南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (6): 1163-1173.doi: 10.12122/j.issn.1673-4254.2025.06.06
收稿日期:2025-01-05
出版日期:2025-06-20
发布日期:2025-06-27
通讯作者:
李荣
E-mail:731997813@qq.com;nflirong@163.com
作者简介:李嘉豪,在读硕士研究生,E-mail: 731997813@qq.com
基金资助:
Jiahao LI( ), Ruiting XIAN, Rong LI(
), Ruiting XIAN, Rong LI( )
)
Received:2025-01-05
Online:2025-06-20
Published:2025-06-27
Contact:
Rong LI
E-mail:731997813@qq.com;nflirong@163.com
Supported by:摘要:
目的 探讨中链酰基辅酶A脱氢酶(ACADM)对乳腺癌细胞侵袭和转移能力的影响及潜在作用机制。 方法 采用Kaplan-Meier Plotter数据库分析ACADM在乳腺癌组织及正常组织中表达水平与预后关系。通过慢病毒转染构建人乳腺癌MCF-7细胞、T47D细胞ACADM低表达细胞株,并建立原位成瘤及裸鼠尾静脉转移模型。分为ACADM高表达及低表达MCF-7细胞、T47D细胞组,分别进行油红O染色实验、ROS检测、线粒体呼吸链功能检测,ROS清除剂、心磷脂氧化抑制剂Elamipretide、AKT激活剂SC79干预后功能检测,Transwell小室实验、Boyden体外侵袭实验检测细胞迁移及侵袭能力,Western blotting检测相关信号通路蛋白表达水平。 结果 ACADM高表达组的OS要比低表达ACADM更短。ACADM下调表达的MCF-7细胞和T47D细胞中N calnexin、Vimentin、p-P13K和p-AKT蛋白表达下调,游离脂肪酸含量显著增加,活性氧含量也增加,线粒体呼吸链复合物Ⅲ和线粒体呼吸链复合物Ⅴ的活性下降,线粒体内心磷脂明显减少,细胞侵袭能力降低,分别使用ROS清除剂、心磷脂氧化抑制剂Elamipretide、特异性AKT激活剂SC79后可逆转细胞侵袭功能。 结论 下调ACADM可抑制HR阳性乳腺癌细胞迁移、侵袭能力,这一过程是因ACADM低表达导致的脂毒性,损伤线粒体功能,并通过ROS/PI3K/AKT通路实现的。
李嘉豪, 冼瑞婷, 李荣. 下调ACADM介导的脂毒性抑制雌激素受体阳性乳腺癌细胞的侵袭与转移[J]. 南方医科大学学报, 2025, 45(6): 1163-1173.
Jiahao LI, Ruiting XIAN, Rong LI. Down-regulation of ACADM-mediated lipotoxicity inhibits invasion and metastasis of estrogen receptor-positive breast cancer cells[J]. Journal of Southern Medical University, 2025, 45(6): 1163-1173.
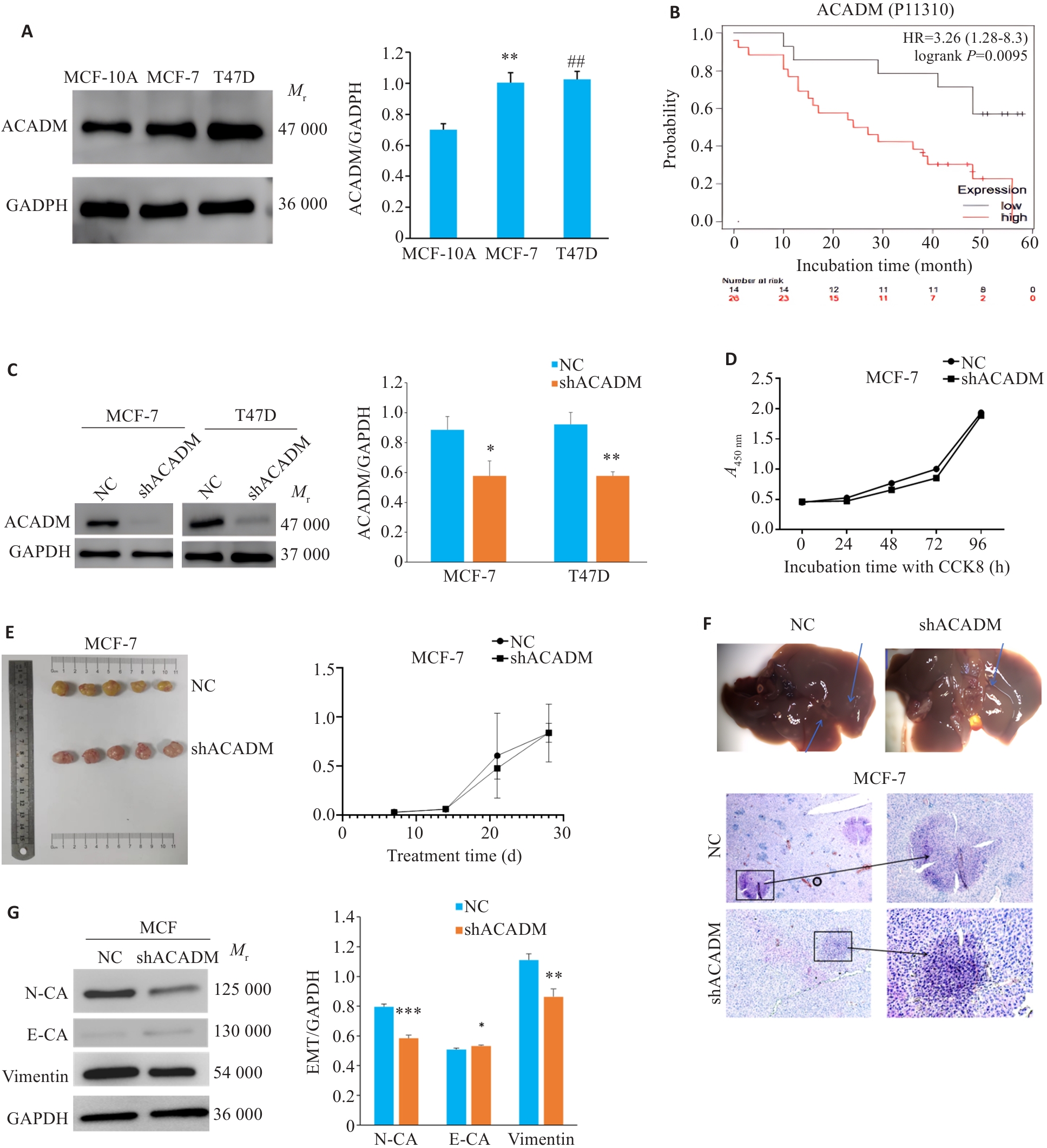
图1 ACADM 在乳腺癌中的表达水平及其对 ER 阳性乳腺癌细胞恶性生物学行为的影响
Fig.1 Expression level of ACADM in breast cancer tissues and its effect on malignant behaviors of estrogen receptor (ER)-positive breast cancer cells. A: Expression levels of ACADM detected by Western blotting in normal breast epithelial cells and ER-positive breast cancer cell lines (**P<0.01, ##P<0.01 vs MCF-10A cells). B: Prognosis of breast cancer patients with low and high ACADM expression obtained from the Kaplan-Meier Plotter bioinformatics website. C: Expression of ACADM protein in MCF-7 and T47D cells were detected by Western blotting after down-regulation of ACADM (*P<0.05, **P<0.01 vs NC). D: CCK8 assay for assessing proliferation of MCF-7 cells after down-regulation of ACADM. E: Changes of tumor size of in situ MCF-7 cell xenograft with ACADM knockdown in nude mice. F: HE staining of hepatic metastases in nude mice with tail vein injection MCF-7 cells with or without ACADM knockdown (Original magnification:×200). G: Western blotting for detecting protein expressions in the EMT signaling pathway in MCF-7 cells with ACADM knockdown. *P<0.05, **P<0.01, ***P<0.001 vs NC.

图2 下调 ACADM 表达后 MCF-7 细胞和 T47D 细胞的脂肪毒性影响细胞的侵袭能力
Fig.2 Down-regulation of ACADM-mediated lipotoxicity attenuates invasiveness of MCF-7 and T47D cells. A: Lipid droplets in shACADM MCF7 and T47D cells detected by Oil red O staining (×100). B: Free fatty acid content in shACADM MCF7 and T47D cells detected using a microplate reader (**P<0.01, ***P<0.001 vs NC). C: Ester CoA contents are increased in shACADM MCF-7 and T47D cells (*P<0.05 vs NC). D:Transwell assay for assessing invasiveness of MCF-7 and T47D cells with ACADM knockdown cultured in 4% serum (**P<0.01 vs shACADM). E: Boyden chamber assay for assessing migration ability of MCF-7 and T47D cells with ACADM knockdown cultured in 4% serum (*P<0.05, **P<0.01 vs shACADM).
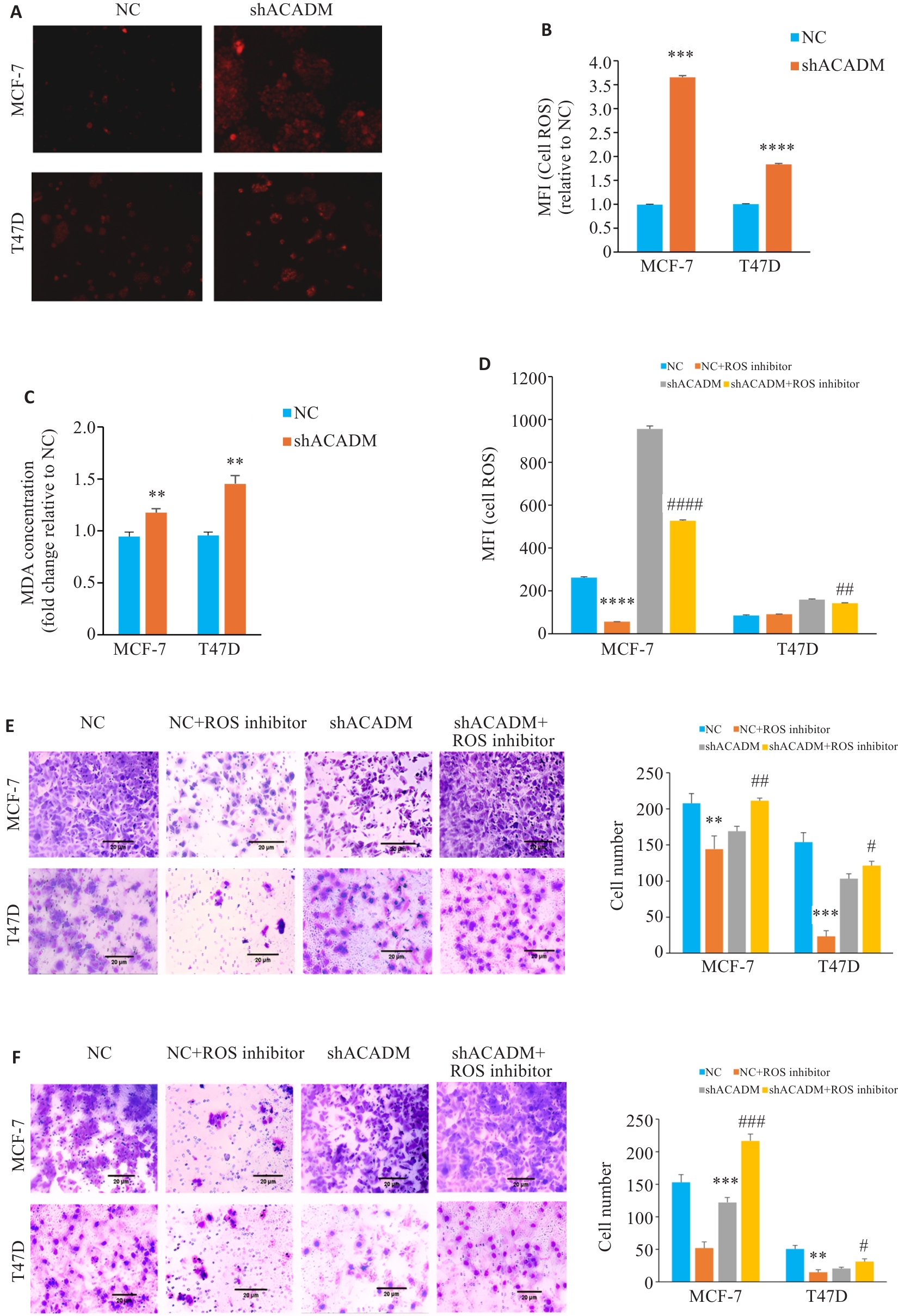
图3 下调 ACADM 导致 ROS 增加影响细胞侵袭能力
Fig.3 ACADM knockdown results in increased levels of ROS in breast cancer cells. A: ROS levels in MCF-7 and T47D cells with ACADM knockdown detected by fluorescence microscopy (×100). B: ROS levels in MCF-7 and T47D cells with ACADM knockdown detected by flow cytometry (***P<0.001, ****P<0.0001 vs NC). C: Lipid oxidation (MDA) in MCF-7 and T47D cells with ACADM knockdown detected by colorimetric method (**P<0.01 vs NC). D: ROS content in MCF-7 and T47D cells with ACADM knockdown detected by flow cytometry after treatment with ROS scavenger (****P<0.0001 vs NC; ##P<0.01, ####P<0.0001 vs shACADM). E: Invasion and migration of MCF-7 and T47D cells with ACADM knockdown detected by Transwell assay after treatment with ROS scavengers (**P<0.01, ***P<0.001 vs NC; #P<0.05, ##P<0.01 vs shACADM). F: Invasion and migration of MCF-7 and T47D cells with ACADM knockdown detected by Boyden chamber assay after treatment with ROS scavengers (**P<0.01, ***P<0.001 vs NC; #P<0.05, ###P<0.001 vs shACADM).
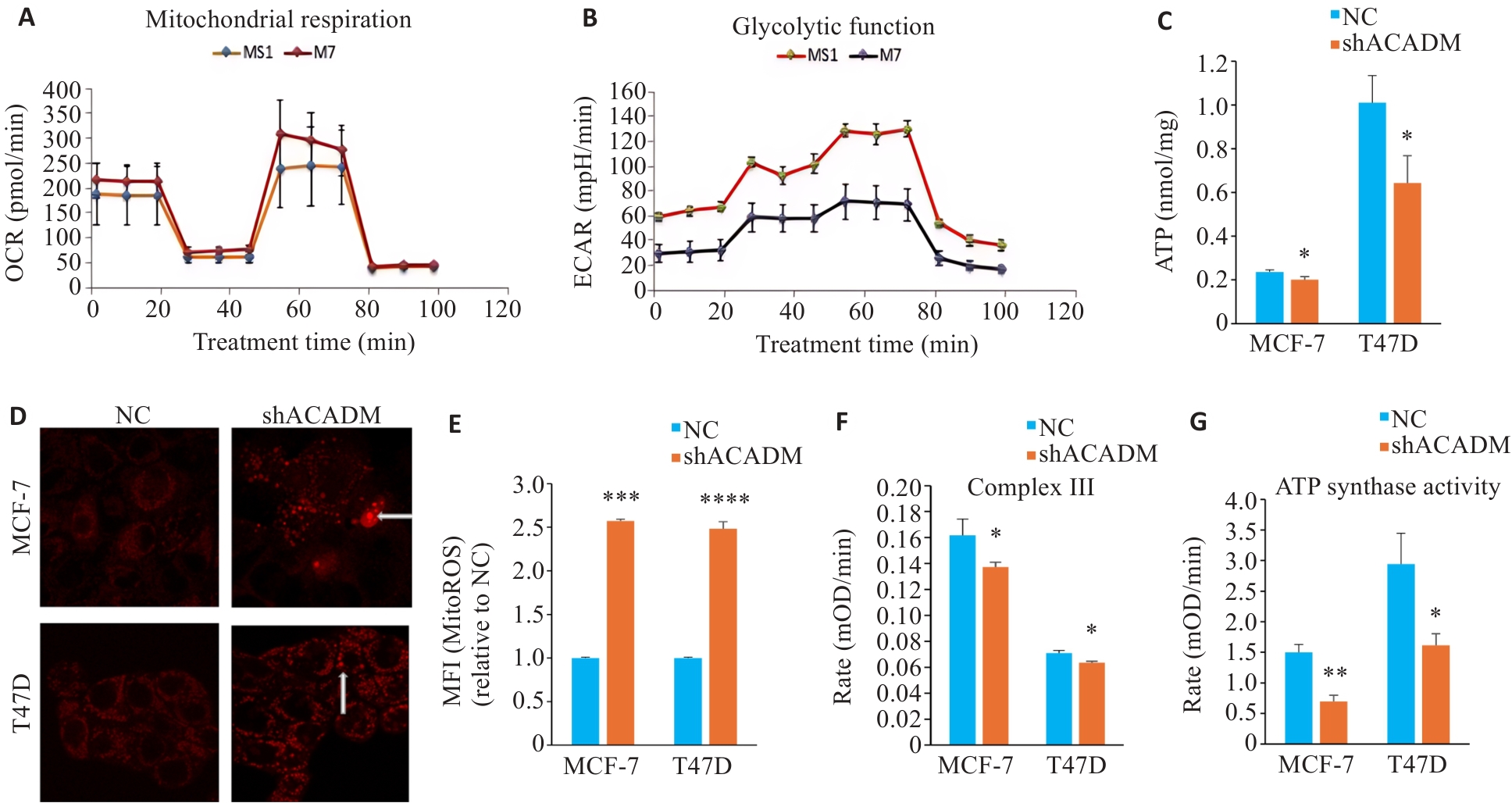
图4 下调ACADM通过损害线粒体功能影响细胞侵袭转移能力
Fig.4 Downregulation of ACADM impairs mitochondrial function in breast cancer cells. A: Seahorse technique for detecting oxygen consumption in MCF-7 cells with ACADM knockdown. B: Glycolysis in MCF-7 cells with ACADM knockdown detected using Seahorse technique. C: ATP content in MCF-7 and T47D cells with ACADM knockdown (*P<0.05 vs NC). D: Mitochondrial ROS in MCF-7 and T47D cells with ACADM knockdown detected by confocal fluorescence microscopy (×100). E: Content of mitochondrial ROS in MCF-7 and T47D with ACADM knockdown detected by flow cytometry (***P<0.001, ****P<0.0001 vs NC). F: Activity of mitochondrial respiratory chain complex III in shACADM MCF-7 and T47D cells (*P<0.05 vs NC). G: Activity of mitochondrial respiratory chain complex V in shACADM MCF-7 and T47D cells (*P<0.05, **P<0.01 vs NC).
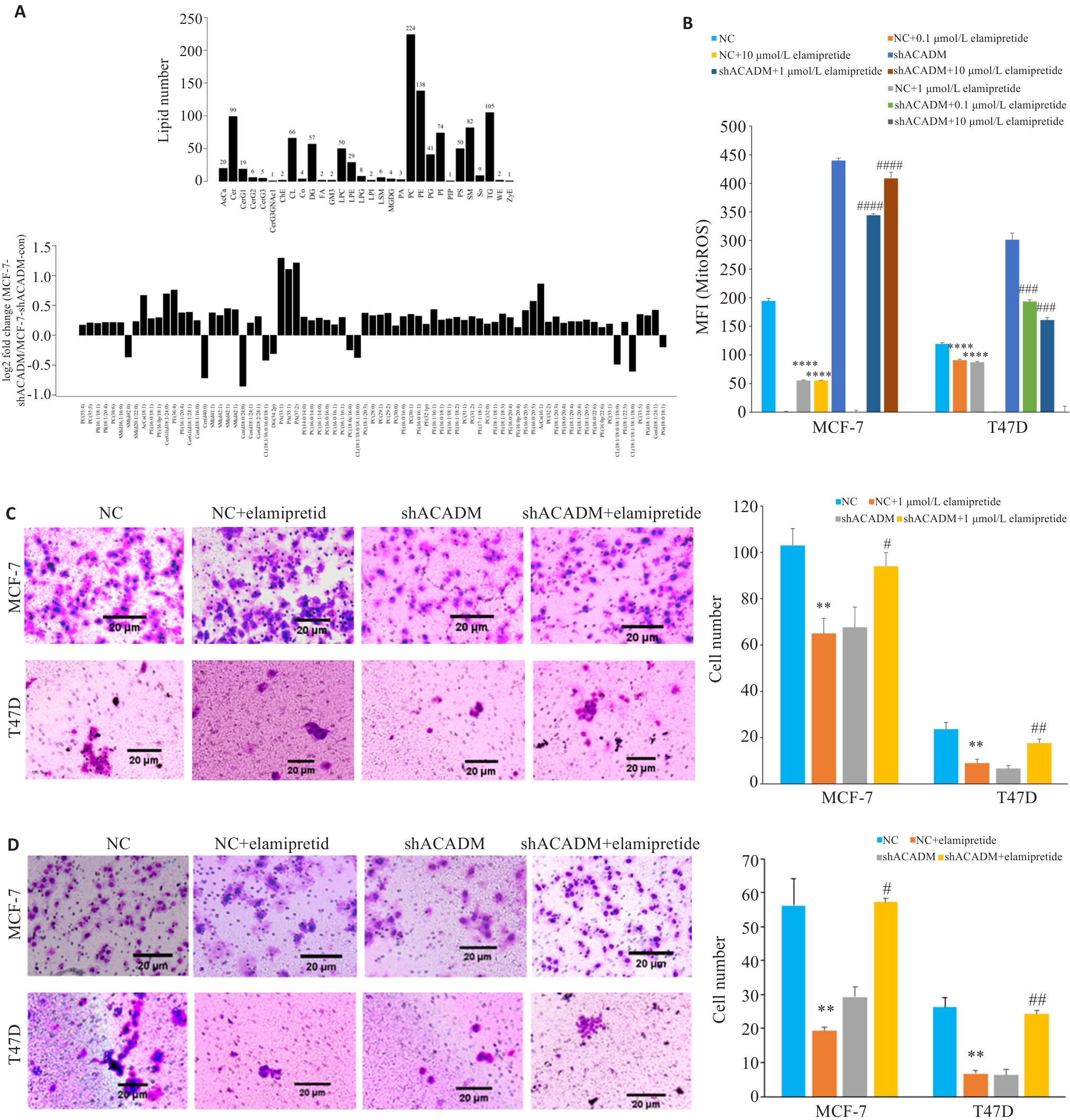
图5 ACADM通过线粒体心磷脂影响细胞侵袭能力
Fig.5 ACADM knockdown reduces mitochondrial cardiolipin to attenuate invasion and migration of breast cancer cells. A: Hierarchical clustering of lipid species in shACADM MCF-7 and T47D cells. B: Contents of mitochondrial ROS detected by flow cytometry in shACADM MCF-7 and T47D cells treated with Elamipretide (****P<0.0001 vs NC; ###P<0.001, ####P<0.0001 vs shACADM). C: Invasion and migration ability of Elamipretide-treated shACADM MCF-7 and T47D cells detected by Transwell assay (**P<0.01 vs NC; #P<0.05, ##P<0.01 vs shACADM). D: Invasion and migration ability of Elamipretide-treated shACADM MCF-7 and T47D cells detected by Boyden chamber assay (**P<0.01 vs NC; #P<0.05, ##P<0.01 vs shACADM).
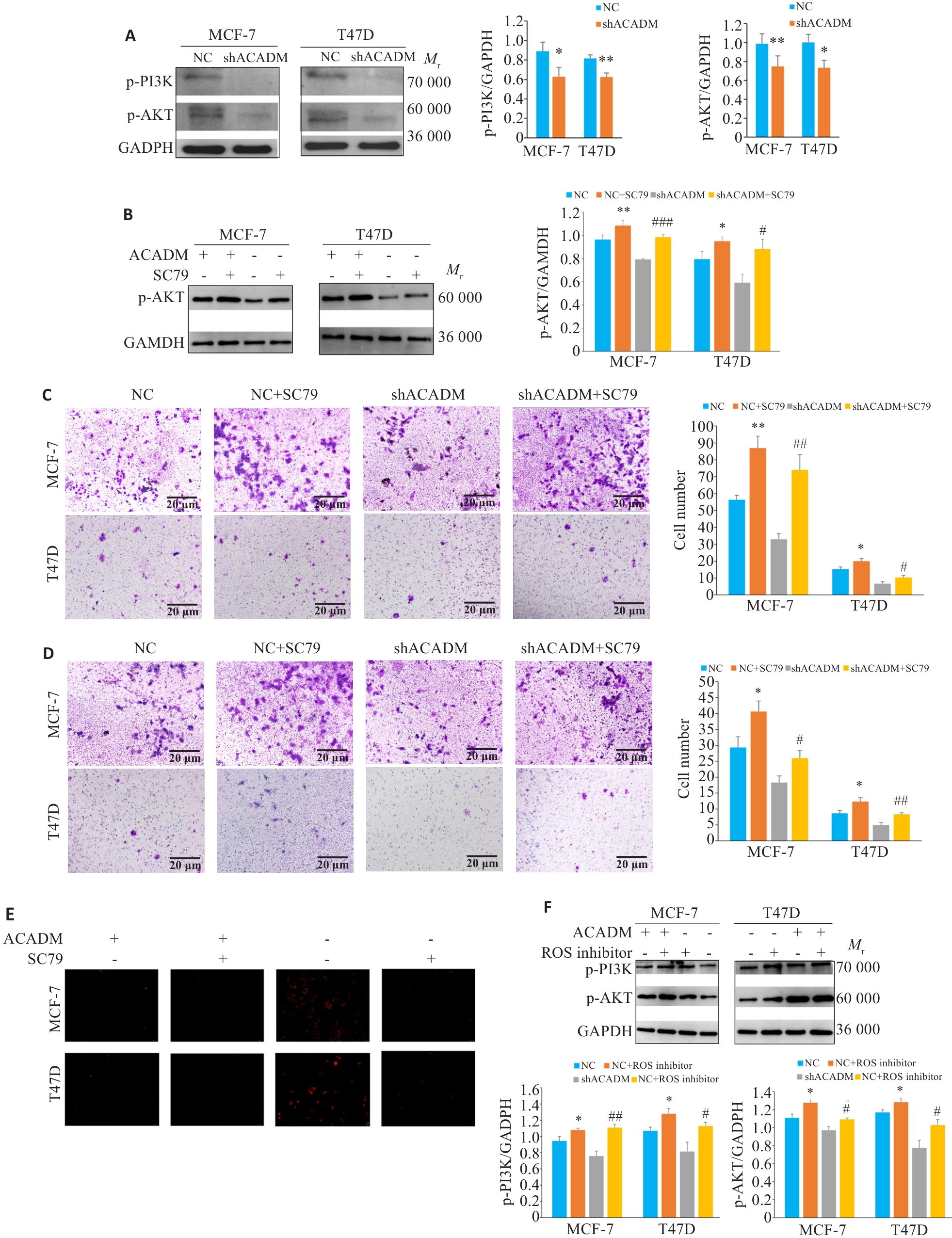
图6 ACADM通过调控PI3K/AKT通路影响细胞侵袭能力
Fig.6 ACADM knockdown affects cell invasion capacity by regulating the PI3K/AKT pathway. A: Expressions of PI3K-AKT pathway proteins in shACADM MCF-7 cells and T47D cells detected by Western blotting (*P<0.05, **P<0.01 vs NC). B: Expression of p-AKT protein in shACADM MCF-7 cells and T47D cells with SC79 treatment detected by Western blotting (*P<0.05, **P<0.01 vs NC; #P<0.05, ###P<0.001 vs shACADM). C: Invasion and metastasis ability of shACADM MCF-7 and T47D cells detected by Transwell assay after SC79 treatment (*P<0.05, **P<0.01vs NC; #P<0.05, ##P<0.01 vs shACADM). D: Invasion and migration ability of shACADM MCF-7 and T47D cells detected by Boyden chamber assay after SC79 treatment (*P<0.05 vs NC; #P<0.05, ##P<0.01 vs shACADM). E: ROS levels in shACADM MCF-7 and T47D cells detected by fluorescence microscope after ROS inhibitor treatment (×100). F: Expressions of PI3K-AKT pathway proteins in shACADM MCF-7 cells and T47D cells treated with ROS inhibitor detected by Western blotting (*P<0.05 vs NC; #P<0.05, ##P<0.01 vs shACADM).
| 1 | International Agency for Research on Cancer. Breast cancer fact sheet[EB/OL]. 2020[2024-07-21]. doi:10.1037/e533512012-001 |
| 2 | Katsura C, Ogunmwonyi I, Kankam HK, et al. Breast cancer: presentation, investigation and management[J]. Br J Hosp Med, 2022, 83(2): 1-7. doi:10.12968/hmed.2021.0459 |
| 3 | Kashyap D, Pal D, Sharma R, et al. Global increase in breast cancer incidence: risk factors and preventive measures[J]. Biomed Res Int, 2022, 2022:9605439. doi:10.1155/2022/9605439 |
| 4 | ydlowskiSP, PoirotM. Lipids, lipid oxidation, and cancer: from biology to therapeutics[J]. Front Oncol, 2024, 14:1414992. doi:10.3389/fonc.2024.1414992 |
| 5 | Dos Santos DZ, de Souza JC, Pimenta TM, et al. The impact of lipid metabolism on breast cancer: a review about its role in tumorigenesis and immune escape[J]. Cell Commun Signal, 2023, 21(1): 161. doi:10.1186/s12964-023-01178-1 |
| 6 | World Health Organization. New global breast cancer initiative highlights renewed commitment to improve survival[EB/OL].2021-03-08[2024-07-21]. |
| 7 | Martin-Perez M, Urdiroz-Urricelqui U, Bigas C, et al. The role of lipids in cancer progression and metastasis[J]. Cell Metab, 2022, 34(11): 1675-99. doi:10.1016/j.cmet.2022.09.023 |
| 8 | Balaban S, Shearer RF, Lee LS, et al. Adipocyte lipolysis links obesity to breast cancer growth: adipocyte-derived fatty acids drive breast cancer cell proliferation and migration[J]. Cancer Metab, 2017, 5: 1. doi:10.1186/s40170-016-0163-7 |
| 9 | Zeng F, Yao MK, Wang Y, et al. Fatty acid β-oxidation promotes breast cancer stemness and metastasis via the miRNA-328-3p-CPT1A pathway[J]. Cancer Gene Ther, 2022, 29(3/4): 383-95. doi:10.1038/s41417-021-00348-y |
| 10 | Dinarvand N, Khanahmad H, Hakimian SM, et al. Evaluation of long-chain acyl-coenzyme A synthetase 4 (ACSL4) expression in human breast cancer[J]. Res Pharm Sci, 2020, 15(1): 48-56. doi:10.4103/1735-5362.278714 |
| 11 | Chen CI, Kuo DY, Chuang HY. FASN inhibition shows the potential for enhancing radiotherapy outcomes by targeting glycolysis, AKT, and ERK pathways in breast cancer[J]. Int J Radiat Biol, 2025, 101(3): 292-303. doi:10.1080/09553002.2024.2446585 |
| 12 | Lu Y, Tian LP, Peng CC, et al. ACLY-induced reprogramming of glycolytic metabolism plays an important role in the progression of breast cancer[J]. Acta Biochim Biophys Sin, 2023, 55(5): 878-81. doi:10.3724/abbs.2023084 |
| 13 | Wang J, Zhang WF, Liu C, et al. Reprogramming of lipid metabolism mediates crosstalk, remodeling, and intervention of microenvironment components in breast cancer[J]. Int J Biol Sci, 2024, 20(5): 1884-904. doi:10.7150/ijbs.92125 |
| 14 | Zipinotti Dos Santos D, de Souza JC, Pimenta TM, et al. The impact of lipid metabolism on breast cancer: a review about its role in tumorigenesis and immune escape[J]. Cell Commun Signal, 2023,21(1):161. doi:10.1186/s12964-023-01178-1 |
| 15 | Kuhajda FP. Fatty acid synthase and cancer: new application of an old pathway[J]. Cancer Res, 2006, 66(12): 5977-80. doi:10.1158/0008-5472.can-05-4673 |
| 16 | Kapur P, Rakheja D, Roy LC, et al. Fatty acid synthase expression in cutaneous melanocytic neoplasms[J]. Mod Pathol, 2005, 18(8): 1107-12. doi:10.1038/modpathol.3800395 |
| 17 | Takahiro T, Shinichi K, Toshimitsu S. Expression of fatty acid synthase as a prognostic indicator in soft tissue sarcomas[J]. Clin Cancer Res, 2003, 9(6): 2204-12. |
| 18 | 俞殷珏, 赵林丰, 李 荣. 中链酰基辅酶A脱氢酶增强乳腺癌细胞的侵袭和转移能力[J]. 南方医科大学学报, 2019, 39(6): 650-6. |
| 19 | Ma APY, Yeung CLS, Tey SK, et al. Suppression of ACADM-mediated fatty acid oxidation promotes hepatocellular carcinoma via aberrant CAV1/SREBP1 signaling[J]. Cancer Res, 2021, 81(13): 3679-92. doi:10.1158/0008-5472.can-20-3944 |
| 20 | Puca F, Yu F, Bartolacci C, et al. Medium-chain acyl-CoA dehydrogenase protects mitochondria from lipid peroxidation in glioblastoma[J]. Cancer Discov, 2021, 11(11): 2904-23. doi:10.1158/2159-8290.cd-20-1437 |
| 21 | Liu YN, Zhu HX, Li TY, et al. Lipid nanoparticle encapsulated oleic acid induced lipotoxicity to hepatocytes via ROS overload and the DDIT3/BCL2/BAX/Caspases signaling in vitro and in vivo [J]. Free Radic Biol Med, 2024, 222: 361-70. doi:10.1016/j.freeradbiomed.2024.06.024 |
| 22 | Mason E, Hindmarch CCT, Dunham-Snary KJ. Medium-chain acyl-COA dehydrogenase deficiency: pathogenesis, diagnosis, and treatment[J]. Endocrinol Diabetes Metab, 2023, 6(1): e385. doi:10.1002/edm2.385 |
| 23 | Wang SS, Fernhoff PM, Hannon WH, et al. Medium chain acyl-CoA dehydrogenase deficiency human genome epidemiology review[J]. Genet Med, 1999, 1(7): 332-9. doi:10.1097/00125817-199911000-00004 |
| 24 | Nishida R, Nukaga S, Kawahara I, et al. Differential effects of three medium-chain fatty acids on mitochondrial quality control and skeletal muscle maturation[J]. Antioxidants, 2024, 13(7): 821. doi:10.3390/antiox13070821 |
| 25 | Onkenhout W, Venizelos V, van der Poel PF, et al. Identification and quantification of intermediates of unsaturated fatty acid metabolism in plasma of patients with fatty acid oxidation disorders[J]. Clin Chem, 1995, 41(10): 1467-74. doi:10.1093/clinchem/41.10.1467 |
| 26 | Fauser JK, Matthews GM, Cummins AG, et al. Induction of apoptosis by the medium-chain length fatty acid lauric acid in colon cancer cells due to induction of oxidative stress[J]. Chemotherapy, 2013, 59(3): 214-24. doi:10.1159/000356067 |
| 27 | Walther TC, Farese RV Jr. Lipid droplets and cellular lipid metabolism[J]. Annu Rev Biochem, 2012, 81: 687-714. doi:10.1146/annurev-biochem-061009-102430 |
| 28 | Kiss E, Kränzlin B, Wagenblaβ K, et al. Lipid droplet accumulation is associated with an increase in hyperglycemia-induced renal damage: prevention by liver X receptors[J]. Am J Pathol, 2013, 182(3): 727-41. doi:10.1016/j.ajpath.2012.11.033 |
| 29 | Peng J, Wang Q, Zhou J, et al. Targeted lipid nanoparticles encapsulating dihydroartemisinin and chloroquine phosphate for suppressing the proliferation and liver metastasis of colorectal cancer[J]. Front Pharmacol, 2021, 12: 720777. doi:10.3389/fphar.2021.720777 |
| 30 | Principe M, Borgoni S, Cascione M, et al. Alpha-enolase (ENO1) controls alpha v/beta 3 integrin expression and regulates pancreatic cancer adhesion, invasion, and metastasis[J]. J Hematol Oncol, 2017, 10(1): 16. doi:10.1186/s13045-016-0385-8 |
| 31 | Zhang ZW, Zhang H, Li DB, et al. Caspase-3-mediated GSDME induced Pyroptosis in breast cancer cells through the ROS/JNK signalling pathway[J]. J Cell Mol Med, 2021, 25(17): 8159-68. doi:10.1111/jcmm.16574 |
| 32 | Osada S, Sakashita F, Hosono Y, et al. Extracellular signal-regulated kinase phosphorylation due to menadione-induced arylation mediates growth inhibition of pancreas cancer cells[J]. Cancer Chemother Pharmacol, 2008, 62(2): 315-20. doi:10.1007/s00280-007-0610-9 |
| 33 | St-Pierre J, Buckingham JA, Roebuck SJ, et al. Topology of superoxide production from different sites in the mitochondrial electron transport chain[J]. J Biol Chem, 2002, 277(47): 44784-90. doi:10.1074/jbc.m207217200 |
| 34 | Hoffman DL, Brookes PS. Oxygen sensitivity of mitochondrial reactive oxygen species generation depends on metabolic conditions[J]. J Biol Chem, 2009, 284(24): 16236-45. doi:10.1074/jbc.m809512200 |
| 35 | Castelli S, Ciccarone F, De Falco P, et al. Adaptive antioxidant response to mitochondrial fatty acid oxidation determines the proliferative outcome of cancer cells[J]. Cancer Lett, 2023, 554: 216010. doi:10.1016/j.canlet.2022.216010 |
| 36 | Vercellino I, Sazanov LA. The assembly, regulation and function of the mitochondrial respiratory chain[J]. Nat Rev Mol Cell Biol, 2022, 23(2):141-161. [37] OnkenhoutW, VenizelosV, van der PoelPF, et al. Identification and quantification of intermediates of unsaturated fatty acid metabolism in plasma of patients with fatty acid oxidation disorders[J]. Clin Chem, 1995, 41(10):1467-74. doi:10.1038/s41580-021-00415-0 |
| 38 | Chen TH, Wang HC, Chang CJ, et al. Mitochondrial glutathione in cellular redox homeostasis and disease manifestation[J]. Int J Mol Sci, 2024, 25(2): 1314. doi:10.3390/ijms25021314 |
| 39 | Vouilloz A, Bourgeois T, Diedisheim M, et al. Impaired unsaturated fatty acid elongation alters mitochondrial function and accelerates metabolic dysfunction-associated steatohepatitis progression[J]. Metabolism, 2025, 162: 156051. doi:10.1016/j.metabol.2024.156051 |
| 40 | Randall EC, Zadra G, Chetta P, et al. Molecular characterization of prostate cancer with associated gleason score using mass spectrometry imaging[J]. Mol Cancer Res, 2019, 17(5): 1155-65. doi:10.1158/1541-7786.mcr-18-1057 |
| [1] | 龚秀莹, 侯顺福, 赵苗苗, 王晓娜, 张致涵, 刘清华, 尹崇高, 李洪利. LncRNA SNHG15通过miR-30b-3p调控COX6B1轴促进肺腺癌细胞增殖、迁移和侵袭的分子机制[J]. 南方医科大学学报, 2025, 45(7): 1498-1505. |
| [2] | 岳雅清, 牟召霞, 王希波, 刘艳. Aurora-A过表达通过激活NF-κBp65/ARPC4信号轴促进宫颈癌细胞的侵袭和转移[J]. 南方医科大学学报, 2025, 45(4): 837-843. |
| [3] | 庆顺杰, 沈智勇. 过表达己糖激酶2通过激活JAK/STAT途径促进结直肠癌细胞的增殖、迁移和侵袭并调节肿瘤免疫微环境[J]. 南方医科大学学报, 2025, 45(3): 542-553. |
| [4] | 陈镝, 吕莹, 郭怡欣, 张怡荣, 王蕊璇, 周小若, 陈雨欣, 武晓慧. 双氢青蒿素可显著增强阿霉素诱导的三阴性乳腺癌细胞凋亡:基于负向调控STAT3/HIF-1α通路[J]. 南方医科大学学报, 2025, 45(2): 254-260. |
| [5] | 褚乔, 王小娜, 续佳颖, 彭荟林, 赵裕琳, 张静, 陆国玉, 王恺. 白头翁皂苷D通过多靶点和多途径抑制三阴性乳腺癌侵袭转移[J]. 南方医科大学学报, 2025, 45(1): 150-161. |
| [6] | 陈孝华, 鲁辉, 王子良, 王炼, 夏勇生, 耿志军, 张小凤, 宋雪, 王月月, 李静, 胡建国, 左芦根. ABI2在胃癌进展和预后中的作用及其调控机制[J]. 南方医科大学学报, 2024, 44(9): 1653-1661. |
| [7] | 薛良军, 谈秋瑜, 许静文, 冯璐, 李文锦, 颜亮, 李玉磊. MiR-6838-5p过表达下调DDR1基因表达抑制乳腺癌MCF-7细胞的增殖[J]. 南方医科大学学报, 2024, 44(9): 1677-1684. |
| [8] | 欧阳明子, 崔佳琦, 王慧, 梁正, 皮大锦, 陈利国, 陈前军, 吴迎朝. 开心散通过减轻前额叶皮质铁死亡缓解小鼠的阿霉素化疗性抑郁[J]. 南方医科大学学报, 2024, 44(8): 1441-1449. |
| [9] | 房锦存, 刘立威, 林俊豪, 陈逢生. CDHR2过表达通过抑制PI3K/Akt通路抑制乳腺癌细胞增殖[J]. 南方医科大学学报, 2024, 44(6): 1117-1125. |
| [10] | 崔芝, 马萃娇, 王倩茹, 陈金豪, 严子阳, 杨建林, 吕亚丰, 曹春雨. 表达 TGF-βⅡ受体的腺相关病毒载体抑制小鼠三阴性乳腺癌4T1细胞的增殖和肺转移[J]. 南方医科大学学报, 2024, 44(5): 818-826. |
| [11] | 夏勇生, 王炼, 陈孝华, 张雨路, 孙奥飞, 陈德利. 过表达TSR2通过下调PI3K/AKT信号通路抑制胃癌细胞的增殖和侵袭[J]. 南方医科大学学报, 2024, 44(5): 913-919. |
| [12] | 黄秋虎, 周 建, 王子珍, 杨 堃, 陈政纲. miR-26b-3p 靶向 CREB1 调控神经胶质瘤细胞的增殖、迁移及侵袭[J]. 南方医科大学学报, 2024, 44(3): 578-584. |
| [13] | 曾佑琴, 陈思雨, 刘燕, 刘奕彤, 张玲, 夏姣, 吴心语, 魏常友, 冷平. AKBA联合阿霉素抑制三阴性乳腺癌细胞MDA-MB-231的增殖、迁移和裸鼠移植瘤生长[J]. 南方医科大学学报, 2024, 44(12): 2449-2460. |
| [14] | 凌潜龙, 纪凯, 陈金业, 管佳佳, 王睿朋, 满文江, 朱冰. SPHK1靶向NF-κB信号通路调控胃癌细胞的迁移与侵袭[J]. 南方医科大学学报, 2024, 44(11): 2163-2171. |
| [15] | 刘雪柔, 杨玉梅, 刘伟, 张振, 周星琦, 谢文宇, 申林, 张梦晓, 李娴, 臧家兰, 李姗姗. 中药泽漆抑制非小细胞肺癌细胞增殖、侵袭、迁移以及促进细胞凋亡[J]. 南方医科大学学报, 2024, 44(10): 1918-1925. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||