南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 655-665.doi: 10.12122/j.issn.1673-4254.2026.03.19
• 基础研究 • 上一篇
收稿日期:2025-08-03
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
左芦根,刘牧林
E-mail:wenhexin66@126.com;zuolugen@126.com;liumulin66@aliyun.com
作者简介:温贺新,硕士,住院医师,E-mail: wenhexin66@126.com
基金资助:
Hexin WEN1( ), Jie LIN2, Lugen ZUO1(
), Jie LIN2, Lugen ZUO1( ), Mulin LIU1(
), Mulin LIU1( )
)
Received:2025-08-03
Online:2026-03-20
Published:2026-03-26
Contact:
Lugen ZUO, Mulin LIU
E-mail:wenhexin66@126.com;zuolugen@126.com;liumulin66@aliyun.com
摘要:
目的 探究珠子草素(Nir)对克罗恩病样结肠炎的作用与机制。 方法 建立2,4,6-三硝基苯磺酸(TNBS,2.5%)诱导的结肠炎小鼠模型,通过疾病活动度活动(DAI)评分、体质量改变、结肠长度、病理学检测评估结肠炎症状改变,AB-PAS、免疫荧光和免疫印迹评估肠屏障功能,TUNEL染色和免疫印迹评估细胞凋亡,流式细胞术分析肠系膜淋巴结中T淋巴细胞亚群(Th1/Th2)变化;ELISA检测TNF-α和IL-10表达水平变化。体外实验采用脂多糖诱导小鼠结肠类器官炎症模型,检测类器官出芽数及屏障蛋白表达。通过网络药理学和体内实验探索验证Nir拮抗结肠炎的潜在机制。 结果 体内实验显示,Nir可改善TNBS诱导结肠炎小鼠体质量下降、DAI升高、结直肠缩短等表型,降低组织炎症评分,减少杯状细胞丢失(P<0.05)及肠上皮凋亡细胞数量。体外实验表明,Nir能增加结肠类器官出芽数目(P<0.05)。免疫荧光和免疫印迹证实,Nir可恢复体内外实验结肠组织紧密连接蛋白(ZO-1、claudin-1)水平,下调促凋亡蛋白C-caspase3和BAX表达,上调抗凋亡蛋白Bcl-2(P<0.05);流式细胞术显示,Nir可降低小鼠肠系膜淋巴结Th1比例,升高Th2比例,改善Th1/Th2失衡,同时下调结肠组织TNF-α、上调IL-10水平(P<0.05);网络药理学预测Nir作用与PI3K/AKT通路相关;免疫印迹发现,Nir可下调p-PI3K、p-AKT和p-p65表达(P<0.05)。并且PI3K/AKT通路激活剂Recilisib可逆转Nir拮抗肠上皮细胞凋亡和调节Th1/Th2平衡的作用。 结论 Nir通过拮抗肠上皮细胞凋亡与调控Th1/Th2平衡来改善克罗恩病样结肠炎,其作用机制与抑制PI3K/AKT通路活性,进而调控下游凋亡信号和炎症免疫分子表达有关。
温贺新, 林洁, 左芦根, 刘牧林. 珠子草素通过拮抗肠上皮细胞凋亡与调控肠道Th1/Th2免疫平衡改善克罗恩病样小鼠结肠炎[J]. 南方医科大学学报, 2026, 46(3): 655-665.
Hexin WEN, Jie LIN, Lugen ZUO, Mulin LIU. Niranthin ameliorates Crohn's disease-like colitis in mice via antagonizing intestinal epithelial cell apoptosis and regulating intestinal Th1/Th2 immune homeostasis[J]. Journal of Southern Medical University, 2026, 46(3): 655-665.
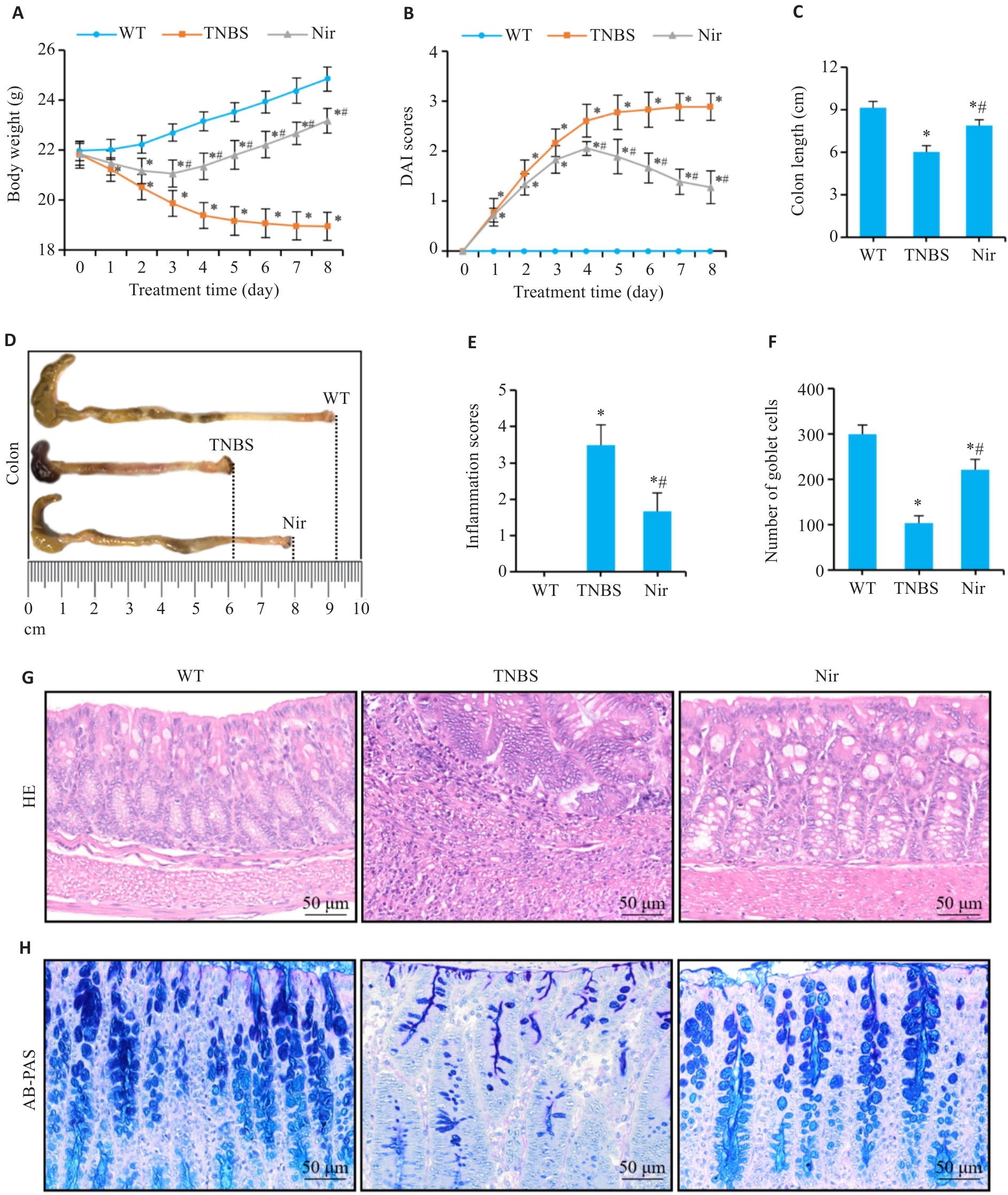
图1 Nir干预对TNBS诱导小鼠结肠炎的作用
Fig.1 Effects of niranthin (Nir) intervention in mice with TNBS-induced colitis. A: Body weight changes. B: DAI scores. C, D: Colon length. E, G: Inflammation scores and HE staining of mouse colon tissues. F, H: Number of goblet cells and AB-PAS staining. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
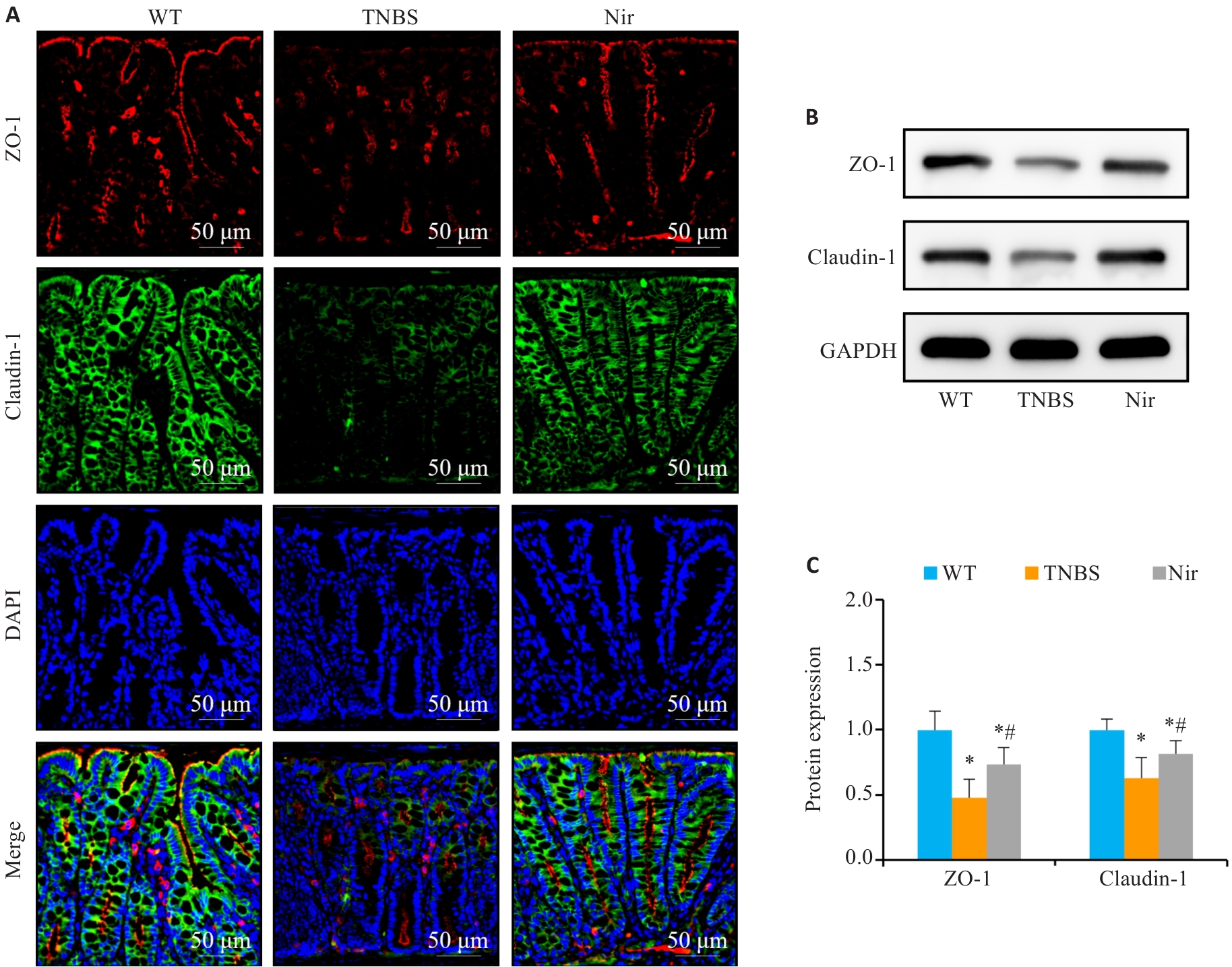
图2 Nir干预对TNBS诱导结肠炎小鼠肠屏障功能的影响
Fig.2 Effects of Nir intervention on intestinal barrier function in TNBS-induced colitis mice. A: Immunofluorescence assay of ZO-1 and claudin-1 expression. B, C: Western blotting for claudin-1 and ZO-1 expressions. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
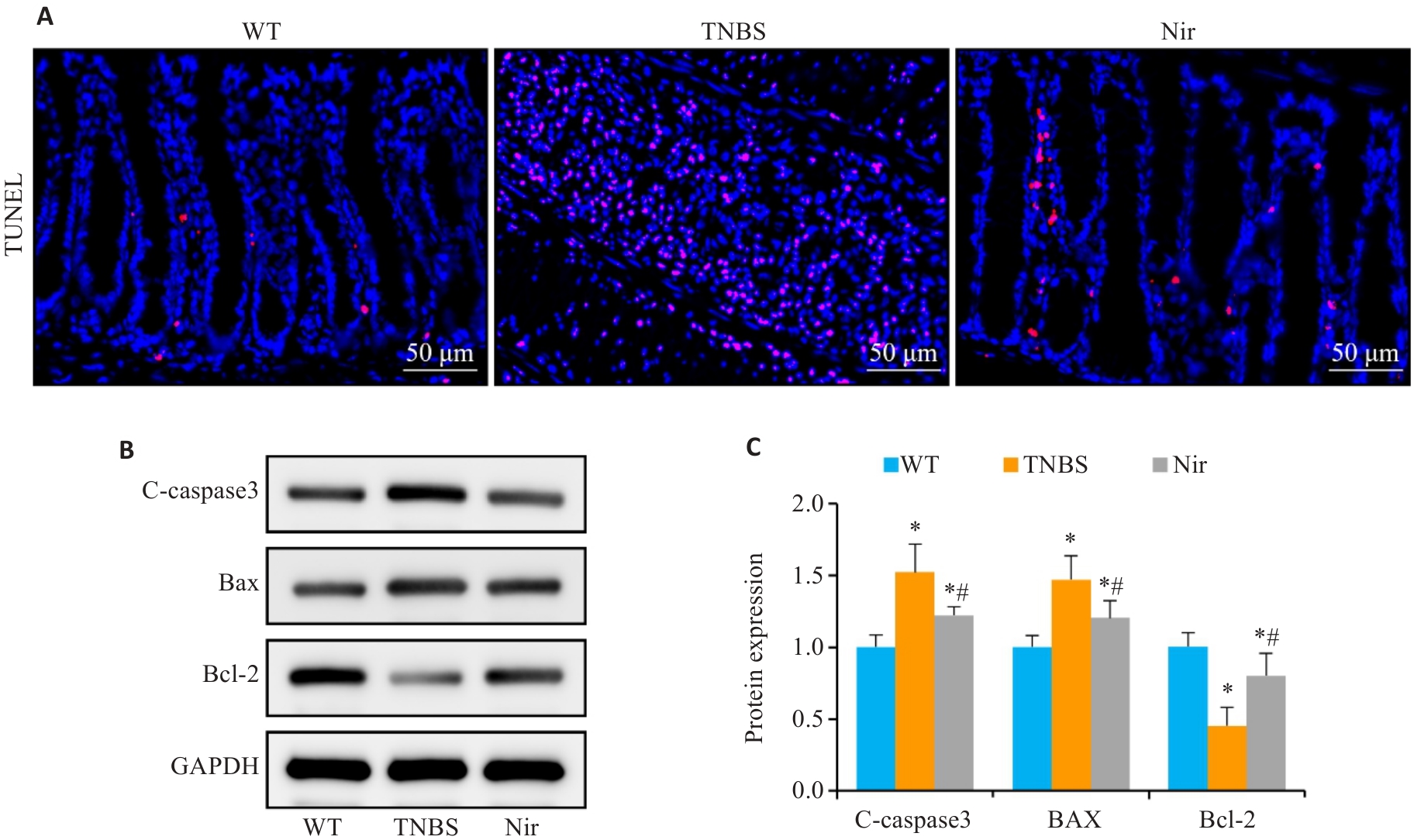
图3 Nir干预对TNBS诱导结肠炎小鼠结肠组织细胞凋亡的影响
Fig.3 The effect of Nir intervention on colonic cell apoptosis in TNBS-induced colitis mice. A: TUNEL staining. B, C: Western blotting for C-caspase-3, Bax and Bcl-2 expressions. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
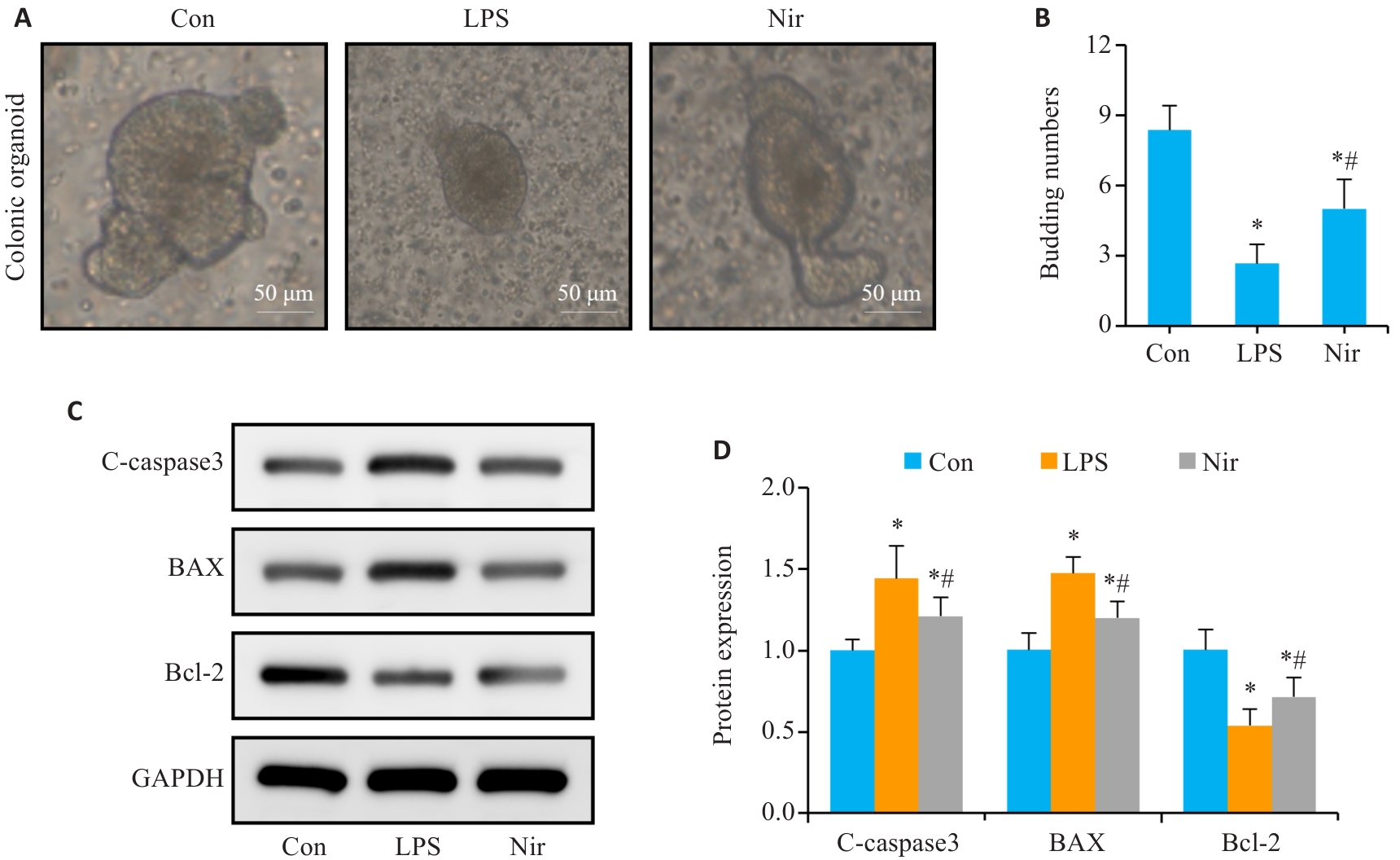
图4 Nir干预对LPS诱导结肠类器官肠上皮细胞凋亡的作用
Fig.4 Effect of Nir on LPS-induced intestinal epithelial cell apoptosis in mouse colon organoids. A: Representative images of colon organoids. B: Number of organoids budding. C, D: Western blotting and bar chart for C-caspase-3, Bax and Bcl-2 expressions. n=6, *P<0.05 vs Con; #P<0.05 vs LPS.
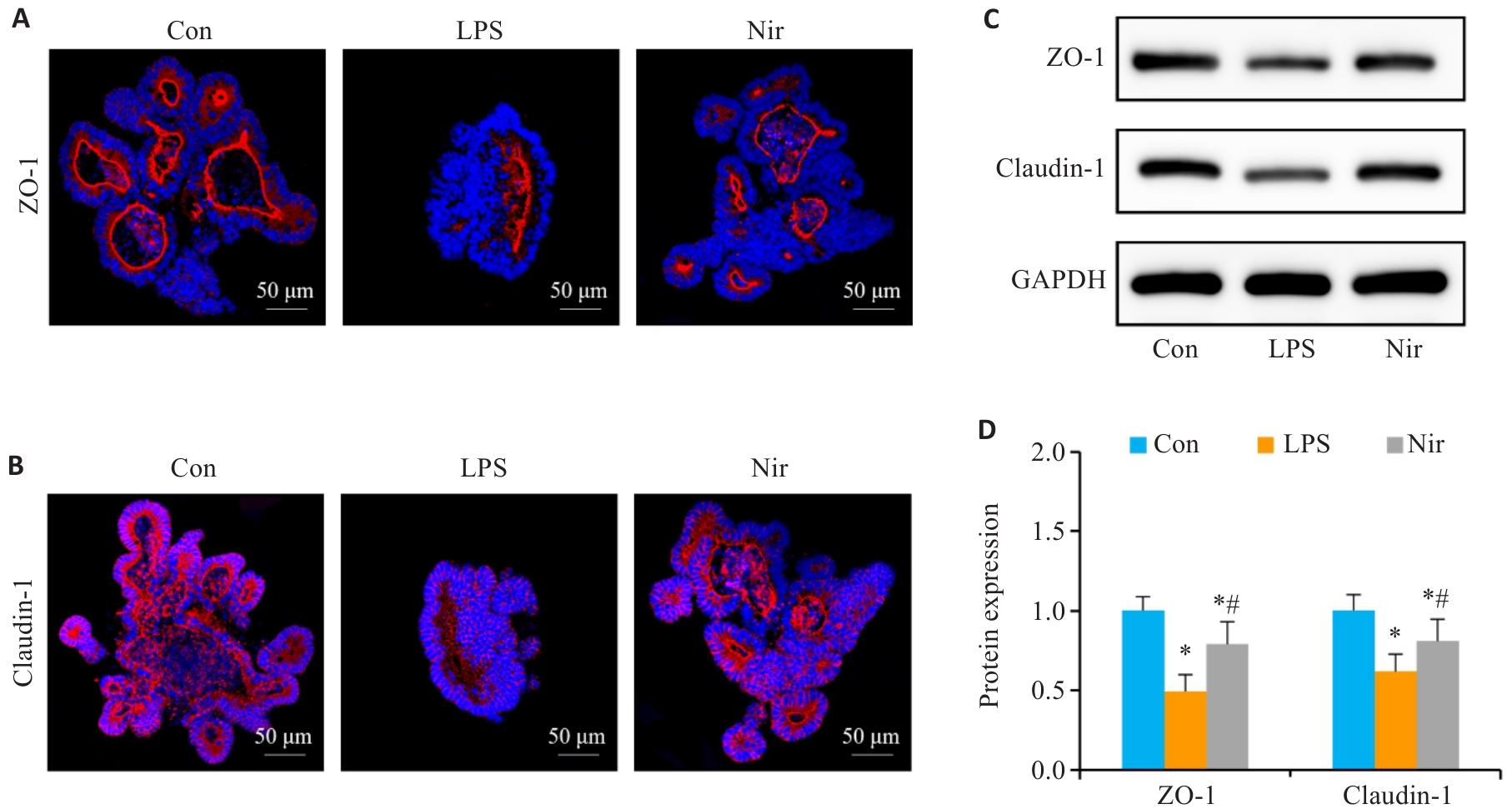
图5 Nir干预对LPS诱导结肠类器官肠屏障功能的影响
Fig.5 Effects of Nir intervention on intestinal barrier function in LPS-induced colonic organoids. A, B: Immunofluorescence assay of ZO-1 and claudin-1 expression. C, D: Western blotting for claudin-1 and ZO-1 expressions. n=6, *P<0.05 vs Con; #P<0.05 vs LPS.
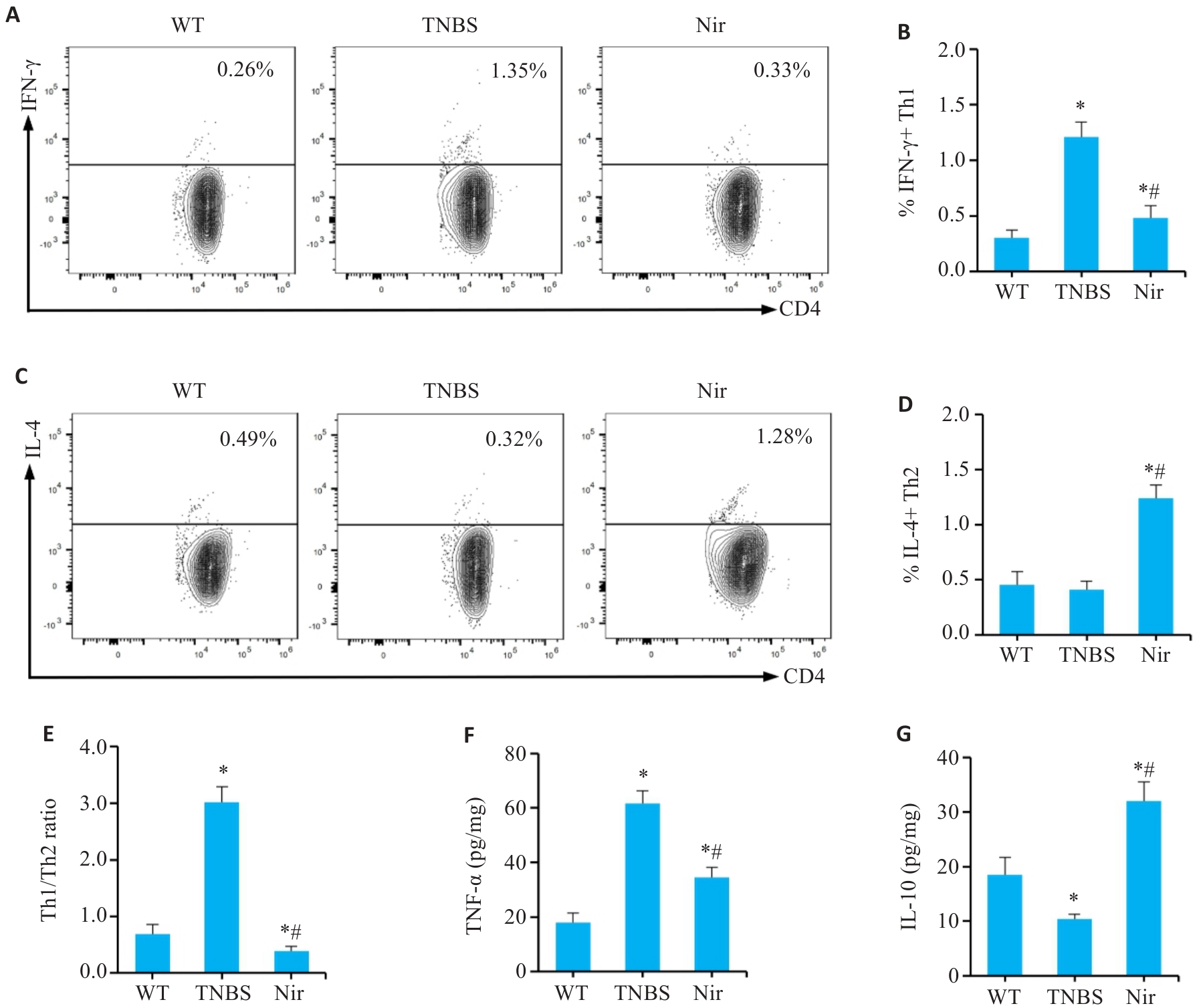
图6 Nir干预对TNBS诱导结肠炎小鼠肠系膜淋巴结Th1/Th2平衡和结肠组织炎症因子水平的影响
Fig.6 Effects of Nir intervention on Th1/Th2 balance in mesenteric lymph nodes and inflammatory factor levels in colonic tissues of TNBS-induced colitis mice. A, B: Proportion of Th1 cells in the mesenteric lymph nodes. C, D: Proportion of Th2 cells in the mesenteric lymph nodes. E: Th1/Th2 ratio. F, G: Expression levels of intestinal mucosal inflammatory mediators. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
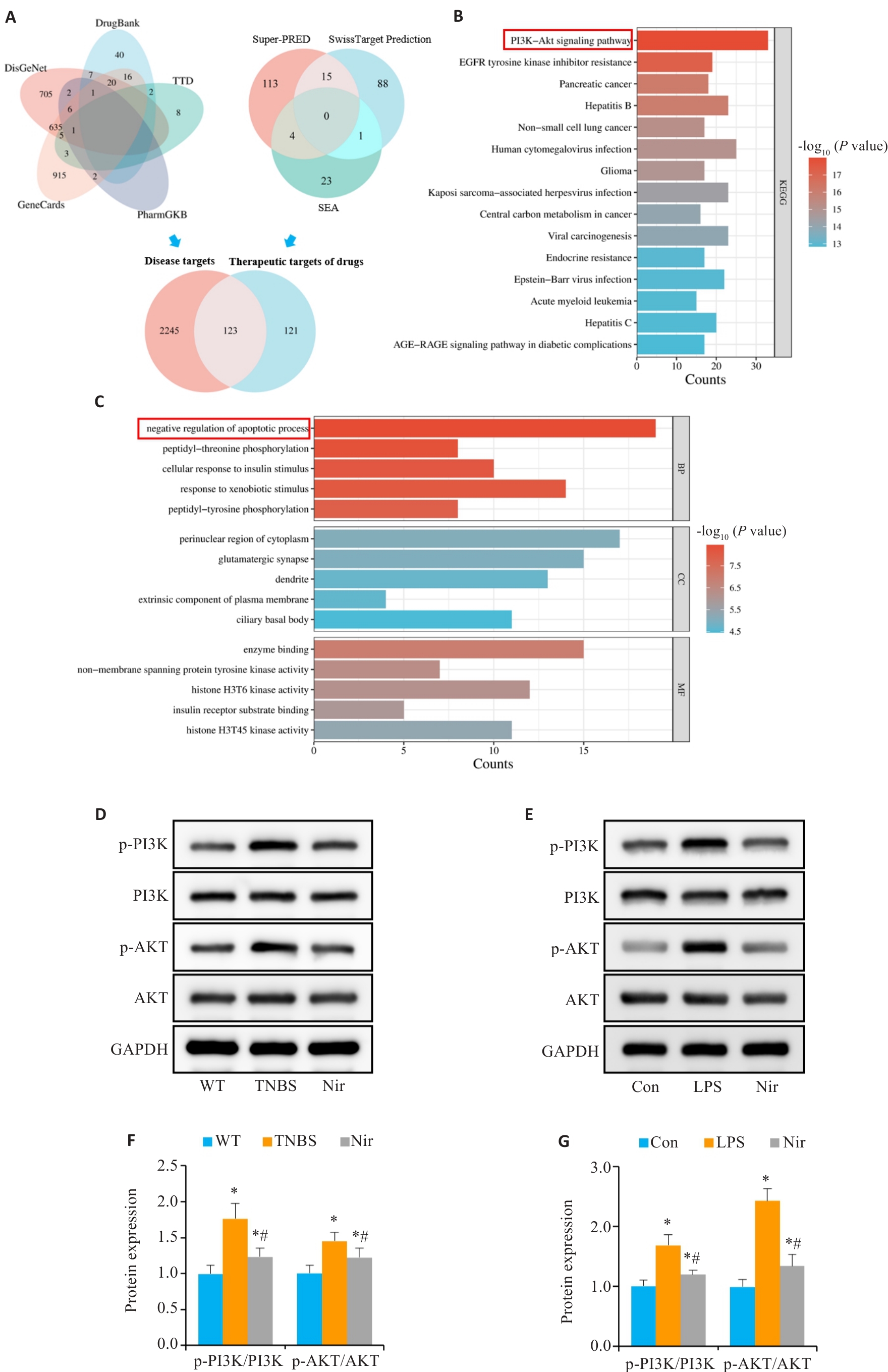
图7 Nir对肠炎的改善作用可能与PI3K/AKT通路相关
Fig.7 Ameliorative effect of Nir on colitis is possibly associated with the PI3K/AKT pathway. A: Venn diagram. B: KEGG enrichment analysis. C: GO enrichment analysis. D, F: Western blotting of p-PI3K, PI3K, p-AKT and AKT expressions in the colon tissue. E, G: Western blotting of p-PI3K, PI3K, p-AKT and AKT expressions in the colonic organoids. n=6, *P<0.05 vs WT; #P<0.05 vs TNBS.
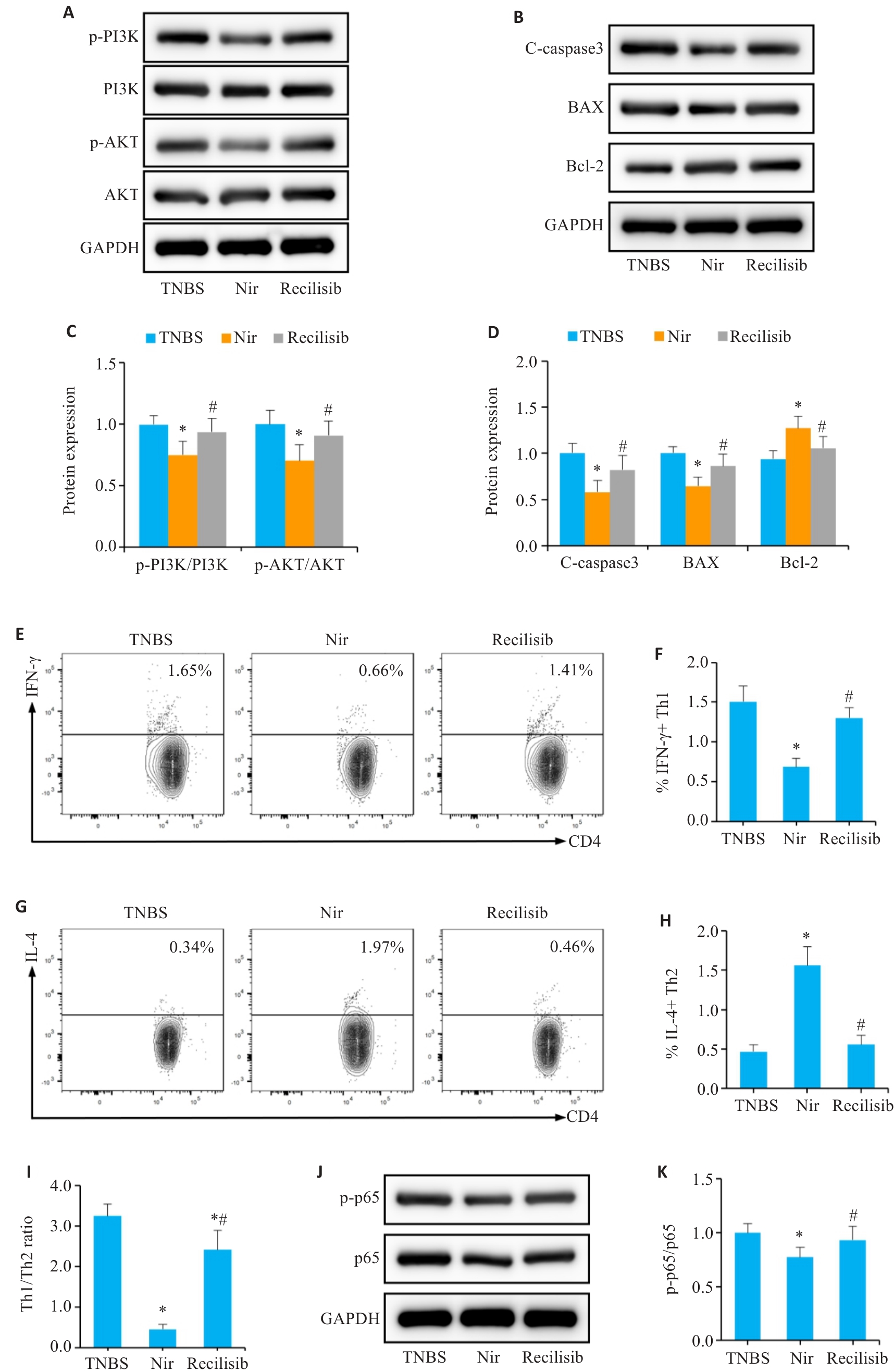
图8 Nir可以通过负调控PI3K/AKT通路恢复免疫稳态与屏障完整性
Fig.8 Nir can restore immune homeostasis and barrier integrity by negatively regulating the PI3K/AKT pathway. A, C: Western blotting of p-PI3K, PI3K, p-AKT and AKT expressions. B, D: Western blotting for C-caspase-3, Bax and Bcl-2 expressions. E, F: Proportion of Th1 cells in the mesenteric lymph nodes. G, H: Proportion of Th2 cells in the mesenteric lymph nodes. I: Th1/Th2 ratio. J, K: Western blotting for p-p65 and p65 expressions. n=6, *P<0.05 vs TNBS; #P<0.05 vs Nir.
| [1] | Torres J, Mehandru S, Colombel JF, et al. Crohn's disease[J]. Lancet, 2017, 389(10080): 1741-55. doi:10.1016/s0140-6736(16)31711-1 |
| [2] | 郭亚慧, 牛巍巍, 张晓岚. 炎症性肠病诊断与治疗的共识意见(2018年, 北京): 克罗恩病部分解读[J]. 临床荟萃, 2018, 33(12): 1077-9, 1082. doi:10.3969/j.issn.1004-583X.2018.12.016 |
| [3] | Veauthier B, Hornecker JR. Crohn's disease: diagnosis and management[J]. Am Fam Physician, 2018, 98(11): 661-9. |
| [4] | Edogawa S, Edwinson AL, Peters SA, et al. Serine proteases as luminal mediators of intestinal barrier dysfunction and symptom severity in IBS[J]. Gut, 2020, 69(1): 62-73. doi:10.1136/gutjnl-2018-317416 |
| [5] | 陈鑫明, 刘 洋, 何运胜, 等. 克罗恩病发病影响因素的研究进展[J]. 现代消化及介入诊疗, 2023, 28(7): 912-8. |
| [6] | Xavier RJ, Podolsky DK. Unravelling the pathogenesis of inflammatory bowel disease[J]. Nature, 2007, 448(7152): 427-34. doi:10.1038/nature06005 |
| [7] | Di Tommaso N, Gasbarrini A, Ponziani FR. Intestinal barrier in human health and disease[J]. Int J Environ Res Public Health, 2021, 18(23): 12836. doi:10.3390/ijerph182312836 |
| [8] | Dunleavy KA, Raffals LE, Camilleri M. Intestinal barrier dysfunction in inflammatory bowel disease: underpinning pathogenesis and therapeutics[J]. Dig Dis Sci, 2023, 68(12): 4306-20. doi:10.1007/s10620-023-08122-w |
| [9] | Farzaei MH, Bahramsoltani R, Abdolghaffari AH, et al. A mechanistic review on plant-derived natural compounds as dietary supplements for prevention of inflammatory bowel disease[J]. Expert Rev Gastroenterol Hepatol, 2016, 10(6): 745-58. doi:10.1586/17474124.2016.1145546 |
| [10] | 李晓花, 杨文玉, 王 剑. 傣药“芽害巴” (珠子草)化学成分研究[J]. 亚太传统医药, 2019, 15(2): 59-63. doi:10.11954/ytctyy.201902017 |
| [11] | Kassuya CAL, Silvestre A, Menezes-de-Lima O, et al. Antiin-flammatory and antiallodynic actions of the lignan niranthin isolated from Phyllanthus amarus Evidence for interaction with platelet activating factor receptor[J]. Eur J Pharmacol, 2006, 546(1/2/3): 182-8. doi:10.1016/j.ejphar.2006.07.025 |
| [12] | Kassuya CAL, Leite DFP, de Melo LV, et al. Anti-inflammatory properties of extracts, fractions and lignans isolated from Phyllanthus amarus[J]. Planta Med, 2005, 71(8): 721-6. doi:10.1055/s-2005-871258 |
| [13] | Ismail EN, Jantan I, Vidyadaran S, et al. Phyllanthus amarus prevents LPS-mediated BV2 microglial activation via MyD88 and NF-κB signaling pathways[J]. BMC Complement Med Ther, 2020, 20(1): 202. doi:10.1186/s12906-020-02961-0 |
| [14] | Alagan A, Jantan I, Kumolosasi E, et al. Protective effects of Phyllanthus amarus against lipopolysaccharide-induced neuroin-flammation and cognitive impairment in rats[J]. Front Pharmacol, 2019, 10: 632. doi:10.3389/fphar.2019.00632 |
| [15] | Harikrishnan H, Jantan I, Haque MA, et al. Anti-inflammatory effects of Phyllanthus amarus Schum. & Thonn. through inhibition of NF‑κB, MAPK, and PI3K-Akt signaling pathways in LPS-induced human macrophages[J]. BMC Complement Altern Med, 2018, 18(1): 224. doi:10.1186/s12906-018-2289-3 |
| [16] | Harikrishnan H, Jantan I, Alagan A, et al. Modulation of cell signaling pathways by Phyllanthus amarus and its major constituents: potential role in the prevention and treatment of inflammation and cancer[J]. Inflammopharmacology, 2020, 28(1): 1-18. doi:10.1007/s10787-019-00671-9 |
| [17] | Harikrishnan H, Jantan I, Haque MA, et al. Anti-inflammatory effects of hypophyllanthin and niranthin through downregulation of NF-κB/MAPKs/PI3K-Akt signaling pathways[J]. Inflammation, 2018, 41(3): 984-95. doi:10.1007/s10753-018-0752-4 |
| [18] | Chowdhury S, Mukherjee T, Mukhopadhyay R, et al. The lignan niranthin poisons Leishmania donovani topoisomerase IB and favours a Th1 immune response in mice[J]. EMBO Mol Med, 2012, 4(10): 1126-43. doi:10.1002/emmm.201201316 |
| [19] | Wirtz S, Popp V, Kindermann M, et al. Chemically induced mouse models of acute and chronic intestinal inflammation[J]. Nat Protoc, 2017, 12(7): 1295-309. doi:10.1038/nprot.2017.044 |
| [20] | Gong ZW, Lao DY, Wu Y, et al. Inhibiting PI3K/Akt-signaling pathway improves neurobehavior changes in anti-NMDAR encephalitis mice by ameliorating blood-brain barrier disruption and neuronal damage[J]. Cell Mol Neurobiol, 2023, 43(7): 3623-37. doi:10.1007/s10571-023-01371-3 |
| [21] | Zuo LG, Geng ZJ, Song X, et al. Browning of mesenteric white adipose tissue in Crohn's disease: a new pathological change and therapeutic target[J]. J Crohns Colitis, 2023, 17(8): 1179-92. doi:10.1093/ecco-jcc/jjad046 |
| [22] | Zuo LG, Li J, Zhang XF, et al. Aberrant mesenteric adipose extracellular matrix remodelling is involved in adipocyte dysfunction in Crohn's disease: the role of TLR-4-mediated macrophages[J]. J Crohns Colitis, 2022, 16(11): 1762-76. doi:10.1093/ecco-jcc/jjac087 |
| [23] | Paone P, Cani PD. Mucus barrier, mucins and gut microbiota: the expected slimy partners?[J]. Gut, 2020, 69(12): 2232-43. doi:10.1136/gutjnl-2020-322260 |
| [24] | Wang ZC, Shen J. The role of goblet cells in Crohn's disease[J]. Cell Biosci, 2024, 14(1): 43. doi:10.1186/s13578-024-01220-w |
| [25] | Kaminsky LW, Al-Sadi R, Ma TY. IL-1β and the intestinal epithelial tight junction barrier[J]. Front Immunol, 2021, 12: 767456. doi:10.3389/fimmu.2021.767456 |
| [26] | Schlegel N, Boerner K, Waschke J. Targeting desmosomal adhesion and signalling for intestinal barrier stabilization in inflammatory bowel diseases-Lessons from experimental models and patients[J]. Acta Physiol (Oxf), 2021, 231(1): e13492. doi:10.1111/apha.13492 |
| [27] | Saez A, Gomez-Bris R, Herrero-Fernandez B, et al. Innate lymphoid cells in intestinal homeostasis and inflammatory bowel disease[J]. Int J Mol Sci, 2021, 22(14): 7618. doi:10.3390/ijms22147618 |
| [28] | Gomez-Bris R, Saez A, Herrero-Fernandez B, et al. CD4 T-cell subsets and the pathophysiology of inflammatory bowel disease[J]. Int J Mol Sci, 2023, 24(3): 2696. doi:10.3390/ijms24032696 |
| [29] | Foerster EG, Mukherjee T, Cabral-Fernandes L, et al. How autophagy controls the intestinal epithelial barrier[J]. Autophagy, 2022, 18(1): 86-103. doi:10.1080/15548627.2021.1909406 |
| [30] | Guo NK, She H, Tan L, et al. Nano parthenolide improves intestinal barrier function of sepsis by inhibiting apoptosis and ROS via 5-HTR2A[J]. Int J Nanomedicine, 2023, 18: 693-709. doi:10.2147/ijn.s394544 |
| [31] | Woznicki JA, Saini NS, Flood P, et al. TNF-α synergises with IFN-γ to induce caspase-8-JAK1/2-STAT1-dependent death of intestinal epithelial cells[J]. Cell Death Dis, 2021, 12(10): 864. doi:10.1038/s41419-021-04151-3 |
| [32] | Zhang Y, Yang X, Ge XH, et al. Puerarin attenuates neurological deficits via Bcl-2/Bax/cleaved caspase-3 and Sirt3/SOD2 apoptotic pathways in subarachnoid hemorrhage mice[J]. Biomed Pharmacother, 2019, 109: 726-33. doi:10.1016/j.biopha.2018.10.161 |
| [33] | Sha JY, Chen KC, Liu ZB, et al. Ginseng-DF ameliorates intestinal mucosal barrier injury and enhances immunity in immuno-suppressed mice by regulating MAPK/NF‑κB signaling pathways[J]. Eur J Nutr, 2024, 63(5): 1487-500. doi:10.1007/s00394-024-03378-y |
| [34] | Mei XP, Zhou HK, Song ZW, et al. PCSK6 mediates Th1 differentiation and promotes chronic colitis progression and mucosal barrier injury via STAT1[J]. Aging (Albany NY), 2023, 15(10): 4363-73. doi:10.18632/aging.204739 |
| [35] | Ma BW, Athari SS, Mehrabi Nasab E, et al. PI3K/AKT/mTOR and TLR4/MyD88/NF‑κB signaling inhibitors attenuate pathological mechanisms of allergic asthma[J]. Inflammation, 2021, 44(5): 1895-907. doi:10.1007/s10753-021-01466-3 |
| [36] | Li ZH, Sun QM, Liu QY, et al. Compound 511 ameliorates MRSA-induced lung injury by attenuating morphine-induced immuno-suppression in mice via PI3K/AKT/mTOR pathway[J]. Phytomedicine, 2023, 108: 154475. doi:10.1016/j.phymed.2022.154475 |
| [37] | Wang JC, Hu KL, Cai XY, et al. Targeting PI3K/AKT signaling for treatment of idiopathic pulmonary fibrosis[J]. Acta Pharm Sin B, 2022, 12(1): 18-32. doi:10.1016/j.apsb.2021.07.023 |
| [38] | Lin CY, Tsai PH, Kandaswami CC, et al. Role of tissue transglutaminase 2 in the acquisition of a mesenchymal-like phenotype in highly invasive A431 tumor cells[J]. Mol Cancer, 2011, 10: 87. doi:10.1186/1476-4598-10-87 |
| [1] | 宋淇乐, 苗益恺, 冯小桐, 王一凡, 刘伟, 魏琪, 于新汝, 陈文文, 付晓艳. 硒代胱氨酸通过诱导活性氧产生启动氧化应激损伤抑制结肠癌细胞生长[J]. 南方医科大学学报, 2026, 46(3): 532-540. |
| [2] | 杨佳瑶, 何玉莲, 郭延垒, 尚芳红, 花雷, 阳勇, 张小梅, 魏江平. 党参-茯苓配伍调控ERα/PI3K/Akt信号通路改善单侧颈总动脉永久结扎痴呆大鼠的认知障碍[J]. 南方医科大学学报, 2026, 46(2): 247-258. |
| [3] | 邱佳惠, 陈萌, 满如, 陈鑫, 邱东瑞, 常启同, 马洪玉. 化瘀通便汤通过抑制PI3K/Akt信号通路促进慢传输型便秘大鼠肠道Cajal间质细胞的胞葬作用[J]. 南方医科大学学报, 2026, 46(2): 293-300. |
| [4] | 李斌杰, 周晓芳, 朗晓猛, 康欣, 刘建平. 泄浊解毒方通过调节Th17/Treg免疫平衡改善大鼠溃疡性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 335-344. |
| [5] | 尹林, 张可妮, 乔通, 牛民主, 殷丽霞, 刘馨悦, 耿志军, 李静, 胡建国. 藜芦酸通过激活Nrf2/HO-1信号通路减轻氧化应激缓解葡聚糖硫酸钠诱导的小鼠实验性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 403-411. |
| [6] | 黄林林, 郑旺, 胡建国, 宋雪, 陶露, 耿志军, 李静, 左芦根, 葛思堂. 马鞭草苷通过抑制PI3K-AKT通路减轻肠上皮炎症改善小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2026, 46(2): 423-433. |
| [7] | 乔通, 尹林, 张可妮, 牛民主, 黄菊, 耿志军, 李静, 胡建国. 茯苓新酸A通过调节AMPK/mTOR介导的自噬来减轻葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2026, 46(1): 131-140. |
| [8] | 赵铖, 李稳, 郑宝寿, 王光明, 肖芝松, 李云鹏. lncRNA SNHG12与ELAVL1相互作用激活PI3K/AKT信号通路促进前列腺癌细胞多西他赛的耐药机制[J]. 南方医科大学学报, 2026, 46(1): 183-190. |
| [9] | 张淑芬, 黄添容, 杨灿洪, 陈家镒, 吕田明, 张嘉发. 莱菔硫烷通过抑制Aβ42寡聚体激活的U87细胞中MAPK/NF-κB信号通路降低反应性星形胶质细胞介导的SH-SY5Y凋亡[J]. 南方医科大学学报, 2026, 46(1): 191-199. |
| [10] | 沙桐, 王文研, 宣佳斌, 吴洁, 石能贤, 何劲, 胡鸿彬, 张耀元. 基于Th1/Th2细胞因子检测的脓毒症免疫状态分型及预后分析:一项回顾性研究[J]. 南方医科大学学报, 2026, 46(1): 6-22. |
| [11] | 赵锦燕, 彭娇, 林明和, 朱晓勤, 黄彬, 林久茂. 清解扶正颗粒通过抑制线粒体依赖的凋亡、激活AMPK-PGC-1α通路缓解5-氟尿嘧啶引起的骨骼肌损伤[J]. 南方医科大学学报, 2026, 46(1): 94-103. |
| [12] | 王莹, 李静, 王伊迪, 华明钰, 胡玮彬, 张晓智. 原发性肝癌患者的临床结局与治疗反应预测模型:基于失巢凋亡和免疫基因[J]. 南方医科大学学报, 2025, 45(9): 1967-1979. |
| [13] | 饶璐, 丁家和, 魏江平, 阳勇, 张小梅, 王计瑞. 槐花通过抑制PI3K/AKT通路减轻炎症反应治疗银屑病[J]. 南方医科大学学报, 2025, 45(9): 1989-1996. |
| [14] | 陈丹丹, 任乾千, 吕梦林, 张宝文, 刘醒然, 张蒙, 王阳, 寇现娟. 天麻钩藤饮通过抑制坏死性凋亡通路改善帕金森病小鼠的运动功能障碍[J]. 南方医科大学学报, 2025, 45(8): 1571-1580. |
| [15] | 常笑语, 张瀚文, 曹红亭, 侯玲, 孟鑫, 陶虹, 罗彦, 李光华. 热应激对大鼠胸主动脉内皮细胞生物钟基因 Bmal1和细胞周期蛋白表达水平的影响[J]. 南方医科大学学报, 2025, 45(7): 1353-1362. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||