南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 871-879.doi: 10.12122/j.issn.1673-4254.2026.04.15
• • 上一篇
董保龙1,2( ), 彭江山1,2, 李进茹3, 孟云1,2, 杨晓军1,2(
), 彭江山1,2, 李进茹3, 孟云1,2, 杨晓军1,2( )
)
收稿日期:2025-07-14
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
杨晓军
E-mail:1193757679@qq.com;yangxjmd@aliyun.com
作者简介:董保龙,硕士,主治医师,E-mail: 1193757679@qq.com
基金资助:
Baolong DONG1,2( ), Jiangshan PENG1,2, Jinru LI3, Yun MENG1,2, Xiaojun YANG1,2(
), Jiangshan PENG1,2, Jinru LI3, Yun MENG1,2, Xiaojun YANG1,2( )
)
Received:2025-07-14
Online:2026-04-20
Published:2026-04-24
Contact:
Xiaojun YANG
E-mail:1193757679@qq.com;yangxjmd@aliyun.com
摘要:
目的 miRNA与胃癌(GC)发生发展相关,探讨miR-593-5p靶向Polo-likekinase1(PLK1)对人GC细胞生物学功能的影响。 方法 培养4种人胃癌细胞系(MGC-803、AGS、HGC27、MKN-45)及正常的人胃黏膜上皮细胞系(GES-1)。采用反转录-聚合酶链反应(RT-PCR)技术分别检测miR-593-5p、PLK1 在各细胞系中RNA的表达情况,并筛选出一株miR-593-5p表达量较高和一株miR-593-5p表达量较低的细胞系做为工具细胞用于转染及功能实验。通过生物信息学网站TargetScans Human 7.2检测miR-593-5p与PLK1基因二者是否存在结合位点。采用脂质体转染胃癌工具细胞,构建miR-593-5p模拟物及其miR-593-5p 模拟物阴性对照分别瞬时转染至miR-593-5p表达量低的细胞系MGC-803中,将miR-593-5p抑制物及其抑制物阴性对照分别瞬时转染至miR-593-5p表达量高的细胞系MKN-45中,构建过表达和抑制miR-593-5p的胃癌细胞株。荧光显微镜下观察转染荧光效果,并采用RT-PCR技术验证并检测miR-593-5p是否转染成功。蛋白质免疫印迹实验检测分别过表达及抑制miR-593-5p后胃癌细胞系MGC-803和MKN-45中PLK1蛋白表达水平。采用划痕实验、Transwell实验、CCK-8实验及流式细胞技术分别检测在胃癌细胞系中成功过表达和抑制miR-593-5p 前后对胃癌细胞迁移、侵袭、增殖及凋亡能力的影响。 结果 PCR结果显示,与正常胃黏膜细胞相比,GC细胞系中miR-593-5p的显著表达降低且PLK1的表达增加(P<0.05),其中MGC-803细胞系miR-593-5p的表达最低,MKN-45细胞系miR-593-5p的表达最高。经生物信息学网站TargetScansHuman7.2检测miR-593-5p与PLK1基因存在相应的3’UTRs结合位点。在MGC803细胞系中过表达miR-593-5p与对照组相比较,PLK1蛋白的表达减少(P<0.05),细胞迁移、侵袭和增殖能力降低、细胞凋亡率增加(P<0.05)。在MKN-45细胞系中沉默miR-593-5p与对照组相比较,PLK1蛋白的表达增加。细胞迁移、侵袭和增殖的能力增加、细胞凋亡率降低(P<0.05)。 结论 过表达miR-593-5p可以抑制GC细胞的迁移、侵袭和增殖,并促进GC细胞的凋亡,其作用机制可能与PLK1表达的靶向下调有关。
董保龙, 彭江山, 李进茹, 孟云, 杨晓军. 过表达miR-593-5p通过靶向PLK1抑制胃癌细胞的迁移、侵袭、增殖并促进其凋亡[J]. 南方医科大学学报, 2026, 46(4): 871-879.
Baolong DONG, Jiangshan PENG, Jinru LI, Yun MENG, Xiaojun YANG. Overexpression of miR-593-5p inhibits migration, invasion and proliferation and promotes apoptosis of gastric cancer cells by targeting PLK1[J]. Journal of Southern Medical University, 2026, 46(4): 871-879.
| Primer name | Primer sequence (5'-3') |
|---|---|
| hsa-miR-593-5p-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGCTGAGCA |
| hsa-miR-593-5p-S | ACACTCCAGCTGGGAGGCACCAGCCAGGCATTG |
| hsa-miR-593-5p-A | TGGTGTCGTGGAGTCG |
| U6-S | CTCGCTTCGGCAGCACA |
| U6-A | AACGCTTCACGAATTTGCGT |
| H-PLK1-S | CGACTTCGTGTTCGTGGTGT |
| H-PLK1-A | GATGAATAACTCGGTTTCGGTG |
| H-GAPDH-S | GGAAGCTTGTCATCAATGGAAATC |
| H-GAPDH-A | TGATGACCCTTTTGGCTCCC |
表1 引物序列
Tab.1 Primer sequences for RT-qPCR
| Primer name | Primer sequence (5'-3') |
|---|---|
| hsa-miR-593-5p-RT | CTCAACTGGTGTCGTGGAGTCGGCAATTCAGTTGAGGCTGAGCA |
| hsa-miR-593-5p-S | ACACTCCAGCTGGGAGGCACCAGCCAGGCATTG |
| hsa-miR-593-5p-A | TGGTGTCGTGGAGTCG |
| U6-S | CTCGCTTCGGCAGCACA |
| U6-A | AACGCTTCACGAATTTGCGT |
| H-PLK1-S | CGACTTCGTGTTCGTGGTGT |
| H-PLK1-A | GATGAATAACTCGGTTTCGGTG |
| H-GAPDH-S | GGAAGCTTGTCATCAATGGAAATC |
| H-GAPDH-A | TGATGACCCTTTTGGCTCCC |
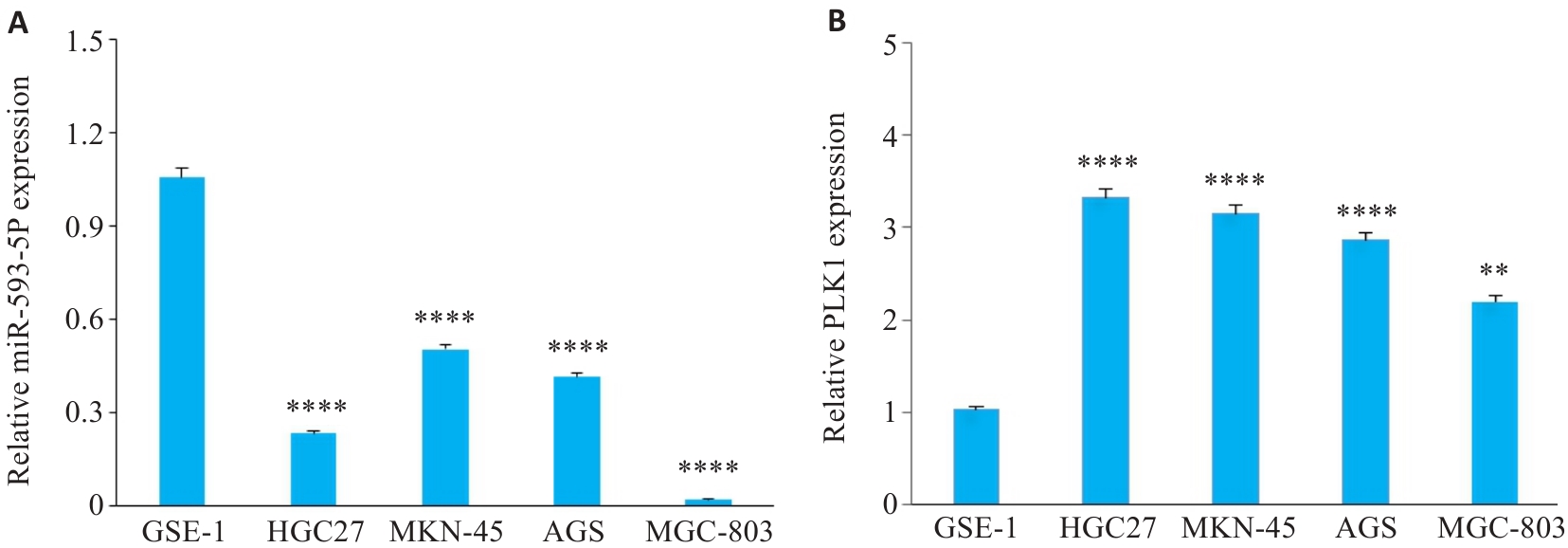
图2 GC细胞系中miR-593-5p和PLK1的差异表达
Fig.2 Differential expressions of miR-593-5p and PLK1in gastric cancer (GC) cell lines. A: miR-593-5p mRNA levels in GC cell lines. B: PLK1 mRNA leveld in GC cell lines. **P<0.01, ****P<0.0001 vs GES-1.
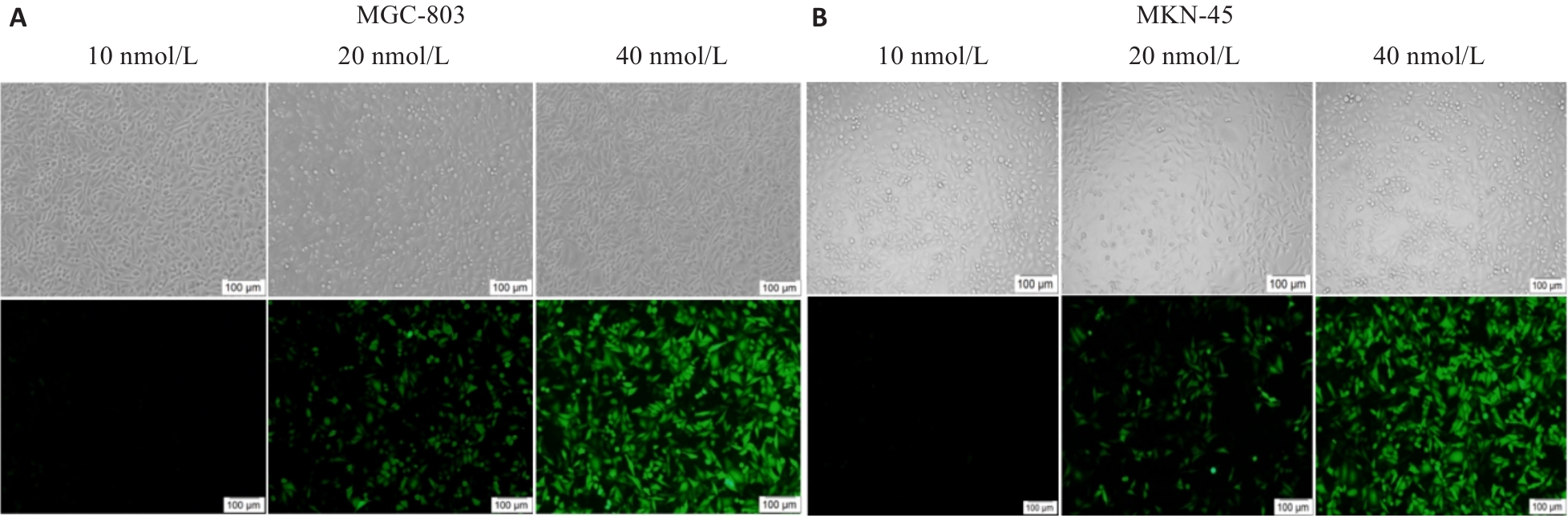
图3 荧光显微下观测转染荧光图
Fig.3 Fluorescence microscopy of the transfected cells. A, B: Transfection efficiency under different concentrations of miR-593-5p mimic or inhibitor in MGC803 cells and MKN-45 cell line (Scale bar=100 µm).
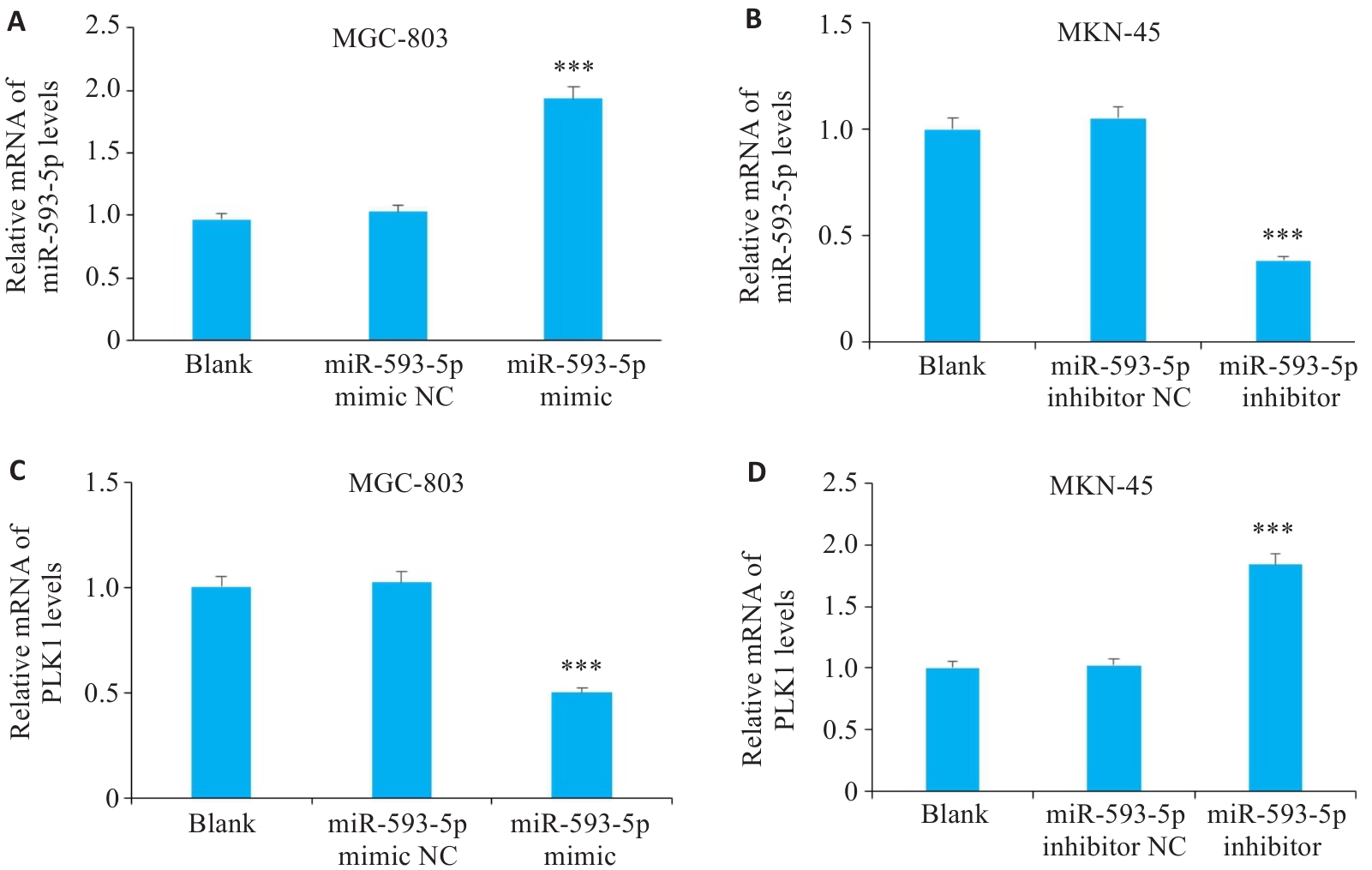
图4 过表达或敲低miR-593-5p后GC细胞系中miR-593-5p及PLK1 mRNA表达
Fig.4 Expressions of miR-593-5p and PLK1 mRNA in GC cell lines after overexpression or knockdown of miR-593-5p. A,C:n miR-593-5p and PLK1 mRNA levels after overexpression of miR-593-5p in MGC-803 cells. B,D: miR-593-5p and PLK1 mRNA levels after inhibiting miR-593-5p in MGC-803 cells. ***P<0.001 vs Blank and miR-593-5p mimic NC or miR-593-5p inhibitor NC.
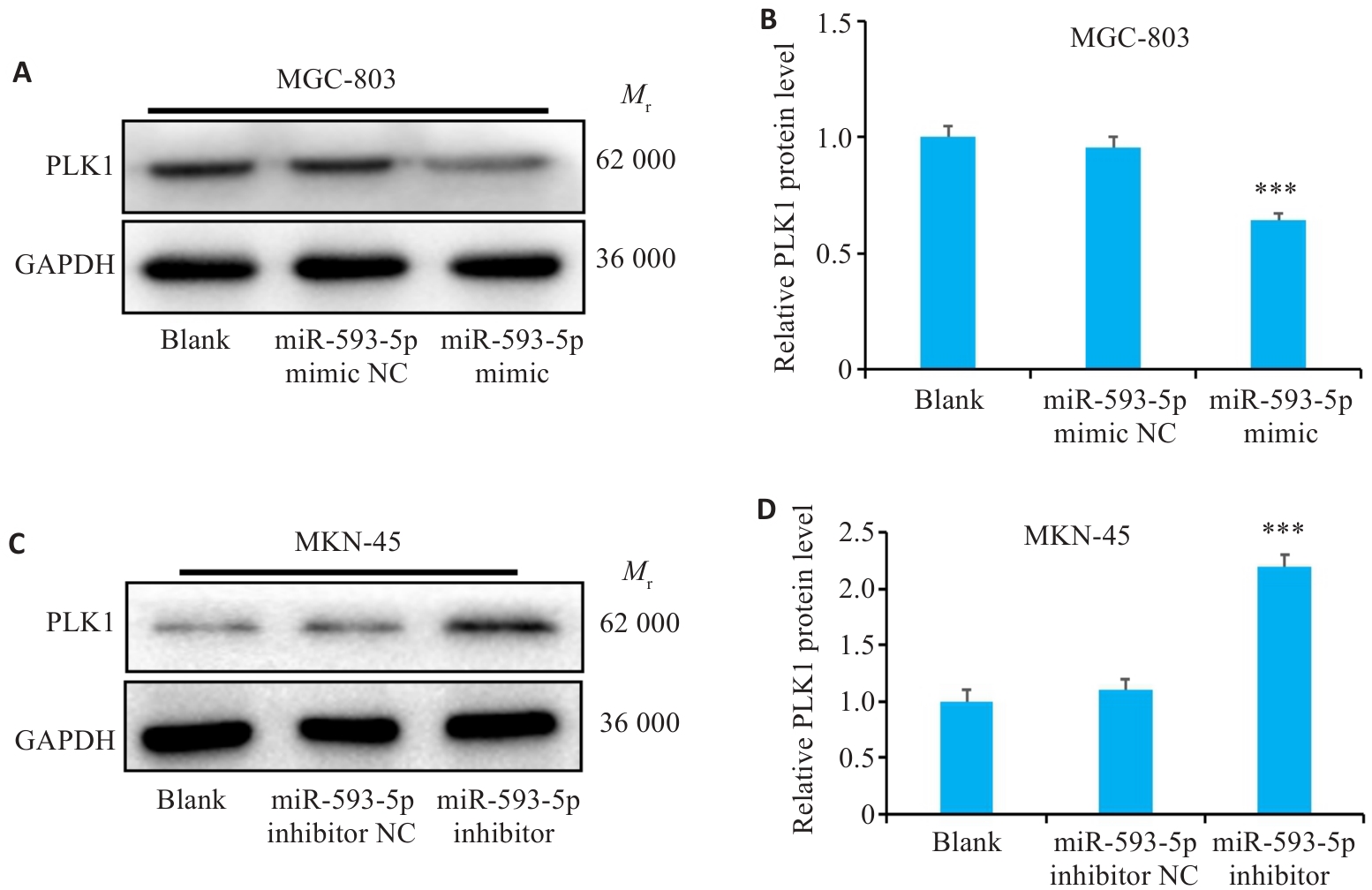
图5 过表达或敲低miR-593-5p后GC细胞系中PLK1 蛋白水平表达
Fig.5 Changes in expression of PLK1 protein in GC cells after overexpression or inhibition of miR-593-5p. A, B: Expression of PLK1 protein in MGC-803 cells transfected with miR-593-5p mimic. C, D: Expression of PLK1 protein in MKN-45 cells transfected with miR-593-5p inhibitor. ***P<0.001 vs Blank and miR-593-5p mimic NC or miR-593-5p inhibitor NC.
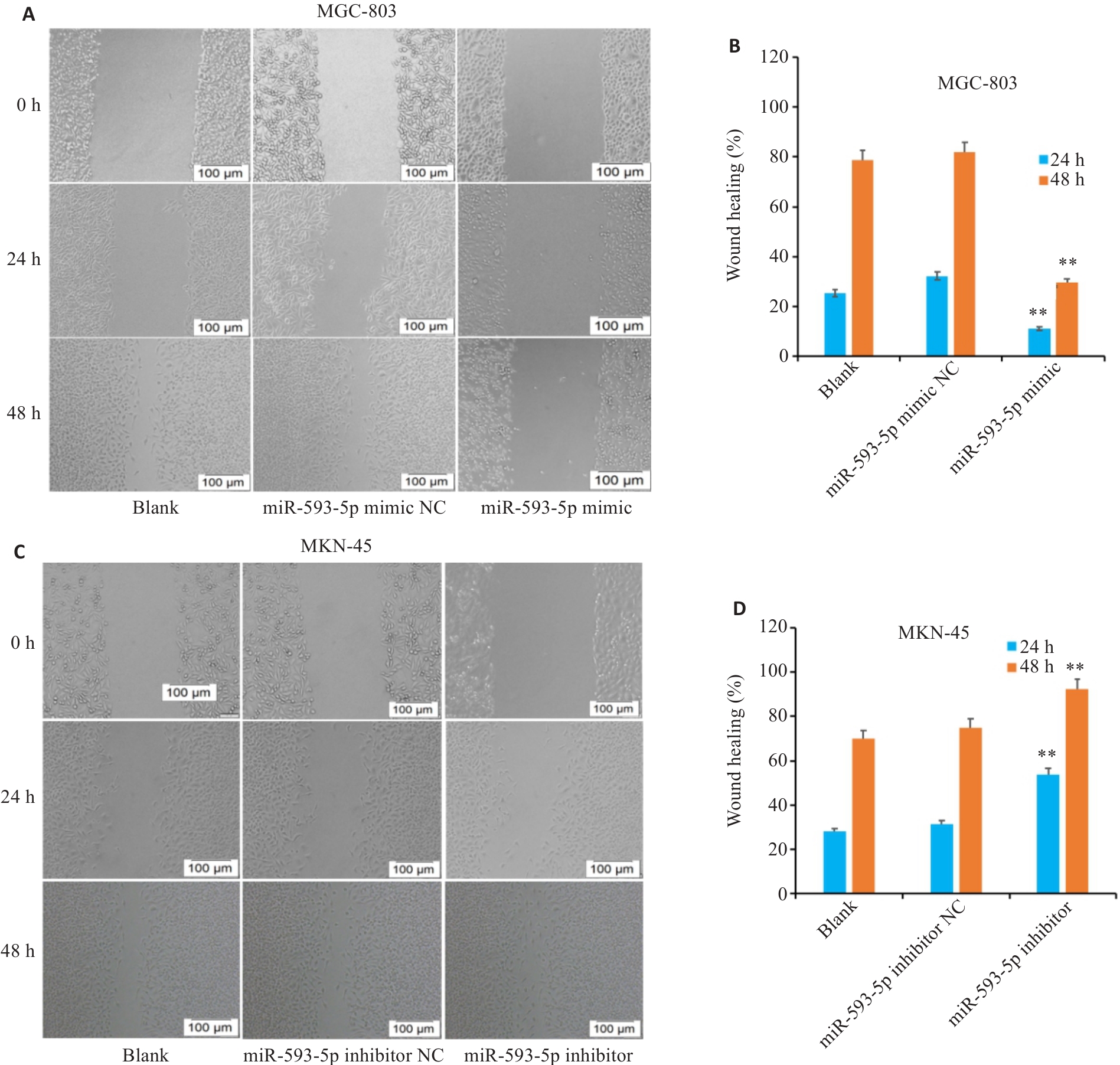
图6 miR-593-5p表达水平对胃癌细胞迁移能力的影响
Fig.6 Scratch healing assay for assessing the effect of miR-593-5p expression level on GC cell migration ability. A, B: Overexpression of miR-593-5p lowers migration ability of MGC-803 cells. C, D: Inhibition of miR-593-5p promotes migration of MKN-45 cells. **P<0.01 vs Blank and miR-593-5p mimic NC or miR-593-5p inhibitor NC.
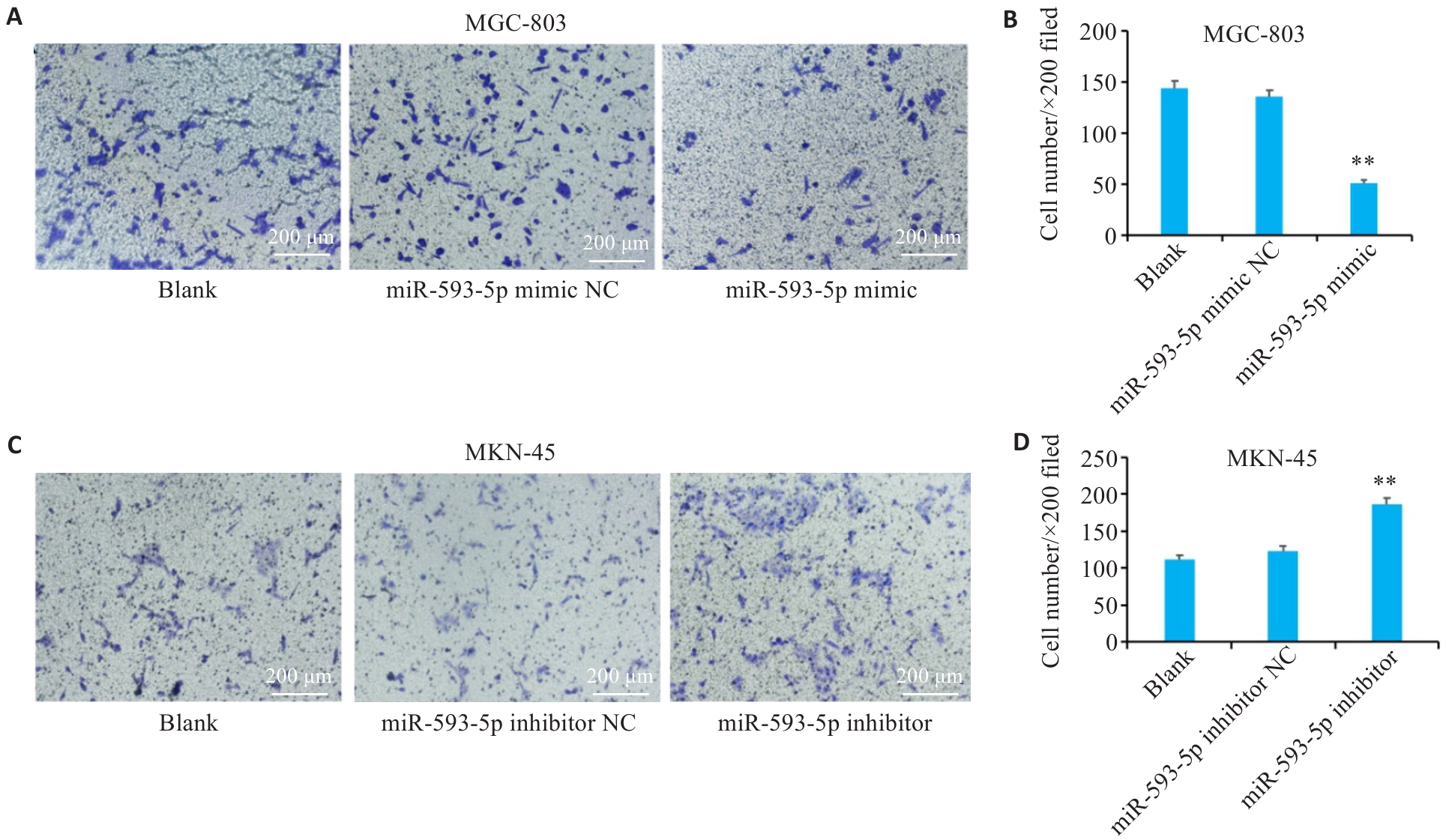
图7 miR-593-5p表达水平对胃癌细胞侵袭能力的影响
Fig 7 Transwell assay for assessing the effect of miR-593-5p expression level on GC cell invasion. A, B: Overexpression of miR-593-5p suppresses invasion of MGC-803 cells. C, D: Inhibition of miR-593-5p promotes invasion of MKN-45 cells (Scale bar=200 µm). **P<0.01 vs Blank and miR-593-5p mimic NC or miR-593-5p inhibitor NC.
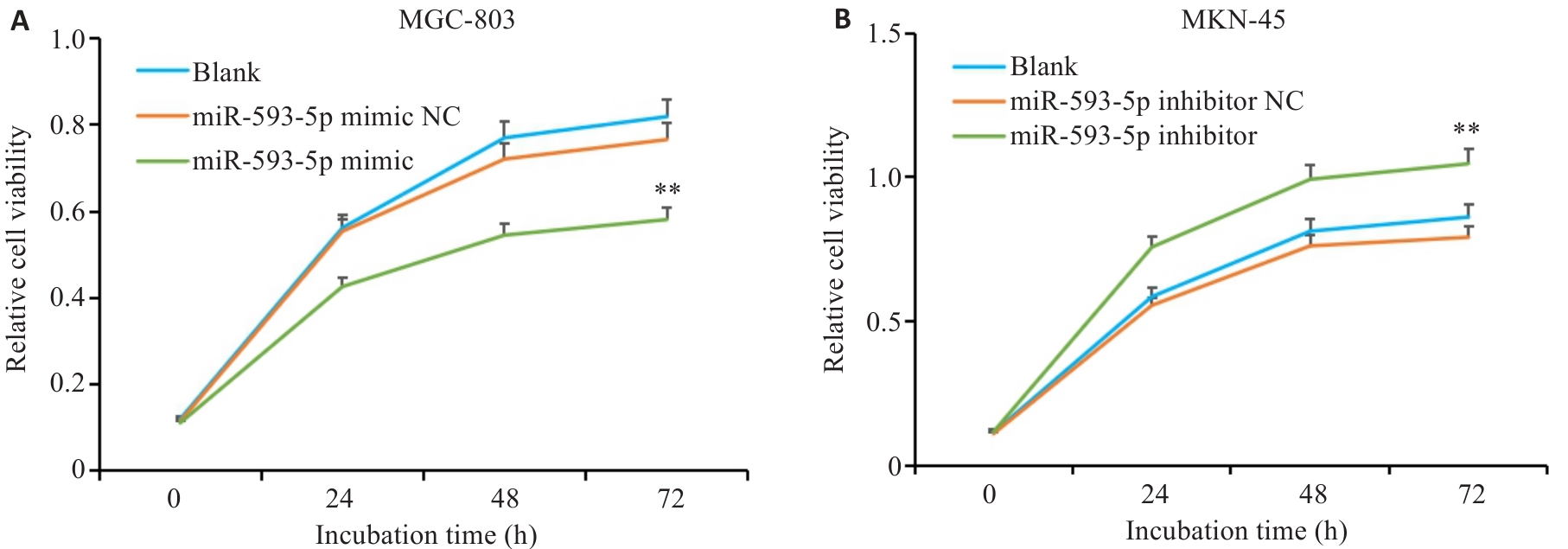
图8 miR-593-5p表达水平对GC细胞增殖能力的影响
Fig.8 CCK-8 assay for assessing the effect of miR-593-5p expression level on GC cell proliferation. A: Overexpression of miR-593-5p inhibits proliferation of MGC-803 cells. B: Inhibition of miR-593-5p promotes proliferation of MKN-45 cells. **P<0.01 vs Blank and miR-593-5p mimic NC or miR-593-5p inhibitor NC.
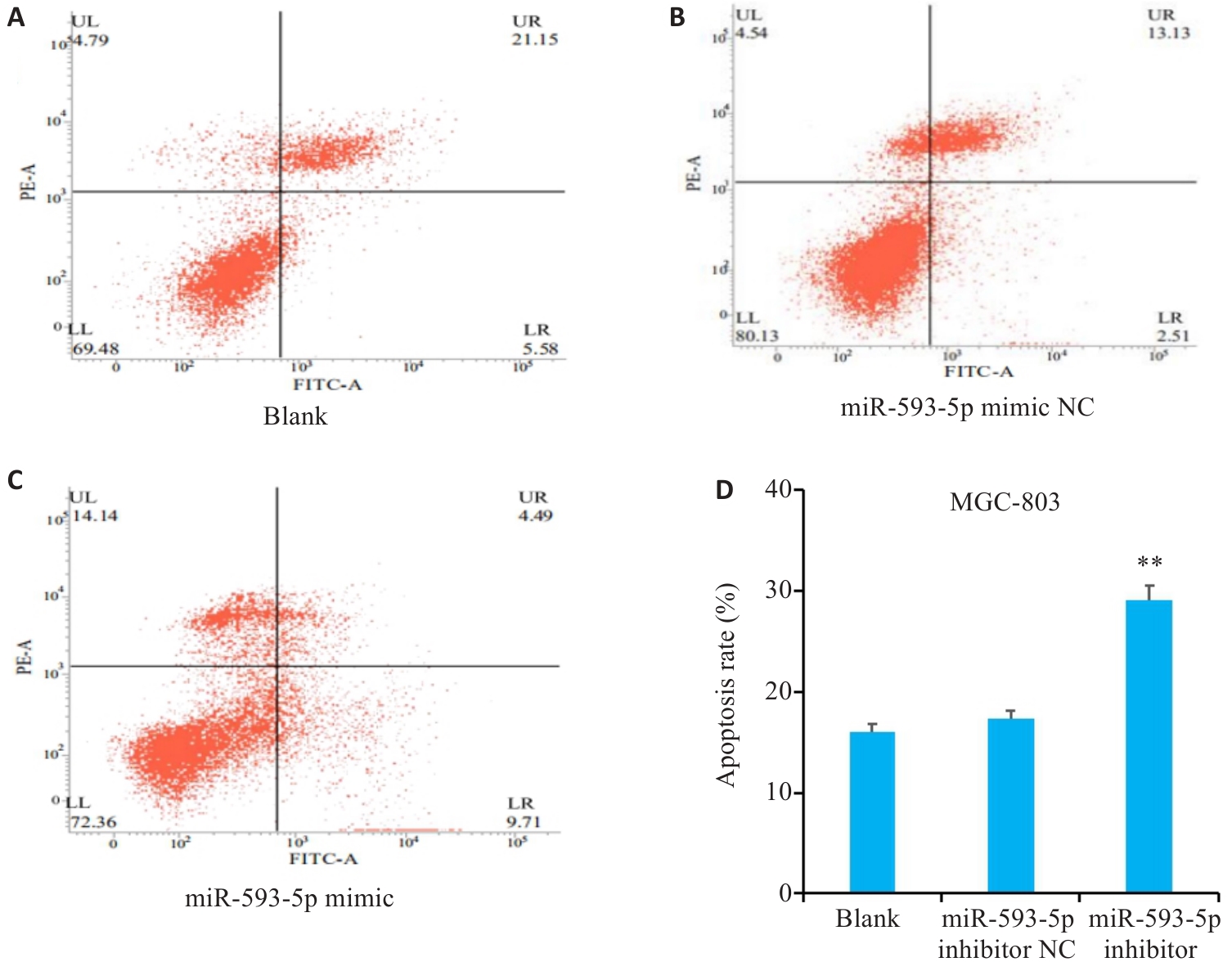
图9 过表达miR-593-5p表达水平对GC细胞凋亡能力的影响
Fig.9 Effect of miR-593-5p expression level on apoptosis of GC cells detected by flow cytcytometry. A-D: Overexpression of miR-593-5p promotes apoptosis of MGC-803 cells. **P<0.01 vs Blank and miR-593-5p mimic NC.
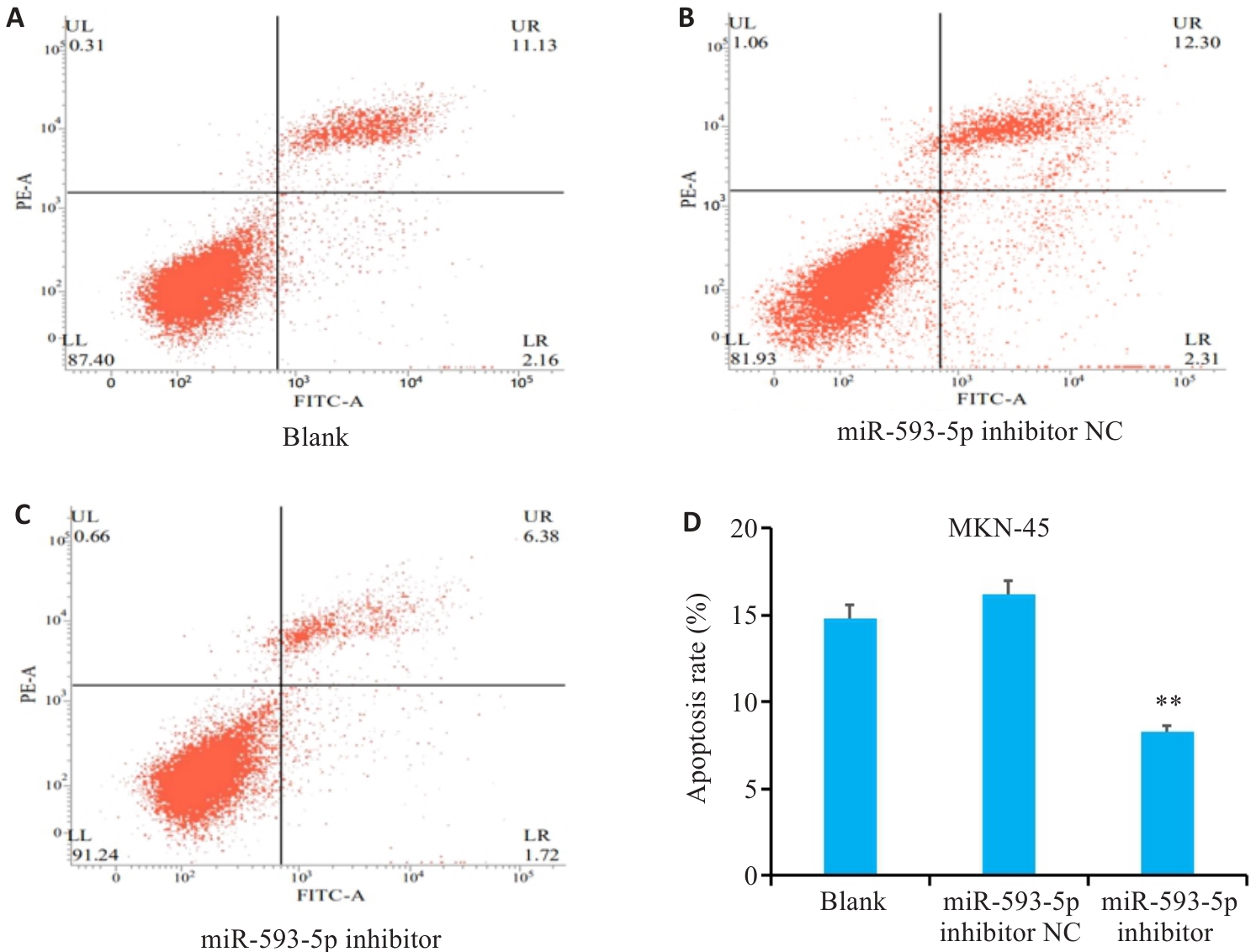
图10 降低miR-593-5p表达水平对GC细胞凋亡能力的影响
Fig.10 Effect of miR-593-5p expression level on apoptosis of GC cells detected by flow cytcytometry. A-D: Inhibiting miR-593-5p suppresses apoptosis of MKN-45 cells. **P<0.01 vs Blank and miR-593-5p inhibitor NC.
| [1] | Yasuda T, Wang Y. Gastric cancer immunosuppressive micro-environment heterogeneity: implications for therapy development[J]. Trends Cancer, 2024, 10(7): 627-42. doi:10.1016/j.trecan.2024.03.008 |
| [2] | López MJ, Carbajal J, Alfaro AL, et al. Characteristics of gastric cancer around the world[J]. Crit Rev Oncol Hematol, 2023, 181: 103841. doi:10.1016/j.critrevonc.2022.103841 |
| [3] | Ferlay J, Colombet M, Soerjomataram I, et al. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods[J]. Int J Cancer, 2019, 144(8): 1941-53. doi:10.1002/ijc.31937 |
| [4] | Zhang KC, Chen L. Chinese consensus on the diagnosis and treatment of gastric cancer with liver metastases[J]. Ther Adv Med Oncol, 2020, 12: 1758835920904803. doi:10.1177/1758835920904803 |
| [5] | Tan ZY. Recent advances in the surgical treatment of advanced gastric cancer: a review[J]. Med Sci Monit, 2019, 25: 3537-41. doi:10.12659/msm.916475 |
| [6] | Yang YQ, Ma YF, Xiang XY, et al. The prognostic value of the lymph node ratio for local advanced gastric cancer patients with intensity-modulated radiation therapy and concurrent chemotherapy after radical gastrectomy in China[J]. Radiat Oncol, 2020, 15(1): 237. doi:10.1186/s13014-020-01687-0 |
| [7] | Nakayama I, Takahari D. The role of angiogenesis targeted therapies in metastatic advanced gastric cancer: a narrative review[J]. J Clin Med, 2023, 12(9): 3226. doi:10.3390/jcm12093226 |
| [8] | Charalampakis N, Economopoulou P, Kotsantis I, et al. Medical management of gastric cancer: a 2017 update[J]. Cancer Med, 2018, 7(1): 123-33. doi:10.1002/cam4.1274 |
| [9] | Guan WL, He Y, Xu RH. Gastric cancer treatment: recent progress and future perspectives[J]. J Hematol Oncol, 2023, 16(1): 57. doi:10.1186/s13045-023-01451-3 |
| [10] | Mulrane L, Klinger R, McGee SF, et al. microRNAs: a new class of breast cancer biomarkers[J]. Expert Rev Mol Diagn, 2014, 14(3): 347-63. doi:10.1586/14737159.2014.901153 |
| [11] | Hwang H, Chang HR, Baek D. Determinants of functional microRNA targeting[J]. Mol Cells, 2023, 46(1): 21-32. doi:10.14348/molcells.2023.2157 |
| [12] | Yoo M, Choi DC, Murphy A, et al. microRNA-593-5p contributes to cell death following exposure to 1-methyl-4-phenylpyridinium by targeting PTEN-induced putative kinase 1[J]. J Biol Chem, 2023, 299(5): 104709. doi:10.1016/j.jbc.2023.104709 |
| [13] | Zhang HB, Shen B, Ma ZC, et al. miR-593-5p inhibited proliferation and migration of lung adenocarcinoma by targeting ICAM-1[J]. Eur Rev Med Pharmacol Sci, 2020, 24(8): 4298-305. doi:10.26355/eurrev_202004_21010 |
| [14] | Qu W, Huang WZ, Yang F, et al. Long noncoding RNA LINC00461 mediates cisplatin resistance of rectal cancer via miR-593-5p/CCND1 axis[J]. Biomed Pharmacother, 2020, 124: 109740. doi:10.1016/j.biopha.2019.109740 |
| [15] | Yang B, Jing C, Wang J, et al. Identification of microRNAs associated with lymphangiogenesis in human gastric cancer[J]. Clin Transl Oncol, 2014, 16(4): 374-9. doi:10.1007/s12094-013-1081-6 |
| [16] | Yu H, Wei WY, Cao WL, et al. Regulation of cell proliferation and metastasis by microRNA-593-5p in human gastric cancer[J]. Onco Targets Ther, 2018, 11: 7429-40. doi:10.2147/ott.s178151 |
| [17] | Strebhardt K. Multifaceted polo-like kinases: drug targets and antitargets for cancer therapy[J]. Nat Rev Drug Discov, 2010, 9(8): 643-60. doi:10.1038/nrd3184 |
| [18] | Kalous J, Aleshkina D. Multiple roles of PLK1 in mitosis and meiosis[J]. Cells, 2023, 12(1): 187. doi:10.3390/cells12010187 |
| [19] | Yan L, Zhang Y, Li K, et al. miR-593-5p inhibit cell proliferation by targeting PLK1 in non small cell lung cancer cells[J]. Pathol Res Pract, 2020, 216(2): 152786. doi:10.1016/j.prp.2019.152786 |
| [20] | Wang YA, Yan QJ, Fan CM, et al. Overview and countermeasures of cancer burden in China[J]. Sci China Life Sci, 2023, 66(11): 2515-26. doi:10.1007/s11427-022-2240-6 |
| [21] | Chen WQ, Zheng RS, Baade PD, et al. Cancer statistics in China, 2015[J]. CA Cancer J Clin, 2016, 66(2): 115-32. doi:10.3322/caac.21338 |
| [22] | Sekiguchi M, Oda I, Matsuda T, et al. Epidemiological trends and future perspectives of gastric cancer in eastern Asia[J]. Digestion, 2022, 103(1): 22-8. doi:10.1159/000518483 |
| [23] | 沈梦迪, 赵 娜, 邓晓晶, 等. COX6B2在胃癌组织中高表达并影响患者的远期预后: 基于抑制p53信号调控胃癌细胞的增殖及细胞周期[J]. 南方医科大学学报, 2024, 44(2): 289-97. doi:10.12122/j.issn.1673-4254.2024.02.11 |
| [24] | 马金珠, 朱益平, 王 箴, 等. miR-593通过调控PLK1基因的表达抑制结肠癌细胞的增殖[J]. 南方医科大学学报, 2019, 39(2): 144-9. doi:10.12122/j.issn.1673-4254.2019.09.03 |
| [25] | 钟腾猛, 黄俊玲, 李广志, 等. miR-593-5p调控肝癌细胞增殖、迁移及侵袭的机制[J]. 河北医药, 2020, 42(20): 3050-4, 3059. doi:10.3969/j.issn.1002-7386.2020.20.002 |
| [26] | Zhao TF, Wang SY, Zou XZ, et al. miR-593-5p promotes the development of hypoxic-induced pulmonary hypertension via targeting PLK1[J]. Eur Rev Med Pharmacol Sci, 2019, 23(8): 3495-502. doi:10.26355/eurrev_201904_17715 |
| [27] | Sekimoto N, Suzuki Y, Sugano S. Decreased KPNB1 expression is induced by PLK1 inhibition and leads to apoptosis in lung adenocarcinoma[J]. J Cancer, 2017, 8(19): 4125-40. doi:10.7150/jca.21802 |
| [28] | Chen MJ, Zhang SP, Wang F, et al. DLGAP5 promotes lung adenocarcinoma growth via upregulating PLK1 and serves as a therapeutic target[J]. J Transl Med, 2024, 22(1): 209. doi:10.1186/s12967-024-04910-8 |
| [29] | Iliaki S, Beyaert R, Afonina IS. Polo-like kinase 1 (PLK1) signaling in cancer and beyond[J]. Biochem Pharmacol, 2021, 193: 114747. doi:10.1016/j.bcp.2021.114747 |
| [30] | Zhang ZZ, Cheng LJ, Li J, et al. Targeting Plk1 sensitizes pancreatic cancer to immune checkpoint therapy[J]. Cancer Res, 2022, 82(19): 3532-48. doi:10.1158/0008-5472.can-22-0018 |
| [31] | Ataseven D, Taştemur Ş, Yulak F, et al. GSK461364A suppresses proliferation of gastric cancer cells and induces apoptosis[J]. Toxicol Vitro, 2023, 90: 105610. doi:10.1016/j.tiv.2023.105610 |
| [32] | Cai XP, Chen LD, Song HB, et al. PLK1 promotes epithelial-mesenchymal transition and metastasis of gastric carcinoma cells[J]. Am J Transl Res, 2016, 8(10): 4172-83. |
| [1] | 王煜煌, 王文锐, 程淑洁, 冯云龙, 张卓, 施其英, 李雨佳, 胡茜文, 吴明彩. 槲皮素通过调控Hippo通路蛋白YAP表达抑制胃癌细胞增殖与迁移并促进其凋亡[J]. 南方医科大学学报, 2026, 46(2): 374-384. |
| [2] | 王强, 柴志欣, 邓玉露, 张志葳, 龚英, 高圣, 冯平锋. cGAS-STING激动剂cGAMP可增强自然杀伤细胞的抗胃癌效应[J]. 南方医科大学学报, 2026, 46(2): 434-442. |
| [3] | 李艳宇, 李晨, 戴传君, 郭润之, 韩浩宇, 卢林明, 周芳芳, 支慧. 皖南蝮蛇抑瘤组分-Ι通过调控RAI14抑制顺铂耐药胃癌细胞的增殖与侵袭[J]. 南方医科大学学报, 2026, 46(1): 113-121. |
| [4] | 王子良, 陈孝华, 杨晶晶, 严晨, 张志郅, 黄炳轶, 赵萌, 刘嵩, 葛思堂, 左芦根, 陈德利. 高表达SURF4通过抑制紧密连接蛋白表达促进胃癌细胞的恶性生物学行为[J]. 南方医科大学学报, 2025, 45(8): 1732-1742. |
| [5] | 陈鑫源, 吴成挺, 李瑞迪, 潘雪芹, 张耀丹, 陶俊宇, 林才志. 双术汤通过P53/SLC7A11/GPX4通路诱导胃癌细胞铁死亡[J]. 南方医科大学学报, 2025, 45(7): 1363-1371. |
| [6] | 吴璇, 方家敏, 韩玮玮, 陈琳, 孙菁, 金齐力. 高表达PRELID1促进胃癌细胞上皮间质转化并与不良预后相关[J]. 南方医科大学学报, 2025, 45(7): 1535-1542. |
| [7] | 侯鑫睿, 张振东, 曹明远, 杜予心, 王小平. 红景天苷靶向miR-1343-3p-OGDHL/PDHB糖代谢轴抑制胃癌细胞的体内外增殖[J]. 南方医科大学学报, 2025, 45(6): 1226-1239. |
| [8] | 张毅, 沈昱, 万志强, 陶嵩, 柳亚魁, 王栓虎. CDKN3高表达促进胃癌细胞的迁移和侵袭:基于调控p53/NF-κB信号通路和抑制胃癌细胞凋亡[J]. 南方医科大学学报, 2025, 45(4): 853-861. |
| [9] | 黄晴晴, 张文静, 张小凤, 王炼, 宋雪, 耿志军, 左芦根, 王月月, 李静, 胡建国. 高表达MYO1B促进胃癌细胞增殖、迁移和侵袭并与患者的不良预后有关[J]. 南方医科大学学报, 2025, 45(3): 622-631. |
| [10] | 宋雪, 陈悦, 张敏, 张诺, 左芦根, 李静, 耿志军, 张小凤, 王月月, 王炼, 胡建国. GPSM2在胃癌组织中高表达并通过促进肿瘤细胞的增殖影响患者预后[J]. 南方医科大学学报, 2025, 45(2): 229-238. |
| [11] | 周仁杰, 杨晶晶, 宋博文, 陈孝华, 王炼, 王月月, 左芦根, 朱冰. 高表达PSMD11促进胃癌细胞上皮-间质转化进程并影响患者预后[J]. 南方医科大学学报, 2025, 45(12): 2747-2755. |
| [12] | 宋博文, 周仁杰, 徐盈, 施金冉, 张志郅, 李静, 耿志军, 宋雪, 王炼, 王月月, 左芦根. TMCO1在胃癌中高表达与患者不良预后相关并通过抑制调亡促进肿瘤恶性进展[J]. 南方医科大学学报, 2025, 45(11): 2385-2393. |
| [13] | 姜雪凝, 黄晴晴, 徐盈, 王舜印, 张小凤, 王炼, 王月月, 左芦根. 高表达YEATS2通过激活Wnt/β-catenin通路促进胃癌细胞上皮-间质转化进程[J]. 南方医科大学学报, 2025, 45(11): 2416-2426. |
| [14] | 鲁辉, 宋博文, 施金冉, 王舜印, 陈孝华, 杨晶晶, 葛思堂, 左芦根. 高表达SF3B3促进胃癌细胞恶性增殖并与患者不良预后相关[J]. 南方医科大学学报, 2025, 45(10): 2240-2249. |
| [15] | 陈孝华, 鲁辉, 王子良, 王炼, 夏勇生, 耿志军, 张小凤, 宋雪, 王月月, 李静, 胡建国, 左芦根. ABI2在胃癌进展和预后中的作用及其调控机制[J]. 南方医科大学学报, 2024, 44(9): 1653-1661. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||