南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 838-847.doi: 10.12122/j.issn.1673-4254.2026.04.12
• • 上一篇
范东伟1( ), 李煊赫2, 姚廷敬2, 金从稳1, 王兴亮1, 黄诚1(
), 李煊赫2, 姚廷敬2, 金从稳1, 王兴亮1, 黄诚1( )
)
收稿日期:2025-08-13
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
黄诚
E-mail:docfan@ wahvc.edu.cn;huangcheng@wahvc.edu.cn
作者简介:范东伟,在读博士研究生,主治医师,E-mail: docfan@ wahvc.edu.cn
基金资助:
Dongwei FAN1( ), Xuanhe LI2, Tingjing YAO2, Congwen JIN1, Xingliang WANG1, Cheng HUANG1(
), Xuanhe LI2, Tingjing YAO2, Congwen JIN1, Xingliang WANG1, Cheng HUANG1( )
)
Received:2025-08-13
Online:2026-04-20
Published:2026-04-24
Contact:
Cheng HUANG
E-mail:docfan@ wahvc.edu.cn;huangcheng@wahvc.edu.cn
摘要:
目的 探讨毛兰素对乳腺癌细胞的增殖、迁移、侵袭和凋亡的影响及潜在的分子机制。 方法 分别用0、12.5、25、50、100 nmol/L毛兰素处理乳腺癌细胞系T-47D和MCF-7,采用CCK-8法检测12、24、36、48、72 h的细胞活力;克隆形成、细胞划痕、Transwell侵袭和细胞衰老实验分别检测毛兰素对T-47D和MCF-7细胞增殖、迁移、侵袭能力以及细胞衰老的影响。通过毛兰素mRNA微阵列以及Enrichr数据库探索毛兰素的生物学功能。通过流式细胞术检测不同浓度毛兰素对T-47D和MCF-7细胞凋亡率的影响。通过Western blotting检测细胞凋亡、上皮-间充质转化(EMT)以及Wnt/β-catenin通路相关蛋白的表达情况。 结果 毛兰素呈浓度依赖性抑制T-47D和MCF-7细胞活力、增殖、迁移与侵袭能力,并促进细胞衰老(P<0.05)。毛兰素相关mRNA微阵列分析结果显示,共有1064个差异表达基因(DEGs)(P<0.05),其中948个表达上调基因,116个表达下调基因。GO富集分析结果显示,毛兰素的DEGs主要富集在EMT的调节、含胶原蛋白的细胞外基质及钙离子结合等功能中;KEGG和WikiPathways富集分析结果显示,毛兰素可能通过调节PI3K-Akt信号通路、Wnt/β-catenin信号通路以及细胞凋亡影响乳腺癌的发生与进展。流式细胞术结果显示,毛兰素呈浓度依赖性诱导乳腺癌细胞凋亡(P<0.05)。Western blotting结果显示,随着毛兰素浓度升高,促凋亡蛋白Bax、Caspase3表达上升,而抗凋亡蛋白Bcl-2表达下降(P<0.05);EMT相关蛋白Snail、N-cadherin、β-catenin表达下降(P<0.05);Wnt/β-catenin信号相关蛋白TCF4、Cyclin D1、c-Myc表达下降(P<0.05);与100 nmol/L毛兰素组相比,100 nmol/L毛兰素+Wnt/β-catenin激动剂组TCF4、Cyclin D1、c-Myc表达增加(P<0.05)。 结论 毛兰素能抑制乳腺癌细胞增殖、迁移和侵袭,并诱导细胞衰老和凋亡,其分子机制可能与阻断Wnt/β-catenin信号通路,协同诱导细胞凋亡并逆转EMT过程有关。
范东伟, 李煊赫, 姚廷敬, 金从稳, 王兴亮, 黄诚. 毛兰素通过阻断Wnt/β-catenin信号抑制乳腺癌细胞增殖和迁移[J]. 南方医科大学学报, 2026, 46(4): 838-847.
Dongwei FAN, Xuanhe LI, Tingjing YAO, Congwen JIN, Xingliang WANG, Cheng HUANG. Erianin inhibits proliferation and migration of breast cancer cells in vitro by inhibiting Wnt/β-catenin signaling[J]. Journal of Southern Medical University, 2026, 46(4): 838-847.
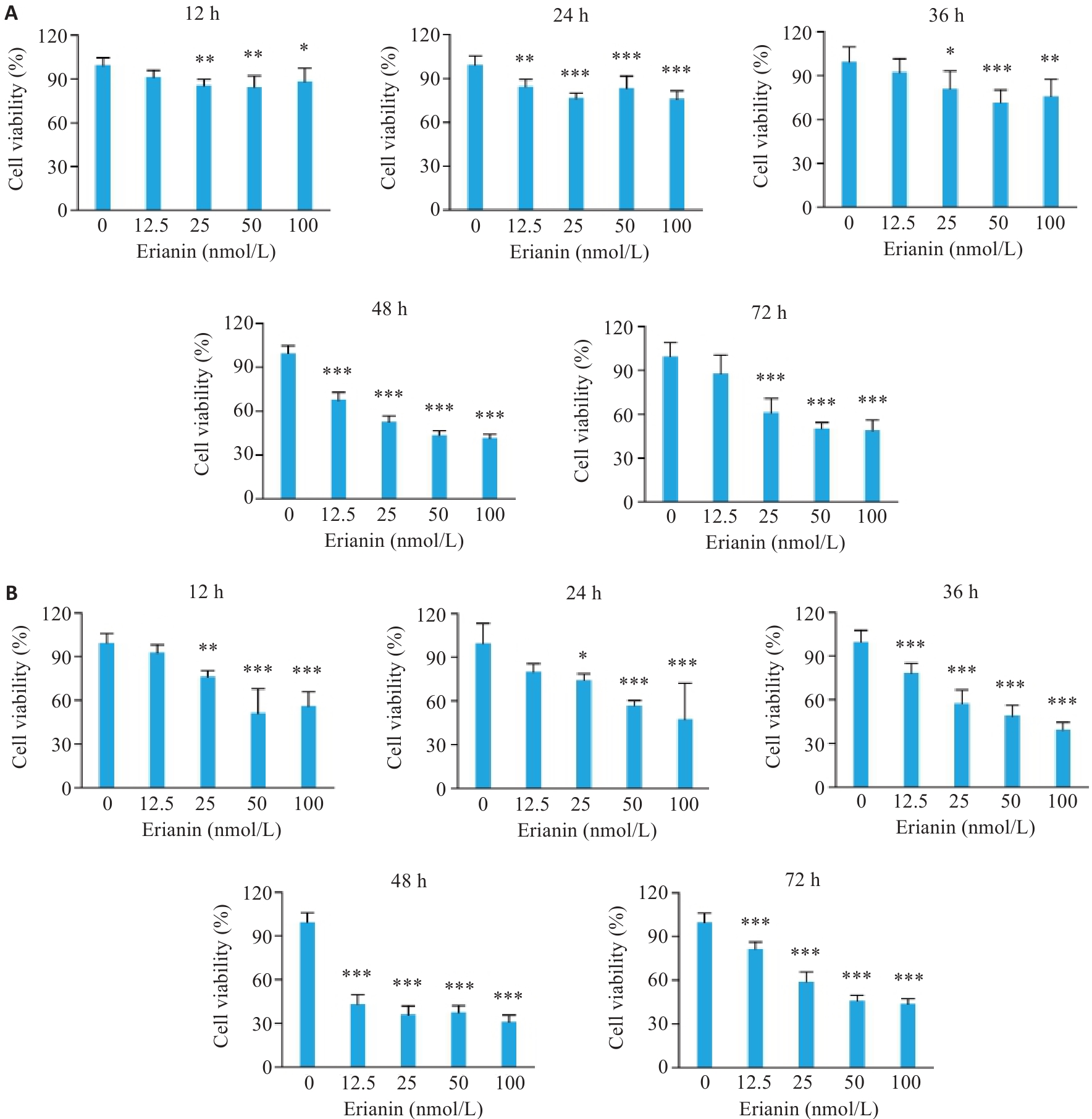
图1 毛兰素对T-47D和MCF-7细胞活力的影响
Fig.1 Effect of erianin on viability of T-47D and MCF-7 cells. A: CCK-8 assay results of T-47D cells treated with different concentrations of erianin for 12, 24, 36, 48, and 72 h. B: CCK-8 assay results of MCF-7 cells treated with different concentrations of erianin for 12, 24, 36, 48, and 72 h. Data are presented as Mean±SD (n=6). *P<0.05, **P<0.01, ***P<0.001 vs 0 nmol/L.
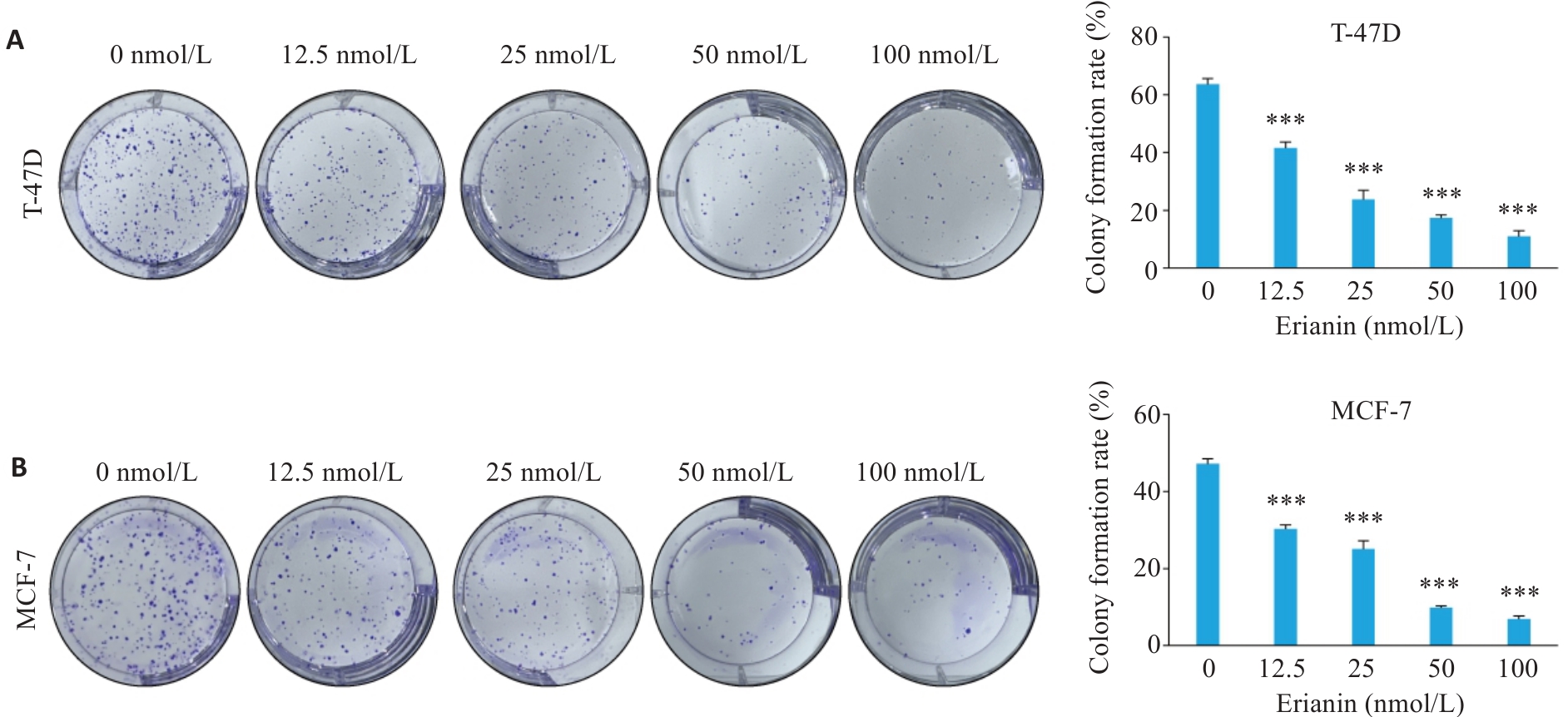
图2 毛兰素对T-47D和MCF-7细胞增殖能力的影响
Fig.2 Effect of erianin on proliferative capacity of T-47D and MCF-7 cells. A: Clone formation assay results of T-47D cells treated with different concentrations of erianin for 48 h. B: Clone formation assay results of MCF-7 cells treated with different concentrations of erianin for 48 h. Data are presented as Mean±SD (n=3). ***P<0.001 vs 0 nmol/L.
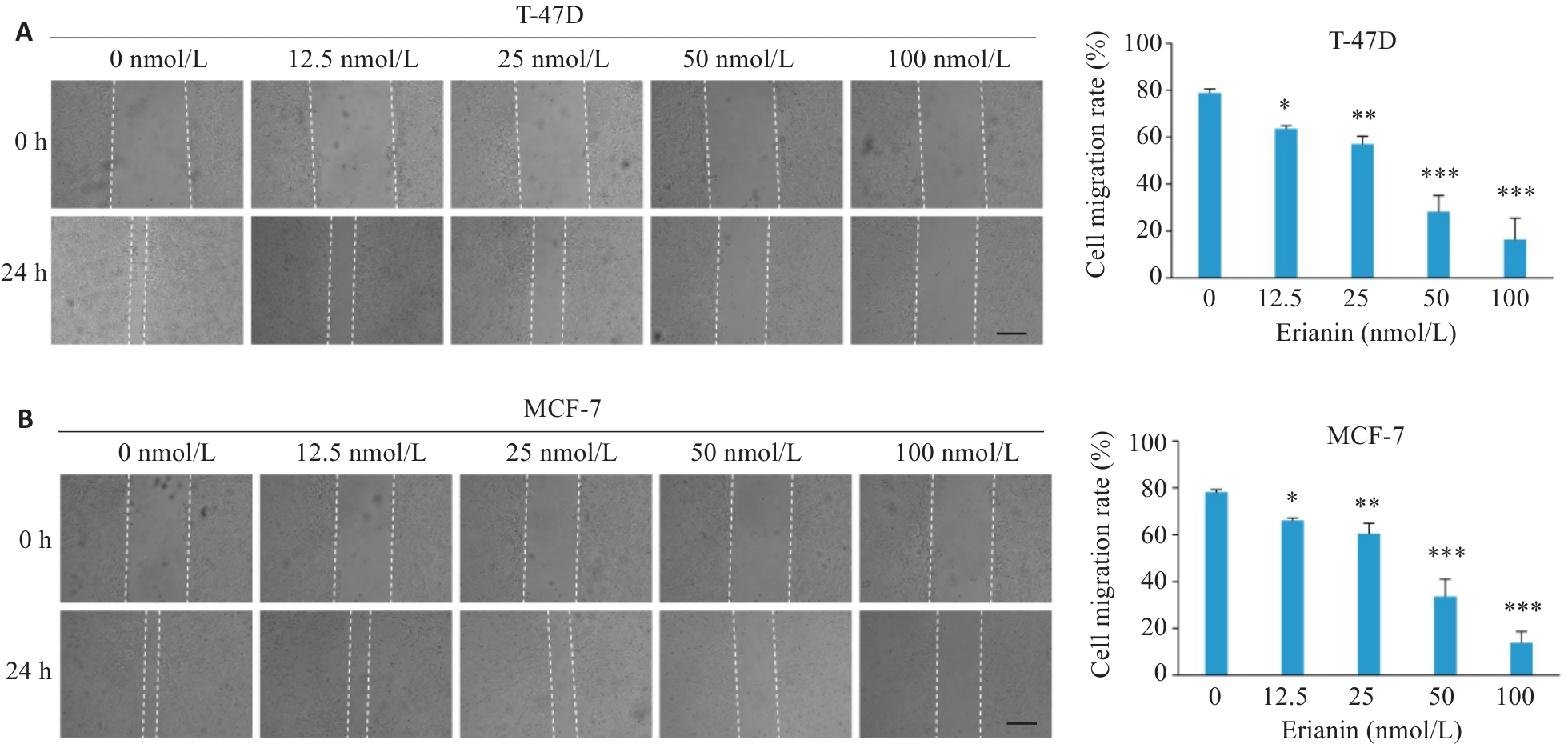
图3 毛兰素对T-47D和MCF-7细胞迁移能力的影响
Fig.3 Effect of erianin on migration of T-47D and MCF-7 cells. A: Wound healing assay results of T-47D cells treated with different concentrations of erianin for 48 h. B: Wound healing assay results of MCF-7 cells treated with different concentrations of erianin for 48 h. Scale bar=200 μm. Data are presented as Mean±SD (n=3). *P<0.05, **P<0.01, ***P<0.001 vs 0 nmol/L.
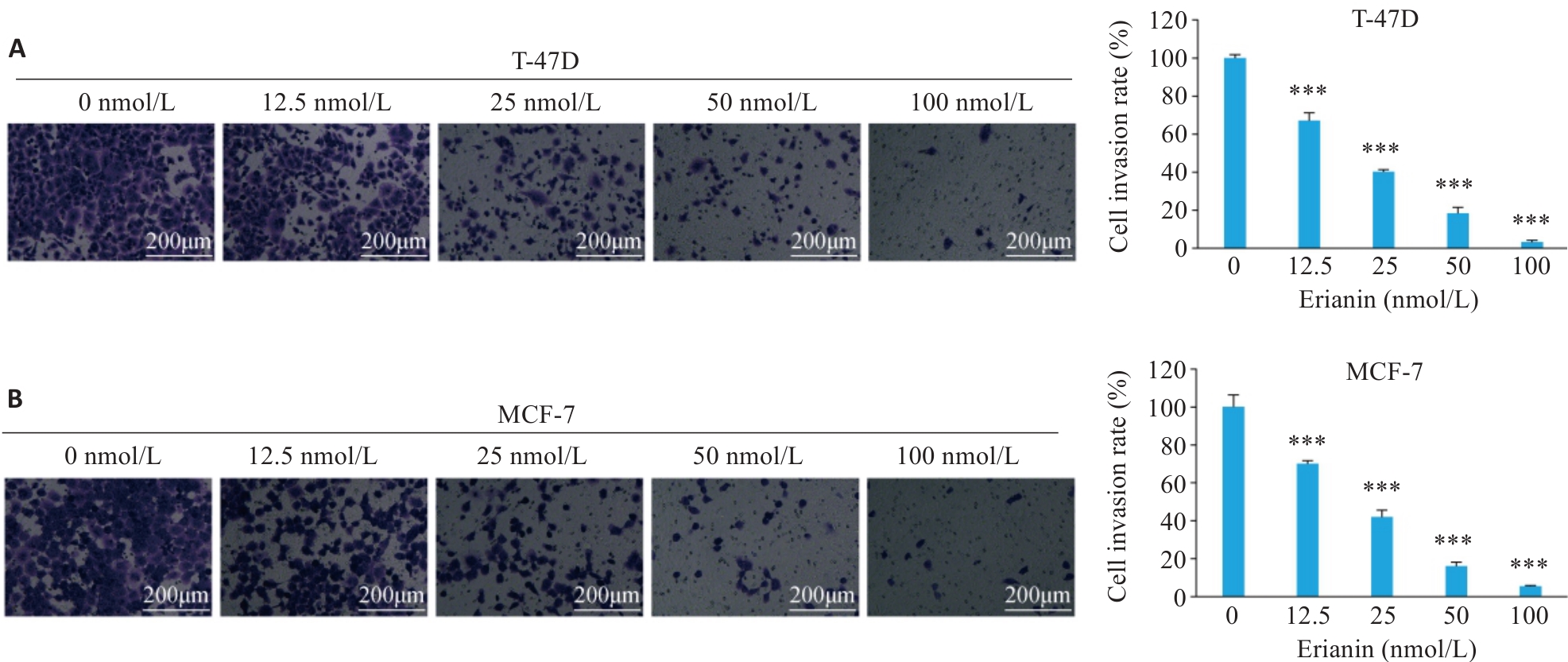
图4 毛兰素对T-47D和MCF-7细胞侵袭能力的影响
Fig.4 Effect of erianin on invasion of T-47D and MCF-7 cells. A: Transwell invasion assay results of T-47D cells treated with different concentrations of erianin for 48 h. B: Transwell invasion assay results of MCF-7 cells treated with different concentrations of erianin for 48 h. Data are presented as Mean±SD (n=3). ***P<0.001 vs 0 nmol/L.
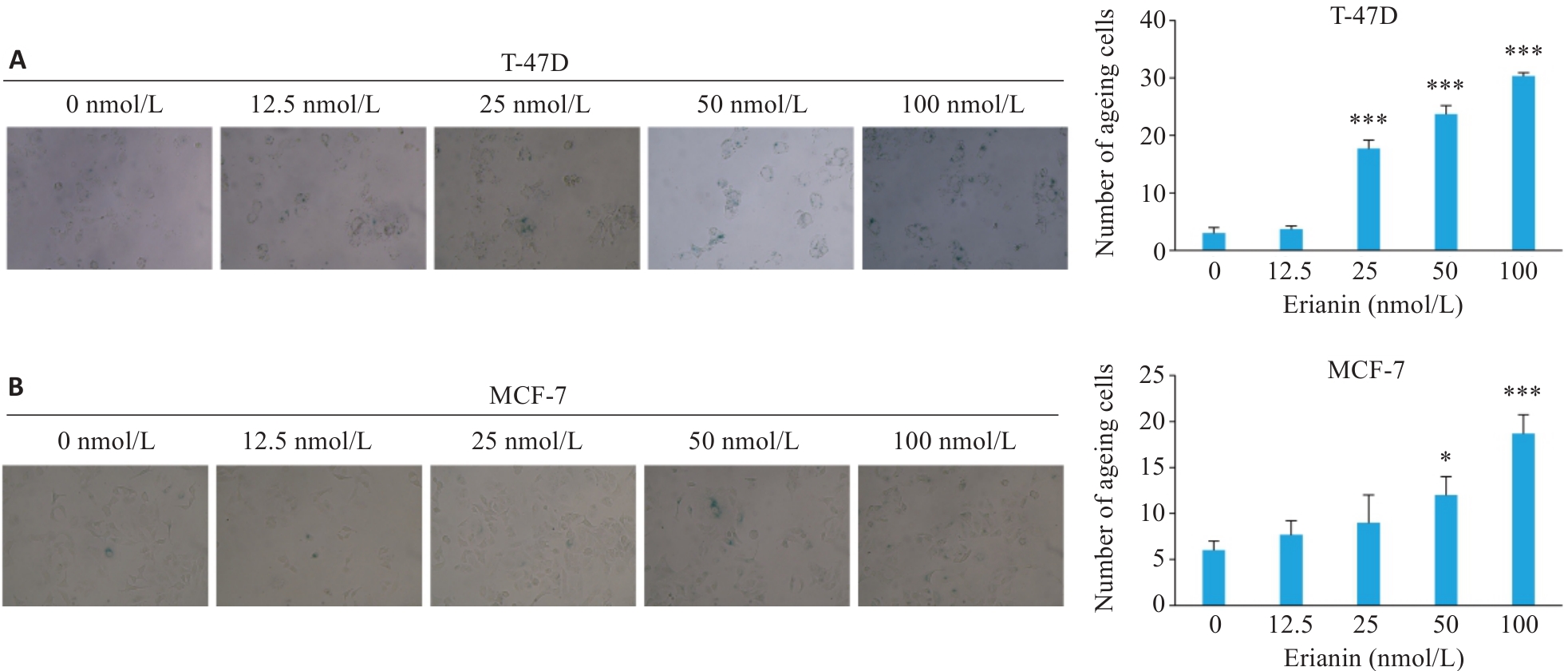
图5 毛兰素对T-47D和MCF-7细胞衰老能力的影响
Fig.5 Effect of erianin on cellular senescence in T-47D and MCF-7 cells (Original magnification: ×200). A: Senescence-associated β-galactosidase staining results of T-47D cells treated with different concentrations of erianin for 48 h. B: Senescence-associated β-galactosidase staining results of MCF-7 cells treated with different concentrations of erianin for 48 h. Data are presented as Mean±SD (n=3). *P<0.05, ***P<0.001 vs 0 nmol/L.

图7 毛兰素DEGs的生物学功能富集分析
Fig.7 Biological functional enrichment analysis of DEGs regulated by erianin. A: GO enrichment analysis of DEGs in breast cancer. B: KEGG and WikiPathways enrichment analysis of DEGs in breast cancer.
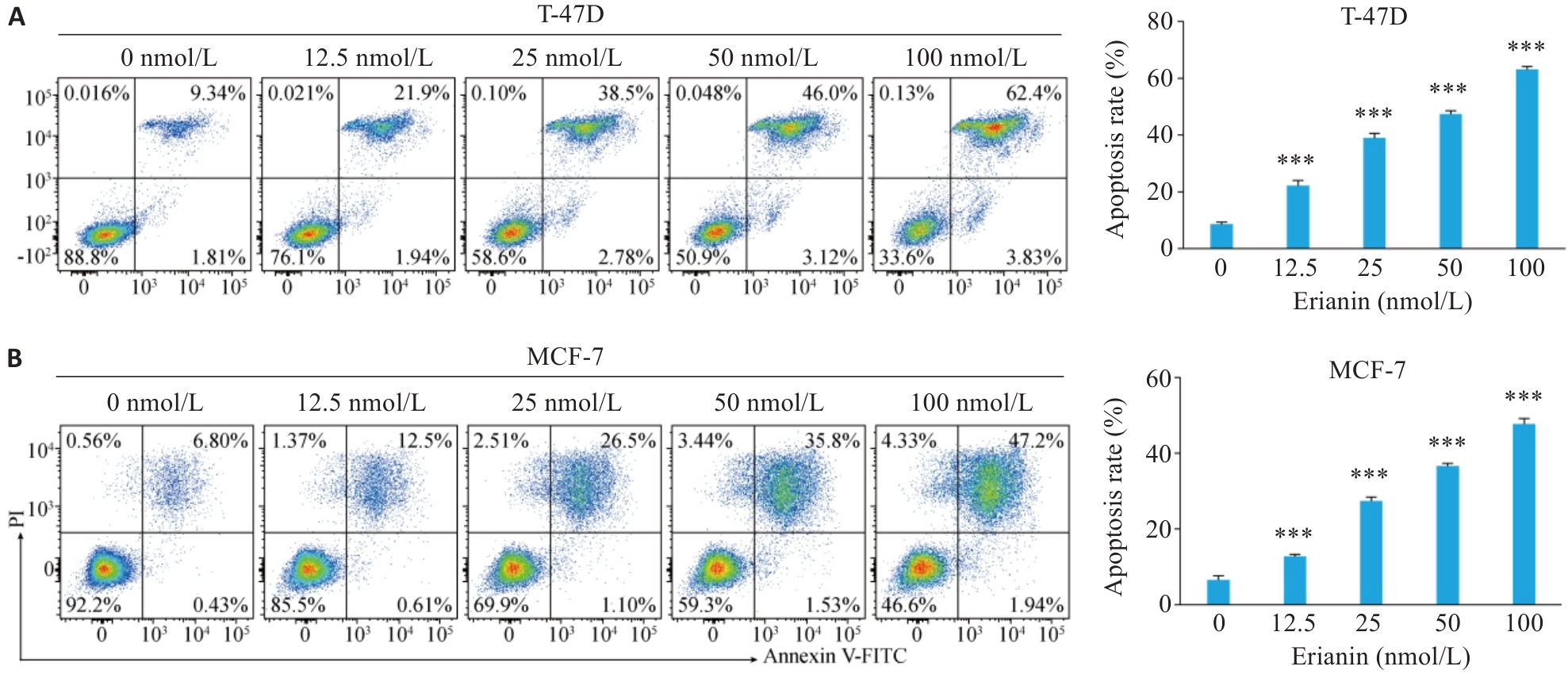
图8 毛兰素对T-47D和MCF-7细胞凋亡的影响
Fig.8 Effect of erianin on apoptosis of T-47D and MCF-7 cells. A: Apoptosis rate of T-47D cells treated with different concentrations of erianin for 48 h analyzed by flow cytometry. B: Apoptosis rate of MCF-7 cells treated with different concentrations of erianin for 48 h analyzed by flow cytometry. Data are presented as Mean±SD (n=3). ***P<0.001 vs 0 nmol/L.
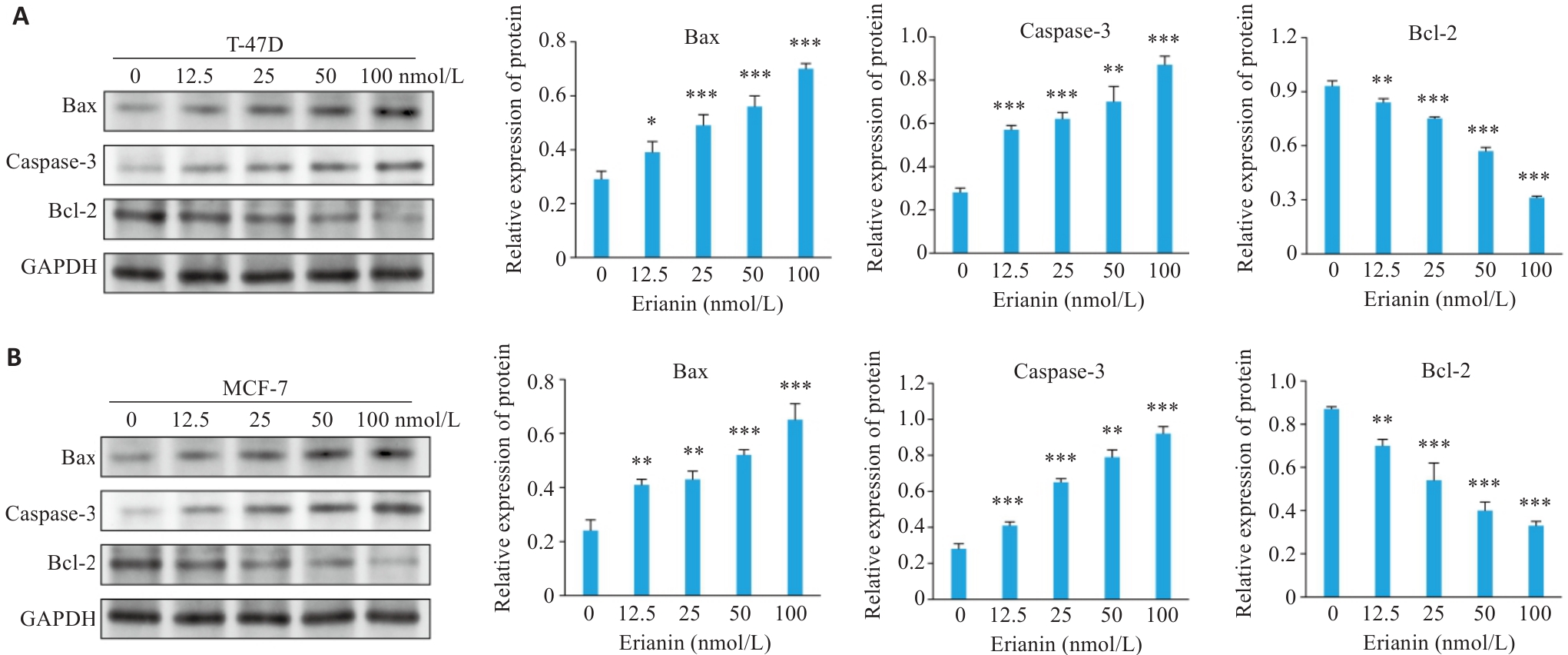
图9 毛兰素对乳腺癌细胞中凋亡相关蛋白表达水平影响
Fig.9 Effect of erianin on expression levels of apoptosis-related proteins in breast cancer cells. A: Western blotting of Bax, caspase-3, and Bcl-2 expression in T-47D cells treated with erianin. B: Western blotting of Bax, caspase-3, and Bcl-2 expression in MCF-7 cells treated with erianin. Data are presented as Mean±SD (n=3). *P<0.05, **P<0.01, ***P<0.001 vs 0 nmol/L.
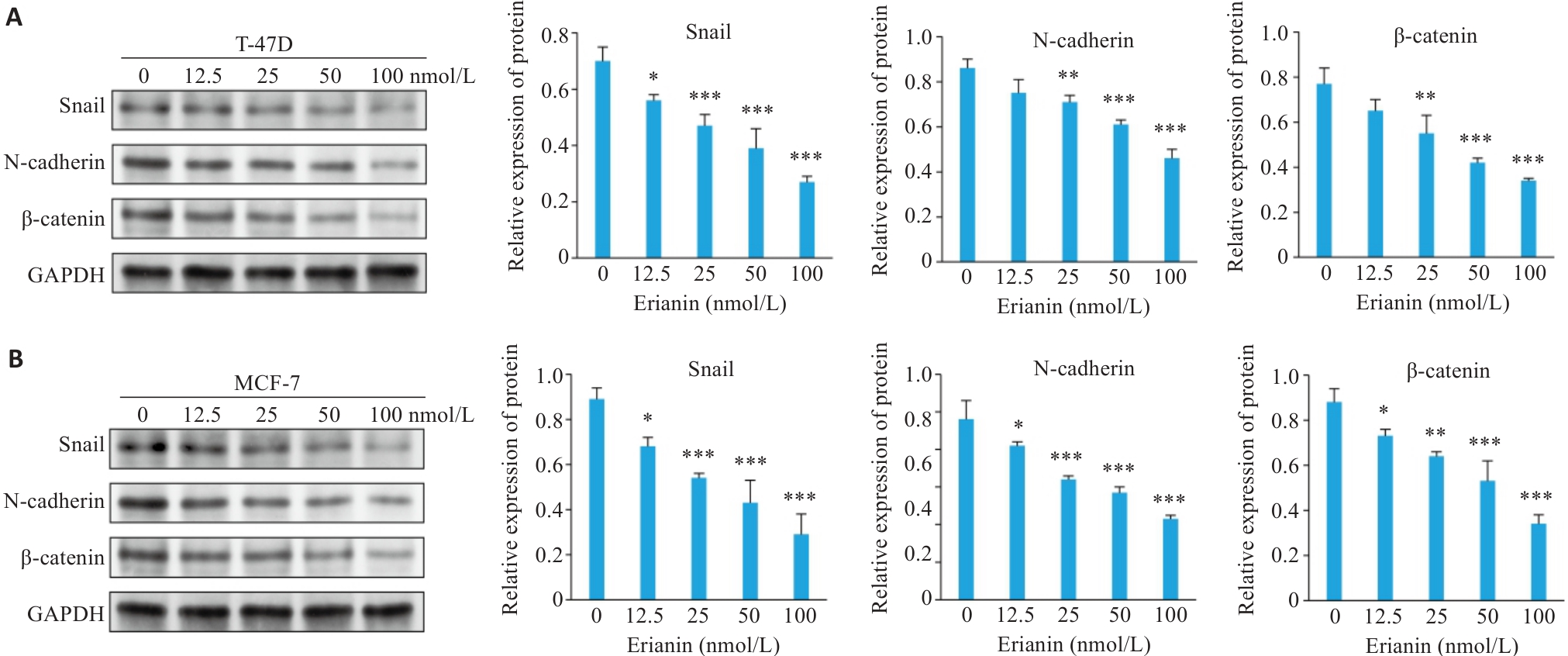
图10 毛兰素对乳腺癌细胞中EMT标志蛋白表达水平影响
Fig.10 Effect of erianin on expression levels of EMT marker proteins in breast cancer cells. A: Western blotting of Snail, N-cadherin, and β-catenin expression in T-47D cells treated with erianin. B: Western blotting of Snail, N-cadherin, and β-catenin expression in MCF-7 cells treated with erianin. Data are presented as Mean±SD (n=3). *P<0.05, **P<0.01, ***P<0.001 vs 0 nmol/L.
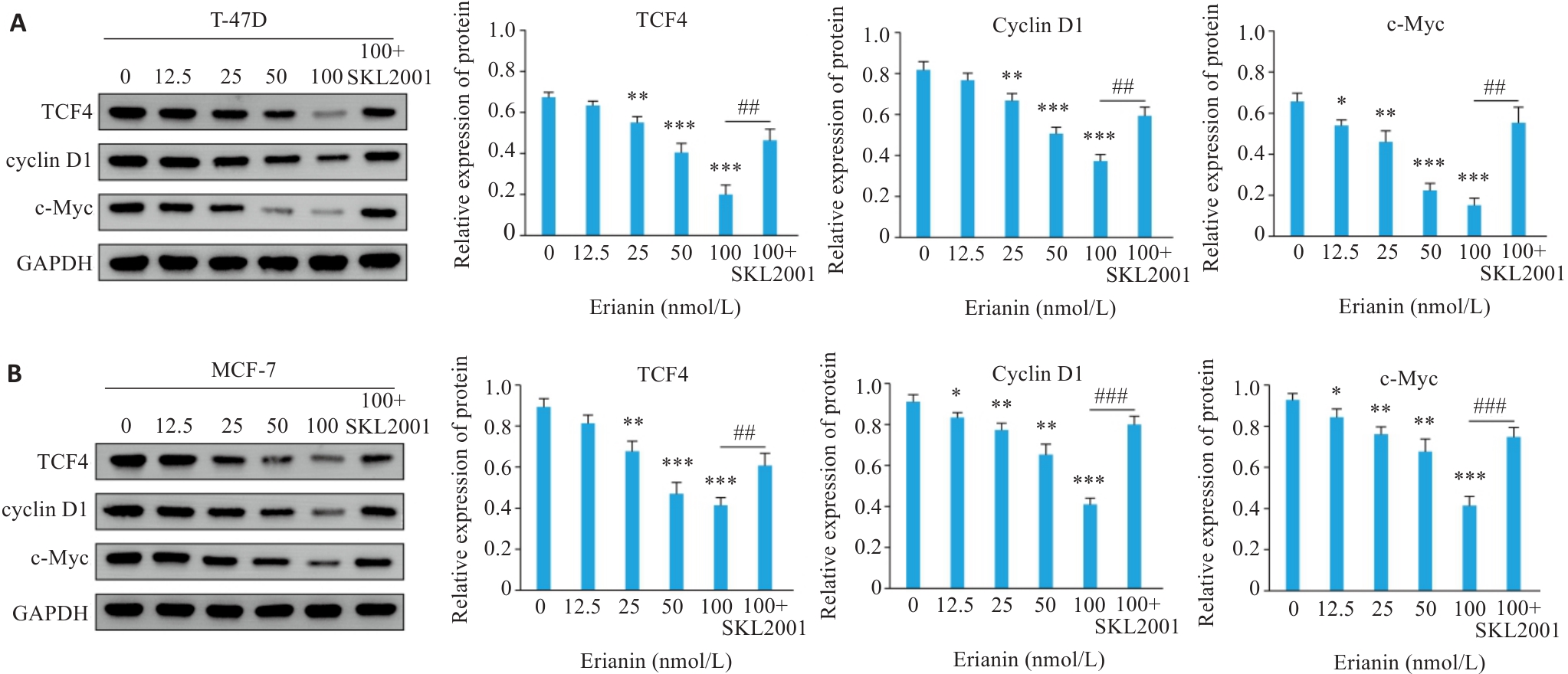
图11 毛兰素对乳腺癌细胞Wnt/β-catenin信号的影响
Fig.11 Effect of erianin on the Wnt/β-catenin signaling pathway in breast cancer cells. A: Western blotting of TCF4, Cyclin D1, and c-Myc expression in T-47D cells treated with erianin. B: Western blotting of TCF4, Cyclin D1, and c-Myc expression in MCF-7 cells treated with erianin. Data are presented as Mean±SD (n=3). *P<0.05, **P<0.01, ***P<0.001 vs 0 nmol/L. ##P<0.01, ###P<0.001.
| [1] | Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2024, 74(3): 229-63. doi:10.3322/caac.21834 |
| [2] | Zhang X. Molecular classification of breast cancer: relevance and challenges[J]. Arch Pathol Lab Med, 2023, 147(1): 46-51. doi:10.5858/arpa.2022-0070-ra |
| [3] | Gadaleta E, Thorn GJ, Ross-Adams H, et al. Field cancerization in breast cancer[J]. J Pathol, 2022, 257(4): 561-74. doi:10.1002/path.5902 |
| [4] | Mortezaee K, Ahmadi A, Haghi-Aminjan H, et al. Thyroid function following breast cancer chemotherapy: a systematic review[J]. J Cell Biochem, 2019, 120(8): 12101-7. doi:10.1002/jcb.28771 |
| [5] | Giaquinto AN, Sung H, Newman LA, et al. Breast cancer statistics 2024[J]. CA A Cancer J Clinicians, 2024, 74(6): 477-95. doi:10.3322/caac.21863 |
| [6] | Yan L, Zhang Z, Liu Y, et al. Anticancer activity of erianin: cancer-specific target prediction based on network pharmacology[J]. Front Mol Biosci, 2022, 9: 862932. doi:10.3389/fmolb.2022.862932 |
| [7] | Zheng YT, Zheng YL, Chen HP, et al. Erianin triggers ferroptosis in colorectal cancer cells by facilitating the ubiquitination and degradation of GPX4[J]. Phytomedicine, 2025, 139: 156465. doi:10.1016/j.phymed.2025.156465 |
| [8] | Deng Y, Zhong L, Zhao Y, et al. A Dendrobium chrysotoxum extract erianin induce AML cells death by activating PPARɑ and downregulating PI3K/AKT signaling pathways[J]. Toxicon, 2025, 261: 108371. doi:10.1016/j.toxicon.2025.108371 |
| [9] | Hong J, Xie ZY, Yang FY, et al. Erianin suppresses proliferation and migration of cancer cells in a pyruvate carboxylase-dependent manner[J]. Fitoterapia, 2022, 157: 105136. doi:10.1016/j.fitote.2022.105136 |
| [10] | Li M, Kang SY, Deng XM, et al. Erianin inhibits the progression of triple-negative breast cancer by suppressing SRC-mediated cholesterol metabolism[J]. Cancer Cell Int, 2024, 24(1): 166. doi:10.1186/s12935-024-03332-2 |
| [11] | Zheng JH, He WL, Chen Y, et al. Erianin serves as an NFATc1 inhibitor to prevent breast cancer-induced osteoclastogenesis and bone destruction[J]. J Adv Res, 2025, 69: 399-411. doi:10.1016/j.jare.2025.09.035 |
| [12] | Song P, Gao ZR, Bao YG, et al. Wnt/β-catenin signaling pathway in carcinogenesis and cancer therapy[J]. J Hematol Oncol, 2024, 17(1): 46. doi:10.1186/s13045-024-01563-4 |
| [13] | Li H, Li J, Zhang Y, et al. The therapeutic effect of traditional Chinese medicine on breast cancer through modulation of the Wnt/ β‑catenin signaling pathway[J]. Front Pharmacol, 2024, 15: 1401979. doi:10.3389/fphar.2024.1401979 |
| [14] | Saini H, Basu P, Nesari T, et al. Therapeutic and pharmacological efficacy of plant-derived bioactive compounds in targeting breast cancer[J]. Am J Transl Res, 2024, 16(5): 1499-520. doi:10.62347/nuzn4999 |
| [15] | Khan AW, Farooq M, Haseeb M, et al. Role of plant-derived active constituents in cancer treatment and their mechanisms of action[J]. Cells, 2022, 11(8): 1326. doi:10.3390/cells11081326 |
| [16] | Li G, Zhang H, Lai H, et al. Erianin: a phytoestrogen with therapeutic potential[J]. Front Pharmacol, 2023, 14: 1197056. doi:10.3389/fphar.2023.1197056 |
| [17] | Liu RX, Qiu MH, Deng XX, et al. Erianin inhibits the progression of pancreatic cancer by directly targeting AKT and ASK1[J]. Cancer Cell Int, 2024, 24(1): 348. doi:10.1186/s12935-024-03533-9 |
| [18] | Wei X, Liu Q, Liu L, et al. Discovery of the natural bibenzyl compound erianin in Dendrobium inhibiting the growth and EMT of gastric cancer through downregulating the LKB1-SIK2/3-PARD3 pathway[J]. Int J Mol Sci, 2024, 25(14): 7973. doi:10.3390/ijms25147973 |
| [19] | Tang LX, Ruan YL, Wang BB, et al. Erianin inhibits the progression of DDP-resistant lung adenocarcinoma by regulating the Wnt/β-catenin pathway and activating the caspase-3 for apoptosis in vitro and in vivo [J]. Hereditas, 2024, 161(1): 48. doi:10.1186/s41065-024-00351-x |
| [20] | Morana O, Wood W, Gregory CD. The apoptosis paradox in cancer[J]. Int J Mol Sci, 2022, 23(3): 1328. doi:10.3390/ijms23031328 |
| [21] | Al-Zubaidy HFS, Majeed SR, Al-Koofee DAF. Evaluation of Bax and BCL 2 genes polymorphisms in Iraqi women with breast cancer[J]. Arch Razi Inst, 2022, 77(2):799-808. doi:10.22092/ARI.2022.357090.1968 |
| [22] | Dou H, Yu PY, Liu YQ, et al. Recent advances in caspase-3, breast cancer, and traditional Chinese medicine: a review[J]. J Chemother, 2024, 36(5): 370-88. doi:10.1080/1120009x.2023.2278014 |
| [23] | Tan Y, Luo X, Lv W, et al. Tumor-derived exosomal components: the multifaceted roles and mechanisms in breast cancer metastasis[J]. Cell Death Dis, 2021, 12(6): 547. doi:10.1038/s41419-021-03825-2 |
| [24] | Park M, Kim D, Ko S, et al. Breast cancer metastasis: mechanisms and therapeutic implications[J]. Int J Mol Sci, 2022, 23(12): 6806. doi:10.3390/ijms23126806 |
| [25] | Brabletz S, Schuhwerk H, Brabletz T, et al. Dynamic EMT: a multi‑ tool for tumor progression[J]. EMBO J, 2021, 40(18): 108647. doi:10.15252/embj.2021108647 |
| [26] | Kong L, He Q, Ma D, et al. Ezetimibe inhibits the migration and invasion of triple-negative breast cancer cells by targeting TGFβ2 and EMT[J]. FEBS Open Bio, 2024, 14(5): 831-42. doi:10.1002/2211-5463.13797 |
| [27] | Park SJ, Jung HJ. Bufotalin suppresses proliferation and metastasis of triple-negative breast cancer cells by promoting apoptosis and inhibiting the STAT3/EMT axis[J]. Molecules, 2023, 28(19): 6783. doi:10.3390/molecules28196783 |
| [28] | Wu XZ, Que HY, Li QF, et al. Wnt/β‑catenin mediated signaling pathways in cancer: recent advances, and applications in cancer therapy[J]. Mol Cancer, 2025, 24(1): 171. doi:10.1186/s12943-025-02363-1 |
| [29] | Xue WH, Yang L, Chen CX, et al. Wnt/β‑catenin-driven EMT regulation in human cancers[J]. Cell Mol Life Sci, 2024, 81(1): 79. doi:10.1007/s00018-023-05099-7 |
| [30] | Yu P, Xu TT, Ma WM, et al. PRMT6-mediated transcriptional activation of ythdf2 promotes glioblastoma migration, invasion, and emt via the wnt-β-catenin pathway[J]. J Exp Clin Cancer Res, 2024, 43(1): 116. doi:10.1186/s13046-024-03038-3 |
| [31] | Luo P, Huang C, Guo J, et al. Network pharmacology and anticancer mechanism study of Dendrobium nobile dendrobine in the treatment of colorectal cancer[J]. Discov Oncol, 2024, 15(1): 768. doi:10.1007/s12672-024-01641-6 |
| [32] | Kang H, Sun Y, Hu X, et al. Gigantol inhibits proliferation and enhanced oxidative stress-mediated apoptosis through modulating of Wnt/β‑catenin signaling pathway in HeLa cells[J]. J Biochem Mol Toxicol, 2022, 36(1): e22944. doi:10.1002/jbt.22944 |
| [33] | Li H, Wang Z, Chen X, et al. Resveratrol downregulated PRDX4 expression to inhibit the progression of renal cell carcinoma via Wnt/β-catenin pathway[J]. Food Sci Nutr, 2025, 13(6): e70352. doi:10.1002/fsn3.70352 |
| [34] | Ma QM, Yu JL, Zhang X, et al. Wnt/β-catenin signaling pathway-a versatile player in apoptosis and autophagy[J]. Biochimie, 2023, 211: 57-67. doi:10.1016/j.biochi.2023.03.001 |
| [35] | Albalawi YA, Narasipura SD, Al-Harthi L. Wnt/β‑catenin protects lymphocytes from HIV-mediated apoptosis via induction of bcl-xL[J]. Viruses, 2022, 14(7): 1469. doi:10.3390/v14071469 |
| [36] | Wang Y, He J, Zhang JW, et al. Cell migration induces apoptosis in osteosarcoma cell via inhibition of Wnt-β-catenin signaling pathway[J]. Colloids Surf B Biointerfaces, 2023, 223: 113142. doi:10.1016/j.colsurfb.2023.113142 |
| [1] | 梁芷晴, 潘富珍, 邓利强, 麦哲芬, 马云, 施传坚, 付卫明. 生脉散通过调控乳酸/Wnt/β-catenin/LDHA通路改善非小细胞肺癌奥希替尼耐药的作用机制[J]. 南方医科大学学报, 2026, 46(3): 523-531. |
| [2] | 张力莹, 张同贞, 赵鑫. 基于乳腺影像报告和数据系统的DWI和T2WI形态评估对乳腺病变的诊断价值[J]. 南方医科大学学报, 2025, 45(9): 1809-1817. |
| [3] | 马思源, 张博超, 浦春. Circ_0000437通过靶向let-7b-5p/CTPS1轴促进乳腺癌细胞的增殖、侵袭、迁移及上皮间质转化[J]. 南方医科大学学报, 2025, 45(8): 1682-1696. |
| [4] | 张兆君, 吴琼, 谢苗苗, 叶洳吟, 耿晨晨, 石纪雯, 杨清玲, 王文锐, 石玉荣. 层状双氢氧化物负载si-NEAT1通过miR-133b/PD-L1轴调控乳腺癌紫杉醇耐药及巨噬细胞极化[J]. 南方医科大学学报, 2025, 45(8): 1718-1731. |
| [5] | 李嘉豪, 冼瑞婷, 李荣. 下调ACADM介导的脂毒性抑制雌激素受体阳性乳腺癌细胞的侵袭与转移[J]. 南方医科大学学报, 2025, 45(6): 1163-1173. |
| [6] | 陈镝, 吕莹, 郭怡欣, 张怡荣, 王蕊璇, 周小若, 陈雨欣, 武晓慧. 双氢青蒿素可显著增强阿霉素诱导的三阴性乳腺癌细胞凋亡:基于负向调控STAT3/HIF-1α通路[J]. 南方医科大学学报, 2025, 45(2): 254-260. |
| [7] | 张芡, 刘博文, 雷丽, 王晔, 张馨月, 毛樟坤, 唐鹏, 张金梅, 杨佳宜, 彭彦茜, 刘泽. 丝氨酸蛋白酶抑制剂E1过表达通过诱导M2型巨噬细胞极化促进三阴性乳腺癌细胞增殖与紫杉醇耐药[J]. 南方医科大学学报, 2025, 45(12): 2551-2560. |
| [8] | 孙心悦, 王宽宇, 王钢, 代清泉, 陈静, 孔祥定, 栾佳. 扶正消岩颗粒通过调控AKT1/BAD/BCL-2通路改善乳腺癌化疗期癌因性疲乏[J]. 南方医科大学学报, 2025, 45(12): 2646-2657. |
| [9] | 张明亮, 孙非凡, 韩卓琪, 高越, 罗毅. HOTAIR rs920778多态性调控乳腺癌易感性及HER2靶向治疗耐药的临床研究[J]. 南方医科大学学报, 2025, 45(10): 2270-2276. |
| [10] | 褚乔, 王小娜, 续佳颖, 彭荟林, 赵裕琳, 张静, 陆国玉, 王恺. 白头翁皂苷D通过多靶点和多途径抑制三阴性乳腺癌侵袭转移[J]. 南方医科大学学报, 2025, 45(1): 150-161. |
| [11] | 薛良军, 谈秋瑜, 许静文, 冯璐, 李文锦, 颜亮, 李玉磊. MiR-6838-5p过表达下调DDR1基因表达抑制乳腺癌MCF-7细胞的增殖[J]. 南方医科大学学报, 2024, 44(9): 1677-1684. |
| [12] | 欧阳明子, 崔佳琦, 王慧, 梁正, 皮大锦, 陈利国, 陈前军, 吴迎朝. 开心散通过减轻前额叶皮质铁死亡缓解小鼠的阿霉素化疗性抑郁[J]. 南方医科大学学报, 2024, 44(8): 1441-1449. |
| [13] | 房锦存, 刘立威, 林俊豪, 陈逢生. CDHR2过表达通过抑制PI3K/Akt通路抑制乳腺癌细胞增殖[J]. 南方医科大学学报, 2024, 44(6): 1117-1125. |
| [14] | 崔芝, 马萃娇, 王倩茹, 陈金豪, 严子阳, 杨建林, 吕亚丰, 曹春雨. 表达 TGF-βⅡ受体的腺相关病毒载体抑制小鼠三阴性乳腺癌4T1细胞的增殖和肺转移[J]. 南方医科大学学报, 2024, 44(5): 818-826. |
| [15] | 曾佑琴, 陈思雨, 刘燕, 刘奕彤, 张玲, 夏姣, 吴心语, 魏常友, 冷平. AKBA联合阿霉素抑制三阴性乳腺癌细胞MDA-MB-231的增殖、迁移和裸鼠移植瘤生长[J]. 南方医科大学学报, 2024, 44(12): 2449-2460. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||