南方医科大学学报 ›› 2024, Vol. 44 ›› Issue (10): 1898-1909.doi: 10.12122/j.issn.1673-4254.2024.10.08
收稿日期:2024-05-31
出版日期:2024-10-20
发布日期:2024-10-31
通讯作者:
汪向海
E-mail:liyonghua562023@163.com;wxhwxpcyy@163.com
作者简介:李泳华,在读硕士研究生,E-mail: liyonghua562023@163.com
基金资助:
Yonghua LI( ), Xinran XI, Meng ZHANG, Xun WU, Xianghai WANG(
), Xinran XI, Meng ZHANG, Xun WU, Xianghai WANG( )
)
Received:2024-05-31
Online:2024-10-20
Published:2024-10-31
Contact:
Xianghai WANG
E-mail:liyonghua562023@163.com;wxhwxpcyy@163.com
摘要:
目的 探讨LINC00467对肺腺癌增殖和转移的影响及参与细胞自噬的机制。 方法 体外培养人支气管上皮细胞16HBE和人肺腺癌细胞A549和H1299,A549和H1299细胞经慢病毒shlinc00467和shNC感染、自噬抑制剂3-甲基腺嘌呤(3-MA)和AMPK抑制剂BML-275处理。分别设置shNC组、shlinc00467组、shNC+3-MA组、shlinc00467+3-MA组、shNC+BML-275组和shlinc00467+BML-275组。实时荧光定量PCR检测细胞中LINC00467的表达,TCGA数据分析组织中LINC00467的表达以及对肺腺癌患者生存率和临床分期的影响,克隆形成实验检测细胞增殖情况,Transwell实验测定细胞迁移和侵袭能力,免疫荧光染色检测LC3的表达,Western blotting检测蛋白表达,GSEA富集分析LINC00467与自噬通路的相关性。 结果 与16HBE细胞相比,LINC00467在A549和H1299细胞中表达增加(P<0.001)。与癌旁组织相比,肺腺癌组织中LINC00467高表达(P<0.001)且随着临床分期增加表达量增加(P<0.05),LINC00467高表达导致患者不良的总体生存率(OS,P=0.049)和第一阶段进展率(FP,P=0.026)。与shNC组相比,shlinc00467感染的A549和H1299细胞中LINC00467表达降低(P<0.001)。与shNC组相比,shlinc00467导致A549和H1299细胞克隆形成数(P<0.01)、迁移细胞数(P<0.001)、侵袭细胞数(P<0.001)减少、p-mTOR/mTOR(P<0.05)及p62(P<0.01)蛋白表达减少;p-AMPK/AMPK(P<0.05)和LC3II/I(P<0.05)增加;GSEA提示了LINC00467对自噬通路的抑制作用(|NES|>1,P<0.05,FDR<0.25)。 结论 LINC00467能促进肺腺癌细胞增殖和转移,可能是通过抑制AMPK/mTOR信号通路介导的自噬实现的。
李泳华, 奚欣然, 张萌, 吴勋, 汪向海. LINC00467高表达通过抑制AMPK/mTOR通路抑制细胞自噬促进肺腺癌细胞的增殖和转移[J]. 南方医科大学学报, 2024, 44(10): 1898-1909.
Yonghua LI, Xinran XI, Meng ZHANG, Xun WU, Xianghai WANG. High expression of LINC00467 promotes proliferation and metastasis of lung adenocarcinoma cells by suppressing autophagy via inhibiting the AMPK/mTOR pathway[J]. Journal of Southern Medical University, 2024, 44(10): 1898-1909.
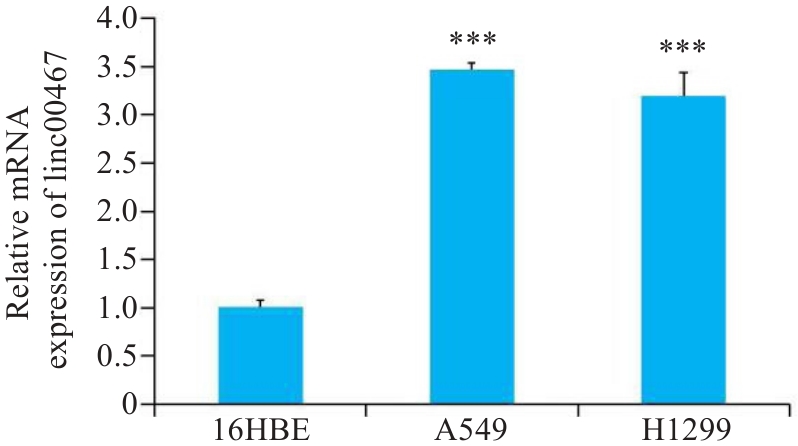
图1 linc00467在肺腺癌细胞中的表达
Fig.1 Expression of LINC00467 in human bronchial epithelial cells 16HBE and lung adenocarcinoma cell lines A549 and H1299. ***P<0.001 vs 16HBE cells.
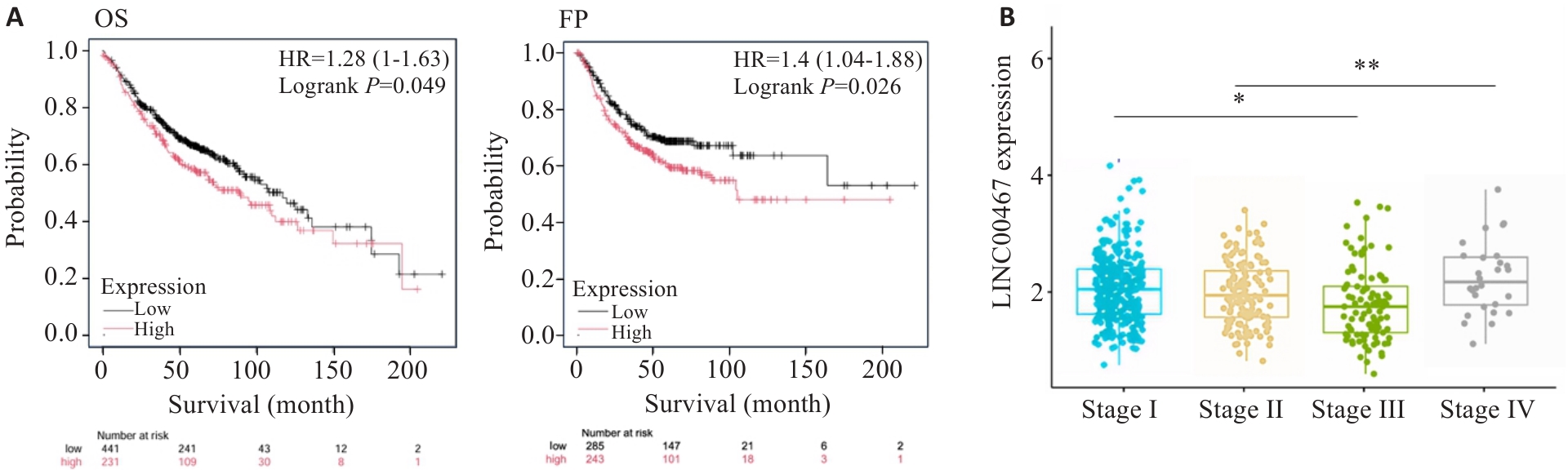
图3 LINC00467与肺腺癌患者生存率和临床分期的关系
Fig.3 Correlation of LINC00467 expression level with survival rates (A) and clinical stages (B) of lung adenocarcinoma patients. *P<0.05 vs Stage I group; **P<0.01 vs Stage II group.
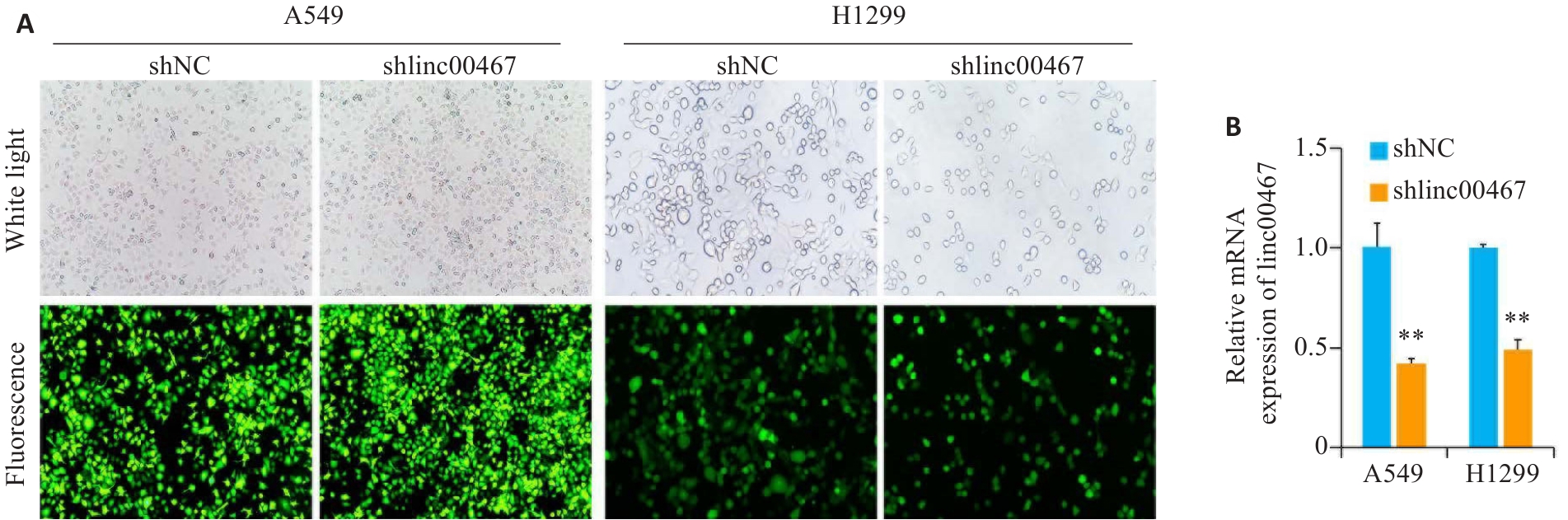
图4 A549和H1299细胞中linc00467的敲低效率
Fig.4 Interference efficiency of Shlinc00467 in A549 and H1299 cells. A: Transfection effect of Shlinc00467 (Original magnification: ×10); B: Quantification of the interference efficiency. **P<0.01 vs shNC group.
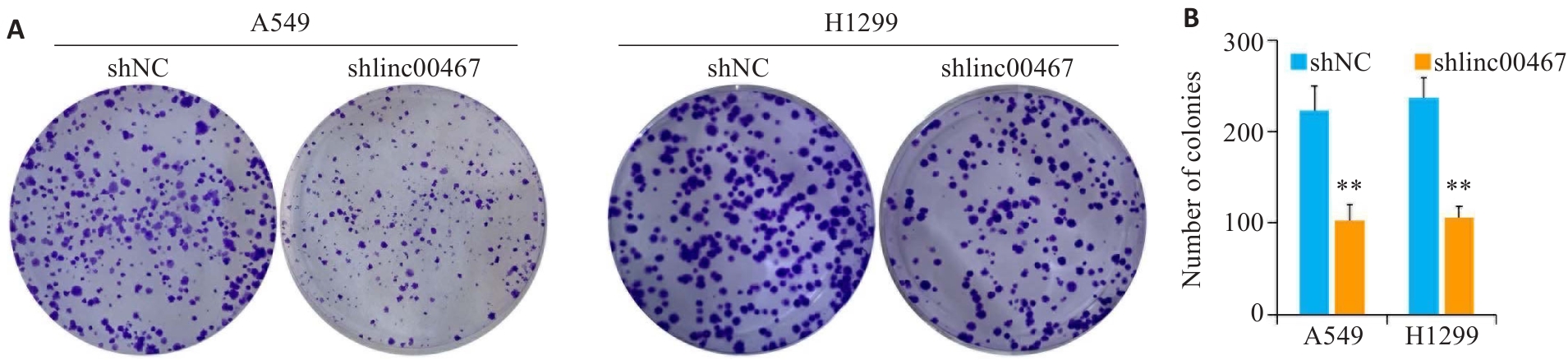
图5 敲低LINC00467对肺腺癌细胞增殖的影响
Fig.5 Effect of LINC00467 knockdown on proliferation of lung adenocarcinoma cells. A: Colony formation assay for assessing cell proliferation. B: Quantification of the colony number. **P<0.01 vs shNC group.

图6 敲低LINC00467对肺腺癌细胞转移的影响
Fig.6 Effect of LINC00467 knockdown on lung adenocarcinoma cell metastasis. A: Transwell assay for assessing cell migration and invasion (×10). B, C: Numbers of migrating and invading cells. ***P<0.001 vs shNC group.
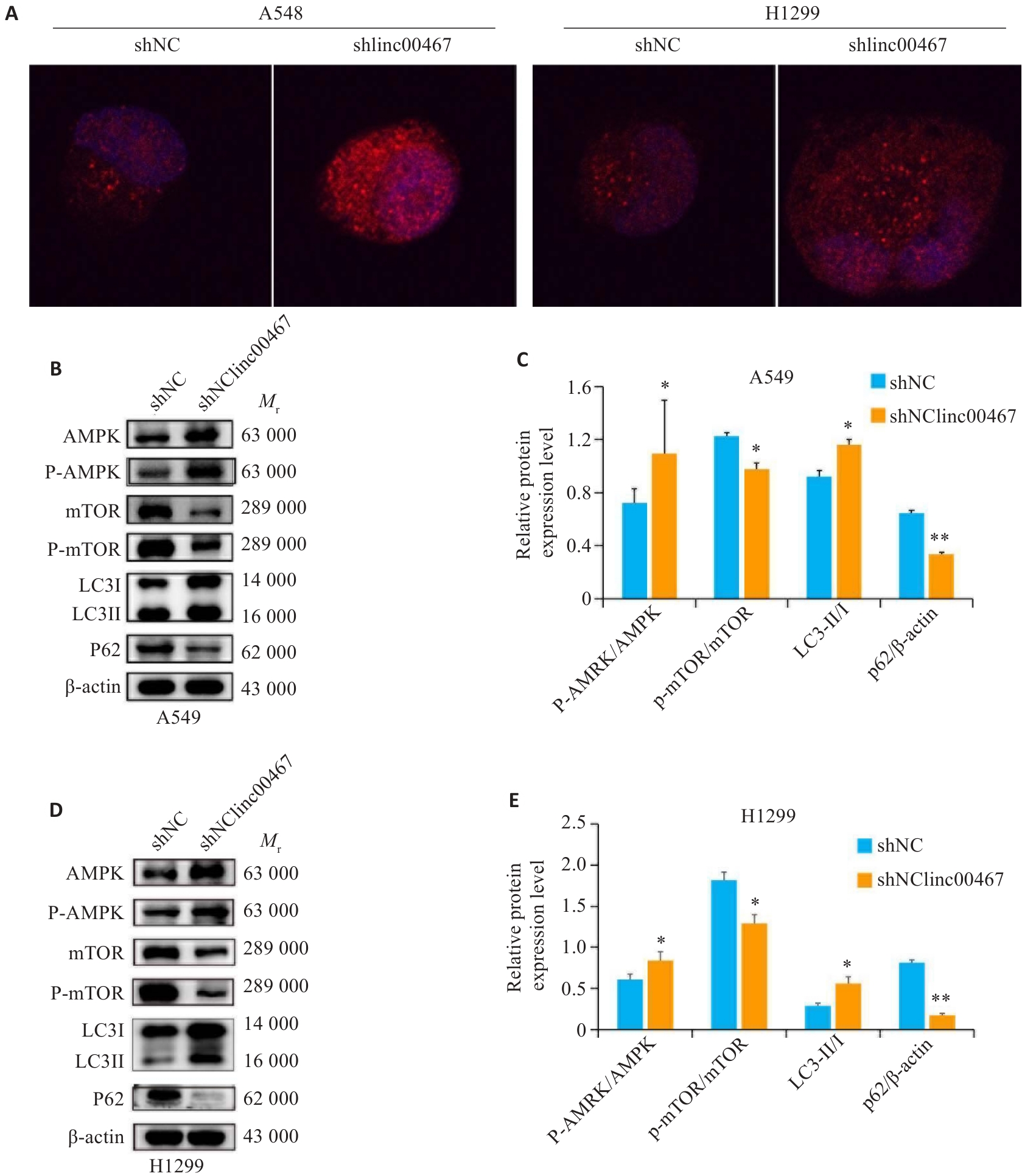
图7 敲低LINC00467对肺腺癌细胞自噬和AMPK/mTOR信号通路的影响
Fig.7 Effects of LINC00467 knockdown on autophagy and AMPK/mTOR signaling pathways in lung adenocarcinoma cells. A: Immunofluorescence staining for detecting LC3 expression (×40). B, D: Western blotting of AMPK, p-AMPK, mTOR, p-mTOR, and LC3II/I proteins. C, E: Expression levels of p-AMPK/AMPK, p-mTOR/mTOR, LC3II/I, and p62. *P<0.05, **P<0.01 vs shNC group.
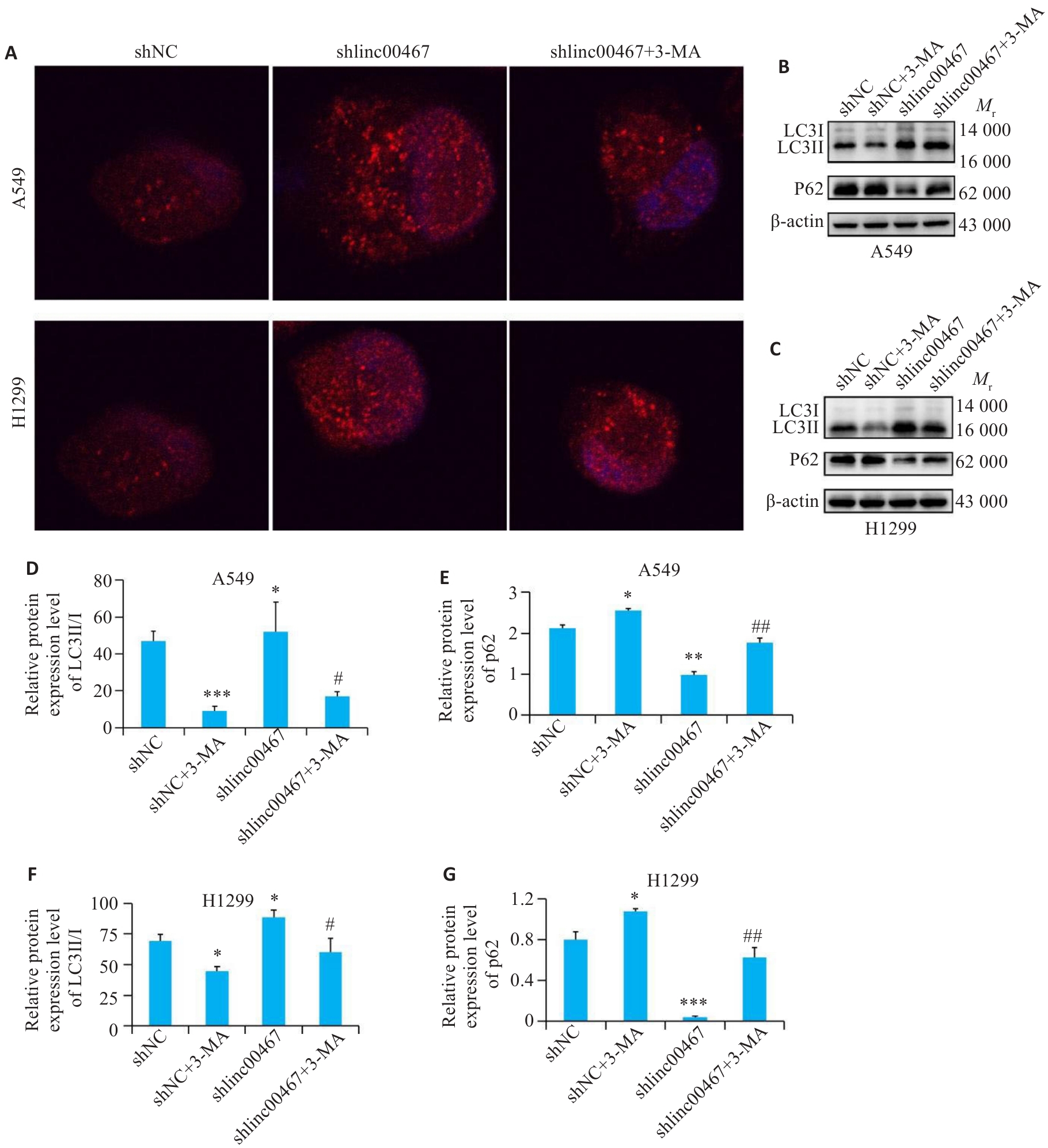
图9 自噬抑制剂对肺腺癌细胞自噬的影响
Fig.9 Effects of the autophagy inhibitor on autophagy of lung adenocarcinoma cells with LINC00467 knockdown. A: Immunofluorescence staining for LC3 expression (×40). B, C: Western blotting for detecting LC3II, LCI and p62 proteins. D-G: Quantification of LC3II/I and p62 protein levels.*P<0.05, **P<0.01, ***P<0.001 vs shNC group; #P<0.05, ##P<0.01 vs shlinc00467 group.
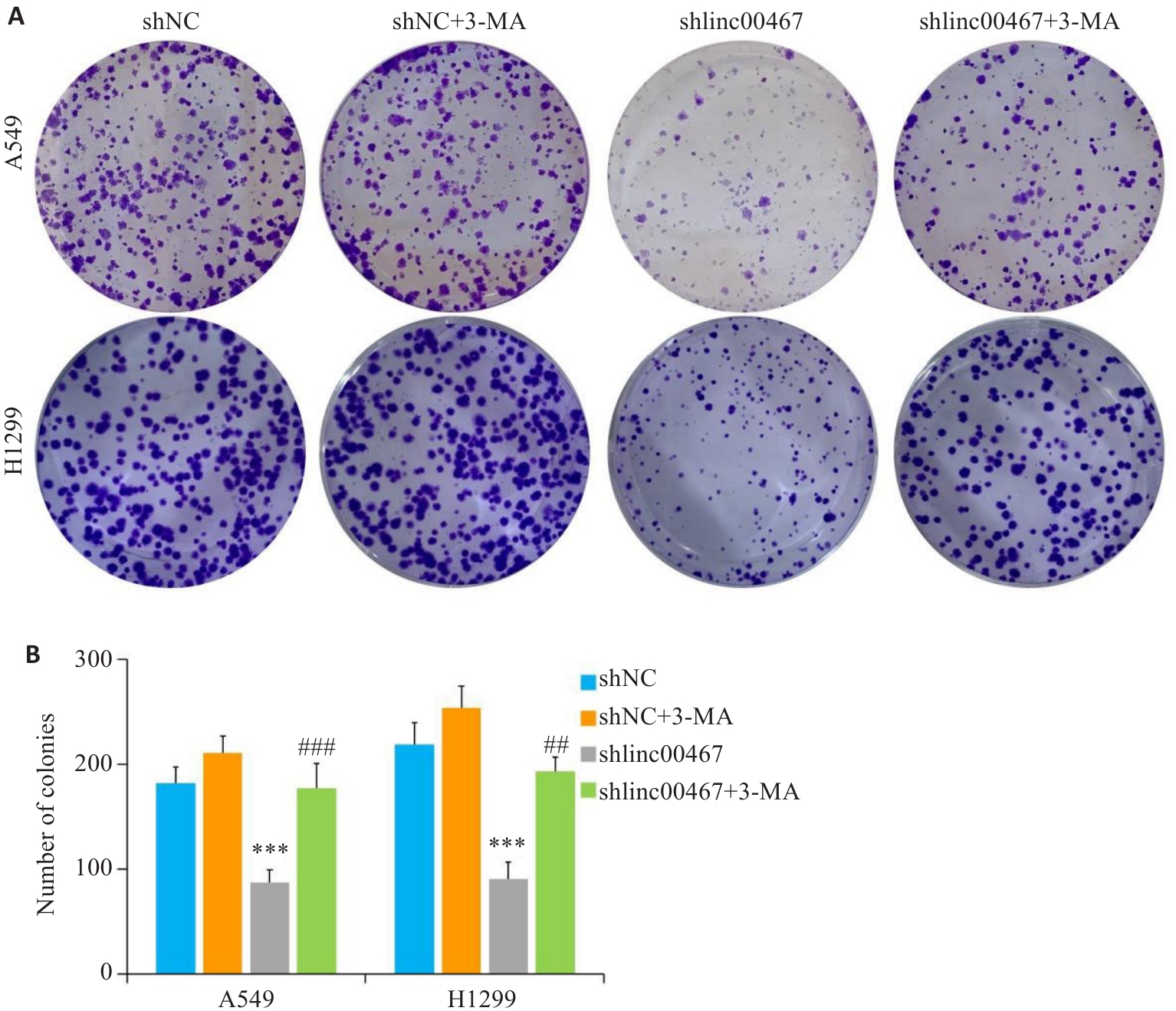
图10 LINC00467通过调控自噬影响肺腺癌细胞增殖
Fig.10 LINC00467 knockdown inhibits proliferation of lung adenocarcinoma cells by suppressing autophagy. A: Colony formation assay for evaluating cell proliferation. B: Quantification of the colony number. ***P<0.001 vs shNC group; ###P<0.01 vs shlinc00467 group.
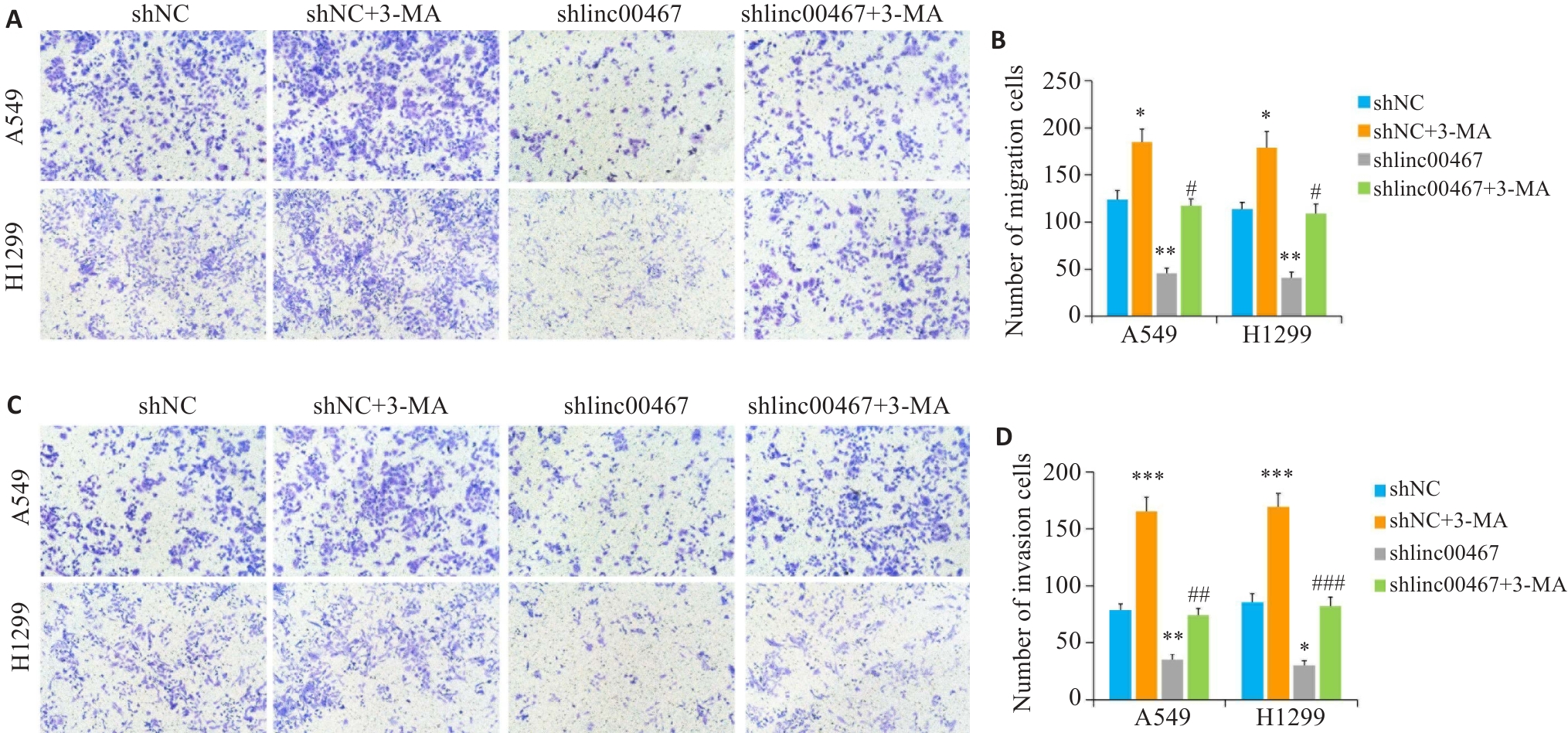
图11 LINC00467通过调控自噬影响肺腺癌细胞转移
Fig.11 LINC00467 knockdown suppresses lung adenocarcinoma cell metastasis by regulating autophagy. A, C: Transwell assay for assessing cell migration and invasion (×10). B, D: Numbers of migrating and invading cells. **P<0.01 vs shNC group; #P<0.05, ##P<0.01, ###P<0.001 vs shlinc00467 group.
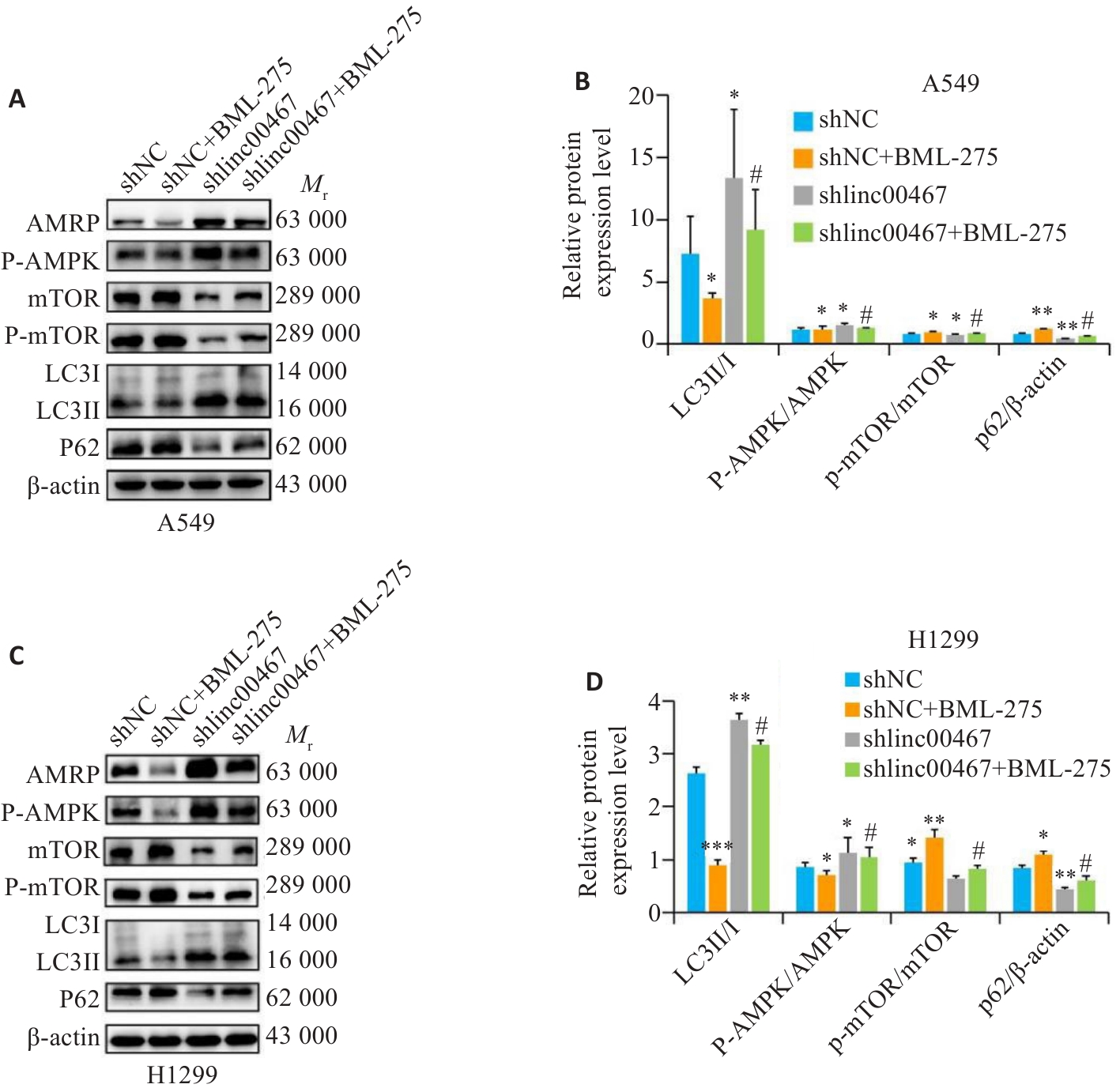
图12 LINC00467通过调控AMPK/mTOR信号通路影响肺腺癌细胞自噬
Fig.12 LINC00467 knockdown suppresses autophagy in lung adenocarcinoma cells by inhibiting the AMPK/mTOR signaling pathway. A,C: Western blotting for detecting AMPK, p-AMPK, mTOR, p-mTOR, LC3II, LC3I, and p62 proteins. B, D: Quantification of p-AMPK/AMPK, p-mTOR/mTOR, LC3II/I, and p62 protein levels. *P<0.05, **P<0.01 vs shNC group; #P<0.05 vs shlinc00467 group.
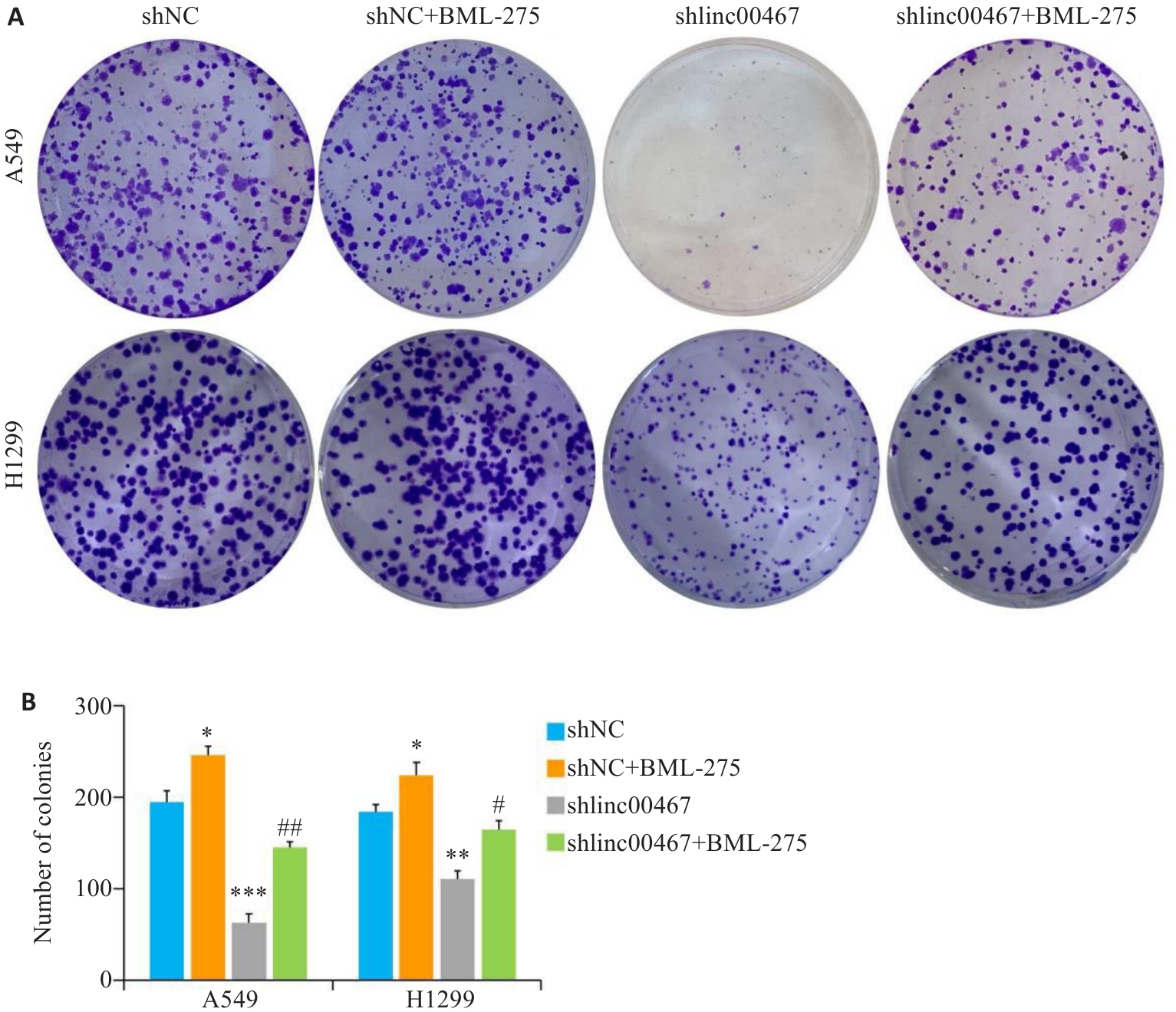
图13 LINC00467通过调控AMPK/mTOR信号通路影响肺腺癌细胞增殖
Fig.13 LINC00467 knockdown suppresses lung adenocarcinoma cell proliferation by inhibiting the AMPK/mTOR signaling pathway. A: Colony formation assay for assessing cell proliferation. B: Quantification of colony formation number. *P<0.05, **P<0.01, ***P<0.001 vs shNC group; #P<0.05, ##P<0.01 vs shlincC00467 group.

图14 LINC00467通过调控AMPK/mTOR信号通路影响肺腺癌细胞迁移
Fig.14 LINC00467 knockdown suppresses lung adenocarcinoma cell migration by inhibiting the AMPK/mTOR signaling pathway. A, C: Transwell assay for assessing cell migration and invasion (×10). B, D: Number of migrating and invading cells. *P<0.05, **P<0.01 vs shNC group; ##P<0.01 vs shlinc00467 group.
| 1 | Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2024, 74(3): 229-63. |
| 2 | Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024[J]. CA Cancer J Clin, 2024, 74(1): 12-49. |
| 3 | Sasaki T, Kuno H, Hiyama T, et al. 2021 WHO classification of lung cancer: molecular biology research and radiologic-pathologic correlation[J]. Radiographics, 2024, 44(3): e230136. |
| 4 | Li ST, Wang AX, Wu YY, et al. Targeted therapy for non-small-cell lung cancer: new insights into regulated cell death combined with immunotherapy[J]. Immunol Rev, 2024, 321(1): 300-34. |
| 5 | Taniue K, Akimitsu N. The functions and unique features of LncRNAs in cancer development and tumorigenesis[J]. Int J Mol Sci, 2021, 22(2): 632. |
| 6 | Ahmad M, Weiswald LB, Poulain L, et al. Involvement of lncRNAs in cancer cells migration, invasion and metastasis: cytoskeleton and ECM crosstalk[J]. J Exp Clin Cancer Res, 2023, 42(1): 173. |
| 7 | Wu D, Li RF, Liu JY, et al. Long noncoding RNA LINC00467: role in various human cancers[J]. Front Genet, 2022, 13: 892009. |
| 8 | Wang W, Bo H, Liang YM, et al. LINC00467 is upregulated by DNA copy number amplification and hypomethylation and shows ceRNA potential in lung adenocarcinoma[J]. Front Endocrinol, 2021, 12: 802463. |
| 9 | Ding H, Luo YC, Hu K, et al. Linc00467 promotes lung adenocarcinoma proliferation via sponging miR-20b-5p to activate CCND1 expression[J]. Onco Targets Ther, 2019, 12: 6733-43. |
| 10 | Wang XH, Liu HB, Shen KK, et al. Long intergenic non-coding RNA 00467 promotes lung adenocarcinoma proliferation, migration and invasion by binding with EZH2 and repressing HTRA3 expression[J]. Mol Med Rep, 2019, 20(1): 640-54. |
| 11 | Yang JL, Liu YD, Mai XS, et al. STAT1-induced upregulation of LINC00467 promotes the proliferation migration of lung adenocarcinoma cells by epigenetically silencing DKK1 to activate Wnt/β‑catenin signaling pathway[J]. Biochem Biophys Res Commun, 2019, 514(1): 118-26. |
| 12 | Yamamoto H, Matsui T. Molecular mechanisms of macroautophagy, microautophagy, and chaperone-mediated autophagy[J]. Nippon Ika Daigaku Zasshi, 2024, 91(1): 2-9. |
| 13 | Debnath J, Gammoh N, Ryan KM. Autophagy and autophagy-related pathways in cancer[J]. Nat Rev Mol Cell Biol, 2023, 24: 560-75. |
| 14 | Biswas U, Roy R, Ghosh S, et al. The interplay between autophagy and apoptosis: its implication in lung cancer and therapeutics[J]. Cancer Lett, 2024, 585: 216662. |
| 15 | Alers S, Löffler AS, Wesselborg S, et al. Role of AMPK-mTOR-Ulk1/2 in the regulation of autophagy: cross talk, shortcuts, and feedbacks[J]. Mol Cell Biol, 2012, 32(1): 2-11. |
| 16 | Li YJ, Chen YY. AMPK and autophagy[J]. Adv Exp Med Biol, 2019, 1206: 85-108. |
| 17 | Noda T. Regulation of autophagy through TORC1 and mTORC1[J]. Biomolecules, 2017, 7(3): 52. |
| 18 | Mesko S, Gomez D. Proton therapy in non-small cell lung cancer[J]. Curr Treat Options Oncol, 2018, 19(12): 76. |
| 19 | Qi C, Ma JM, Sun JJ, et al. The role of molecular subtypes and immune infiltration characteristics based on disulfidptosis-associated genes in lung adenocarcinoma[J]. Aging, 2023, 15(11): 5075-95. |
| 20 | Yao ZT, Yang YM, Sun MM, et al. New insights into the interplay between long non-coding RNAs and RNA-binding proteins in cancer[J]. Cancer Commun, 2022, 42(2): 117-40. |
| 21 | Chen XY, Luo Q, Xiao YN, et al. LINC00467: an oncogenic long noncoding RNA[J]. Cancer Cell Int, 2022, 22(1): 303. |
| 22 | Ghafouri-Fard S, Khoshbakht T, Hussen BM, et al. A review on the role of LINC00467 in the carcinogenesis[J]. Cancer Cell Int, 2022, 22(1): 319. |
| 23 | Changizian M, Nourisanami F, Hajpoor V, et al. LINC00467: a key oncogenic long non-coding RNA[J]. Clin Chim Acta, 2022, 536: 112-25. |
| 24 | Zhu YX, Li JJ, Bo H, et al. LINC00467 is up-regulated by TDG-mediated acetylation in non-small cell lung cancer and promotes tumor progression[J]. Oncogene, 2020, 39(38): 6071-84. |
| 25 | Chang YX, Yang LS. LINC00467 promotes cell proliferation and stemness in lung adenocarcinoma by sponging miR-4779 and miR-7978[J]. J Cell Biochem, 2020, 121(8/9): 3691-9. |
| 26 | Liu SZ, Yao SJ, Yang H, et al. Autophagy: regulator of cell death[J]. Cell Death Dis, 2023, 14(10): 648. |
| 27 | Sedlackova L, Kataura T, Sarkar S, et al. Metabolic function of autophagy is essential for cell survival[J]. Autophagy, 2023, 19(8): 2395-7. |
| 28 | Bai ZS, Peng YL, Ye XY, et al. Autophagy and cancer treatment: four functional forms of autophagy and their therapeutic applications[J]. J Zhejiang Univ Sci B, 2022, 23(2): 89-101. |
| 29 | Sun T. Long noncoding RNAs act as regulators of autophagy in cancer[J]. Pharmacol Res, 2018, 129: 151-5. |
| 30 | Wu JC, Huang XB, Li XF, et al. Suppression of the long non-coding RNA LINC01279 triggers autophagy and apoptosis in lung cancer by regulating FAK and SIN3A[J]. Discov Oncol, 2024, 15(1): 3. |
| 31 | Zhao HJ, Wang YH, Wu X, et al. FAM83A antisense RNA 1 (FAM83A-AS1) silencing impairs cell proliferation and induces autophagy via MET-AMPKɑ signaling in lung adenocarcinoma[J]. Bioengineered, 2022, 13(5): 13312-27. |
| 32 | Xiao L, Li Y, Zeng XF, et al. Silencing of LOC389641 impairs cell proliferation and induces autophagy via EGFR/MET signaling in lung adenocarcinoma[J]. Aging, 2020, 13(2): 2539-52. |
| 33 | Song Y, Du JK, Lu PL, et al. LncRNA NFYC-AS1 promotes the development of lung adenocarcinomas through autophagy, apoptosis, and MET/c-Myc oncogenic proteins[J]. Ann Transl Med, 2021, 9(21): 1621. |
| 34 | González A, Hall MN, Lin SC, et al. AMPK and TOR: the Yin and Yang of cellular nutrient sensing and growth control[J]. Cell Metab, 2020, 31(3): 472-92. |
| 35 | Fukuda T, Shiozaki K. Multiplexed suppression of TOR complex 1 induces autophagy during starvation[J]. Autophagy, 2021, 17(7): 1794-5. |
| 36 | Yuan WB, Fang WY, Zhang R, et al. Therapeutic strategies targeting AMPK-dependent autophagy in cancer cells[J]. Biochim Biophys Acta Mol Cell Res, 2023, 1870(7): 119537. |
| 37 | Liu XW, Xiao ZD, Han L, et al. LncRNA NBR2 engages a metabolic checkpoint by regulating AMPK under energy stress[J]. Nat Cell Biol, 2016, 18(4): 431-42. |
| 38 | Saha S, Zhang Y, Wilson B, et al. The tumor-suppressive long noncoding RNA DRAIC inhibits protein translation and induces autophagy by activating AMPK[J]. J Cell Sci, 2021, 134(24): jcs259306. |
| [1] | 李玮怡, 江露, 张宗星, 陈丹, 包卓玛, 黄丽, 袁林. 强骨康疏方通过抑制HIF-1α/BNIP3自噬信号通路减少类风湿性关节炎大鼠的破骨细胞分化[J]. 南方医科大学学报, 2025, 45(7): 1389-1396. |
| [2] | 谢婷, 王云云, 郭婷, 袁春华. 雷氏大疣蛛多肽毒素组分通过激活促凋亡通路和协同作用抑制癌细胞增殖[J]. 南方医科大学学报, 2025, 45(7): 1460-1470. |
| [3] | 龚秀莹, 侯顺福, 赵苗苗, 王晓娜, 张致涵, 刘清华, 尹崇高, 李洪利. LncRNA SNHG15通过miR-30b-3p调控COX6B1轴促进肺腺癌细胞增殖、迁移和侵袭的分子机制[J]. 南方医科大学学报, 2025, 45(7): 1498-1505. |
| [4] | 王心恒, 邵小涵, 李童童, 张璐, 杨勤军, 叶卫东, 童佳兵, 李泽庚, 方向明. 平喘宁方通过调控HMGB1/Beclin-1轴介导的自噬改善患寒哮证大鼠的气道炎症[J]. 南方医科大学学报, 2025, 45(6): 1153-1162. |
| [5] | 翁诺舟, 谭彬, 曾文涛, 古家宇, 翁炼基, 郑克鸿. 过表达RGL1通过激活CDC42/RAC1复合体上调运动型黏着斑组装促进结直肠癌转移[J]. 南方医科大学学报, 2025, 45(5): 1031-1038. |
| [6] | 曾玉梅, 李继科, 黄仲曦, 周毅波. 绒毛样蛋白VILL通过与LMO7蛋白相互作用抑制鼻咽癌细胞的增殖[J]. 南方医科大学学报, 2025, 45(5): 954-961. |
| [7] | 岳雅清, 牟召霞, 王希波, 刘艳. Aurora-A过表达通过激活NF-κBp65/ARPC4信号轴促进宫颈癌细胞的侵袭和转移[J]. 南方医科大学学报, 2025, 45(4): 837-843. |
| [8] | 张毅, 沈昱, 万志强, 陶嵩, 柳亚魁, 王栓虎. CDKN3高表达促进胃癌细胞的迁移和侵袭:基于调控p53/NF-κB信号通路和抑制胃癌细胞凋亡[J]. 南方医科大学学报, 2025, 45(4): 853-861. |
| [9] | 董妍妍, 张可敬, 储俊, 储全根. 抵当汤含药血清通过PI3K/Akt/mTOR信号通路增强高糖诱导的大鼠肾小球内皮细胞自噬[J]. 南方医科大学学报, 2025, 45(3): 461-469. |
| [10] | 庆顺杰, 沈智勇. 过表达己糖激酶2通过激活JAK/STAT途径促进结直肠癌细胞的增殖、迁移和侵袭并调节肿瘤免疫微环境[J]. 南方医科大学学报, 2025, 45(3): 542-553. |
| [11] | 廖茗, 钟文华, 张冉, 梁娟, 徐文陶睿, 万文珺, 吴超, 李曙. 源自蛇毒的蛋白C激活剂通过调控HIF-1α抑制BNIP3活性氧生成保护人脐静脉内皮细胞免受缺氧-复氧损伤[J]. 南方医科大学学报, 2025, 45(3): 614-621. |
| [12] | 黄晴晴, 张文静, 张小凤, 王炼, 宋雪, 耿志军, 左芦根, 王月月, 李静, 胡建国. 高表达MYO1B促进胃癌细胞增殖、迁移和侵袭并与患者的不良预后有关[J]. 南方医科大学学报, 2025, 45(3): 622-631. |
| [13] | 邹金华, 王惠, 张冬艳. SLC1A5通过促进M2型巨噬细胞极化促进肝癌进展[J]. 南方医科大学学报, 2025, 45(2): 269-284. |
| [14] | 宾禹, 李子雯, 左素微, 孙思诺, 李敏, 宋佳茵, 林旭, 薛刚, 吴靖芳. 载脂蛋白C1高表达通过激活JAK2/STAT3信号通路促进甲状腺乳头状癌细胞的增殖并抑制凋亡[J]. 南方医科大学学报, 2025, 45(2): 359-370. |
| [15] | 曹周芳, 汪元, 王梦娜, 孙玥, 刘菲菲. LINC00837/miR-671-5p/SERPINE2功能轴促进类风湿关节炎成纤维细胞样滑膜细胞的恶性病理学过程[J]. 南方医科大学学报, 2025, 45(2): 371-378. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||