Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 541-549.doi: 10.12122/j.issn.1673-4254.2026.03.08
Jie XIAO( ), Lingyue JIN, Lina DUAN, Ying GONG(
), Lingyue JIN, Lina DUAN, Ying GONG( ), Haixia LI(
), Haixia LI( )
)
Received:2025-09-26
Online:2026-03-20
Published:2026-03-26
Contact:
Ying GONG, Haixia LI
E-mail:x011218jie@163.com;gongy3@mail2.sysu.edu.cn;yingchun1220@163.com
Supported by:Jie XIAO, Lingyue JIN, Lina DUAN, Ying GONG, Haixia LI. Streptococcus salivarius-derived extracellular vesicles exacerbate progression of lupus nephritis by activating natural killer cells[J]. Journal of Southern Medical University, 2026, 46(3): 541-549.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.08
| Primer name | Sequence (5'-3') |
|---|---|
| GAPDH -FOR | CTGTTCGACAGTCAGCCGCATC |
| GAPDH -REV | GCGCCCAATACGACCAAATCCG |
| TNF-FOR | GAGGCCAAGCCCTGGTATG |
| TNF-REV | CGGGCCGATTGATCTCAGC |
| GZMB-FOR | CCCTGGGAAAACACTCACACA |
| GZMB-REV | GCACAACTCAATGGTACTGTCG |
| IL2-FOR | AACTCCTGTCTTGCATTGCAC |
| IL2-REV | GCTCCAGTTGTAGCTGTGTTT |
| Perforin-FOR | GACTGCCTGACTGTCGAGG |
| Perforin-REV | TCCCGGTAGGTTTGGTGGAA |
| NKG2D-FOR | CCTTGACCGAAAGTTACTGTGG |
| NKG2D-REV | GGCTGGCATTTTGAGACATACAA |
| NKG2A-FOR | AGCTCCATTTTAGCAACTGAACA |
| NKG2A-REV | CAACTATCGTTACCACAGAGGC |
Tab.1 Primer sequences for qPCR
| Primer name | Sequence (5'-3') |
|---|---|
| GAPDH -FOR | CTGTTCGACAGTCAGCCGCATC |
| GAPDH -REV | GCGCCCAATACGACCAAATCCG |
| TNF-FOR | GAGGCCAAGCCCTGGTATG |
| TNF-REV | CGGGCCGATTGATCTCAGC |
| GZMB-FOR | CCCTGGGAAAACACTCACACA |
| GZMB-REV | GCACAACTCAATGGTACTGTCG |
| IL2-FOR | AACTCCTGTCTTGCATTGCAC |
| IL2-REV | GCTCCAGTTGTAGCTGTGTTT |
| Perforin-FOR | GACTGCCTGACTGTCGAGG |
| Perforin-REV | TCCCGGTAGGTTTGGTGGAA |
| NKG2D-FOR | CCTTGACCGAAAGTTACTGTGG |
| NKG2D-REV | GGCTGGCATTTTGAGACATACAA |
| NKG2A-FOR | AGCTCCATTTTAGCAACTGAACA |
| NKG2A-REV | CAACTATCGTTACCACAGAGGC |
| General information | SLE group (n=35) | HC group (n=38) | P | |
|---|---|---|---|---|
| Gender [n (%)] | Female | 30 (85.71%) | 31 (81.58%) | 0.756 |
| Male | 5 (14.29%) | 7 (18.42%) | ||
| Age (year) | 36.57±12.50 | 39.34±11.84 | 0.334 | |
| WBC (109/L) | 6.07±2.81 | 5.66±1.17 | 0.422 | |
| LYM (109/L) | 1.53±0.76 | 2.20±0.47 | <0.001 | |
| NEU (109/L) | 4.12±2.09 | 3.12±0.93 | 0.043 | |
| RBC (1012/L) | 4.12±0.67 | 4.89±0.32 | <0.001 | |
| HGB (g/L) | 113.48±14.73 | 135.67±17.63 | <0.001 | |
| HCT (L/L) | 0.37±0.38 | 0.42±0.43 | <0.001 | |
| PLT (109/L) | 243.42±82.22 | 255.64±59.45 | 0.512 | |
| SLEDAI | 6.74±3.35 | - | - | |
| WBC: White blood cell; LYM: Lymphocyte; NEU: Neutrophil; RBC: Red blood cell; HGB: Hemoglobin; HCT: Hematocrit; PLT: Platelet. Data are presented as Mean±SD. P values were calculated by Mann-Whitney U test or t test. | ||||
Tab.2 Clinical characteristics of the SLE patients and healthy control subjects (Mean±SD)
| General information | SLE group (n=35) | HC group (n=38) | P | |
|---|---|---|---|---|
| Gender [n (%)] | Female | 30 (85.71%) | 31 (81.58%) | 0.756 |
| Male | 5 (14.29%) | 7 (18.42%) | ||
| Age (year) | 36.57±12.50 | 39.34±11.84 | 0.334 | |
| WBC (109/L) | 6.07±2.81 | 5.66±1.17 | 0.422 | |
| LYM (109/L) | 1.53±0.76 | 2.20±0.47 | <0.001 | |
| NEU (109/L) | 4.12±2.09 | 3.12±0.93 | 0.043 | |
| RBC (1012/L) | 4.12±0.67 | 4.89±0.32 | <0.001 | |
| HGB (g/L) | 113.48±14.73 | 135.67±17.63 | <0.001 | |
| HCT (L/L) | 0.37±0.38 | 0.42±0.43 | <0.001 | |
| PLT (109/L) | 243.42±82.22 | 255.64±59.45 | 0.512 | |
| SLEDAI | 6.74±3.35 | - | - | |
| WBC: White blood cell; LYM: Lymphocyte; NEU: Neutrophil; RBC: Red blood cell; HGB: Hemoglobin; HCT: Hematocrit; PLT: Platelet. Data are presented as Mean±SD. P values were calculated by Mann-Whitney U test or t test. | ||||
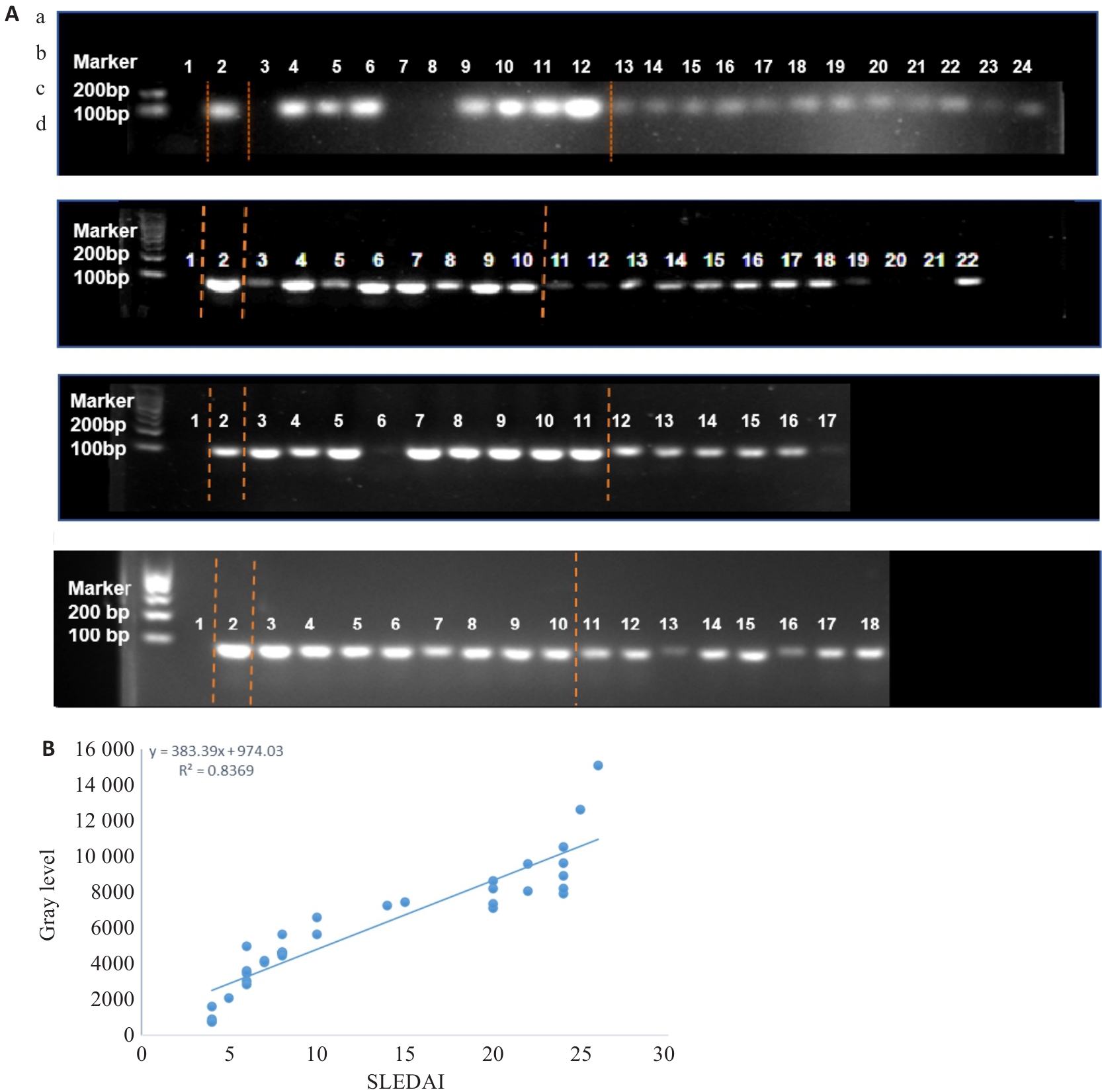
Fig.1 Agarose gel electrophoresis of fecal Streptococcus salivarius (SS) nucleic acids. A: Results of agarose gel electrophoresis. Lane 1: Deionized water (negative control); Lane 2: SS standard strain (positive control). a: Lanes 3-12 are SLE group, and lanes 13-24 are HC group; b: Lanes 3-10 are SLE group, and lanes 11-22 are HC group; c: Lanes 3-11 are SLE group, and lanes 12-17 are HC group; d: Lanes 3-10 are SLE group, and lanes 11-18 are HC group. B: Correlation analysis between the grayscale value of fecal SS nucleic acids from SLE patients and the SLEDAI score.
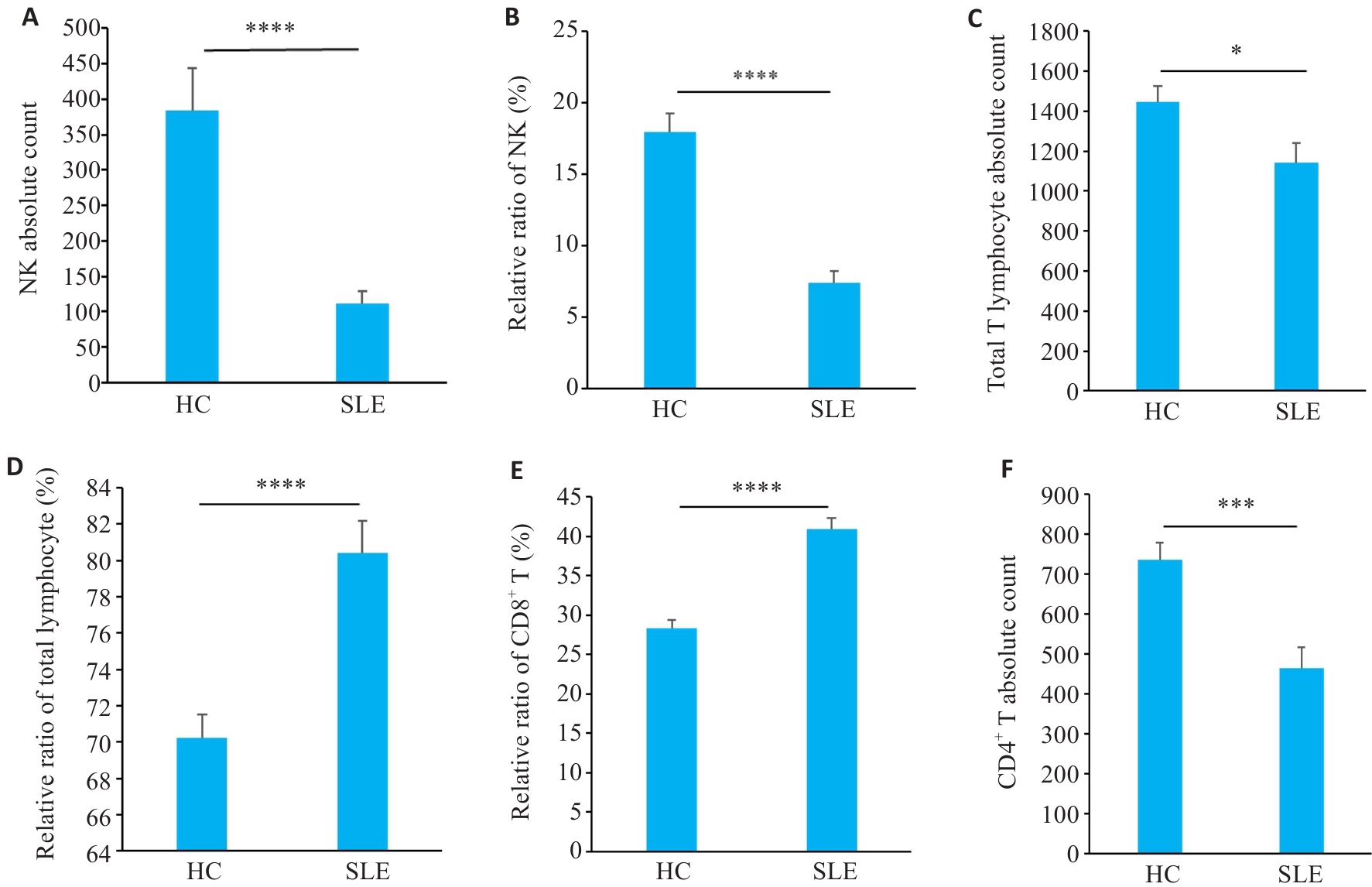
Fig.2 Distribution of peripheral blood immune cell subsets in SLE patients. A, B: NK cell proportions and absolute counts in SLE patients and HC groups. C, D: Total T cell absolute counts and subset proportions. E: CD8+ T cell subset proportions. F: CD4+ T cell absolute counts (n=35 in SLE group, n=38 in HC group). *P<0.05, ***P<0.001, ****P<0.0001.
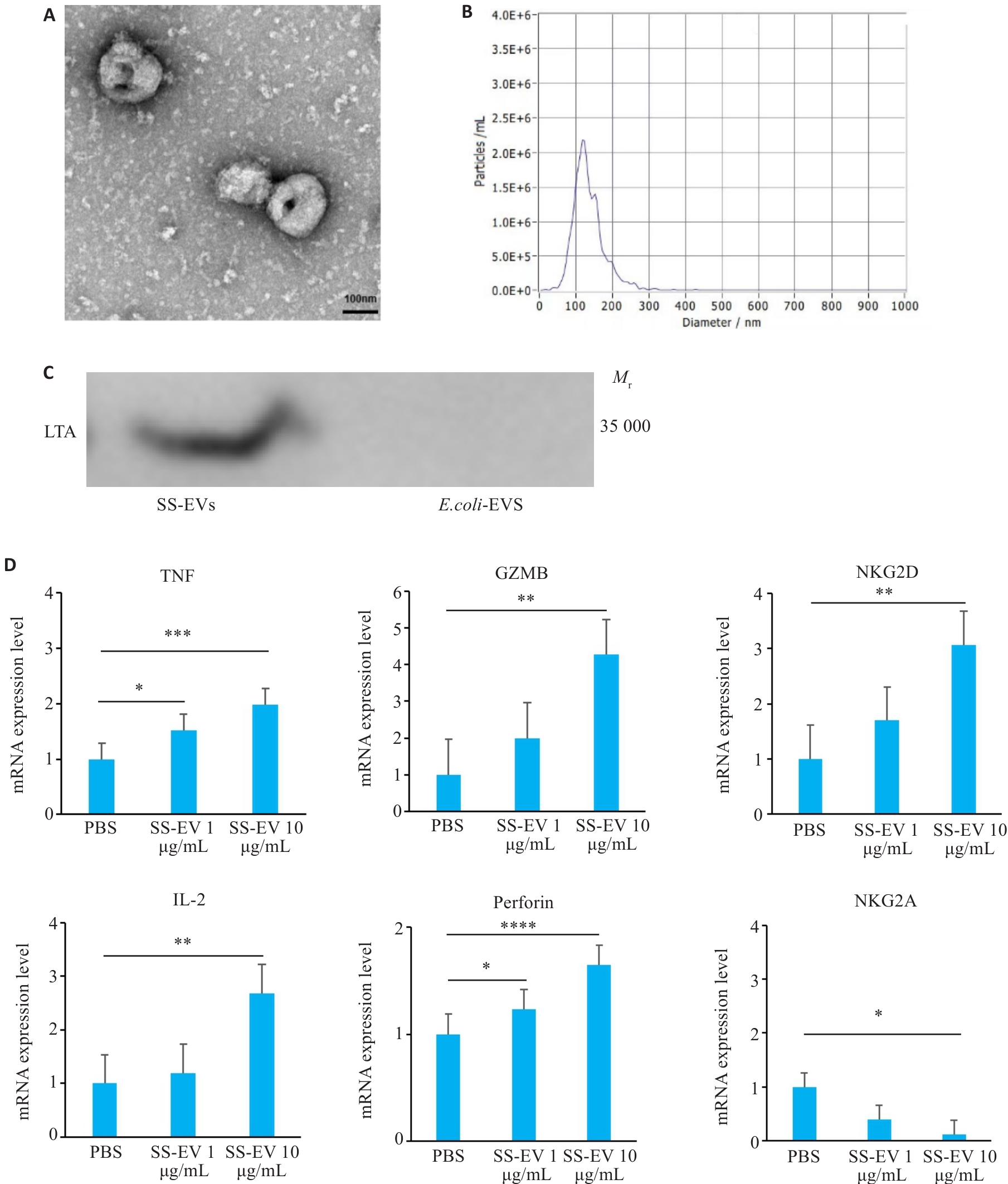
Fig.3 Physical properties of Streptococcus salivarius-derived extracellular vesicles (SS-EVs)and their regulatory effect on NK cell receptors. A: Transmission electron microscopy (TEM) of SS-EVs (Scale bar=100 nm). B: NTA analysis showing size distribution of SS-EVs (100-200 nm). C: Western blotting of LTA protein expression in SS-EVs. D: qPCR analysis of mRNA expression changes of the activating receptors (TNF, GZMB, NKG2D, IL-2, and Perforin) and inhibitory receptors (NKG2A) in NK cells co-cultured with SS-EVs (n=3). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
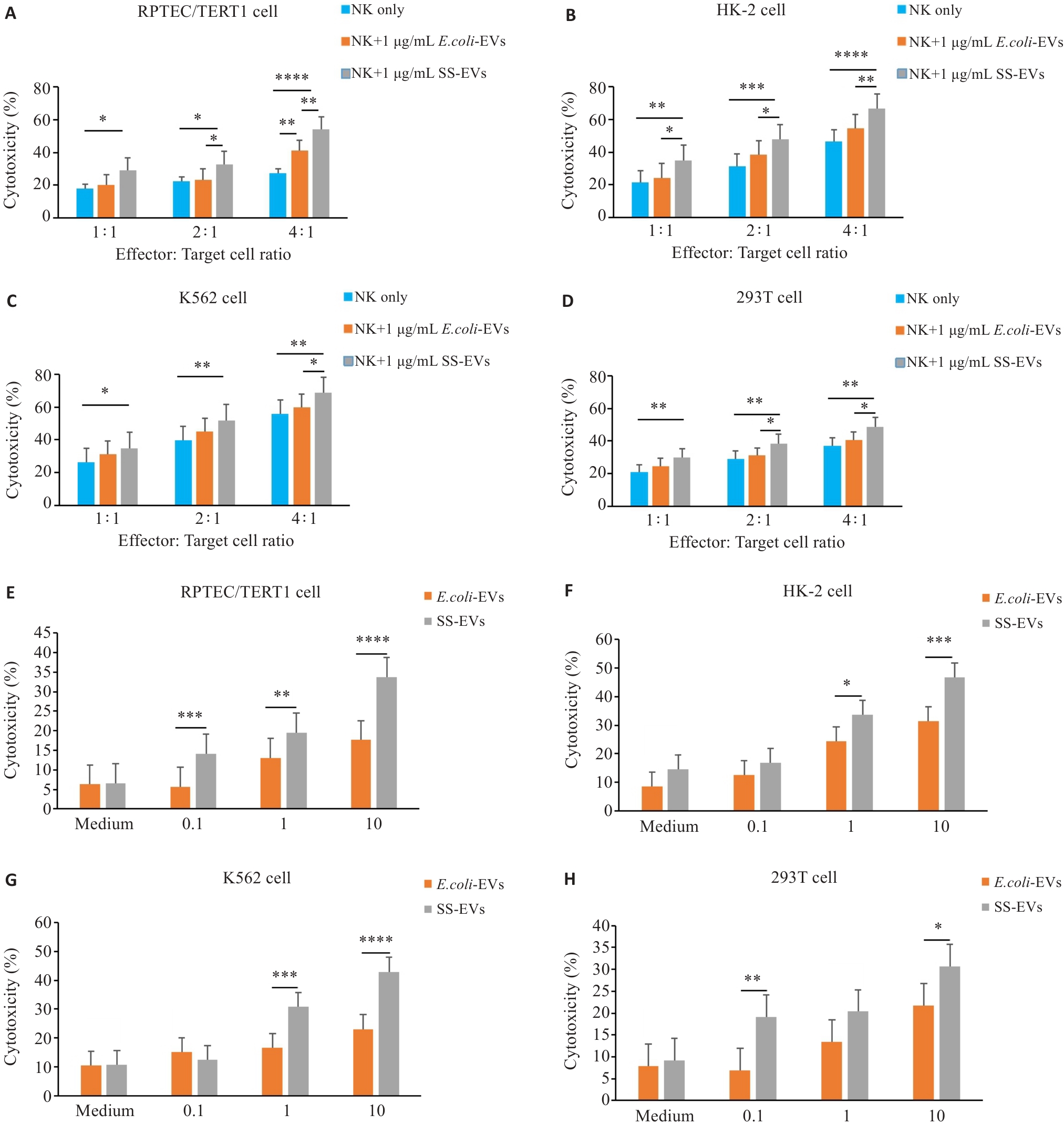
Fig.4 SS-EVs enhance cytotoxic effect of NK cells on target cells. A-D: Relationship between NK cell to target cell ratio (1:1 to 4:1) and NK cell cytotoxicity at a fixed SS-EV concentration (1 μg/mL) (n=3). E-H: Flow cytometry analysis of the effects of varying SS-EV concentrations (0.1-10 μg/mL) on NK cell cytotoxicity against 4 target cells (n=3). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
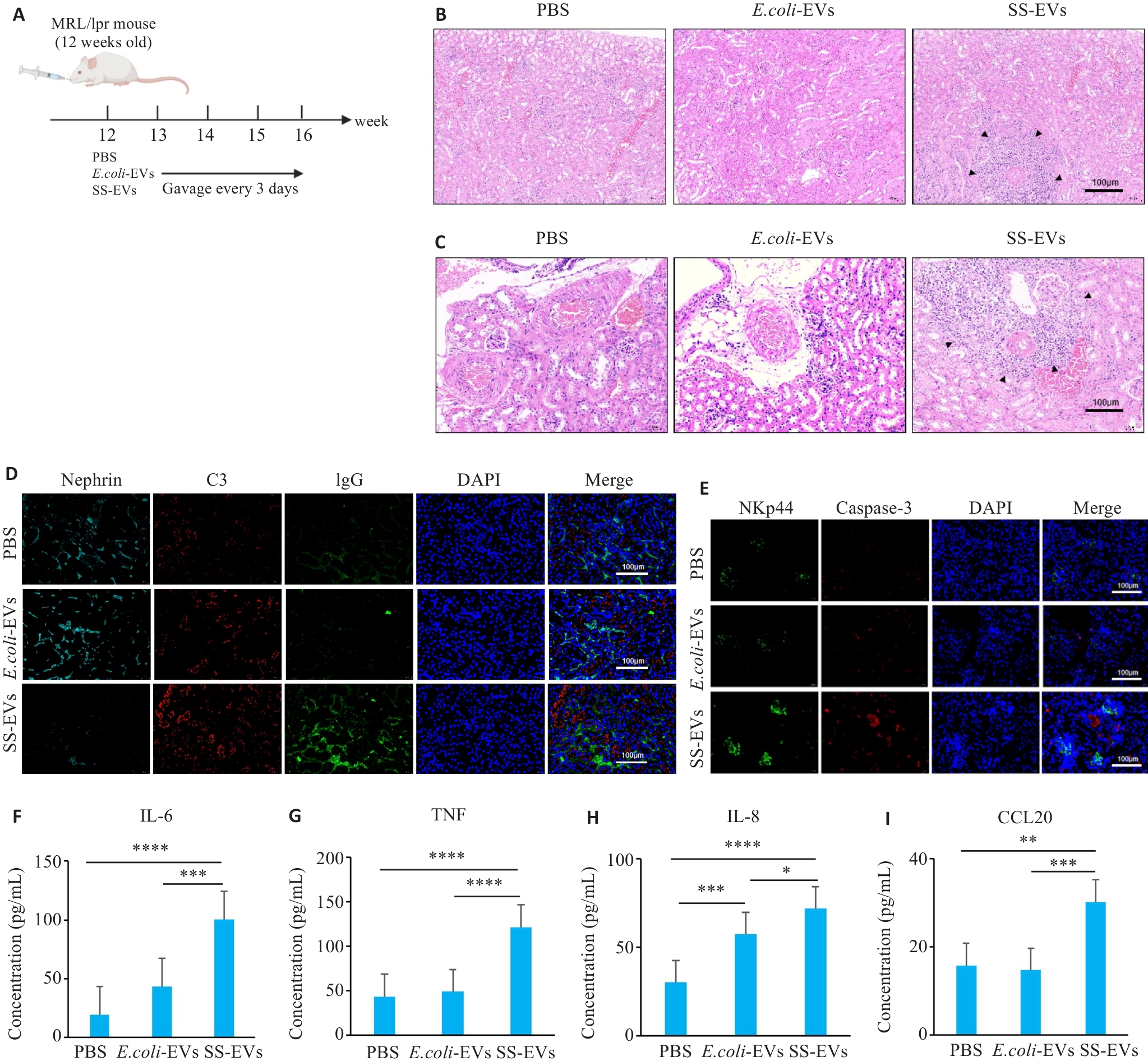
Fig.5 SS-EVs gavage aggravates kidney pathological damage in MRL/lpr mice. A: Schematic diagram of animal grouping and gavage. B: Significant inflammatory cell infiltration in the kidneys of mice gavaged with SS-EVs. C: Significant perivascular inflammatory cell infiltration in the kidneys of mice gavaged with SS-EVs. D: SS-EVs damage the renal medulla and promote immune complex deposition. E:SS-EVs induce NK cell infiltration in the kidneys and renal apoptosis. F-I: ELISA for IL-6, TNF, IL-8, and CCL20 levels in mouse serum (n=6). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
| [1] | Siegel CH, Sammaritano LR. Systemic lupus erythematosus: a review[J]. JAMA, 2024, 331(17): 1480-91. doi:10.1001/jama.2024.2315 |
| [2] | Cojocaru M, Cojocaru IM, Silosi I, et al. Manifestations of systemic lupus erythematosus[J]. Maedica (Bucur), 2011, 6(4): 330-6. |
| [3] | Weckerle CE, Niewold TB. The unexplained female predominance of systemic lupus erythematosus: clues from genetic and cytokine studies[J]. Clin Rev Allergy Immunol, 2011, 40(1): 42-9. doi:10.1007/s12016-009-8192-4 |
| [4] | McMurray RW, May W. Sex hormones and systemic lupus erythematosus: review and meta-analysis[J]. Arthritis Rheum, 2003, 48(8): 2100-10. doi:10.1002/art.11105 |
| [5] | Rosario C, Seguro L, Vasconcelos C, et al. Is there a cure for systemic lupus erythematosus[J]? Lupus, 2013, 22(5): 417-21. doi:10.1177/0961203313479839 |
| [6] | Yu HT, Nagafuchi Y, Fujio K. Clinical and immunological biomarkers for systemic lupus erythematosus[J]. Biomolecules, 2021, 11(7): 928. doi:10.3390/biom11070928 |
| [7] | Suárez-Fueyo A, Bradley SJ, Tsokos GC. T cells in systemic lupus erythematosus[J]. Curr Opin Immunol, 2016, 43: 32-8. doi:10.1016/j.coi.2016.09.001 |
| [8] | Karrar S, Cunninghame Graham DS. Abnormal B cell development in systemic lupus erythematosus: what the genetics tell us[J]. Arthritis Rheumatol, 2018, 70(4): 496-507. doi:10.1002/art.40396 |
| [9] | Perera Arachchige AS. Human NK cells: From development to effector functions[J]. Innate Immun, 2021, 27(3): 212-29. doi:10.1177/17534259211001512 |
| [10] | Mace EM. Human natural killer cells: Form, function, and development[J]. J Allergy Clin Immunol, 2023, 151(2): 371-85. doi:10.1016/j.jaci.2022.09.022 |
| [11] | Jamal E, Azmy E, Ayed M, et al. Clinical impact of percentage of natural killer cells and natural killer-like T cell population in acute myeloid leukemia[J]. J Hematol, 2020, 9(3): 62-70. doi:10.14740/jh655 |
| [12] | Ramírez-Labrada A, Pesini C, Santiago L, et al. All about (NK cell-mediated) death in two acts and an unexpected encore: initiation, execution and activation of adaptive immunity[J]. Front Immunol, 2022, 13: 896228. doi:10.3389/fimmu.2022.896228 |
| [13] | Paul S, Lal G. The molecular mechanism of natural killer cells function and its importance in cancer immunotherapy[J]. Front Immunol, 2017, 8: 1124. doi:10.3389/fimmu.2017.01124 |
| [14] | Wang W, Erbe AK, Hank JA, et al. NK cell-mediated antibody-dependent cellular cytotoxicity in cancer immunotherapy[J]. Front Immunol, 2015, 6: 368. doi:10.3389/fimmu.2015.00368 |
| [15] | Hill GR, Betts BC, Tkachev V, et al. Current concepts and advances in graft-versus-host disease immunology[J]. Annu Rev Immunol, 2021, 39: 19-49. doi:10.1146/annurev-immunol-102119-073227 |
| [16] | Xiao J, Luo YH, Duan LN, et al. Exploring differential gene expression and biomarker potential in systemic lupus erythematosus: a retrospective study[J]. PeerJ, 2025, 13: e19891. doi:10.7717/peerj.19891 |
| [17] | Duarte-García A, Hocaoglu M, Valenzuela-Almada M, et al. Rising incidence and prevalence of systemic lupus erythematosus: a population-based study over four decades[J]. Ann Rheum Dis, 2022, 81(9): 1260-6. doi:10.1136/annrheumdis-2022-222276 |
| [18] | Fu YF, Lyu J, Wang SS. The role of intestinal microbes on intestinal barrier function and host immunity from a metabolite perspective[J]. Front Immunol, 2023, 14: 1277102. doi:10.3389/fimmu.2023.1277102 |
| [19] | Bechler KK, Stolyar L, Steinberg E, et al. Predicting patients who are likely to develop Lupus Nephritis of those newly diagnosed with Systemic Lupus Erythematosus[J]. AMIA Annu Symp Proc, 2023, 2022: 221-30. |
| [20] | Singh BK, Singh S. Systemic lupus erythematosus and infections[J]. Reumatismo, 2020, 72(3): 154-69. doi:10.4081/reumatismo.2020.1303 |
| [21] | Jin LY, Xiao J, Luo YH, et al. Exploring gut microbiota in systemic lupus erythematosus: insights and biomarker discovery potential[J]. Clin Rev Allergy Immunol, 2025, 68(1): 42. doi:10.1007/s12016-025-09051-4 |
| [22] | Yan R, Jiang HY, Gu SL, et al. Fecal metabolites were altered, identified as biomarkers and correlated with disease activity in patients with systemic lupus erythematosus in a GC-MS-based metabolomics study[J]. Front Immunol, 2020, 11: 2138. doi:10.3389/fimmu.2020.02138 |
| [23] | Kaci G, Goudercourt D, Dennin V, et al. Anti-inflammatory properties of Streptococcus salivarius, a commensal bacterium of the oral cavity and digestive tract[J]. Appl Environ Microbiol, 2014, 80(3): 928-34. doi:10.1128/aem.03133-13 |
| [24] | Wescombe PA, Hale JDF, Heng NCK, et al. Developing oral probiotics from Streptococcus salivarius [J]. Future Microbiol, 2012, 7(12): 1355-71. doi:10.2217/fmb.12.113 |
| [25] | García-Ríos P, Pecci-Lloret MP, Oñate-Sánchez RE. Oral manifestations of systemic lupus erythematosus: a systematic review[J]. Int J Environ Res Public Health, 2022, 19(19): 11910. doi:10.3390/ijerph191911910 |
| [26] | Duan LN, Lin WY, Zhang Y, et al. Exosomes in autoimmune diseases: a review of mechanisms and diagnostic applications[J]. Clin Rev Allergy Immunol, 2025, 68(1): 5. doi:10.1007/s12016-024-09013-2 |
| [27] | Muñoz-Echeverri LM, Benavides-López S, Geiger O, et al. Bacterial extracellular vesicles: biotechnological perspective for enhanced productivity[J]. World J Microbiol Biotechnol, 2024, 40(6): 174. doi:10.1007/s11274-024-03963-7 |
| [28] | Ou ZH, Bo ST, Huang XY, et al. Single-particle analysis of circulating bacterial extracellular vesicles reveals their biogenesis, changes in blood and links to intestinal barrier[J]. J Extracell Vesicles, 2023, 12(12): e12395. doi:10.1002/jev2.12395 |
| [29] | Nie XK, Li QQ, Chen XY, et al. Bacterial extracellular vesicles: Vital contributors to physiology from bacteria to host[J]. Microbiol Res, 2024, 284: 127733. doi:10.1016/j.micres.2024.127733 |
| [30] | Gao Y, Zhang YK, Liu XG. Rheumatoid arthritis: pathogenesis and therapeutic advances[J]. MedComm (2020), 2024, 5(3): e509. doi:10.1002/mco2.509 |
| [31] | Mueller AL, Payandeh Z, Mohammadkhani N, et al. Recent advances in understanding the pathogenesis of rheumatoid arthritis: new treatment strategies[J]. Cells, 2021, 10(11): 3017. doi:10.3390/cells10113017 |
| [32] | Hong MK, Li Z, Liu HH, et al. Fusobacterium nucleatum aggravates rheumatoid arthritis through FadA-containing outer membrane vesicles[J]. Cell Host Microbe, 2023, 31(5): 798-810.e7. doi:10.1016/j.chom.2023.03.018 |
| [33] | Salgueiro VC, Passemar C, Vázquez-Iniesta L, et al. Extracellular vesicles in mycobacteria: new findings in biogenesis, host-pathogen interactions, and diagnostics[J]. mBio, 2024, 15(5): e0255223. doi:10.1128/mbio.02552-23 |
| [34] | Kucuksezer UC, Aktas Cetin E, Esen F, et al. The role of natural killer cells in autoimmune diseases[J]. Front Immunol, 2021, 12: 622306. doi:10.3389/fimmu.2021.622306 |
| [35] | Novelli L, Barbati C, Capuano C, et al. KLRG1 is reduced on NK cells in SLE patients, inversely correlates with disease activity and is modulated by hydroxychloroquine in vitro [J]. Lupus, 2023, 32(4): 549-59. doi:10.1177/09612033231160979 |
| [36] | Seow WK, Lam JHC, Tsang AKL, et al. Oral Streptococcus species in pre-term and full-term children-a longitudinal study[J]. Int J Paediatr Dent, 2009, 19(6): 406-11. doi:10.1111/j.1365-263x.2009.01003.x |
| [37] | Iraci N, Leonardi T, Gessler F, et al. Focus on extracellular vesicles: physiological role and signalling properties of extracellular membrane vesicles[J]. Int J Mol Sci, 2016, 17(2): 171. doi:10.3390/ijms17020171 |
| [38] | Rafieezadeh D, Rafieezadeh A. Extracellular vesicles and their therapeutic applications: a review article (part1)[J]. Int J Physiol Pathophysiol Pharmacol, 2024, 16(1): 1-9. doi:10.62347/qpag5693 |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||