南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (7): 1460-1470.doi: 10.12122/j.issn.1673-4254.2025.07.12
收稿日期:2025-04-01
出版日期:2025-07-20
发布日期:2025-07-17
通讯作者:
袁春华
E-mail:424464387@qq.com;yuanch@smu.edu.cn
作者简介:谢 婷,硕士,教师,E-mail: 424464387@qq.com
基金资助:
Ting XIE( ), Yunyun WANG, Ting GUO, Chunhua YUAN(
), Yunyun WANG, Ting GUO, Chunhua YUAN( )
)
Received:2025-04-01
Online:2025-07-20
Published:2025-07-17
Contact:
Chunhua YUAN
E-mail:424464387@qq.com;yuanch@smu.edu.cn
Supported by:摘要:
目的 研究雷氏大疣蛛毒素对癌细胞增殖抑制的选择性和有效组分。 方法 采用CCK-8法检测共培养48 h后雷氏大疣蛛毒素对癌细胞增殖的影响;应用流式细胞术检测癌细胞凋亡率;采用caspase试剂盒检测癌细胞内caspase-8和9的表达水平;通过凝胶过滤层析和高效液相色谱(HPLC)将雷氏大疣蛛粗毒分为蛋白质组分、多肽组分、小分子化合物组分3个部分;通过蛋白质组学对蛋白质和多肽组分进行鉴定;采用核磁共振、质谱结合HPLC解析小分子化合物组分中各物质的结构。 结果 雷氏大疣蛛粗毒对乳腺癌MCF7和鼻咽癌(SUNE1、HONE1)细胞表现出很强的浓度依赖性增殖抑制作用,半抑制浓度(IC50)分别为2.14 ±0.29、1.57±0.14、2.85±0.15 µg/mL;对多种胃癌和肠癌细胞系表现出较强的增殖抑制作用,对胃癌HGC27细胞的IC50为3.02±0.27 µg/mL,对肠癌SW620细胞的IC50为3.02±0.28 µg/mL。选用MCF7细胞来研究增殖抑制机制和筛选有效组分,发现粗毒可能通过激活caspase-8介导的信号通路诱导MCF7细胞凋亡。粗毒中蛋白质组分对MCF7细胞的增殖抑制作用很弱,10 µg/mL浓度仅抑制12.1%±1.9%。多肽组分抑制作用较强,IC50为6.41±0.31 µg/mL。多肽组分相对分子质量集中在10 000左右,主要为与巨型上户蛛毒素同源的多肽;小分子组分未表现出增殖抑制作用,主要为核苷酸代谢物。多肽组分和小分子组分质量比4∶1混合抑制作用明显增强。 结论 雷氏大疣蛛毒素对不同癌细胞的抑制活性有很大差异,发挥对癌细胞增殖抑制作用的组分为多肽组分,可能与核苷酸代谢物存在协同作用。
谢婷, 王云云, 郭婷, 袁春华. 雷氏大疣蛛多肽毒素组分通过激活促凋亡通路和协同作用抑制癌细胞增殖[J]. 南方医科大学学报, 2025, 45(7): 1460-1470.
Ting XIE, Yunyun WANG, Ting GUO, Chunhua YUAN. The peptide toxin components and nucleotide metabolites in Macrothele raveni venom synergistically inhibit cancer cell proliferation by activating the pro-apoptotic pathways[J]. Journal of Southern Medical University, 2025, 45(7): 1460-1470.
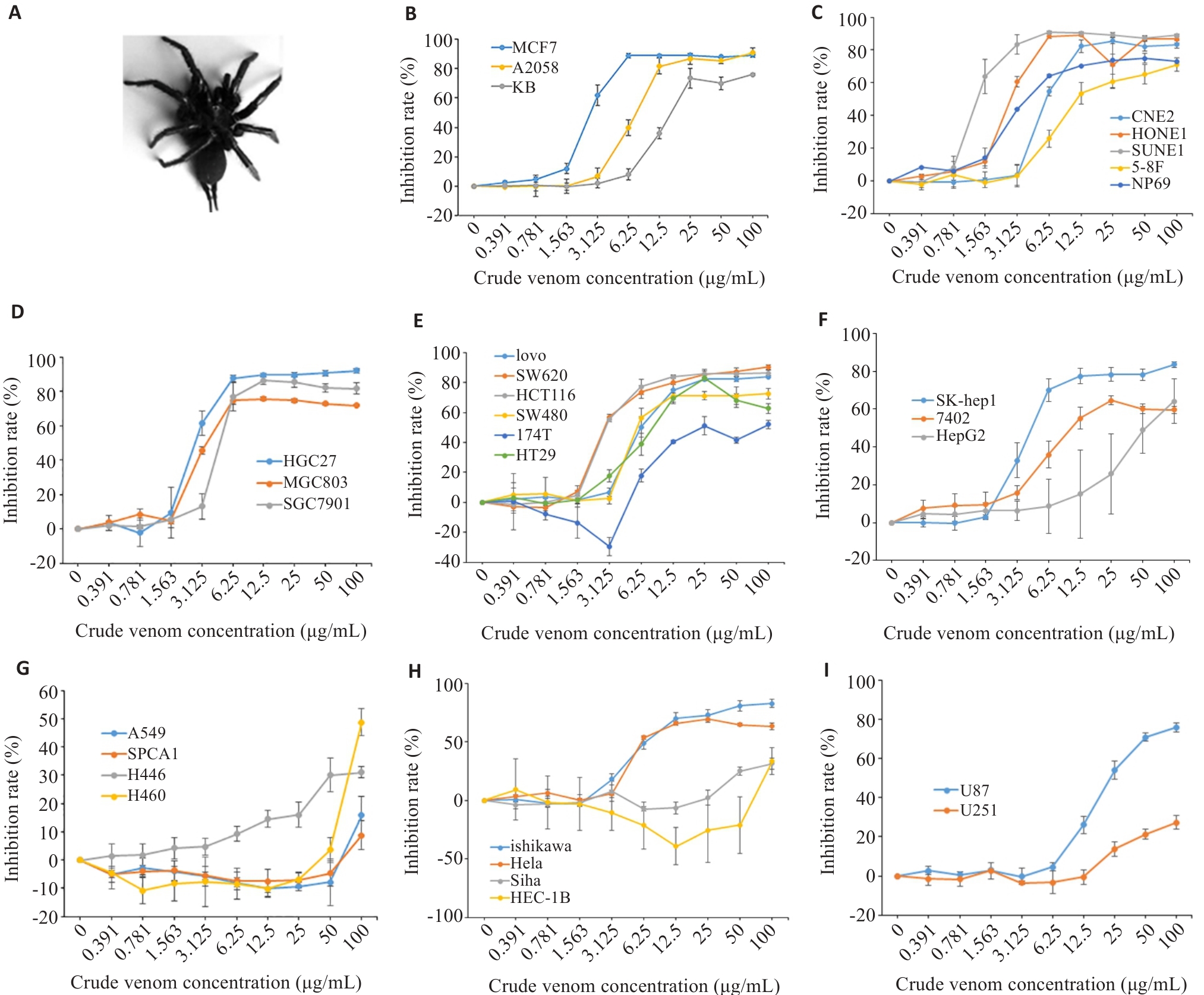
图1 雷氏大疣蛛毒素对癌细胞的增殖抑制作用
Fig.1 Inhibitory effects of Macrothele raven venom in different cancer cells assessed using CCK-8 assay at 48 h. A: The spider Macrothele raven. B: Inhibitory effects of the venom on proliferation of human breast cancer MCF7 cells, A2058 human Melanoma cells, and human oral epidermoid carcinoma KB cells. C-G: Inhibitory effects of the venom on proliferation of nasopharyngeal carcinoma cells (C), gastric cancer cells (D), colon cancer cells (E), hepatoma cells (F), and lung cancer cells (G). H: Inhibitory effects of the venom on human endometrial cancer Ishikawa cells, human cervical cancer HeLa cells, Siha cells, and human endometrial adenocarcinoma HEC-1B cells. I: Inhibitory effects of the venom on human glioma cells.
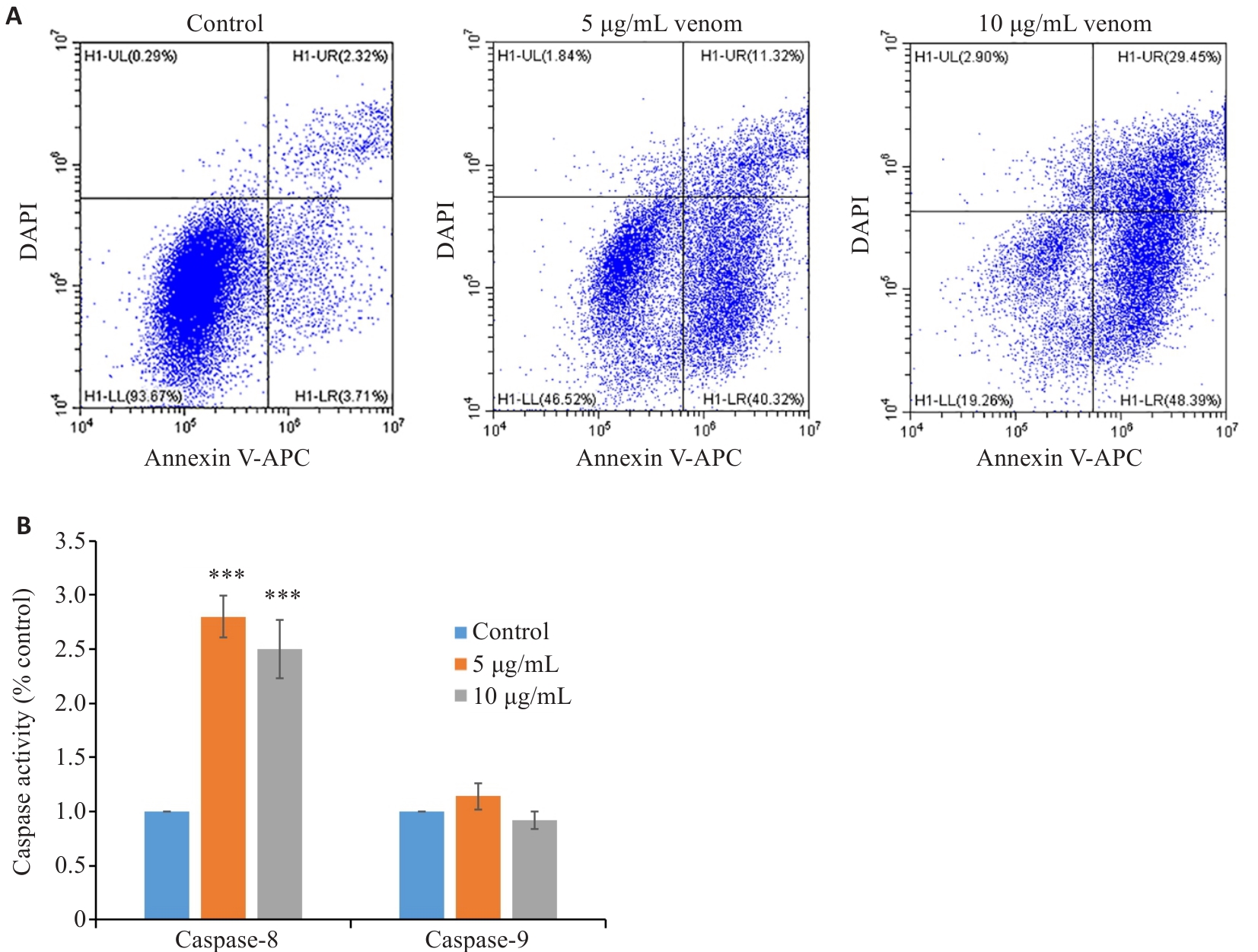
图3 雷氏大疣蛛粗毒诱导MCF7 细胞凋亡
Fig.3 Apoptosis of MCF7 cells following treatment with Macrothele raven venom. A: Flow cytometry of MCF7 cells treated with 5 and 10 µg/mL venom for 24 h. B: Analysis of caspase-8 and 9 activities in MCF7 cells treated with the venom at 5 and 10 µg/mL for 24 h (n=3). ***P<0.001 vs Control.
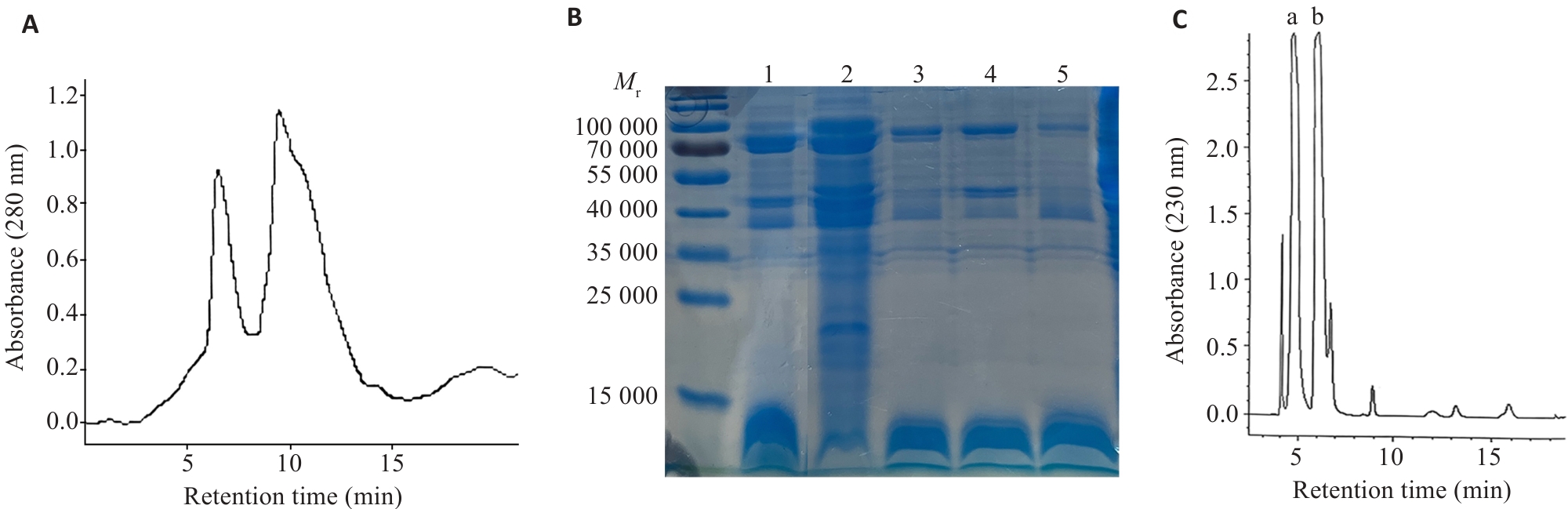
图4 雷氏大疣蛛毒素的分离纯化和SDS-PAGE分子量测定
Fig.4 Isolation, purification, and SDS-PAGE molecular weight determination of Macrothele raven venom. A: Separation of the crude venom by gel filtration (Sephadex G-75 column, 10 mm×100 mm). B: Analysis of the eluted fractions by SDS-PAGE with Coomassie Brilliant Blue staining. 1: Crude venom; 2: Fraction eluted at 5 min; 3, 4, 5: Fractions eluted at 10 min of different batches. C: Separation of the small molecule compounds from the crude venom by reversed-phase HPLC.
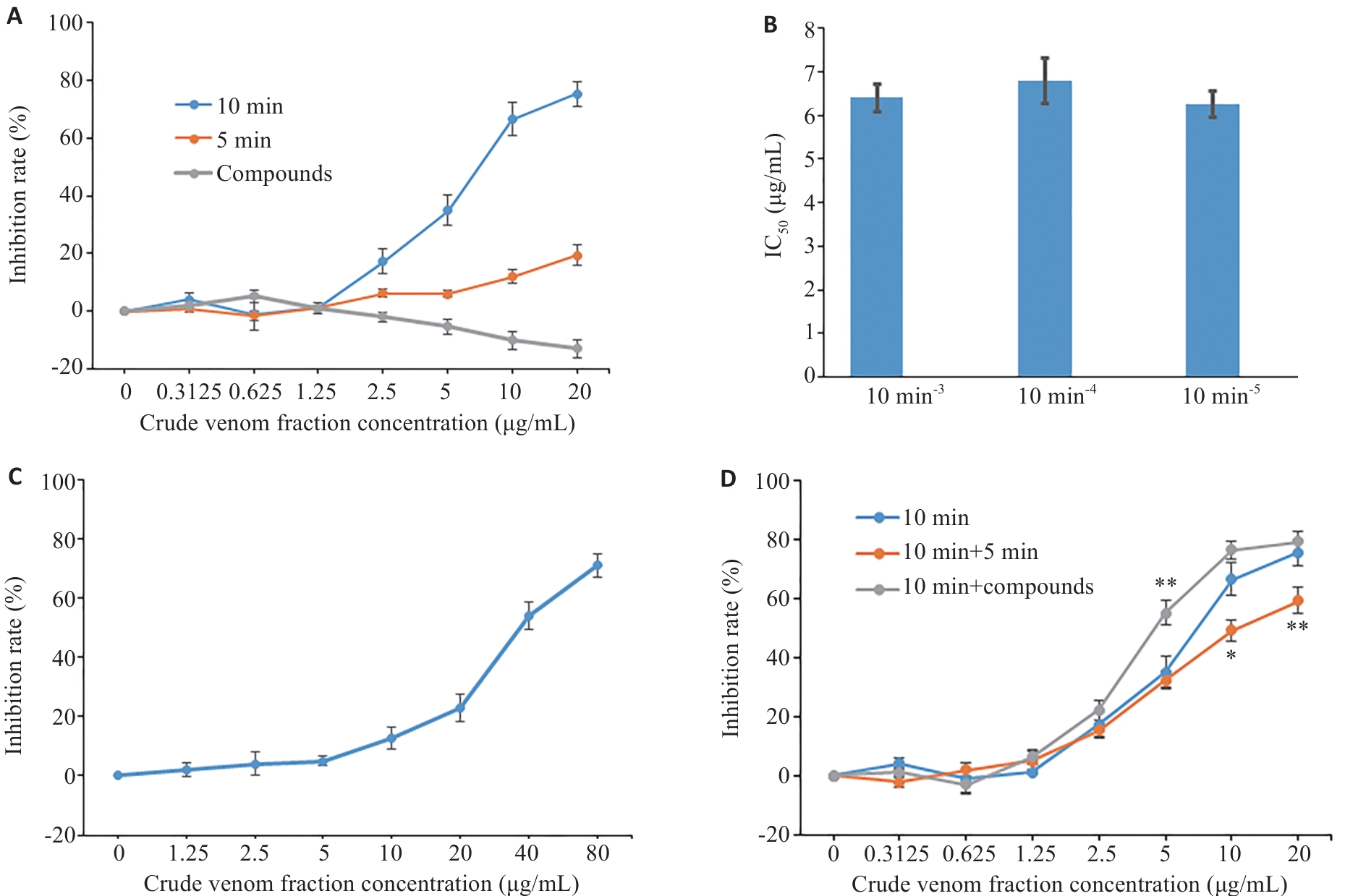
图5 雷氏大疣蛛毒素组分对乳腺癌MCF7细胞的增殖抑制作用
Fig.5 Inhibitory effects of the fractions from Macrothele raveni crude venom on proliferation of MCF-7 cells. A: Inhibitory effects of the 5-min and 10-min fractions separated by gel filtration and small compounds separated by HPLC on human breast cancer MCF-7 cells. B: Inhibitory effects of the fractions eluted at 10 min of different batches on MCF7 cells. C: Inhibitory effects of the 10 min-3 fraction on HEK-293 cells. D: Inhibitory effects of the combinations on human breast cancer MCF-7 cells. Data are presented as Mean±SD (n=3). *P<0.05, **P<0.01 vs 10-min peptide fraction.
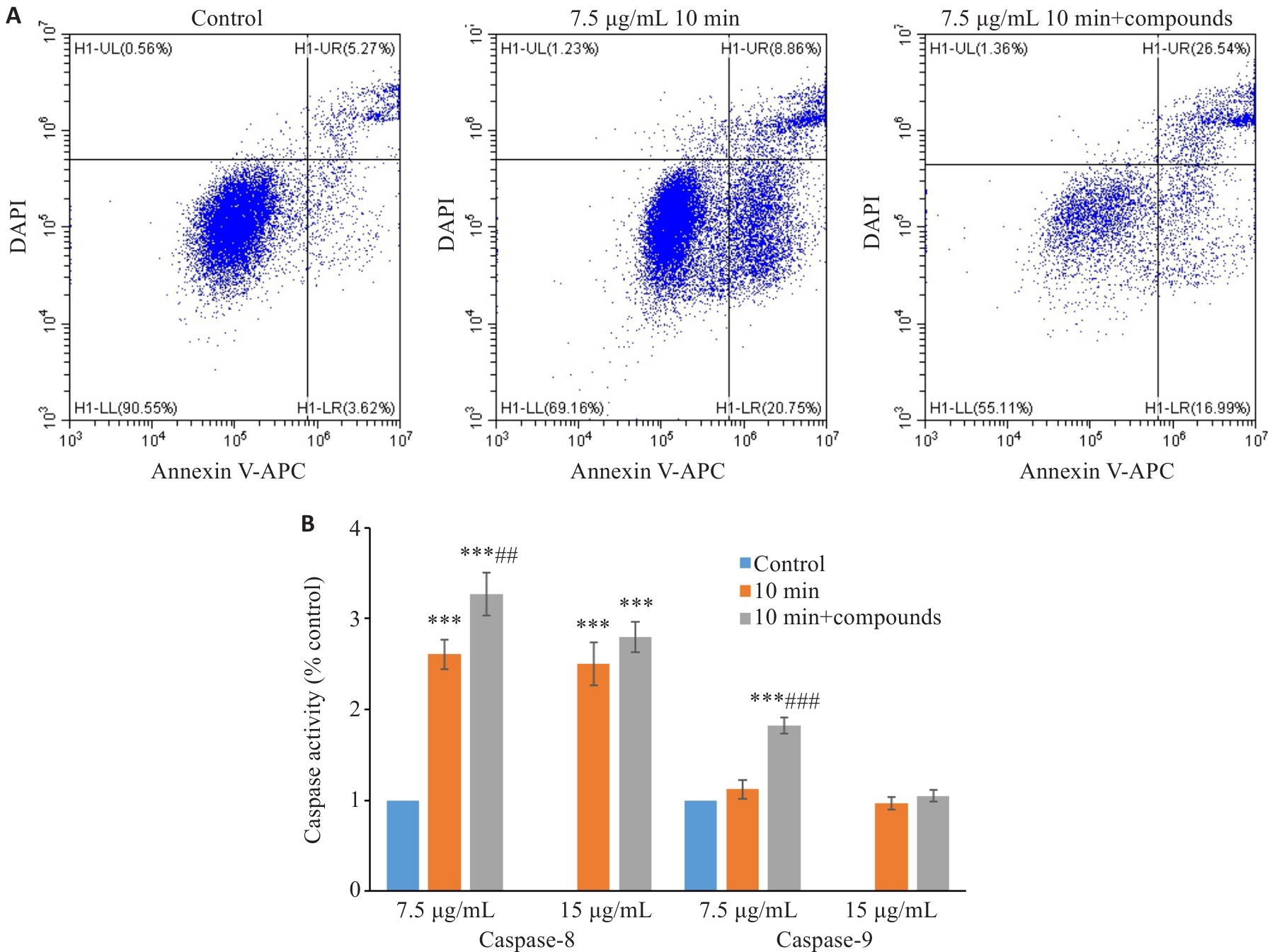
图6 小分子化合物促进多肽诱导的MCF7 细胞凋亡
Fig.6 The small molecule compounds from Macrothele raveni venom promoted peptide-induced apoptosis of MCF7 cells. A: Apoptosis of MCF7 cells treated with 7.5 µg/mL venom fractions for 24 h analyzed by flow cytometry (10 min: 10 min peptide fraction). B: Analysis of the activities of caspase 8 and 9 in MCF7 cells treated with the venom fractions at 7.5 and 15 µg/mL for 24 h (n=3). ***P<0.001 vs Control, ##P<0.01, ###P<0.001 vs 10-min peptide fraction.
| Accession No. | Protein | #PSMs | MW | Score sequest HT |
|---|---|---|---|---|
| Q9NFL4 | Hemocyanin G chain | 8 | 71.766 | 17.8 |
| P02241 | Hemocyanin D chain | 9 | 72.077 | 17.6 |
| P02242 | Hemocyanin E chain | 8 | 71.63 | 15.3 |
| P41340 | Actin-3 | 6 | 41.781 | 11.7 |
| Q9NFH9 | Hemocyanin B chain | 7 | 72.052 | 10.7 |
| P14750 | Hemocyanin A chain | 4 | 72.273 | 6.0 |
| P0C2V1 | U15-hexatoxin-Mg1a | 2 | 6.946 | 4.7 |
| P80476 | Hemocyanin AA6 chain | 3 | 71.74 | 3.8 |
| W4VS99 | Neprilysin-1 | 1 | 82.004 | 2.4 |
| B4GEL3 | E3 UFM1-protein ligase 1 homolog | 1 | 87.486 | 2.1 |
| Q9NFL6 | Hemocyanin C chain | 2 | 72.521 | 2.1 |
| Q9VL52 | Putative ATP-dependent RNA helicase | 1 | 168.374 | 2.0 |
表1 凝胶过滤层析5 min组分中鉴定的蛋白质
Tab.1 Identified proteins in the 5-min component separated by gel filtration chromatography
| Accession No. | Protein | #PSMs | MW | Score sequest HT |
|---|---|---|---|---|
| Q9NFL4 | Hemocyanin G chain | 8 | 71.766 | 17.8 |
| P02241 | Hemocyanin D chain | 9 | 72.077 | 17.6 |
| P02242 | Hemocyanin E chain | 8 | 71.63 | 15.3 |
| P41340 | Actin-3 | 6 | 41.781 | 11.7 |
| Q9NFH9 | Hemocyanin B chain | 7 | 72.052 | 10.7 |
| P14750 | Hemocyanin A chain | 4 | 72.273 | 6.0 |
| P0C2V1 | U15-hexatoxin-Mg1a | 2 | 6.946 | 4.7 |
| P80476 | Hemocyanin AA6 chain | 3 | 71.74 | 3.8 |
| W4VS99 | Neprilysin-1 | 1 | 82.004 | 2.4 |
| B4GEL3 | E3 UFM1-protein ligase 1 homolog | 1 | 87.486 | 2.1 |
| Q9NFL6 | Hemocyanin C chain | 2 | 72.521 | 2.1 |
| Q9VL52 | Putative ATP-dependent RNA helicase | 1 | 168.374 | 2.0 |
| Accession No. | Protein | #PSMs | MW | Score sequest HT |
|---|---|---|---|---|
| P68423 | U4-theraphotoxin-Hs1a | 59 | 3.134 | 254.7 |
| P0C2V2 | U15-hexatoxin-Mg1b | 32 | 6.904 | 101.0 |
| P0C2V1 | U15-hexatoxin-Mg1a | 30 | 6.946 | 88.0 |
| Q75WH4 | U10-hexatoxin-Mg1a | 9 | 8.876 | 35.2 |
| Q75WH1 | Mu-hexatoxin-Mg1c | 14 | 11.64 | 34.2 |
| P02241 | Hemocyanin D chain | 8 | 72.077 | 26.9 |
| Q9NFL4 | Hemocyanin G chain | 8 | 71.766 | 25.2 |
| P83558 | Mu-hexatoxin-Mg1a | 8 | 14.179 | 24.0 |
| Q75WG7 | U13-hexatoxin-Mg1a | 8 | 13.18 | 21.2 |
| P0DL75 | RTX-VII | 5 | 8.706 | 18.8 |
| P83561 | Beta-hexatoxin-Mg1a | 5 | 8.785 | 17.5 |
| P14750 | Hemocyanin A chain | 5 | 72.273 | 15.2 |
| P02242 | Hemocyanin E chain | 4 | 71.63 | 13.4 |
| P02572 | Actin-42A | 5 | 41.797 | 10.5 |
| Q9NFH9 | Hemocyanin B chain | 6 | 72.052 | 10.2 |
| P83559 | Mu-hexatoxin-Mg2a | 3 | 5.229 | 8.9 |
| P56676 | Mu/omega-theraphotoxin-Hs1a | 2 | 9.313 | 5.5 |
| P61233 | U5-hexatoxin-Mr1a(Raventoxin-1) | 1 | 4.845 | 4.3 |
| C0JB02 | Phospholipase D | 1 | 30.78 | 4.0 |
| Q9NFL6 | Hemocyanin C chain | 2 | 72.521 | 4.0 |
| P82959 | U1-theraphotoxin-Hs1a | 1 | 9.423 | 3.5 |
| Q86C49 | U1-theraphotoxin-Hs1f | 1 | 9.162 | 3.4 |
表2 凝胶过滤层析10 min组分中鉴定的多肽和蛋白质
Tab.2 Identified peptides and proteins in the 10-min component separated by gel filtration chromatography
| Accession No. | Protein | #PSMs | MW | Score sequest HT |
|---|---|---|---|---|
| P68423 | U4-theraphotoxin-Hs1a | 59 | 3.134 | 254.7 |
| P0C2V2 | U15-hexatoxin-Mg1b | 32 | 6.904 | 101.0 |
| P0C2V1 | U15-hexatoxin-Mg1a | 30 | 6.946 | 88.0 |
| Q75WH4 | U10-hexatoxin-Mg1a | 9 | 8.876 | 35.2 |
| Q75WH1 | Mu-hexatoxin-Mg1c | 14 | 11.64 | 34.2 |
| P02241 | Hemocyanin D chain | 8 | 72.077 | 26.9 |
| Q9NFL4 | Hemocyanin G chain | 8 | 71.766 | 25.2 |
| P83558 | Mu-hexatoxin-Mg1a | 8 | 14.179 | 24.0 |
| Q75WG7 | U13-hexatoxin-Mg1a | 8 | 13.18 | 21.2 |
| P0DL75 | RTX-VII | 5 | 8.706 | 18.8 |
| P83561 | Beta-hexatoxin-Mg1a | 5 | 8.785 | 17.5 |
| P14750 | Hemocyanin A chain | 5 | 72.273 | 15.2 |
| P02242 | Hemocyanin E chain | 4 | 71.63 | 13.4 |
| P02572 | Actin-42A | 5 | 41.797 | 10.5 |
| Q9NFH9 | Hemocyanin B chain | 6 | 72.052 | 10.2 |
| P83559 | Mu-hexatoxin-Mg2a | 3 | 5.229 | 8.9 |
| P56676 | Mu/omega-theraphotoxin-Hs1a | 2 | 9.313 | 5.5 |
| P61233 | U5-hexatoxin-Mr1a(Raventoxin-1) | 1 | 4.845 | 4.3 |
| C0JB02 | Phospholipase D | 1 | 30.78 | 4.0 |
| Q9NFL6 | Hemocyanin C chain | 2 | 72.521 | 4.0 |
| P82959 | U1-theraphotoxin-Hs1a | 1 | 9.423 | 3.5 |
| Q86C49 | U1-theraphotoxin-Hs1f | 1 | 9.162 | 3.4 |
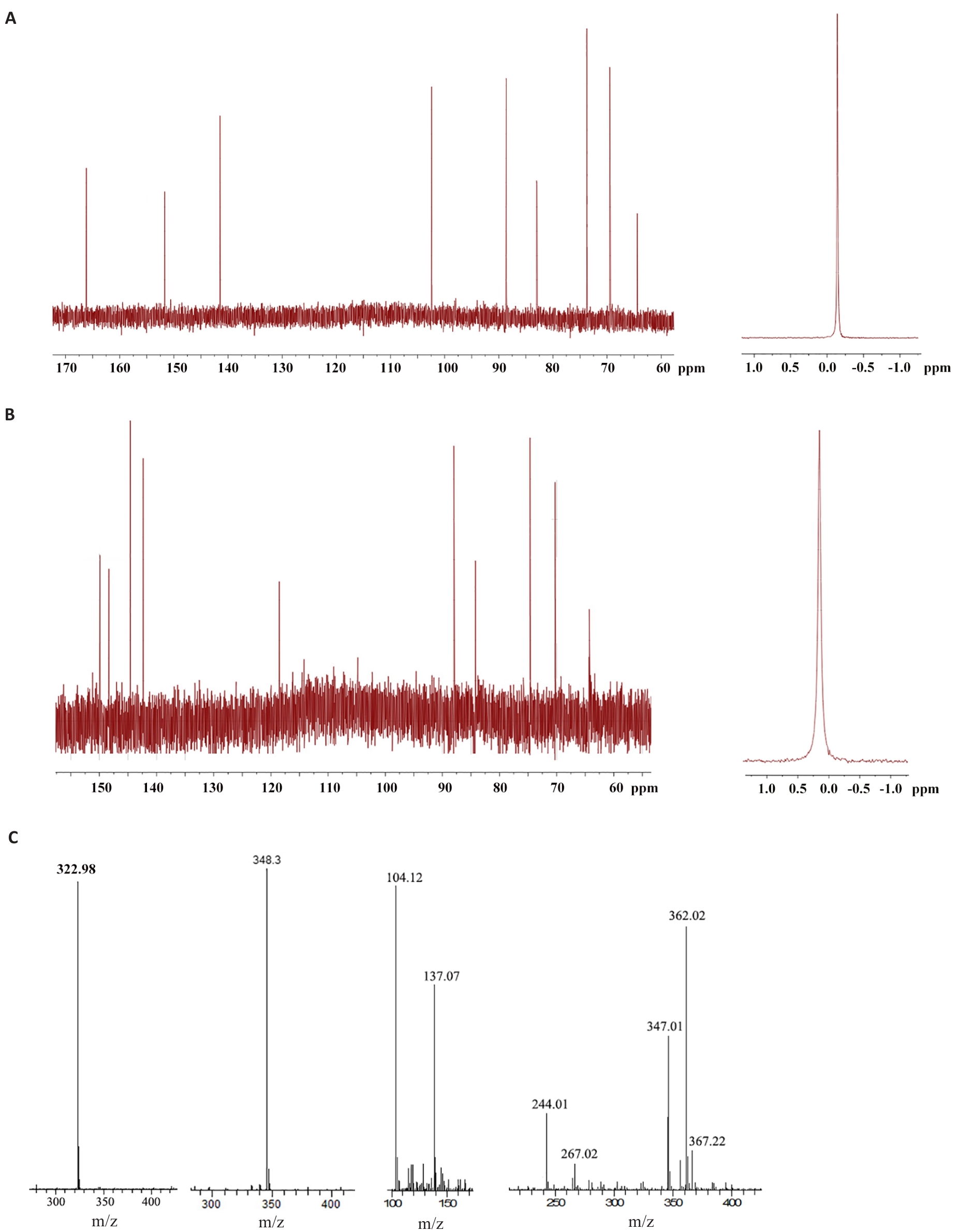
图7 雷氏大疣蛛毒素小分子化合物结构解析
Fig.7 Structural analysis of small molecule compounds isolated from Macrothele raveni venom. A: 600 MHz 13C NMR (left) and 31P NMR (right) spectrum of sample of peak a in Fig.4C. B: 600 MHz 13C NMR (left) and 31P NMR (right) spectrum of sample of peak b in Fig.4C. C: Mass spectrometry of the small molecule compounds isolated from Macrothele raveni venom.
| ID | a peak | UMP | b peak | AMP |
|---|---|---|---|---|
| 1 | 64.4 | 64.4 | 64.30 | 64.25 |
| 2 | 69.5 | 71 | 64.33 | 64.28 |
| 3 | 73.74 | 74.9 | 70.27 | 70.31 |
| 4 | 83.01 | 84.9 | 74.66 | 74.67 |
| 5 | 88.63 | 89.4 | 84.21 | 84.27 |
| 6 | 102.41 | 103.5 | 84.27 | 84.32 |
| 7 | 141.47 | 143 | 87.99 | 87.91 |
| 8 | 151.69 | 152.7 | 118.54 | 118.55 |
| 9 | 166.16 | 167.1 | 142.35 | 142.21 |
| 10 | 144.61 | 145.10 | ||
| 11 | 148.35 | 148.40 | ||
| 12 | 149.91 | 150.25 |
表3 13C NMR 化学位移(ppm):样本数据与文献数据或标准品数据比较
Tab.3 13C NMR chemical shifts (ppm): comparison between sample and literature or standard data
| ID | a peak | UMP | b peak | AMP |
|---|---|---|---|---|
| 1 | 64.4 | 64.4 | 64.30 | 64.25 |
| 2 | 69.5 | 71 | 64.33 | 64.28 |
| 3 | 73.74 | 74.9 | 70.27 | 70.31 |
| 4 | 83.01 | 84.9 | 74.66 | 74.67 |
| 5 | 88.63 | 89.4 | 84.21 | 84.27 |
| 6 | 102.41 | 103.5 | 84.27 | 84.32 |
| 7 | 141.47 | 143 | 87.99 | 87.91 |
| 8 | 151.69 | 152.7 | 118.54 | 118.55 |
| 9 | 166.16 | 167.1 | 142.35 | 142.21 |
| 10 | 144.61 | 145.10 | ||
| 11 | 148.35 | 148.40 | ||
| 12 | 149.91 | 150.25 |
| Sample | Small-molecule compounds | Standards | |||
|---|---|---|---|---|---|
| MW | Retention time (min) | Standards | MW | Retention time (min) | |
| 1 | 322.98 (-) | 3.41 | UMP | 324.2 | 3.43 |
| 2 | 348.3 (-) | 3.64 | AMP | 347.2 | 3.62 |
| 3 | 104.12 (+) | 15.03 | GABA | 103.1 | 15.08 |
| 4 | 137.07 (+) | 4.14 | Hypoxanthine | 136.1 | 4.11 |
| 5 | 244.01 (-) | 4.91 | |||
| 6 | 267.02 (-) | 4.45 | Inosine | 268.2 | 4.41 |
| 7 | 347.01 (-) | 3.54 | IMP | 348.2 | 3.52 |
| 8 | 362.02 (-) | 3.71 | GMP | 363.2 | 3.77 |
| 9 | 367.22 (-) | 5.74 | |||
表4 雷氏大疣蛛毒素中小分子组分的分子量和洗脱保留时间与标准品比较。
Tab.4 Comparison relative molecular mass and retention time between small-molecule compounds from Macrothele raveni venom and standards
| Sample | Small-molecule compounds | Standards | |||
|---|---|---|---|---|---|
| MW | Retention time (min) | Standards | MW | Retention time (min) | |
| 1 | 322.98 (-) | 3.41 | UMP | 324.2 | 3.43 |
| 2 | 348.3 (-) | 3.64 | AMP | 347.2 | 3.62 |
| 3 | 104.12 (+) | 15.03 | GABA | 103.1 | 15.08 |
| 4 | 137.07 (+) | 4.14 | Hypoxanthine | 136.1 | 4.11 |
| 5 | 244.01 (-) | 4.91 | |||
| 6 | 267.02 (-) | 4.45 | Inosine | 268.2 | 4.41 |
| 7 | 347.01 (-) | 3.54 | IMP | 348.2 | 3.52 |
| 8 | 362.02 (-) | 3.71 | GMP | 363.2 | 3.77 |
| 9 | 367.22 (-) | 5.74 | |||
| [1] | Tan KK, Bay BH, Gopalakrishnakone P. L-amino acid oxidase from snake venom and its anticancer potential[J]. Toxicon, 2018, 144: 7-13. doi:10.1016/j.toxicon.2018.01.015 |
| [2] | Ullah A. Structure-function studies and mechanism of action of snake venom L-amino acid oxidases[J]. Front Pharmacol, 2020, 11: 110. doi:10.3389/fphar.2020.00110 |
| [3] | Akhtar B, Muhammad F, Sharif A, et al. Mechanistic insights of snake venom disintegrins in cancer treatment[J]. Eur J Pharmacol, 2021, 899: 174022. doi:10.1016/j.ejphar.2021.174022 |
| [4] | Schönthal AH, Swenson SD, Chen TC, et al. Preclinical studies of a novel snake venom-derived recombinant disintegrin with antitumor activity: a review[J]. Biochem Pharmacol, 2020, 181: 114149. doi:10.1016/j.bcp.2020.114149 |
| [5] | Dardevet L, Rani D, Aziz TA, et al. Chlorotoxin: a helpful natural scorpion peptide to diagnose glioma and fight tumor invasion[J]. Toxins (Basel), 2015, 7(4): 1079-101. doi:10.3390/toxins7041079 |
| [6] | Duan XF, Zou HY, Yang JZ, et al. Melittin-incorporated nanomedicines for enhanced cancer immunotherapy[J]. J Control Release, 2024, 375: 285-99. doi:10.1016/j.jconrel.2024.08.047 |
| [7] | Zhang P, Luo W, Zhang ZX, et al. A lipid-sensitive spider peptide toxin exhibits selective anti-leukemia efficacy through multimodal mechanisms[J]. Adv Sci (Weinh), 2024, 11(32): e2404937. doi:10.1002/advs.202404937 |
| [8] | Buglione A, Alloisio G, Ciaccio C, et al. GsMTx-4 venom toxin antagonizes biophysical modulation of metastatic traits in human osteosarcoma cells[J]. Eur J Cell Biol, 2025, 104(1): 151469. doi:10.1016/j.ejcb.2024.151469 |
| [9] | 胡增祥, 杜昱蕾, 刘全喜, 等. 雷氏大疣蛛蜘蛛毒素对人肺癌细胞A549增殖的影响[J]. 中国肺癌杂志, 2010, 13(10): 933-6. doi:10.3779/j.issn.1009-3419.2010.10.02 |
| [10] | 高 莉, 沈金宝, 孙 捷, 等. 雷氏大疣蛛毒液对人肝癌HepG2细胞p21基因表达的影响[J]. 生理学报, 2007, 59(1): 58-62. doi:10.3321/j.issn:0371-0874.2007.01.010 |
| [11] | Liu ZH, Zhao Y, Li J, et al. The venom of the spider Macrothele raveni induces apoptosis in the myelogenous leukemia K562 cell line[J]. Leuk Res, 2012, 36(8): 1063-6. doi:10.1016/j.leukres.2012.02.025 |
| [12] | Gottlieb B, Trifiro M, Batist G. Why tumor genetic heterogeneity may require rethinking cancer genesis and treatment[J]. Trends Cancer, 2021, 7(5): 400-9. doi:10.1016/j.trecan.2020.10.013 |
| [13] | Fruman DA, Chiu H, Hopkins BD, et al. The PI3K pathway in human disease[J]. Cell, 2017, 170(4): 605-35. doi:10.1016/j.cell.2017.07.029 |
| [14] | Bi SY, Chen W, Fang YY, et al. Cancer cell-selective membrane receptor clustering driven by VEGF secretion for in vivo therapy[J]. J Am Chem Soc, 2023, 145(9): 5041-52. doi:10.1021/jacs.2c10428 |
| [15] | Dobrzyńska I, Szachowicz-Petelska B, Sulkowski S, et al. Changes in electric charge and phospholipids composition in human colorectal cancer cells[J]. Mol Cell Biochem, 2005, 276(1): 113-9. doi:10.1007/s11010-005-3557-3 |
| [16] | Chang Y, Chen JY, Yang J, et al. Targeting the Cell Membrane by Charge-Reversal Amphiphilic Pillar[5]arene for the Selective Killing of Cancer Cells[J]. ACS Appl Mater Interfaces, 2019, 11(42): 38497-502. doi:10.1021/acsami.9b13492 |
| [17] | Li XL, Chen ZW, Li YM, et al. Optical tweezers study of membrane fluidity in small cell lung cancer cells[J]. Opt Express, 2021, 29(8): 11976. doi:10.1364/oe.420288 |
| [18] | Sahoo G, Samal D, Khandayataray P, et al. A review on caspases: key regulators of biological activities and apoptosis[J]. Mol Neurobiol, 2023, 60(10): 5805-37. doi:10.1007/s12035-023-03433-5 |
| [19] | Yuan JY, Ofengeim D. A guide to cell death pathways[J]. Nat Rev Mol Cell Biol, 2024, 25(5): 379-95. doi:10.1038/s41580-023-00689-6 |
| [20] | Ramaswamy M, Efimova EV, Martinez O, et al. IG20 (MADD splice variant-5), a proapoptotic protein, interacts with DR4/DR5 and enhances TRAIL-induced apoptosis by increasing recruitment of FADD and caspase-8 to the DISC[J]. Oncogene, 2004, 23(36): 6083-94. doi:10.1038/sj.onc.1207804 |
| [21] | Capera J, Pérez-Verdaguer M, Peruzzo R, et al. A novel mitochondrial Kv1.3-caveolin axis controls cell survival and apoptosis[J]. eLife, 2021, 10: e69099. doi:10.7554/elife.69099 |
| [22] | Teisseyre A, Chmielarz M, Uryga A, et al. Co-application of statin and flavonoids as an effective strategy to reduce the activity of voltage-gated potassium channels Kv1.3 and induce apoptosis in human leukemic T cell line jurkat[J]. Molecules, 2022, 27(10): 3227. doi:10.3390/molecules27103227 |
| [23] | Xiao Z, You X, Chen MZ, et al. A novel spider toxin as a selective antagonist of the Kv1 subfamily of voltage-gated potassium channels through gating modulation[J]. J Biol Chem, 2025, 301(4): 108341. doi:10.1016/j.jbc.2025.108341 |
| [24] | Liang SP. An overview of peptide toxins from the venom of the Chinese bird spider Selenocosmia huwena Wang [=Ornithoctonus huwena (Wang)[J]. Toxicon, 2004, 43(5): 575-85. doi:10.1016/j.toxicon.2004.02.005 |
| [25] | Xiao X, Luo XQ, Huang CL, et al. Transcriptome analysis reveals the peptide toxins diversity of Macrothele palpator venom[J]. Int J Biol Macromol, 2023, 253(Pt 1): 126577. doi:10.1016/j.ijbiomac.2023.126577 |
| [26] | Corzo G, Gilles N, Satake H, et al. Distinct primary structures of the major peptide toxins from the venom of the spider Macrothele gigas that bind to sites 3 and 4 in the sodium channel[J]. FEBS Lett, 2003, 547(1/2/3): 43-50. doi:10.1016/s0014-5793(03)00666-5 |
| [27] | Titaux-Delgado G, Carrillo E, Mendoza A, et al. Successful refolding and NMR structure of rMagi3: a disulfide-rich insecticidal spider toxin[J]. Protein Sci, 2018, 27(3): 692-701. doi:10.1002/pro.3363 |
| [28] | Suchyna TM, Johnson JH, Hamer K, et al. Identification of a peptide toxin from Grammostola spatulata spider venom that blocks cation-selective stretch-activated channels[J]. J Gen Physiol, 2000, 115(5): 583-98. doi:10.1085/jgp.115.5.583 |
| [29] | Boța M, Vlaia L, Jîjie AR, et al. Exploring synergistic interactions between natural compounds and conventional chemotherapeutic drugs in preclinical models of lung cancer[J]. Pharmaceuticals (Basel), 2024, 17(5): 598. doi:10.3390/ph17050598 |
| [30] | He M, Liao QY, Liu D, et al. Dihydroergotamine mesylate enhances the anti-tumor effect of sorafenib in liver cancer cells[J]. Biochem Pharmacol, 2023, 211: 115538. doi:10.1016/j.bcp.2023.115538 |
| [31] | Kumar S, Arora A, Chaudhary R, et al. Recent advances in the synthesis of acyclic nucleosides and their therapeutic applications[J]. Top Curr Chem (Cham), 2024, 382(4): 34. doi:10.1007/s41061-024-00476-7 |
| [32] | Sigmond J, Peters GJ. Pyrimidine and purine analogues, effects on cell cycle regulation and the role of cell cycle inhibitors to enhance their cytotoxicity[J]. Nucleosides Nucleotides Nucleic Acids, 2005, 24(10/11/12): 1997-2022. doi:10.1080/15257770500269556 |
| [1] | 于滢, 涂丽, 刘洋, 宋雪翼, 邵倩倩, 唐小龙. TGF-β通过miR-23a-3p/IRF1轴下调主要组织相容性复合体I类表达促进肝癌免疫逃逸[J]. 南方医科大学学报, 2025, 45(7): 1397-1408. |
| [2] | 黄萃园, 孙运平, 李文强, 刘丽, 王伟, 张静. Nlrp6过表达通过调控AMPK-Srebp1c轴抑制脂质合成抑制肝癌细胞的增殖[J]. 南方医科大学学报, 2024, 44(10): 1910-1917. |
| [3] | 汪虹晓, 陶德韬, 马俊杰, 张东林, 沈左媛, 邓超, 周静萍. 顺铂诱导TNF-α自分泌引发头颈部鳞状癌细胞RIP1/RIP3/MLKL的坏死性凋亡[J]. 南方医科大学学报, 2024, 44(10): 1947-1954. |
| [4] | 辛 辰, 王笑影, 李 响, 陈 宇, 王 雪, 宁佳曦, 杨 适, 王忠琼. LncRNA SOX2OT靶向SIRT1/自噬通路增强胆管癌细胞5-FU耐药[J]. 南方医科大学学报, 2024, 44(1): 187-193. |
| [5] | 潘中武, 李思琪, 王耀辉, 刘海军, 桂 琳, 董博翰. 低HMGB1含量的肿瘤细胞裂解物增强树突状细胞的抗肺癌作用[J]. 南方医科大学学报, 2023, 43(6): 906-914. |
| [6] | 辛 辰, 王笑影, 李 响, 陈 宇, 王 雪, 宁佳曦, 杨 适, 王忠琼. 沉默SIRT1降低胆管癌细胞对 5-氟尿嘧啶的耐药:基于抑制FOXO1/Rab7自噬通路[J]. 南方医科大学学报, 2023, 43(3): 454-459. |
| [7] | 张玉心, 高美佳, 朱美林, 李红梅, 马 涛, 吴成柱. 补骨脂乙素通过多途径诱导人乳腺癌MCF-7细胞死亡[J]. 南方医科大学学报, 2022, 42(6): 878-885. |
| [8] | 徐 璐, 钱 峰, 孙 磊. 三硫二苄基抑制头颈部鳞状癌细胞HN30的增殖并诱导其凋亡[J]. 南方医科大学学报, 2021, 41(6): 847-853. |
| [9] | 朱晨露, 杜家如, 姚言雪, 武丹丹, 苑 敏, 干 露, 童旭辉. 沉默ATG5及ATG7可抑制顺铂介导的耐顺铂睾丸癌细胞的自噬和增殖[J]. 南方医科大学学报, 2021, 41(5): 657-663. |
| [10] | 赵 珂,郭玉刚,霍 正,马国辉,张 桂,邢雨欣,徐 茜. 食管鳞癌患者血清中lncRNA-TUSC7的表达及与细胞侵袭转移的关系[J]. 南方医科大学学报, 2020, 40(05): 661-669. |
| [11] | 朱云,李成,林鑫盛,孙晶晶,程旸. 白术多糖对肝癌细胞增殖及侵袭的抑制作用及其机制[J]. 南方医科大学学报, 2019, 39(10): 1180-. |
| [12] | 马金珠,朱益平,王箴,昝嘉伟,曹陇,冯遵永,王森林,范倩,颜亮. miR-593 通过调控PLK1基因的表达抑制结肠癌细胞的增殖[J]. 南方医科大学学报, 2019, 39(02): 144-. |
| [13] | 安海燕,林俊豪,孙海涛,许梨梨,苏嘉琪,何春雨,曾嘉敏,梁佩湘,贺松其. 鳖甲煎丸通过RhoA/ROCK信号通路抑制肝癌细胞血管形成拟态的生成[J]. 南方医科大学学报, 2018, 38(08): 997-. |
| [14] | 孙小锦,马琳艳,张梦晓,王颖,张配,蒋琛琛,刘浩. 氯喹通过miR-26b调控Mcl-1诱导人肝癌HepG2细胞凋亡[J]. 南方医科大学学报, 2018, 38(04): 409-. |
| [15] | 张韫琪,蔡云,刘媛,赵博欣,李国锋. 甘草次酸选择性抑制肝癌细胞的增殖[J]. 南方医科大学学报, 2018, 38(04): 477-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||