南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 907-922.doi: 10.12122/j.issn.1673-4254.2026.04.19
• • 上一篇
马扬1( ), 郭龙辉1(
), 郭龙辉1( ), 王锦帼2, 肖文萍1, 吕运成3(
), 王锦帼2, 肖文萍1, 吕运成3( ), 范晓明4,5(
), 范晓明4,5( )
)
收稿日期:2025-09-14
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
吕运成,范晓明
E-mail:mayang@stu.glmc.edu.cn;guolonghui@stu.glmu.edu.cn;anthony0723@163.com;fanxiaom1987@glmc.edu.cn
作者简介:马 扬,在读硕士研究生,E-mail: mayang@stu.glmc.edu.cn基金资助:
Yang MA1( ), Longhui GUO1(
), Longhui GUO1( ), Jinguo WANG2, Wenpin XIAO1, Yuncheng LV3(
), Jinguo WANG2, Wenpin XIAO1, Yuncheng LV3( ), Xiaoming FAN4,5(
), Xiaoming FAN4,5( )
)
Received:2025-09-14
Online:2026-04-20
Published:2026-04-24
Contact:
Yuncheng LV, Xiaoming FAN
E-mail:mayang@stu.glmc.edu.cn;guolonghui@stu.glmu.edu.cn;anthony0723@163.com;fanxiaom1987@glmc.edu.cn
摘要:
目的 通过生物信息学、网络药理学及动物实验,探讨灯盏乙素(Scu)治疗代谢相关脂肪性肝病(MASLD)的靶点及机制。 方法 从GEO数据库GSE89632鉴定MASLD差异表达基因(DEGs)。利用PharmMapper预测Scu靶点,Cytoscape构建药效团-靶点网络。R语言的clusterProfiler包进行GO和KEGG通路分析。STRING工具构建PPI网络,cytoHubba和MCODE确定关键治疗(KT)基因。进行分子对接及分子动力学模拟验证。将SD大鼠随机分为对照组、模型组、Scu低/中/高剂量(50、100、200 mg·kg-1·d-1)组及辛伐他汀(5 mg·kg-1·d-1)组(n=10)。12周高脂高糖饮食造模后,给药组连续灌胃8周。 结果 共鉴定出810个DEGs 和12个PT基因。GO和KEGG分析指向炎症调控、细胞因子应答、纤维化和代谢相关通路。分子对接和动力学模拟验证了MMP7和LCN2为关键靶点。动物实验显示,Scu剂量依赖改善MASLD大鼠胰岛素抵抗、肝功能,脂质及炎症相关因子,以及肝脏病理改变,高剂量Scu效果优于辛伐他汀(P<0.05)。q-PCR及Western blotting检测发现Scu以剂量依赖的方式上调MMP7的表达,下调LCN2的表达(P<0.05)。 结论 Scu可能通过调控MMP7和LCN2发挥抗MASLD的作用。
马扬, 郭龙辉, 王锦帼, 肖文萍, 吕运成, 范晓明. 灯盏乙素通过调控MMP7与LCN2改善大鼠代谢功能障碍相关脂肪性肝病[J]. 南方医科大学学报, 2026, 46(4): 907-922.
Yang MA, Longhui GUO, Jinguo WANG, Wenpin XIAO, Yuncheng LV, Xiaoming FAN. Scutellarin improves metabolic dysfunction-associated steatotic liver disease in rats by regulating MMP7 and LCN2[J]. Journal of Southern Medical University, 2026, 46(4): 907-922.
| Gene name | Primer name | Primer sequence |
|---|---|---|
| MMP7 | MMP7-F | F: CTCTCTGGGTCTGGGTCACT |
| MMP7-R | R: AAGGGCGTTTGCTCATTCCA | |
| LCN2 | LCN2-F | F: GCTGTCGCTACTGGATCAGA |
| LCN2-R | R: TCGCTCCTTCAGTTCATCGG | |
| GAPDH | GAPDH-F | F: GACATGCCGCCTGGAGAAAC |
| GAPDH-R | R: AGCCCAGGATGCCCTTTAGT |
表1 引物序列
Tab.1 Primer sequence
| Gene name | Primer name | Primer sequence |
|---|---|---|
| MMP7 | MMP7-F | F: CTCTCTGGGTCTGGGTCACT |
| MMP7-R | R: AAGGGCGTTTGCTCATTCCA | |
| LCN2 | LCN2-F | F: GCTGTCGCTACTGGATCAGA |
| LCN2-R | R: TCGCTCCTTCAGTTCATCGG | |
| GAPDH | GAPDH-F | F: GACATGCCGCCTGGAGAAAC |
| GAPDH-R | R: AGCCCAGGATGCCCTTTAGT |
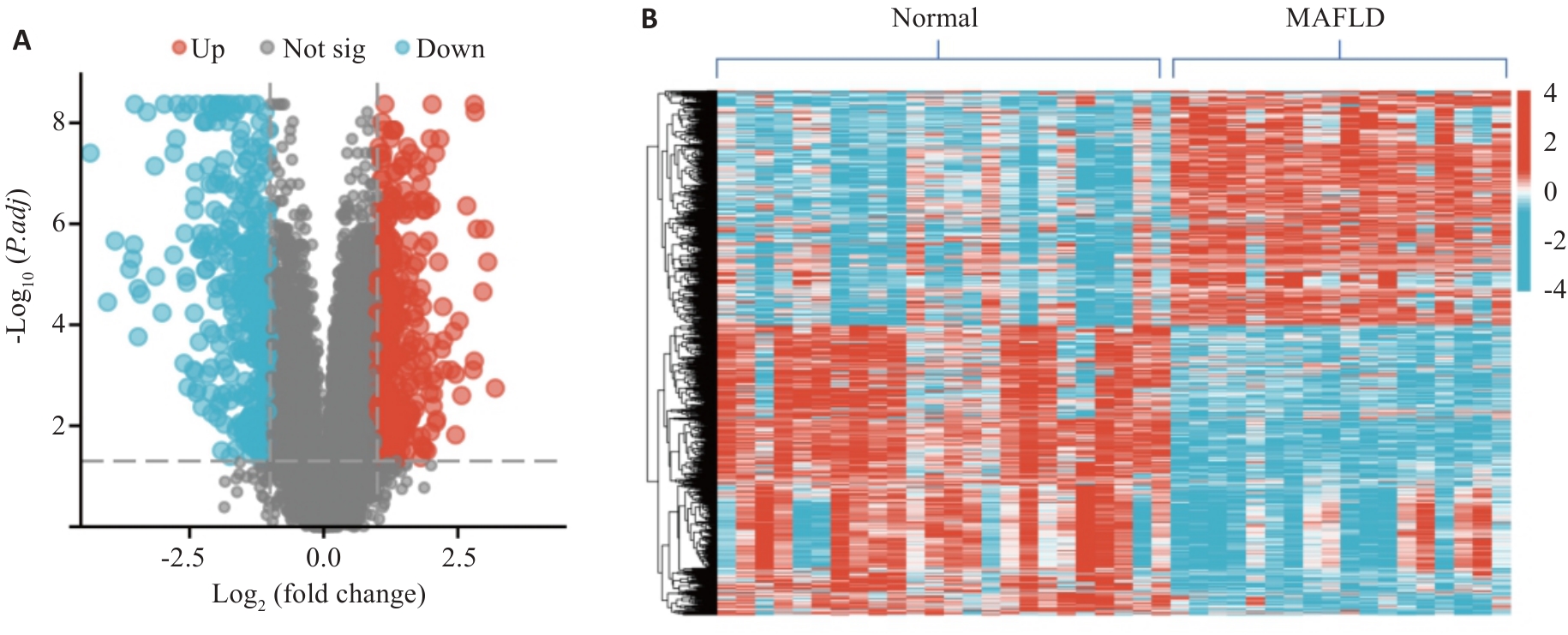
图1 GSE89632数据集差异基因的火山图和热图
Fig.1 Volcano map and heat map of the differentially expressed genes (DEGs) in the GSE89632 dataset. A: Volcano map of the detected genes. Each point represents a gene. Blue dots represent down-regulated genes, and red dots represent up-regulated genes. The screening criteria for important genes are: |Log2FC|>1 and P.adj<0.05. B: Heat map of DEGs clustering. Blue indicates low expression and red indicates high expression.
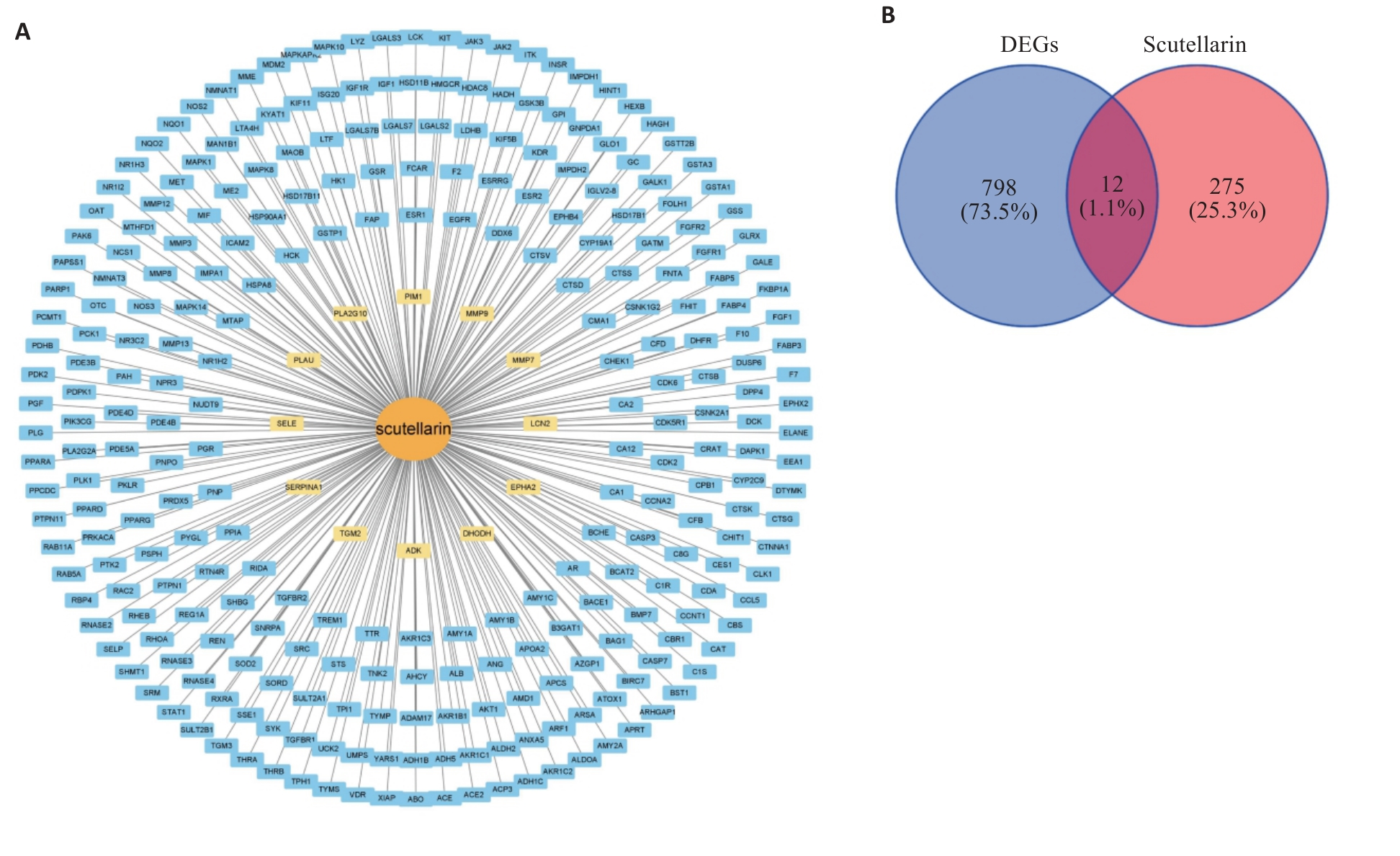
图2 Scu的药效团靶网络
Fig.2 Pharmacophore target network of scutellarin. A: Drug-target network. Scutellarin is located at the center of the network. The potential therapeutic gene (PT gene) for treating MASLD is marked yellow. B: Intersecting DEGs obtained from GSE89632 as the target genes of scutellarin are considered to be the PT genes.
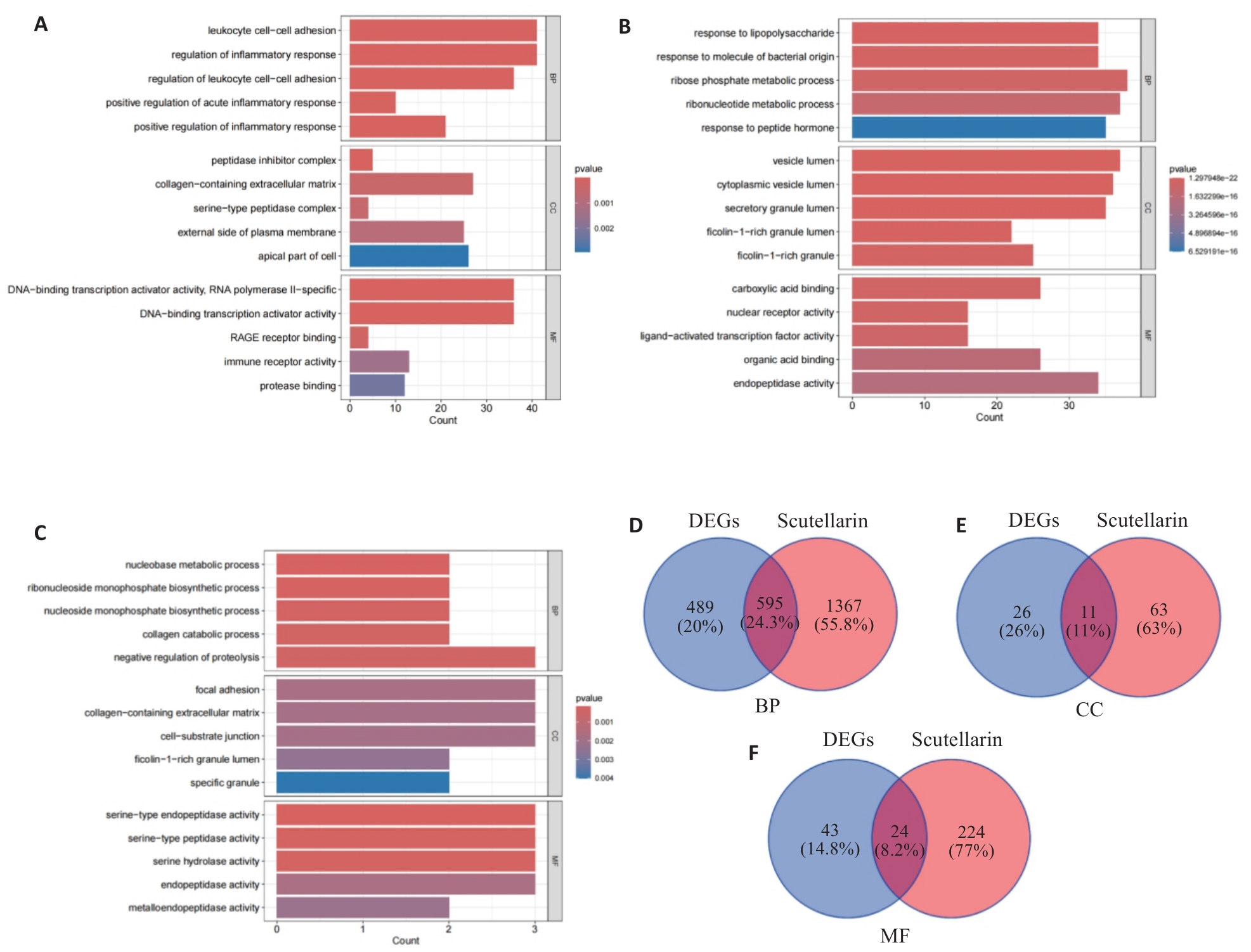
图3 GO功能富集分析
Fig.3 GO functional enrichment analysis. The top 5 are cellular components (CC), biological processes (BP), and molecular functions (MF). A: Rich in MASLD-related DEGs. B: Target gene of S-CU. C: Potential therapeutic genes (PT genes). D: Venn diagrams of GO for DEG and scutellarin (cell component). E: Venn diagrams of GO for DEG and scutellarin (biological process). F: Venn diagrams of GO for DEG and scutellarin (molecular function).
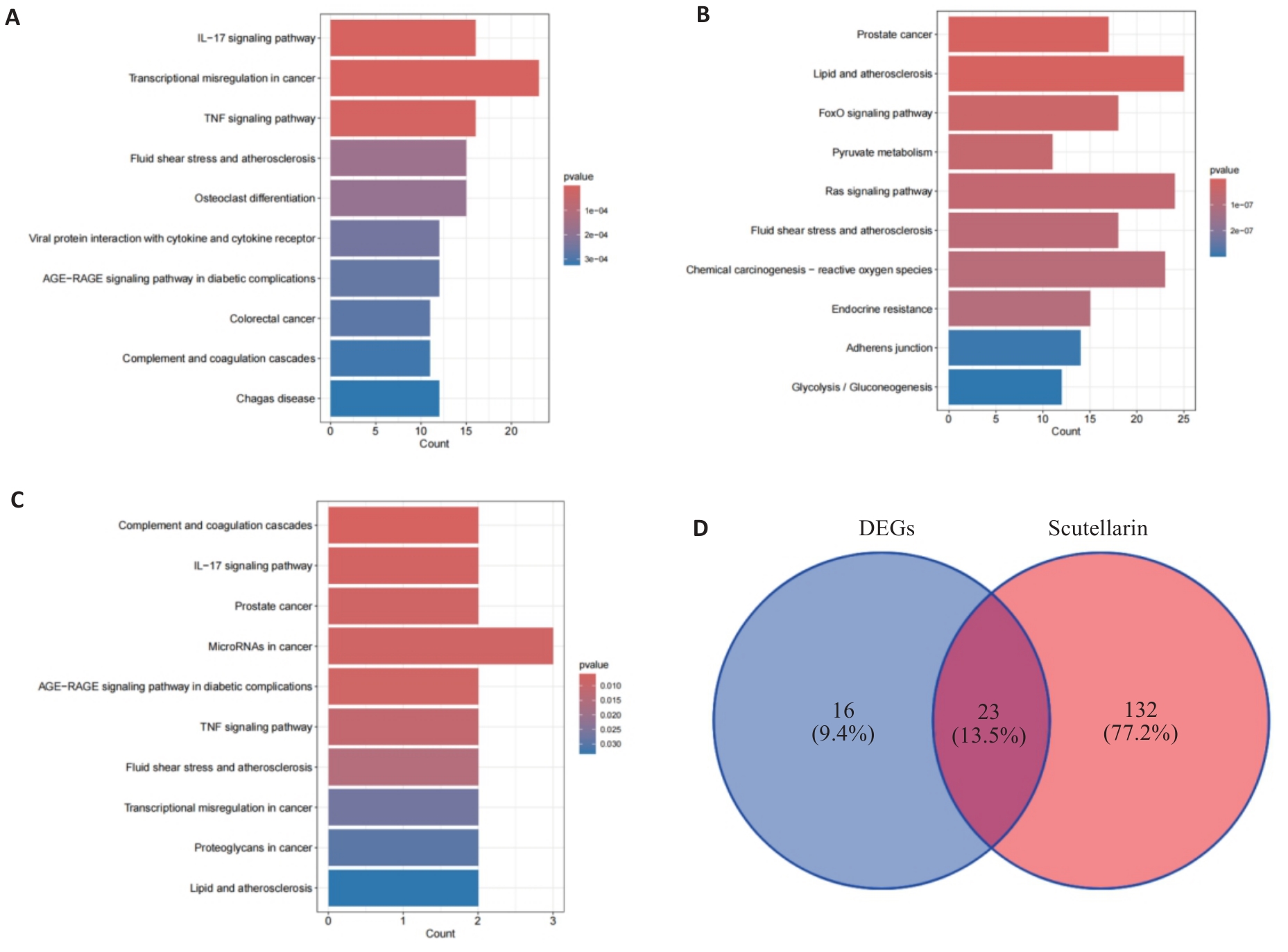
图4 KEGG通路富集分析前10名KEGG
Fig.4 Top 10 KEGGs in KEGG pathway enrichment analysis. A: MASLD-related DEGs. B: Target genes of scutellarin. C: Potential therapeutic genes (PT genes). D: Venn diagrams of the KEGG pathways in DEG and scutellarin.
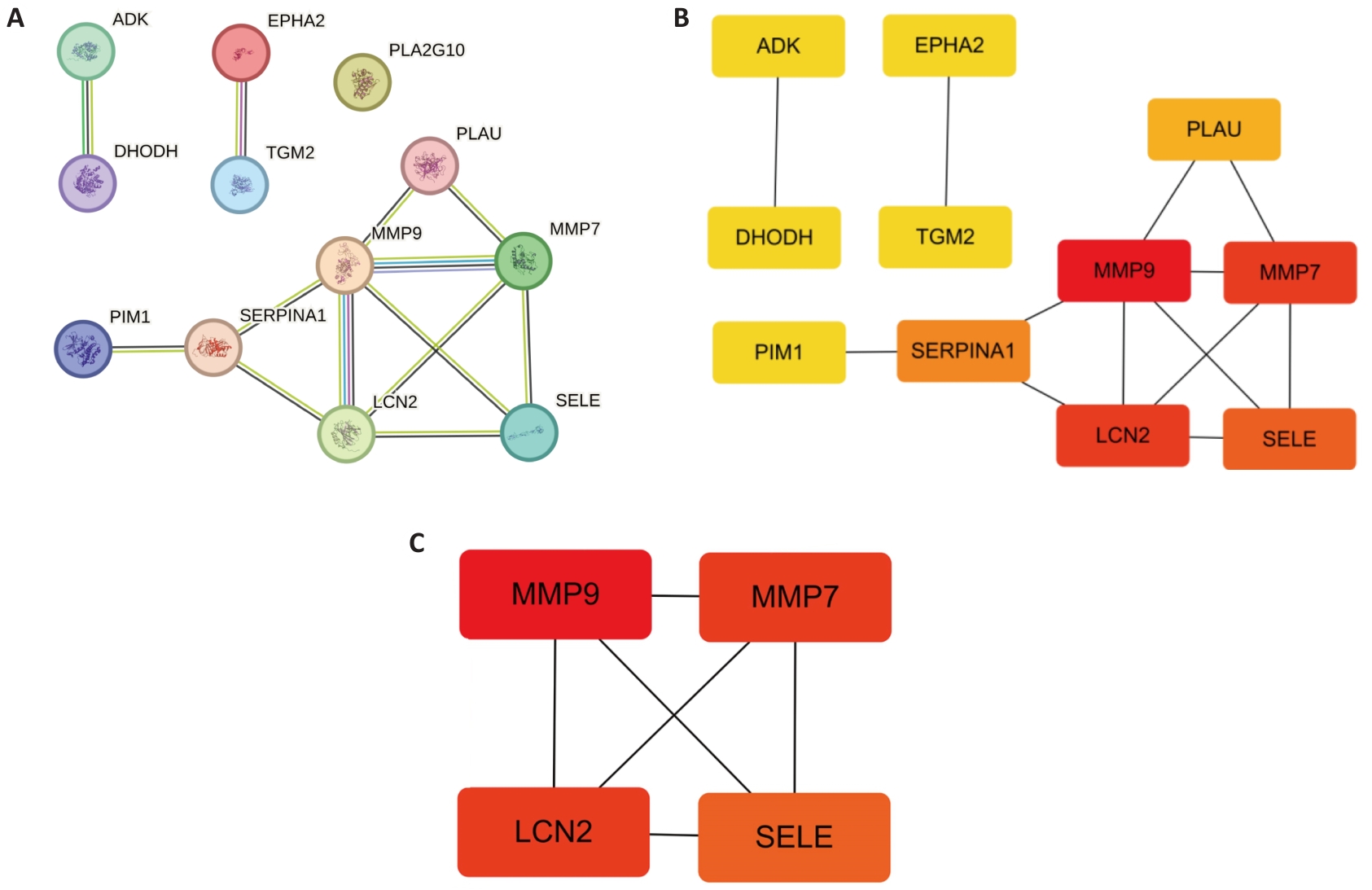
图5 关键治疗基因(KT基因)筛选
Fig.5 Protein-protein interaction diagram of the key therapeutic gene screening. A: PT gene. B: The top 11 key genes calculated by cytoHubba. C: Analysis of the key module genes using MCODE.
| Target | Drug | Binding energy (kcal/mol) | Hydrogen bonds |
|---|---|---|---|
| MMP7 | Scu | -8.3 | 6 |
| MMP9 | Scu | -8.8 | 6 |
| LCN2 | Scu | -8.2 | 2 |
| SELE | Scu | -7.1 | 6 |
表2 Scu与治疗靶点结合能量值
Tab.2 Binding energy value of scutellarin (Scu) to the therapeutic targets
| Target | Drug | Binding energy (kcal/mol) | Hydrogen bonds |
|---|---|---|---|
| MMP7 | Scu | -8.3 | 6 |
| MMP9 | Scu | -8.8 | 6 |
| LCN2 | Scu | -8.2 | 2 |
| SELE | Scu | -7.1 | 6 |
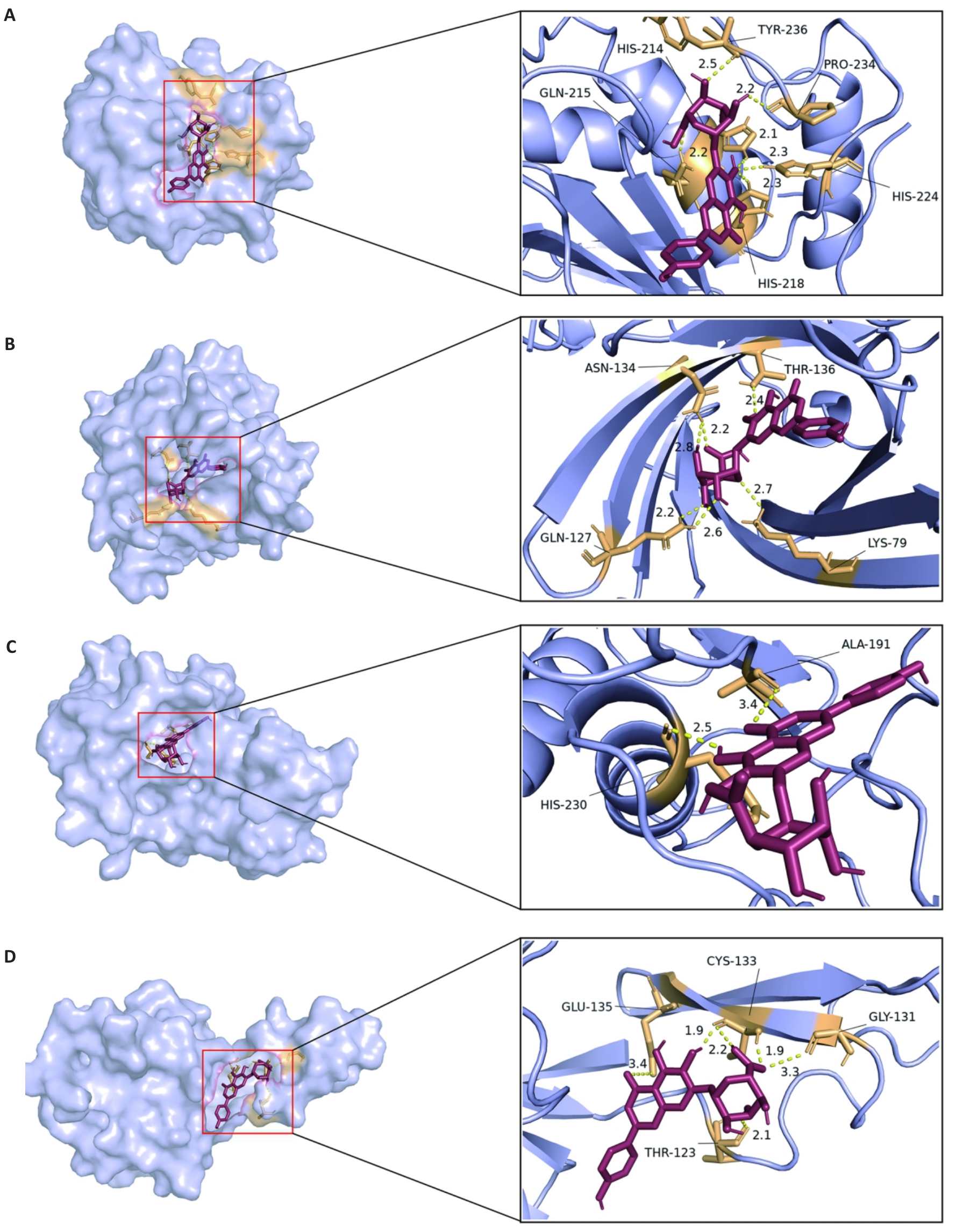
图6 Scu与关键治疗基因(KT基因)蛋白质受体的分子对接
Fig.6 Molecular docking of scutellarin with the protein receptor of the key therapeutic genes. A: Molecular docking display diagram of MMP7. B: Molecular docking display diagram of MMP9. C: Molecular docking display diagram of LCN2. D: Molecular docking display diagram of SELE.
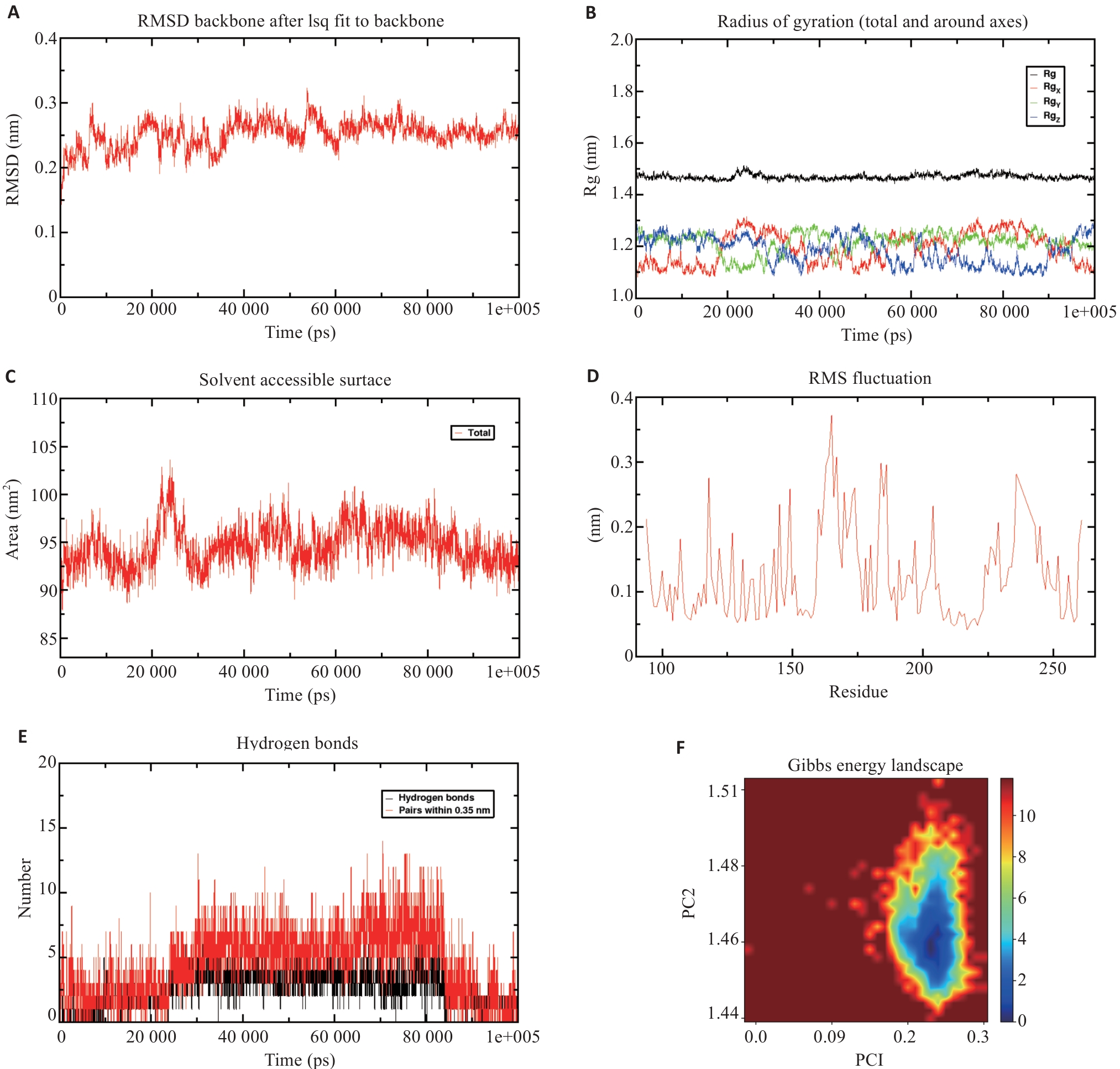
图7 Scu与MMP7的分子动力学模拟
Fig.7 Molecular dynamics simulation of scutellarin and MMP7. A: Root mean square deviation (RMSD) of the protein backbone over time, indicating stabilization after approximately 10 ns. B: Radius of gyration analysis of the protein complex, which stabilizes at 1.46 nm, suggesting a stable complex structure. C: Solvent accessible surface area (SASA) analysis of the complex, remaining stable at approximately 92.5 nm². D: Residue fluctuation analysis of MMP7, showing higher flexibility around residues 165 and 167. E: Hydrogen bond analysis between scutellarin and MMP7, showing an average number of 4.469 hydrogen bonds. F: Free energy landscape.
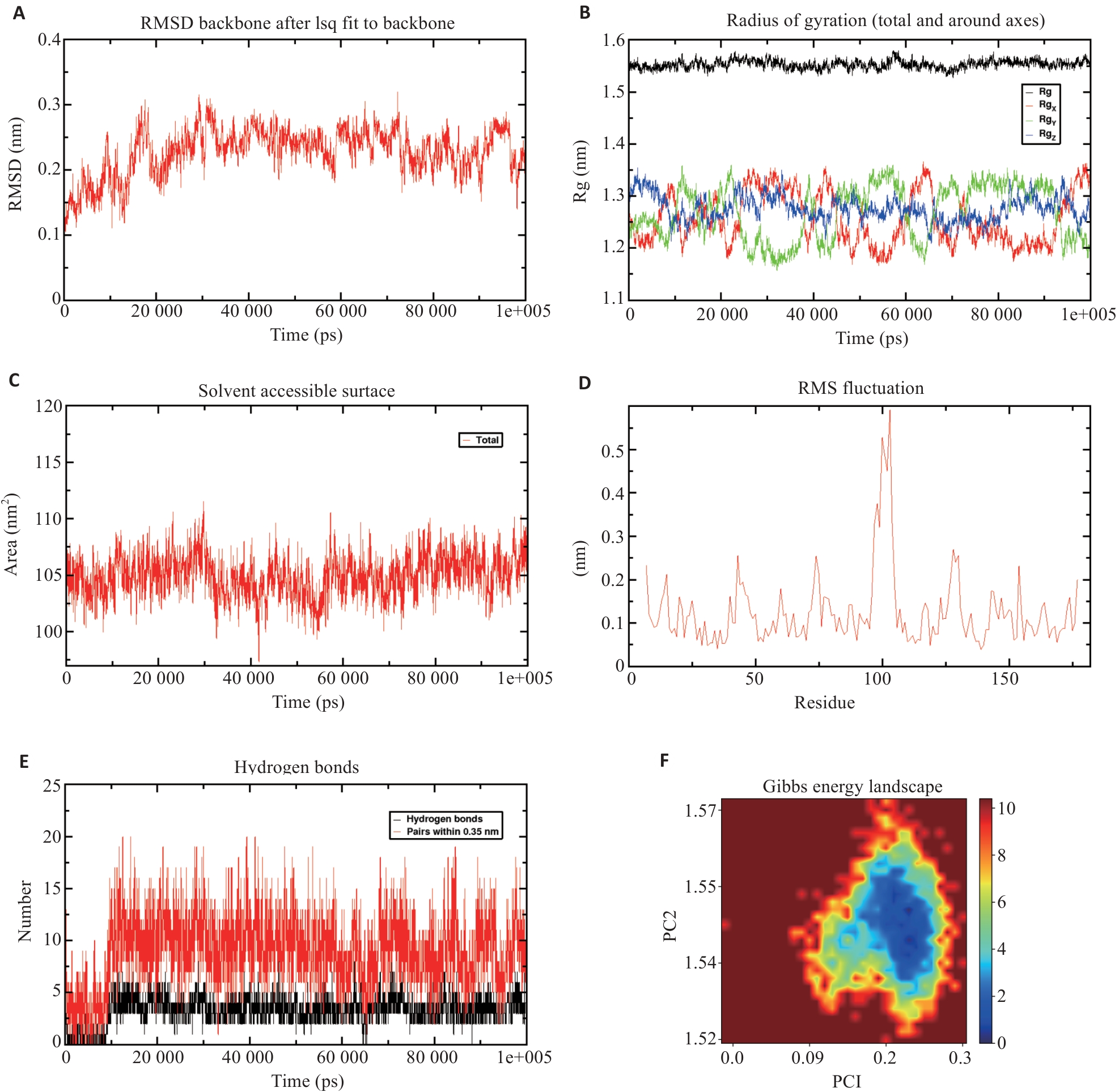
图8 Scu与LCN2的分子动力学模拟
Fig.8 Molecular dynamics simulation of scutellarin and LCN2. A: RMSD of the protein backbone over time, indicating stabilization after approximately 20 ns. B: Radius of gyration analysis of the protein complex, which stabilizes at 1.54 nm, suggesting a stable complex structure. C: SASA analysis of the complex, remaining stable at approximately 107 nm². D: Residue fluctuation analysis of LCN2, showing higher flexibility around residues 100 and 103. E: Hydrogen bond analysis between scutellarin and LCN2, showing an average number of 8.989 hydrogen bonds. F: Free energy landscape.
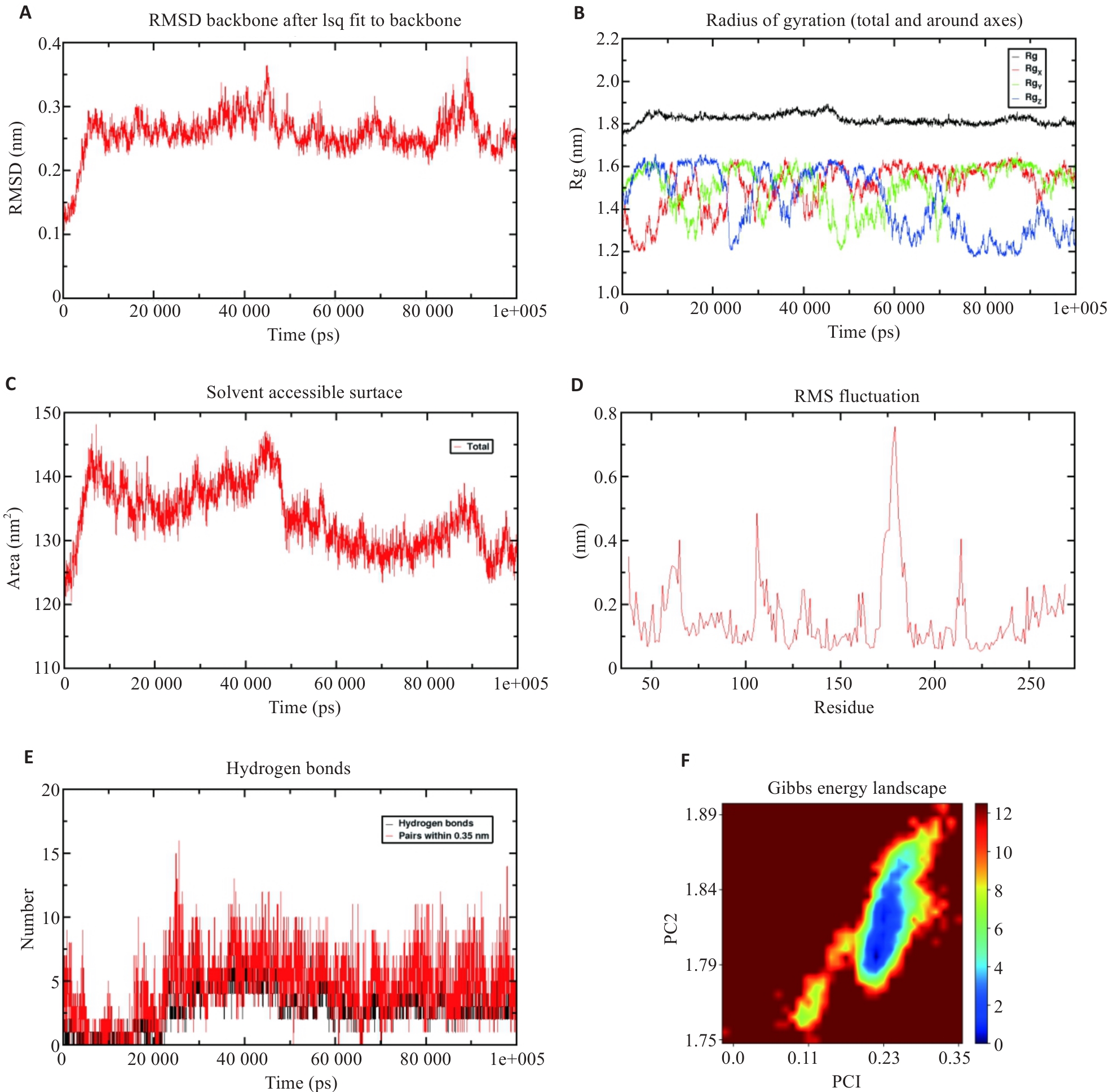
图9 Scu与MMP9的分子动力学模拟
Fig.9 Molecular dynamics simulation of scutellarin and MMP9. A: RMSD of the protein backbone over time, indicating stabilization after approximately 7 ns. B: Radius of gyration analysis of the protein complex, which stabilizes at 1.8 nm, suggesting a stable complex structure. C: SASA analysis of the complex, remaining stable at approximately 128 nm². D: Residue fluctuation analysis of MMP9, showing higher flexibility around residue 179. E: Hydrogen bond analysis between scutellarin and MMP9, with an average number of 4.48 hydrogen bonds. F: Free energy landscape.
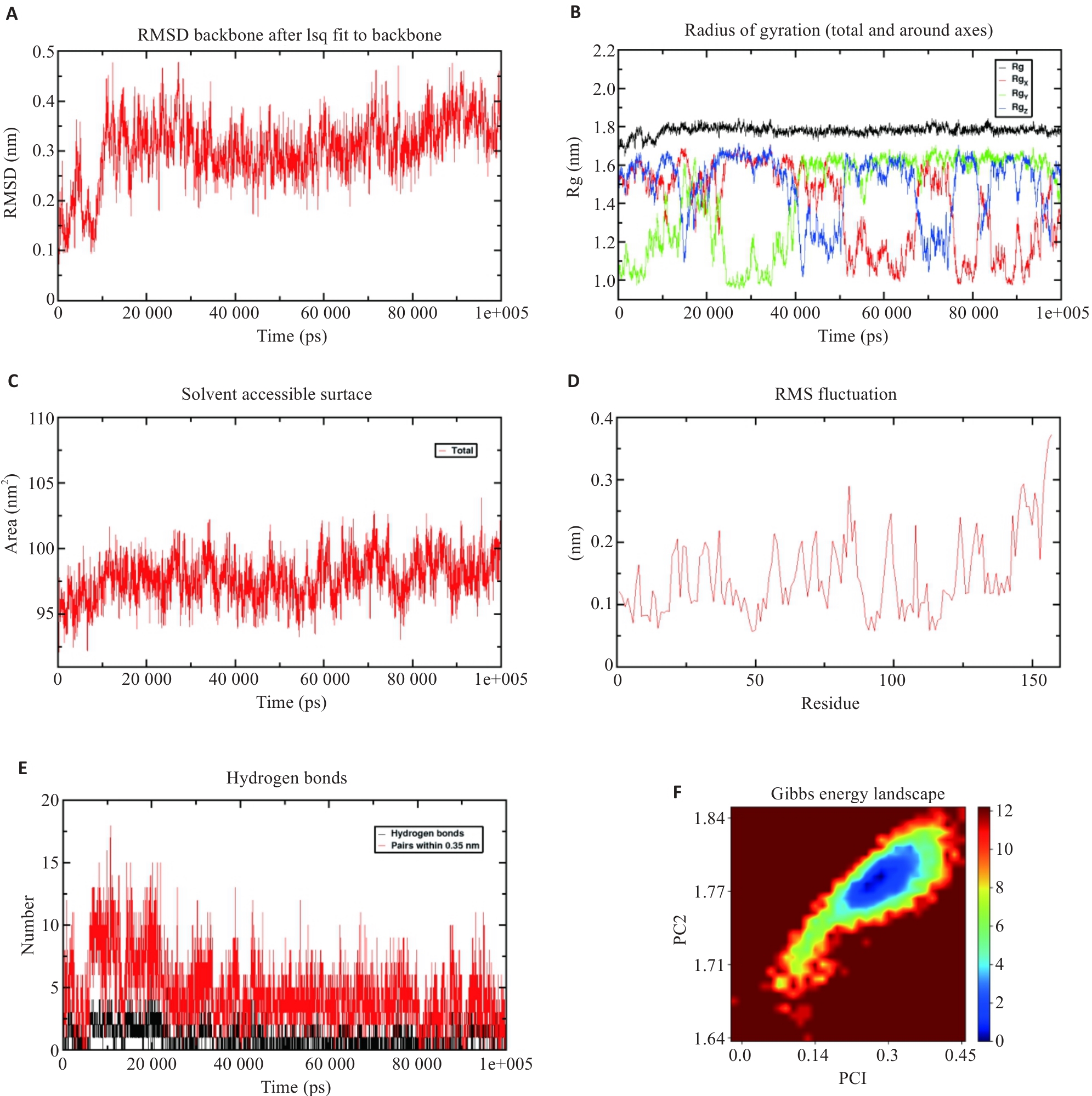
图10 Scu与SELE的分子动力学模拟
Fig.10 Molecular dynamics simulation of scutellarin and SELE. A: RMSD of the protein backbone over time, indicating stabilization after approximately 7 ns. B: Radius of gyration analysis of the protein complex, which stabilizes at 1.8 nm, suggesting a stable complex structure. C: SASA analysis of the complex, remaining stable at approximately 98 nm². D: Residue fluctuation analysis of SELE, showing higher flexibility around residue 84. E: Hydrogen bond analysis between scutellarin and SELE, showing an average number of 4.46 hydrogen bonds. F: Free energy landscape.
| Control | 0 | 0 | 0 | 0 | Normal |
| Model | 3 | 2-3 | 2 | 7 | Marked NASH-like alterations |
| LD Scu | 2 | 2 | 1 | 5 | Moderate improvement |
| MD Scu | 1 | 1 | 1 | 3 | Significant improvement |
| HD Scu | 0-1 | 0-1 | 0 | 1-2 | Nearly normal |
| SIM | 0-1 | 0-1 | 0 | 1-2 | Trend toward normalization |
表3 大鼠肝组织NAS评分
Tab.3 NAS scores of rat liver tissues
| Control | 0 | 0 | 0 | 0 | Normal |
| Model | 3 | 2-3 | 2 | 7 | Marked NASH-like alterations |
| LD Scu | 2 | 2 | 1 | 5 | Moderate improvement |
| MD Scu | 1 | 1 | 1 | 3 | Significant improvement |
| HD Scu | 0-1 | 0-1 | 0 | 1-2 | Nearly normal |
| SIM | 0-1 | 0-1 | 0 | 1-2 | Trend toward normalization |
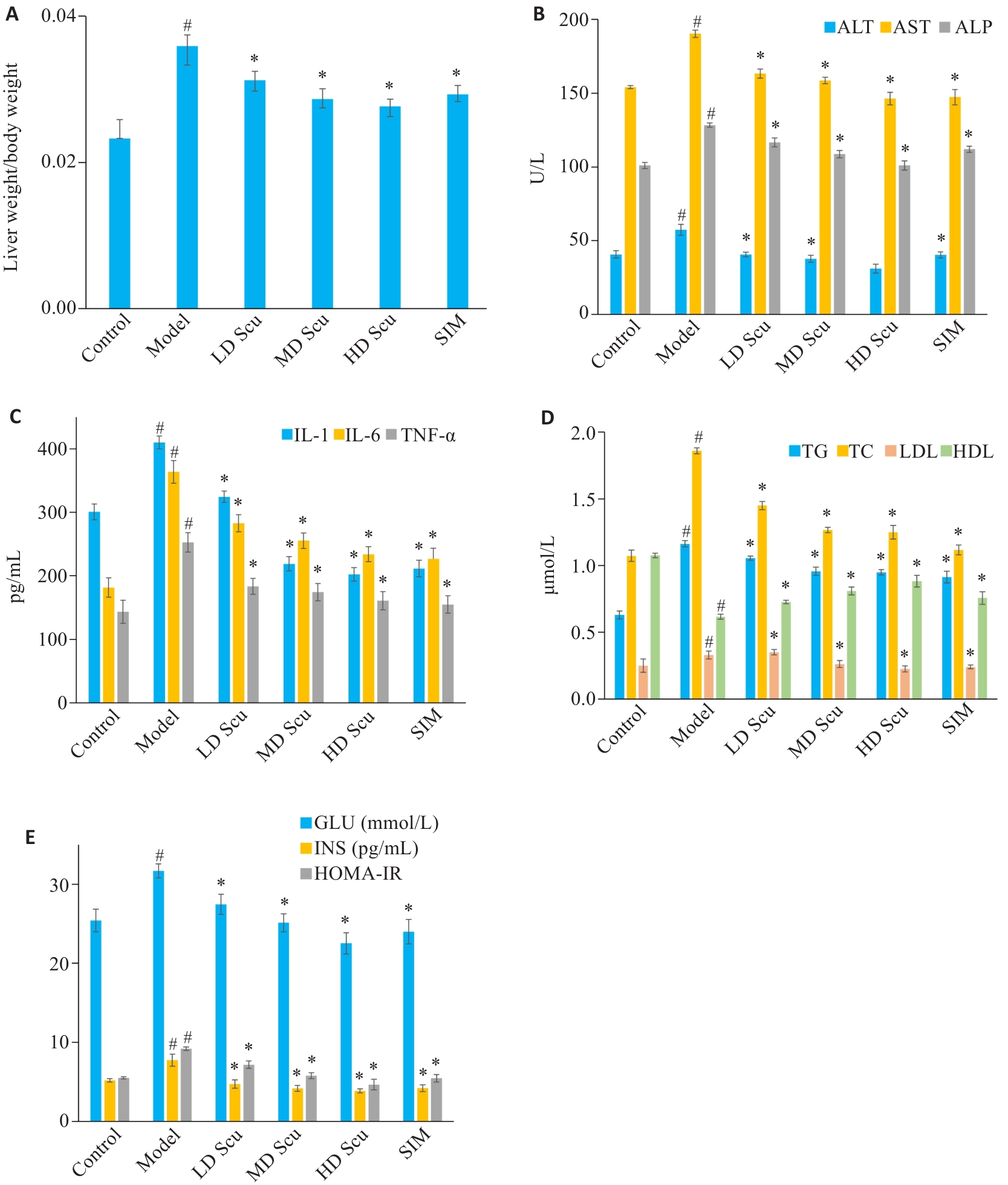
图11 Scu对MASLD大鼠生化指标的影响
Fig.11 Effects of scutellarin on biochemical parameters in MASLD rats. A: Changes in liver-to-body weight ratio across different treatment groups. B: Effects of different scutellarin doses on liver function markers (ALT, AST, and ALP). C: Effects of different scutellarin doses on inflammatory cytokines (IL-1β, IL-6, and TNF-α). D: Effects of different scutellarin doses on serum lipid profiles (TG, TC, LDL, and HDL). E: Effects of different scutellarin doses on blood glucose, insulin levels, and insulin resistance. *P<0.05 vs Model. #P<0.05 vs Control.
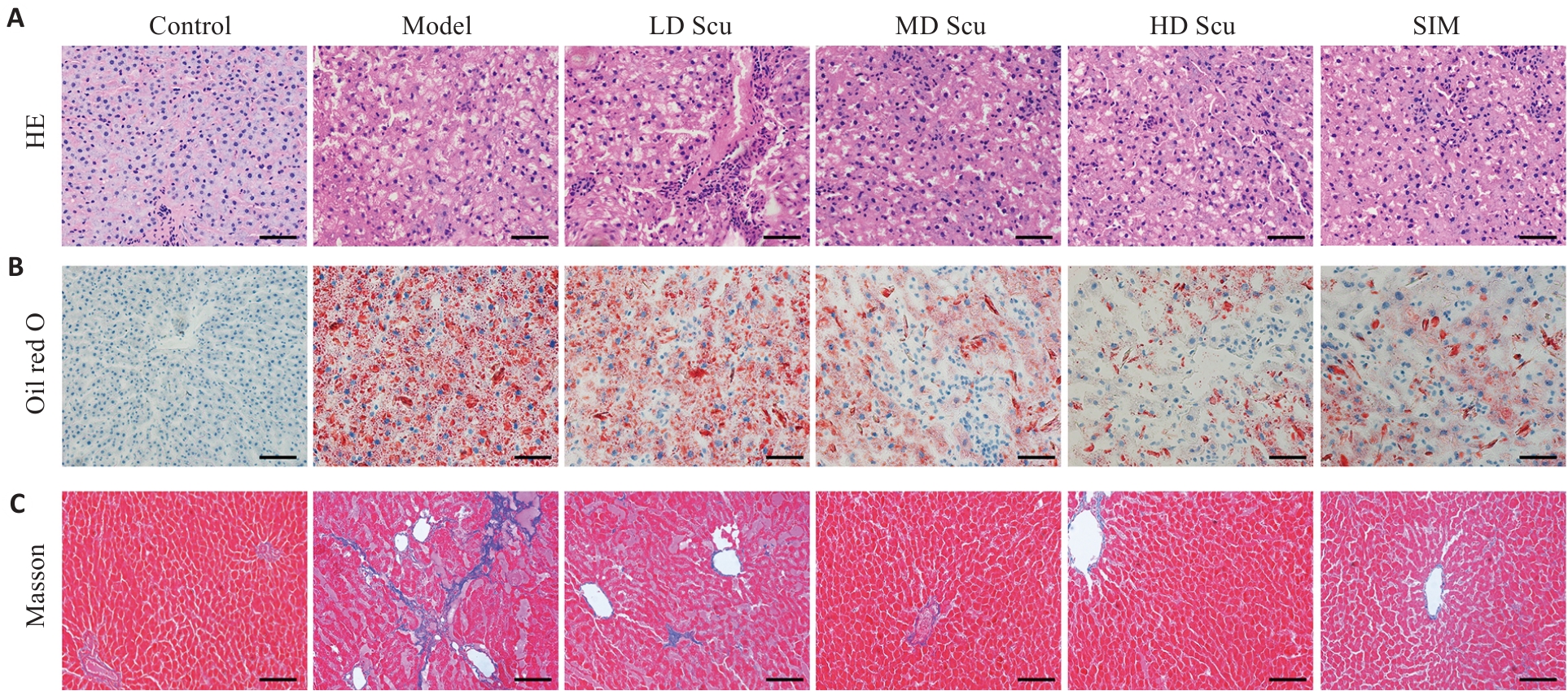
图12 Scu对MASLD大鼠肝组织病理学改变的影响
Fig.12 Effects of scutellarin on histopathological alterations in liver tissues of MASLD rats. A: HE staining of liver sections from different treatment groups, showing a notable increase in fat vacuoles in the MASLD group, which was reduced following scutellarin treatment. B: Oil Red O staining of liver sections from different treatment groups, revealing severe hepatic steatosis in the MASLD group and obvious improvement after scutellarin treatment. C: Masson staining of liver sections from different treatment groups (scale bar=50 μm).
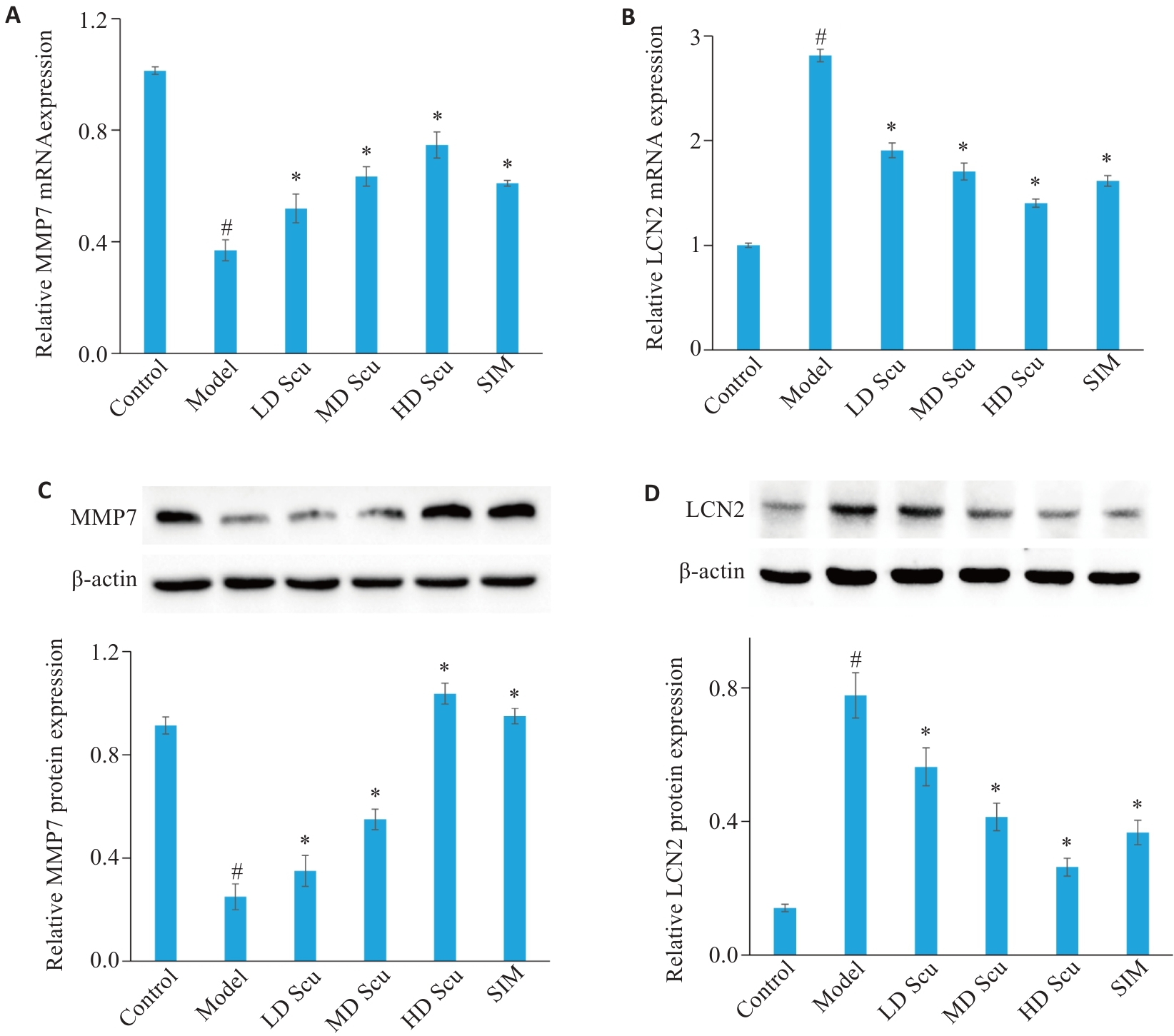
图13 Scu对MASLD大鼠肝组织MMP7和LCN2表达的影响
Fig.13 Effects of scutellarin on MMP7 and LCN2 expression in liver tissues of MASLD rats. A, B: qRT-PCR analysis of MMP7 and LCN2 mRNA expression levels in liver tissues from different treatment groups. C, D: Western blotting analysis of MMP7 and LCN2 protein expression levels in liver tissues from different treatment groups. #P<0.05 vs control group; *P<0.05 vs model group (n=10).
| [1] | Eslam M, Newsome PN, Sarin SK, et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement[J]. J Hepatol, 2020, 73(1): 202-9. doi:10.1016/j.jhep.2020.07.045 |
| [2] | Cotter TG, Rinella M. Nonalcoholic fatty liver disease 2020: the state of the disease[J]. Gastroenterology, 2020, 158(7): 1851-64. doi:10.1053/j.gastro.2020.01.052 |
| [3] | Rong L, Zou JY, Ran W, et al. Advancements in the treatment of non-alcoholic fatty liver disease (NAFLD)[J]. Front Endocrinol, 2023, 13: 1087260. doi:10.3389/fendo.2022.1087260 |
| [4] | Ratziu V, Francque S, Sanyal A. Breakthroughs in therapies for NASH and remaining challenges[J]. J Hepatol, 2022, 76(6): 1263-78. doi:10.1016/j.jhep.2022.04.002 |
| [5] | Wang R, Mao YH, Yu CP, et al. Research progress of natural products with the activity of anti-nonalcoholic steatohepatitis[J]. Mini Rev Med Chem, 2024, 24(21): 1894-929. doi:10.2174/0113895575306598240503054317 |
| [6] | Liu TY, Sun YM, Zhao XY. Research progress on chemical components of Astragalus membranaceus and treatment of metabolic syndrome[J]. Molecules, 2025, 30(18): 3721. doi:10.3390/molecules30183721 |
| [7] | Chen LZ, Pu XM, Li L, et al. Shenling Jianpiwei formula ameliorates metabolic associated fatty liver disease through modulation of the gut microbiota and attenuation of LPS-mediated inflammation[J]. Fitoterapia, 2025, 186: 106862. doi:10.1016/j.fitote.2025.106862 |
| [8] | Guo C, Huang QX, Wang YS, et al. Therapeutic application of natural products: NAD+ metabolism as potential target[J]. Phytomedicine, 2023, 114: 154768. doi:10.1016/j.phymed.2023.154768 |
| [9] | Wen L, He T, Yu AX, et al. Breviscapine: a review on its phytochemistry, pharmacokinetics and therapeutic effects[J]. Am J Chin Med, 2021, 49(6): 1369-97. doi:10.1142/s0192415x21500646 |
| [10] | Yuan T, Yang HY, Li YP, et al. Scutellarin inhibits inflammatory PANoptosis by diminishing mitochondrial ROS generation and blocking PANoptosome formation[J]. Int Immunopharmacol, 2024, 139: 112710. doi:10.1016/j.intimp.2024.112710 |
| [11] | Xie XH, Wang F, Ge WX, et al. Scutellarin attenuates oxidative stress and neuroinflammation in cerebral ischemia/reperfusion injury through PI3K/Akt-mediated Nrf2 signaling pathways[J]. Eur J Pharmacol, 2023, 957: 175979. doi:10.1016/j.ejphar.2023.175979 |
| [12] | Zhou H, Chen X, Chen LZ, et al. Anti-fibrosis effect of scutellarin via inhibition of endothelial-mesenchymal transition on isoprenaline-induced myocardial fibrosis in rats[J]. Molecules, 2014, 19(10): 15611-23. doi:10.3390/molecules191015611 |
| [13] | Mo J, Yang RH, Li F, et al. Scutellarin protects against vascular endothelial dysfunction and prevents atherosclerosis via antioxidation[J]. Phytomedicine, 2018, 42: 66-74. doi:10.1016/j.phymed.2018.03.021 |
| [14] | Zhang XY, Huo ZJ, Luan HL, et al. Scutellarin ameliorates hepatic lipid accumulation by enhancing autophagy and suppressing IRE1α/XBP1 pathway[J]. Phytother Res, 2022, 36(1): 433-47. doi:10.1002/ptr.7344 |
| [15] | Zhang X, Dong ZC, Fan H, et al. Scutellarin prevents acute alcohol-induced liver injury via inhibiting oxidative stress by regulating the Nrf2/HO-1 pathway and inhibiting inflammation by regulating the AKT, p38 MAPK/NF-κB pathways[J]. J Zhejiang Univ SCIENCE B, 2023, 24(7): 617-31. doi:10.1631/jzus.B2200612 |
| [16] | Miao ZM, Lai Y, Zhao YY, et al. Protective property of scutellarin against liver injury induced by carbon tetrachloride in mice[J]. Front Pharmacol, 2021, 12: 710692. doi:10.3389/fphar.2021.710692 |
| [17] | Kanehisa M, Bork P. Bioinformatics in the post-sequence era[J]. Nat Genet, 2003, 33(S3): 305-10. doi:10.1038/ng1109 |
| [18] | Nogales C, Mamdouh ZM, List M, et al. Network pharmacology: curing causal mechanisms instead of treating symptoms[J]. Trends Pharmacol Sci, 2022, 43(2): 136-50. doi:10.1016/j.tips.2021.11.004 |
| [19] | Fan XM, Wang YY, Li XF, et al. Scutellarin alleviates liver injury in type 2 diabetic mellitus by suppressing hepatocyte apoptosis in vitro and in vivo [J]. Chin Herb Med, 2023, 15(4): 542-8. doi:10.1016/j.chmed.2023.03.007 |
| [20] | Wang YY, Fan XM, Fan B, et al. Scutellarin reduce the homocysteine level and alleviate liver injury in type 2 diabetes model[J]. Front Pharmacol, 2020, 11: 538407. doi:10.3389/fphar.2020.538407 |
| [21] | Chen JYS, Chua D, Lim CO, et al. Lessons on drug development: a literature review of challenges faced in nonalcoholic fatty liver disease (NAFLD) clinical trials[J]. Int J Mol Sci, 2023, 24(1): 158. doi:10.3390/ijms24010158 |
| [22] | Thorgersen EB, Barratt-Due A, Haugaa H, et al. The role of complement in liver injury, regeneration, and transplantation[J]. Hepatology, 2019, 70(2): 725-36. doi:10.1002/hep.30508 |
| [23] | Bitto N, Liguori E, La Mura V. Coagulation, microenvironment and liver fibrosis[J]. Cells, 2018, 7(8): 85. doi:10.3390/cells7080085 |
| [24] | Li N, Yamamoto G, Fuji H, et al. Interleukin-17 in liver disease pathogenesis[J]. Semin Liver Dis, 2021, 41(4): 507-15. doi:10.1055/s-0041-1730926 |
| [25] | Hollenbach M. The role of glyoxalase-I (glo-I), advanced glycation endproducts (AGEs), and their receptor (RAGE) in chronic liver disease and hepatocellular carcinoma (HCC)[J]. Int J Mol Sci, 2017, 18(11): 2466. doi:10.3390/ijms18112466 |
| [26] | Walke PB, Bansode SB, More NP, et al. Molecular investigation of glycated insulin-induced insulin resistance via insulin signaling and AGE-RAGE axis[J]. Biochim Biophys Acta BBA Mol Basis Dis, 2021, 1867(2): 166029. doi:10.1016/j.bbadis.2020.166029 |
| [27] | Luan HL, Huo ZJ, Zhao ZF, et al. Scutellarin, a modulator of mTOR, attenuates hepatic insulin resistance by regulating hepatocyte lipid metabolism via SREBP-1c suppression[J]. Phytother Res, 2020, 34(6): 1455-66. doi:10.1002/ptr.6582 |
| [28] | Irvine KM, Okano S, Patel PJ, et al. Serum matrix metalloproteinase 7 (MMP7) is a biomarker of fibrosis in patients with non-alcoholic fatty liver disease[J]. Sci Rep, 2021, 11: 2858. doi:10.1038/s41598-021-82315-z |
| [29] | Zheng CM, Lu KC, Chen YJ, et al. Matrix metalloproteinase-7 promotes chronic kidney disease progression via the induction of inflammasomes and the suppression of autophagy[J]. Biomed Pharmacother, 2022, 154: 113565. doi:10.1016/j.biopha.2022.113565 |
| [30] | Ke B, Fan C, Yang L, et al. Corrigendum: matrix metalloproteinase-7 and kidney fibrosis[J]. Frontiers in Physiology, 2017, 8: 192. doi:10.3389/fphys.2017.00192 |
| [31] | de Almeida LGN, Thode H, Eslambolchi Y, et al. Matrix metalloproteinases: from molecular mechanisms to physiology, pathophysiology, and pharmacology[J]. Pharmacol Rev, 2022, 74(3): 714-70. doi:10.1124/pharmrev.121.000349 |
| [32] | Belaaouaj AA, Li AG, Wun TC, et al. Matrix metalloproteinases cleave tissue factor pathway inhibitor[J]. J Biol Chem, 2000, 275(35): 27123-8. doi:10.1016/s0021-9258(19)61488-2 |
| [33] | Zhang Q, Liu S, Parajuli KR, et al. Interleukin-17 promotes prostate cancer via MMP7-induced epithelial-to-mesenchymal transition[J]. Oncogene, 2017, 36(5): 687-99. doi:10.1038/onc.2016.240 |
| [34] | Rodriguez-Ramiro I, Pastor-Fernández A, López-Aceituno JL, et al. Pharmacological and genetic increases in liver NADPH levels ameliorate NASH progression in female mice[J]. Free Radic Biol Med, 2024, 210: 448-61. doi:10.1016/j.freeradbiomed.2023.11.019 |
| [35] | Deng YL, Lu LQ, Zhu DD, et al. MafG/MYH9-LCN2 axis promotes liver fibrosis through inhibiting ferroptosis of hepatic stellate cells[J]. Cell Death Differ, 2024, 31(9): 1127-39. doi:10.1038/s41418-024-01322-5 |
| [36] | Kim KE, Lee J, Shin HJ, et al. Lipocalin-2 activates hepatic stellate cells and promotes nonalcoholic steatohepatitis in high-fat diet-fed Ob/Ob mice[J]. Hepatology, 2023, 77(3): 888-901. doi:10.1002/hep.32569 |
| [37] | Al Jaberi S, Cohen A, D'Souza C, et al. Lipocalin-2: Structure, function, distribution and role in metabolic disorders[J]. Biomed Pharmacother, 2021, 142: 112002. doi:10.1016/j.biopha.2021.112002 |
| [38] | Sciarretta F, Ceci V, Tiberi M, et al. Lipocalin-2 promotes adipose-macrophage interactions to shape peripheral and central inflam-matory responses in experimental autoimmune encephalomyelitis[J]. Mol Metab, 2023, 76: 101783. doi:10.1016/j.molmet.2023.101783 |
| [39] | Ma HT, Yan XY, Liu JC, et al. Secondary ferroptosis promotes thrombogenesis after venous injury in rats[J]. Thromb Res, 2022, 216: 59-73. doi:10.1016/j.thromres.2022.06.002 |
| [40] | Zhao RY, Wei PJ, Sun X, et al. Role of lipocalin 2 in stroke[J]. Neurobiol Dis, 2023, 179: 106044. doi:10.1016/j.nbd.2023.106044 |
| [41] | Stallhofer J, Friedrich M, Konrad-Zerna A, et al. Lipocalin-2 is a disease activity marker in inflammatory bowel disease regulated by IL-17A, IL-22, and TNF-α and modulated by IL23R genotype status[J]. Inflamm Bowel Dis, 2015: 1. doi:10.1097/mib.0000000000000515 |
| [42] | Peng L, Wen L, Shi QF, et al. Scutellarin ameliorates pulmonary fibrosis through inhibiting NF‑κB/NLRP3-mediated epithelial-mesenchymal transition and inflammation[J]. Cell Death Dis, 2020, 11(11): 978. doi:10.1038/s41419-020-03178-2 |
| [43] | Zhou Y, Gu CL, Zhu Y, et al. Pharmacological effects and the related mechanism of scutellarin on inflammation-related diseases: a review[J]. Front Pharmacol, 2024, 15: 1463140. doi:10.3389/fphar.2024.1463140 |
| [44] | Su YM, Fan XM, Li SM, et al. Scutellarin improves type 2 diabetic cardiomyopathy by regulating cardiomyocyte autophagy and apoptosis[J]. Dis Markers, 2022, 2022: 3058354. doi:10.1155/2022/3058354 |
| [45] | Hou YH, Gu DS, Peng JZ, et al. Ginsenoside Rg1 regulates liver lipid factor metabolism in NAFLD model rats[J]. ACS Omega, 2020, 5(19): 10878-90. doi:10.1021/acsomega.0c00529 |
| [46] | Gu DS, Yi HA, Jiang KR, et al. Transcriptome analysis reveals the efficacy of ginsenoside-Rg1 in the treatment of nonalcoholic fatty liver disease[J]. Life Sci, 2021, 267: 118986. doi:10.1016/j.lfs.2020.118986 |
| [47] | Wang JG, Li SD, Fan XM. Transcriptome analysis revealed the therapeutic effect of scutellarin on MASLD[J]. ACS Omega, 2025, 10(21): 21095-104. doi:10.1021/acsomega.4c09465 |
| [1] | 陶红成, 梁富凯, 黄文波, 范思奇, 曾平. 酒精暴露与股骨头坏死的潜在关联:基于机器学习构建诊断模型[J]. 南方医科大学学报, 2026, 46(3): 559-569. |
| [2] | 向科霖, 章晓云, 黎征鹏, 徐志为, 刘素杰, 柴源. 胞葬相关基因UCP2、EGLN3、IL1B对骨关节炎的诊断价值及中药治疗预测:基于生物信息学及机器学习[J]. 南方医科大学学报, 2026, 46(3): 693-706. |
| [3] | 李钊泳, 周凤华, 孙晓敏, 赵华杉, 金瑶, 何培坤, 贾钰华. 降脂祛斑方多成分协同调控炎症-代谢网络改善2型糖尿病合并高脂血症:网络药理学与临床验证[J]. 南方医科大学学报, 2026, 46(1): 83-93. |
| [4] | 呼琴, 金华. 清肾颗粒通过调控miR-23b及Nrf2通路改善慢性肾脏病湿热证患者的肾功能:基于网络药理学和临床试验[J]. 南方医科大学学报, 2025, 45(9): 1867-1879. |
| [5] | 杨子为, 吕畅, 董柱, 计书磊, 毕生辉, 张雪花, 王晓武. 金樱子通过调控Src-AKT1轴抑制肺动脉高压平滑肌增殖[J]. 南方医科大学学报, 2025, 45(9): 1889-1902. |
| [6] | 云琦, 杜若丽, 贺玉莹, 张贻欣, 王佳慧, 叶红伟, 李正红, 高琴. 肉桂酸通过抑制TLR4减轻阿霉素诱导的小鼠心肌损伤铁死亡的发生[J]. 南方医科大学学报, 2025, 45(9): 1946-1958. |
| [7] | 王莹, 李静, 王伊迪, 华明钰, 胡玮彬, 张晓智. 原发性肝癌患者的临床结局与治疗反应预测模型:基于失巢凋亡和免疫基因[J]. 南方医科大学学报, 2025, 45(9): 1967-1979. |
| [8] | 饶璐, 丁家和, 魏江平, 阳勇, 张小梅, 王计瑞. 槐花通过抑制PI3K/AKT通路减轻炎症反应治疗银屑病[J]. 南方医科大学学报, 2025, 45(9): 1989-1996. |
| [9] | 张晶晶, 冯松, 张达利, 薛剑, 周超, 刘鹏程, 付双楠, 宫嫚, 冯卉, 张宁. 乙型肝炎病毒-代谢相关脂肪性肝病共病患者脂质代谢紊乱与口腔微生物组及代谢产物变化相关[J]. 南方医科大学学报, 2025, 45(9): 2034-2045. |
| [10] | 陈鑫源, 吴成挺, 李瑞迪, 潘雪芹, 张耀丹, 陶俊宇, 林才志. 双术汤通过P53/SLC7A11/GPX4通路诱导胃癌细胞铁死亡[J]. 南方医科大学学报, 2025, 45(7): 1363-1371. |
| [11] | 王立明, 陈宏睿, 杜燕, 赵鹏, 王玉洁, 田燕歌, 刘新光, 李建生. 益气滋肾方通过抑制PI3K/Akt/NF-κB通路改善小鼠慢性阻塞性肺疾病的炎症反应[J]. 南方医科大学学报, 2025, 45(7): 1409-1422. |
| [12] | 朱胤福, 李怡燃, 王奕, 黄颖而, 龚昆翔, 郝文波, 孙玲玲. 桂枝茯苓丸活性成分常春藤皂苷元通过抑制JAK2/STAT3通路抑制宫颈癌细胞的生长[J]. 南方医科大学学报, 2025, 45(7): 1423-1433. |
| [13] | 庞金龙, 赵新丽, 张振, 王豪杰, 周星琦, 杨玉梅, 李姗姗, 常小强, 李锋, 李娴. 皮肤黑色素瘤中MMRN2高表达促进肿瘤细胞的侵袭和迁移并与不良预后相关[J]. 南方医科大学学报, 2025, 45(7): 1479-1489. |
| [14] | 王康, 李海宾, 余靖, 孟源, 张虹丽. ELFN1高表达是结肠癌的预后生物标志物并促进结肠癌细胞的增殖转移[J]. 南方医科大学学报, 2025, 45(7): 1543-1553. |
| [15] | 何丽君, 陈晓菲, 闫陈昕, 师林. 扶正化积汤治疗非小细胞肺癌的分子机制:基于网络药理学及体外实验验证[J]. 南方医科大学学报, 2025, 45(6): 1143-1152. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||