南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 675-685.doi: 10.12122/j.issn.1673-4254.2026.03.21
• 基础研究 • 上一篇
收稿日期:2025-06-29
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
汪元
E-mail:3253806681@qq.com;echowang0268@126.com
作者简介:孙梦雨,在读硕士研究生,E-mail:3253806681@qq.com
基金资助:
Mengyu SUN1( ), Yuan WANG2(
), Yuan WANG2( ), Feifei LIU1
), Feifei LIU1
Received:2025-06-29
Online:2026-03-20
Published:2026-03-26
Contact:
Yuan WANG
E-mail:3253806681@qq.com;echowang0268@126.com
摘要:
目的 研究黄芩清热除痹胶囊(HQC)调控LncRNA EBLN3P/miR-369-3p/NFIX 轴干预类风湿关节炎(RA)中JAK/STAT驱动的滑膜血管生成的作用机制。 方法 通过优化干预策略构建类风湿关节炎成纤维样滑膜细胞/人脐静脉内皮细胞(RA-FLS/HUVEC)协同培养模型,采用梯度浓度含HQC血清(2.5%~30%,干预24~72 h)、LncRNA EBLN3P过表达及基因-药物联合处理,利用CCK-8法筛选共培养体系的最佳血清浓度与干预时长。通过EdU增殖实验、Transwell侵袭实验及细胞划痕实验评估HQC对RA-FLS病理活化的抑制作用,ELISA定量分析血管生成因子(VEGF、FGF2)及基质金属蛋白酶(MMP9、MMP2)表达水平,管腔形成实验联合免疫荧光技术检测RA-FLS诱导的HUVEC血管生成能力及内皮标志物(CD34、CD105)表达变化,qRT-PCR及Western blotting解析信号通路关键分子(LncEBLN3P、miR-369-3p、NFIX、JAK2、STAT3、p-JAK2、p-STAT3)的mRNA或蛋白表达动态。 结果 共培养体系优化作用条件(细胞比例5:1,时间48 h)模型中,与对照组相比,模型组RA-FLS的恶性行为(增殖、侵袭及迁移能力)显著增强(P<0.01),促血管生成因子(VEGF、FGF2)与基质降解酶(MMP2、MMP9)表达显著上调(P<0.01),LncEBLN3P/miR-369-3p/NFIX轴异常激活,miR-369-3p表达受抑制(P<0.01),而LncEBLN3P、NFIX及下游JAK2/STAT3通路关键分子(JAK2、STAT3、p-JAK2、p-STAT3)表达均显著升高(P<0.01)。经HQC干预后,模型组RA-FLS恶性行为受抑(P<0.01),促血管因子及基质降解酶表达下调(P<0.01),且LncEBLN3P/miR-369-3p/NFIX-JAK/STAT轴被抑制(miR-369-3p表达升高,P<0.01;LncEBLN3P、NFIX、JAK2、STAT3、p-JAK2、p-STAT3表达均下调,P<0.01)。在LncRNA EBLN3P过表达(OE-Lnc)模型中,HQC仍可部分逆转其诱导的病理表型及通路激活(P<0.01 vs OE-Lnc组),但与单纯HQC处理组相比,OE-Lnc+HQC联合处理组对细胞恶性行为及通路活化的抑制作用显著减弱(P<0.01)。 结论 HQC含药血清可能通过靶向LncRNA EBLN3P/miR-369-3p/NFIX轴抑制JAK/STAT通路,从而抑制RA-FLS的促血管生成功能。
孙梦雨, 汪元, 刘菲菲. 黄芩清热除痹胶囊调控LncRNA EBLN3P/miR-369-3p/NFIX轴抑制JAK/STAT通路介导的类风湿关节炎滑膜血管新生[J]. 南方医科大学学报, 2026, 46(3): 675-685.
Mengyu SUN, Yuan WANG, Feifei LIU. Huangqin Qingre Chubi Capsule inhibits JAK/STAT-driven synovial angiogenesis in rheumatoid arthritis by suppressing the LncRNA EBLN3P/miR-369-3p/NFIX axis[J]. Journal of Southern Medical University, 2026, 46(3): 675-685.
| Gene | Amplicon size (bp) | Forward primer (5'→3') | Reverse primer (5'→3') |
|---|---|---|---|
| Hu-β-actin | 96 | CCCTGGAGAAGAGCTACGAG | GGAAGGAAGGCTGGAAGAGT |
| Hu-U6 | 94 | CTCGCTTCGGCAGCACA | AACGCTTCACGAATTTGCGT |
| Hu-Linc-EBLN3P | 151 | GTCCAGTCTTTGAGGACCGA | TGGTTCCTATGCCCAGATCG |
| Hu-NFIX | 80 | ACGGCTGCGATAGAACATGG | GGTGGAACTCATCACACGCT |
| hsa-miR-369-3p | CCGCGCAATACATGGTTG | AGTGCAGGGTCCGAGGTATT | |
| hsa-miR-369-3p RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAAAGAT | ||
| Hu-STAT3 | 124 | GGAGAAACAGGATGGCCCAA | ATCCAAGGGGCCAGAAACTG |
| Hu-JAK2 | 88 | TGAGTTCGAAGCTAGCAGGGC | ACAGTTGTCTCCACCCTCTCC |
表1 引物序列
Tab.1 Primer sequences
| Gene | Amplicon size (bp) | Forward primer (5'→3') | Reverse primer (5'→3') |
|---|---|---|---|
| Hu-β-actin | 96 | CCCTGGAGAAGAGCTACGAG | GGAAGGAAGGCTGGAAGAGT |
| Hu-U6 | 94 | CTCGCTTCGGCAGCACA | AACGCTTCACGAATTTGCGT |
| Hu-Linc-EBLN3P | 151 | GTCCAGTCTTTGAGGACCGA | TGGTTCCTATGCCCAGATCG |
| Hu-NFIX | 80 | ACGGCTGCGATAGAACATGG | GGTGGAACTCATCACACGCT |
| hsa-miR-369-3p | CCGCGCAATACATGGTTG | AGTGCAGGGTCCGAGGTATT | |
| hsa-miR-369-3p RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAAAGAT | ||
| Hu-STAT3 | 124 | GGAGAAACAGGATGGCCCAA | ATCCAAGGGGCCAGAAACTG |
| Hu-JAK2 | 88 | TGAGTTCGAAGCTAGCAGGGC | ACAGTTGTCTCCACCCTCTCC |
| Product name | Manufacturer | Product number | Batch number | Theoretical molecular weight | Antibody source | Dilution ratio | Separation gel concentration |
|---|---|---|---|---|---|---|---|
| β-actin | Zs-BIO | TA-09 | 19AW0505 | 42 000 | mouse | 1:1000 | 10% |
| Goat anti-mouse IgG antibody | Zs-BIO | ZB-2305 | 249760203 | 1:10000 | |||
| Goat anti-rabbit IgG antibody | Zs-BIO | ZB-2301 | 247860216 | 1:10000 | |||
| JAK2 | Affinity | AF6022 | 22V9316 | 131 000 | rabbit | 1:1000 | 10% |
| P-JAK2 | Affinity | AF3024 | 75d7669 | 131 000 | rabbit | 1:1000 | 10% |
| STAT3 | Affinity | AF6294 | 15X8824 | 88 000 | rabbit | 1:1000 | 10% |
| P-STAT3 | Affinity | AF3293 | 74M1478 | 88 000 | rabbit | 1:1000 | 10% |
| NFIX | HUABIO | ER1913-79 | H650014015 | 55 000 | rabbit | 1:1000 | 10% |
表2 Western blotting所用抗体信息
Tab.2 Antibodies for Western blotting
| Product name | Manufacturer | Product number | Batch number | Theoretical molecular weight | Antibody source | Dilution ratio | Separation gel concentration |
|---|---|---|---|---|---|---|---|
| β-actin | Zs-BIO | TA-09 | 19AW0505 | 42 000 | mouse | 1:1000 | 10% |
| Goat anti-mouse IgG antibody | Zs-BIO | ZB-2305 | 249760203 | 1:10000 | |||
| Goat anti-rabbit IgG antibody | Zs-BIO | ZB-2301 | 247860216 | 1:10000 | |||
| JAK2 | Affinity | AF6022 | 22V9316 | 131 000 | rabbit | 1:1000 | 10% |
| P-JAK2 | Affinity | AF3024 | 75d7669 | 131 000 | rabbit | 1:1000 | 10% |
| STAT3 | Affinity | AF6294 | 15X8824 | 88 000 | rabbit | 1:1000 | 10% |
| P-STAT3 | Affinity | AF3293 | 74M1478 | 88 000 | rabbit | 1:1000 | 10% |
| NFIX | HUABIO | ER1913-79 | H650014015 | 55 000 | rabbit | 1:1000 | 10% |
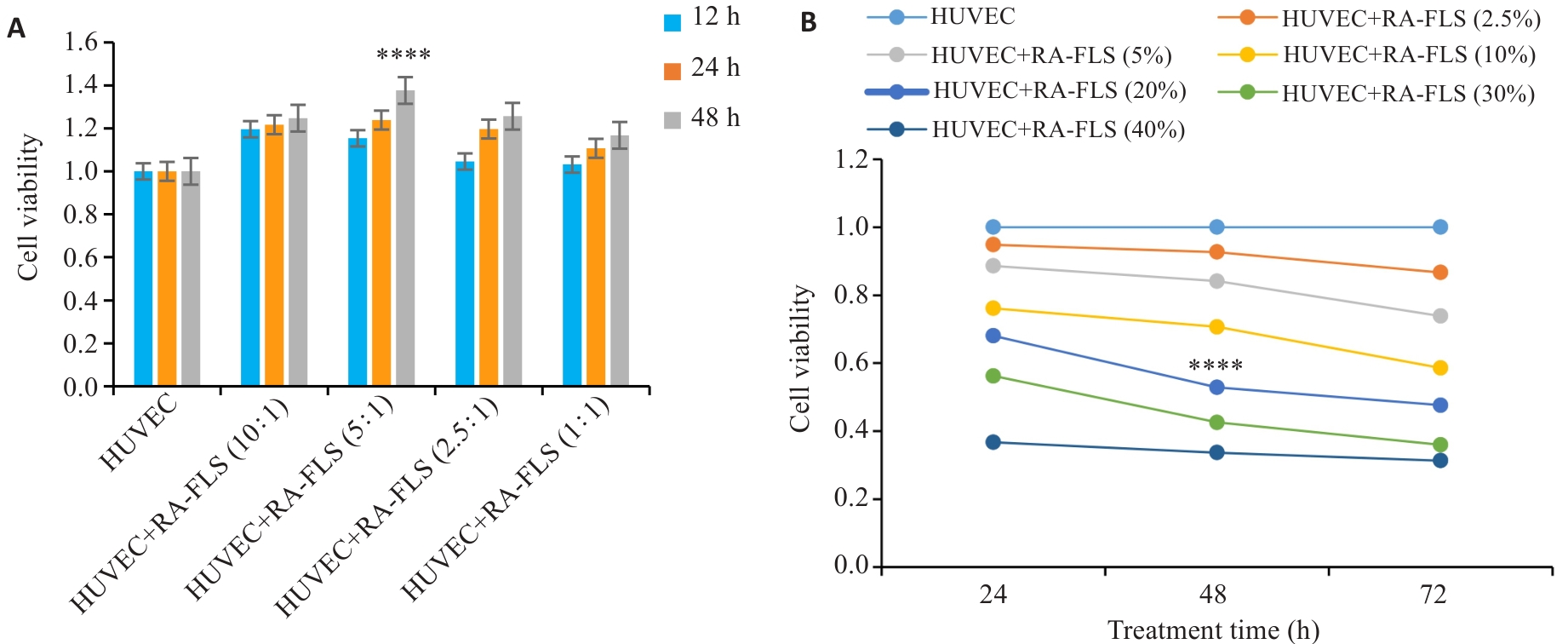
图1 RA-FLS/HUVEC共培养体系建立及HQC给药参数优化
Fig. 1 Establishment of RA-FLS/HUVEC co-culture system and optimization of HQC treatment protocol.A: CCK-8 assay for determining optimal co-culture time (n=6). ****P<0.0001 vs the same time point in other groups. B: Dose-time effect analysis of HQC-medicated serum (n=6). ****P<0.0001 vs all other groups.
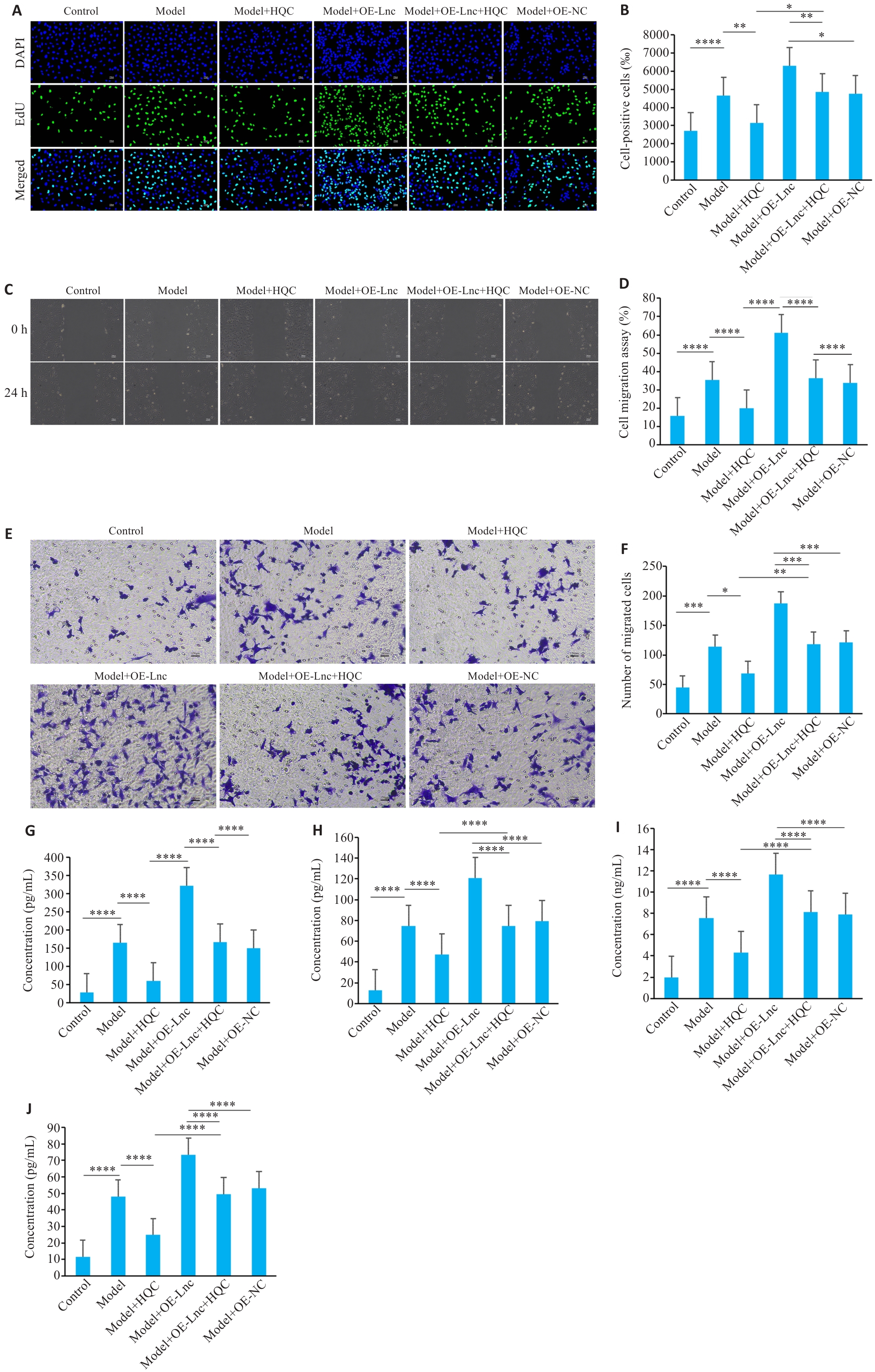
图2 HQC抑制RA-FLS的病理活化、血管生成因子及基质金属蛋白酶表达
Fig.2 HQC inhibits pathological activation, angiogenesis factor, and matrix metalloproteinase expression in RA-FLS. A, B: EdU assay for assessing cell proliferation (n=3) using Click Chemistry fluorescent staining (Original magnification, ×200). C, D: Scratch assay for assessing cell migration ability (n=3,×100). E, F: Transwell assay for assessing cell invasion ability (n=3) with crystal violet staining (×200). G-I: Detection of the concentrations of angiogenic factors and matrix-degrading enzymes by ELISA (n=6). ****P<0.0001, ***P<0.001, **P<0.01, * P<0.05.
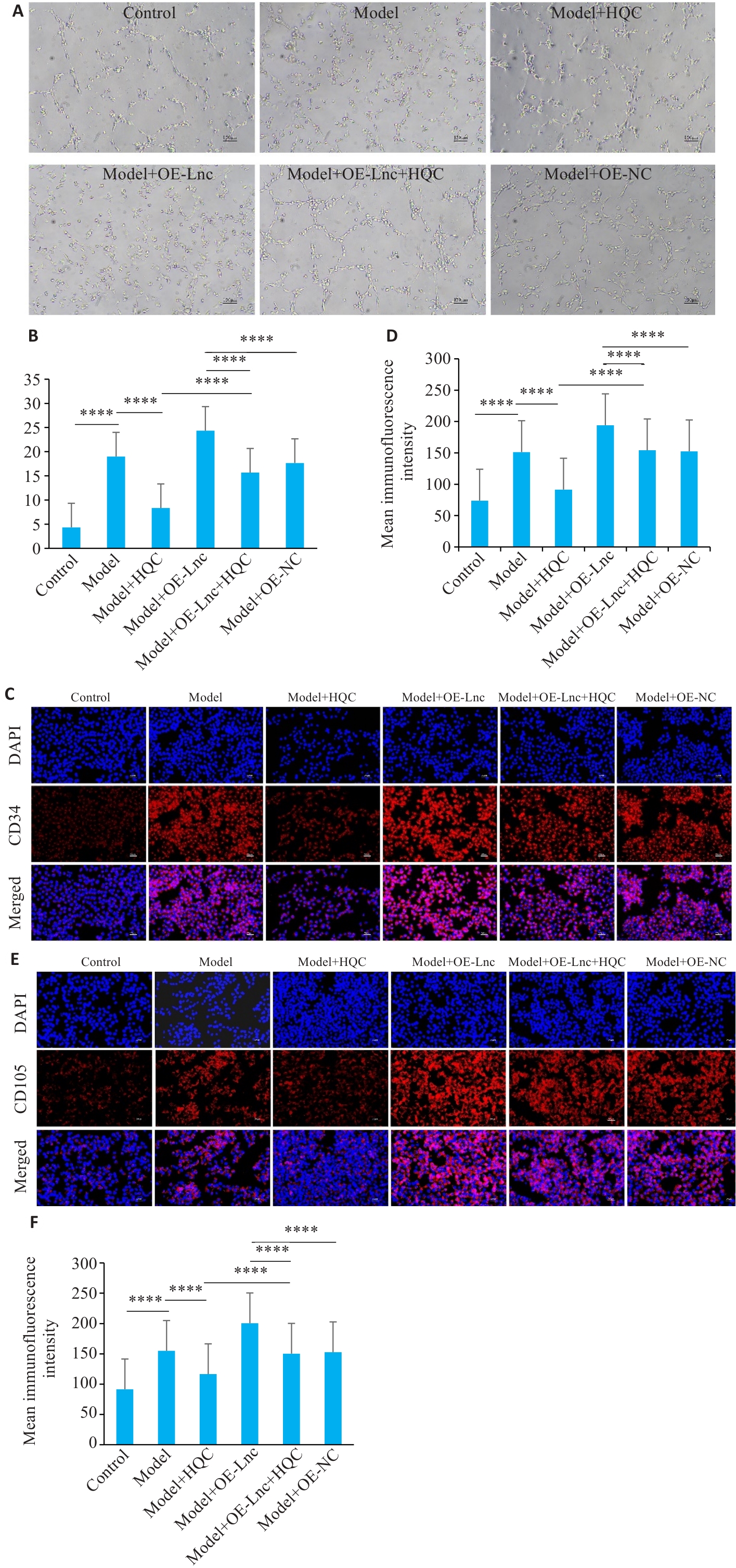
图3 HQC抑制RA-FLS诱导的HUVEC血管生成和血管内皮标志物表达
Fig.3 HQC inhibits RA-FLS-induced HUVEC angiogenesis and vascular endothelial marker expression. A, B: Angiogenesis assay of testing HUVEC tubule formation ability (n=3,×200). C, D: CD34 immunofluorescence quantification (n=3) using a dual-color indirect immunofluo-rescence staining method (×200). E, F: CD105 immunofluorescence quantification (n=3) using a dual-color indirect immunofluorescence staining method (×200). ****P<0.0001.
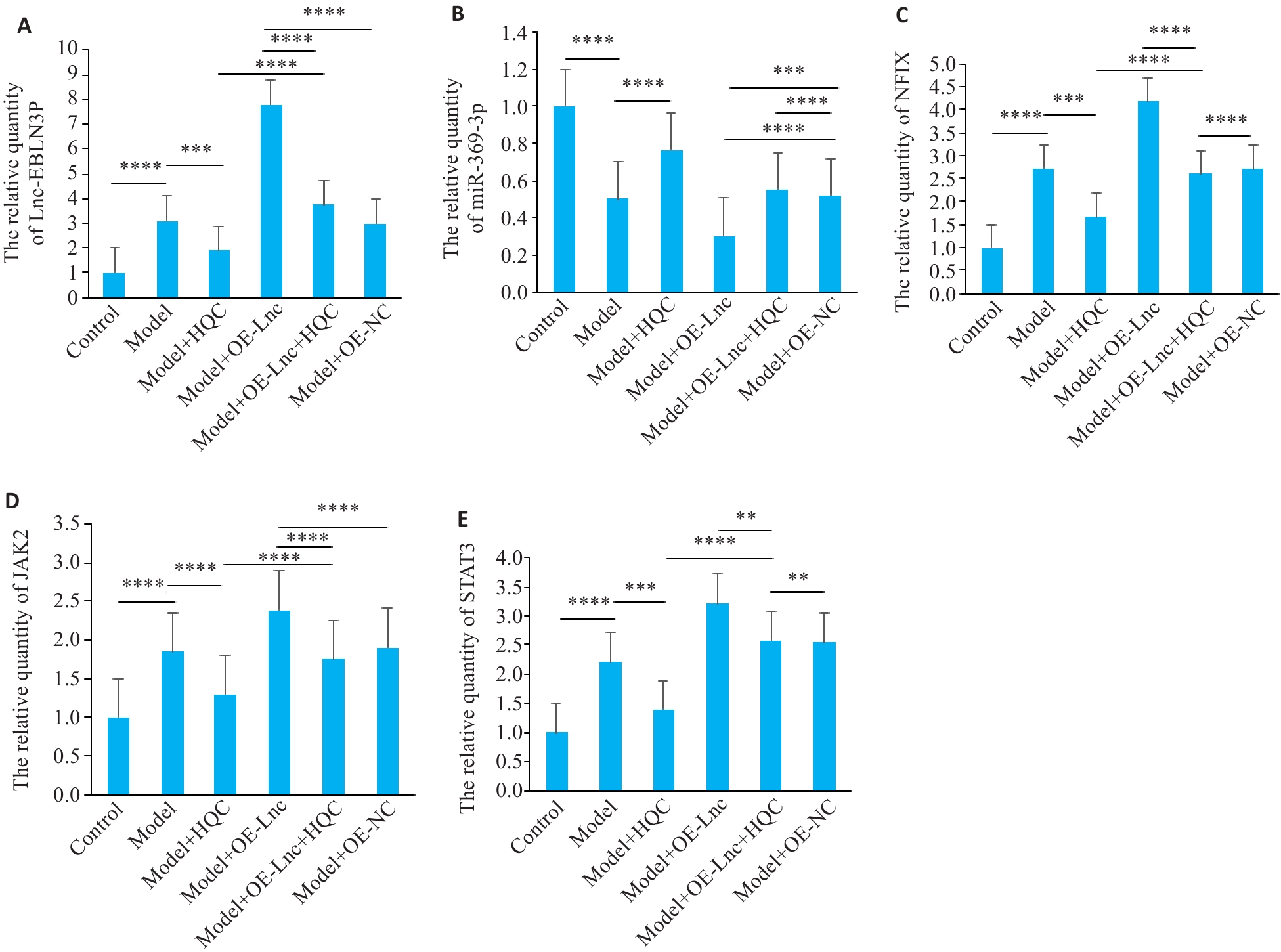
图4 HQC下调RA-FLS诱导的HUVEC信号转导通路关键分子的表达
Fig.4 HQC downregulates expressions of key molecules in the RA-FLS-induced HUVEC signal transduction pathway. A-E: qRT-PCR detection of molecular expression of regulatory axes and their targeted signaling pathways (n=6). ****P<0.0001, ***P<0.001, **P<0.01.
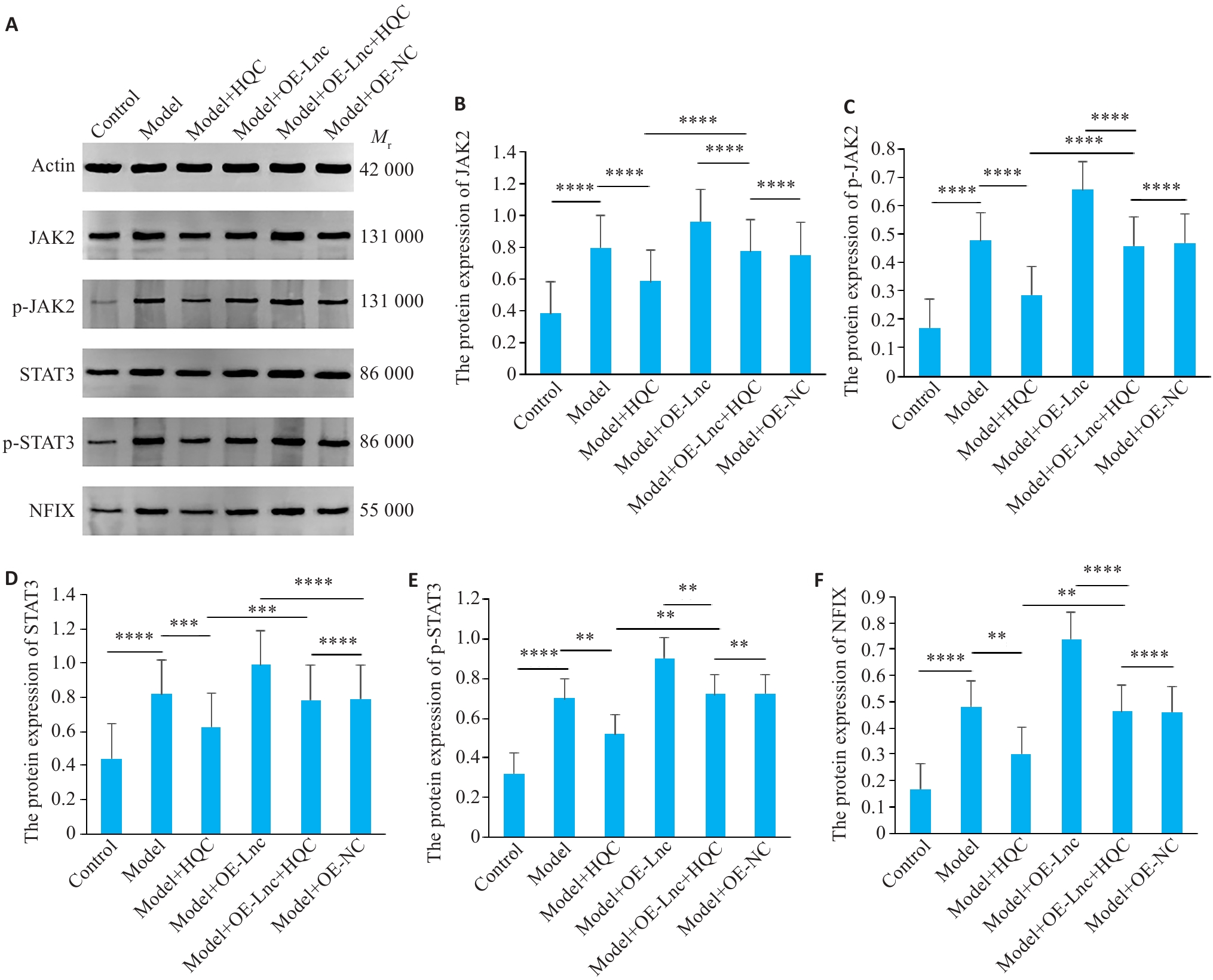
图5 HQC抑制RA-FLS诱导的HUVEC信号转导相关蛋白活性
Fig.5 HQC inhibits RA-FLS-induced HUVEC signal transduction-related protein activity. A: Western blots of the proteins. B-F: Expression levels of the pathway-related proteins (n=3).****P<0.0001, ***P<0.001, **P<0.01.
| [1] | Jang S, Kwon EJ, Lee JJ. Rheumatoid arthritis: pathogenic roles of diverse immune cells[J]. Int J Mol Sci, 2022, 23(2): 905. doi:10.3390/ijms23020905 |
| [2] | Zheng YX, Wei K, Jiang P, et al. Macrophage polarization in rheumatoid arthritis: signaling pathways, metabolic reprogramming, and crosstalk with synovial fibroblasts[J]. Front Immunol, 2024, 15: 1394108. doi:10.3389/fimmu.2024.1394108 |
| [3] | Tsaltskan V, Firestein GS. Targeting fibroblast-like synoviocytes in rheumatoid arthritis[J]. Curr Opin Pharmacol, 2022, 67: 102304. doi:10.1016/j.coph.2022.102304 |
| [4] | Simon LS, Taylor PC, Choy EH, et al. The Jak/STAT pathway: a focus on pain in rheumatoid arthritis[J]. Semin Arthritis Rheum, 2021, 51(1): 278-84. doi:10.1016/j.semarthrit.2020.10.008 |
| [5] | Xia X, He C, Xue Z, et al. Single cell immunoprofile of synovial fluid in rheumatoid arthritis with TNF/JAK inhibitor treatment[J]. Nat Commun, 2025, 16(1): 2152. doi:10.1038/s41467-025-57361-0 |
| [6] | Xu D, Jiang Y, Yang L, et al. Long noncoding RNAs expression profile and functional networks in rheumatoid arthritis[J]. Oncotarget, 2017, 8(56): 95280-92. doi:10.18632/oncotarget.20036 |
| [7] | Chan JJ, Tay Y. Noncoding RNA: RNA regulatory networks in cancer[J]. Int J Mol Sci, 2018, 19(5): E1310. doi:10.3390/ijms19051310 |
| [8] | Fan Z, Gao S, Chen Y, et al. Integrative analysis of competing endogenous RNA networks reveals the functional lncRNAs in heart failure[J]. J Cell Mol Med, 2018, 22(10): 4818-29. doi:10.1111/jcmm.13739 |
| [9] | Li LJ, Zhao W, Tao SS, et al. Competitive endogenous RNA network: potential implication for systemic lupus erythematosus[J]. Expert Opin Ther Targets, 2017, 21(6): 639-48. doi:10.1080/14728222.2017.1319938 |
| [10] | Hu Y, Liu J, Xin L, et al. Huangqin Qingre chubi capsule is associated with reduced risk of readmission in patients with rheumatoid arthritis: a real-world retrospective cohort study[J]. Int J Gen Med, 2023, 16: 4819-34. doi:10.2147/ijgm.s431124 |
| [11] | 王玉亭, 吴雅婕, 黄玉蓉, 等. 基于METTL3-SFRP4/Wnt/β-catenin通路探讨黄芩清热除痹胶囊改善类风湿性关节炎的机制[J]. 2024(11): 3081-94. |
| [12] | 周婉婉. 黄芩清热除痹胶囊通过FZD8: Wnt/β: catenin信号通路改善类风湿性关节炎的机制研究[D]. 合肥: 安徽中医药大学, 2023. |
| [13] | Peng Y, Huang Y, Li H, et al. Huangqin Qingre Chubi Capsule inhibits rheumatoid arthritis by regulating intestinal flora and improving intestinal barrier[J]. Front Pharmacol, 2024, 15: 1422245. doi:10.3389/fphar.2024.1422245 |
| [14] | Wang X, Chang J, Zhou G, et al. The traditional Chinese medicine compound Huangqin Qingre chubi capsule inhibits the pathogenesis of rheumatoid arthritis through the CUL4B/wnt pathway[J]. Front Pharmacol, 2021, 12: 750233. doi:10.3389/fphar.2021.750233 |
| [15] | Wang ST, Zeng XX, Gui P, et al. LncRNA EBLN3P facilitates osteosarcoma metastasis by enhancing annexin A3 mRNA stability and recruiting HuR[J]. Ann Surg Oncol, 2023, 30(13): 8690-703. doi:10.1245/s10434-023-14032-y |
| [16] | Sun MX, An HY, Sun YB, et al. LncRNA EBLN3P attributes methotrexate resistance in osteosarcoma cells through miR-200a-3p/O-GlcNAc transferase pathway[J]. J Orthop Surg Res, 2022, 17(1): 557. doi:10.1186/s13018-022-03449-y |
| [17] | Cieśla M, Darmochwał‑Kolarz D, Pałka A, et al. Expression of NEAT1, PACERR, and GAS5 circulating long noncoding RNAs can be associated with disease activity in rheumatoid arthritis patients[J]. Pol Arch Intern Med, 2024, 134(12): 16866. doi:10.20452/pamw.16866 |
| [18] | Wang Y, Hou L, Yuan X, et al. LncRNA NEAT1 targets fibroblast-like synoviocytes in rheumatoid arthritis via the miR-410-3p/YY1 axis[J]. Front Immunol, 2020, 11: 1975. doi:10.3389/fimmu.2020.01975 |
| [19] | Zhou Q, Liu J, Qi Y, et al. Jianpi Qingre Tongluo prescription alleviates the senescence-associated secretory phenotype with osteoarthritis by regulating STAG1/TP53/P21 signaling pathway[J]. J Ethnopharmacol, 2025, 337(pt 3): 118953. doi:10.1016/j.jep.2024.118953 |
| [20] | Yan Y, Zhang LB, Ma R, et al. Jolkinolide B ameliorates rheumatoid arthritis by regulating the JAK2/STAT3 signaling pathway[J]. Phytomedicine, 2024, 124: 155311. doi:10.1016/j.phymed.2023.155311 |
| [21] | Molendijk M, Hazes JM, Lubberts E. From patients with arthralgia, pre-RA and recently diagnosed RA: what is the current status of understanding RA pathogenesis[J]? RMD Open, 2018, 4(1): e000256. doi:10.1136/rmdopen-2016-000256 |
| [22] | Mor A, Abramson SB, Pillinger MH. The fibroblast-like synovial cell in rheumatoid arthritis: a key player in inflammation and joint destruction[J]. Clin Immunol, 2005, 115(2): 118-28. doi:10.1016/j.clim.2004.12.009 |
| [23] | Masoumi M, Bashiri H, Khorramdelazad H, et al. Destructive roles of fibroblast-like synoviocytes in chronic inflammation and joint damage in rheumatoid arthritis[J]. Inflammation, 2021, 44(2): 466-79. doi:10.1007/s10753-020-01371-1 |
| [24] | Giannini D, Antonucci M, Petrelli F, Bilia S, Alunno A, Puxeddu I. One year in review 2020: pathogenesis of rheumatoid arthritis. Clin Exp Rheumatol, 2020, 38(3): 387-97. doi:10.55563/clinexprheumatol/j1l5l3 |
| [25] | Chen ZF, Guo X, Wu SM, et al. Huayu Tongbi formula attenuates rheumatoid arthritis by inhibiting the HIF1A/VEGFA/ANGPT axis and suppressing angiogenesis[J]. Phytomedicine, 2025, 139: 156479. doi:10.1016/j.phymed.2025.156479 |
| [26] | Aihaiti Y, Song Cai Y, Tuerhong X, et al. Therapeutic effects of naringin in rheumatoid arthritis: network pharmacology and experimental validation[J]. Front Pharmacol, 2021, 12: 672054. doi:10.3389/fphar.2021.672054 |
| [27] | Baldini C, Moriconi FR, Galimberti S, Libby P, De Caterina R. The JAK-STAT pathway: an emerging target for cardiovascular disease in rheumatoid arthritis and myeloproliferative neoplasms. Eur Heart J. 2021,42(42):4389-4400. doi:10.1093/eurheartj/ehab447 |
| [28] | Bridges MC, Daulagala AC, Kourtidis A. LNCcation: lncRNA localization and function[J]. J Cell Biol, 2021, 220(2): e202009045. doi:10.1083/jcb.202009045 |
| [29] | Sun L, Yang K, Wang L, et al. LncRNA MIAT suppresses inflammation in LPS-induced J774A.1 macrophages by promoting autophagy through miR-30a-5p/SOCS1 axi[J]. Sci Rep, 2024, 14(1): 22608. doi:10.1038/s41598-024-73607-1 |
| [30] | Selim AM, Elsabagh YA, El-Sawalhi MM, et al. Serum lncRNA ITGB2-AS1 and ICAM-1 as novel biomarkers for rheumatoid arthritis and osteoarthritis diagnosis[J]. BMC Med Genom, 2024, 17(1): 247. doi:10.1186/s12920-024-01993-6 |
| [31] | Qiu H, Liu M, Shi X, et al. LncRNA HOTAIR inhibits the progression of fibroblast-like synoviocytes by sponging miRNA-106b-5p in rheumatoid arthritis[J]. Autoimmunity, 2022, 55(8): 567-76. doi:10.1080/08916934.2022.2126460 |
| [32] | Zhang Z, Zhang W, Wen QW, et al. Associations of genetic polymorphisms within MALAT1, UCA1, FAM211A-AS1 and AC000111.6 with genetic susceptibility to rheumatoid arthritis. Autoimmunity. 2020,53(7):408-414. doi:10.1080/08916934.2020.1818230 |
| [33] | Zhang M, Lu N, Guo XY, et al. Influences of the lncRNA TUG1-miRNA-34a-5p network on fibroblast-like synoviocytes (FLSs) dysfunction in rheumatoid arthritis through targeting the lactate dehydrogenase A (LDHA)[J]. J Clin Lab Anal, 2021, 35(9): e23969. doi:10.1002/jcla.23969 |
| [34] | Yang Z, Lin SD, Zhan F, et al. LncRNA GAS5 alleviates rheumatoid arthritis through regulating miR-222-3p/Sirt1 signalling axis[J]. Autoimmunity, 2021, 54(1): 13-22. doi:10.1080/08916934.2020.1846183 |
| [35] | Dai S, Li N, Zhou M, et al. LncRNA EBLN3P promotes the progression of osteosarcoma through modifying the miR-224-5p/Rab10 signaling axis[J]. Sci Rep, 2021, 11(1): 1992. doi:10.1038/s41598-021-81641-6 |
| [36] | Tang H, Huang H, Guo Z, et al. Heavy ion-responsive lncRNA EBLN3P functions in the radiosensitization of non-small cell lung cancer cells mediated by TNPO1[J]. Cancers: Basel, 2023, 15(2): 511. doi:10.3390/cancers15020511 |
| [37] | Xu XH, Song W, Li JH, et al. Long non-coding RNA EBLN3P regulates UHMK1 expression by sponging miR-323a-3p and promotes colorectal cancer progression[J]. Front Med: Lausanne, 2021, 8: 651600. doi:10.3389/fmed.2021.651600 |
| [38] | 陈瑞莲, 刘 健, 汪 元, 等. 黄芩清热除痹胶囊对湿热痹阻型类风湿关节炎患者临床疗效观察[J]. 中药药理与临床, 2025, 41(8): 78-81. |
| [39] | 徐昌萍, 曹云祥, 刘 健, 等. 黄芩清热除痹胶囊抑制TLR4/NF-κB信号通路对活动期类风湿关节炎的影响和机制[J]. 中华中医药杂志, 2025, 40(4): 2042-6. |
| [40] | 李 舒, 万 磊, 赵 磊, 等. 黄芩清热除痹胶囊治疗类风湿关节炎临床观察及对血清M1、M2型炎症因子影响[J]. 中药药理与临床, 2022, 38(2): 190-4. |
| [41] | SunMengyu, Yuan Wang, LIU FEIFEI, et al. LncRNA EBLN3P regulates the JAK/STAT signaling pathway through the miR-369-3p/NFIX axis: a potential therapeutic target for synovial revascul-arization in rheumatoid arthritis. Authorea. April 06, 2025. doi: 10.22541/au.174395356.60440021/v1 . |
| [1] | 马思源, 张博超, 浦春. Circ_0000437通过靶向let-7b-5p/CTPS1轴促进乳腺癌细胞的增殖、侵袭、迁移及上皮间质转化[J]. 南方医科大学学报, 2025, 45(8): 1682-1696. |
| [2] | 李玮怡, 江露, 张宗星, 陈丹, 包卓玛, 黄丽, 袁林. 强骨康疏方通过抑制HIF-1α/BNIP3自噬信号通路减少类风湿性关节炎大鼠的破骨细胞分化[J]. 南方医科大学学报, 2025, 45(7): 1389-1396. |
| [3] | 高志, 吴傲, 胡仲翔, 孙培养. 类风湿性关节炎中氧化应激与免疫浸润的生物信息学分析[J]. 南方医科大学学报, 2025, 45(4): 862-870. |
| [4] | 曹周芳, 汪元, 王梦娜, 孙玥, 刘菲菲. LINC00837/miR-671-5p/SERPINE2功能轴促进类风湿关节炎成纤维细胞样滑膜细胞的恶性病理学过程[J]. 南方医科大学学报, 2025, 45(2): 371-378. |
| [5] | 李亚辉, 杨欣, 姚血明, 黄聪. 豨莶丸治疗类风湿关节炎的分子机制:基于蛋白质组学[J]. 南方医科大学学报, 2025, 45(11): 2330-2339. |
| [6] | 徐买元, 李妮, 李嘉懿, 张涛, 马俐文, 林涛, 余浩楠, 吴宁, 吴遵秋, 黄丽. 葛根素通过调控TAK1介导的TLR4/NF-κB信号通路减轻大鼠类风湿关节炎症状[J]. 南方医科大学学报, 2025, 45(10): 2231-2239. |
| [7] | 展俊平, 黄硕, 孟庆良, 范围, 谷慧敏, 崔家康, 王慧莲. 缺氧微环境下补阳还五汤通过抑制BNIP3-PI3K/Akt通路抑制类风湿关节炎滑膜成纤维细胞的线粒体自噬[J]. 南方医科大学学报, 2025, 45(1): 35-42. |
| [8] | 杨锐, 舒翊秦, 文晖杰, 蔡熹, 王震, 张棽, 向阳, 吴昊. 枫杨总黄酮通过抑制中性粒细胞胞外陷阱网的释放减轻大鼠类风湿关节炎[J]. 南方医科大学学报, 2024, 44(9): 1645-1652. |
| [9] | 张先恒, 刘健, 韩琦, 陈一鸣, 丁香, 陈晓露. 黄芩清热除痹胶囊通过PTEN/PI3K/AKT信号通路改善痛风性关节炎大鼠的炎症反应及尿酸、脂质代谢失衡[J]. 南方医科大学学报, 2024, 44(8): 1450-1458. |
| [10] | 陈莉莉, 吴天宇, 张铭, 丁子夏, 张妍, 杨依清, 郑佳倩, 张小楠. 类风湿关节炎的潜在生物标志物及其免疫调控机制:基于GEO数据库[J]. 南方医科大学学报, 2024, 44(6): 1098-1108. |
| [11] | 王帆帆, 刘 健, 方妍妍, 文建庭, 贺明玉, 韩 琦, 李 旭. 中医药治疗可降低类风湿关节炎合并血小板升高患者再入院的风险:一项匹配队列研究[J]. 南方医科大学学报, 2023, 43(9): 1548-1557. |
| [12] | 宗世烨, 周 静, 蔡伟伟, 余 芸, 王 颖, 宋宜宁, 程静文, 李宇会, 高 艺, 吴百海, 咸 郝, 魏 芳. 小檗碱抑制类风湿关节炎患者的成纤维样滑膜细胞的自噬并促进其凋亡:基于下调ROS/mTOR信号通路[J]. 南方医科大学学报, 2023, 43(4): 552-559. |
| [13] | 刘天阳, 周学平, 黄传兵, 周玲玲, 谌 曦, 万 磊, 纵瑞凯, 范海霞, 孙 玥, 俞志超, 汤忠富, 徐耿瑞, 周子译. 清络通痹方调控类风湿关节炎“免疫-骨侵蚀”的机制[J]. 南方医科大学学报, 2023, 43(10): 1706-1714. |
| [14] | 唐 娟, 陈 娟, 林国新, 张 浩, 桂 明, 李楠楠, 谷依红, 罗林娟, 孙 剑. 中性粒细胞/淋巴细胞、血小板/淋巴细胞比值是评价托法替尼治疗后类风湿关节炎活动性的有效指标[J]. 南方医科大学学报, 2023, 43(10): 1651-1656. |
| [15] | 周思聪, 杨 威, 曾 丽, 曹春浩, 袁 速, 荣晓凤. 大黄素对胶原诱导性关节炎大鼠的骨保护作用:基于抑制铁死亡和降解基质金属蛋白酶[J]. 南方医科大学学报, 2023, 43(10): 1776-1781. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||