Journal of Southern Medical University ›› 2025, Vol. 45 ›› Issue (7): 1353-1362.doi: 10.12122/j.issn.1673-4254.2025.07.01
Xiaoyu CHANG1( ), Hanwen ZHANG2(
), Hanwen ZHANG2( ), Hongting CAO2, Ling HOU2, Xin MENG1, Hong TAO2, Yan LUO3(
), Hongting CAO2, Ling HOU2, Xin MENG1, Hong TAO2, Yan LUO3( ), Guanghua LI1,2(
), Guanghua LI1,2( )
)
Received:2024-06-21
Accepted:2025-04-25
Online:2025-07-20
Published:2025-07-17
Contact:
Yan LUO, Guanghua LI
E-mail:xiaoxyyu@163.com;1912118742@qq.com;wyzxsh@hotmail.com;ghlee0404@163.com
About author:First author contact:CHANG Xiaoyu and ZHANG Hanwen contributed equally to this work
Supported by:Xiaoyu CHANG, Hanwen ZHANG, Hongting CAO, Ling HOU, Xin MENG, Hong TAO, Yan LUO, Guanghua LI. Heat stress affects expression levels of circadian clock gene Bmal1 and cyclins in rat thoracic aortic endothelial cells[J]. Journal of Southern Medical University, 2025, 45(7): 1353-1362.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2025.07.01
| Gene | Sequence (5'-3') | Length (bp) |
|---|---|---|
| Bmal1 | 108 | |
| Forward | ACCCATACACAGAAGCAAACTACA | |
| Reverse | GGTCACATCCTACGACAAACAAA | |
| 140 | ||
| Forward | CCTAGACTTCGAGCAAGAGA | |
| Reverse | GGAAGGAAGGCTGGAAGA |
Tab.1 Primer sequences and the expected product sizes
| Gene | Sequence (5'-3') | Length (bp) |
|---|---|---|
| Bmal1 | 108 | |
| Forward | ACCCATACACAGAAGCAAACTACA | |
| Reverse | GGTCACATCCTACGACAAACAAA | |
| 140 | ||
| Forward | CCTAGACTTCGAGCAAGAGA | |
| Reverse | GGAAGGAAGGCTGGAAGA |
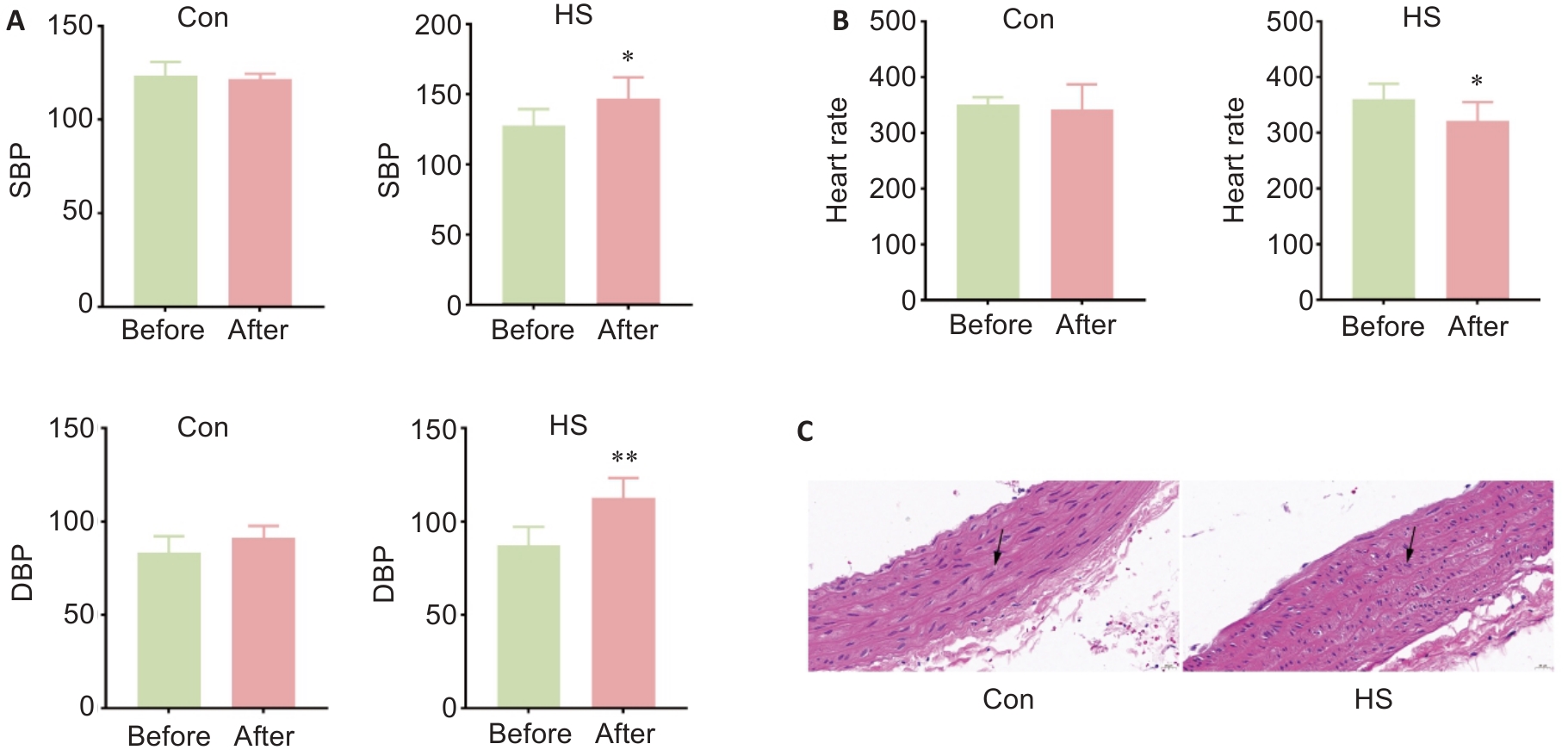
Fig.1 Effect of heat stress on blood pressure, heart rate and structure of the thoracic aorta in rats. A: Blood pressure of the rats before and after heat stress. B: Heart rate of the rats before and after heat stress. C: HE staining of the thoracic aorta of the rats (Original magnification:×40). Data are presented as Mean±SD from 8 independent measurements. *P<0.05, **P<0.01 vs control (Con). SBP: Systolic blood pressure; DBP: Diastolic blood pressure; HS: Heat stress.
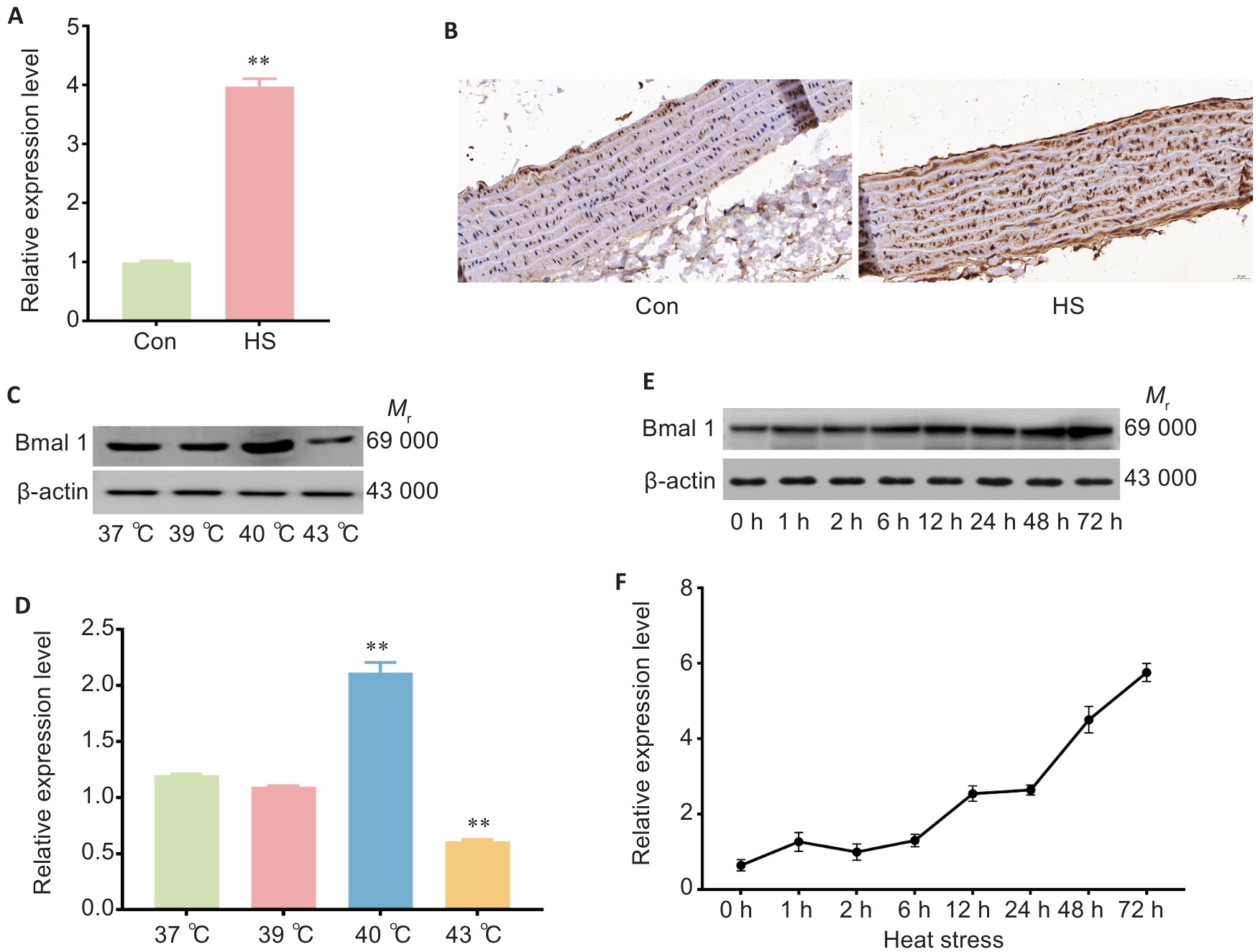
Fig.2 Heat stress increases Bmal1 expression in rat thoracic aorta and rat thoracic aortic endothelial cells (RTAECs). A: Bmal1 mRNA expression levels of in rat thoracic aorta. B: Immunohistochemical detection of Bmal1 protein in rat thoracic aorta (×40). C, D: Western blotting and quantitative analysis of Bmal1 protein expression in RTAECs with heat stress at different temperatures. E, F: Western blotting and quantitative analysis of Bmal1 protein expression in RTAECs with heat stress of different lengths. Data are presented as Mean±SD from 3 independent experiments. *P<0.05, **P<0.01 vs Con.
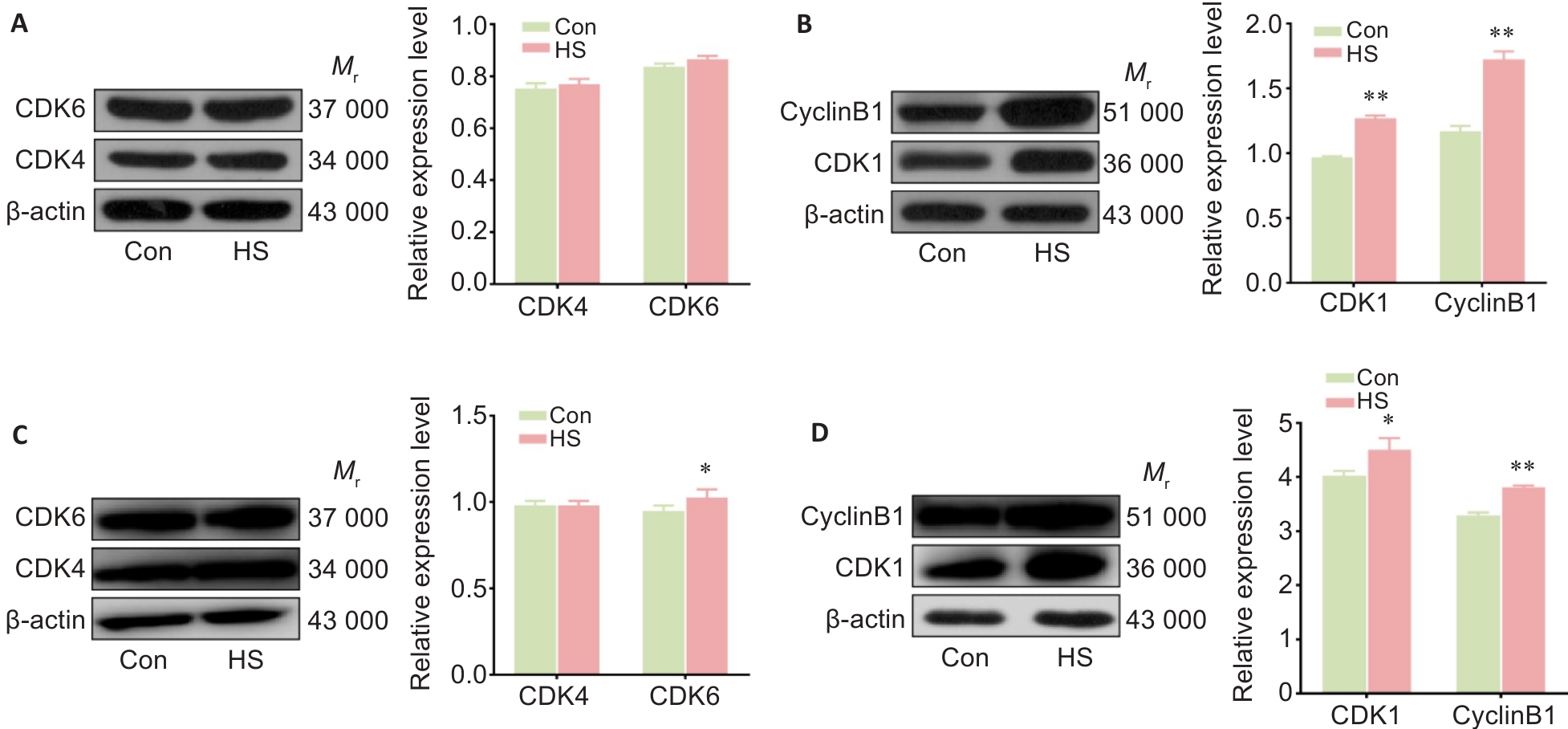
Fig.3 Heat stress causes changes in cyclin expression and cell cycle progression in rat thoracic aorta and RTAECs. A, B: Western blotting for detecting the expressions of CDK4, CDK6, cyclin B1 and CDK1 proteins in rat thoracic aorta. C, D: Western blotting for detecting CDK4, CDK6, cyclin B1 and CDK1 proteins in RTAECs. Data are presented as Mean±SD from 3 independent experiments. *P<0.05, **P<0.01 vs Con.
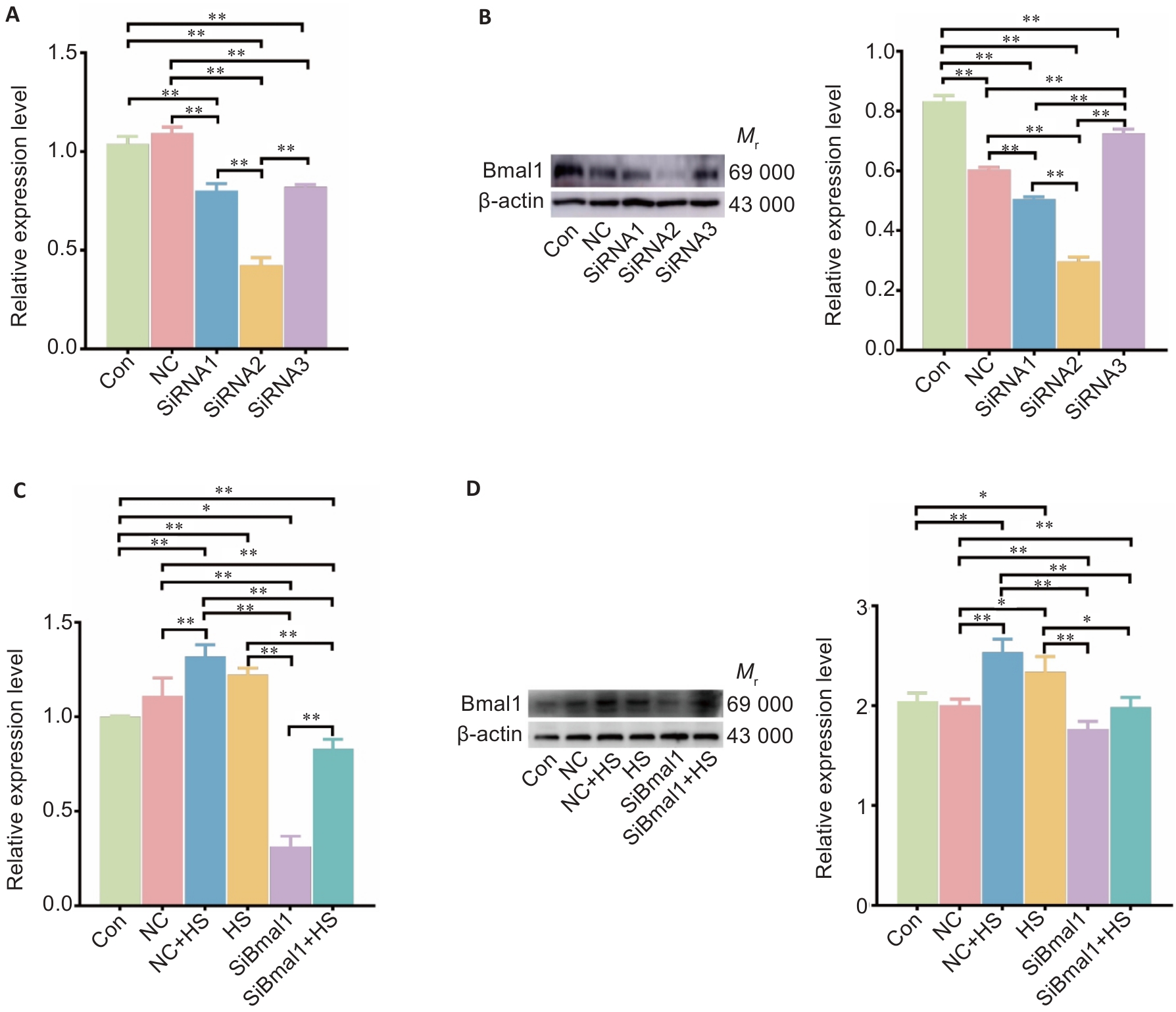
Fig.4 Efficiency of the siRNA constructs for Bmal1 knockdown and effect of siRNA2 on mRNA and protein Bmal1 expressions in RTAECs. A: Bmal1 mRNA expression levels in RTAECs transfected with different si-Bmal1 constructs. B: Western blotting for detecting Bmal1 protein expressions in the transfected cells. C: Bmal1 mRNA expression levels in RTAECs with heat stress, transfection with si-Bmal1, or both. D: Western blotting for detecting Bmal1 protein expression in different treatment groups. Data are presented as Mean±SD from 3 independent experiments. *P<0.05, **P<0.01. NC: Negative control.
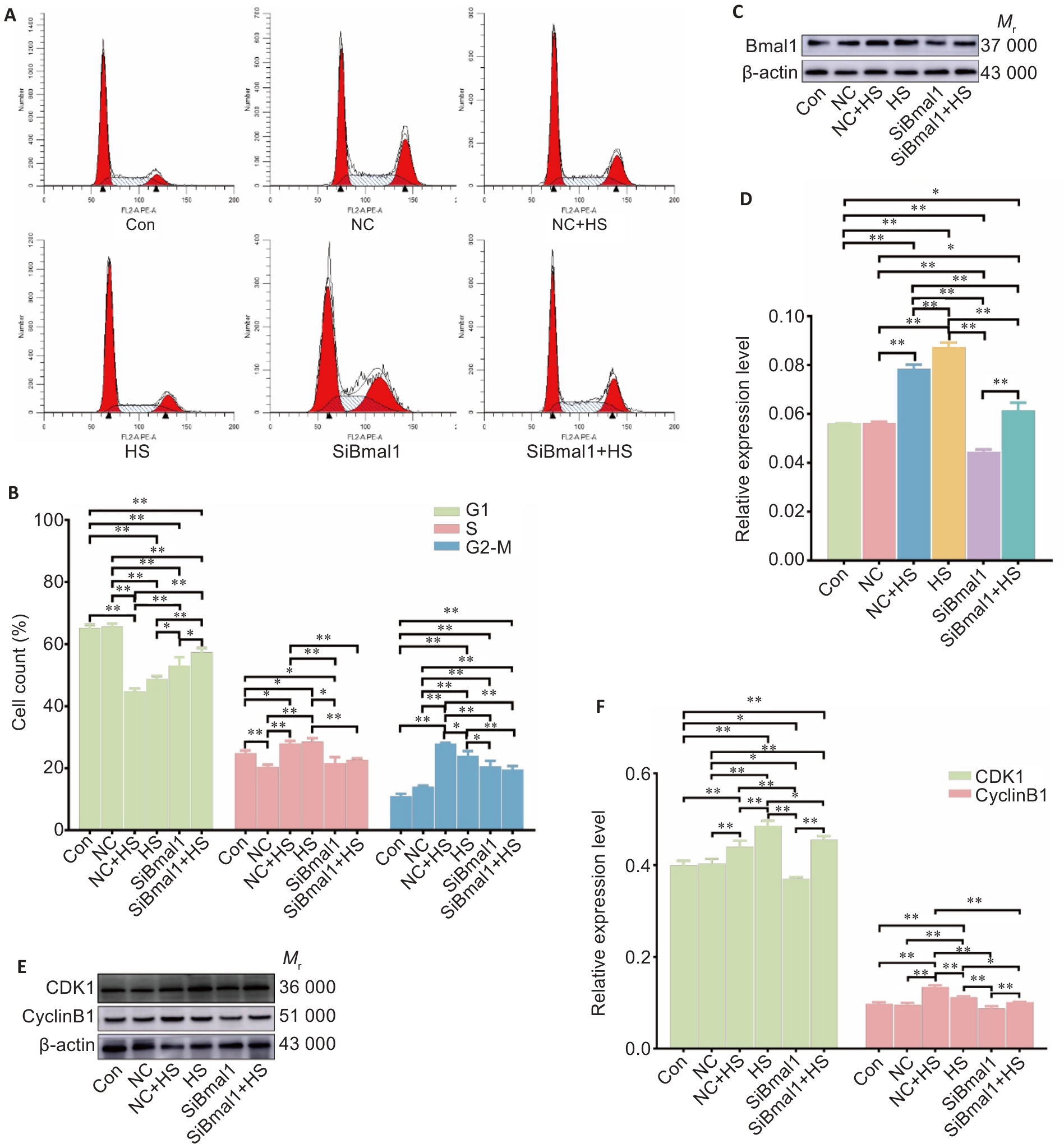
Fig.5 Bmal1 knockdown reverses heat stress-induced changes in cyclin expression and cell cycle in RTAECs. A, B: Cell cycle distribution in treated RTAECs. C-F: Western blotting for detecting CDK6, CDK1 and cyclin B1 protein expressions in the treated cells. Data are presented as Mean±SD from 3 independent experiments. *P<0.05, **P<0.01.
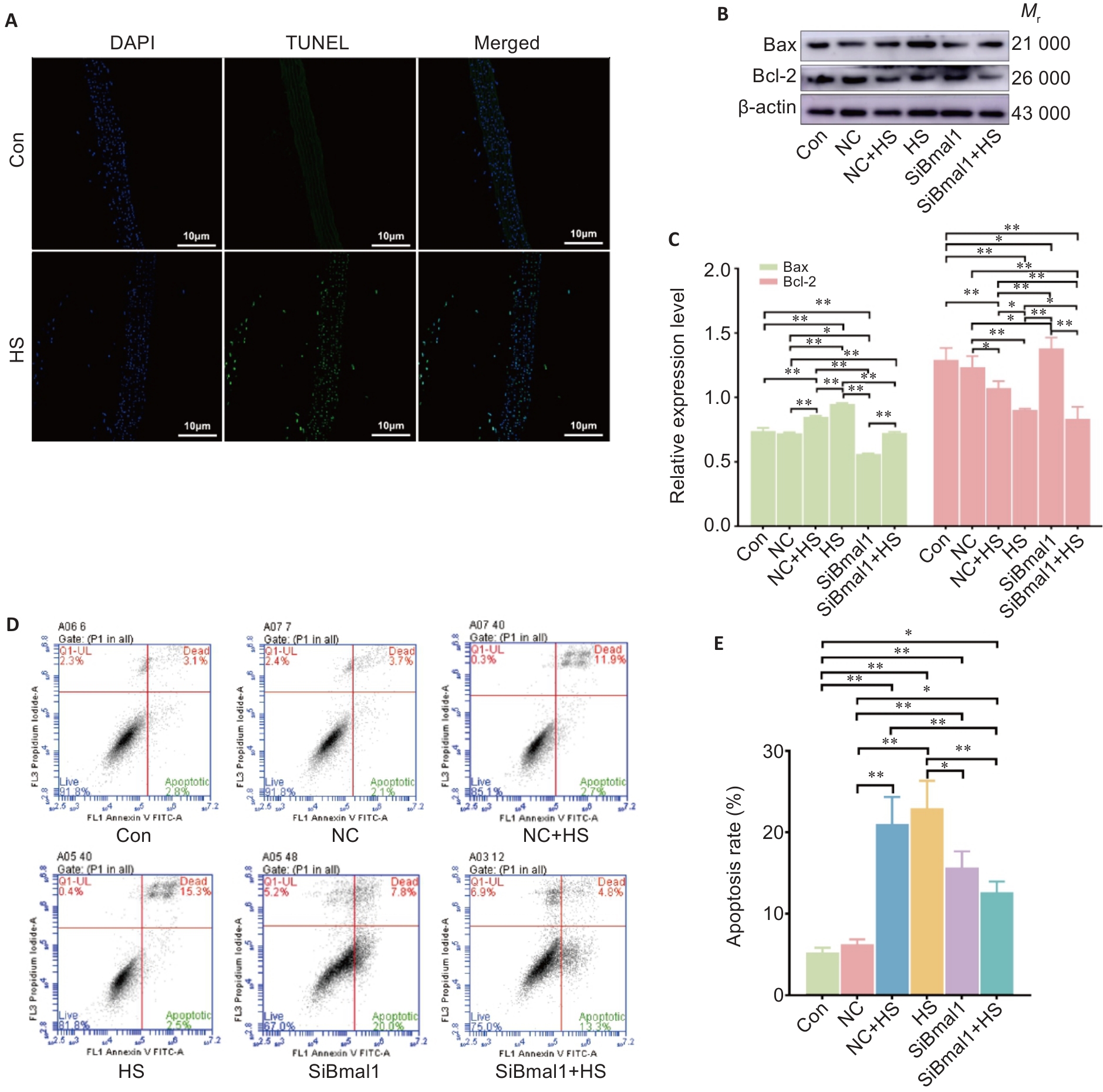
Fig.6 Bmal1 knockdown suppresses heat stress-induced apoptosis in RTAECs. A: TUNEL assay for assessing apoptosis in rat thoracic aorta. B, C: Western blotting for detecting Bax and Bcl-2 protein expressions in RTAECs. D, E: Flow cytometric analysis of apoptosis of RTAECs in different groups. Data are presented as Mean±SD from 3 independent experiments. *P<0.05, **P<0.01.
| [1] | Liu J, Varghese BM, Hansen A, et al. Heat exposure and cardiovascular health outcomes: a systematic review and meta-analysis [J]. Lancet Planet Health, 2022, 6(6): e484-95. doi:10.1016/s2542-5196(22)00117-6 |
| [2] | Tang LP, Li WH, Liu YL, et al. Heat stress aggravates intestinal inflammation through TLR4-NF-κB signaling pathway in Ma chickens infected with Escherichia coli O157:H7 [J]. Poult Sci, 2021, 100(5): 101030. doi:10.1016/j.psj.2021.101030 |
| [3] | Stern N, Sowers JR, McGinty D, et al. Circadian rhythm of plasma renin activity in older normal and essential hypertensive men: relation with inactive renin, aldosterone, cortisol and REM sleep[J]. J Hypertens, 1986, 4(5): 543-50. doi:10.1097/00004872-198610000-00005 |
| [4] | Minamisawa M, Izawa A, Motoki H, et al. Prognostic significance of neuroadrenergic dysfunction for cardiovascular events in patients with acute myocardial infarction [J]. Circ J, 2015, 79(10): 2238-2245. doi:10.1253/circj.cj-15-0265 |
| [5] | Welz PS, Zinna VM, Symeonidi A, et al. BMAL1-driven tissue clocks respond independently to light to maintain homeostasis[J]. Cell, 2019, 177(6): 1436-47. e12. doi:10.1016/j.cell.2019.05.009 |
| [6] | Xie M, Tang Q, Nie J, et al. BMAL1-downregulation aggravates Porphyromonas gingivalis-induced atherosclerosis by encouraging oxidative stress [J]. Circ Res, 2020, 126(6): e15-29. doi:10.1161/circresaha.119.315502 |
| [7] | Takeda N, Maemura K. Circadian clock and vascular disease [J]. Hypertens Res, 2010, 33(7): 645-51. doi:10.1038/hr.2010.68 |
| [8] | Elliott WJ. Circadian variation in the timing of stroke onset: a meta-analysis [J]. Stroke, 1998, 29(5): 992-6. doi:10.1161/01.str.29.5.992 |
| [9] | Cannon CP, McCabe CH, Stone PH, et al. Circadian variation in the onset of unstable angina and non-Q-wave acute myocardial infarction (the TIMI III Registry and TIMI IIIB) [J]. Am J Cardiol, 1997, 79(3): 253-8. doi:10.1016/s0002-9149(97)00743-1 |
| [10] | Liu L, Michowski W, Kolodziejczyk A, et al. The cell cycle in stem cell proliferation, pluripotency and differentiation [J]. Nat Cell Biol, 2019, 21(9): 1060-7. doi:10.1038/s41556-019-0384-4 |
| [11] | Tokunaga Y, Otsuyama KI, Hayashida N. Cell cycle regulation by heat shock transcription factors [J]. Cells, 2022, 11(2): 203. doi:10.3390/cells11020203 |
| [12] | Rotinen M. "Defining the independence of the liver circadian clock" & "BMAL1-driven tissue clocks respond independently to light to maintain homeostasis" [J]. Front Neurosci, 2020, 14: 107. doi:10.3389/fnins.2020.00107 |
| [13] | Gallego M, Virshup DM. Post-translational modifications regulate the ticking of the circadian clock [J]. Nat Rev Mol Cell Biol, 2007, 8(2): 139-48. doi:10.1038/nrm2106 |
| [14] | Stojkovic K, Wing SS, Cermakian N. A central role for ubiquitination within a circadian clock protein modification code [J]. Front Mol Neurosci, 2014, 7: 69. doi:10.3389/fnmol.2014.00069 |
| [15] | Trott AJ, Menet JS. Regulation of circadian clock transcriptional output by CLOCK:BMAL1 [J]. PLoS Genet, 2018, 14(1): e1007156. doi:10.1371/journal.pgen.1007156 |
| [16] | Balsalobre A, Damiola F, Schibler U. A serum shock induces circadian gene expression in mammalian tissue culture cells [J]. Cell, 1998, 93(6): 929-37. doi:10.1016/s0092-8674(00)81199-x |
| [17] | Maemura K, Layne MD, Watanabe M, et al. Molecular mechanisms of morning onset of myocardial infarction [J]. Ann N Y Acad Sci, 2001, 947: 398-402. doi:10.1111/j.1749-6632.2001.tb03972.x |
| [18] | McNamara P, Seo SB, Rudic RD, et al. Regulation of CLOCK and MOP4 by nuclear hormone receptors in the vasculature: a humoral mechanism to reset a peripheral clock [J]. Cell, 2001, 105(7): 877-89. doi:10.1016/s0092-8674(01)00401-9 |
| [19] | Nonaka H, Emoto N, Ikeda K, et al. Angiotensin II induces circadian gene expression of clock genes in cultured vascular smooth muscle cells [J]. Circulation, 2001, 104(15):1746-8. doi:10.1161/hc4001.098048 |
| [20] | Bray MS, Young ME. Diurnal variations in myocardial metabolism [J]. Cardiovasc. Res., 2008, 79(2): 228-37. doi:10.1093/cvr/cvn054 |
| [21] | Deng W, Zhu S, Zeng L, et al. The circadian clock controls immune checkpoint pathway in sepsis [J]. Cell Rep, 2018, 24(2):366-78. doi:10.1016/j.celrep.2018.06.026 |
| [22] | Young ME, Brewer RA, Peliciari-Garcia RA, et al. Cardiomyocyte-specific BMAL1 plays critical roles in metabolism, signaling, and maintenance of contractile function of the heart [J]. J Biol Rhythms, 2014, 29(4): 257-76. doi:10.1177/0748730414543141 |
| [23] | Morris CJ, Purvis TE, Hu K, et al. Circadian misalignment increases cardiovascular disease risk factors in humans [J]. Proc Natl Acad Sci U S A, 2016, 113(10):E1402-11. doi:10.1073/pnas.1516953113 |
| [24] | Anea CB, Zhang M, Stepp DW, et al. Vascular disease in mice with a dysfunctional circadian clock [J]. Circulation, 2009, 119(11): 1510-7. doi:10.1161/circulationaha.108.827477 |
| [25] | Kovanen L, Donner K, Kaunisto M, et al. CRY1, CRY2 and PRKCDBP genetic variants in metabolic syndrome [J]. Hypertens Res, 2015, 38(3): 186-92. doi:10.1038/hr.2014.157 |
| [26] | Anea CB, Ali MI, Osmond JM, et al. Matrix metalloproteinase 2 and 9 dysfunction underlie vascular stiffness in circadian clock mutant mice [J]. Arterioscler Thromb Vasc Biol, 2010, 30(12): 2535-43. doi:10.1161/atvbaha.110.214379 |
| [27] | Pan X, Bradfield CA, Hussain MM. Global and hepatocyte-specific ablation of Bmal1 induces hyperlipidaemia and enhances atherosclerosis [J]. Nat Commun, 2016, 7: 13011. doi:10.1038/ncomms13011 |
| [28] | Hsu PY, Harmer SL. Wheels within wheels: the plant circadian system [J]. Trends Plant Sci, 2014, 19(4): 240-9. doi:10.1016/j.tplants.2013.11.007 |
| [29] | Wenden B, Kozma-Bognár L, Edwards KD, et al. Light inputs shape the Arabidopsis circadian system [J]. Plant J, 2011, 66(3): 480-91. doi:10.1111/j.1365-313x.2011.04505.x |
| [30] | Ki Y, Ri H, Lee H, et al. Warming up your tick-tock: temperature-dependent regulation of circadian clocks [J]. Neuroscientist, 2015, 21(5): 503-18. doi:10.1177/1073858415577083 |
| [31] | Mwimba M, Karapetyan S, Liu L, et al. Daily humidity oscillation regulates the circadian clock to influence plant physiology [J]. Nat Commun, 2018, 9(1): 4290. doi:10.1038/s41467-018-06692-2 |
| [32] | Buhr ED, Yoo SH, Takahashi JS. Temperature as a universal resetting cue for mammalian circadian oscillators [J]. Science, 2010, 330(6002): 379-85. doi:10.1126/science.1195262 |
| [33] | Terman JS, Remé CE, Terman M. Rod outer segment disk shedding in rats with lesions of the suprachiasmatic nucleus [J]. Brain Res, 1993, 605(2): 256-64. doi:10.1016/0006-8993(93)91748-h |
| [34] | Collis SJ, Boulton SJ. Emerging links between the biological clock and the DNA damage response [J]. Chromosoma, 2007, 116(4): 331-9. doi:10.1007/s00412-007-0108-6 |
| [35] | Gavet O, Pines J. Progressive activation of CyclinB1-Cdk1 coordinates entry to mitosis [J]. Dev Cell, 2010, 18(4): 533-43. doi:10.1016/j.devcel.2010.02.013 |
| [36] | Fung TK, Poon RY. A roller coaster ride with the mitotic cyclins [J]. Semin Cell Dev Biol, 2005, 16(3): 335-42. doi:10.1016/j.semcdb.2005.02.014 |
| [37] | Pagano M, Pepperkok R, Verde F, et al. Cyclin A is required at two points in the human cell cycle [J]. EMBO J, 1992, 11(3): 961-71. doi:10.1002/j.1460-2075.1992.tb05135.x |
| [38] | Schrader LA, Ronnekleiv-Kelly SM, Hogenesch JB, et al. Circadian disruption, clock genes, and metabolic health [J]. J Clin Invest, 2024, 134(14): e170998. doi:10.1172/jci170998 |
| [39] | Moravčík R, Olejárová S, Zlacká J, et al. Effect of miR-34a on the expression of clock and clock-controlled genes in DLD1 and Lovo human cancer cells with different backgrounds with respect to p53 functionality and 17β-estradiol-mediated regulation [J]. PLoS One, 2023, 18(10): e0292880. doi:10.1371/journal.pone.0292880 |
| [40] | Farshadi E, Yan J, Leclere P, et al. The positive circadian regulators CLOCK and BMAL1 control G2/M cell cycle transition through Cyclin B1 [J]. Cell Cycle, 2019, 18(1): 16-33. doi:10.1080/15384101.2018.1558638 |
| [41] | Maemura K, de la Monte SM, Chin MT, et al. CLIF, a novel cycle-like factor, regulates the circadian oscillation of plasminogen activator inhibitor-1 gene expression [J]. J Biol Chem, 2000, 275(47): 36847-51. doi:10.1074/jbc.c000629200 |
| [42] | 常笑语, 朱玲勤, 张瀚文, 等.热暴露对大鼠心脏与肝脏组织细胞凋亡及钟基因Bmal1的影响 [J]. 宁夏医科大学学报, 2022, 44(5): 433-7. 450. doi:10.16050/j.cnki.issn1674-6309.2022.05.001 |
| [1] | Yujia YANG, Lifang YANG, Yaling WU, Zhaoda DUAN, Chunze YU, Chunyun WU, Jianyun YU, Li YANG. Cannabidiol inhibits neuronal endoplasmic reticulum stress and apoptosis in rats with multiple concussions by regulating the PERK-eIF2α-ATF4-CHOP pathway [J]. Journal of Southern Medical University, 2025, 45(6): 1240-1250. |
| [2] | Yue CHEN, Linyu XIAO, Lü REN, Xue SONG, Jing LI, Jianguo HU. Monotropein improves motor function of mice with spinal cord injury by inhibiting the PI3K/AKT signaling pathway to suppress neuronal apoptosis [J]. Journal of Southern Medical University, 2025, 45(4): 774-784. |
| [3] | Fei CHU, Xiaohua CHEN, Bowen SONG, Jingjing YANG, Lugen ZUO. Moslosooflavone ameliorates dextran sulfate sodium-induced colitis in mice by suppressing intestinal epithelium apoptosis via inhibiting the PI3K/AKT signaling pathway [J]. Journal of Southern Medical University, 2025, 45(4): 819-828. |
| [4] | Yi ZHANG, Yu SHEN, Zhiqiang WAN, Song TAO, Yakui LIU, Shuanhu WANG. High expression of CDKN3 promotes migration and invasion of gastric cancer cells by regulating the p53/NF-κB signaling pathway and inhibiting cell apoptosis [J]. Journal of Southern Medical University, 2025, 45(4): 853-861. |
| [5] | Di CHEN, Ying LÜ, Yixin GUO, Yirong ZHANG, Ruixuan WANG, Xiaoruo ZHOU, Yuxin CHEN, Xiaohui WU. Dihydroartemisinin enhances doxorubicin-induced apoptosis of triple negative breast cancer cells by negatively regulating the STAT3/HIF-1α pathway [J]. Journal of Southern Medical University, 2025, 45(2): 254-260. |
| [6] | Yu BIN, Ziwen LI, Suwei ZUO, Sinuo SUN, Min LI, Jiayin SONG, Xu LIN, Gang XUE, Jingfang WU. High expression of apolipoprotein C1 promotes proliferation and inhibits apoptosis of papillary thyroid carcinoma cells by activating the JAK2/STAT3 signaling pathway [J]. Journal of Southern Medical University, 2025, 45(2): 359-370. |
| [7] | Shuo LIU, Jing LI, Xingwang WU. Swertiamarin ameliorates 2,4,6-trinitrobenzenesulfonic acid-induced colitis in mice by inhibiting intestinal epithelial cell apoptosis [J]. Journal of Southern Medical University, 2024, 44(8): 1545-1552. |
| [8] | Xiaofan CONG, Teng CHEN, Shuo LI, Yuanyuan WANG, Longyun ZHOU, Xiaolong LI, Pei ZHANG, Xiaojin SUN, Surong ZHAO. Dihydroartemisinin enhances sensitivity of nasopharyngeal carcinoma HNE1/DDP cells to cisplatin-induced apoptosis by promoting ROS production [J]. Journal of Southern Medical University, 2024, 44(8): 1553-1560. |
| [9] | Linyu XIAO, Ting DUAN, Yongsheng XIA, Yue CHEN, Yang SUN, Yibo XU, Lei XU, Xingzhou YAN, Jianguo HU. Linarin inhibits microglia activation-mediated neuroinflammation and neuronal apoptosis in mouse spinal cord injury by inhibiting the TLR4/NF-κB pathway [J]. Journal of Southern Medical University, 2024, 44(8): 1589-1598. |
| [10] | Mengdong ZHENG, Yan LIU, Jiaojiao LIU, Qiaozhen KANG, Ting WANG. Effect of deletion of protein 4.1R on proliferation, apoptosis and glycolysis of hepatocyte HL-7702 cells [J]. Journal of Southern Medical University, 2024, 44(7): 1355-1360. |
| [11] | Yuanguo WANG, Peng ZHANG. Ferroptosis suppressor genes are highly expressed in esophageal cancer to inhibit tumor cell ferroptosis [J]. Journal of Southern Medical University, 2024, 44(7): 1389-1396. |
| [12] | Zhijun REN, Jianxin DIAO, Yiting WANG. Xionggui Decoction alleviates heart failure in mice with myocardial infarction by inhibiting oxidative stress-induced cardiomyocyte apoptosis [J]. Journal of Southern Medical University, 2024, 44(7): 1416-1424. |
| [13] | Guiling CHEN, Xiaofeng LIAO, Pengtao SUN, Huan CEN, Shengchun SHU, Bijing LI, Jinhua LI. Solasonine promotes apoptosis of non-small cell lung cancer cells by regulating the Bcl-2/Bax/caspase-3 pathway [J]. Journal of Southern Medical University, 2024, 44(6): 1109-1116. |
| [14] | Lingjun LU, Xiaodi YANG, Huaping ZHANG, Yuan LIANG, Xiulan SHI, Xin ZHOU. Recombinant Schistosoma japonicum cystatin alleviates acute liver injury in mice by inhibiting endoplasmic reticulum stress, inflammation and hepatocyte apoptosis [J]. Journal of Southern Medical University, 2024, 44(6): 1126-1134. |
| [15] | Guoxin LIANG, Hongyue TANG, Chang GUO, Mingming ZHANG. MiR-224-5p overexpression inhibits oxidative stress by regulating the PI3K/Akt/FoxO1 axis to attenuate hypoxia/reoxygenation-induced cardiomyocyte injury [J]. Journal of Southern Medical University, 2024, 44(6): 1173-1181. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||