南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (6): 1226-1239.doi: 10.12122/j.issn.1673-4254.2025.06.12
侯鑫睿1,2( ), 张振东1,2, 曹明远1,2, 杜予心1,2, 王小平1(
), 张振东1,2, 曹明远1,2, 杜予心1,2, 王小平1( )
)
收稿日期:2025-03-21
出版日期:2025-06-20
发布日期:2025-06-27
通讯作者:
王小平
E-mail:362380328@qq.com;xpwang@xzmz.edu.cn
作者简介:侯鑫睿,在读硕士研究生,E-mail: 362380328@qq.com
基金资助:
Xinrui HOU1,2( ), Zhendong ZHANG1,2, Mingyuan CAO1,2, Yuxin DU1,2, Xiaoping WANG1(
), Zhendong ZHANG1,2, Mingyuan CAO1,2, Yuxin DU1,2, Xiaoping WANG1( )
)
Received:2025-03-21
Online:2025-06-20
Published:2025-06-27
Contact:
Xiaoping WANG
E-mail:362380328@qq.com;xpwang@xzmz.edu.cn
Supported by:摘要:
目的 探讨红景天苷抗胃肿瘤(GC)机制,强调糖代谢重编程途径。 方法 高通量测序结合生物信息学分析预测红景天苷作用后显著差异表达的miRNA-靶mRNA,mRNA-mRNA相互作用关系,RNA结合蛋白免疫共沉淀实验验证miRNA-mRNA互作,蛋白质免疫共沉淀(Co-IP)实验验证下游靶mRNA-mRNA互作。通过体外及体内实验探究红景天苷与miR-1343-3p对GC的作用机制。体外实验通过CCK-8法确定红景天苷的IC50值并分低、中、高剂量处理细胞,结合克隆实验、RT-qPCR、Western blotting、ELISA及ATP检测评估药物对增殖、基因/蛋白/下游分子表达(miRNA、mRNA、丙酮酸、乙酰CoA)及能量代谢的影响;进一步以脂质体法转染miR-1343-3p模拟物(miR-1343-3p mimic)、抑制剂(miR-1343-3p inhibitor)、靶向OGDHL的siRNA(si-OGDHL)及其阴性对照(NC mimic/NC inhibitor/si-NC)至GC细胞,联合红景天苷处理,通过相同方法验证其协同作用。体内实验通过构建荷瘤裸鼠模型,给予红景天苷和miR-1343-3p激动剂(miR-1343-3p agomir)干预,分析瘤块体积/质量变化及靶基因、蛋白表达与代谢状态。实验分组包括空白对照、单药组及联合组,系统解析红景天苷与miRNA的调控网络。 结果 生物信息学证实,miR-1343-3p与三羧酸(TCA)循环关键酶α-酮戊二酸脱氢酶复合体亚基(OGDHL)呈负相关(P<0.01),且直接相互作用。OGDHL与丙酮酸脱氢酶E1亚基-β(PDHB)在胃癌组织中同步高表达,且二者存在相互作用。体外与体内实验证实,红景天苷以剂量/时间依赖性抑制GC细胞增殖,红景天苷作用GC细胞后,重要抑癌因子miR-1343-3p升高,受其调控的OGDHL的基因表达明显下调,导致与之互作的糖酵解酶蛋白PDHB稳态失衡,丙酮酸氧化脱羧减少,产物乙酰CoA和ATP生成明显减少(P<0.05)。 结论 红景天苷可能通过调控miR-1343-3p-OGDHL/PDHB酶复合体-丙酮酸糖代谢通路抑制GC细胞的增殖,为红景天苷抗肿瘤机制提供了新的见解,为开发基于代谢酶复合体协同调控癌症的精准疗法提供理论依据。
侯鑫睿, 张振东, 曹明远, 杜予心, 王小平. 红景天苷靶向miR-1343-3p-OGDHL/PDHB糖代谢轴抑制胃癌细胞的体内外增殖[J]. 南方医科大学学报, 2025, 45(6): 1226-1239.
Xinrui HOU, Zhendong ZHANG, Mingyuan CAO, Yuxin DU, Xiaoping WANG. Salidroside inhibits proliferation of gastric cancer cells by regulating the miR-1343-3p-OGDHL/PDHB glucose metabolic axis[J]. Journal of Southern Medical University, 2025, 45(6): 1226-1239.
| Primer name | Direction | Sequence (5'→3') |
|---|---|---|
| GAPDH | Forward | TGACATCAAGAAGGTGGTGAAGCAG |
| Reverse | GTGTCGCTGTTGAAGTCAGAGGAG | |
| U6 | Forward | GCTTCGGCAGCACATATACTAAAAT |
| Reverse | CGCTTCACGAATTTGCGTGTCAT | |
| miR-1343-3p | Forward | CGCGGCCTTAATGCTAATTGTGA |
| Reverse | AGTGCAGGGTCCGAGGTATT | |
| OGDHL | Forward | ATCACTCTGTCGCTGGTTGCC |
| Reverse | CGCTCAGGTGGAAGGTCTCATATAC | |
| PDHB | Forward | TTCTCCATGCAAGCCATTGACCAG |
| Reverse | CTACACCTGCTGAGGCACCATTG | |
| miR-1343-3p mimic | Sense | CUCCUGGGGCCCGCACUCUCGCUU |
| Antisense | AAGCGAGAGUGCGGGCCCAGGAG | |
| NC mimic | Sense | UCACAACCUCCUAGAAAGAGUAGA |
| Antisense | UCUACUCUUUCUAGGAGGUUGUGA | |
| miR-1343-3p inhibitor | Antisense | AAGCGAGAGUGCGGGCCCCAGGAG |
| NC inhibitor | Antisense | UCUACUCUUUCUAGGAGGUUGUGA |
| miR1343-3p agomir | sense | CUCCUGGGGCCCGCACUCUCGCUU |
| antisense | AAGCGAGAGUGCGGGCCCAGGAG | |
| si-OGDHL | Sense | CCCUCAGACUUGAUCACAAdTdT |
| Antisense | UUGUGAUCAAGUCUGAGGGdTdT | |
| si-NC | Sense | UUCUCCGAACGUGUCACGUdTdT |
| Antisense | ACGUGACACGUUCGGAGAAdTdT |
表1 引物和寡核苷酸序列
Tab.1 Primer and oligonucleotide sequences
| Primer name | Direction | Sequence (5'→3') |
|---|---|---|
| GAPDH | Forward | TGACATCAAGAAGGTGGTGAAGCAG |
| Reverse | GTGTCGCTGTTGAAGTCAGAGGAG | |
| U6 | Forward | GCTTCGGCAGCACATATACTAAAAT |
| Reverse | CGCTTCACGAATTTGCGTGTCAT | |
| miR-1343-3p | Forward | CGCGGCCTTAATGCTAATTGTGA |
| Reverse | AGTGCAGGGTCCGAGGTATT | |
| OGDHL | Forward | ATCACTCTGTCGCTGGTTGCC |
| Reverse | CGCTCAGGTGGAAGGTCTCATATAC | |
| PDHB | Forward | TTCTCCATGCAAGCCATTGACCAG |
| Reverse | CTACACCTGCTGAGGCACCATTG | |
| miR-1343-3p mimic | Sense | CUCCUGGGGCCCGCACUCUCGCUU |
| Antisense | AAGCGAGAGUGCGGGCCCAGGAG | |
| NC mimic | Sense | UCACAACCUCCUAGAAAGAGUAGA |
| Antisense | UCUACUCUUUCUAGGAGGUUGUGA | |
| miR-1343-3p inhibitor | Antisense | AAGCGAGAGUGCGGGCCCCAGGAG |
| NC inhibitor | Antisense | UCUACUCUUUCUAGGAGGUUGUGA |
| miR1343-3p agomir | sense | CUCCUGGGGCCCGCACUCUCGCUU |
| antisense | AAGCGAGAGUGCGGGCCCAGGAG | |
| si-OGDHL | Sense | CCCUCAGACUUGAUCACAAdTdT |
| Antisense | UUGUGAUCAAGUCUGAGGGdTdT | |
| si-NC | Sense | UUCUCCGAACGUGUCACGUdTdT |
| Antisense | ACGUGACACGUUCGGAGAAdTdT |
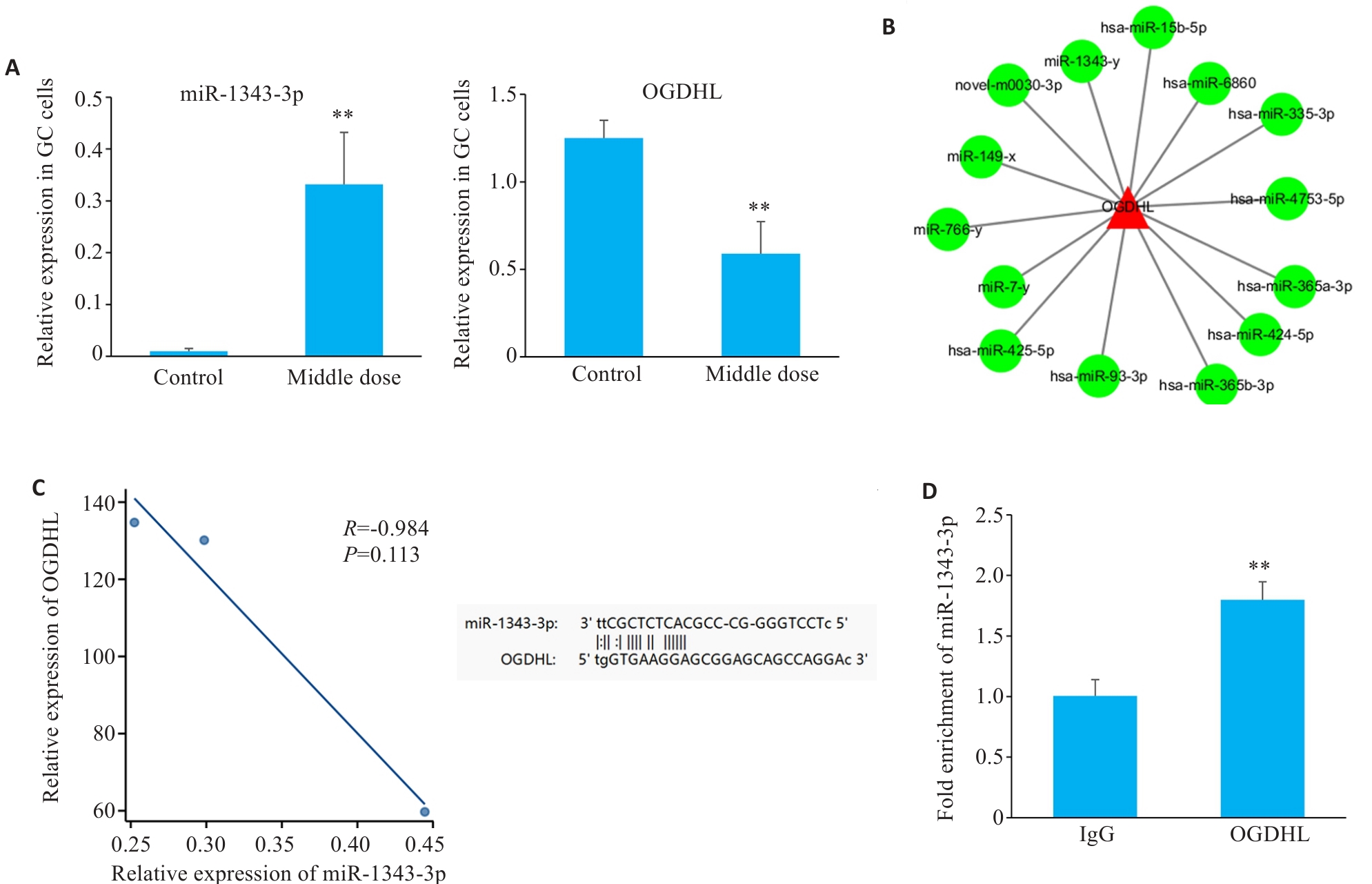
图1 高通量测序结合生物信息学分析miR-1343-3p与OGDHL相互作用关系
Fig.1 High-throughput sequencing combined with bioinformatics analysis for interaction between miR-1343-3p and OGDHL. A: Changes in miR-1343-3p and OGDHL in human gastric cancer cells after salidroside treatment detected by high-throughput sequencing. B: miRNA network and target gene analysis of the interaction between OGDHL and miR-1343-3p. C: Pearson correlation and genomic locus analysis of the association of OGDHL with miR-1343-3p in human gastric cancer cells. D: RNA immunoprecipitation assay for validating the interaction between OGDHL and miR-1343-3p. **P<0.01 vs Control/Ig G group (n=3).
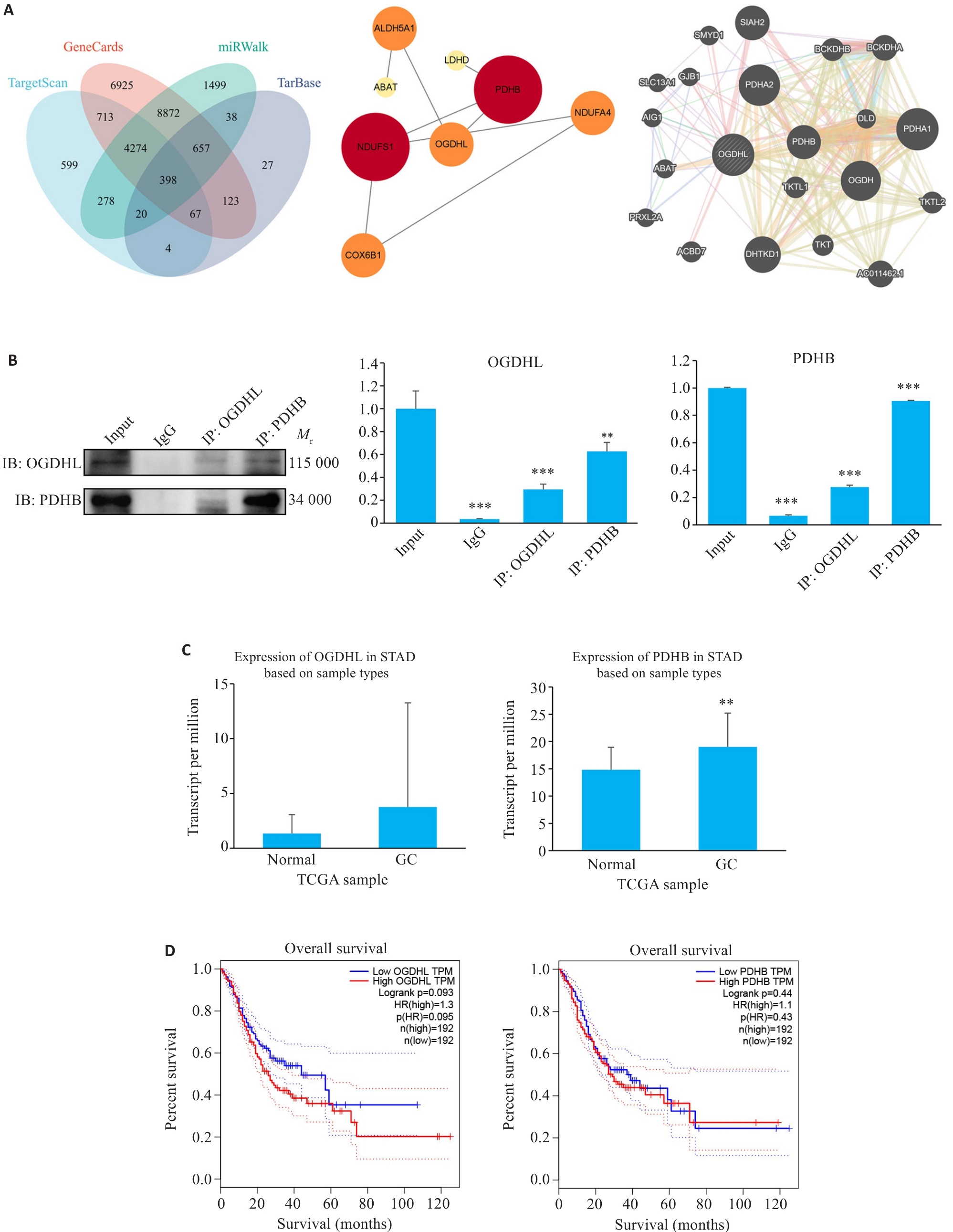
图2 OGDHL与PDHB蛋白的相互作用关系
Fig.2 Interaction between OGDHL and PDHB proteins. A: Bioinformatics analysis of the OGDHL-PDHB interaction network. B: Co-immunoprecipitation assay for confirming their binding (**P<0.01, ***P<0.001 vs Input; n=3). C: Differential expression of OGDHL and PDHB in gastric cancer (GC) tissues from The Cancer Genome Atlas (TCGA) (**P<0.01 vs Normal; n=3). D: Gene Expression Profiling Interactive Analysis (GEPIA) showing a negative correlation between high OGDHL/PDHB expression and clinical outcomes of GC patients, highlighting a significantly shorter overall survival (OS) of patients in high-expression groups.
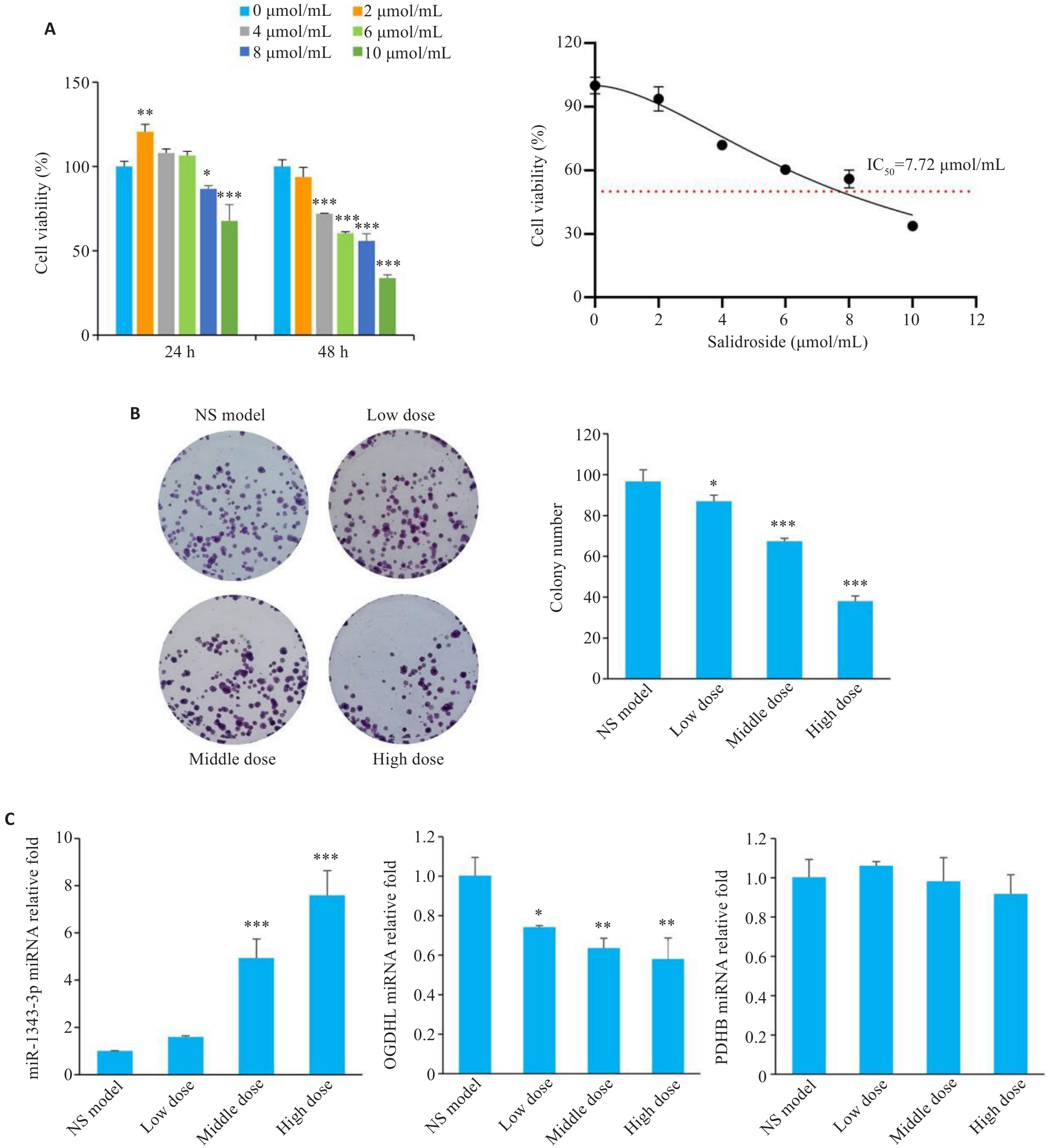
图3 红景天苷通过miR-1343-3p-OGDHL/PDHB轴抑制胃癌细胞的增殖
Fig.3 Salidroside inhibits proliferation of gastric cancer cells through the miR-1343-3p-OGDHL/PDHB axis. A: CCK-8 assay for assessing viability of MGC-803 cells treated with different concentrations of salidroside for 24 h/48 h. B: Colony formation assay of GC cells after salidroside treatment. C: qRT-PCR analysis showing changes in miR-1343-3p, OGDHL, and PDHB in GC cells after salidroside treatment. *P<0.05, **P<0.01, ***P<0.001 vs 0 μmol/mL or NS model (n=3).
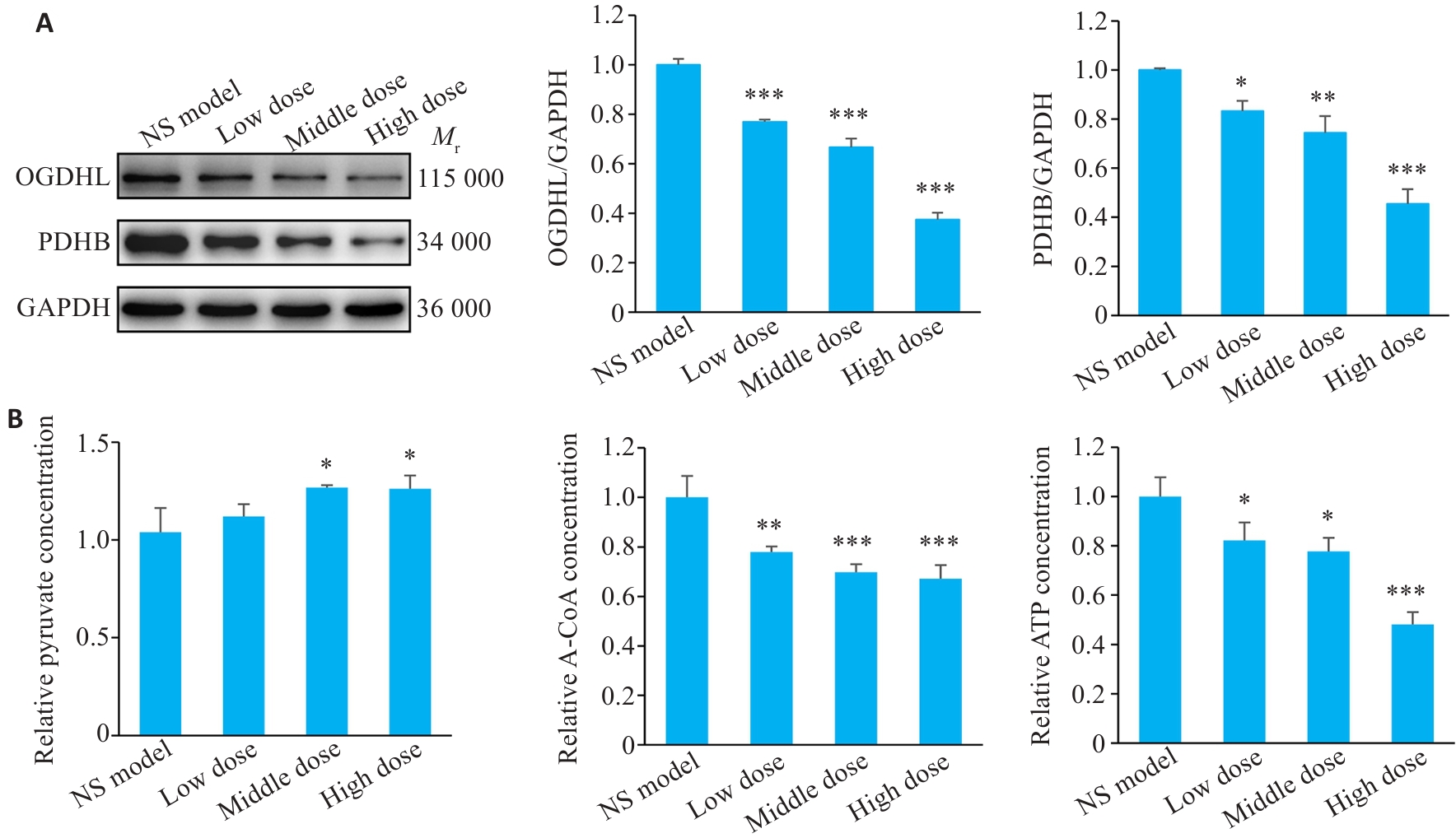
图4 红景天苷作用后观察下游蛋白分子能量的表达
Fig.4 Expression of downstream protein molecules and energy-related metabolites post-salidroside treatment. A: Western blotting results. B: Detection results of pyruvate, acetyl-CoA, and ATP in salidroside-treated GC cells using biochemical assay kits. *P<0.05, **P<0.01, ***P<0.001 vs NS model (n=3).
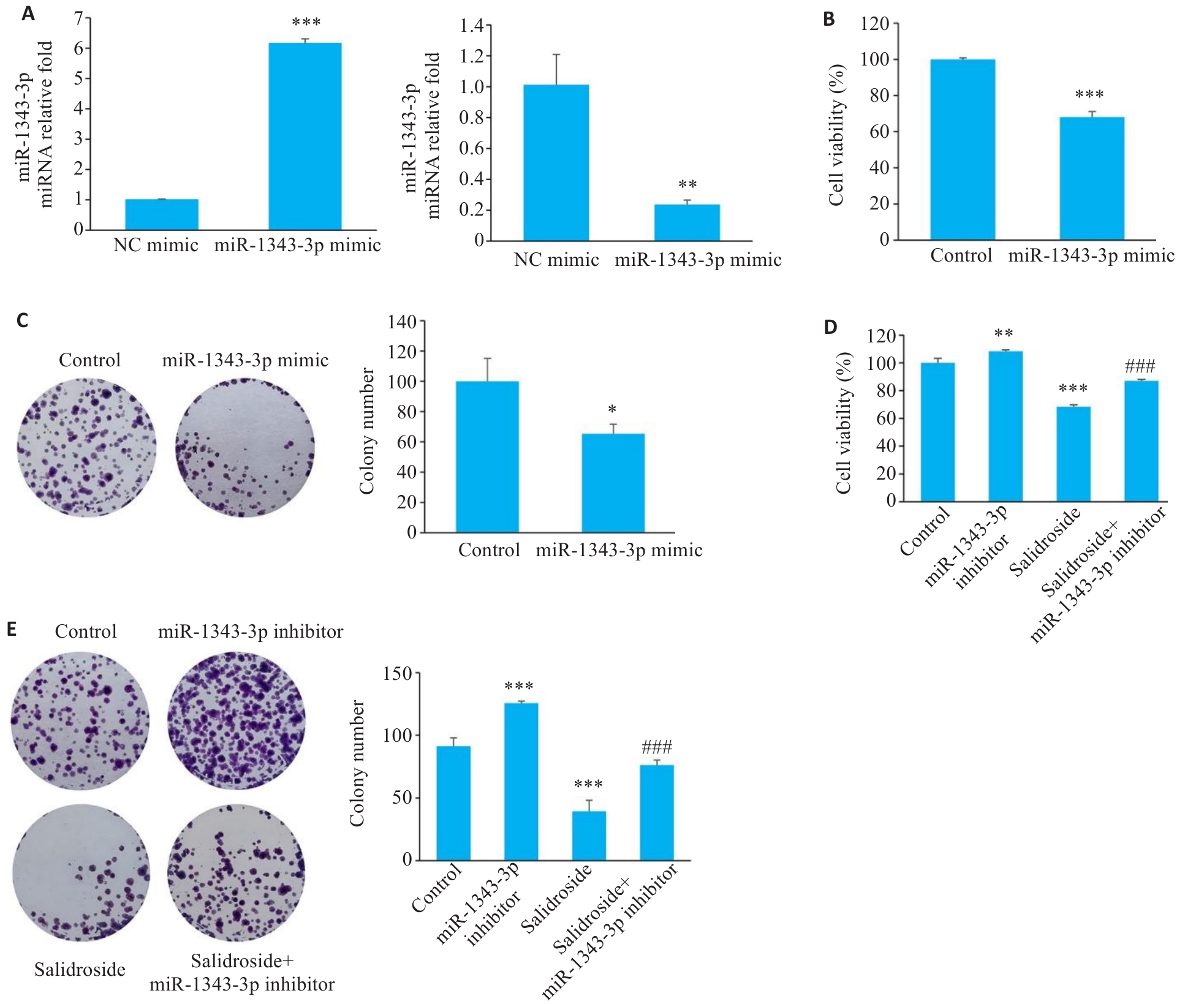
图5 miR-1343-3p mimic与miR-1343-3p inhibitor在细胞中的转染效率及细胞的功能性变化
Fig.5 Transfection efficiency and functional changes of miR-1343-3p mimic/inhibitor in human GC cells. A: Successful transfection of miR-1343-3p mimic or inhibitor into human GC cells (***P<0.01 vs NC mimic, **P<0.001 vs NC inhibitor; n=3). B-E: Effects of miR-1343-3p mimic/inhibitor on cell viability and clonogenic ability of GC cells (*P<0.05, **P<0.01, ***P<0.01 vs Control; ###P<0.001 vs Salidroside; n=3).
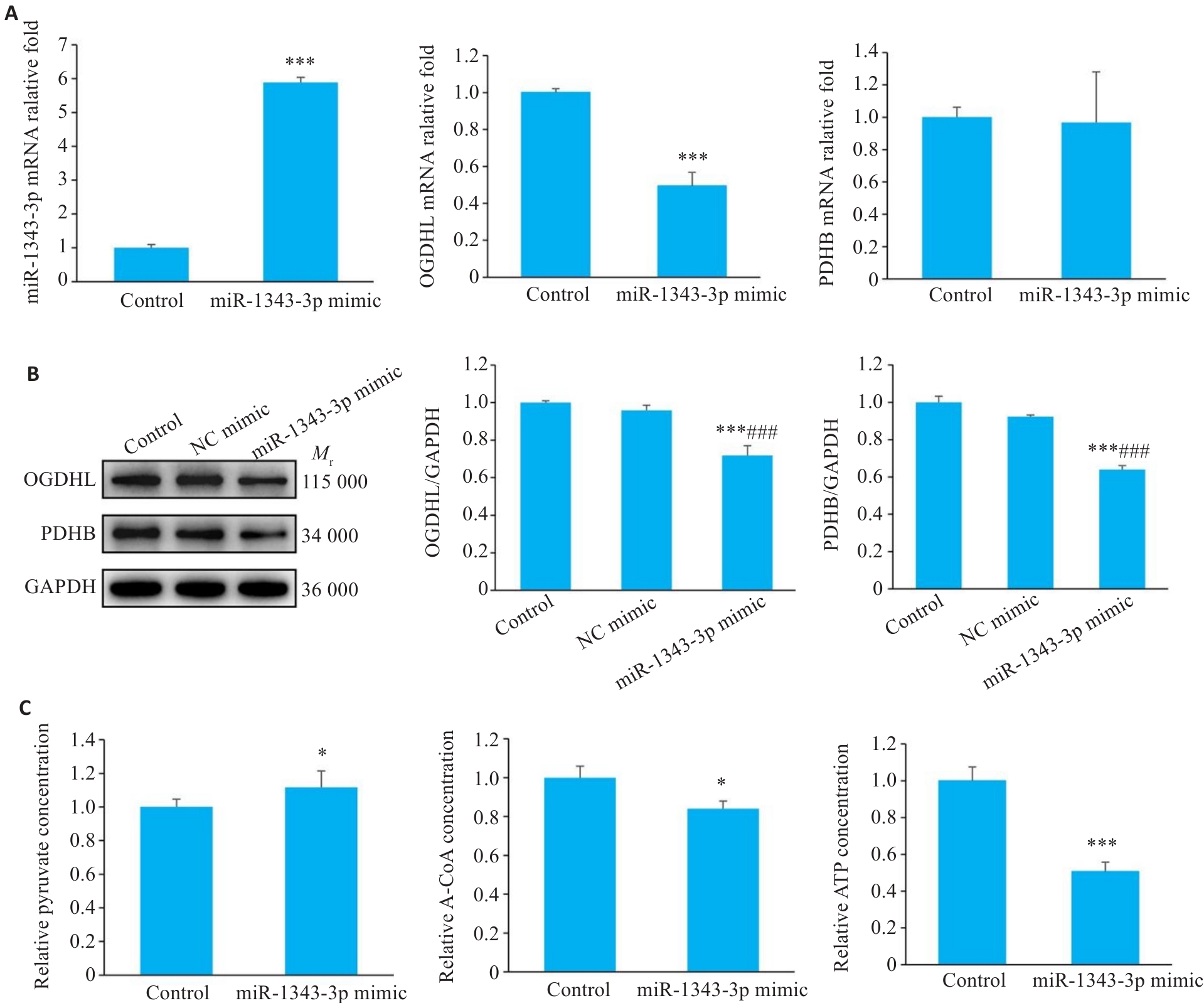
图6 miR-1343-3p mimic协同红景天苷调控胃癌细胞的生长
Fig.6 Synergistic regulation of GC cell growth by miR-1343-3p mimics and salidroside. A-C: Results of qRT-PCR, Western blotting, and biochemical assays for assessing changes in miR-1343-3p, OGDHL, PDHB, pyruvate, acetyl-CoA, and ATP levels in GC cells treated with miR-1343-3p mimic or inhibitors. *P<0.05, ***P<0.001 vs Control; ###P<0.001 vs NC mimic (n=3).
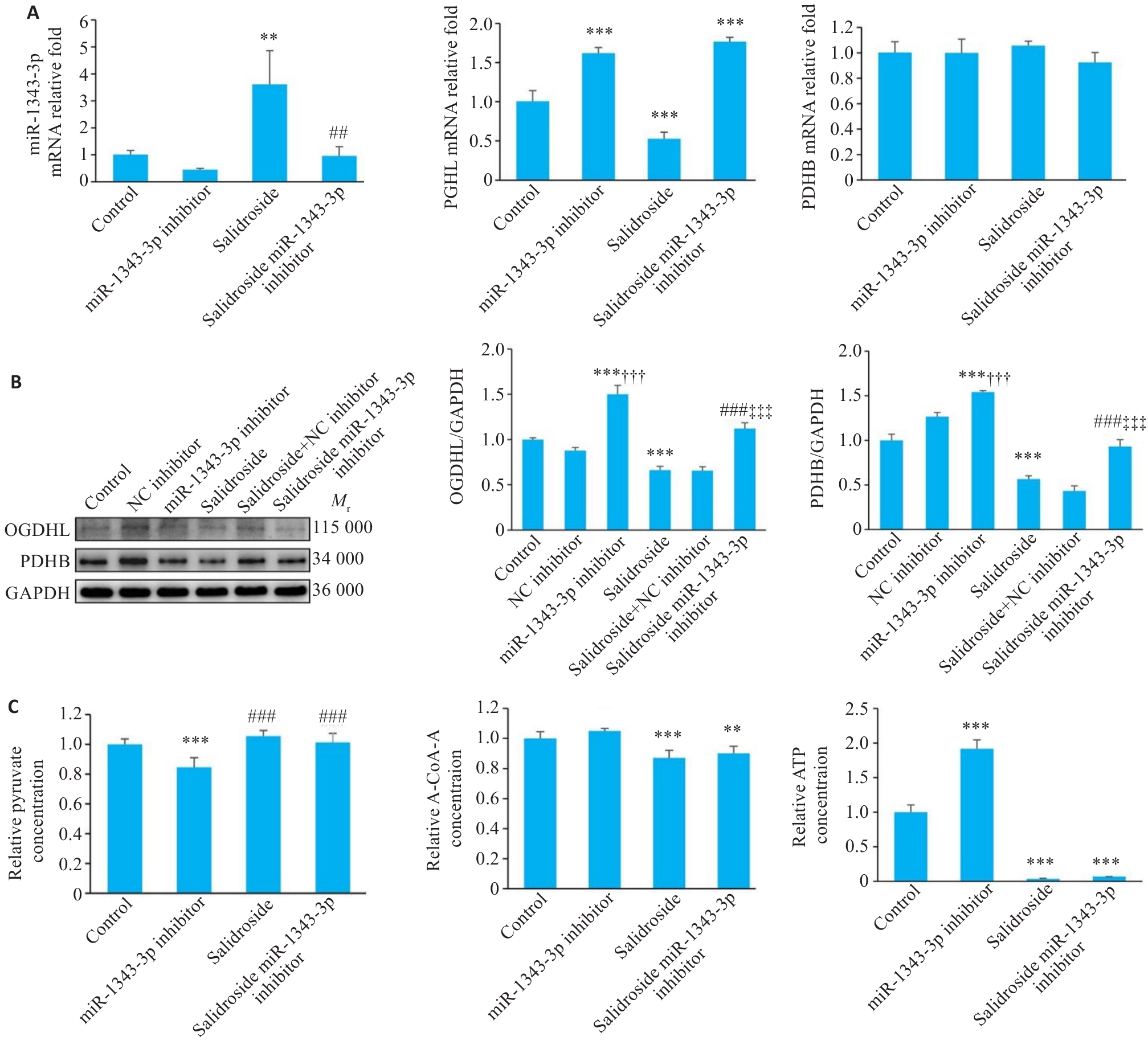
图7 miR-1343-3p inhibitor逆转了红景天苷调控胃癌细胞生长的抑制作用
Fig.7 miR-1343-3p inhibitor reverses salidroside-mediated suppression of GC cell growth. A-C: Results of qRT-PCR, Western blotting, and biochemical detection miR-1343-3p, OGDHL, PDHB, pyruvate, acetyl-CoA, and ATP levels in GC cells following miR-1343-3p inhibitor treatment. **P<0.01, ***P<0.001 vs Control, ##P<0.01, ###P<0.001 vs Salidroside; †††P<0.001 vs NC inhibitor, ‡‡‡P<0.001 vs Salidroside+NC inhibitor; C: ###P<0.001 vs miR-1343-3p inhibitor (n=3).
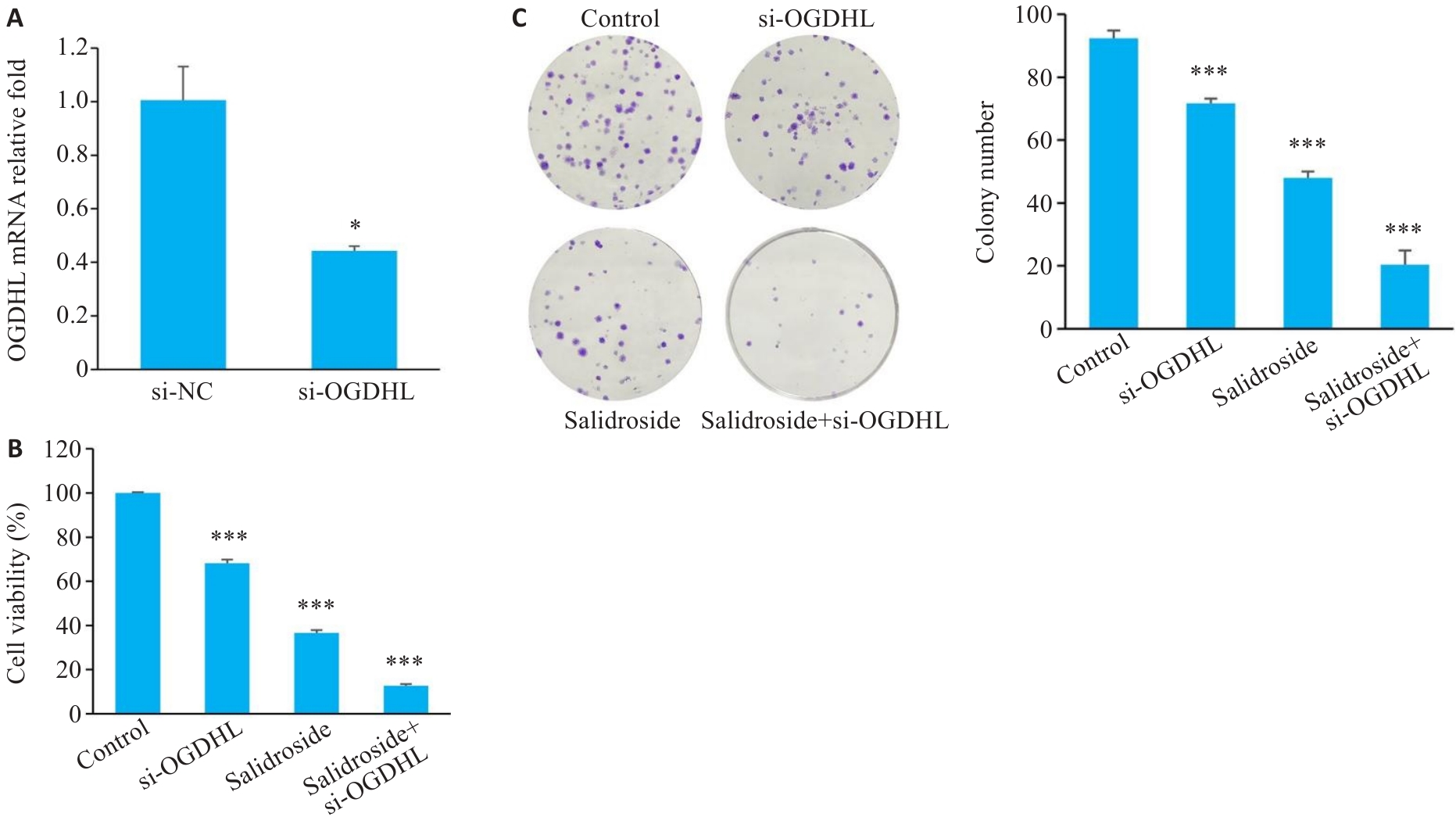
图8 si-OGDHL在细胞中的转染效率及形态学变化
Fig.8 Transfection efficiency and functional changes of si-OGDHL in human GC cells. A: Successful transfection of si-OGDHL into GC cells. B, C: Effects of si-OGDHL on cell viability and clonogenic ability in GC cells. *P<0.05, ***P<0.001 vs Control (n=3).
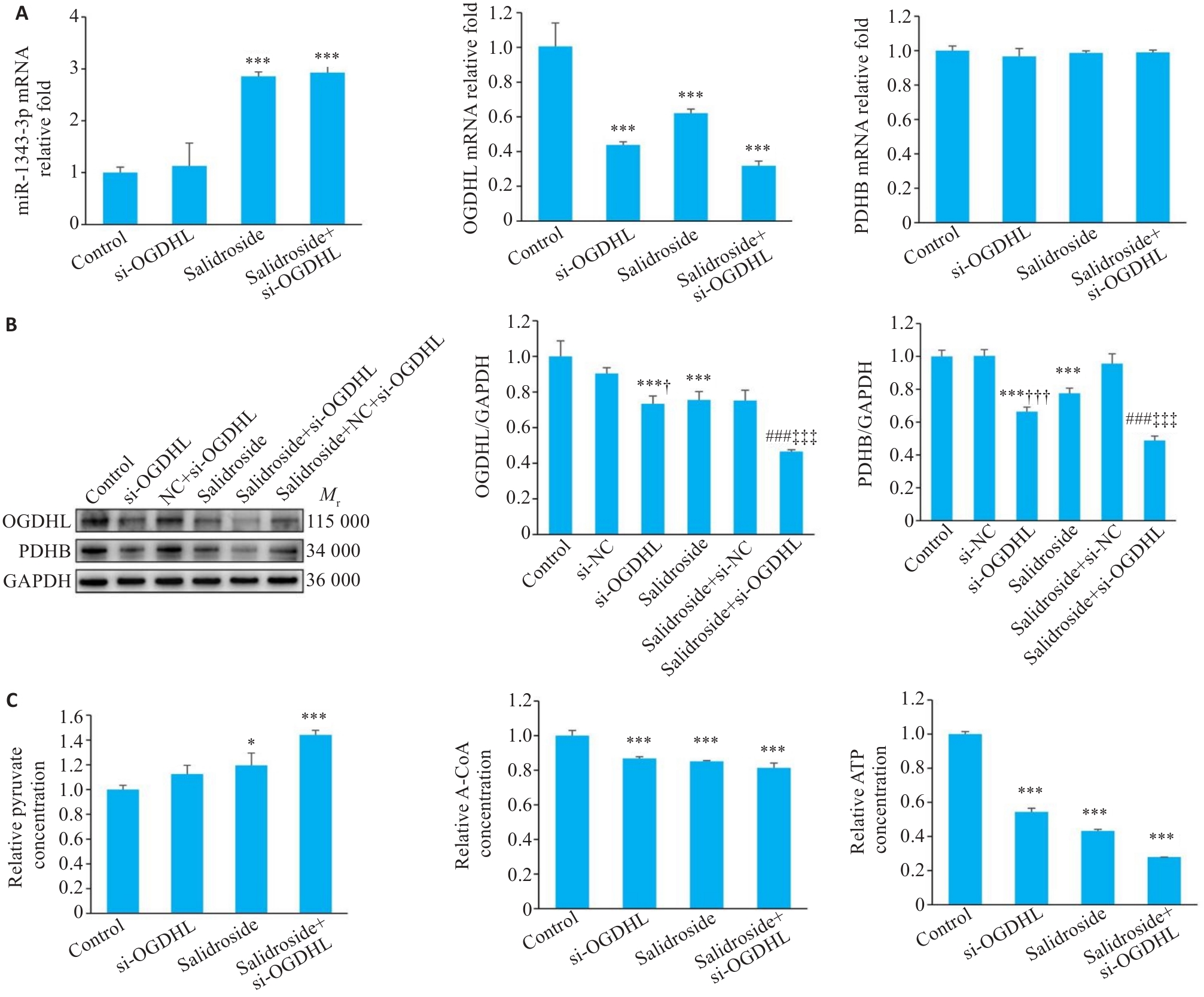
图9 敲低OGDHL协同红景天苷抑制胃癌细胞生长
Fig.9 Knockdown of OGDHL synergizes with salidroside to inhibit GC cell growth. A-C: Results of qRT-PCR, Western blotting, and assay kits for miR-1343-3p, OGDHL, PDHB, pyruvate, acetyl-CoA, and ATP levels in GC cells treated with si-OGDHL. *P<0.05, ***P<0.001 vs Control; ###P<0.001 vs Salidroside; †P<0.05, †††P<0.001 vs si-NC, ‡‡‡P<0.001 vs Salidroside+si-NC (n=3).
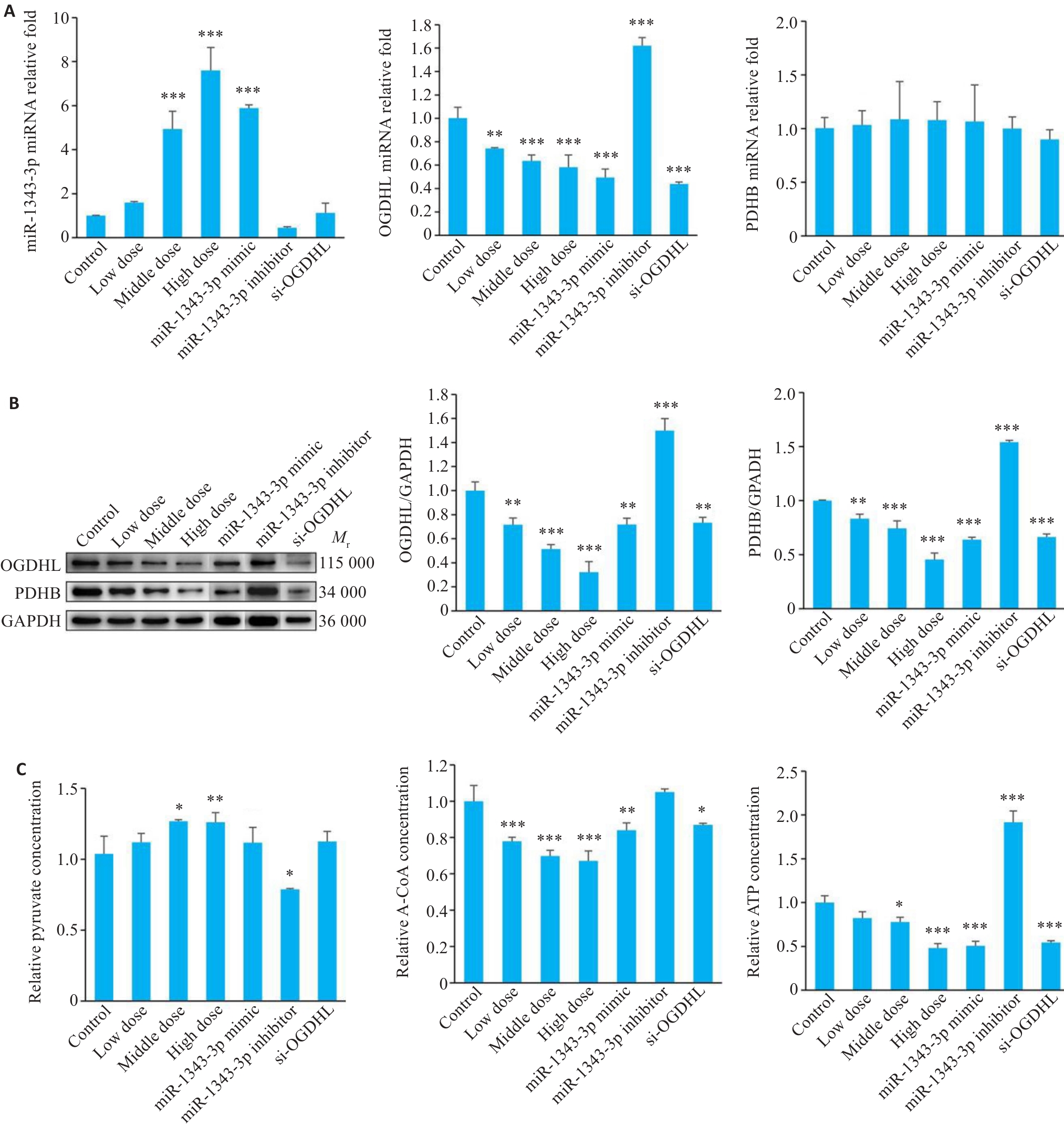
图10 不同方法处理组作用后,胃癌细胞中miR-1343-3p, OGDHL, PDHB及丙酮酸、乙酰CoA和ATP水平的变化
Fig.10 Changes in miR-1343-3p, OGDHL, PDHB, pyruvate, acetyl-CoA, and ATP levels in GC cells following different treatments. A-C:Results of qRT-PCR, Western blotting and biochemical assay kits for detecting miR-1343-3p, OGDHL, PDHB, pyruvate, acetyl-CoA and ATP levels in GC cells treated with salidroside, miR-1343-3p mimic, miR-1343-3p inhibitor and si-OGDHL. *P<0.05, **P<0.01, ***P<0.001 vs Control (n=3).
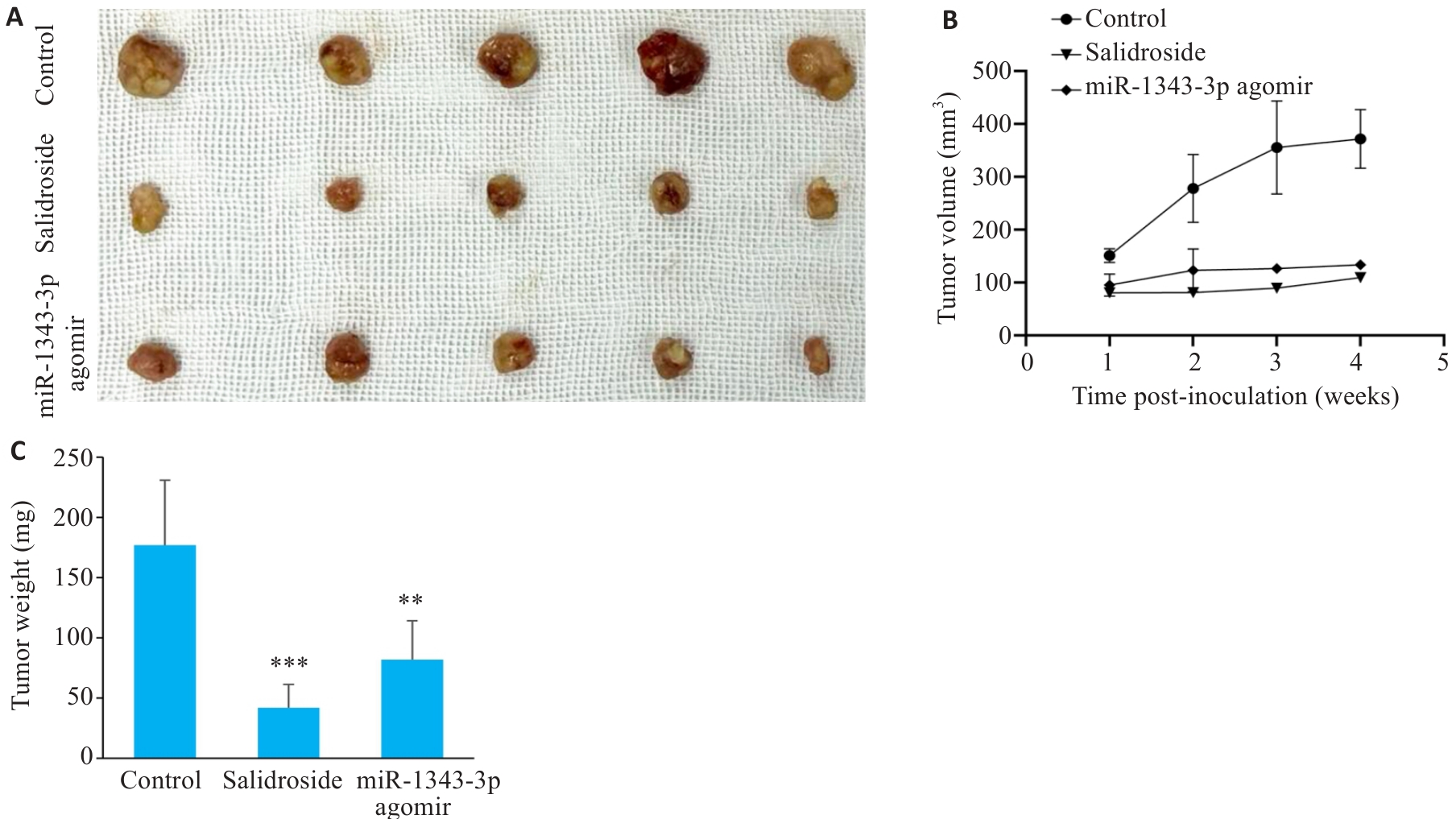
图11 异种移植GC细胞后观察活体内瘤块形态变化
Fig.11 Observation of morphological changes in tumor masses after xenografting of GC cells in nude mice. A-C: Gross appearance and comparison of volume/weight in tumor masses treated with salidroside and miR-1343-3p overexpression. **P<0.01, ***P<0.001 vs Control.
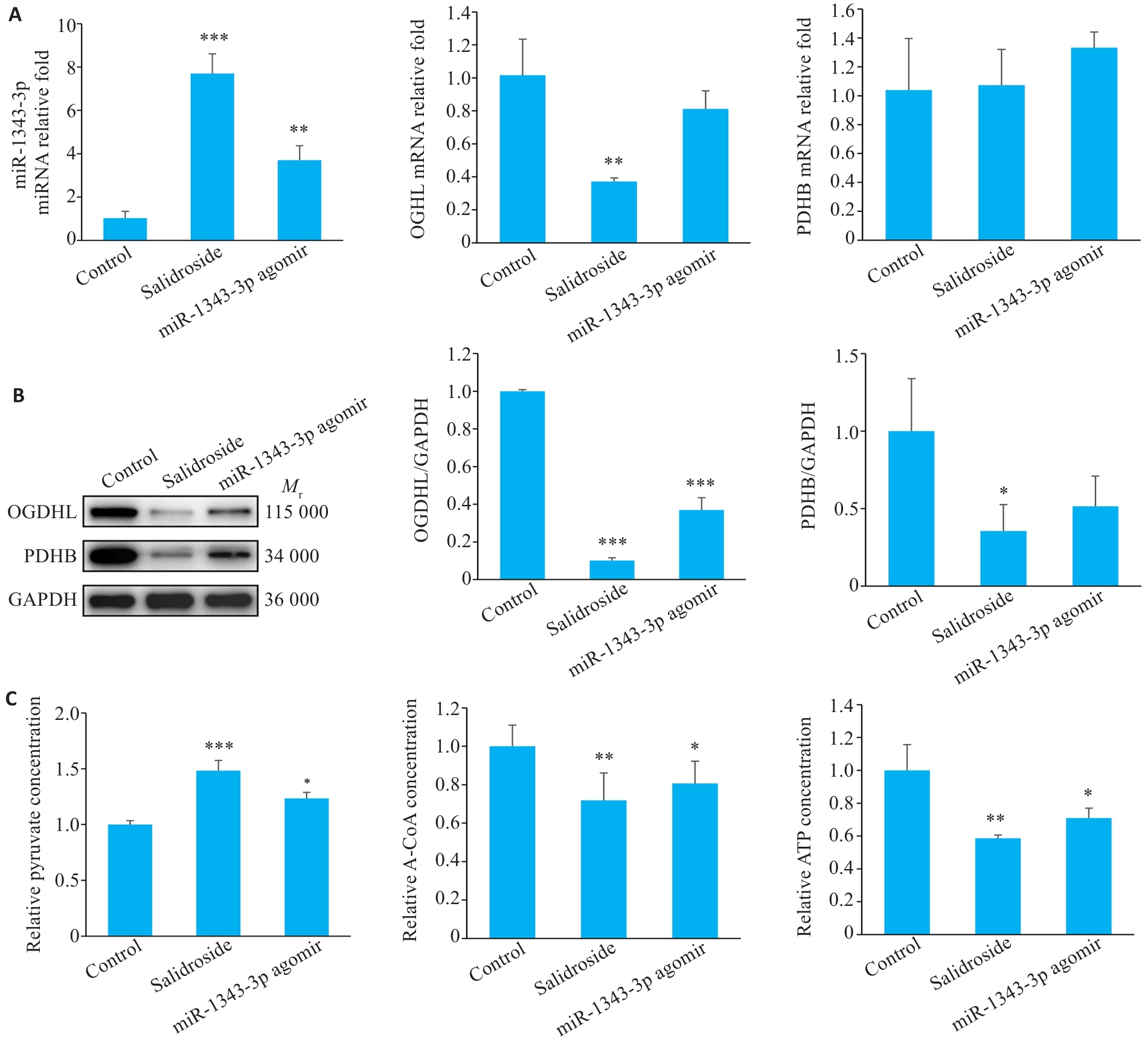
图12 红景天苷同miR-1343-3p在活体内抑制胃癌肿瘤生长
Fig.12 Salidroside and miR-1343-3p inhibit gastric cancer tumor growth in nude mice. A-C: Changes in miR-1343-3p levels, OGDHL and PDHB expression, and concentrations of pyruvate, acetyl-CoA, and ATP in tumor tissues analyzed by qRT-PCR, Western blotty, and biochemical kits. *P<0.05, **P<0.01, ***P<0.001 vs Control.
| 1 | Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-49. doi:10.3322/caac.21660 |
| 2 | Han BF, Zheng RS, Zeng HM, et al. Cancer incidence and mortality in China, 2022[J]. J Natl Cancer Cent, 2024, 4(1): 47-53. doi:10.1016/j.jncc.2024.01.006 |
| 3 | He SY, Xia CF, Li H, et al. Cancer profiles in China and comparisons with the USA: a comprehensive analysis in the incidence, mortality, survival, staging, and attribution to risk factors[J]. Sci China Life Sci, 2024, 67(1): 122-31. doi:10.1007/s11427-023-2423-1 |
| 4 | Greene J, Segaran A, Lord S. Targeting OXPHOS and the electron transport chain in cancer; Molecular and therapeutic implications[J]. Semin Cancer Biol, 2022, 86(Pt 2): 851-9. doi:10.1016/j.semcancer.2022.02.002 |
| 5 | Jiang B, Feng LF, Yang T, et al. Combination of chloroquine diphosphate and salidroside induces human liver cell apoptosis via regulation of mitochondrial dysfunction and autophagy[J]. Mol Med Rep, 2023, 27(2): 37. doi:10.3892/mmr.2022.12924 |
| 6 | Rong L, Li ZD, Leng X, et al. Salidroside induces apoptosis and protective autophagy in human gastric cancer AGS cells through the PI3K/Akt/mTOR pathway[J]. Biomed Pharmacother, 2020, 122: 109726. doi:10.1016/j.biopha.2019.109726 |
| 7 | Kang DY, Sp N, Kim DH, et al. Salidroside inhibits migration, invasion and angiogenesis of MDA-MB 231 TNBC cells by regulating EGFR/Jak2/STAT3 signaling via MMP2[J]. Int J Oncol, 2018, 53(2): 877-85. doi:10.3892/ijo.2018.4430 |
| 8 | Warburg O. On the origin of cancer cells[J]. Science, 1956, 123(3191): 309-14. doi:10.1126/science.123.3191.309 |
| 9 | Vaupel P, Multhoff G. Revisiting the Warburg effect: historical dogma versus current understanding[J]. J Physiol, 2021, 599(6): 1745-57. doi:10.1113/JP278810 |
| 10 | Liu XL, Ma RX, Yi B, et al. microRNAs are involved in the development and progression of gastric cancer[J]. Acta Pharmacol Sin, 2021, 42(7): 1018-26. doi:10.1038/s41401-020-00540-0 |
| 11 | Shen XJ, Zhu XY, Hu PX, et al. Knockdown circZNF131 inhibits cell progression and glycolysis in gastric cancer through miR-186-5p/PFKFB2 axis[J]. Biochem Genet, 2022, 60(5): 1567-84. doi:10.1007/s10528-021-10165-5 |
| 12 | Wang XP, Zhang ZD, Cao XL. Salidroside inhibited the proliferation of gastric cancer cells through up-regulating tumor suppressor miR-1343-3p and down-regulating MAP3K6/MMP24 signal molecules[J]. Cancer Biol Ther, 2024, 25(1): 2322206. doi:10.1080/15384047.2024.2322206 |
| 13 | Sumegi B, Srere PA. Complex I binds several mitochondrial NAD-coupled dehydrogenases[J]. J Biol Chem, 1984, 259(24): 15040-5. doi:10.1016/s0021-9258(17)42511-7 |
| 14 | Liu G, Wu XY, Chen J. Identification and validation of a glycolysis-related gene signature for depicting clinical characteristics and its relationship with tumor immunity in patients with colon cancer[J]. Aging, 2022, 14(21): 8700-18. doi:10.18632/aging.204226 |
| 15 | Hoque MO, Kim MS, Ostrow KL, et al. Genome-wide promoter analysis uncovers portions of the cancer methylome[J]. Cancer Res, 2008, 68(8): 2661-70. doi:10.1158/0008-5472.CAN-07-5913 |
| 16 | Khalaj-Kondori M, Hosseinnejad M, Hosseinzadeh A, et al. Aberrant hypermethylation of OGDHL gene promoter in sporadic colorectal cancer[J]. Curr Probl Cancer, 2020, 44(1): 100471. doi:10.1016/j.currproblcancer.2019.03.001 |
| 17 | Liu YT, Leng P, Liu Y, et al. Crosstalk between methylation and ncRNAs in breast cancer: therapeutic and diagnostic implications[J]. Int J Mol Sci, 2022, 23(24): 15759. doi:10.3390/ijms232415759 |
| 18 | Patel MS, Nemeria NS, Furey W, et al. The pyruvate dehydrogenase complexes: structure-based function and regulation[J]. J Biol Chem, 2014, 289(24): 16615-23. doi:10.1074/jbc.R114.563148 |
| 19 | Perham RN. Domains, motifs, and linkers in 2-oxo acid dehydrogenase multienzyme complexes: a paradigm in the design of a multifunctional protein[J]. Biochemistry, 1991, 30(35): 8501-12. doi:10.1021/bi00099a001 |
| 20 | Yonashiro R, Eguchi K, Wake M, et al. Pyruvate dehydrogenase PDH-E1β controls tumor progression by altering the metabolic status of cancer cells[J]. Cancer Res, 2018, 78(7): 1592-603. doi:10.1158/0008-5472.can-17-1751 |
| 21 | Wang YH, Patti GJ. The Warburg effect: a signature of mitochondrial overload[J]. Trends Cell Biol, 2023, 33(12): 1014-20. doi:10.1016/j.tcb.2023.03.013 |
| 22 | Yang SX, Wang LS, Zeng Y, et al. Salidroside alleviates cognitive impairment by inhibiting ferroptosis via activation of the Nrf2/GPX4 axis in SAMP8 mice[J]. Phytomedicine, 2023, 114: 154762. doi:10.1016/j.phymed.2023.154762 |
| 23 | Park JH, Pyun WY, Park HW. Cancer metabolism: phenotype, signaling and therapeutic targets[J]. Cells, 2020, 9(10): 2308. doi:10.3390/cells9102308 |
| 24 | Zhao JY, Feng KR, Wang F, et al. A retrospective overview of PHGDH and its inhibitors for regulating cancer metabolism[J]. Eur J Med Chem, 2021, 217: 113379. doi:10.1016/j.ejmech.2021.113379 |
| 25 | Min HY, Lee HY. Oncogene-driven metabolic alterations in cancer[J]. Biomol Ther, 2018, 26(1): 45-56. doi:10.4062/biomolther.2017.211 |
| 26 | Hu J, Huang LJ, Ding Q, et al. Long noncoding RNA HAGLR sponges miR-338-3p to promote 5-Fu resistance in gastric cancer through targeting the LDHA-glycolysis pathway[J]. Cell Biol Int, 2022, 46(2): 173-84. doi:10.1002/cbin.11714 |
| 27 | Liu HP, Chen K, Wang L, et al. miR-613 inhibits Warburg effect in gastric cancer by targeting PFKFB2[J]. Biochem Biophys Res Commun, 2019, 515(1): 37-43. doi:10.1016/j.bbrc.2019.05.001 |
| 28 | Pavlides S, Whitaker-Menezes D, Castello-Cros R, et al. The reverse Warburg effect: aerobic glycolysis in cancer associated fibroblasts and the tumor stroma[J]. Cell Cycle, 2009, 8(23): 3984-4001. doi:10.4161/cc.8.23.10238 |
| 29 | Mookerjee SA, Gerencser AA, Nicholls DG, et al. Quantifying intracellular rates of glycolytic and oxidative ATP production and consumption using extracellular flux measurements[J]. J Biol Chem, 2017, 292(17): 7189-207. doi:10.1074/jbc.M116.774471 |
| 30 | Annison EF, Lindsay DB, White RR. Metabolic interrelations of glucose and lactate in sheep[J]. Biochem J, 1963, 88(2): 243-8. doi:10.1042/bj0880243 |
| 31 | Hui S, Ghergurovich JM, Morscher RJ, et al. Glucose feeds the TCA cycle via circulating lactate[J]. Nature, 2017, 551(7678): 115-8. doi:10.1038/nature24057 |
| 32 | Sharif T, Martell E, Dai C, et al. Phosphoglycerate dehydrogenase inhibition induces p-mTOR-independent autophagy and promotes multilineage differentiation in embryonal carcinoma stem-like cells[J]. Cell Death Dis, 2018, 9(10): 990. doi:10.1038/s41419-018-0997-8 |
| 33 | Shen LL, Zhang JF, Zheng ZT, et al. PHGDH inhibits ferroptosis and promotes malignant progression by upregulating SLC7A11 in bladder cancer[J]. Int J Biol Sci, 2022, 18(14): 5459-74. doi:10.7150/ijbs.74546 |
| 34 | Popovic D, Vucic D, Dikic I. Ubiquitination in disease pathogenesis and treatment[J]. Nat Med, 2014, 20(11): 1242-53. doi:10.1038/nm.3739 |
| 35 | Cockram PE, Kist M, Prakash S, et al. Ubiquitination in the regulation of inflammatory cell death and cancer[J]. Cell Death Differ, 2021, 28(2): 591-605. doi:10.1038/s41418-020-00708-5 |
| [1] | 陈鑫源, 吴成挺, 李瑞迪, 潘雪芹, 张耀丹, 陶俊宇, 林才志. 双术汤通过P53/SLC7A11/GPX4通路诱导胃癌细胞铁死亡[J]. 南方医科大学学报, 2025, 45(7): 1363-1371. |
| [2] | 吴璇, 方家敏, 韩玮玮, 陈琳, 孙菁, 金齐力. 高表达PRELID1促进胃癌细胞上皮间质转化并与不良预后相关[J]. 南方医科大学学报, 2025, 45(7): 1535-1542. |
| [3] | 张毅, 沈昱, 万志强, 陶嵩, 柳亚魁, 王栓虎. CDKN3高表达促进胃癌细胞的迁移和侵袭:基于调控p53/NF-κB信号通路和抑制胃癌细胞凋亡[J]. 南方医科大学学报, 2025, 45(4): 853-861. |
| [4] | 黄晴晴, 张文静, 张小凤, 王炼, 宋雪, 耿志军, 左芦根, 王月月, 李静, 胡建国. 高表达MYO1B促进胃癌细胞增殖、迁移和侵袭并与患者的不良预后有关[J]. 南方医科大学学报, 2025, 45(3): 622-631. |
| [5] | 宋雪, 陈悦, 张敏, 张诺, 左芦根, 李静, 耿志军, 张小凤, 王月月, 王炼, 胡建国. GPSM2在胃癌组织中高表达并通过促进肿瘤细胞的增殖影响患者预后[J]. 南方医科大学学报, 2025, 45(2): 229-238. |
| [6] | 陈孝华, 鲁辉, 王子良, 王炼, 夏勇生, 耿志军, 张小凤, 宋雪, 王月月, 李静, 胡建国, 左芦根. ABI2在胃癌进展和预后中的作用及其调控机制[J]. 南方医科大学学报, 2024, 44(9): 1653-1661. |
| [7] | 叶梦楠, 武鸿美, 梅琰, 张庆玲. CREM在胃癌中高表达并与患者的不良预后相关[J]. 南方医科大学学报, 2024, 44(9): 1776-1782. |
| [8] | 耿志军, 杨晶晶, 牛民主, 刘馨悦, 施金冉, 刘亦珂, 姚新宇, 张雨路, 张小凤, 胡建国. 桑黄酮G通过调控PI3K/AKT/mTOR通路抑制胃癌细胞的生长、迁移和侵袭[J]. 南方医科大学学报, 2024, 44(8): 1476-1484. |
| [9] | 庞一丹, 刘雅, 陈思嫒, 张荆雷, 曾今, 潘元明, 安娟. SPAG5在胃癌细胞恶性增殖中的生物学作用[J]. 南方医科大学学报, 2024, 44(8): 1497-1507. |
| [10] | 夏勇生, 王炼, 陈孝华, 张雨路, 孙奥飞, 陈德利. 过表达TSR2通过下调PI3K/AKT信号通路抑制胃癌细胞的增殖和侵袭[J]. 南方医科大学学报, 2024, 44(5): 913-919. |
| [11] | 杨晶晶, 殷丽霞, 段婷, 牛民主, 何震东, 陈心蕊, 张小凤, 李静, 耿志军, 左芦根. 胃癌组织中高表达ATP5A1与患者术后的不良预后和肿瘤细胞的糖代谢有关[J]. 南方医科大学学报, 2024, 44(5): 974-980. |
| [12] | 梁一豪, 赖颖君, 袁燕文, 袁 炜, 张锡波, 张拔山, 卢志锋. 基于GEO数据库筛选胃癌差异表达基因及其功能和通路富集分析[J]. 南方医科大学学报, 2024, 44(3): 605-616. |
| [13] | 沈梦迪, 赵 娜, 邓晓晶, 邓 敏. COX6B2在胃癌组织中高表达并影响患者的远期预后:基于抑制p53信号调控胃癌细胞的增殖及细胞周期[J]. 南方医科大学学报, 2024, 44(2): 289-297. |
| [14] | 张 诺, 张 震, 张雨路, 宋 雪, 张小凤, 李 静, 左芦根, 胡建国. PCID2在胃癌组织中高表达并通过调控细胞周期进程和增殖影响患者预后[J]. 南方医科大学学报, 2024, 44(2): 324-332. |
| [15] | 张文静, 张 诺, 杨 子, 张小凤, 孙奥飞, 王 炼, 宋 雪, 耿志军, 李 静, 胡建国. BZW1 高表达促进胃癌细胞的侵袭和转移:基于调控Wnt//β-catenin通路和促进上皮间质转化[J]. 南方医科大学学报, 2024, 44(2): 354-362. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||