Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (2): 412-422.doi: 10.12122/j.issn.1673-4254.2026.02.19
Mengxia YIN1( ), Ruichen LI1, Kaibo YANG1, Weijie JIAO1,2(
), Ruichen LI1, Kaibo YANG1, Weijie JIAO1,2( ), Xinguang LIU3,4(
), Xinguang LIU3,4( )
)
Received:2025-07-10
Online:2026-02-20
Published:2026-03-10
Contact:
Weijie JIAO, Xinguang LIU
E-mail:ymx6524@163.com;weijie_jiao@hactcm.edu.cn;xgliu2016@126.com
Mengxia YIN, Ruichen LI, Kaibo YANG, Weijie JIAO, Xinguang LIU. Xuebijing Injection in synergy with linezolid alleviates inflammatory injury in mice with MRSA pneumonia by inhibiting the Hla-NLRP3 pathway[J]. Journal of Southern Medical University, 2026, 46(2): 412-422.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.02.19
| Gene | Primer sequences (5'-3') | |
|---|---|---|
| agrA | F | CTCACCTCACACTCCTGCTG |
| R | AGAACCTCACAGAGCGTCAC | |
| RNAIII | F | TTCACTGTGTCGATAATCCA |
| R | GGAAGGAGTGATTTCAATGG | |
| Hla | F | CGCGGATCCGCAGATTCTGATATTAATATTAAAAC |
| R | CCGCTCGAGTTAATTTGTCATTTCTTCTTTTTC | |
| 16sRNA | F | TGGAGCATGTGGTTTAATTCGA |
| R | TGCGGGACTTAACCCAACA | |
| NLRP3 | F | CTCACCTCACACTCCTGCTG |
| R | AGAACCTCACAGAGCGTCAC | |
| ASC | F | CTGCAGATGGACCCCATAGAC |
| R | GTGAGCTCCAAGCCATACCC | |
| Caspase-1 | F | TGACCGAGTGGTTCCCTCAAG |
| R | GACGTGTACGAGTGGGTGTT | |
| GAPDH | F | ACAGCAACAGGGTGGTGGAC |
| R | TTTGAGGGTGCAGCGAACTT |
Tab.1 Primers for RT-qPCR amplification of the target genes
| Gene | Primer sequences (5'-3') | |
|---|---|---|
| agrA | F | CTCACCTCACACTCCTGCTG |
| R | AGAACCTCACAGAGCGTCAC | |
| RNAIII | F | TTCACTGTGTCGATAATCCA |
| R | GGAAGGAGTGATTTCAATGG | |
| Hla | F | CGCGGATCCGCAGATTCTGATATTAATATTAAAAC |
| R | CCGCTCGAGTTAATTTGTCATTTCTTCTTTTTC | |
| 16sRNA | F | TGGAGCATGTGGTTTAATTCGA |
| R | TGCGGGACTTAACCCAACA | |
| NLRP3 | F | CTCACCTCACACTCCTGCTG |
| R | AGAACCTCACAGAGCGTCAC | |
| ASC | F | CTGCAGATGGACCCCATAGAC |
| R | GTGAGCTCCAAGCCATACCC | |
| Caspase-1 | F | TGACCGAGTGGTTCCCTCAAG |
| R | GACGTGTACGAGTGGGTGTT | |
| GAPDH | F | ACAGCAACAGGGTGGTGGAC |
| R | TTTGAGGGTGCAGCGAACTT |
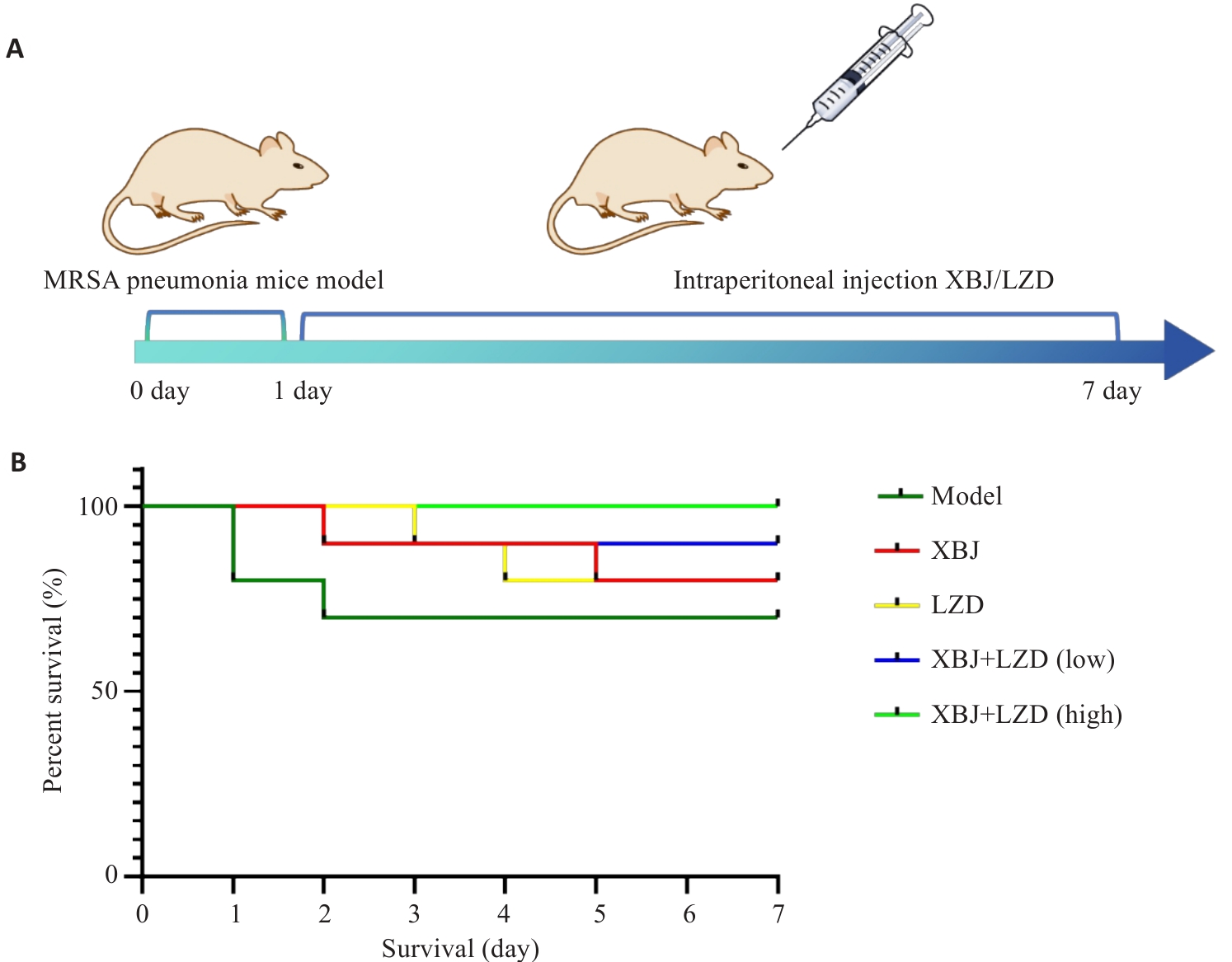
Fig.1 Animal experiment workflow and survival rates of the mice in each group. A: Timeline of MRSA pneumonia mouse modeling and interventions. B: Survival curves of the mice (n=10).
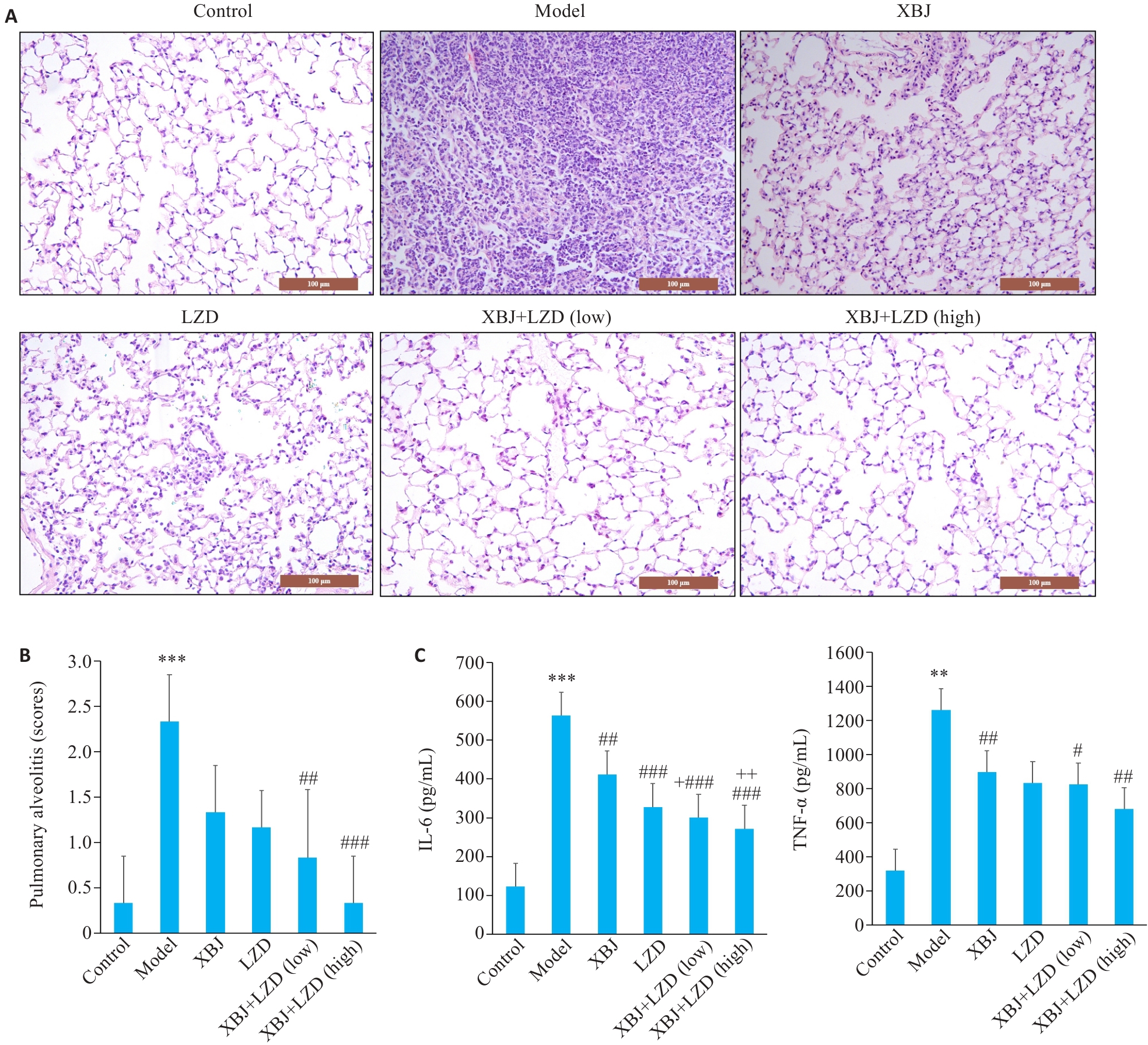
Fig.2 Evaluation of lung injury and expression of lung inflammatory cytokines in each group. A: HE staining of the lung tissues (scale bar=100 μm). B: Lung injury score. C: ELISA analysis of inflammatory cytokines in each group (n=6). **P<0.01,***P<0.001 vs Control; #P<0.05, ##P<0.01, ###P<0.001 vs Model; +P< 0.05,++P<0.01 vs XBJ.
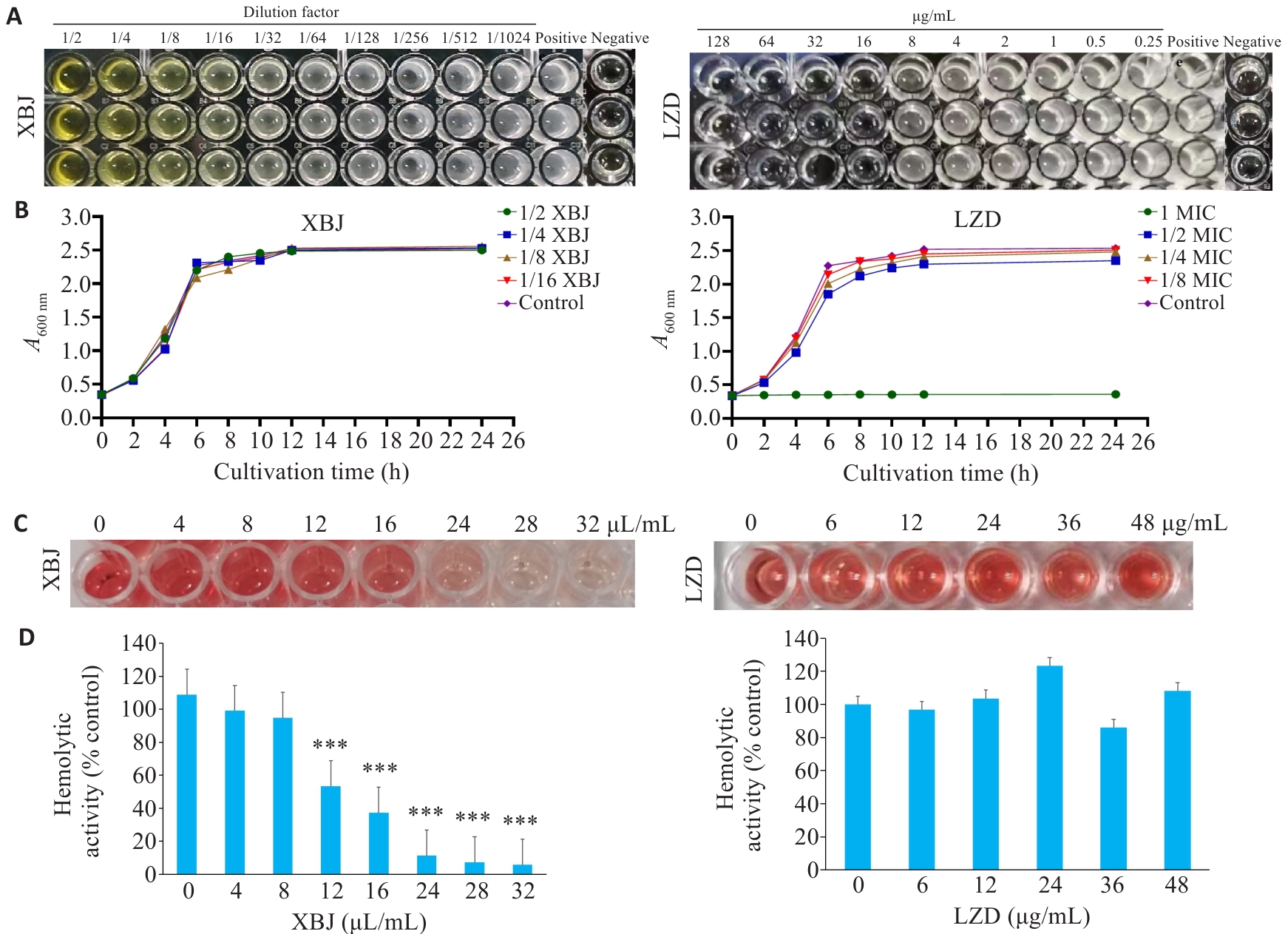
Fig.3 Minimum inhibitory concentrations (MIC) of XBJ and LZD against MRSA and their effects on growth and hemolytic activity of MRSA. A: MIC of XBJ and LZD. B: Growth curves of MRSA treated with XBJ and LZD. C, D: Qualitative analysis of hemolytic activity of MRSA treated with XBJ and LZD (n=3). ***P<0.001 vs 0 μg/mL, 0 μL/mL.
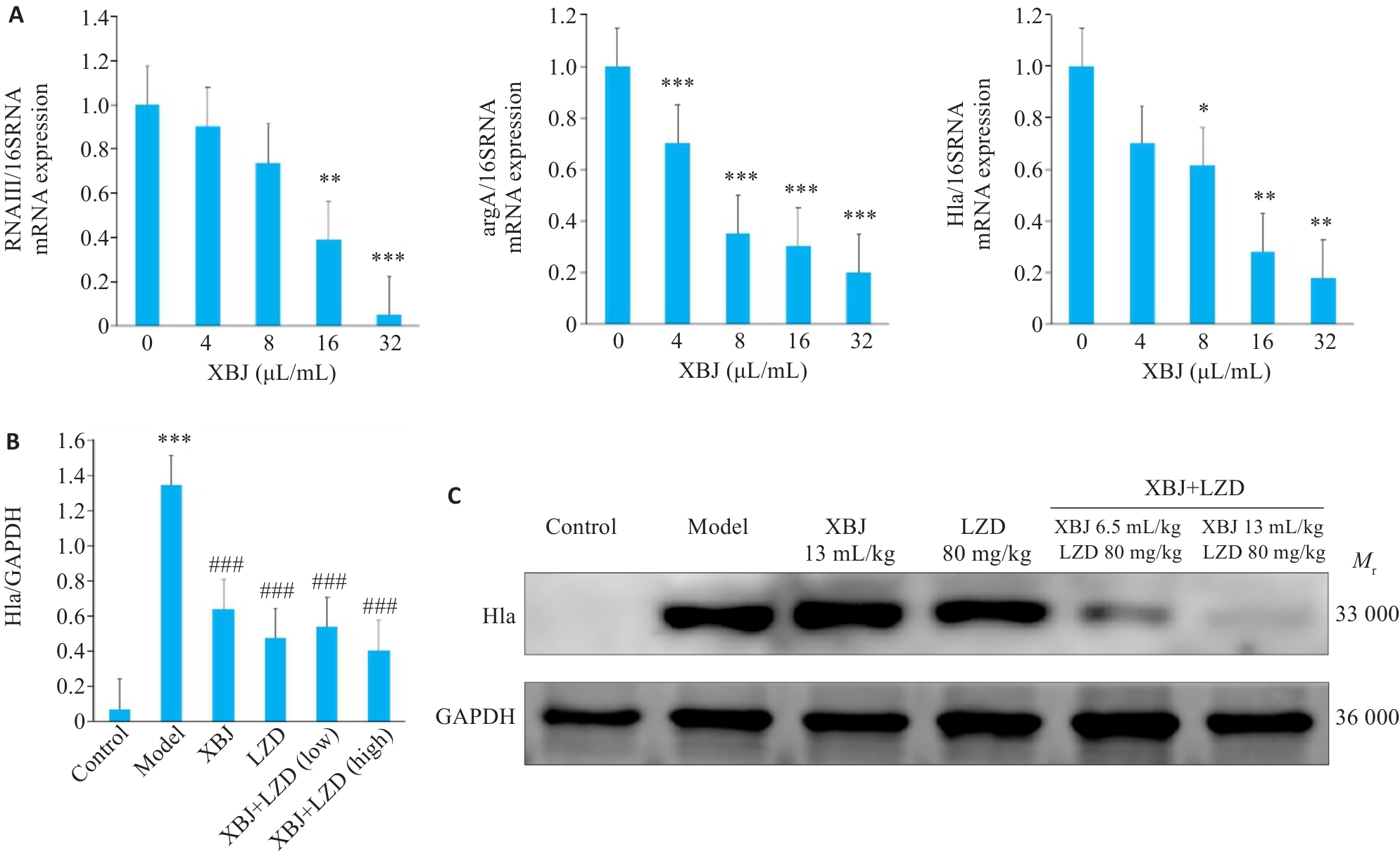
Fig.4 Pulmonary RNAIII, argA and Hla mRNA expressions and Hla protein expression in the mouse models treated with XBJ and LZD. A: qPCR for detecting RNAIII, argA and Hla mRNA in the lung tissues of mice. B: Bar chart of gray values of Hla protein bands in Western blotting. C: Western blots of Hla protein in mouse lung tissues. n=3, *P<0.05, **P<0.01, ***P<0.001 vs 0 μL/mL or Control; ###P<0.001 vs Model.
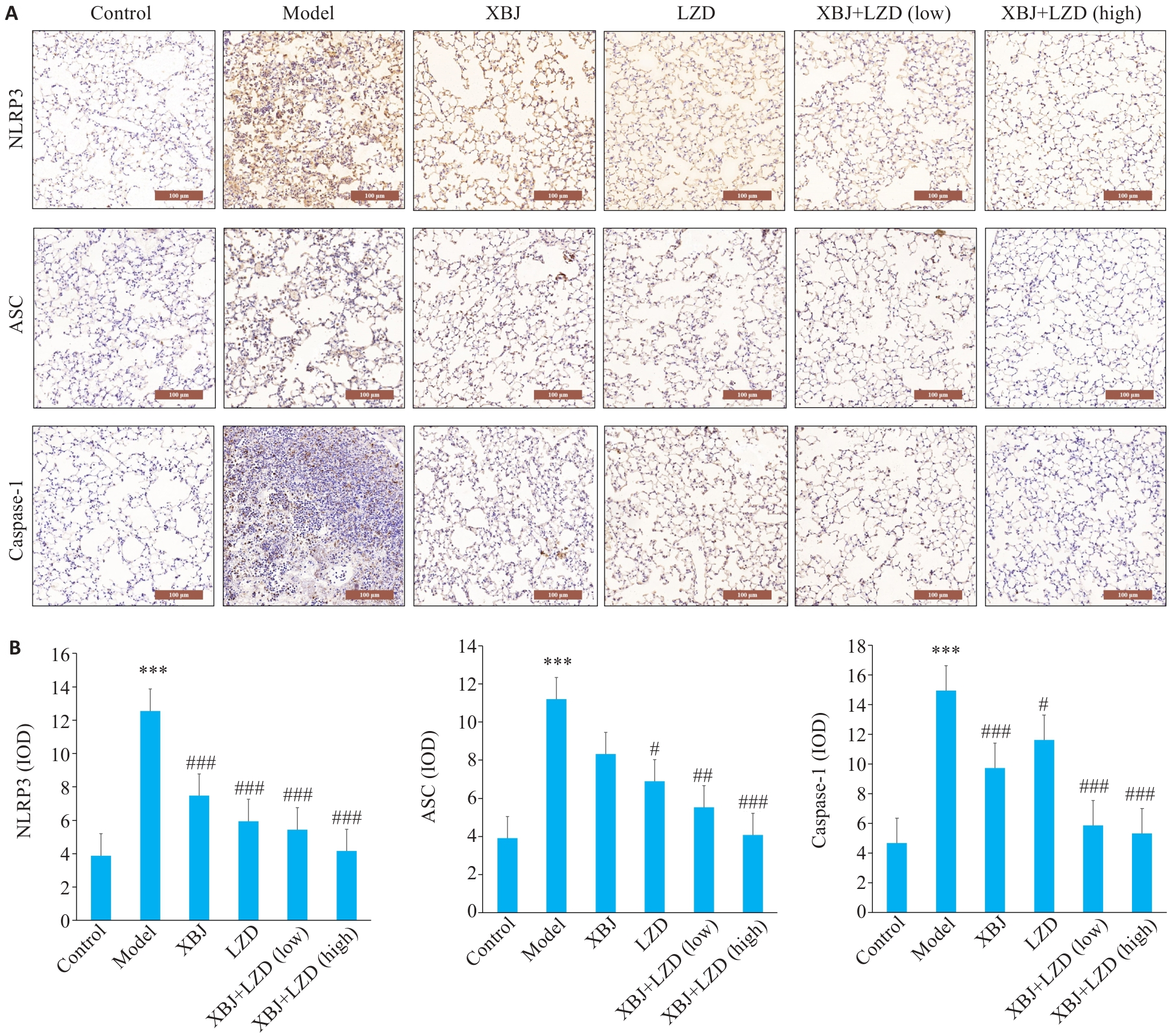
Fig.5 Immunohistochemistry and quantification of NLRP3 inflammasome-related pathway proteins. A: Immunohistochemical staining images of NLRP3, ASC and caspase-1 in mouse lung tissues (Scale bar=100 μm). B: Integrated optical density (IOD) analysis of protein expressions. n=6, ***P<0.001 vs Control; #P<0.05, ##P<0.01, ###P<0.001 vs Model.
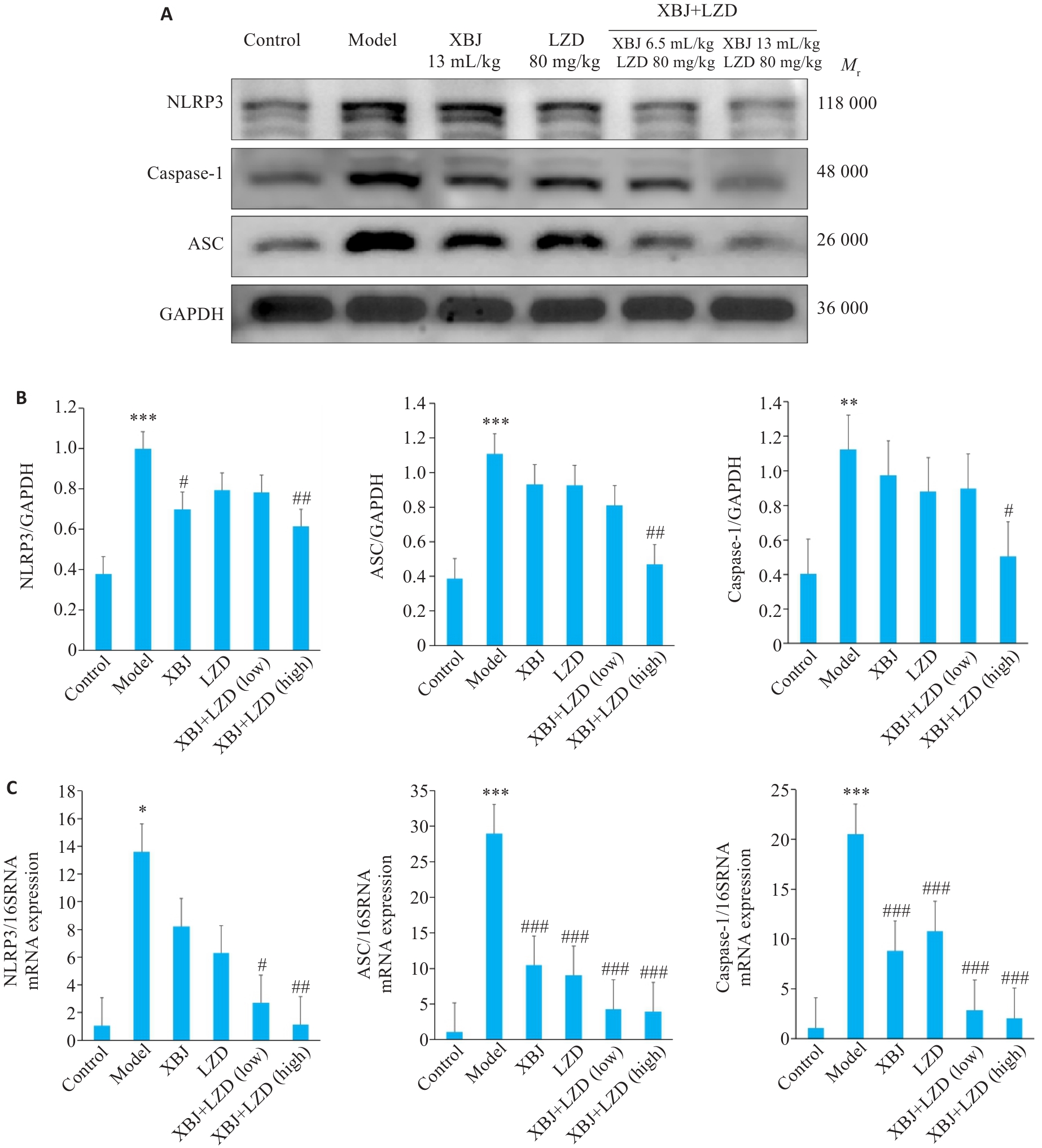
Fig.6 Results of Western blotting and qPCR for detecting expressions of NLEP3, ASC and caspase-1 proteins and mRNAs in mouse lung tissues.. A: Western blotting for detecting NLEP3, ASC and caspase-1 protein expressions in mouse lung tissues. B: Bar chart of gray values of the protein bands. C: qPCR for detecting NLEP3, ASC and caspase-1 mRNA expressions in the lung tissues of the mice. n=3,*P<0.05, **P<0.01,***P<0.001 vs Control; #P<0.05, ##P<0.01, ###P<0.001 vs Model.
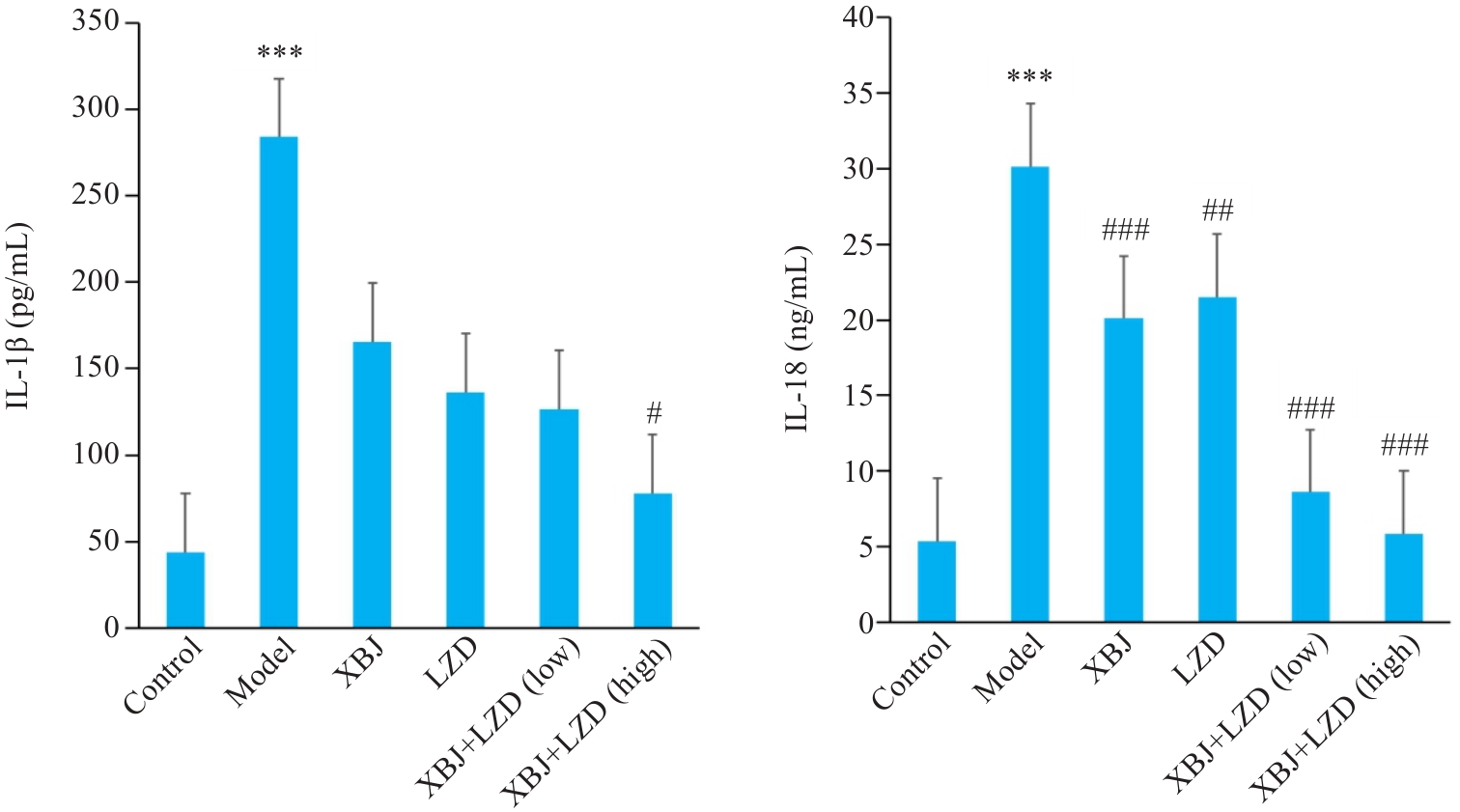
Fig.7 Levels of IL-1β and IL-18 in the lung homogenates of the mice in each group analyzed using ELISA. n=6, ***P<0.001 vs Control; #P<0.05, ##P<0.01, ###P<0.001 vs Model.
| [1] | Keim KC, Horswill AR. Staphylococcus aureus[J]. Trends Microbiol, 2023, 31(12): 1300-1. doi:10.1016/j.tim.2023.07.001 |
| [2] | Hou Z, Liu L, Wei J, et al. Progress in the prevalence, classification and drug resistance mechanisms of methicillin-resistant Staphylococcus aureus [J]. Infect Drug Resist, 2023, 16: 3271-92. doi:10.2147/idr.s412308 |
| [3] | Parker D, Ryan CL, Alonzo F, et al. CD4+ T cells promote the pathogenesis of Staphylococcus aureus pneumonia[J]. J Infect Dis, 2015, 211(5): 835-45. doi:10.1093/infdis/jiu525 |
| [4] | Köck R, Becker K, Cookson B, et al. Methicillin-resistant Staphylococcus aureus (MRSA): burden of disease and control challenges in Europe[J]. Euro Surveill, 2010, 15(41): 19688. doi:10.2807/ese.15.41.19688-en |
| [5] | Shoaib M, Aqib AI, Muzammil I, et al. MRSA compendium of epidemiology, transmission, pathophysiology, treatment, and prevention within one health framework[J]. Front Microbiol, 2022, 13: 1067284. doi:10.3389/fmicb.2022.1067284 |
| [6] | 郭 燕, 胡付品, 朱德妹, 等. 2023年CHINET中国细菌耐药监测[J]. 中国感染与化疗杂志, 2024, 24(6): 627-37. |
| [7] | 张美琦, 王浩嘉, 李艺颖, 等. 血必净注射液对多种病毒的抑制作用及机制研究[J]. 药物评价研究, 2022, 45(9): 1697-705. |
| [8] | 钟南山. 血必净注射液治疗新型冠状病毒感染的肺炎疗效的前瞻性队列研究[M]. 天津:天津红日药业股份有限公司, 2021. |
| [9] | Liu BG, Yuan XL, He DD, et al. Research progress on the oxazolidinone drug linezolid resistance[J]. Eur Rev Med Pharmacol Sci, 2020, 24(18): 9274-81. |
| [10] | Chen H, Du Y, Xia Q, et al. Role of linezolid combination therapy for serious infections: review of the current evidence[J]. Eur J Clin Microbiol Infect Dis, 2020, 39(6): 1043-52. doi:10.1007/s10096-019-03801-x |
| [11] | 杨俊明. 血必净联合利奈唑胺治疗重症肺炎患者的临床疗效[J]. 当代医学, 2022, 28(14): 21-4. |
| [12] | 邱佳男. 血必净联合利奈唑胺对重症肺炎患者血清学指标及细菌清除率的影响[J]. 中国药物经济学, 2021, 16(2): 46-8, 52. |
| [13] | 何晨晗, 师莹莹, 郑天元, 等. 血必净注射液联合利奈唑胺治疗重症肺炎有效性及安全性的Meta分析[J]. 中草药, 2023, 54(14): 4615-22. |
| [14] | Arifin WN, Zahiruddin WM. Sample size calculation in animal studies using resource equation approach[J]. Malays J Med Sci, 2017, 24(5): 101-5. doi:10.21315/mjms2017.24.5.11 |
| [15] | Matute-Bello G, Downey G, Moore BB, et al. An official American Thoracic Society workshop report: features and measurements of experimental acute lung injury in animals[J]. Am J Respir Cell Mol Biol, 2011, 44(5): 725-38. doi:10.1165/rcmb.2009-0210st |
| [16] | 胡宇华, 赵欣彤, 李天磊, 等. 噁唑烷酮类抗菌药物研究进展[J]. 药学学报, 2022, 57(11): 3276-91. |
| [17] | 柴明思. 利奈唑胺与氨基糖苷类抗生素对耐甲氧西林抗药性金黄色葡萄球菌肺炎的疗效比较[J]. 吉林医学, 2022, 43(9): 2426-9. |
| [18] | 王 峰, 李建生, 王守富, 等. 肺炎克雷伯杆菌肺炎多器官功能障碍老龄大鼠模型的建立[J]. 中国中医药现代远程教育, 2008, (4): 317-9. |
| [19] | 广承灵, 张笑恺, 王 于, 等. 金黄色葡萄球菌感染非抗生素治疗手段研究进展[J]. 中国抗生素杂志, 2025, 50(1): 15-21. |
| [20] | Wächter H, Yörük E, Becker K, et al. Correlations of host and bacterial characteristics with clinical parameters and survival in Staphylococcus aureus bacteremia[J]. J Clin Med, 2021, 10(7): 1371. doi:10.3390/jcm10071371 |
| [21] | Surewaard BGJ, Thanabalasuriar A, Zeng Z, et al. α-toxin induces platelet aggregation and liver injury during Staphylococcus aureus sepsis[J]. Cell Host Microbe, 2018, 24(2): 271-84.e3. doi:10.1016/j.chom.2018.06.017 |
| [22] | Du Y, Liu L, Zhang C, et al. Two residues in Staphylococcus aureus α-hemolysin related to hemolysis and self-assembly[J]. Infect Drug Resist, 2018, 11: 1271-4. doi:10.2147/idr.s167779 |
| [23] | 李 詹, 谈 玲, 丁曼琳, 等. 川乌水提取物及其单体成分对金黄色葡萄球菌α-溶血素分泌的抑制作用[J]. 中国药业, 2024, 33(13): 51-5. |
| [24] | Greenberg M, Kuo D, Jankowsky E, et al. Small-molecule AgrA inhibitors F12 and F19 act as antivirulence agents against Gram-positive pathogens[J]. Sci Rep, 2018, 8(1): 14578. doi:10.1038/s41598-018-32829-w |
| [25] | Ragle BE, Karginov VA, Bubeck Wardenburg J. Prevention and treatment of Staphylococcus aureus pneumonia with a beta-cyclodextrin derivative[J]. Antimicrob Agents Chemother, 2010, 54(1): 298-304. doi:10.1128/aac.00973-09 |
| [26] | McGilligan VE, Gregory-Ksander MS, Li D, et al. Staphylococcus aureus activates the NLRP3 inflammasome in human and rat conjunctival goblet cells[J]. PLoS One, 2013, 8(9): e74010. doi:10.1371/journal.pone.0074010 |
| [27] | Peters XQ, Omolo CA, Govender T. Paving the way for NLRP3 inflammasome inactivation using ADAM10 and alpha-hemolysin: an In silico investigation[J]. ACS Omega, 2025, 10(8): 7981-8. doi:10.1021/acsomega.4c09015 |
| [28] | Ren M, Chen J, Xu H, et al. Ergolide covalently binds NLRP3 and inhibits NLRP3 inflammasome-mediated pyroptosis[J]. Int Immunopharmacol, 2023, 120: 110292. doi:10.1016/j.intimp.2023.110292 |
| [29] | Kebaier C, Chamberland RR, Allen IC, et al. Staphylococcus aureus α‑hemolysin mediates virulence in a murine model of severe pneumonia through activation of the NLRP3 inflammasome[J]. J Infect Dis, 2012, 205(5): 807-17. doi:10.1093/infdis/jir846 |
| [30] | Muñoz-Planillo R, Franchi L, Miller LS, et al. A critical role for hemolysins and bacterial lipoproteins in Staphylococcus aureus-induced activation of the Nlrp3 inflammasome[J]. J Immunol, 2009, 183(6): 3942-8. doi:10.4049/jimmunol.0900729 |
| [31] | Lepak AJ, Marchillo K, Pichereau S, et al. Comparative pharmacodynamics of the new oxazolidinone tedizolid phosphate and linezolid in a neutropenic murine Staphylococcus aureus pneumonia model[J]. Antimicrob Agents Chemother, 2012, 56(11): 5916-22. doi:10.1128/aac.01303-12 |
| [32] | Shekhar A, Di Lucrezia R, Jerye K, et al. Highly potent quino-xalinediones inhibit α‑hemolysin and ameliorate Staphylococcus aureus lung infections[J]. Cell Host Microbe, 2025, 33(4): 560-72.e21. doi:10.1016/j.chom.2025.03.006 |
| [1] | Chenfei LIU, Wei ZHANG, Yao ZENG, Yan LIANG, Mengting WANG, Mingfang ZHANG, Xinyuan LI, Fengchao WANG, Yanqing YANG. 2,6-dimethoxy-1,4-benzoquinone alleviates dextran sulfate sodium-induced ulcerative colitis in mice by suppressing NLRP3 inflammasome activation [J]. Journal of Southern Medical University, 2025, 45(8): 1654-1662. |
| [2] | Feifei SHANG, Xiaoke SHI, Yao ZENG, Xunqian TAO, Tianzhen LI, Yan LIANG, Yanqin YANG, Chuanwang SONG. Avitinib suppresses NLRP3 inflammasome activation and ameliorates septic shock in mice [J]. Journal of Southern Medical University, 2025, 45(8): 1697-1705. |
| [3] | Guoyong LI, Renling LI, Yiting LIU, Hongxia KE, Jing LI, Xinhua WANG. Therapeutic mechanism of Arctium lappa extract for post-viral pneumonia pulmonary fibrosis: a metabolomics, network pharmacology analysis and experimental verification [J]. Journal of Southern Medical University, 2025, 45(6): 1185-1199. |
| [4] | Zhengwang ZHU, Linlin WANG, Jinghan ZHAO, Ruixue MA, Yuchun YU, Qingchun CAI, Bing WANG, Pingsheng ZHU, Mingsan MIAO. Tuihuang Mixture improves α‑naphthylisothiocyanate-induced cholestasis in rats by inhibiting NLRP3 inflammasomes via regulating farnesoid X receptor [J]. Journal of Southern Medical University, 2025, 45(4): 718-724. |
| [5] | Zehan LI, Meng LIANG, Gencheng HAN, Xuewu ZHANG. Therapeutic effects of inulin-type oligosaccharides of Morinda officinalis on Streptococcus pneumoniae meningitis in mice [J]. Journal of Southern Medical University, 2025, 45(3): 577-586. |
| [6] | Jingyu CHEN, Jinhu ZOU, Bingliang ZHOU, Xuefeng GAO, Pengwei HUANG, Hong CAO. Indole-3-acetic acid alleviates Cryptococcus neoformans-induced pyroptosis in cerebral microvascular endothelial cells by regulating stress granule-mediated NLRP3 inflammasome activation [J]. Journal of Southern Medical University, 2025, 45(12): 2679-2689. |
| [7] | Mingyuan LI, Wei ZHANG, Mengqing HUA. Bardoxolone methyl alleviates acute liver injury in mice by inhibiting NLRP3 inflammasome activation [J]. Journal of Southern Medical University, 2024, 44(9): 1662-1669. |
| [8] | Wei ZHANG, Mengmeng DENG, Yao ZENG, Chenfei LIU, Feifei SHANG, Wenhao XU, Haoyi JIANG, Fengchao WANG, Yanqing YANG. 2,6-dimethoxy-1,4-benzoquinone alleviates septic shock in mice by inhibiting NLRP3 inflammasome activation [J]. Journal of Southern Medical University, 2024, 44(6): 1024-1032. |
| [9] | WANG Hui, JIANG Xiaoyu, LI Feiyu. Construction and characterization of a modA gene mutant strain of Klebsiella pneumoniae [J]. Journal of Southern Medical University, 2024, 44(4): 748-756. |
| [10] | XIE Jichen, MA Renhui, LI Moran, LI Bei, XIONG Lina. Effect of intestinal nitrate on growth of Klebsiella pneumoniae and its regulatory mechanism [J]. Journal of Southern Medical University, 2024, 44(4): 757-764. |
| [11] | FANG Shangping, SUN Renke, SU Hui, ZHAI Kecheng, XIANG Yu, GAO Yangmengna, GUO Wenjun. Chlorogenic acid alleviates acute kidney injury in septic mice by inhibiting NLRP3 inflammasomes and the caspase-1 canonical pyroptosis pathway [J]. Journal of Southern Medical University, 2024, 44(2): 317-323. |
| [12] | LING Xuguang, XU Wenwen, PANG Guanlai, HONG Xuxing, LIU Fengqin, LI Yang. Tea polyphenols ameliorates acute lung injury in septic mice by inhibiting NLRP3 inflammasomes [J]. Journal of Southern Medical University, 2024, 44(2): 381-386. |
| [13] | LIU Yunze, LI Chengrun, GUO Juntang, LIU Yang. A clinical-radiomics nomogram for differentiating focal organizing pneumonia and lung adenocarcinoma [J]. Journal of Southern Medical University, 2024, 44(2): 397-404. |
| [14] | Xiaoling SU, Daoyong LIAO, Chao LI, Li CHEN, Jingyun WANG, Tian GAN, Haodang LUO, Ning WU, Jun HE. Protective effect of Streptococcus salivarius K12 against Mycoplasma pneumoniae infection in mice [J]. Journal of Southern Medical University, 2024, 44(12): 2300-2307. |
| [15] | CAO Hairuo, ZHANG Wei, LI Mingyuan, YANG Yanqing, LI Yuyun. Isodopharicin C inhibits NLRP3 inflammasome activation and alleviates septic shock in mice [J]. Journal of Southern Medical University, 2023, 43(9): 1476-1484. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||