8] | 张珊苑, 蔡巧燕, 祁江晗, 殷恺馨, 何晨晨, 高铸烨, 张铃, 褚剑锋. 清心解瘀颗粒抗动脉粥样硬化的药效学及调控机制[J]. 南方医科大学学报, 2024, 44(8): 1518-1528. |
| [9] | 肖林雨, 段婷, 夏勇生, 陈悦, 孙洋, 许轶博, 徐磊, 闫兴洲, 胡建国. 蒙花苷通过抑制TLR4/NF-κB通路抑制小鼠脊髓损伤后小胶质细胞活化介导的神经炎症和神经元凋亡[J]. 南方医科大学学报, 2024, 44(8): 1589-1598. |
| [10] | 陶怀祥, 骆金光, 闻志远, 虞亘明, 苏萧, 王鑫玮, 关翰, 陈志军. STING高表达通过调控TLR4/NF-κB/NLRP3通路和影响炎症与凋亡水平促进小鼠肾脏缺血再灌注损伤[J]. 南方医科大学学报, 2024, 44(7): 1345-1354. |
| [11] | 何 程, 陈 炜, 张念志, 栾 军, 王三凤, 张 尤. 参七虫草方通过ASS1/src/STAT3信号通路改善肺纤维化大鼠的炎症反应[J]. 南方医科大学学报, 2024, 44(4): 644-651. |
| [12] | 包汉生, 王苏童, 吕穆杰, 王永成, 姜 萍, 李 晓. 激活α7nAchR促进肥胖小鼠的脂肪稳态和米色脂肪生成及产热作用[J]. 南方医科大学学报, 2024, 44(3): 499-506. |
| [13] | 吴广阳, 宋添力, 唐 浪, 王一民, 刘 绪, 黄 胜. 竹节参总皂苷缓解CCl4诱导的大鼠急性肝损伤:基于调控PI3K/Akt/NF-κB信号通路[J]. 南方医科大学学报, 2024, 44(2): 244-251. |
| [14] | 牛民主, 殷丽霞, 段婷, 黄菊, 李静, 耿志军, 胡建国, 宋传旺. 川续断皂苷VI通过抑制PI3K/AKT/NF-κB通路拮抗肠上皮细胞凋亡缓解TNBS诱导的小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2024, 44(12): 2335-2346. |
| [15] | 孙秀颀, 蔡静, 张安邦, 庞博, 陈春艳, 查琪琪, 全菲, 叶涛. 电针预处理通过抑制NF-kB/NLRP3信号通路介导炎症和凋亡改善大鼠脑卒中后痉挛[J]. 南方医科大学学报, 2024, 44(11): 2102-2109. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||