南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 403-411.doi: 10.12122/j.issn.1673-4254.2026.02.18
• • 上一篇
尹林1,2( ), 张可妮1, 乔通1, 牛民主2, 殷丽霞1, 刘馨悦3, 耿志军3, 李静1, 胡建国1(
), 张可妮1, 乔通1, 牛民主2, 殷丽霞1, 刘馨悦3, 耿志军3, 李静1, 胡建国1( )
)
收稿日期:2025-06-05
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
胡建国
E-mail:yljykbbmu@163.com;jghu9200@bbmu.edu.cn
作者简介:尹 林,在读硕士研究生,E-mail: yljykbbmu@163.com
基金资助:
Lin YIN1,2( ), Keni ZHANG1, Tong QIAO1, Minzhu NIU2, Lixia YIN1, Xinyue LIU3, Zhijun GENG3, Jing LI1, Jianguo HU1(
), Keni ZHANG1, Tong QIAO1, Minzhu NIU2, Lixia YIN1, Xinyue LIU3, Zhijun GENG3, Jing LI1, Jianguo HU1( )
)
Received:2025-06-05
Online:2026-02-20
Published:2026-03-10
Contact:
Jianguo HU
E-mail:yljykbbmu@163.com;jghu9200@bbmu.edu.cn
摘要:
目的 探讨藜芦酸(VA)通过调控Nrf2/HO-1信号通路改善葡聚糖硫酸钠(DSS)诱导的结肠炎氧化应激损伤及肠屏障功能障碍的分子机制。 方法 将30只雄性C57BL/6小鼠随机分为对照组(WT组)、DSS模型组及VA治疗组,10只/组。通过评估体质量变化、疾病活动指数(DAI)、结肠长度及组织病理学损伤。ELISA检测结肠组织中炎症因子(TNF-α、IL-6、IL-10)水平,以及分析氧化应激标志物(SOD、GSH、MDA、COX-2)的表达。免疫荧光及Western blotting检测紧密连接蛋白(ZO-1、Claudin-1)及Nrf2/HO-1通路相关蛋白的表达。体外构建H₂O₂(150 μmol/L)诱导的Caco-2细胞氧化应激模型,流式细胞术及DCFH-DA荧光探针检测活性氧(ROS)蓄积,并联合Nrf2抑制剂ML385验证VA的调控机制。 结果 VA干预缓解DSS小鼠体质量下降(P<0.05)、结肠缩短(P<0.05)及DAI评分升高(P<0.05),以及修复肠黏膜隐窝结构、紧密连接蛋白(ZO-1和Claudin-1)表达(P<0.05)和增加杯状细胞数量(P<0.05)。VA降低结肠组织中促炎因子TNF-α、IL-6水平(P<0.05),同时上调抗炎因子IL-10表达水平(P<0.05)。VA逆转DSS诱导的SOD和GSH活性降低,以及MDA和COX-2水平升高(P<0.05)。在H₂O₂诱导的Caco-2细胞模型中,流式细胞术和免疫荧光染色结果提示:VA降低ROS阳性细胞率和细胞内ROS蓄积;免疫荧光染色和Western blotting数据分析显示:VA增加了Claudin-1和ZO-1蛋白的表达。机制上,VA促进Nrf2及下游HO-1蛋白表达(P<0.05),以及Nrf2抑制剂ML385部分拮抗了VA对H₂O₂诱导的ROS阳性细胞比例的下降。 结论 VA通过激活Nrf2/HO-1通路增强抗氧化防御、抑制炎症反应并修复肠屏障功能,为IBD治疗提供新策略。
尹林, 张可妮, 乔通, 牛民主, 殷丽霞, 刘馨悦, 耿志军, 李静, 胡建国. 藜芦酸通过激活Nrf2/HO-1信号通路减轻氧化应激缓解葡聚糖硫酸钠诱导的小鼠实验性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 403-411.
Lin YIN, Keni ZHANG, Tong QIAO, Minzhu NIU, Lixia YIN, Xinyue LIU, Zhijun GENG, Jing LI, Jianguo HU. Veratric acid relieves oxidative stress and DSS-induced colitis in mice by activating the Nrf2/HO-1 signaling pathway[J]. Journal of Southern Medical University, 2026, 46(2): 403-411.
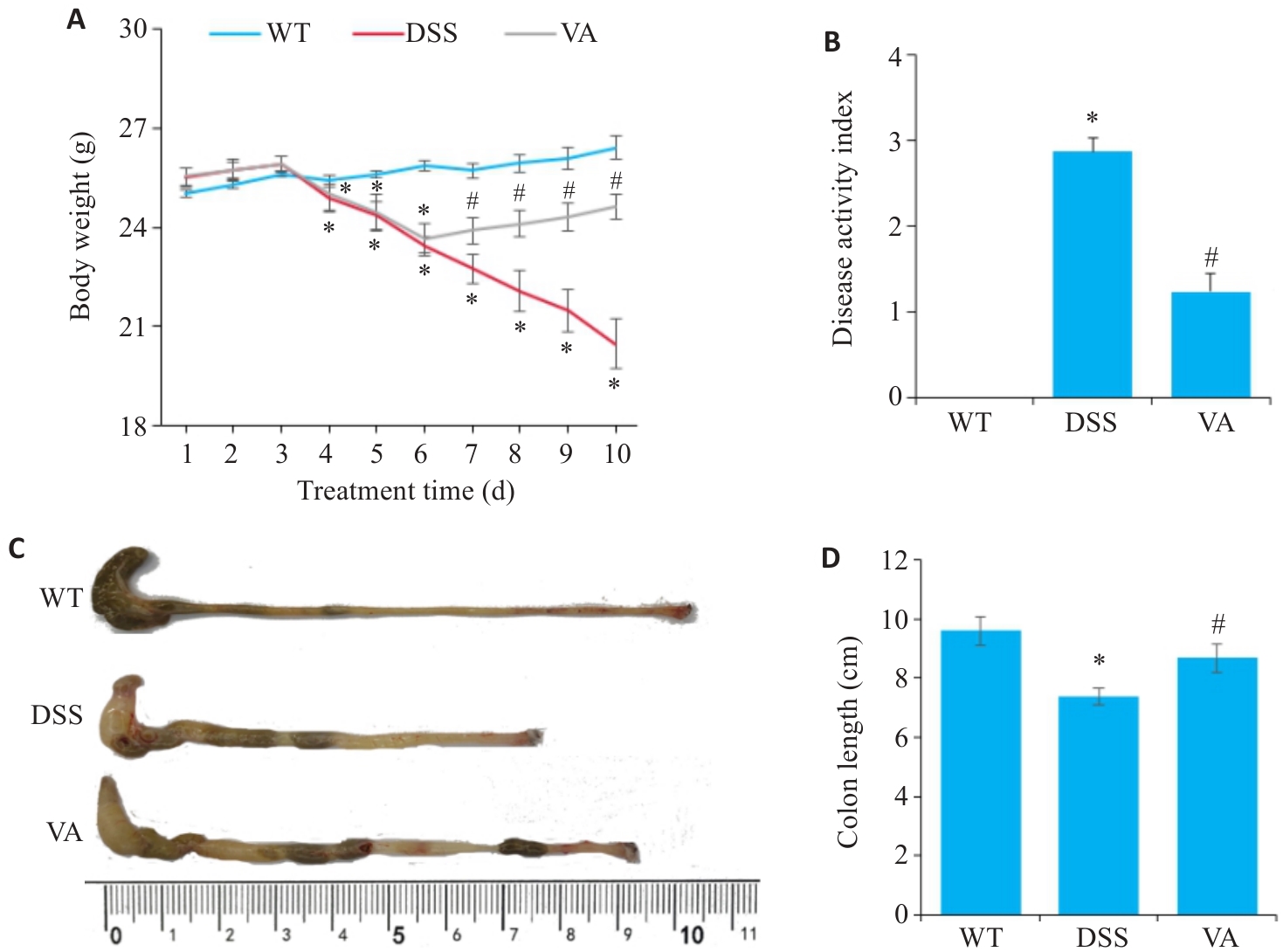
图1 VA对DSS诱导的小鼠肠炎症状的影响
Fig.1 Effect of VA on DSS-induced colitis symptoms in mice (n=10). A: Changes of body weight. B: Changes of DAI scores. C, D: Comparison of colon length among different groups. *P<0.05 vs WT group; #P<0.05 vs DSS group.
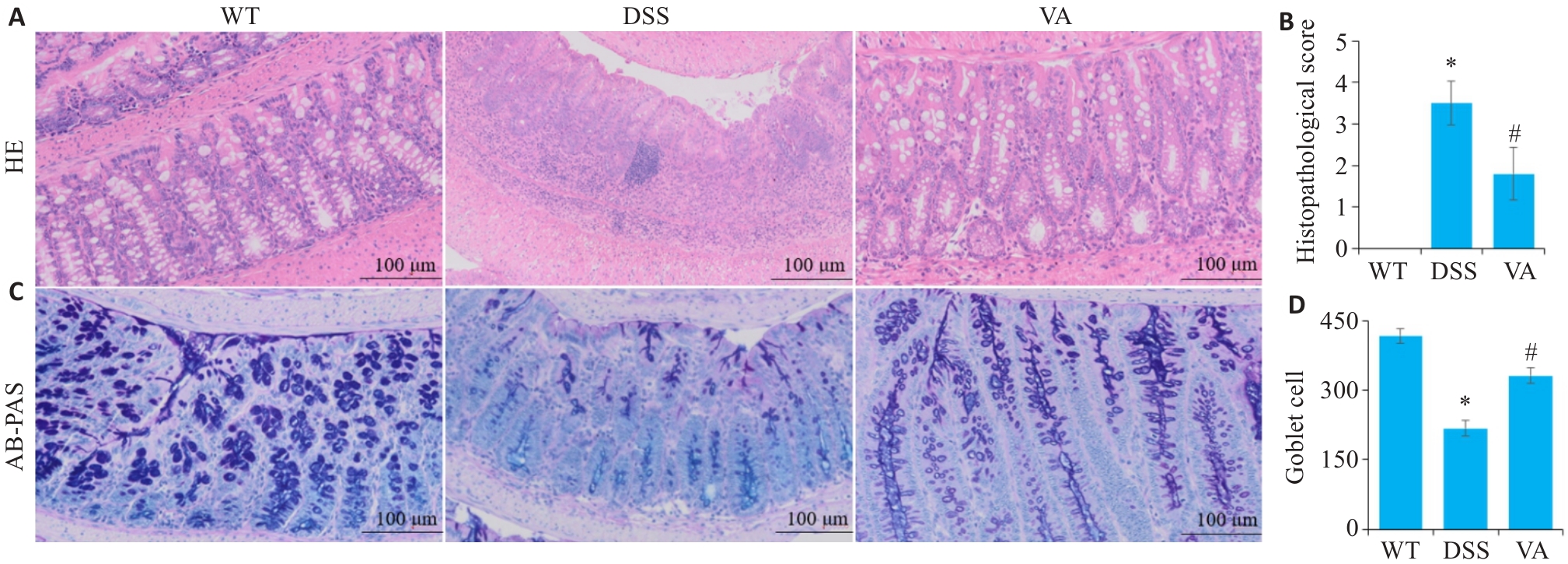
图2 VA对DSS小鼠肠组织病理学的影响
Fig.2 Effect of VA on intestinal histopathology in DSS-treated mice (n=10). A: HE staining and inflammatory scores of the intestinal tissues of mice in each group. B: Histopathological scores of mice in each group. C: AB-PAS staining of the intestinal tissues in each group. D: Comparison of goblet cell counts in each group. *P<0.05 vs WT group; #P<0.05 vs DSS group.
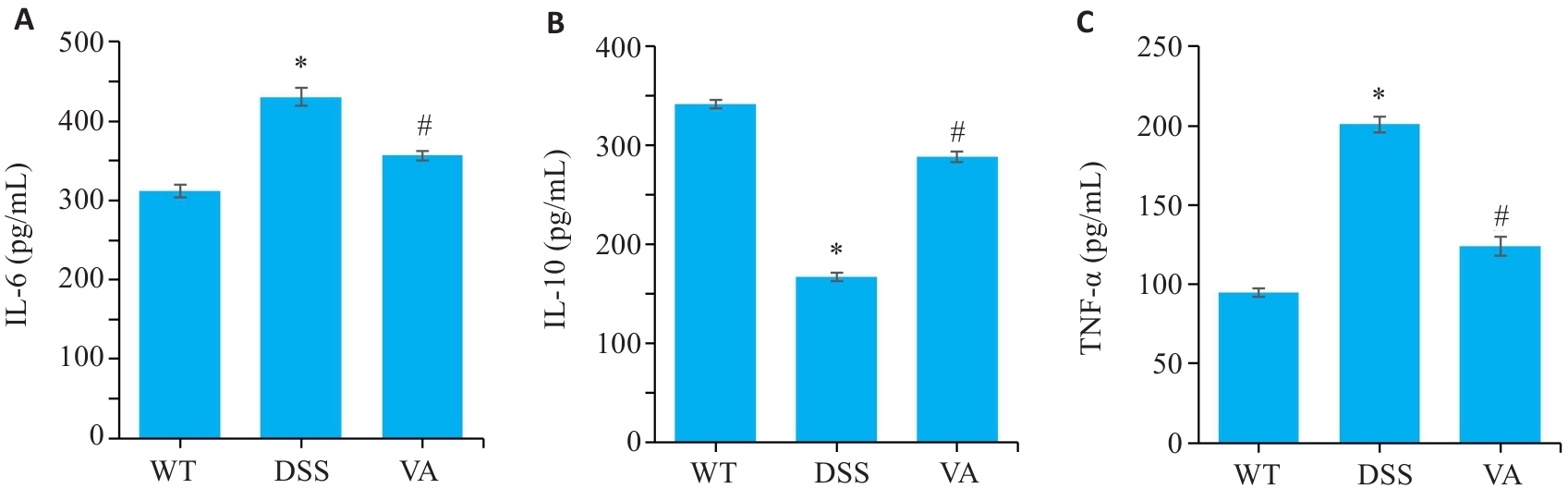
图3 VA对DSS小鼠结肠组织中炎症因子表达的影响
Fig.3 Effect of VA on expressions of inflammatory cytokines in the colonic tissues of DSS-treated mice (n=10). A: Levels of interleukin-6 (IL-6) in the colonic mucosa of the mice in each group detected by enzyme-linked immunosorbent assay (ELISA). B: Levels of interleukin-10 (IL-10) in the colonic mucosa of the mice in each group detected by ELISA. C: Levels of tumor necrosis factor-α (TNF-α) in the colonic mucosa of the mice in each group detected by ELISA. *P<0.05 vs WT group; #P<0.05 vs DSS group.
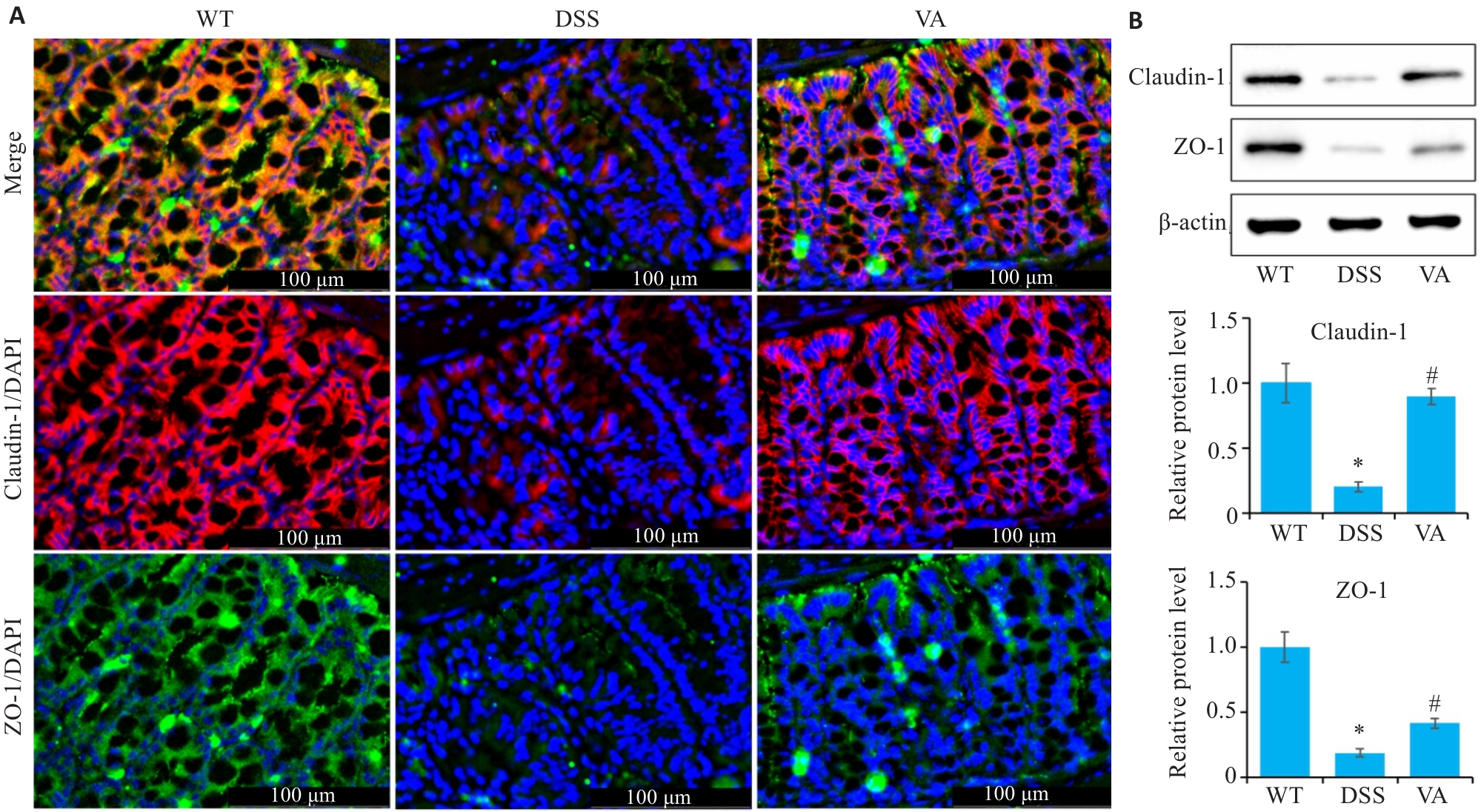
图4 VA对DSS小鼠结肠组织中紧密连接蛋白的影响
Fig.4 Effect of VA on tight junction proteins in the colonic tissues of DSS-induced mice (n=10). A: Immunofluorescence staining for claudin-1 and ZO-1 in the colons of the mice in each group. B: Western blotting for detecting expression levels of claudin-1 and ZO-1 in the colons of the mice in each group. *P<0.05 vs WT group; #P<0.05 vs DSS group.
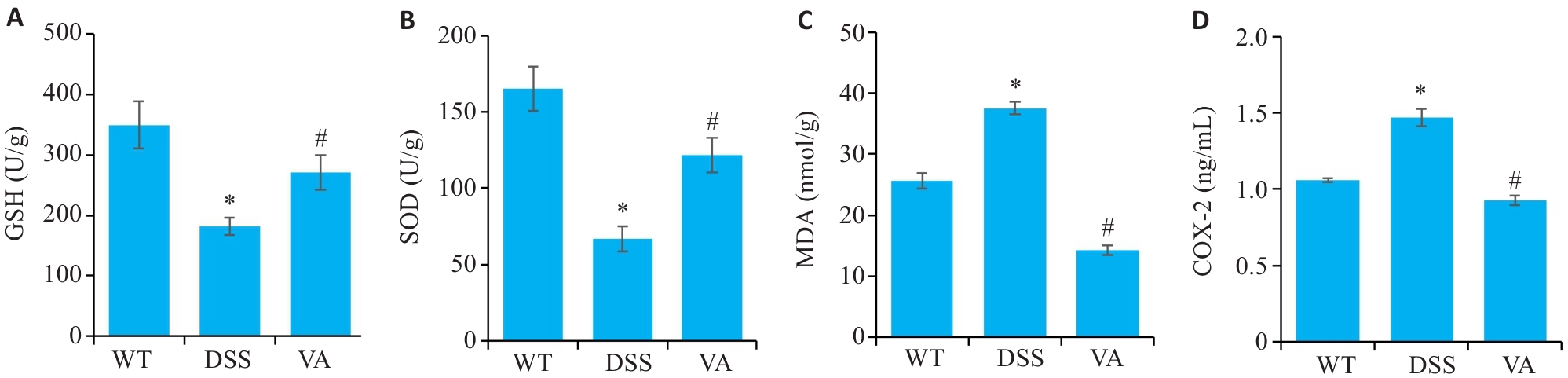
图5 VA对DSS小鼠结肠组织中氧化应激的调控作用
Fig.5 Regulatory effect of VA on oxidative stress in the colonic tissues of DSS-induced mice (n=10). A: Detection results of glutathione (GSH) in each group. B: levels of superoxide dismutase (SOD) in each group. C: Malondialdehyde (MDA) levels in each group. D: Cyclooxygenase-2 (COX-2) levels in each group. *P<0.05 vs WT group; #P<0.05 vs DSS group.
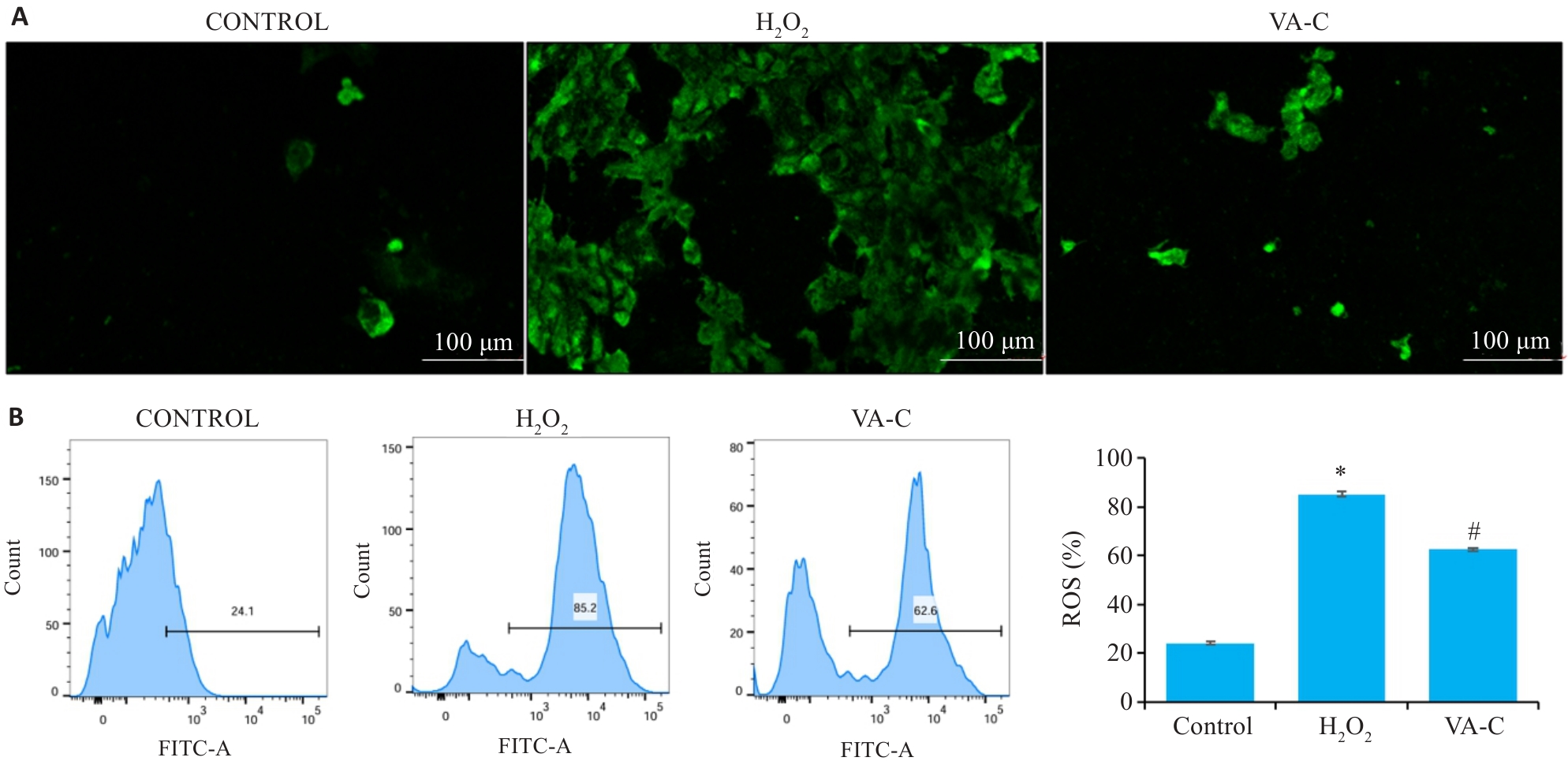
图6 VA对H₂O₂诱导的Caco-2细胞活性氧的影响
Fig.6 Effect of VA on reactive oxygen species (ROS) production induced by H₂O₂ in Caco-2 cells (n=10). A: ROS staining with the fluorescent probe (DCFH-DA) in each group. B: ROS detection by flow cytometry in each group. *P<0.05 vs Control group. #P<0.05 vs H₂O₂ group.
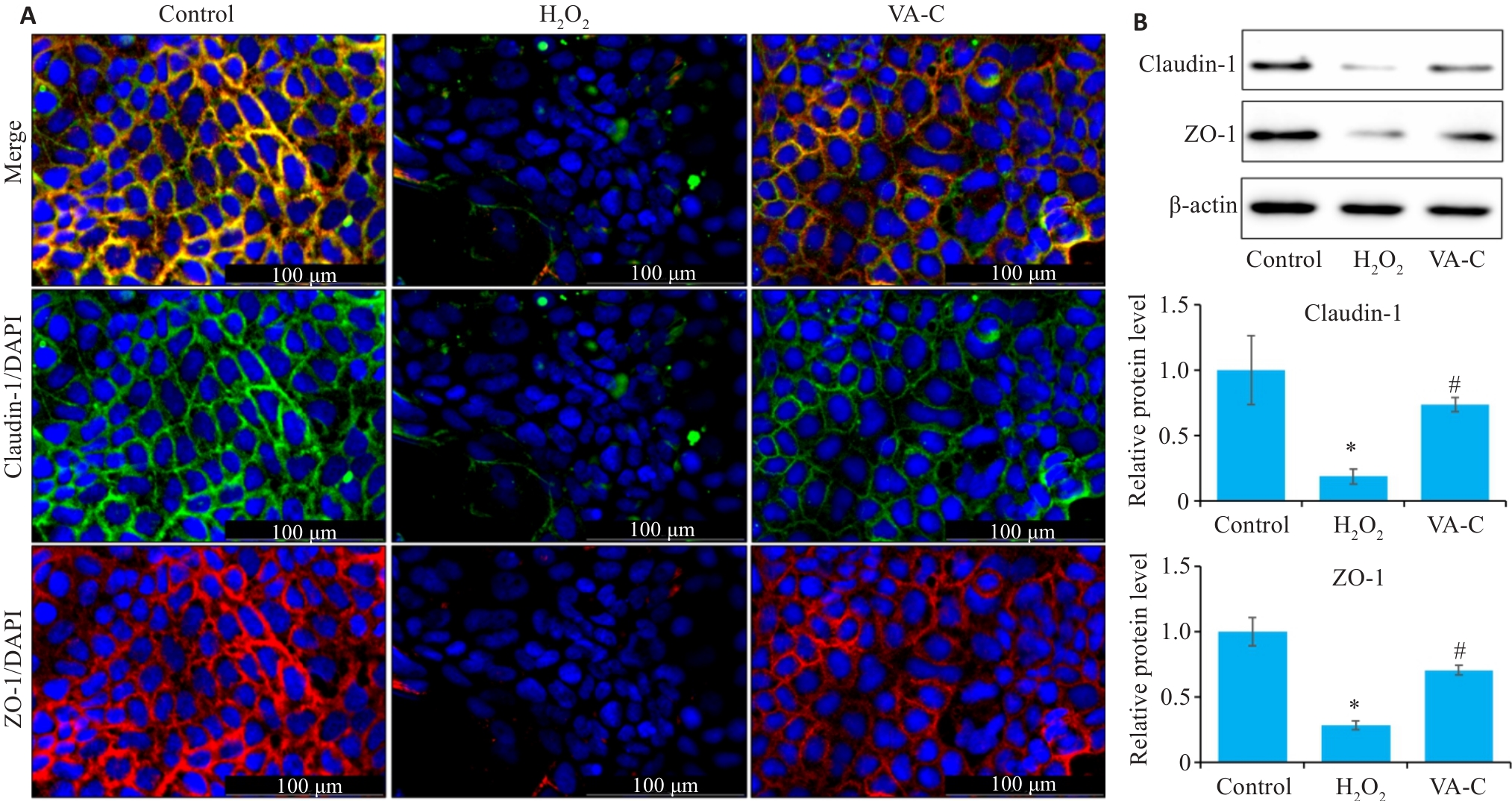
图7 VA 对H₂O₂诱导的Caco-2细胞肠屏障的影响
Fig.7 Effect of VA on intestinal barrier function of H₂O₂-induced Caco-2 cells (n=10). A: Immunofluorescence staining for claudin-1 and ZO-1 in each group. B: Western blotting for detecting expression levels of claudin-1 and ZO-1 in each group. *P<0.05 vs Control group; #P<0.05 vs H₂O₂ group.
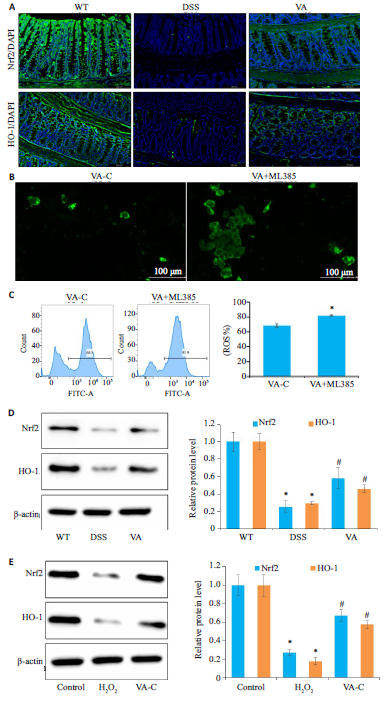
图8 VA 通过激活Nrf2/HO-1通路改善结肠炎小鼠肠道氧化应激损伤
Fig.8 VA alleviates intestinal oxidativestress in colitis mice by activating the Nrf2/HO-1 pathway. A: Immunofluorescencestaining of Nrf2/HO-1 in the colons of themice in each group. B: Intracellular ROSdetection using the fluorescent probe(DCFH-DA) in VA treatment group andML385 inhibitor group. C: IntracellularROS detection by flow cytometry in VAtreatment group and ML385 inhibitorgroup. D: Western blotting for detectingcolonic expression levels of Nrf2/HO-1 ofthe mice in each group. E: Western blottingfor detecting esxpression levels of Nrf2/HO-1 in Caco-2 cells in each group. *P<0.05vs (WT/VA-C/control) group. #PP<0.05 vs(DSS/H₂O₂) group.
| [1] | Bisgaard TH, Allin KH, Keefer L, et al. Depression and anxiety in inflammatory bowel disease: epidemiology, mechanisms and treatment[J]. Nat Rev Gastroenterol Hepatol, 2022, 19(11): 717-26. doi:10.1038/s41575-022-00634-6 |
| [2] | Xu S, Li XZ, Zhang SH, et al. Oxidative stress gene expression, DNA methylation, and gut microbiota interaction trigger Crohn's disease: a multi-omics Mendelian randomization study[J]. BMC Med, 2023, 21(1): 179. doi:10.1186/s12916-023-02878-8 |
| [3] | Sahoo DK, Heilmann RM, Paital B, et al. Oxidative stress, hormones, and effects of natural antioxidants on intestinal inflammation in inflammatory bowel disease[J]. Front Endocrinol (Lausanne), 2023, 14: 1217165. doi:10.3389/fendo.2023.1217165 |
| [4] | Wang S, Dong ZX, Wan XJ. Global, regional, and national burden of inflammatory bowel disease and its associated Anemia, 1990 to 2019 and predictions to 2050: an analysis of the global burden of disease study 2019[J]. Autoimmun Rev, 2024, 23(3): 103498. doi:10.1016/j.autrev.2023.103498 |
| [5] | Chen YY, Cui WW, Li X, et al. Interaction between commensal bacteria, immune response and the intestinal barrier in inflammatory bowel disease[J]. Front Immunol, 2021, 12: 761981. doi:10.3389/fimmu.2021.761981 |
| [6] | Hsu NY, Nayar S, Gettler K, et al. NOX1 is essential for TNFα-induced intestinal epithelial ROS secretion and inhibits M cell signatures[J]. Gut, 2023, 72(4): 654-62. doi:10.1136/gutjnl-2021-326305 |
| [7] | Zheng MY, Han R, Yuan YL, et al. The role of Akkermansia muciniphila in inflammatory bowel disease: Current knowledge and perspectives[J]. Front Immunol, 2023, 13: 1089600. doi:10.3389/fimmu.2022.1089600 |
| [8] | Yu QW, Chen SY, Tang HW, et al. Veratric acid alleviates liver ischemia/reperfusion injury by activating the Nrf2 signaling pathway[J]. Int Immunopharmacol, 2021, 101(Pt B): 108294. doi:10.1016/j.intimp.2021.108294 |
| [9] | Saravanakumar M, Raja B. Veratric acid, a phenolic acid attenuates blood pressure and oxidative stress in L-NAME induced hypertensive rats[J]. Eur J Pharmacol, 2011, 671(1/2/3): 87-94. doi:10.1016/j.ejphar.2011.08.052 |
| [10] | Bian WC, Wei LL, Wang KX. Carthamin yellow alleviates dextran sodium sulfate-induced ulcerative colitis by repairing the intestinal barrier and activating the Nrf2/GPX4 axis[J]. Int Immuno-pharmacol, 2024, 141: 113020. doi:10.1016/j.intimp.2024.113020 |
| [11] | Huang LY, Qian WW, Xu YH, et al. Mesenteric adipose tissue contributes to intestinal fibrosis in Crohn’s disease through the ATX-LPA axis[J]. J Crohns Colitis, 2022, 16(7): 1124-39. doi:10.1093/ecco-jcc/jjac017 |
| [12] | Schultz M, Tonkonogy SL, Sellon RK, et al. IL-2-deficient mice raised under germfree conditions develop delayed mild focal intestinal inflammation[J]. Am J Physiol, 1999, 276(6): G1461-72. doi:10.1152/ajpgi.1999.276.6.g1461 |
| [13] | Biasi F, Leonarduzzi G, Oteiza PI, et al. Inflammatory bowel disease: mechanisms, redox considerations, and therapeutic targets[J]. Antioxid Redox Signal, 2013, 19(14): 1711-47. doi:10.1089/ars.2012.4530 |
| [14] | Farzaei MH, Bahramsoltani R, Abdolghaffari AH, et al. A mechanistic review on plant-derived natural compounds as dietary supplements for prevention of inflammatory bowel disease[J]. Expert Rev Gastroenterol Hepatol, 2016, 10(6): 745-58. doi:10.1586/17474124.2016.1145546 |
| [15] | Wang QB, Sun LY, Gong ZD, et al. Veratric acid inhibits LPS-induced IL-6 and IL-8 production in human gingival fibroblasts[J]. Inflammation, 2016, 39(1): 237-42. doi:10.1007/s10753-015-0243-9 |
| [16] | Guo XX, Xu Y, Geng RX, et al. Curcumin alleviates dextran sulfate sodium-induced colitis in mice through regulating gut microbiota[J]. Mol Nutr Food Res, 2022, 66(8): e2100943. doi:10.1002/mnfr.202100943 |
| [17] | Jayakumar T, Huang CJ, Yen TL, et al. Activation of Nrf2 by esculetin mitigates inflammatory responses through suppression of NF-κB signaling cascade in RAW 264.7 cells[J]. Molecules, 2022, 27(16): 5143. doi:10.3390/molecules27165143 |
| [18] | Gao W, Guo L, Yang Y, et al. Dissecting the crosstalk between Nrf2 and NF-κB response pathways in drug-induced toxicity[J]. Front Cell Dev Biol, 2022, 9: 809952. doi:10.3389/fcell.2021.809952 |
| [19] | Dong LJ, Xie JW, Wang YY, et al. Mannose ameliorates experimental colitis by protecting intestinal barrier integrity[J]. Nat Commun, 2022, 13(1): 4804. doi:10.1038/s41467-022-32505-8 |
| [20] | Kinchen J, Chen HH, Parikh K, et al. Structural remodeling of the human colonic mesenchyme in inflammatory bowel disease[J]. Cell, 2018, 175(2): 372-86.e17. doi:10.1016/j.cell.2018.08.067 |
| [21] | Neurath MF, Artis D, Becker C. The intestinal barrier: a pivotal role in health, inflammation, and cancer[J]. Lancet Gastroenterol Hepatol, 2025, 10(6): 573-92. doi:10.1016/s2468-1253(24)00390-x |
| [22] | Li MQ, Lv RZ, Wang CZ, et al. Tricholoma matsutake-derived peptide WFNNAGP protects against DSS-induced colitis by ameliorating oxidative stress and intestinal barrier dysfunction[J]. Food Funct, 2021, 12(23): 11883-97. doi:10.1039/d1fo02806e |
| [23] | Almousa AA, Meurens F, Krol ES, et al. Linoorbitides and enterolactone mitigate inflammation-induced oxidative stress and loss of intestinal epithelial barrier integrity[J]. Int Immuno-pharmacol, 2018, 64: 42-51. doi:10.1016/j.intimp.2018.08.012 |
| [24] | Yang XW, Mao ZM, Huang YR, et al. Reductively modified albumin attenuates DSS-Induced mouse colitis through rebalancing systemic redox state[J]. Redox Biol, 2021, 41: 101881. doi:10.1016/j.redox.2021.101881 |
| [25] | Gustafsson JK, Johansson MEV. The role of goblet cells and mucus in intestinal homeostasis[J]. Nat Rev Gastroenterol Hepatol, 2022, 19(12): 785-803. doi:10.1038/s41575-022-00675-x |
| [26] | Kim YS, Ho SB. Intestinal goblet cells and mucins in health and disease: recent insights and progress[J]. Curr Gastroenterol Rep, 2010, 12(5): 319-30. doi:10.1007/s11894-010-0131-2 |
| [27] | Zhang MM, Wu CC. The relationship between intestinal goblet cells and the immune response[J]. Biosci Rep, 2020, 40(10): BSR20201471. doi:10.1042/bsr20201471 |
| [28] | Yang X, Li GL, Lou PB, et al. Excessive nucleic acid R-loops induce mitochondria-dependent epithelial cell necroptosis and drive spontaneous intestinal inflammation[J]. Proc Natl Acad Sci USA, 2024, 121(1): e2307395120. doi:10.1073/pnas.2307395120 |
| [29] | Arda-Pirincci P, Aykol-Celik G. Galectin-1 reduces the severity of dextran sulfate sodium (DSS)-induced ulcerative colitis by suppressing inflammatory and oxidative stress response[J]. Bosn J Basic Med Sci, 2020, 20(3): 319-28. |
| [30] | Pan TL, Liu SQ, Liao QC, et al. Dietary supplement of veratric acid alleviates liver steatosis and reduces abdominal fat deposition in broilers[J]. Poult Sci, 2024, 103(12): 104406. doi:10.1016/j.psj.2024.104406 |
| [31] | Yan B, Mao XJ, Hu SS, et al. Spermidine protects intestinal mucosal barrier function in mice colitis via the AhR/Nrf2 and AhR/STAT3 signaling pathways[J]. Int Immunopharmacol, 2023, 119: 110166. doi:10.1016/j.intimp.2023.110166 |
| [32] | Ni J, Wu GD, Albenberg L, et al. Gut microbiota and IBD: causation or correlation[J]? Nat Rev Gastroenterol Hepatol, 2017, 14(10): 573-84. doi:10.1038/nrgastro.2017.88 |
| [33] | Qiu P, Ishimoto T, Fu LF, et al. The gut microbiota in inflammatory bowel disease[J]. Front Cell Infect Microbiol, 2022, 12: 733992. doi:10.3389/fcimb.2022.733992 |
| [1] | 彭煜策, 姜毅, 马丹, 何岸, 吕鼎一, 罗明昊, 罗素新. 异牡荆素通过促进SIRT3表达减轻糖尿病小鼠的心肌氧化应激损伤[J]. 南方医科大学学报, 2026, 46(2): 316-324. |
| [2] | 王静娴, 任自敬, 周佩洋. S1PR5激动与过表达通过调控氧化应激增强脑微血管内皮细胞屏障功能抵抗氧糖剥夺/复氧复糖损伤[J]. 南方医科大学学报, 2025, 45(7): 1451-1459. |
| [3] | 牛民主, 殷丽霞, 乔通, 尹林, 张可妮, 胡建国, 宋传旺, 耿志军, 李静. 旱莲苷A通过调控JAK2/STAT3通路抑制M1型巨噬细胞极化改善葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2025, 45(6): 1297-1306. |
| [4] | 张安邦, 孙秀颀, 庞博, 吴远华, 时靖宇, 张宁, 叶涛. 电针预处理通过调节肠道-大脑轴及Nrf2/HO-1信号通路抑制铁死亡减轻大鼠脑缺血再灌注损伤[J]. 南方医科大学学报, 2025, 45(5): 911-920. |
| [5] | 储菲, 陈孝华, 宋博文, 杨晶晶, 左芦根. 苏荠宁黄酮通过抑制PI3K/AKT信号通路拮抗肠上皮细胞凋亡改善小鼠实验性结肠炎[J]. 南方医科大学学报, 2025, 45(4): 819-828. |
| [6] | 高志, 吴傲, 胡仲翔, 孙培养. 类风湿性关节炎中氧化应激与免疫浸润的生物信息学分析[J]. 南方医科大学学报, 2025, 45(4): 862-870. |
| [7] | 黄鹏伟, 陈洁, 邹金虎, 高雪锋, 曹虹. 槲皮素促进应激颗粒G3BP1解聚改善HIV-1 gp120诱导的星形胶质细胞神经毒性[J]. 南方医科大学学报, 2025, 45(2): 304-312. |
| [8] | 卢晓宇, 刘智慧, 刘烨, 庞天霄, 卞蓉, 郭玲, 何学红. 参芪泄浊饮通过调控Rap1/MAPK/FoxO3a信号通路改善氧化应激及炎症反应延缓大鼠肾纤维化[J]. 南方医科大学学报, 2025, 45(12): 2585-2597. |
| [9] | 陶露, 陈悦, 黄林林, 郑旺, 宋雪, 项平, 胡建国. 珠子草素通过调控p38/JNK信号通路抑制肠上皮细胞凋亡保护肠屏障改善克罗恩病样肠炎[J]. 南方医科大学学报, 2025, 45(11): 2483-2495. |
| [10] | 林淑娴, 郭丽娜, 马燕, 熊尧, 何盈犀, 许欣筑, 盛雯, 许素哗, 邱峰. 植物乳植杆菌ZG03通过其代谢物短链脂肪酸缓解斑马鱼的氧化应激[J]. 南方医科大学学报, 2025, 45(10): 2223-2230. |
| [11] | 张钰明, 夏士程, 张淋淋, 陈梦茜, 刘晓婧, 高琴, 叶红伟. 金银花提取物对小鼠阿霉素肝脏损伤的保护作用[J]. 南方医科大学学报, 2024, 44(8): 1571-1581. |
| [12] | 任志军, 刁建新, 王奕婷. 芎归汤通过抑制氧化应激诱导的心肌凋亡减轻小鼠心梗后心衰引起的心肌损伤[J]. 南方医科大学学报, 2024, 44(7): 1416-1424. |
| [13] | 梁国新, 唐红悦, 郭畅, 张明明. miR-224-5p调控PI3K/Akt/FoxO1轴抑制氧化应激减轻缺氧/复氧诱导的心肌细胞损伤[J]. 南方医科大学学报, 2024, 44(6): 1173-1181. |
| [14] | 陈国栋, 罗素新. 秋水仙碱通过激活AMPK减轻小鼠心肌缺血再灌注损伤[J]. 南方医科大学学报, 2024, 44(2): 226-235. |
| [15] | 凌旭光, 徐雯雯, 庞观来, 洪旭星, 刘凤芹, 李 洋. 茶多酚通过抑制NLRP3炎症小体改善脓毒症小鼠的急性肺损伤[J]. 南方医科大学学报, 2024, 44(2): 381-386. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||