南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 825-837.doi: 10.12122/j.issn.1673-4254.2026.04.11
• • 上一篇
金成桓1,2( ), 洪宗超1,2(
), 洪宗超1,2( ), 段雪云3, 范恒4, 杨晶鑫1,2, 秦佳美2, 邹丽艳2, 覃孟渊5
), 段雪云3, 范恒4, 杨晶鑫1,2, 秦佳美2, 邹丽艳2, 覃孟渊5
收稿日期:2025-09-25
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
洪宗超
E-mail:1659939107@qq.com;1286069643@qq.com
作者简介:金成桓,在读硕士研究生,E-mail: 1659939107@qq.com
基金资助:
Chenghuan JIN1,2( ), Zongchao HONG1,2(
), Zongchao HONG1,2( ), Xueyun DUAN3, Heng FAN4, Jingxing YANG1,2, Jiamei QIN2, Liyan ZOU2, Mengyuan QIN5
), Xueyun DUAN3, Heng FAN4, Jingxing YANG1,2, Jiamei QIN2, Liyan ZOU2, Mengyuan QIN5
Received:2025-09-25
Online:2026-04-20
Published:2026-04-24
Contact:
Zongchao HONG
E-mail:1659939107@qq.com;1286069643@qq.com
Supported by:摘要:
目的 探讨化结消瘤方(HJF)及其活性成分熊果酸(Ursolic acid)通过铜死亡抗结直肠癌(CRC)的分子机制。 方法 采用蛋白质组学分析化结消瘤方对结直肠癌蛋白表达谱的影响;血清药物化学探索化结消瘤方抗结直肠癌的潜在活性物质;通过网络药理学和分子对接预测熊果酸与铜死亡相关靶点的相互作用;采用MTT、划痕、克隆形成、Western blotting等细胞生物学实验验证熊果酸对HCT-116和LOVO细胞增殖、迁移及铁氧还蛋白1(FDX1)、溶质载体家族31成员1(SLC31A1)、二氢硫辛酸转乙酰基酶(DLAT)、谷胱甘肽(GSH)、丙二醛(MDA)、丙酮酸、Cu2+等铜死亡相关指标的影响。 结果 化结消瘤方调控628个差异表达蛋白,涉及炎症、免疫、代谢等通路;熊果酸为其主要入血成分,与铜死亡关键蛋白FDX1具有强结合潜力(对接分数106.813),可抑制结肠癌细胞增殖与迁移,诱导Cu2+(P<0.05)、MDA(P<0.05)和丙酮酸(P<0.01)积累,降低GSH水平(P<0.01),抑制DLAT表达并上调FDX1和SLC31A1表达。 结论 熊果酸作为化结消瘤方发挥抗结直肠癌作用的关键活性成分之一,通过靶向FDX1在结直肠癌细胞中诱导铜死亡,从而发挥抗肿瘤作用。
金成桓, 洪宗超, 段雪云, 范恒, 杨晶鑫, 秦佳美, 邹丽艳, 覃孟渊. 化结消瘤方中的熊果酸通过诱导铜死亡抑制结直肠癌细胞生长[J]. 南方医科大学学报, 2026, 46(4): 825-837.
Chenghuan JIN, Zongchao HONG, Xueyun DUAN, Heng FAN, Jingxing YANG, Jiamei QIN, Liyan ZOU, Mengyuan QIN. Ursolic acid in Huajie Xiaoliu Formula inhibits colorectal cancer cell growth by inducing cuproptosis[J]. Journal of Southern Medical University, 2026, 46(4): 825-837.
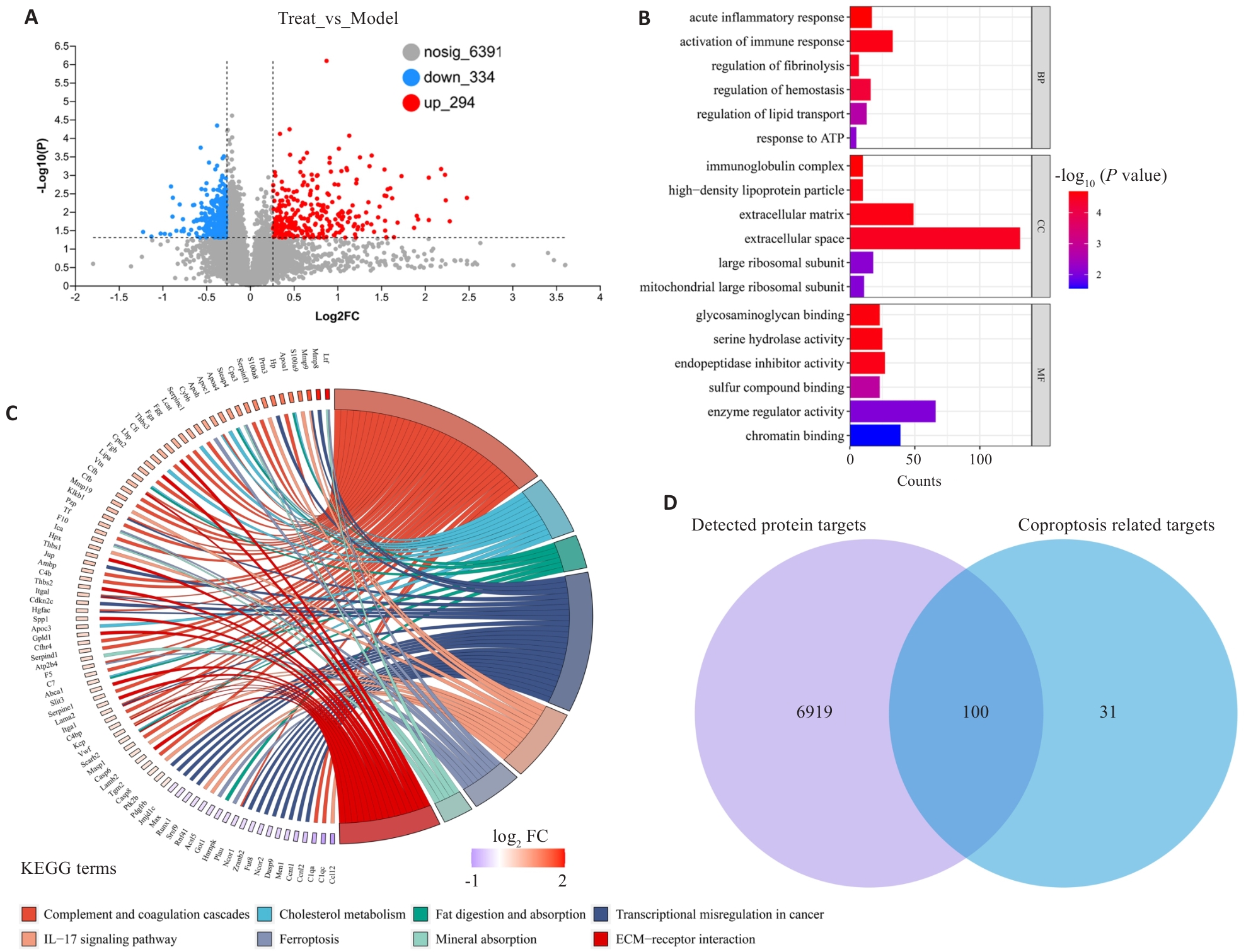
图1 化结消瘤方抗结直肠癌的蛋白组学研究
Fig.1 Proteomic study of HJF in treatment for CRC. A: Volcano plot for high-throughput detection of protein expression, including 334 downregulated proteins (blue) and 294 upregulated proteins (red); B: Partial results of GO enrichment of the differentially expressed proteins. C: Partial results of KEGG enrichment of the differentially expressed proteins. D: Intersection diagram of the differentially expressed proteins and cuproptosis-related proteins detected by proteomics, showing a total of 100 proteins related to cuproptosis.
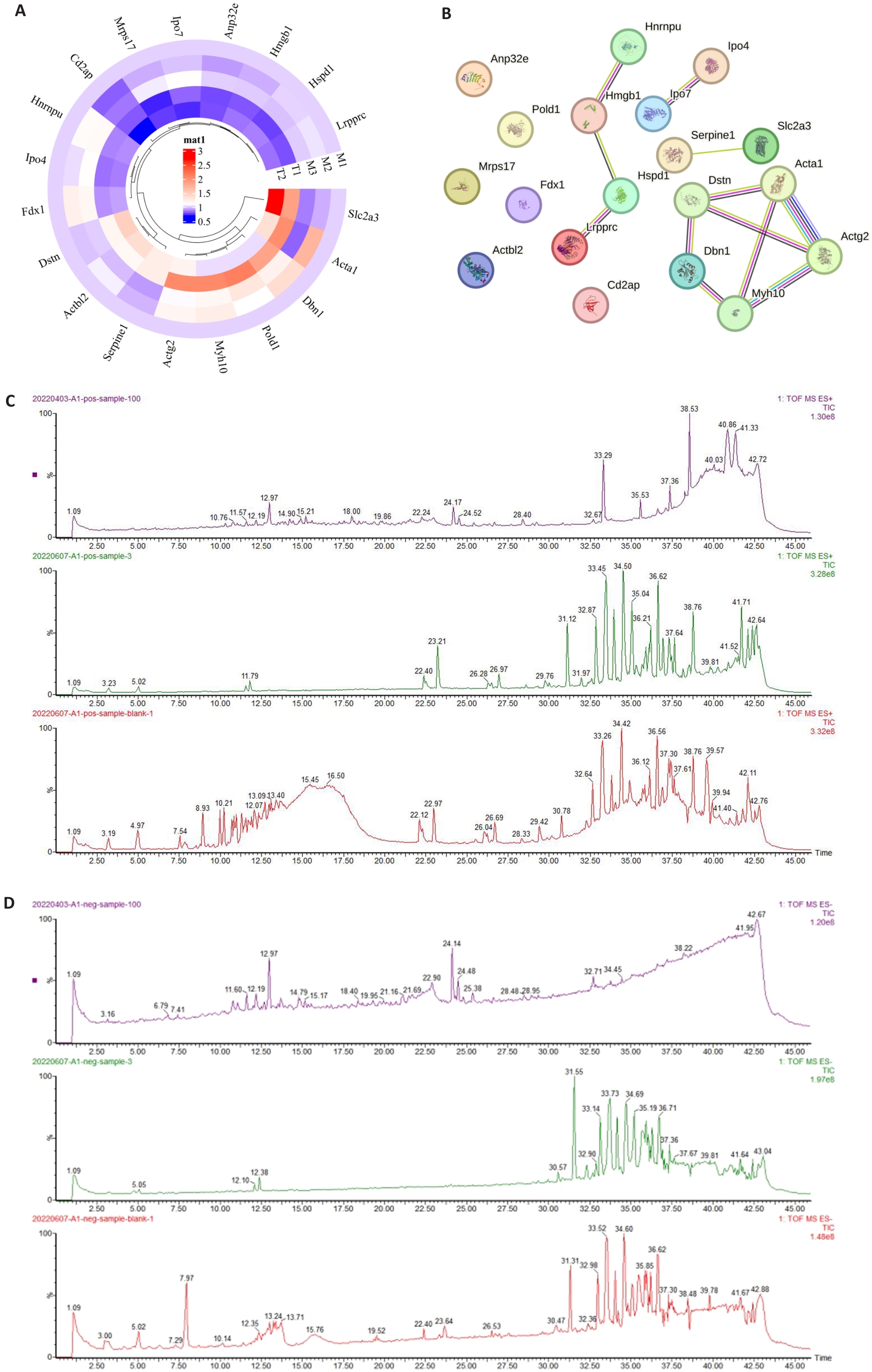
图2 铜死亡相关差异表达蛋白及LC-MS/MS分析谱图
Fig.2 Differential expression of cuproptosis-related proteins and LC-MS/MS analysis spectrum. A: Heat map of differential expression of 19 cuproptosis-related proteins. B: Display of interactions between 19 cuproptosis-related proteins. C: TIC spectrum of LC-MS/MS in positive ion mode (from top to bottom: LC-MS/MS analysis results of blank serum, drug-containing serum, and HJF). D: TIC spectrum of LC-MS/MS in negative ion mode (from top to bottom: LC-MS/MS analysis results of blank serum, drug-containing serum, and HJF).
| No. | RT (min) | Name | Formula | Theoretical molecular weight | Measured molecular weight |
|---|---|---|---|---|---|
| 1 | 3.41 | Protocatechuic acid | C7H6O4 | 154.0266 | [M-H]- 153.0170 |
| 2 | 4.71 | Deacetylasperulosidic acid methyl ester | C17H24O11 | 404.1319 | [M+Na]+ 427.1221 [M-H]- 403.1210 |
| 3 | 6.36 | Chlorogenic acid | C16H18O9 | 354.0951 | [M+H]+ 355.1083 [M-H]- 353.0847 |
| 4 | 6.88 | Asperulosidic acid | C18H24O12 | 432.1268 | [M+Na]+ 455.1141 [M-H]- 431.1172 |
| 5 | 9.98 | Ferulic acid | C10H10O4 | 194.0579 | [M+H]+ 195.0636 [M-H]- 193.0475 |
| 6 | 10.86 | Calycosin-7-O-β-D-glucoside | C22H22O10 | 446.1213 | [M+H]+ 447.1302 [M-H]- 445.1112 |
| 7 | 11.64 | Scutellarin | C21H18O12 | 462.0798 | [M+H]+ 463.0850 [M-H]- 461.0727 |
| 8 | 12.07 | Rutoside | C27H30O16 | 610.1534 | [M+H]+ 611.1584 [M-H]- 609.1432 |
| 9 | 13.00 | Hesperidin | C28H34O15 | 610.1898 | [M+H]+ 611.1989 [M-H]- 609.1786 |
| 10 | 13.78 | Isoliquiritin | C21H22O9 | 418.1264 | [M+H]+ 419.1331 [M-H]- 417.1166 |
| 11 | 13.96 | Ononin | C22H22O9 | 430.1264 | [M+H]+ 431.1321 [M-H]- 429.1211 |
| 12 | 15.26 | 10-Deacetylbaccatin III | C29H36O10 | 544.2308 | [M+H]+ 545.2370 [M-H]- 543.2204 |
| 13 | 15.41 | Calycosin | C16H12O5 | 284.0685 | [M+H]+ 285.0760 [M-H]- 283.0602 |
| 14 | 18.62 | Formononetin | C16H12O4 | 268.0736 | [M+H]+ 269.0824 [M-H]- 267.0657 |
| 15 | 19.83 | Heterophyllin B | C40H58N8O8 | 778.4378 | [M+H]+ 779.4432 [M-H]- 777.4281 |
| 16 | 20.22 | 6-Gingerol | C17H26O4 | 294.1831 | [M+Na]+ 317.1717 |
| 17 | 23.40 | Paclitaxel | C47H51NO14 | 853.3310 | [M+H]+ 854.3370 [M-H]- 852.3248 |
| 18 | 24.21 | Glycyrrhizic acid | C42H62O16 | 822.4038 | [M+H]+ 823.4129 [M-H]- 821.3976 |
| 19 | 33.88 | Pachymic acid | C33H52O5 | 528.3815 | [M+H]+ 529.3902 [M-H]- 527.3729 |
| 20 | 35.03 | Ursolic Acid | C30H48O3 | 456.3603 | [M+H]+ 457.3694 [M-H]- 455.3524 |
表1 化结消瘤方中的20个入血成分
Tab.1 Identification of 20 metabolites of Huajie Xiaoliu Formula (HJF) in plasma by LC- MS analysis
| No. | RT (min) | Name | Formula | Theoretical molecular weight | Measured molecular weight |
|---|---|---|---|---|---|
| 1 | 3.41 | Protocatechuic acid | C7H6O4 | 154.0266 | [M-H]- 153.0170 |
| 2 | 4.71 | Deacetylasperulosidic acid methyl ester | C17H24O11 | 404.1319 | [M+Na]+ 427.1221 [M-H]- 403.1210 |
| 3 | 6.36 | Chlorogenic acid | C16H18O9 | 354.0951 | [M+H]+ 355.1083 [M-H]- 353.0847 |
| 4 | 6.88 | Asperulosidic acid | C18H24O12 | 432.1268 | [M+Na]+ 455.1141 [M-H]- 431.1172 |
| 5 | 9.98 | Ferulic acid | C10H10O4 | 194.0579 | [M+H]+ 195.0636 [M-H]- 193.0475 |
| 6 | 10.86 | Calycosin-7-O-β-D-glucoside | C22H22O10 | 446.1213 | [M+H]+ 447.1302 [M-H]- 445.1112 |
| 7 | 11.64 | Scutellarin | C21H18O12 | 462.0798 | [M+H]+ 463.0850 [M-H]- 461.0727 |
| 8 | 12.07 | Rutoside | C27H30O16 | 610.1534 | [M+H]+ 611.1584 [M-H]- 609.1432 |
| 9 | 13.00 | Hesperidin | C28H34O15 | 610.1898 | [M+H]+ 611.1989 [M-H]- 609.1786 |
| 10 | 13.78 | Isoliquiritin | C21H22O9 | 418.1264 | [M+H]+ 419.1331 [M-H]- 417.1166 |
| 11 | 13.96 | Ononin | C22H22O9 | 430.1264 | [M+H]+ 431.1321 [M-H]- 429.1211 |
| 12 | 15.26 | 10-Deacetylbaccatin III | C29H36O10 | 544.2308 | [M+H]+ 545.2370 [M-H]- 543.2204 |
| 13 | 15.41 | Calycosin | C16H12O5 | 284.0685 | [M+H]+ 285.0760 [M-H]- 283.0602 |
| 14 | 18.62 | Formononetin | C16H12O4 | 268.0736 | [M+H]+ 269.0824 [M-H]- 267.0657 |
| 15 | 19.83 | Heterophyllin B | C40H58N8O8 | 778.4378 | [M+H]+ 779.4432 [M-H]- 777.4281 |
| 16 | 20.22 | 6-Gingerol | C17H26O4 | 294.1831 | [M+Na]+ 317.1717 |
| 17 | 23.40 | Paclitaxel | C47H51NO14 | 853.3310 | [M+H]+ 854.3370 [M-H]- 852.3248 |
| 18 | 24.21 | Glycyrrhizic acid | C42H62O16 | 822.4038 | [M+H]+ 823.4129 [M-H]- 821.3976 |
| 19 | 33.88 | Pachymic acid | C33H52O5 | 528.3815 | [M+H]+ 529.3902 [M-H]- 527.3729 |
| 20 | 35.03 | Ursolic Acid | C30H48O3 | 456.3603 | [M+H]+ 457.3694 [M-H]- 455.3524 |
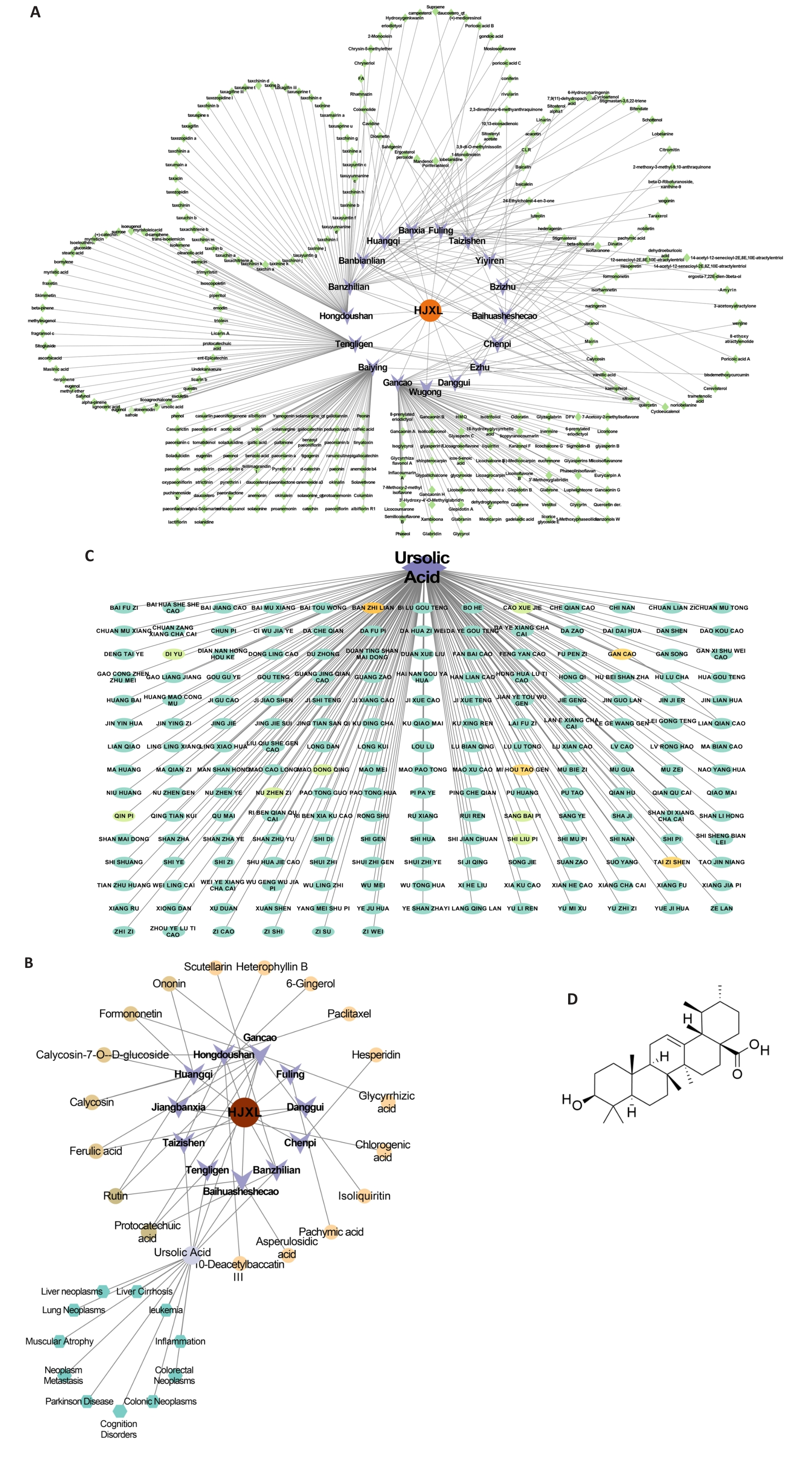
图3 化结消瘤方的网络药理学分析
Fig.3 Network pharmacology analysis related to HJF. A: Network construction results of HJFTCM-Chemical Components. B: Network construction results of HJF-TCM-Enter blood chemical components. C: Display of Chinese herbal medicines containing ursolic acid. D: Chemical structure of ursolic acid.
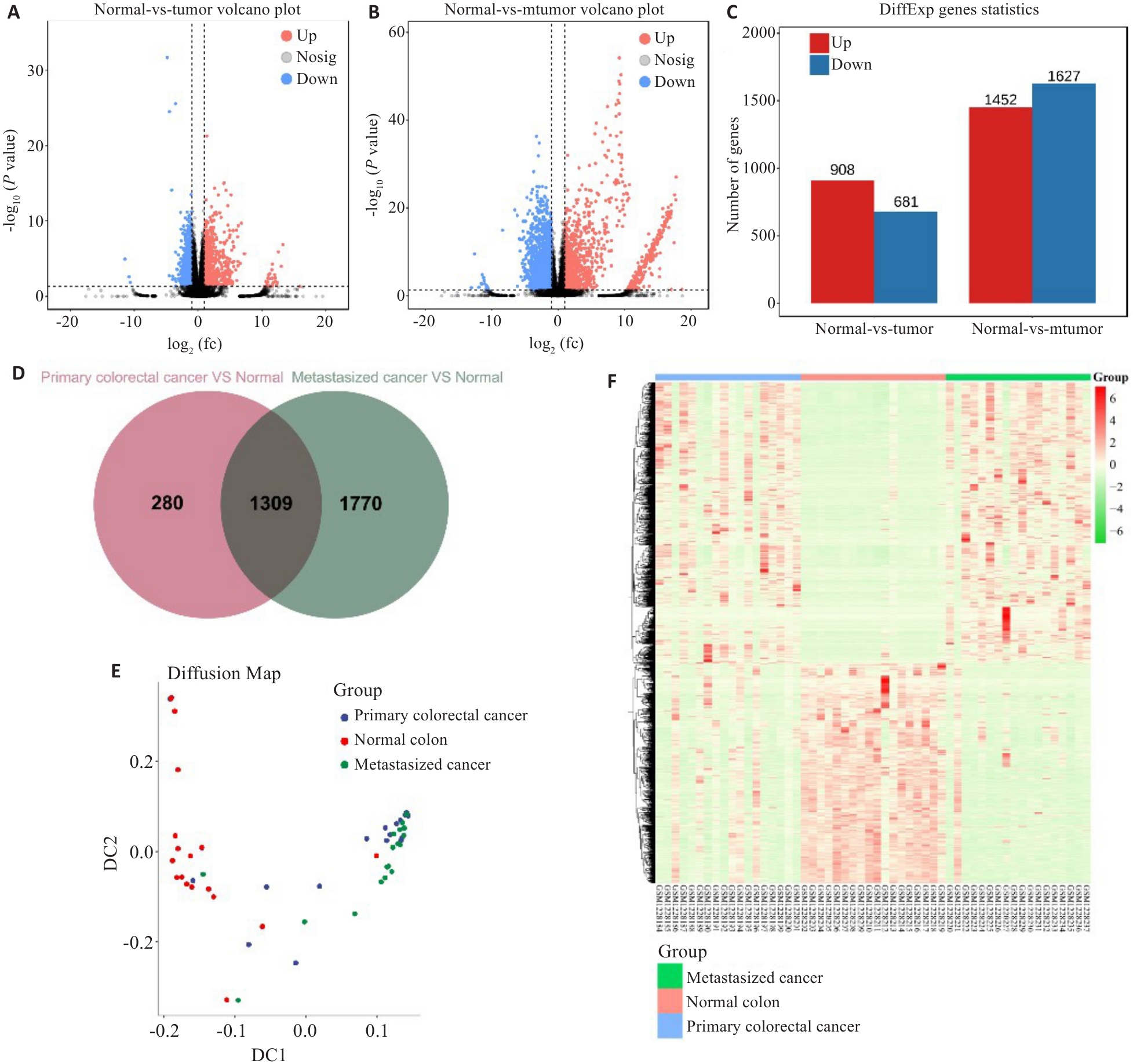
图4 公共数据集分析
Fig.4 Public data analysis. A: Volcano map of differential gene expression in primary colon cancer and normal tissue. B: Volcano map of differential gene expression in metastatic colon cancer and normal tissue. C: Statistical chart of differentially expressed genes in colon cancer. D: There are a total of 1309 differentially expressed genes in primary colon cancer and metastatic colon cancer. E: Diffusion mapping of 1309 differentially expressed genes in normal tissues, primary colon cancer, and metastatic colon cancer. F: Expression heatmap of 1309 genes in normal tissues, primary colon cancer, and metastatic colon cancer.
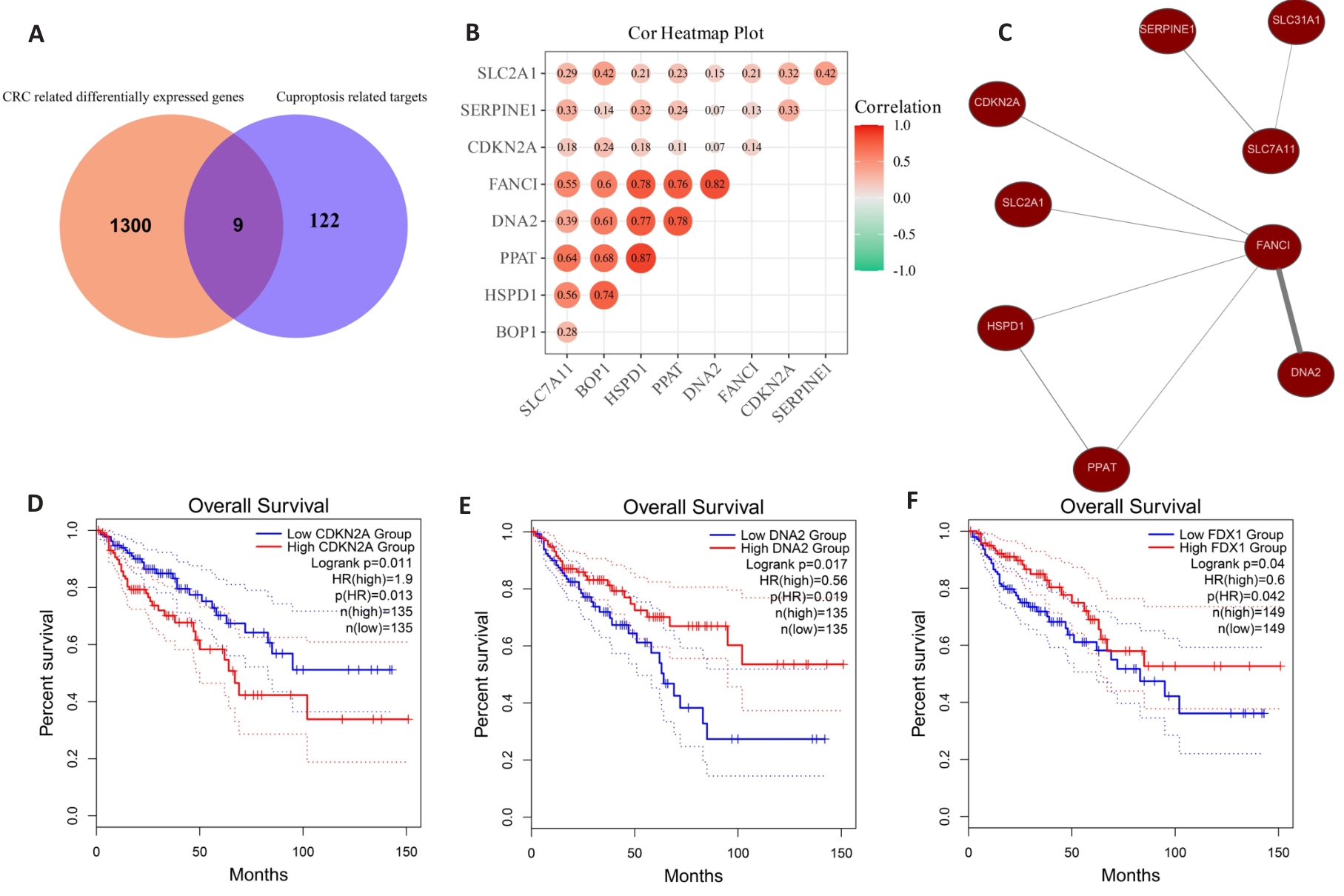
图5 公共数据集中铜死亡相关基因的筛选
Fig.5 Identification of cuproptosis-related genes in public data. A: Among the 1309 differentially expressed genes, 9 genes are associated with cuprotosis. B: Correlation heatmap of 9 cuprotosis-related genes. C: Co-expression networks of 9 cuprotosis-related genes. D: Patients with low expression of CDKN2A gene are more likely to have longer overall survival. E: Patients with high expression of DNA2 gene are more likely to have longer overall survival. F: Patients with high expression of FDX1 gene are more likely to have longer overall survival.
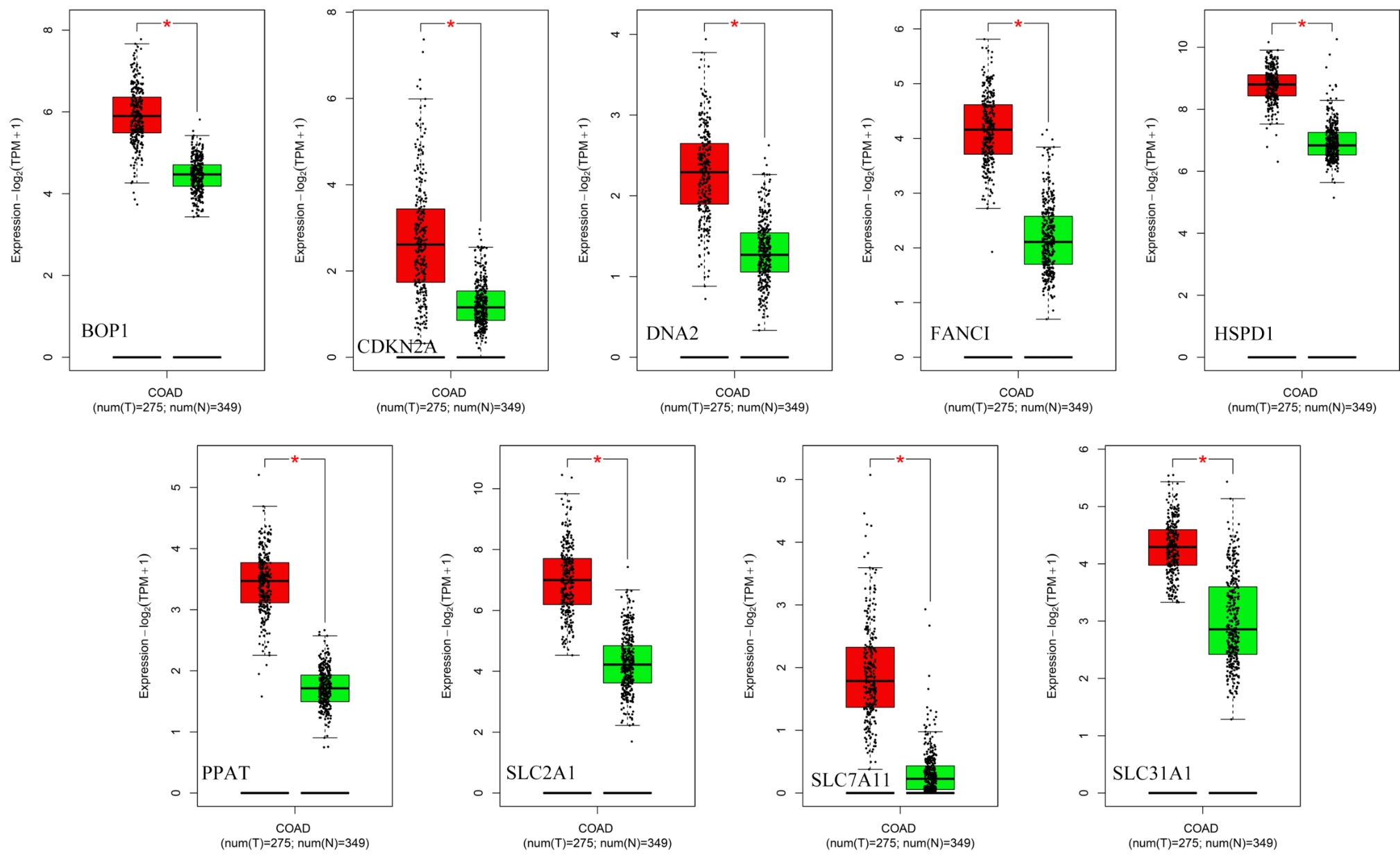
图6 基于GEPIA2验证分析结直肠癌中的基因表达差异
Fig.6 GEPIA2 analysis of genes that are significantly differentially expressed in CRC. Red color represents tumor tissue, and green color represents normal tissue.
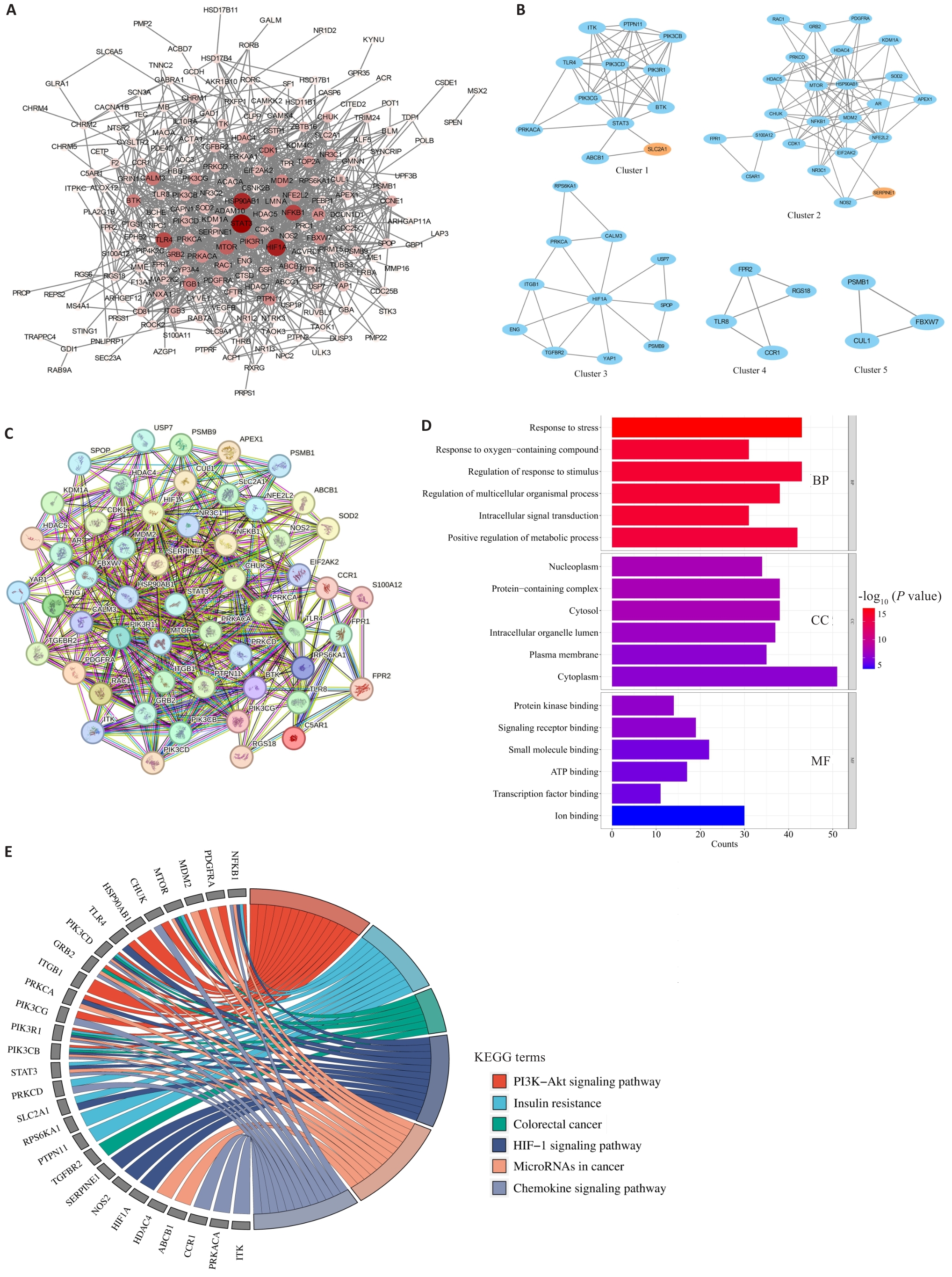
图7 熊果酸的网络药理学分析
Fig.7 Network pharmacology analysis of ursolic acid. A: PPI network of 241 predicted targets of ursolic acid. B: 5 clusters analyzed by MCODE. C: PPI network of 54 genes in 5 clusters. D: GO analysis results of 54 genes. E: KEGG analysis results of 54 genes.
| Gene cluster | Score | Node | Edge | Genes |
|---|---|---|---|---|
| Cluster 1 | 7.455 | 12 | 41 | ABCB1, BTK, ITK, PIK3CB, PIK3CD, PIK3CG, PIK3R1, PRKACA, PTPN11, SLC2A1, STAT3, TLR4 |
| Cluster 2 | 7.13 | 24 | 82 | APEX1, AR, C5AR1, CDK1, CHUK, EIF2AK2, FPR1, GRB2, HDAC4, HDAC5, HSP90AB1, KDM1A, MDM2, MTOR, NFE2L2, NFKB1, NOS2, NR3C1, PDGFRA, PRKCD, RAC1, S100A12, SERPINE1, SOD2 |
| Cluster 3 | 3.8 | 11 | 19 | CALM3, ENG, HIF1A, ITGB1, PRKCA, PSMB9, RPS6KA1, SPOP, TGFBR2, USP7, YAP1 |
| Cluster 4 | 3.333 | 4 | 5 | CCR1, FPR2, RGS18, TLR8 |
| Cluster 5 | 3 | 3 | 3 | CUL1, FBXW7, PSMB1 |
表2 预测靶点PPI网络的MCODE分析结果
Tab.2 Clusters of the predicted target in the PPI network
| Gene cluster | Score | Node | Edge | Genes |
|---|---|---|---|---|
| Cluster 1 | 7.455 | 12 | 41 | ABCB1, BTK, ITK, PIK3CB, PIK3CD, PIK3CG, PIK3R1, PRKACA, PTPN11, SLC2A1, STAT3, TLR4 |
| Cluster 2 | 7.13 | 24 | 82 | APEX1, AR, C5AR1, CDK1, CHUK, EIF2AK2, FPR1, GRB2, HDAC4, HDAC5, HSP90AB1, KDM1A, MDM2, MTOR, NFE2L2, NFKB1, NOS2, NR3C1, PDGFRA, PRKCD, RAC1, S100A12, SERPINE1, SOD2 |
| Cluster 3 | 3.8 | 11 | 19 | CALM3, ENG, HIF1A, ITGB1, PRKCA, PSMB9, RPS6KA1, SPOP, TGFBR2, USP7, YAP1 |
| Cluster 4 | 3.333 | 4 | 5 | CCR1, FPR2, RGS18, TLR8 |
| Cluster 5 | 3 | 3 | 3 | CUL1, FBXW7, PSMB1 |
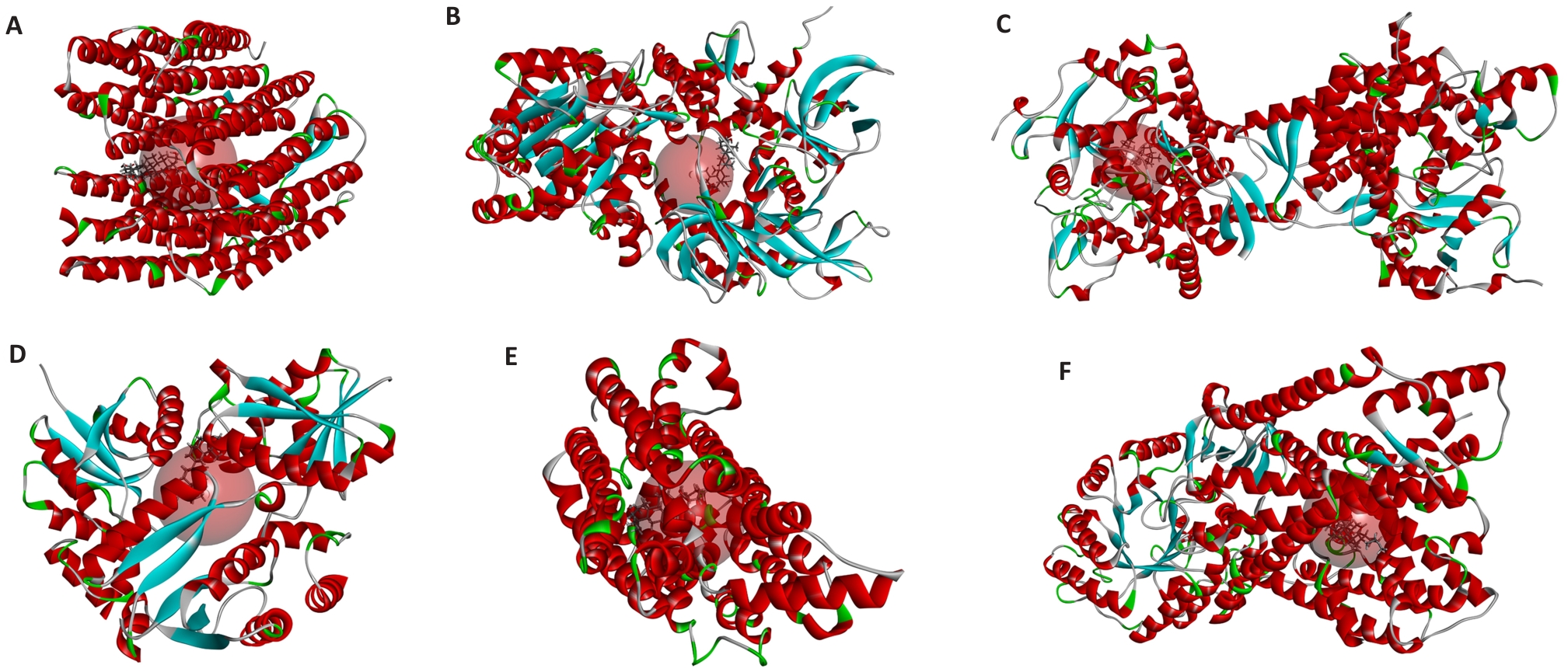
图8 熊果酸与各蛋白的分子对接结果
Fig.8 Molecular docking between ursolic acid and its targets. A: BOP1-ursolic acid; B: DNA2-ursolic acid; C: FDX1-ursolic acid; D: PPAT-ursolic acid; E: SLC2A1-ursolic acid; F: SLC7A11-ursolic acid.
| Target | PDB code | LiDock Score |
|---|---|---|
| BOP1 | 4qi1 | 78.5132 |
| CDKN2A | 7ozt | N/A |
| DNA2 | 5ean | 56.8153 |
| FANCI | 6tnf | N/A |
| FDX1 | 3n9y | 106.813 |
| HSPD1 | 6mrc | N/A |
| PPAT | 4nat | 58.2893 |
| SERPINE1 | 3cvm | N/A |
| SLC2A1 | 4pyp | 113.927 |
| SLC7A11 | 7ccs | 102.503 |
| SLC31A1 | 2ls2 | N/A |
表3 熊果酸与各靶点分子对接的得分
Tab.3 Scoring of ursolic acid-protein binding
| Target | PDB code | LiDock Score |
|---|---|---|
| BOP1 | 4qi1 | 78.5132 |
| CDKN2A | 7ozt | N/A |
| DNA2 | 5ean | 56.8153 |
| FANCI | 6tnf | N/A |
| FDX1 | 3n9y | 106.813 |
| HSPD1 | 6mrc | N/A |
| PPAT | 4nat | 58.2893 |
| SERPINE1 | 3cvm | N/A |
| SLC2A1 | 4pyp | 113.927 |
| SLC7A11 | 7ccs | 102.503 |
| SLC31A1 | 2ls2 | N/A |
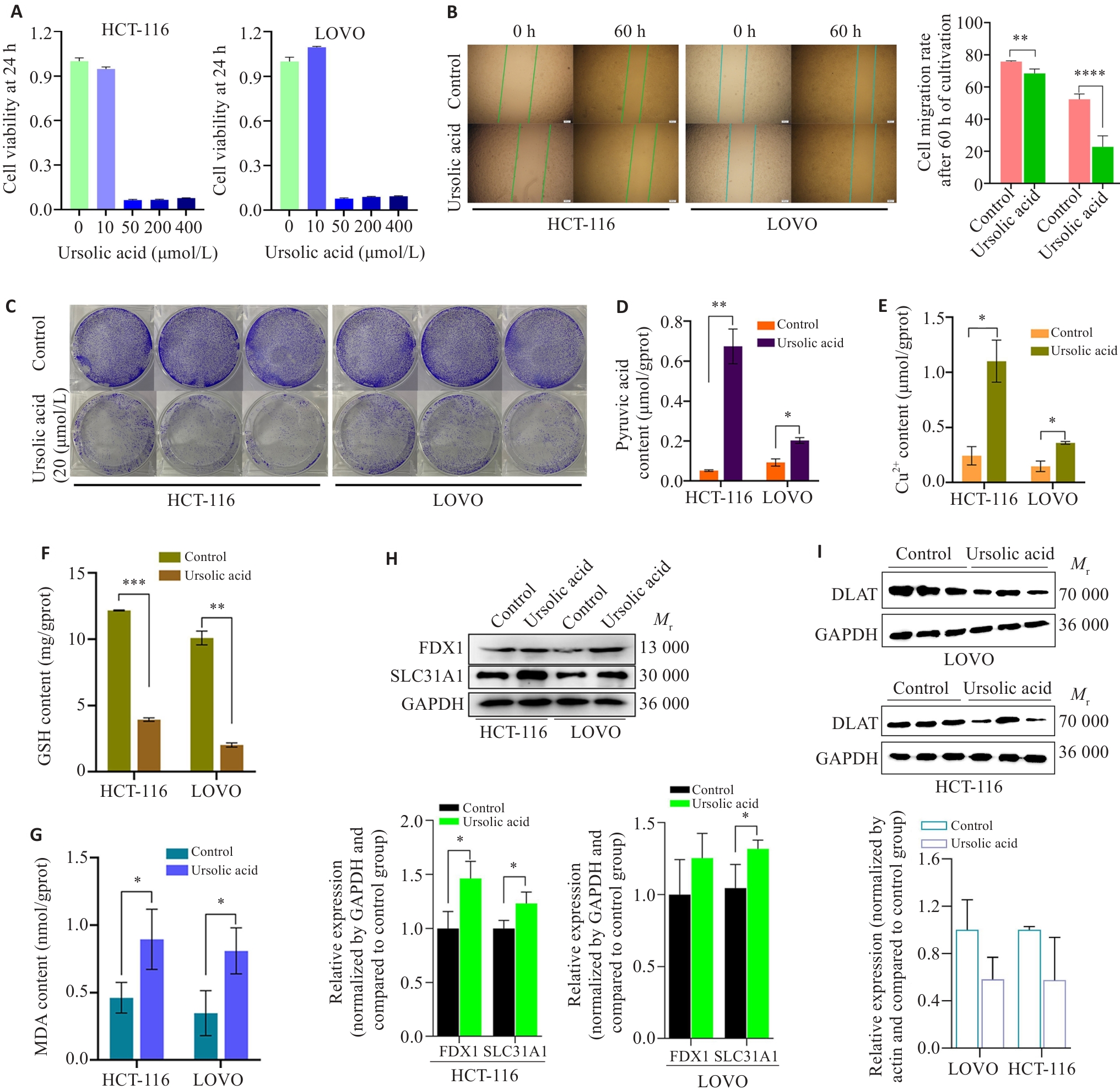
图9 熊果酸对结直肠癌细胞铜死亡的调控作用
Fig.9 Regulatory effect of ursolic acid on cuproptosis in CRC. A: Ursolic acid significantly inhibits the growth of HCT-116 and LoVo cells. B: Ursolic acid reduces migration of colon cancer cells. C: Ursolic acid inhibits clone formation of colon cancer cells. D-G: Changes in pyruvate (D), Cu2+ (E), GSH (F), and MDA (G) before and after ursolic acid intervention. H: Ursolic acid upregulates the expression of SLC31A1 and FDX1. I: Ursolic acid inhibits the expression of DLAT. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
| [1] | Shin AE, Giancotti FG, Rustgi AK. Metastatic colorectal cancer: mechanisms and emerging therapeutics[J]. Trends Pharmacol Sci, 2023, 44(4): 222-36. doi:10.1016/j.tips.2023.01.003 |
| [2] | 刘超越, 王 汉, 杜 宇, 等. 中药干预相关信号通路防治结直肠癌及癌前病变研究进展[J]. 中成药, 2026, 48(2): 534-42. |
| [3] | Biller LH, Schrag D. Diagnosis and treatment of metastatic colorectal cancer: a review[J]. JAMA, 2021, 325(7): 669-85. doi:10.1001/jama.2021.0106 |
| [4] | Patel SG, Karlitz JJ, Yen T, et al. The rising tide of early-onset colorectal cancer: a comprehensive review of epidemiology, clinical features, biology, risk factors, prevention, and early detection[J]. Lancet Gastroenterol Hepatol, 2022, 7(3): 262-74. doi:10.1016/s2468-1253(21)00426-x |
| [5] | 宁思明, 孙 宝, 晋亚楠, 等. 1990—2021年中国结直肠癌疾病负担研究及2022—2041年变化趋势分析[J]. 现代肿瘤医学, 2025, 33(9): 1579-87. |
| [6] | 桂 杨, 范 恒. 化结消瘤汤通过调节肠道ILC3细胞缓解结肠炎相关结直肠癌小鼠肠道屏障功能损伤[J]. 时珍国医国药, 2025, 36(18): 3416-22. |
| [7] | 唐佩莉. 医院制剂化结消瘤颗粒的药学及其抗结肠癌作用机制的初步研究[D]. 武汉: 湖北中医药大学, 2023. |
| [8] | Xie JM, Yang YN, Gao YB, et al. Cuproptosis: mechanisms and links with cancers[J]. Mol Cancer, 2023, 22(1): 46. doi:10.1186/s12943-023-01732-y |
| [9] | Chen LY, Min JX, Wang FD. Copper homeostasis and cuproptosis in health and disease[J]. Signal Transduct Target Ther, 2022, 7: 378. doi:10.1038/s41392-022-01229-y |
| [10] | Solier S, Müller S, Cañeque T, et al. A druggable copper-signalling pathway that drives inflammation[J]. Nature, 2023, 617(7960): 386-94. doi:10.1038/s41586-023-06017-4 |
| [11] | Li XC, Zeng X, Yang WQ, et al. Impacts of copper deficiency on oxidative stress and immune function in mouse spleen[J]. Nutrients, 2024, 17(1): 117. doi:10.3390/nu17010117 |
| [12] | Zhong GL, Li L, Li YX, et al. Cuproptosis is involved in copper-induced hepatotoxicity in chickens[J]. Sci Total Environ, 2023, 866: 161458. doi:10.1016/j.scitotenv.2023.161458 |
| [13] | Xu YZ, Liu SY, Zeng LL, et al. An enzyme-engineered nonporous copper(I) coordination polymer nanoplatform for cuproptosis-based synergistic cancer therapy[J]. Adv Mater, 2022, 34(43): e2204733. doi:10.1002/adma.202204733 |
| [14] | Yang LF, Zhang YW, Wang Y, et al. Ferredoxin 1 is a cuproptosis-key gene responsible for tumor immunity and drug sensitivity: a pan-cancer analysis[J]. Front Pharmacol, 2022, 13: 938134. doi:10.3389/fphar.2022.938134 |
| [15] | Xue Q, Kang R, Klionsky DJ, et al. Copper metabolism in cell death and autophagy[J]. Autophagy, 2023, 19(8): 2175-95. doi:10.1080/15548627.2023.2200554 |
| [16] | Tang PL, Sun WJ, Xie SF, et al. A Chinese medicine, Huajie Xiaoliu formula, enhances the anti-colon cancer effect of tegafur, gimeracil, and oteracil potassium capsules through bcl-2/bax signaling pathway[J]. Nat Prod Commun, 2023, 18(8): 1934578X231185201. doi:10.1177/1934578x231185201 |
| [17] | Gu XR, Chen Y, Hu XY, et al. Exploring autophagy-related gene expression in hepatocellular carcinoma via TCGA GEPIA2 and HPA databases: implications for prognosis[J]. J Hepatocell Carcinoma, 2025, 12: 1557-86. doi:10.2147/jhc.s520917 |
| [18] | Li CW, Tang ZF, Zhang WJ, et al. GEPIA2021: integrating multiple deconvolution-based analysis into GEPIA[J]. Nucleic Acids Res, 2021, 49(W1): W242-6. doi:10.1093/nar/gkab418 |
| [19] | Wang WY, Mo WT, Hang ZS, et al. Cuproptosis: harnessing transition metal for cancer therapy[J]. ACS Nano, 2023, 17(20): 19581-99. doi:10.1021/acsnano.3c07775 |
| [20] | Pan ZW, Huang L, Gan YY, et al. The molecular mechanisms of cuproptosis and small-molecule drug design in diabetes mellitus[J]. Molecules, 2024, 29(12): 2852. doi:10.3390/molecules29122852 |
| [21] | Tao XY, Wang HR, Wang Q, et al. Marine natural product chagosendine C induces cuproptosis in colorectal cancer cells by targeting FDX1[J]. J Am Chem Soc, 2025, 147(41): 37089-103. |
| [22] | 魏 婷, 丁洋洋, 张佳佳, 等. 铜死亡诱导剂FDX1、硫辛酸与冠状动脉病变严重程度的相关性[J]. 南方医科大学学报, 2024, 44(2): 308-16. doi:10.12122/j.issn.1673-4254.2024.02.13 |
| [23] | Liu WQ, Lin WR, Yan L, et al. Copper homeostasis and cuproptosis in cancer immunity and therapy[J]. Immunol Rev, 2024, 321(1): 211-27. doi:10.1111/imr.13276 |
| [24] | Li K, Wu LL, Wang H, et al. Apoptosis and cuproptosis co-activated Copper-based metal-organic frameworks for cancer therapy[J]. J Nanobiotechnology, 2024, 22(1): 546. doi:10.1186/s12951-024-02828-3 |
| [25] | Zhang P, Zhou CT, Ren XY, et al. Inhibiting the compensatory elevation of xCT collaborates with disulfiram/copper-induced GSH consumption for cascade ferroptosis and cuproptosis[J]. Redox Biol, 2024, 69: 103007. doi:10.1016/j.redox.2023.103007 |
| [26] | Yang Y, Liang SY, Geng HE, et al. Proteomics revealed the crosstalk between copper stress and cuproptosis, and explored the feasibility of curcumin as anticancer copper ionophore[J]. Free Radic Biol Med, 2022, 193: 638-47. doi:10.1016/j.freeradbiomed.2022.11.023 |
| [27] | Liu SW, Yan WN, Zhang WY, et al. Nanoenhanced-cuproptosis results from the synergy of calcium overload and GSH depletion with the increasing of intracellular Ca/Mn/Cu ions[J]. Adv Sci, 2025, 12(13): 2412067. doi:10.1002/advs.202412067 |
| [28] | He G, Zhang YY, Feng YJ, et al. SBFI26 induces triple-negative breast cancer cells ferroptosis via lipid peroxidation[J]. J Cell Mol Med, 2024, 28(7): e18212. doi:10.1111/jcmm.18212 |
| [29] | Kim MJ, Lee H, Chanda D, et al. The role of pyruvate metabolism in mitochondrial quality control and inflammation[J]. Mol Cells, 2023, 46(5): 259-67. doi:10.14348/molcells.2023.2128 |
| [30] | Yang XL, Zhang XQ, Shen KF, et al. Cuproptosis-related genes signature and validation of differential expression and the potential targeting drugs in temporal lobe epilepsy[J]. Front Pharmacol, 2023, 14: 1033859. doi:10.3389/fphar.2023.1033859 |
| [31] | Huang Z, Liu Y, Zeng Y, et al. Mitochondrial thermal proteome profiling reveals aberrant activation of pyruvate dehydrogenase upon cuproptosis[J]. Cell Rep, 2025, 44(7): 115937. doi:10.1016/j.celrep.2025.115937 |
| [1] | 刘超群, 宁紫燕, 吴江华, 刘魏魏, 林创, 许嘉玮, 周蕊, 赵亮. 结直肠癌细胞分泌牙本质涎磷蛋白通过整合素αvβ3依赖途径诱导奥沙利铂耐药[J]. 南方医科大学学报, 2026, 46(3): 479-488. |
| [2] | 翁诺舟, 谭彬, 曾文涛, 古家宇, 翁炼基, 郑克鸿. 过表达RGL1通过激活CDC42/RAC1复合体上调运动型黏着斑组装促进结直肠癌转移[J]. 南方医科大学学报, 2025, 45(5): 1031-1038. |
| [3] | 马振南, 刘福全, 赵雪峰, 张晓微. DTX2促进奥沙利铂耐药的结直肠癌细胞增殖、侵袭和上皮间质转化[J]. 南方医科大学学报, 2025, 45(4): 829-836. |
| [4] | 孙红燕, 卢国庆, 付程文, 徐梦文, 朱小翌, 邢国权, 刘乐强, 柯雨菲, 崔乐妹, 陈睿旸, 王磊, 康品方, 唐碧. 槲皮素通过调控L型钙通道改善糖尿病大鼠心肌损伤[J]. 南方医科大学学报, 2025, 45(3): 531-541. |
| [5] | 庆顺杰, 沈智勇. 过表达己糖激酶2通过激活JAK/STAT途径促进结直肠癌细胞的增殖、迁移和侵袭并调节肿瘤免疫微环境[J]. 南方医科大学学报, 2025, 45(3): 542-553. |
| [6] | 喻珍妮, 高竟哲, 孙惠, 冯芹, 那效旗, 张宁, 沈昆双, 王媛媛, 王喜军. 肠道菌群、T细胞在结直肠癌发病中的因果关联:孟德尔随机化分析[J]. 南方医科大学学报, 2025, 45(12): 2756-2766. |
| [7] | 纪凯, 于冠宇, 周乐其, 张天帅, 凌潜龙, 满文江, 朱冰, 张卫. HNRNPA1基因在结直肠癌组织中高表达及其潜在的诊断和治疗价值[J]. 南方医科大学学报, 2024, 44(9): 1685-1695. |
| [8] | 张银亮, 骆泽谭, 赵睿, 赵娜, 徐志东, 奥迪, 丛古一, 刘新宇, 郑海伦. 血根碱通过调控STUB1/GPX4诱导直肠癌细胞发生铁死亡[J]. 南方医科大学学报, 2024, 44(8): 1537-1544. |
| [9] | 郗雪艳, 邓婷, 杜伯雨. 结直肠成纤维细胞通过激活ERK信号通路促进结直肠癌细胞的恶性生物学行为[J]. 南方医科大学学报, 2024, 44(10): 1866-1873. |
| [10] | 颜 畅, 刘 爽, 宋庆志, 胡艺冰. 二甲双胍通过抑制线粒体氧化磷酸化降低结直肠癌干细胞的自我更新能力[J]. 南方医科大学学报, 2023, 43(8): 1279-1286. |
| [11] | 魏 可, 石纪雯, 肖雨寒, 王文锐, 杨清玲, 陈昌杰. miR-30e-5p过表达促进结直肠癌细胞的增殖和迁移:基于下调PTEN激活CXCL12轴[J]. 南方医科大学学报, 2023, 43(7): 1081-1092. |
| [12] | 张雪芳, 陈延华, 李宗恒, 尚 靖, 袁泽婷, 邓皖利, 骆 莺, 韩 娜, 殷佩浩, 殷 军. 六神丸治疗小鼠结肠炎相关性结直肠癌的作用机制:基于网络药理学和体内验证方法[J]. 南方医科大学学报, 2023, 43(7): 1051-1062. |
| [13] | 邓 婷, 杜伯雨, 郗雪艳. 结直肠癌细胞通过激活成纤维细胞的ERK通路诱导癌症相关成纤维细胞的形成[J]. 南方医科大学学报, 2023, 43(6): 943-951. |
| [14] | 马振南, 赵雪峰, 张晓微, 许广大, 刘福全. DTX2通过Notch2/Akt轴促进结直肠癌细胞的迁移和侵袭[J]. 南方医科大学学报, 2023, 43(3): 340-348. |
| [15] | 王旋成, 朱一帆, 周海琳, 黄宗声, 陈鸿炜, 张嘉豪, 杨珊伊, 陈广辉, 张淇淞. 血清非靶向代谢组学联合靶向胆汁酸代谢组学筛查结直肠癌的潜在生物标志物[J]. 南方医科大学学报, 2023, 43(3): 443-453. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||