南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 479-488.doi: 10.12122/j.issn.1673-4254.2026.03.01
• 基础研究 •
刘超群1,2( ), 宁紫燕2(
), 宁紫燕2( ), 吴江华1,2, 刘魏魏2, 林创1, 许嘉玮3, 周蕊1,2(
), 吴江华1,2, 刘魏魏2, 林创1, 许嘉玮3, 周蕊1,2( ), 赵亮1,2(
), 赵亮1,2( )
)
收稿日期:2025-10-28
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
周蕊,赵亮
E-mail:2436965229@qq.com;13277483682@163.com;yaruisunny@sina.com;liangsmu@foxmail.com
作者简介:刘超群,博士,医师,E-mail: 2436965229@qq.com基金资助:
Chaoqun LIU1,2( ), Ziyan NING2(
), Ziyan NING2( ), Jianghua WU1,2, Weiwei LIU2, Chuang LIN1, Jiawei XU3, Rui ZHOU1,2(
), Jianghua WU1,2, Weiwei LIU2, Chuang LIN1, Jiawei XU3, Rui ZHOU1,2( ), Liang ZHAO1,2(
), Liang ZHAO1,2( )
)
Received:2025-10-28
Online:2026-03-20
Published:2026-03-26
Contact:
Rui ZHOU, Liang ZHAO
E-mail:2436965229@qq.com;13277483682@163.com;yaruisunny@sina.com;liangsmu@foxmail.com
Supported by:摘要:
目的 探索牙本质涎磷蛋白(DSPP)对结直肠癌奥沙利铂治疗效果的影响,及其作用机制,评估靶向DSPP对结直肠癌的治疗疗效。 方法 将奥沙利铂耐药HCT8细胞(OxR/HCT8)、结直肠癌类器官(PDTOs)、HCT116及SW620细胞设为sgNC对照组与sgDSPP敲除组,以CCK-8实验检测敲除DSPP后细胞对奥沙利铂的敏感性;将HCT116、SW620细胞设为sgNC、sgDSPP敲除组,Caco2及HCT8细胞设为oeNC对照、oeDSPP过表达组、oeDSPP过表达+Cyclo(整合素αvβ3抑制剂)组,Western blotting验证OxR/HCT8中 DSPP的表达水平,DSPP过表达、敲除细胞株构建以及对MAPK信号通路的调控。体内实验:采用4~5周龄BALB/c-nu裸鼠构建皮下成瘤模型,随机分3组(5只/组):Glu对照组、L-OHP(奥沙利铂)组、L-OHP+α-DSPP(DSPP单克隆抗体)联合组,验证联合治疗疗效。构建结直肠癌PDX模型,其中3例以4~5周龄NOD-SCID小鼠分4组(5~6只/组):Glu组、α-DSPP组、L-OHP组、L-OHP+α-DSPP组,验证靶向DSPP对奥沙利铂敏感性的影响;其余2例分6组(4~6只/组),新增Cyclo组及L-OHP+Cyclo组,对比靶向DSPP与整合素αvβ3的作用差异。免疫荧光染色、Co-IP、免疫组织化学染色实验分析DSPP与整合素αvβ3相互作用;HE染色及免疫荧光实验验证类器官模型的成功构建;免疫组织化学染色分析DSPP在结直肠癌奥沙利铂治疗敏感与耐药患者中的表达强弱。 结果 免疫组织化学染色结果显示,与对奥沙利铂敏感结直肠癌患者相比(n=30),DSPP表达水平在奥沙利铂耐药患者(n=30)中显著升高(P<0.001);OxR/HCT8细胞中DSPP表达上调,并且在OxR/HCT8细胞、CRC类器官模型、HCT116以及SW620细胞中敲除DSPP均能增加奥沙利铂敏感性;Co-IP及免疫荧光实验证实肿瘤细胞DSPP与整合素αvβ3二者间具有相互结合及共定位;免疫组织化学染色发现结直肠癌患者肿瘤组织中DSPP表达较高时,整合素αvβ3表达水平也相对较高;DSPP可以上调MAPK信号通路中ERK及P53的磷酸化水平,而整合素αvβ3抑制剂能有效逆转DSPP的这一调控作用;裸鼠及PDX模型证实靶向DSPP及整合素αvβ3可以抑制肿瘤的生长(P<0.05),减小肿瘤的重量(P<0.05),增加结直肠癌奥沙利铂疗效(P<0.05),而DSPP单克隆抗体的促进作用优于αvβ3靶向抑制剂。 结论 DSPP通过整合素αvβ3调控结直肠癌奥沙利铂耐药的新机制,确立DSPP有望成为提高晚期结直肠癌患者对奥沙利铂治疗敏感性的新靶点。
刘超群, 宁紫燕, 吴江华, 刘魏魏, 林创, 许嘉玮, 周蕊, 赵亮. 结直肠癌细胞分泌牙本质涎磷蛋白通过整合素αvβ3依赖途径诱导奥沙利铂耐药[J]. 南方医科大学学报, 2026, 46(3): 479-488.
Chaoqun LIU, Ziyan NING, Jianghua WU, Weiwei LIU, Chuang LIN, Jiawei XU, Rui ZHOU, Liang ZHAO. Tumor-secreted dentin sialophosphoprotein induces oxaliplatin resistance in colorectal cancer through an integrin αvβ3-dependent pathway[J]. Journal of Southern Medical University, 2026, 46(3): 479-488.
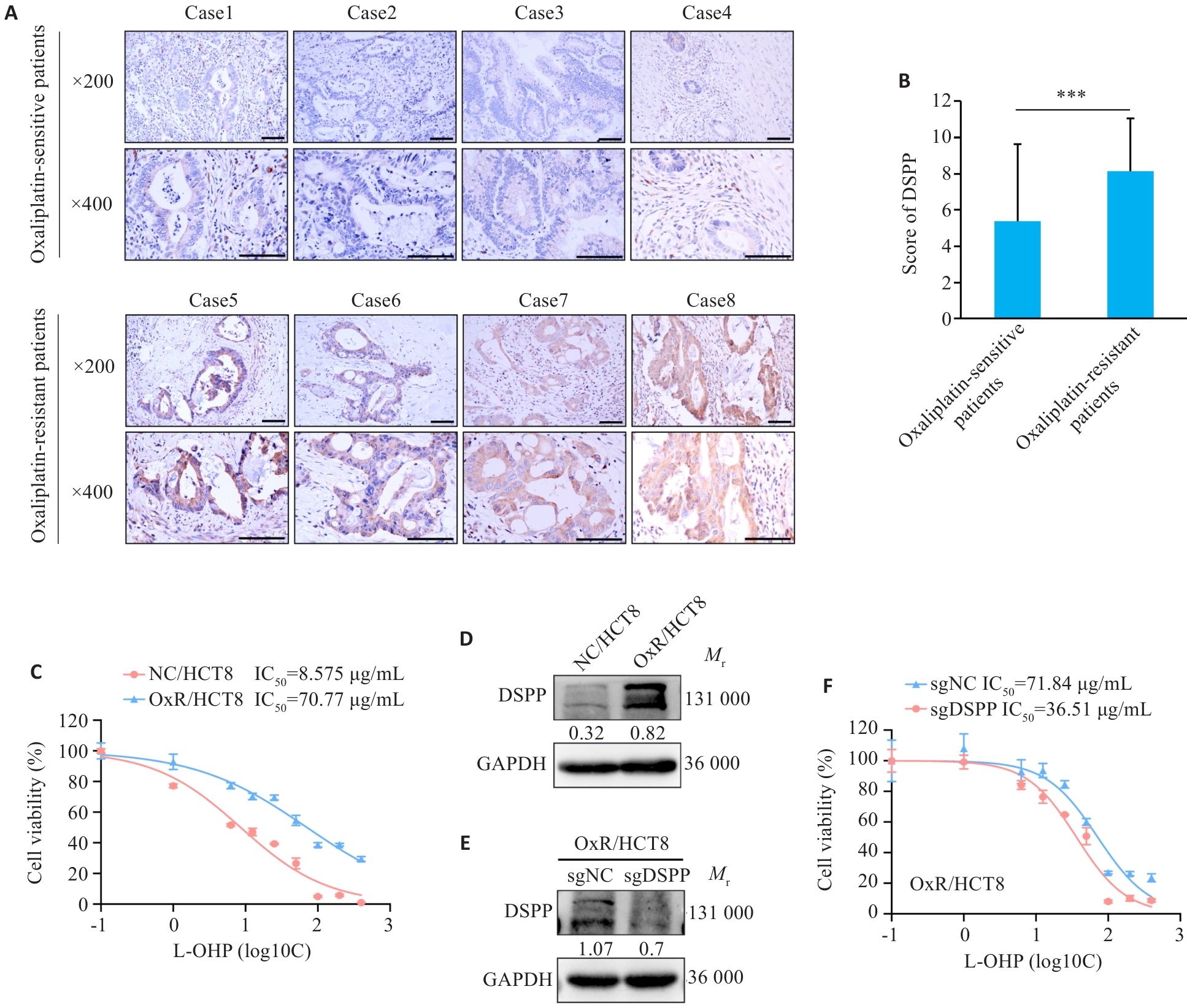
图1 人结直肠癌患者中DSPP的表达与奥沙利铂敏感性的相关性
Fig.1 DSPP correlates with oxaliplatin responsiveness in colorectal cancer (CRC). A: Immunohistochemical (IHC) images showing DSPP expression in tumors from oxaliplatin (L-OHP)‑sensitive and ‑resistant CRC patients (***P<0.001). B: Quantification of DSPP IHC scores in L-OHP-sensitive and -resistant CRC tumors (n=30). C: CCK-8 assay for assessing viability of L-OHP-resistant HCT8 cell line (n=4). D: Western blotting of DSPP in L-OHP-sensitive HCT8 and L-OHP-resistant HCT8 cells. E: Western blotting confirming stable DSPP knockout in HCT8 cells. F: CCK-8 assay showing increased L-OHP sensitivity in DSPP-knockout HCT8 cells (n=4).
| Characteristic | Level | Oxaliplatin-sensitive group (n=30) | Oxaliplatin-resistant group (n=30) | P |
|---|---|---|---|---|
表1 结直肠癌样本中奥沙利铂敏感组与耐药组基线特征表
Tab.1 Baseline characteristics of oxaliplatin-sensitive and oxaliplatin-resistant colorectal cancer (CRC) samples
| Characteristic | Level | Oxaliplatin-sensitive group (n=30) | Oxaliplatin-resistant group (n=30) | P |
|---|---|---|---|---|
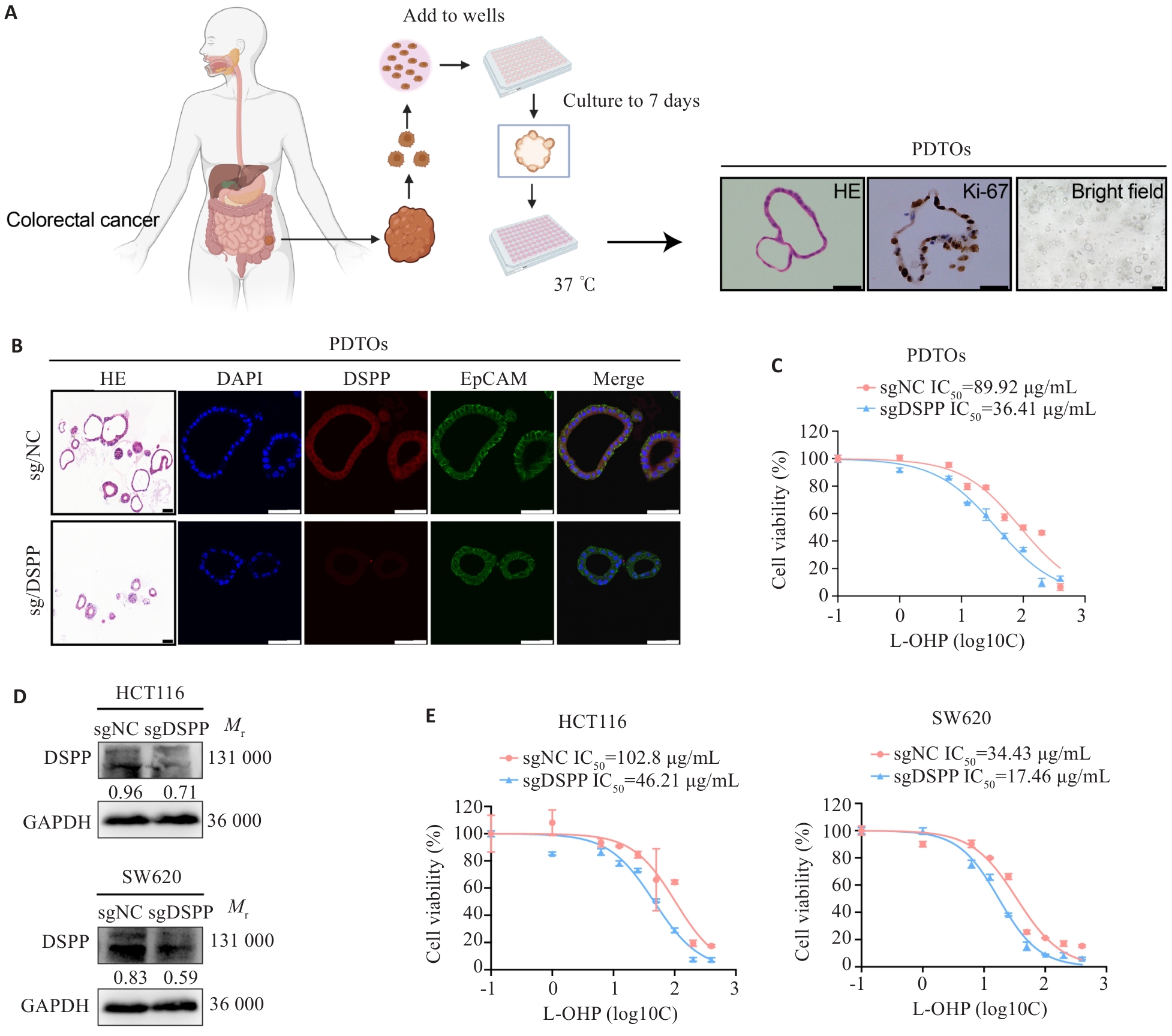
图2 DSPP促进结直肠癌类器官和细胞系对奥沙利铂耐药
Fig.2 DSPP depletion enhances oxaliplatin resistance in human CRC organoids and cell lines. A: Representative schematic of the establishment and characterization of patient-derived tumor organoids (PDTOs). Scale bar=100 μm. B: HE and immunofluorescence staining showing DSPP expression in edited PDTOs. Scale bar=100 μm. C: CCK-8 assay showing significantly increased L-OHP sensitivity in DSPP-knockout PDTOs (n=4). D: Western blotting showing the efficiency of DSPP knockout in HCT116 and SW620 cells. E: CCK-8 assays showing increased L-OHP sensitivity in DSPP knockout HCT116 and SW620 cells (n=4).
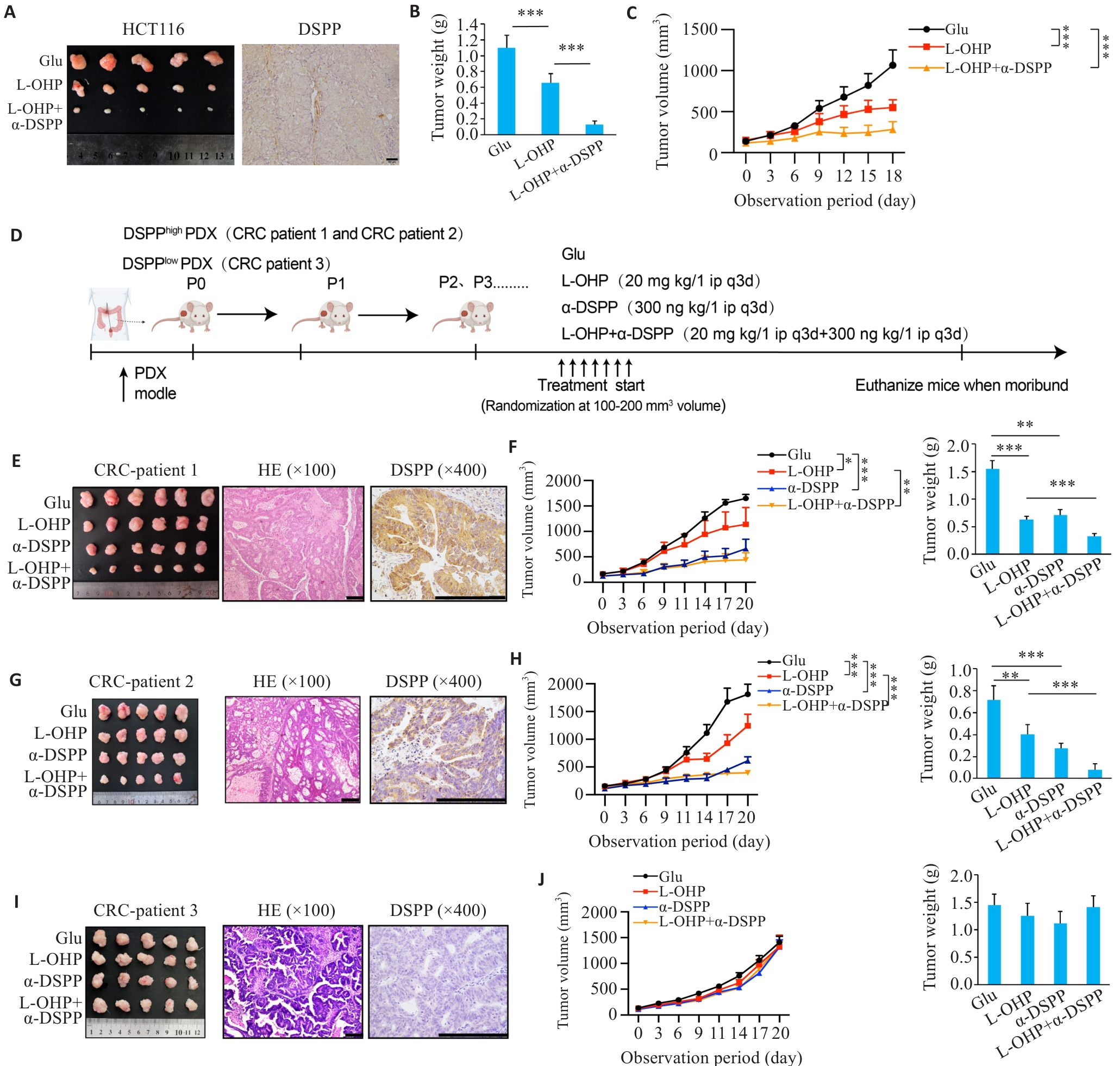
图3 靶向DSPP增强奥沙利铂在裸鼠模型和人源肿瘤异种移植模型中的疗效
Fig.3 Targeting DSPP augments oxaliplatin efficacy in xenograft and PDX models.A: Representative subcutaneous xenograft tumors and IHC demonstrating the efficacy of combining L-OHP with a DSPP-targeting therapy (n=5). Scale bar=50 μm. B: Endpoint tumor weights in each group (n=5). C: Tumor growth curves over time in each group (n=5). D: Schematic diagram of patient-derived xenograft (PDX) establishment and treatment regimens. E, G, I: Representative macroscopic tumors, HE staining, and DSPP IHC from 3 independent PDX cases (n=5 or 6). Scale bar=200 μm. F, H, J: Tumor growth curves and tumor weights in the corresponding PDX cases. *P<0.05, **P<0.01, ***P<0.001.
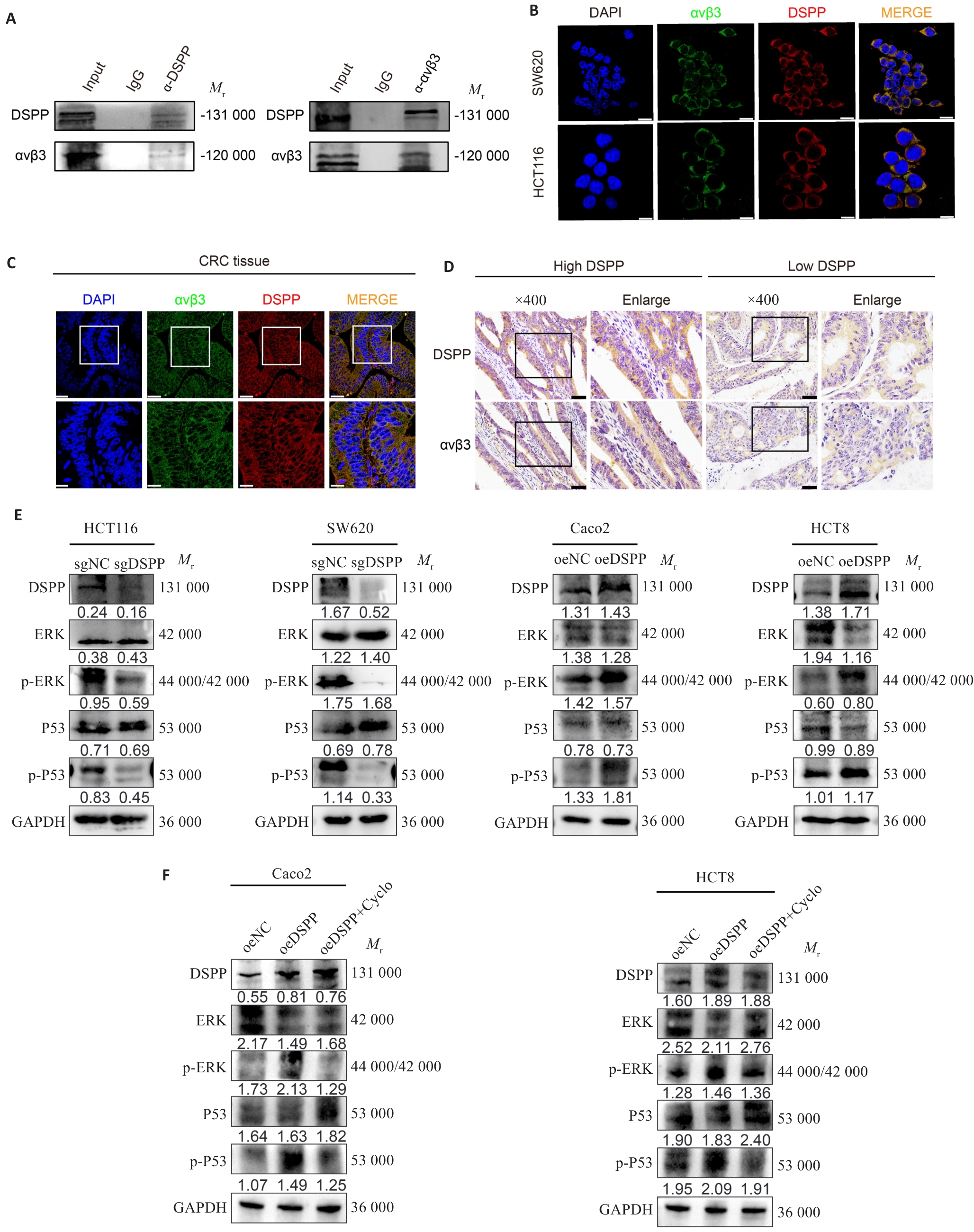
图4 DSPP与整合素αvβ3相互作用激活MAPK信号通路
Fig.4 DSPP interacts with integrin αvβ3 and modulates MAPK signaling. A, B: Co-immunoprecipitation (Co-IP) and immunofluorescence images showing the interaction and subcellular co-localization of DSPP and integrin αvβ3 in CRC cells. Scale bar=10 μm. C: Immunofluorescence images showing co-localization of DSPP and αvβ3 in CRC tissues. Scale bar=50 μm. D: IHC showing the correlation between DSPP and integrin αvβ3 expression in human CRC specimens. Scale bar=50 μm. E, F: Western blotting showing that DSPP regulates MAPK/ERK and p53 pathways in the indicated CRC cell lines, and the integrin-targeted inhibitor Cyclo (at a concentration of 38.33 ng/mL) reverses DSPP-induced upregulation of ERK and P53 phosphorylation levels.
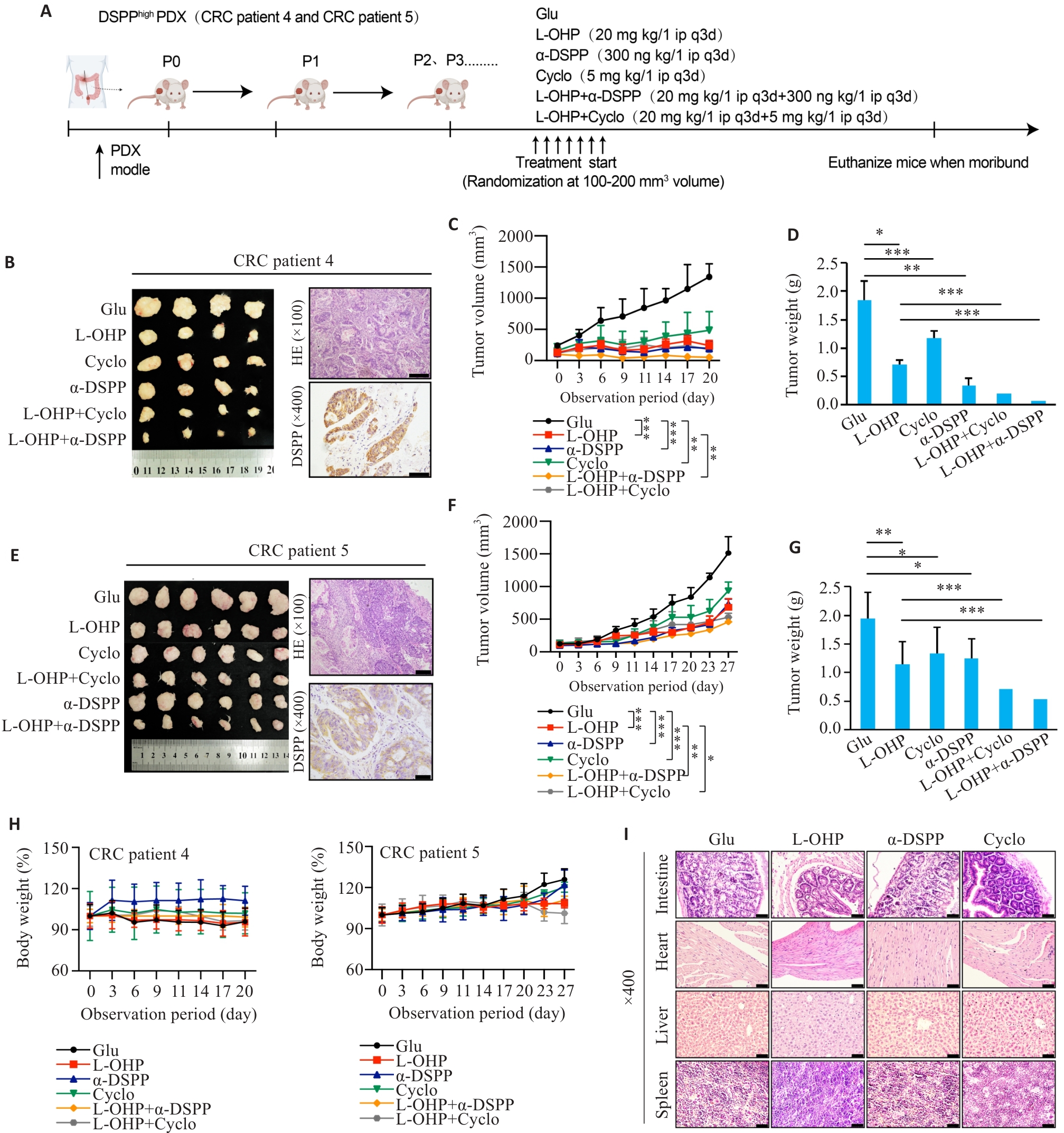
图5 PDX模型证实靶向DSPP、整合素αvβ3增强结直肠癌PDX奥沙利铂化疗敏感性
Fig.5 Targeting DSPP or integrin αvβ3 enhances oxaliplatin efficacy in CRC PDX models. A: Schematic diagram of the PDX experimental design for evaluating L-OHP combined with DSPP-targeting antibody or αvβ3 inhibitor Cyclo(-RGDfK). B, E: Representative macroscopic tumors, HE staining, and DSPP IHC from two independent PDX cases (n=4-6). Scale bar=100 μm. C, F: Tumor growth curves for the indicated treatment groups (n=4-6). D, G: Endpoint tumor weights in each group (n=4-6). H: Body-weight monitoring of PDX mice across different treatment groups, showing no significant toxicity (n=4-6). I: HE staining of major organs (intestine, liver, heart, and spleen) from PDX mice across the treatment groups. Scale bar=50 μm. *P<0.05, **P<0.01, ***P<0.001.
| [1] | Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025[J]. CA A Cancer J Clin, 2025, 75(1): 10-45. doi:10.3322/caac.21871 |
| [2] | Filho AM, Laversanne M, Ferlay J, et al. The GLOBOCAN 2022 cancer estimates: Data sources, methods, and a snapshot of the cancer burden worldwide[J]. Int J Cancer, 2025, 156(7): 1336-46. doi:10.1002/ijc.35278 |
| [3] | Han BF, Zheng RS, Zeng HM, et al. Cancer incidence and mortality in China, 2022[J]. J Natl Cancer Cent, 2024, 4(1): 47-53. doi:10.1016/j.jncc.2024.01.006 |
| [4] | Ciardiello F, Ciardiello D, Martini G, et al. Clinical management of metastatic colorectal cancer in the era of precision medicine[J]. CA Cancer J Clin, 2022, 72(4): 372-401. doi:10.3322/caac.21728 |
| [5] | Van Cutsem E, Cervantes A, Adam R, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer[J]. Ann Oncol, 2016, 27(8): 1386-422. |
| [6] | Bellahcène A, Castronovo V, Ogbureke KUE, et al. Small integrin-binding ligand N-linked glycoproteins (SIBLINGs): multifunctional proteins in cancer[J]. Nat Rev Cancer, 2008, 8(3): 212-26. doi:10.1038/nrc2345 |
| [7] | Fan SC, Gao HS, Sun LZ, et al. Knockdown of DSPP inhibits the migration and invasion of glioma cells[J]. Pathol Res Pract, 2018, 214(12): 2025-30. doi:10.1016/j.prp.2018.09.024 |
| [8] | Gu Y, Taifour T, Bui T, et al. Osteopontin is a therapeutic target that drives breast cancer recurrence[J]. Nat Commun, 2024, 15(1): 9174. doi:10.1038/s41467-024-53023-9 |
| [9] | Aseervatham J, Ogbureke KUE. Effects of DSPP and MMP20 silencing on adhesion, metastasis, angiogenesis, and epithelial-mesenchymal transition proteins in oral squamous cell carcinoma cells[J]. Int J Mol Sci, 2020, 21(13): 4734. doi:10.3390/ijms21134734 |
| [10] | Nikitakis NG, Gkouveris I, Aseervatham J, et al. DSPP-MMP20 gene silencing downregulates cancer stem cell markers in human oral cancer cells[J]. Cell Mol Biol Lett, 2018, 23: 30. doi:10.1186/s11658-018-0096-y |
| [11] | Liu D, Luo M, Hu J, et al. Osteopontin enhances cisplatin resistance of human A549 lung cancer cells via stimulating the PI3K signaling pathway and upregulating ERCC1 expression[J]. Transl Cancer Res, 2020, 9(5): 3258-65. doi:10.21037/tcr.2020.03.60 |
| [12] | Panda VK, Mishra B, Nath AN, et al. Osteopontin: a key multifaceted regulator in tumor progression and immunomodulation[J]. Biomedicines, 2024, 12(7): 1527. doi:10.3390/biomedicines12071527 |
| [13] | Qin YC, Yan X, Yuan XL, et al. Osteopontin promotes gastric cancer progression via phosphatidylinositol-3-kinase/protein kinase B/mammalian target of rapamycin signaling pathway[J]. World J Gastrointest Oncol, 2023, 15(9): 1544-55. doi:10.4251/wjgo.v15.i9.1544 |
| [14] | Gu M, Zheng XY. Osteopontin and vasculogenic mimicry formation are associated with response to neoadjuvant chemotherapy in advanced breast cancer[J]. OncoTargets Ther, 2017, 10: 4121-7. doi:10.2147/ott.s129414 |
| [15] | Göthlin Eremo A, Lagergren K, Othman L, et al. Evaluation of SPP1/osteopontin expression as predictor of recurrence in tamoxifen treated breast cancer[J]. Sci Rep, 2020, 10(1): 1451. doi:10.1038/s41598-020-58323-w |
| [16] | Qian J, LeSavage BL, Hubka KM, et al. Cancer-associated mesothelial cells promote ovarian cancer chemoresistance through paracrine osteopontin signaling[J]. J Clin Invest, 2021, 131(16): e146186. doi:10.1172/jci146186 |
| [17] | Sanchis P, Sabater A, Lechuga J, et al. PKA-driven SPP1 activation as a novel mechanism connecting the bone microenvironment to prostate cancer progression[J]. Oncogene, 2025, 44(38): 3568-79. doi:10.1038/s41388-025-03511-z |
| [18] | Fu YL, Zhang Y, Lei Z, et al. Abnormally activated OPN/integrin αVβ3/FAK signalling is responsible for EGFR-TKI resistance in EGFR mutant non-small-cell lung cancer[J]. J Hematol Oncol, 2020, 13(1): 169. doi:10.1186/s13045-020-01009-7 |
| [19] | Gkouveris I, Nikitakis NG, Aseervatham J, et al. The tumorigenic role of DSPP and its potential regulation of the unfolded protein response and ER stress in oral cancer cells[J]. Int J Oncol, 2018, 53(4): 1743-51. doi:10.3892/ijo.2018.4484 |
| [20] | Faraji SN, Nejatollahi F, Tamaddon AM, et al. Generation and characterization of a specific single-chain antibody against DSPP as a prostate cancer biomarker: Involvement of bioinformatics-based design of novel epitopes[J]. Int Immunopharmacol, 2019, 69: 217-24. doi:10.1016/j.intimp.2019.01.016 |
| [21] | Chaplet M, Waltregny D, Detry C, et al. Expression of dentin sialophosphoprotein in human prostate cancer and its correlation with tumor aggressiveness[J]. Int J Cancer, 2006, 118(4): 850-6. doi:10.1002/ijc.21442 |
| [22] | Mascha EJ, Vetter TR. Significance, errors, power, and sample size: the blocking and tackling of statistics: erratum[J]. Anesth Analg, 2018, 126(4): 1429. doi:10.1213/ane.0000000000002887 |
| [23] | Zhong Y, Long T, Gu CS, et al. MYH9-dependent polarization of ATG9B promotes colorectal cancer metastasis by accelerating focal adhesion assembly[J]. Cell Death Differ, 2021, 28(12): 3251-69. doi:10.1038/s41418-021-00813-z |
| [24] | Liu CQ, Pan ZH, Chen Q, et al. Pharmacological targeting PTK6 inhibits the JAK2/STAT3 sustained stemness and reverses chemoresistance of colorectal cancer[J]. J Exp Clin Cancer Res, 2021, 40(1): 297. doi:10.1186/s13046-021-02059-6 |
| [25] | Lieu C, Kennedy EB, Bergsland E, et al. Duration of oxaliplatin-containing adjuvant therapy for stage III colon cancer: ASCO clinical practice guideline[J]. J Clin Oncol, 2019, 37(16): 1436-47. doi:10.1200/jco.19.00281 |
| [26] | Kee JX, Yau JNN, Kumar Muthuramalingam RP, et al. Colorectal cancer at the crossroads: the good, the bad, and the future of platinum-based drugs[J]. Chem Rev, 2025, 125(21): 10248-341. doi:10.1021/acs.chemrev.5c00041 |
| [27] | Liu XW, Zhang SM, An Y, et al. USP10/XAB2/ANXA2 axis promotes DNA damage repair to enhance chemoresistance to oxaliplatin in colorectal cancer[J]. J Exp Clin Cancer Res, 2025, 44(1): 94. doi:10.1186/s13046-025-03357-z |
| [28] | Ding DB, Li WB, Ren JN, et al. SPP1(high) macrophage-induced T-cell stress promotes colon cancer liver metastasis through SPP1/CD44/PI3K/AKT signaling[J]. J Immunother Cancer, 2025, 13(10): e012330. doi:10.1136/jitc-2025-012330 |
| [29] | Zhang H, Guo M, Chen JH, et al. Osteopontin knockdown inhibits αv, β3 integrin-induced cell migration and invasion and promotes apoptosis of breast cancer cells by inducing autophagy and inactivating the PI3K/Akt/mTOR pathway[J]. Cell Physiol Biochem, 2014, 33(4): 991-1002. doi:10.1159/000358670 |
| [30] | Hidalgo M, Bruckheimer E, Rajeshkumar NV, et al. A pilot clinical study of treatment guided by personalized tumorgrafts in patients with advanced cancer[J]. Mol Cancer Ther, 2011, 10(8): 1311-6. doi:10.1158/1535-7163.mct-11-0233 |
| [31] | Aparicio S, Hidalgo M, Kung AL. Examining the utility of patient-derived xenograft mouse models[J]. Nat Rev Cancer, 2015, 15(5): 311-6. doi:10.1038/nrc3944 |
| [32] | Liu GK, Fan XY, Tang M, et al. Osteopontin induces autophagy to promote chemo-resistance in human hepatocellular carcinoma cells[J]. Cancer Lett, 2016, 383(2): 171-82. doi:10.1016/j.canlet.2016.09.033 |
| [33] | Ding K, Fan L, Chen SJ, et al. Overexpression of osteopontin promotes resistance to cisplatin treatment in HCC[J]. Oncol Rep, 2015, 34(6): 3297-303. doi:10.3892/or.2015.4306 |
| [34] | Di YQ, Zhang X, Wen XQ, et al. MAPK signaling-mediated RFNG phosphorylation and nuclear translocation restrain oxaliplatin-induced apoptosis and ferroptosis[J]. Adv Sci, 2024, 11(38): 2402795. doi:10.1002/advs.202402795 |
| [35] | Lin JF, Liu ZX, Chen DL, et al. Nucleus-translocated GCLM promotes chemoresistance in colorectal cancer through a moonlighting function[J]. Nat Commun, 2025, 16(1): 263. doi:10.1038/s41467-024-55568-1 |
| [1] | 翁诺舟, 谭彬, 曾文涛, 古家宇, 翁炼基, 郑克鸿. 过表达RGL1通过激活CDC42/RAC1复合体上调运动型黏着斑组装促进结直肠癌转移[J]. 南方医科大学学报, 2025, 45(5): 1031-1038. |
| [2] | 马振南, 刘福全, 赵雪峰, 张晓微. DTX2促进奥沙利铂耐药的结直肠癌细胞增殖、侵袭和上皮间质转化[J]. 南方医科大学学报, 2025, 45(4): 829-836. |
| [3] | 张夏玮, 杨晶晶, 温亚男, 刘青阳, 窦立萍, 高春记. METTL3介导的m6A修饰通过调节自噬促进急性髓性白血病细胞中FOXO3表达及蒽环类药物耐药性[J]. 南方医科大学学报, 2025, 45(3): 470-478. |
| [4] | 庆顺杰, 沈智勇. 过表达己糖激酶2通过激活JAK/STAT途径促进结直肠癌细胞的增殖、迁移和侵袭并调节肿瘤免疫微环境[J]. 南方医科大学学报, 2025, 45(3): 542-553. |
| [5] | 喻珍妮, 高竟哲, 孙惠, 冯芹, 那效旗, 张宁, 沈昆双, 王媛媛, 王喜军. 肠道菌群、T细胞在结直肠癌发病中的因果关联:孟德尔随机化分析[J]. 南方医科大学学报, 2025, 45(12): 2756-2766. |
| [6] | 杜越, 张秀森, 周克旭, 金星, 原翔, 高社干. RgpB通过抑制自噬小体与溶酶体融合避免Cx43降解参与食管癌化疗耐药[J]. 南方医科大学学报, 2024, 44(9): 1670-1676. |
| [7] | 纪凯, 于冠宇, 周乐其, 张天帅, 凌潜龙, 满文江, 朱冰, 张卫. HNRNPA1基因在结直肠癌组织中高表达及其潜在的诊断和治疗价值[J]. 南方医科大学学报, 2024, 44(9): 1685-1695. |
| [8] | 张银亮, 骆泽谭, 赵睿, 赵娜, 徐志东, 奥迪, 丛古一, 刘新宇, 郑海伦. 血根碱通过调控STUB1/GPX4诱导直肠癌细胞发生铁死亡[J]. 南方医科大学学报, 2024, 44(8): 1537-1544. |
| [9] | 郗雪艳, 邓婷, 杜伯雨. 结直肠成纤维细胞通过激活ERK信号通路促进结直肠癌细胞的恶性生物学行为[J]. 南方医科大学学报, 2024, 44(10): 1866-1873. |
| [10] | 颜 畅, 刘 爽, 宋庆志, 胡艺冰. 二甲双胍通过抑制线粒体氧化磷酸化降低结直肠癌干细胞的自我更新能力[J]. 南方医科大学学报, 2023, 43(8): 1279-1286. |
| [11] | 魏 可, 石纪雯, 肖雨寒, 王文锐, 杨清玲, 陈昌杰. miR-30e-5p过表达促进结直肠癌细胞的增殖和迁移:基于下调PTEN激活CXCL12轴[J]. 南方医科大学学报, 2023, 43(7): 1081-1092. |
| [12] | 张雪芳, 陈延华, 李宗恒, 尚 靖, 袁泽婷, 邓皖利, 骆 莺, 韩 娜, 殷佩浩, 殷 军. 六神丸治疗小鼠结肠炎相关性结直肠癌的作用机制:基于网络药理学和体内验证方法[J]. 南方医科大学学报, 2023, 43(7): 1051-1062. |
| [13] | 邓 婷, 杜伯雨, 郗雪艳. 结直肠癌细胞通过激活成纤维细胞的ERK通路诱导癌症相关成纤维细胞的形成[J]. 南方医科大学学报, 2023, 43(6): 943-951. |
| [14] | 马振南, 赵雪峰, 张晓微, 许广大, 刘福全. DTX2通过Notch2/Akt轴促进结直肠癌细胞的迁移和侵袭[J]. 南方医科大学学报, 2023, 43(3): 340-348. |
| [15] | 王旋成, 朱一帆, 周海琳, 黄宗声, 陈鸿炜, 张嘉豪, 杨珊伊, 陈广辉, 张淇淞. 血清非靶向代谢组学联合靶向胆汁酸代谢组学筛查结直肠癌的潜在生物标志物[J]. 南方医科大学学报, 2023, 43(3): 443-453. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||