Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 550-558.doi: 10.12122/j.issn.1673-4254.2026.03.09
Xinyi HU1,2,4( ), Weijian WANG4(
), Weijian WANG4( ), Hui LI3,4, Zongzheng CHEN4,5, Xin ZHOU6, Junfei YUAN6, Liang CHEN1,4,5(
), Hui LI3,4, Zongzheng CHEN4,5, Xin ZHOU6, Junfei YUAN6, Liang CHEN1,4,5( )
)
Received:2025-09-08
Online:2026-03-20
Published:2026-03-26
Contact:
Liang CHEN
E-mail:huxydoct@163.com;wangweijian904@163.com;chenliangsmmu@163.com
Supported by:Xinyi HU, Weijian WANG, Hui LI, Zongzheng CHEN, Xin ZHOU, Junfei YUAN, Liang CHEN. A nomogram model for predicting MACE risk following primary percutaneous coronary intervention in STEMI patients: an exploratory study based on serum GSDMD[J]. Journal of Southern Medical University, 2026, 46(3): 550-558.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.09
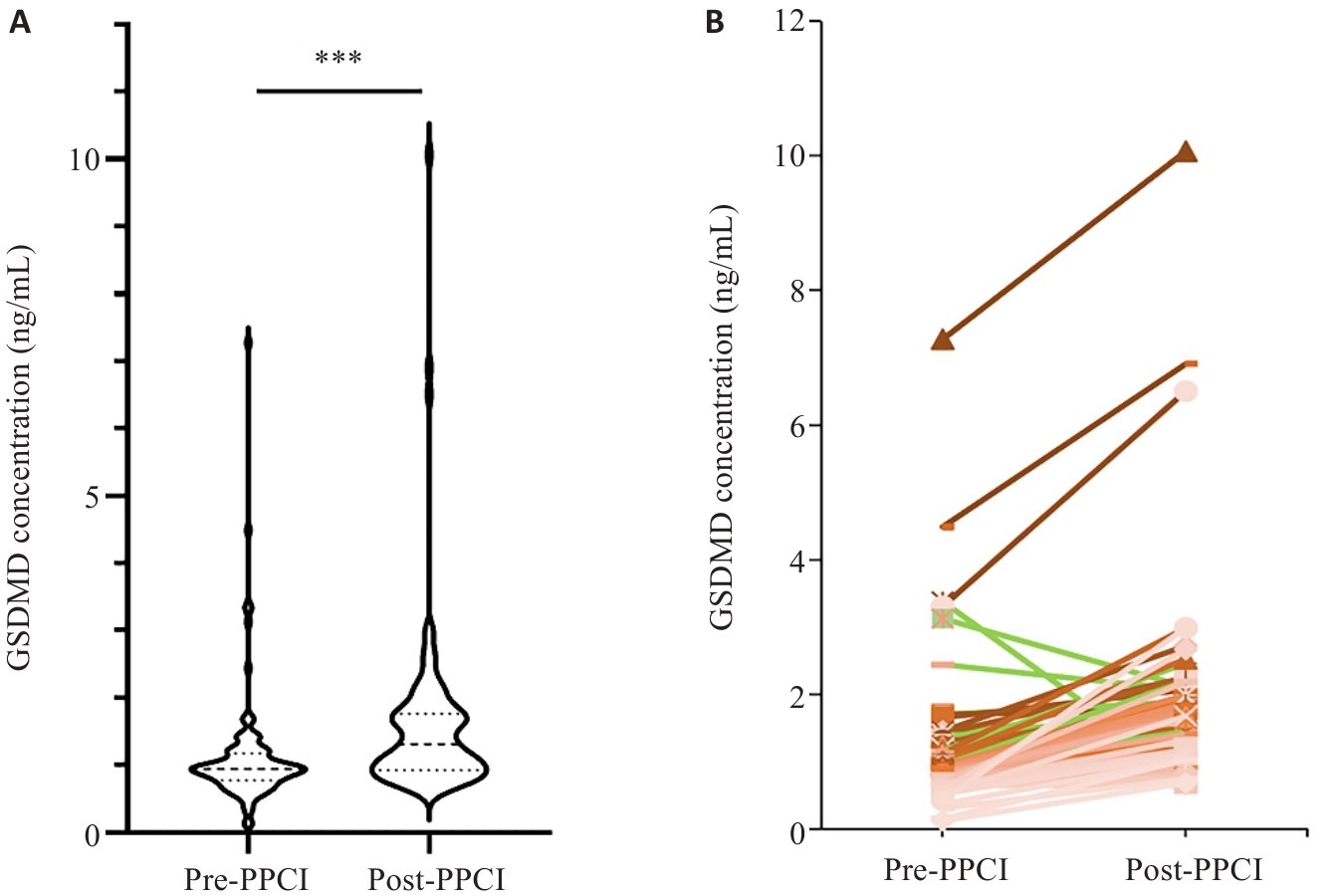
Fig.1 Comparison of pre- and post-PPCI serum GSDMD concentrations in STEMI patients. A: Violin plot showing the distribution of pre- and post-PPCI serum GSDMD concentrations; B: Paired line chart depicting the changes in pre- and post-PPCI serum GSDMD concentrations of the same patient. ***P<0.001.
| Variable | Non-MACE (n=74) | MACE (n=26) | Overall (n=100) | P |
|---|---|---|---|---|
| Pre-PPCI GSDMD | 0.036 | |||
| Low | 46 (82.1%) | 10 (17.9%) | 56 (100.0%) | |
| High | 28 (63.6%) | 16 (36.4%) | 44 (100.0%) | |
| Post-PPCI GSDMD | 0.010 | |||
| Low | 57 (81.4%) | 13 (18.6%) | 70 (100.0%) | |
| High | 17 (56.7%) | 13 (43.3%) | 30 (100.0%) | |
| ΔGSDMD | 0.141 | |||
| Low | 38 (80.9%) | 9(19.1%) | 47 (100.0%) | |
| High | 36 (67.9%) | 17(32.1%) | 53 (100.0%) |
Tab.1 Association of pre-PPCI GSDMD, post-PPCI GSDMD, and ΔGSDMD with 30-day MACE in STEMI patients [n (%)]
| Variable | Non-MACE (n=74) | MACE (n=26) | Overall (n=100) | P |
|---|---|---|---|---|
| Pre-PPCI GSDMD | 0.036 | |||
| Low | 46 (82.1%) | 10 (17.9%) | 56 (100.0%) | |
| High | 28 (63.6%) | 16 (36.4%) | 44 (100.0%) | |
| Post-PPCI GSDMD | 0.010 | |||
| Low | 57 (81.4%) | 13 (18.6%) | 70 (100.0%) | |
| High | 17 (56.7%) | 13 (43.3%) | 30 (100.0%) | |
| ΔGSDMD | 0.141 | |||
| Low | 38 (80.9%) | 9(19.1%) | 47 (100.0%) | |
| High | 36 (67.9%) | 17(32.1%) | 53 (100.0%) |
| Basic clinical characteristics | Non-MACE (n=74) | MACE (n=26) | Overall (n=100) | P |
|---|---|---|---|---|
| Male [n (%)] | 57 (77.0) | 25 (96.2) | 82 (82.0) | 0.029 |
| Age [year, Median (Q1, Q3)] | 59.5 (46.75, 74.0) | 61.5 (49.0, 77.5) | 60.0 (48.5, 74.0) | 0.494 |
| Days of hospitalization [day, Median (Q1, Q3)] | 9.00 (8.00, 11.0) | 10.5 (9.75, 15.0) | 10.0 (8.00, 12.0) | 0.005 |
| Killip classes [n (%)] | 0.000 | |||
| I | 55 (74.3) | 15 (57.7) | 70 (70.0) | |
| II | 15 (20.3) | 4 (15.4) | 19 (19.0) | |
| III | 4 (5.4) | 1 (3.8) | 5 (5.0) | |
| IV | 0 (0.0) | 6 (23.1) | 6 (6.0) | |
| Body weight [kg, Median (Q1, Q3)] | 70.0 (62.0, 77.6) | 75.0 (65.0, 80.0) | 71.5 (63.3, 79.5) | 0.214 |
| Number of stents [n (%)] | 0.121 | |||
| 0 | 14 (18.9) | 2 (7.7) | 16 (16.0) | |
| 1 | 50 (67.6) | 16 (61.5) | 66 (66.0) | |
| 2 | 7 (9.5) | 7 (26.9) | 14 (14.0) | |
| 3 | 3 (4.1) | 1 (3.8) | 4 (4.0) | |
| Diabetes [n (%)] | 27 (36.5) | 9(34.6) | 36 (36.0) | 0.864 |
| Hypertension [n (%)] | 40 (54.1) | 13(50.0) | 53 (53.0) | 0.722 |
| Hyperlipidemia [n (%)] | 13 (17.6) | 2 (7.7) | 15 (15.0) | 0.229 |
| Gensini score [Median (Q1, Q3)] | 52.0 (40.0, 80.0) | 64.0 (44.5, 97.0) | 53.5 (40, 82) | 0.377 |
| LVEF [%, Median (Q1, Q3)]) | 58.0 (48.0, 60.0) | 56.5 (43.0, 59.0) | 57.5 (45.5, 60.0) | 0.241 |
| RBC (×1012/L, Mean±SD) | 4.56±0.705 | 4.48±0.743 | 4.54±0.712 | 0.654 |
| N [×109/L, Median (Q1, Q3)] | 6.43 (4.95, 8.10) | 7.05 (4.76, 8.75) | 6.53 (4.94, 8.29) | 0.755 |
| hsCRP [mg/dL, Median (Q1, Q3)] | 5.27 (2.60, 9.79) | 19.9 (2.99, 52.39) | 6.10 (2.75, 26.65) | 0.021 |
| ALB [g/L, Median (Q1, Q3)] | 39.1 (36.10, 41.80) | 35.2 (31.4, 36.8) | 38.7 (35.5, 41.1) | 0.000 |
| UA [μmol/L, Median (Q1, Q3)] | 359 (291, 432) | 379 (348, 439) | 368 (304, 435) | 0.149 |
| Cr [μmol/L, Median (Q1, Q3)] | 69.0 (60.0, 79.0) | 77.5 (63.0, 100.0) | 71.0 (60.0, 82.0) | 0.058 |
| GGT [U/L, Median (Q1, Q3)] | 25.5 (19.0, 39.0) | 37.0 (24.0, 55.0) | 27.0 (19.5, 48.5) | 0.137 |
| Glu [mmol/L, Median (Q1, Q3)] | 5.96 (5.14, 6.89) | 6.00 (5.01, 8.13) | 5.96 (5.11, 7.65) | 0.590 |
| HbA1c [%, Median (Q1, Q3)] | 5.85 (5.50, 7.20) | 5.80 (5.50, 6.80) | 5.80 (5.50, 7.15) | 0.743 |
| LDL-C [mmol/L, Median (Q1, Q3)] | 2.64 (2.02, 3.14) | 2.61 (2.00, 2.96) | 2.64 (2.00, 3.12) | 0.723 |
| HDL-C [mmol/L, Median (Q1, Q3)] | 0.945 (0.820, 1.112) | 0.840 (0.800, 1.013) | 0.935 (0.810, 1.070) | 0.108 |
| Apo(a) [mg/L, Median (Q1, Q3)] | 130 (60, 270) | 169 (63, 395) | 141 (62, 320) | 0.282 |
| CKMB [U/L, Median (Q1, Q3)] | 72.3 (14.7, 193.2) | 33.1 (15.4, 117.2) | 55.8 (15.3, 157.9) | 0.207 |
| NT-proBNP [pg/mL, Median (Q1, Q3)] | 1905 (651, 2949) | 1539 (848, 3313) | 1864 (719, 3041) | 0.782 |
| cTnI [ng/mL, Median (Q1, Q3)] | 33.8 (7.6, 50.0) | 36.3 (8.4, 50.0) | 33.8 (8.1, 50.0) | 0.918 |
| D-dimer [mg/L FEU, Median (Q1, Q3)] | 0.335 (0.210, 0.540) | 0.485 (0.270, 1.620) | 0.405 (0.215, 0.680) | 0.037 |
| FIB [g/L, Median (Q1, Q3)] | 2.80 (2.50, 3.65) | 3.48 (2.54, 5.85) | 2.94 (2.50, 3.96) | 0.034 |
Tab.2 Comparison of baseline clinical data between the MACE group and non-MACE group
| Basic clinical characteristics | Non-MACE (n=74) | MACE (n=26) | Overall (n=100) | P |
|---|---|---|---|---|
| Male [n (%)] | 57 (77.0) | 25 (96.2) | 82 (82.0) | 0.029 |
| Age [year, Median (Q1, Q3)] | 59.5 (46.75, 74.0) | 61.5 (49.0, 77.5) | 60.0 (48.5, 74.0) | 0.494 |
| Days of hospitalization [day, Median (Q1, Q3)] | 9.00 (8.00, 11.0) | 10.5 (9.75, 15.0) | 10.0 (8.00, 12.0) | 0.005 |
| Killip classes [n (%)] | 0.000 | |||
| I | 55 (74.3) | 15 (57.7) | 70 (70.0) | |
| II | 15 (20.3) | 4 (15.4) | 19 (19.0) | |
| III | 4 (5.4) | 1 (3.8) | 5 (5.0) | |
| IV | 0 (0.0) | 6 (23.1) | 6 (6.0) | |
| Body weight [kg, Median (Q1, Q3)] | 70.0 (62.0, 77.6) | 75.0 (65.0, 80.0) | 71.5 (63.3, 79.5) | 0.214 |
| Number of stents [n (%)] | 0.121 | |||
| 0 | 14 (18.9) | 2 (7.7) | 16 (16.0) | |
| 1 | 50 (67.6) | 16 (61.5) | 66 (66.0) | |
| 2 | 7 (9.5) | 7 (26.9) | 14 (14.0) | |
| 3 | 3 (4.1) | 1 (3.8) | 4 (4.0) | |
| Diabetes [n (%)] | 27 (36.5) | 9(34.6) | 36 (36.0) | 0.864 |
| Hypertension [n (%)] | 40 (54.1) | 13(50.0) | 53 (53.0) | 0.722 |
| Hyperlipidemia [n (%)] | 13 (17.6) | 2 (7.7) | 15 (15.0) | 0.229 |
| Gensini score [Median (Q1, Q3)] | 52.0 (40.0, 80.0) | 64.0 (44.5, 97.0) | 53.5 (40, 82) | 0.377 |
| LVEF [%, Median (Q1, Q3)]) | 58.0 (48.0, 60.0) | 56.5 (43.0, 59.0) | 57.5 (45.5, 60.0) | 0.241 |
| RBC (×1012/L, Mean±SD) | 4.56±0.705 | 4.48±0.743 | 4.54±0.712 | 0.654 |
| N [×109/L, Median (Q1, Q3)] | 6.43 (4.95, 8.10) | 7.05 (4.76, 8.75) | 6.53 (4.94, 8.29) | 0.755 |
| hsCRP [mg/dL, Median (Q1, Q3)] | 5.27 (2.60, 9.79) | 19.9 (2.99, 52.39) | 6.10 (2.75, 26.65) | 0.021 |
| ALB [g/L, Median (Q1, Q3)] | 39.1 (36.10, 41.80) | 35.2 (31.4, 36.8) | 38.7 (35.5, 41.1) | 0.000 |
| UA [μmol/L, Median (Q1, Q3)] | 359 (291, 432) | 379 (348, 439) | 368 (304, 435) | 0.149 |
| Cr [μmol/L, Median (Q1, Q3)] | 69.0 (60.0, 79.0) | 77.5 (63.0, 100.0) | 71.0 (60.0, 82.0) | 0.058 |
| GGT [U/L, Median (Q1, Q3)] | 25.5 (19.0, 39.0) | 37.0 (24.0, 55.0) | 27.0 (19.5, 48.5) | 0.137 |
| Glu [mmol/L, Median (Q1, Q3)] | 5.96 (5.14, 6.89) | 6.00 (5.01, 8.13) | 5.96 (5.11, 7.65) | 0.590 |
| HbA1c [%, Median (Q1, Q3)] | 5.85 (5.50, 7.20) | 5.80 (5.50, 6.80) | 5.80 (5.50, 7.15) | 0.743 |
| LDL-C [mmol/L, Median (Q1, Q3)] | 2.64 (2.02, 3.14) | 2.61 (2.00, 2.96) | 2.64 (2.00, 3.12) | 0.723 |
| HDL-C [mmol/L, Median (Q1, Q3)] | 0.945 (0.820, 1.112) | 0.840 (0.800, 1.013) | 0.935 (0.810, 1.070) | 0.108 |
| Apo(a) [mg/L, Median (Q1, Q3)] | 130 (60, 270) | 169 (63, 395) | 141 (62, 320) | 0.282 |
| CKMB [U/L, Median (Q1, Q3)] | 72.3 (14.7, 193.2) | 33.1 (15.4, 117.2) | 55.8 (15.3, 157.9) | 0.207 |
| NT-proBNP [pg/mL, Median (Q1, Q3)] | 1905 (651, 2949) | 1539 (848, 3313) | 1864 (719, 3041) | 0.782 |
| cTnI [ng/mL, Median (Q1, Q3)] | 33.8 (7.6, 50.0) | 36.3 (8.4, 50.0) | 33.8 (8.1, 50.0) | 0.918 |
| D-dimer [mg/L FEU, Median (Q1, Q3)] | 0.335 (0.210, 0.540) | 0.485 (0.270, 1.620) | 0.405 (0.215, 0.680) | 0.037 |
| FIB [g/L, Median (Q1, Q3)] | 2.80 (2.50, 3.65) | 3.48 (2.54, 5.85) | 2.94 (2.50, 3.96) | 0.034 |
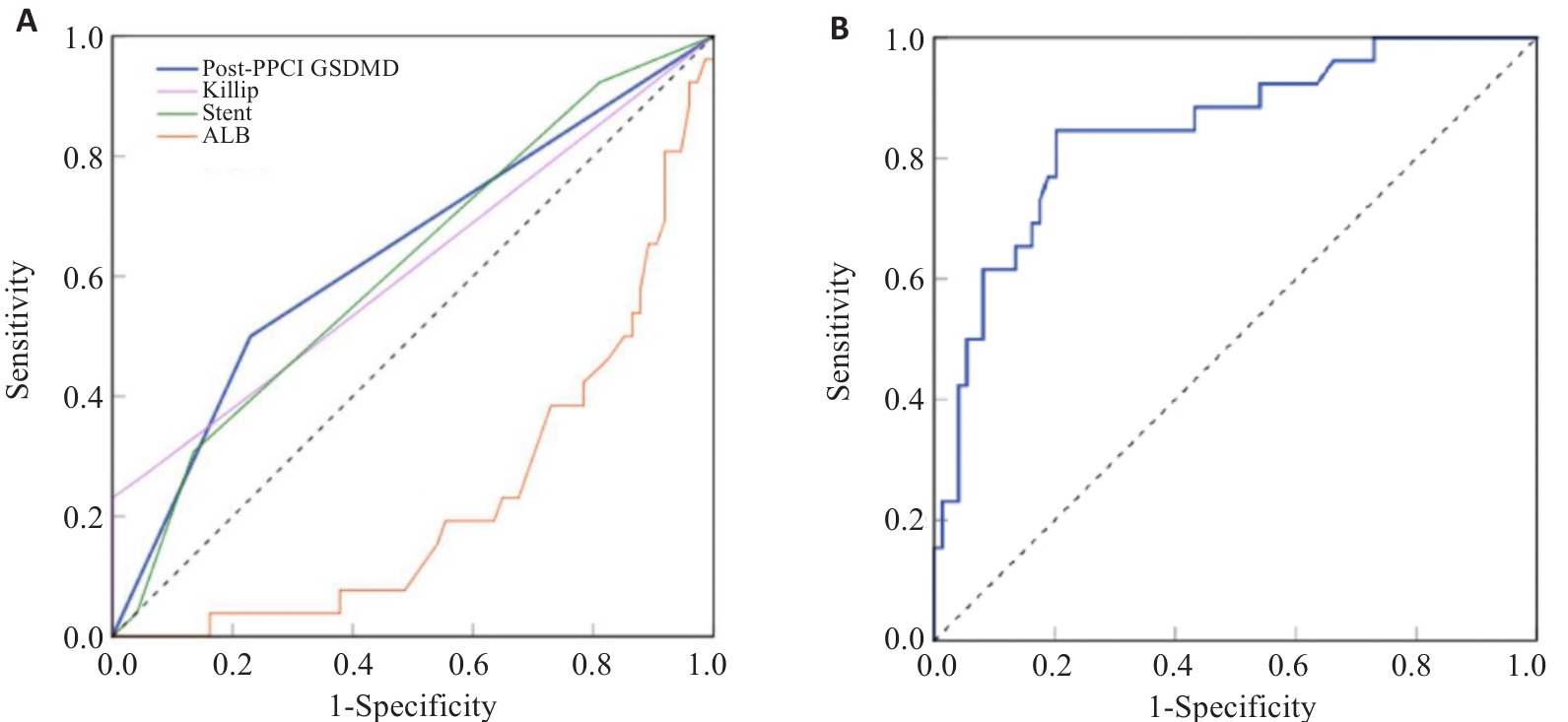
Fig.3 ROC curves for each predictive variable and the nomogram model. A: ROC curve analysis for individual predictors included in the model. Among these predictors, the AUC for GSDMD was 0.635, P=0.041; the AUC for Killip classification was 0.613, P=0.089; the AUC for the number of stents was 0.615, P=0.082; The AUC for albumin was 0.23, P=0.000. B: ROC curve analysis of the nomogram model.
| [1] | Vaduganathan M, Mensah GA, Turco JV, et al. The global burden of cardiovascular diseases and risk: a compass for future health[J]. J Am Coll Cardiol, 2022, 80(25): 2361-71. doi:10.1016/j.jacc.2022.11.005 |
| [2] | Reed GW, Rossi JE, Cannon CP. Acute myocardial infarction[J]. Lancet, 2017, 389(10065): 197-210. doi:10.1016/s0140-6736(16)30677-8 |
| [3] | Iliodromitis K, Seyfarth M, Balogh Z, et al. Anti-inflammatory interventions in coronary artery disease: antipodal responses requiring targeted therapeutic strategies[J]. Basic Res Cardiol, 2025, 120(4): 597-618. doi:10.1007/s00395-025-01121-0 |
| [4] | Barrère-Lemaire S, Vincent A, Jorgensen C, et al. Mesenchymal stromal cells for improvement of cardiac function following acute myocardial infarction: a matter of timing[J]. Physiol Rev, 2024, 104(2): 659-725. doi:10.1152/physrev.00009.2023 |
| [5] | Rao ZP, Zhu YT, Yang P, et al. Pyroptosis in inflammatory diseases and cancer[J]. Theranostics, 2022, 12(9): 4310-29. doi:10.7150/thno.71086 |
| [6] | Burdette BE, Esparza AN, Zhu H, et al. Gasdermin D in pyroptosis[J]. Acta Pharm Sin B, 2021, 11(9): 2768-82. doi:10.1016/j.apsb.2021.02.006 |
| [7] | Toldo S, Abbate A. The role of the NLRP3 inflammasome and pyroptosis in cardiovascular diseases[J]. Nat Rev Cardiol, 2024, 21(4): 219-37. doi:10.1038/s41569-023-00946-3 |
| [8] | Jiang K, Tu ZZ, Chen K, et al. Gasdermin D inhibition confers antineutrophil-mediated cardioprotection in acute myocardial infarction[J]. J Clin Invest, 2022, 132(1): e151268. doi:10.1172/jci151268 |
| [9] | Shi HR, Gao Y, Dong Z, et al. GSDMD-mediated cardiomyocyte pyroptosis promotes myocardial I/R injury[J]. Circ Res, 2021, 129(3): 383-96. doi:10.1161/circresaha.120.318629 |
| [10] | Weng YW, Ye BZ, Lin JH, et al. Elevated circulating levels of gasdermin D are related to acute myocardial infarction and pyrogptosis[J]. BMC Cardiovasc Disord, 2022, 22(1): 554. doi:10.1186/s12872-022-02998-8 |
| [11] | Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction (2018)[J]. Circulation, 2018, 138(20): e618-51. doi:10.1161/cir.0000000000000632 |
| [12] | Moore KJ. Targeting inflammation in CVD: advances and challenges[J]. Nat Rev Cardiol, 2019, 16(2): 74-5. doi:10.1038/s41569-018-0144-3 |
| [13] | Olsen MB, Gregersen I, Sandanger Ø, et al. Targeting the inflammasome in cardiovascular disease[J]. JACC Basic Transl Sci, 2021, 7(1): 84-98. doi:10.1016/j.jacbts.2021.08.006 |
| [14] | Zhou BW, Abbott DW. Chemical modulation of gasdermin D activity: Therapeutic implications and consequences[J]. Semin Immunol, 2023, 70: 101845. doi:10.1016/j.smim.2023.101845 |
| [15] | Ding JJ, Wang K, Liu W, et al. Pore-forming activity and structural autoinhibition of the gasdermin family[J]. Nature, 2016, 535(7610): 111-6. doi:10.1038/nature18590 |
| [16] | Yu P, Zhang X, Liu N, et al. Pyroptosis: mechanisms and diseases[J]. Signal Transduct Target Ther, 2021, 6(1): 128. doi:10.1038/s41392-021-00507-5 |
| [17] | Gu LG, Sun MJ, Li RH, et al. Didymin suppresses microglia pyroptosis and neuroinflammation through the asc/caspase-1/GSDMD pathway following experimental intracerebral hemorrhage[J]. Front Immunol, 2022, 13: 810582. doi:10.3389/fimmu.2022.810582 |
| [18] | He WT, Wan HQ, Hu LC, et al. Gasdermin D is an executor of pyroptosis and required for interleukin-1β secretion[J]. Cell Res, 2015, 25(12): 1285-98. doi:10.1038/cr.2015.139 |
| [19] | Dai Z, Liu WC, Chen XY, et al. Gasdermin D-mediated pyroptosis: mechanisms, diseases, and inhibitors[J]. Front Immunol, 2023, 14: 1178662. doi:10.3389/fimmu.2023.1178662 |
| [20] | Fan XX, Han JB, Zhong LF, et al. Macrophage-derived GSDMD plays an essential role in atherosclerosis and cross talk between macrophages via the mitochondria-STING-IRF3/NF-κB axis[J]. Arterioscler Thromb Vasc Biol, 2024, 44(6): 1365-78. doi:10.1161/atvbaha.123.320612 |
| [21] | Lv JY, Fu ZY, Wang YX, et al. Lingguizhugan decoction ameliorates renal injury secondary to heart failure by improving pyroptosis through TLR4/NF-KB/IRE1α pathway[J]. Phytomedicine, 2025, 143: 156862. doi:10.1016/j.phymed.2025.156862 |
| [22] | Sun WJ, Wang CQ, Cui SH, et al. Association of GSDMD with microvascular-ischemia reperfusion injury after ST-elevation myocardial infarction[J]. Front Cardiovasc Med, 2023, 10: 1138352. doi:10.3389/fcvm.2023.1138352 |
| [23] | Ye XM, Zhang P, Zhang YT, et al. GSDMD contributes to myocardial reperfusion injury by regulating pyroptosis[J]. Front Immunol, 2022, 13: 893914. doi:10.3389/fimmu.2022.893914 |
| [24] | Yanpiset P, Maneechote C, Sriwichaiin S, et al. Gasdermin D-mediated pyroptosis in myocardial ischemia and reperfusion injury: Cumulative evidence for future cardioprotective strategies[J]. Acta Pharm Sin B, 2023, 13(1): 29-53. doi:10.1016/j.apsb.2022.08.007 |
| [25] | Zhang XP, Yang B. The serum levels of gasdermin D in uremic patients and its relationship with the prognosis: a prospective observational cohort study[J]. Ren Fail, 2024, 46(1): 2312534. doi:10.1080/0886022x.2024.2312534 |
| [26] | Wang D, Zhan X, Wu R, et al. Assessment of pyroptosis-related indicators as potential biomarkers and their association with severity in patients with liver cirrhosis[J]. J Inflamm Res, 2021, 14: 3185-96. doi:10.2147/jir.s319213 |
| [27] | Arques S. Human serum albumin in cardiovascular diseases[J]. Eur J Intern Med, 2018, 52: 8-12. doi:10.1016/j.ejim.2018.04.014 |
| [28] | Yoshioka G, Tanaka A, Nishihira K, et al. Prognostic impact of follow-up serum albumin after acute myocardial infarction[J]. ESC Heart Fail, 2021, 8(6): 5456-65. doi:10.1002/ehf2.13640 |
| [29] | Manolis AA, Manolis TA, Melita H, et al. Low serum albumin: a neglected predictor in patients with cardiovascular disease[J]. Eur J Intern Med, 2022, 102: 24-39. doi:10.1016/j.ejim.2022.05.004 |
| [30] | Chen Y, Jiang D, Tao HM, et al. Neutrophils to high-density lipoprotein cholesterol ratio as a new prognostic marker in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention: a retrospective study[J]. BMC Cardiovasc Disord, 2022, 22(1): 434. doi:10.1186/s12872-022-02870-9 |
| [31] | Li XW, Liu Y, Gao MD, et al. Long-term outcomes of 316 patients with STEMI following coronary stent implantation[J]. Am J Transl Res, 2022, 14(6): 4139-45. |
| [32] | Flint C, Cearns M, Opel N, et al. Systematic misestimation of machine learning performance in neuroimaging studies of depression[J]. Neuropsychopharmacology, 2021, 46(8): 1510-7. doi:10.1038/s41386-021-01020-7 |
| [1] | Xiyu GAO, Jing XIAO, Na FENG, Chen GUO, Lifei CAO, Chunyan ZHANG, Yan ZHANG, Tuo HAN. β‑blockers after percutaneous coronary intervention does not reduce risks of all-cause mortality or major adverse cardiovascular events in patients with stable coronary artery disease [J]. Journal of Southern Medical University, 2026, 46(1): 159-165. |
| [2] | Shanyu LUO, Qiang ZHU, Yufei YAN, Zonghong JI, Huajie ZOU, Ruixia ZHANG, Yinggui BA. NLRP3 signaling pathway promotes hepatocyte pyroptosis in mice with nonalcoholic steatohepatitis in hypoxic environment [J]. Journal of Southern Medical University, 2025, 45(9): 2026-2033. |
| [3] | Haiyi ZHOU, Siyi HE, Ruifang HAN, Yongge GUAN, Lijuan DONG, Yang SONG. Moxibustion promotes endometrial repair in rats with thin endometrium by inhibiting the NLRP3/pyroptosis axis via upregulating miR-223-3p [J]. Journal of Southern Medical University, 2025, 45(7): 1380-1388. |
| [4] | Fenlan BIAN, Shiyao NI, Peng ZHAO, Maonanxing QI, Bi TANG, Hongju WANG, Pinfang KANG, Jinjun LIU. Asiaticoside alleviates myocardial ischemia-reperfusion injury in rats by inhibiting NLRP3 inflammasome-mediated pyroptosis [J]. Journal of Southern Medical University, 2025, 45(5): 977-985. |
| [5] | Yalei SUN, Meng LUO, Changsheng GUO, Jing GAO, Kaiqi SU, Lidian CHEN, Xiaodong FENG. Amentoflavone alleviates acute lung injury in mice by inhibiting cell pyroptosis [J]. Journal of Southern Medical University, 2025, 45(4): 692-701. |
| [6] | Zhengwang ZHU, Linlin WANG, Jinghan ZHAO, Ruixue MA, Yuchun YU, Qingchun CAI, Bing WANG, Pingsheng ZHU, Mingsan MIAO. Tuihuang Mixture improves α‑naphthylisothiocyanate-induced cholestasis in rats by inhibiting NLRP3 inflammasomes via regulating farnesoid X receptor [J]. Journal of Southern Medical University, 2025, 45(4): 718-724. |
| [7] | Ju HUANG, Lixia YIN, Minzhu NIU, Zhijun GENG, Lugen ZUO, Jing LI, Jianguo HU. Nodakenin ameliorates TNBS-induced experimental colitis in mice by inhibiting pyroptosis of intestinal epithelial cells [J]. Journal of Southern Medical University, 2025, 45(2): 261-268. |
| [8] | Shicheng XIA, Huifang WEI, Weican HONG, Yuming ZHANG, Feiyang YIN, Yixin ZHANG, Linlin ZHANG, Qin GAO, Hongwei YE. Protective effect of Lonicerae Japonicae Flos extract against doxorubicin-induced myocardial injury in mice and the possible mechanisms [J]. Journal of Southern Medical University, 2025, 45(12): 2527-2540. |
| [9] | Changlong FU, Ruolan CHEN, Shiqi XU, Jinxin YOU, Qing LIN, Yanfeng HUANG. Morinda officinalis polysaccharide delays osteoarthritis mouse chondrocyte degeneration by modulating the glycolysis-pyroptosis axis via targeting the lncRNA XIST [J]. Journal of Southern Medical University, 2025, 45(12): 2541-2550. |
| [10] | Jingyu CHEN, Jinhu ZOU, Bingliang ZHOU, Xuefeng GAO, Pengwei HUANG, Hong CAO. Indole-3-acetic acid alleviates Cryptococcus neoformans-induced pyroptosis in cerebral microvascular endothelial cells by regulating stress granule-mediated NLRP3 inflammasome activation [J]. Journal of Southern Medical University, 2025, 45(12): 2679-2689. |
| [11] | Siyu ZHANG, Linwu RAN, Jin ZENG, Yujiong WANG. Clostridium perfringens Beta1 toxin induces macrophage pyroptosis and ferroptosis through the purinergic receptor P2X7-Ca2+ axis [J]. Journal of Southern Medical University, 2025, 45(10): 2126-2134. |
| [12] | Yiming SUN, Rong ZHANG, Ying MENG, Lei ZHU, Mingqiang LI, Zhe LIU. Coenzyme Q10 alleviates depression-like behaviors in mice with chronic restraint stress by down-regulating the pyroptosis signaling pathway [J]. Journal of Southern Medical University, 2024, 44(5): 810-817. |
| [13] | FANG Shangping, SUN Renke, SU Hui, ZHAI Kecheng, XIANG Yu, GAO Yangmengna, GUO Wenjun. Chlorogenic acid alleviates acute kidney injury in septic mice by inhibiting NLRP3 inflammasomes and the caspase-1 canonical pyroptosis pathway [J]. Journal of Southern Medical University, 2024, 44(2): 317-323. |
| [14] | Duanyi SONG, Yun LI, Xuefang TANG, Hua LI, Kang TAO. Diazepam alleviates pulmonary fibrosis in mice by inhibiting LPS-induced pyroptosis and inflammation via the let-7a-5p/MYD88 axis [J]. Journal of Southern Medical University, 2024, 44(11): 2092-2101. |
| [15] | Li MEI, Lu ZHANG, Di WU, Huanzhang DING, Xinru WANG, Xian ZHANG, Yuhang WEI, Zegeng LI, Jiabing TONG. Liuwei Buqi Formula delays progression of chronic obstructive pulmonary disease in rats by regulating the NLRP3/caspase-1/GSDMD pyroptosis pathway [J]. Journal of Southern Medical University, 2024, 44(11): 2156-2162. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||