Journal of Southern Medical University ›› 2025, Vol. 45 ›› Issue (7): 1451-1459.doi: 10.12122/j.issn.1673-4254.2025.07.11
Previous Articles Next Articles
Jingxian WANG( ), Zijing REN, Peiyang ZHOU(
), Zijing REN, Peiyang ZHOU( )
)
Received:2025-02-22
Online:2025-07-20
Published:2025-07-17
Contact:
Peiyang ZHOU
E-mail:781893244@qq.com;zhoupeiyang@126.com
Jingxian WANG, Zijing REN, Peiyang ZHOU. S1PR5 activation or overexpression enhances barrier function of mouse brain microvascular endothelial cells against OGD/R injury by modulating oxidative stress[J]. Journal of Southern Medical University, 2025, 45(7): 1451-1459.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2025.07.11
| Gene | Targeting sequence (5'-3') |
|---|---|
| siRNA1 | Sense: GCUAUUGCUCGGGGCUGCUATT Antisense: UAGCAGCCCCGAGCAAUAGCTT |
| siRNA2 | Sense: GCUCUACGCCAAGGCCUAUTT Antisense: AUAGGCCUUGGCGUAGAGCTT |
| siRNA3 | Sense: GCUCUACGCCAAGGCCUAUTT Antisense: ACGUGACACGUUCGGAGAATT |
| siNC | Sense: UUCUCCGAACGUGUCACGUTT Antisense: ACGUGACACGUUCGGAGAATT |
Tab.1 S1PR5-specifc siRNA sequences used in this study
| Gene | Targeting sequence (5'-3') |
|---|---|
| siRNA1 | Sense: GCUAUUGCUCGGGGCUGCUATT Antisense: UAGCAGCCCCGAGCAAUAGCTT |
| siRNA2 | Sense: GCUCUACGCCAAGGCCUAUTT Antisense: AUAGGCCUUGGCGUAGAGCTT |
| siRNA3 | Sense: GCUCUACGCCAAGGCCUAUTT Antisense: ACGUGACACGUUCGGAGAATT |
| siNC | Sense: UUCUCCGAACGUGUCACGUTT Antisense: ACGUGACACGUUCGGAGAATT |
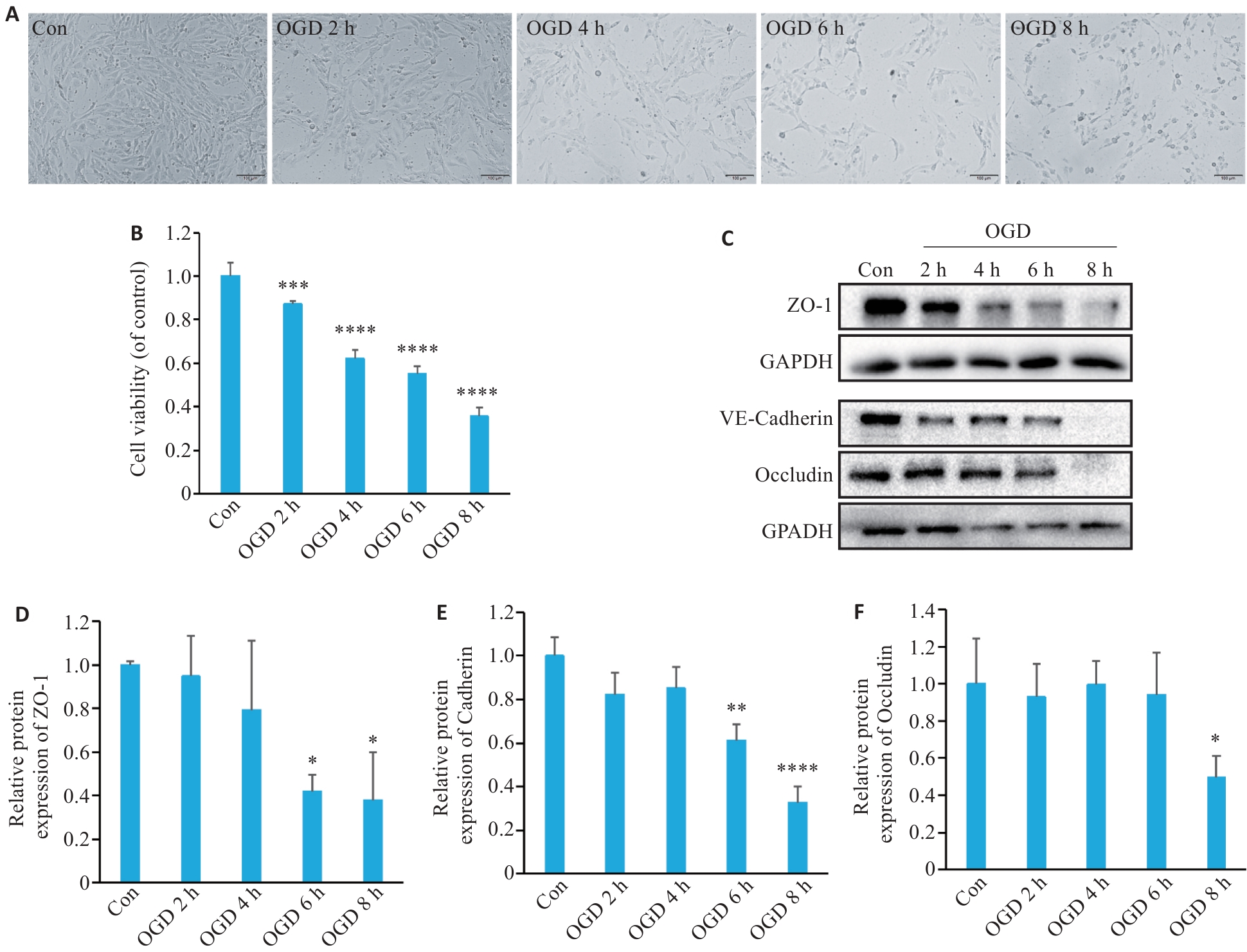
Fig.1 Oxygen-glucose deprivation (OGD) decrease viability of bEnd.3 cells and down-regulate expressions of intercellular tight junction proteins. A: Representative images of bEnd.3 cells after OGD treatment. B: Viability of b.End3 cells assessed with CCK-8 assay (n=6). C: Western blotting of tight junction proteins (ZO-1 and occludin) and VE-cadherin after OGD. D-F: Quantification of ZO-1, VE-cadherin, and occludin protein expression levels (n=3). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 vs Con.
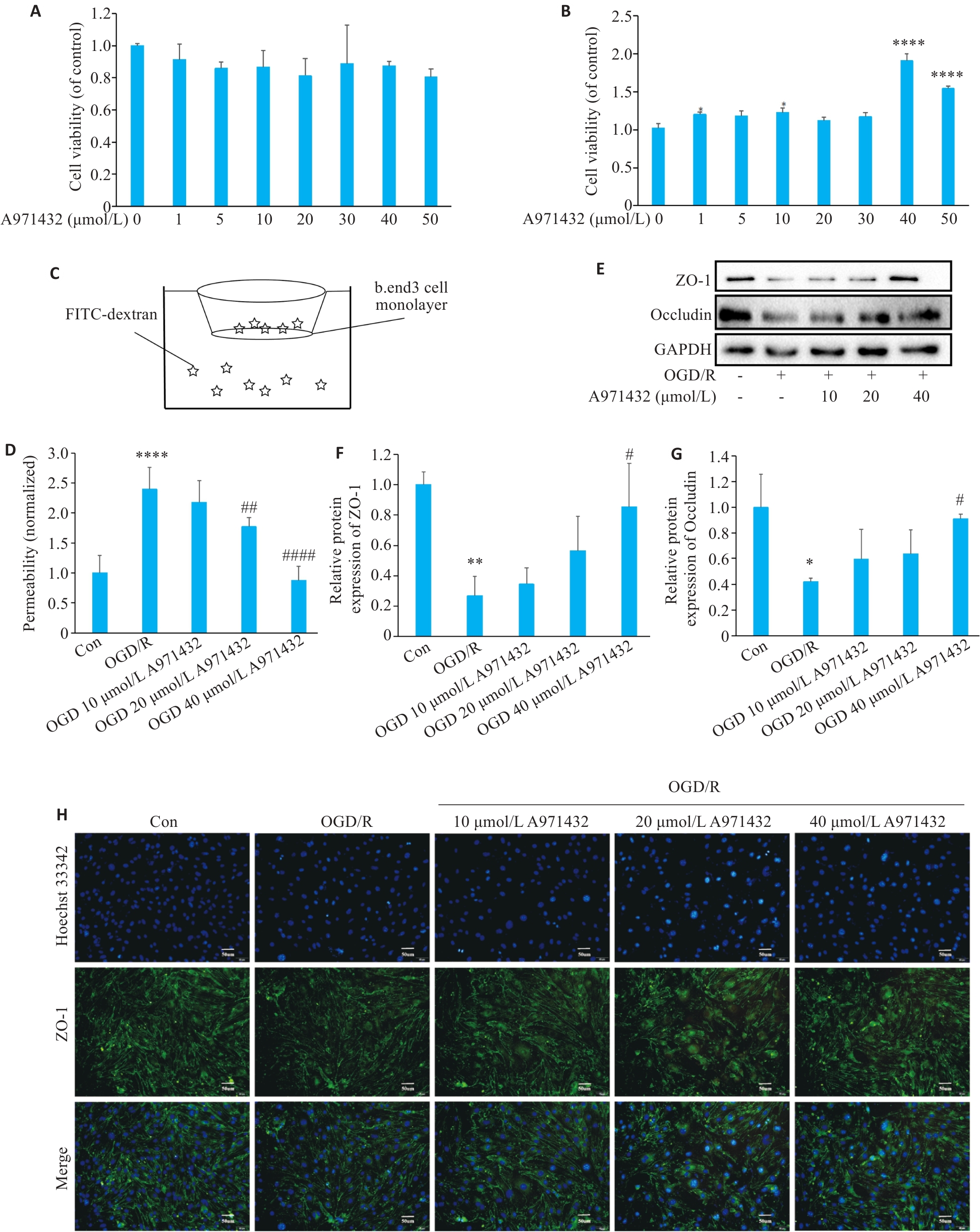
Fig.2 Activation of S1PR5 restores viability of OGD-damaged cells and up-regulates expressions of intercellular tight junction proteins. A: CCK8 assay for assessing viability of cultured bEnd.3 cells treated with different concentrations of A971432. B: CCK8 assay for assessing viability of bEnd.3 cells exposed to OGD after treatment with different concentrations of A971432 (****P<0.0001 vs 0 μmol/L A971432). C: Schematic diagram for detecting endothelial permeability of bEnd.3 cells using FITC-Dextran. D: Detection of endothelial permeability using FITC-Dextran. E: Western blotting of ZO-1 and occluding expressions in bEnd.3 cells with OGD/R after S1PR5 activation. F, G: Quantitative analysis of the expression levels of ZO-1 and occludin. H: Changes in the fluorescence intensity of tight junction proteins caused by activation of S1PR5 in bEnd.3 cells with OGD/R (scale bar= 50 μm). *P<0.05, **P<0.01, ****P<0.0001 vs Con; #P<0.05, ##P<0.01, ####P<0.0001 vs OGD/R.
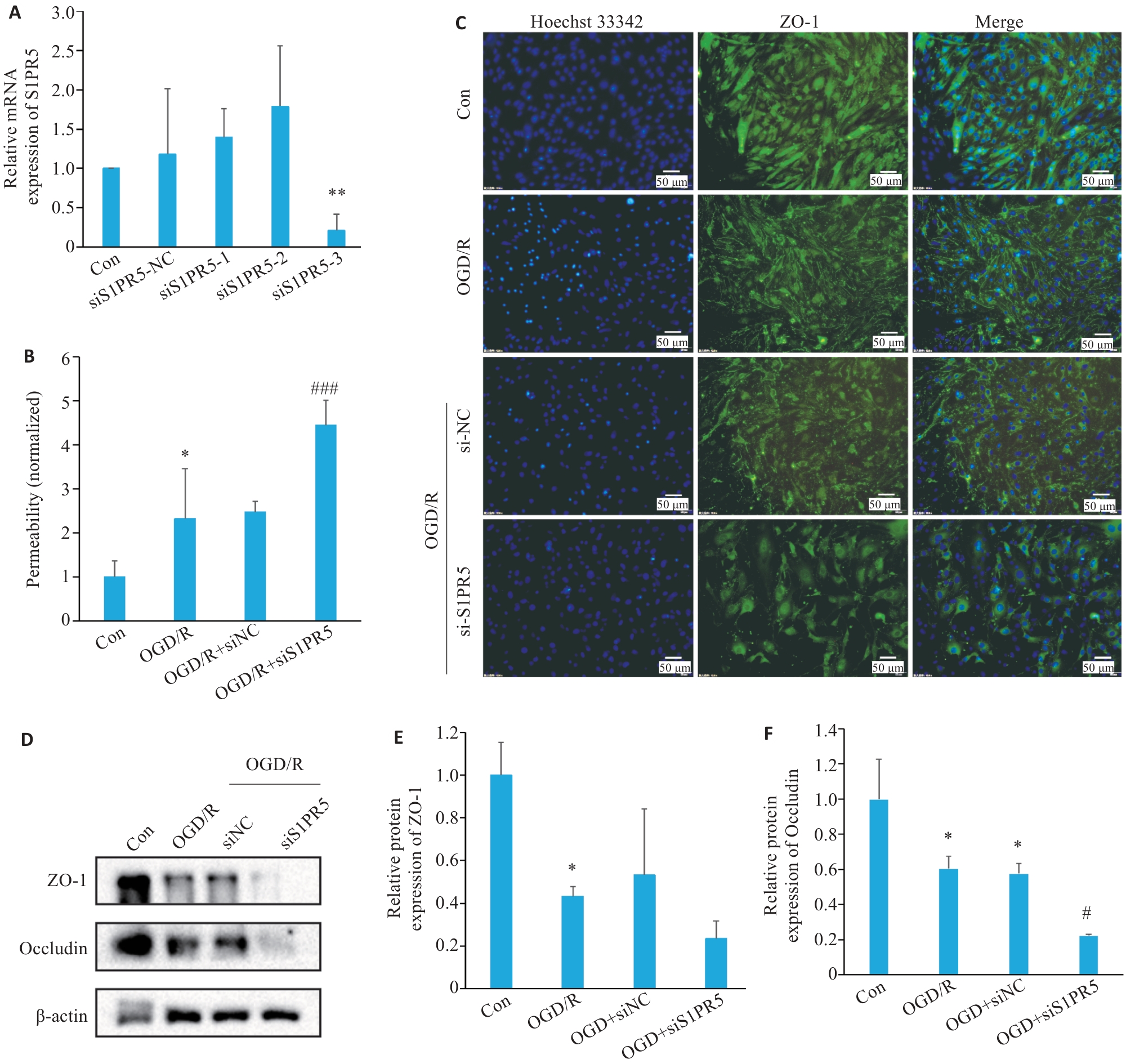
Fig.3 S1PR5 knockdown exacerbates the reduction of cell viability and tight junction protein expressions in OGD-induced bEnd.3 cells. A: Efficiency of S1PR5 knockdown assessed by RT-qPCR. B: Endothelial permeability of the transfected cells detected by FITC-Dextran. C: Alterations in the fluorescence intensity of the tight junction proteins in bEnd.3 cells with S1PR5 knockdown following OGD/R. D-F: Western blotting for detecting the expression levels of ZO-1 and occluding in bEnd.3 cells with OGD/R after S1PR5 knockdown. *P<0.05, **P<0.01 vs Con; #P<0.05, ###P<0.001 vs OGD/R.
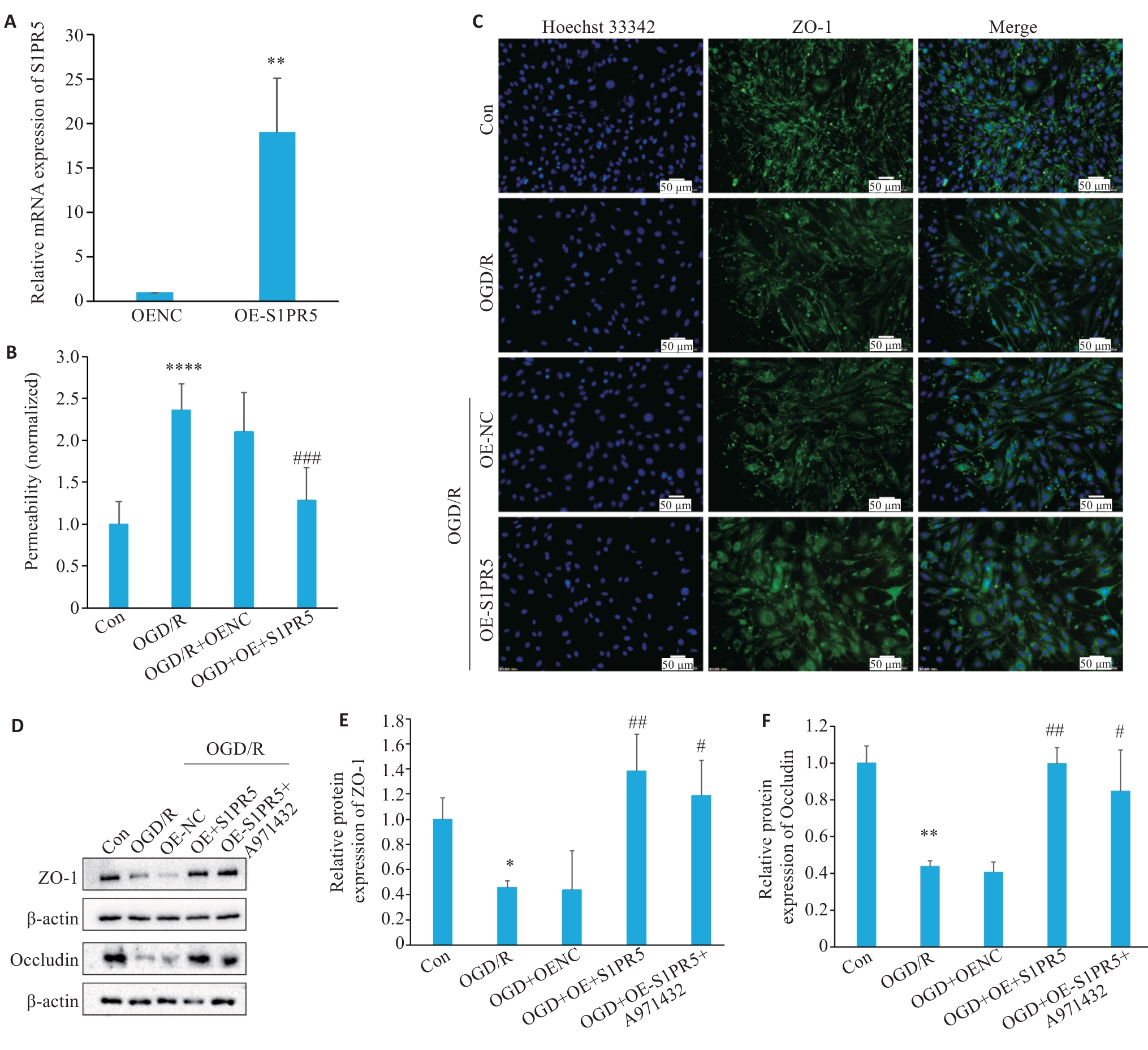
Fig.4 Overexpression of S1PR5 enhances viability and tight junction protein expressions in OGD-induced bEnd.3 cells. A: RT-qPCR for assessing efficiency of lentivirus-mediated S1PR5 overexpression (**P<0.01 vs OENC). B: Endothelial permeability of the cells detected by FITC-Dextran. C: Changes in the fluorescence intensity of tight junction proteins in OGD-induced bEnd.3 cells with S1PR5 overexpression. D-F: Western blotting for detecting expression levels of ZO-1 and occludin in OGD-induced bEnd.3 cells with S1PR5 overexpression. *P<0.05, **P<0.01, ****P<0.0001 vs Con; #P<0.05, ##P<0.01, ###P<0.001 vs OGD/R.
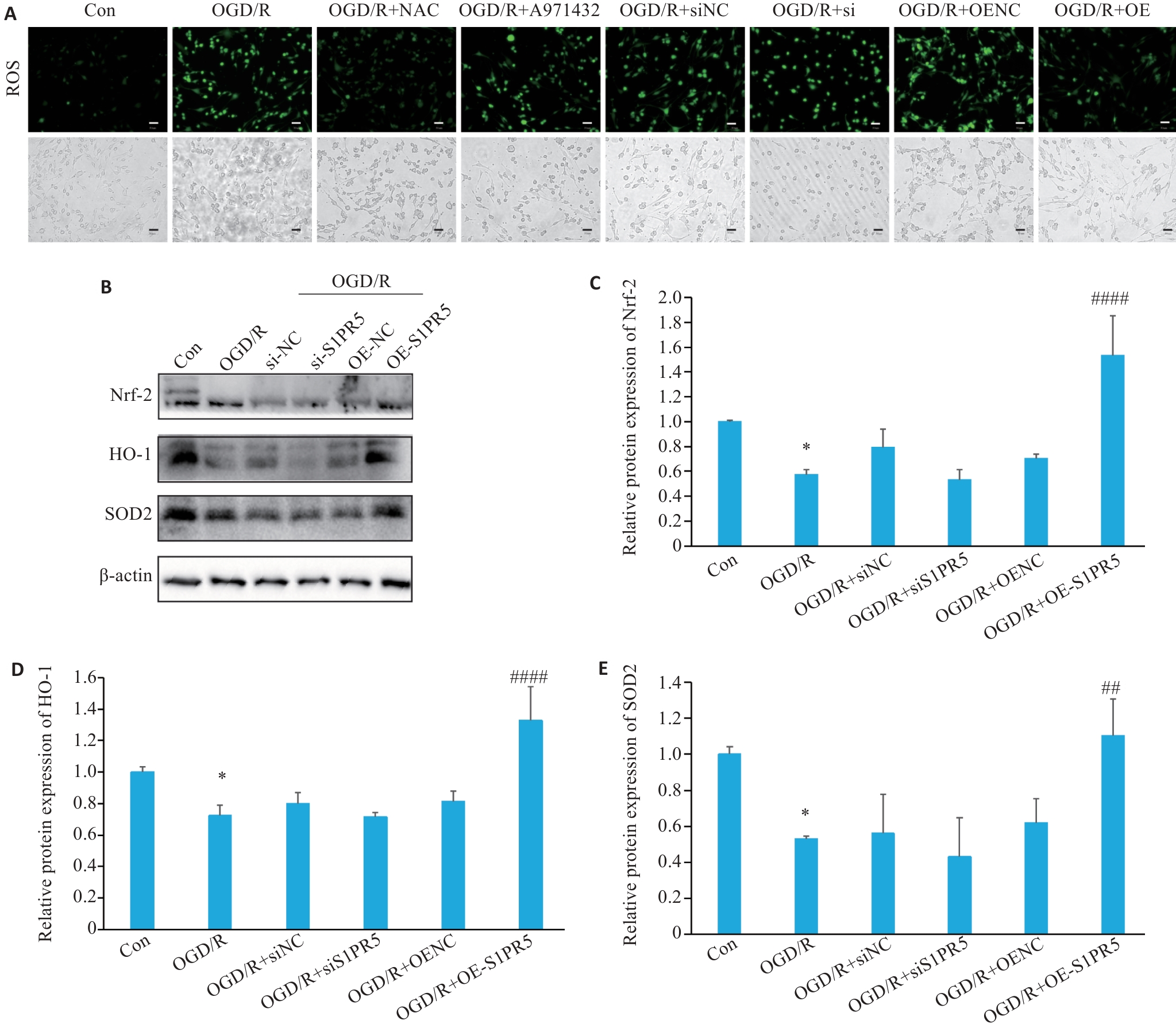
Fig.5 S1PR5 overexpression produces antioxidant effect in OGD-induced bEnd.3 cells via the Nrf-2/HO-1 pathway. A: Effects of S1PR5 knockdown and overexpression on intracellular reactive oxygen species in OGD-induced bEnd.3 cells (scale bar=50 μm). B-E: Western blotting for detecting expression levels of Nrf2, HO-1 and SOD2 in OGD-induced bEnd.3 cells with S1PR5 knockdown or overexpression. *P<0.05 vs Con, ##P<0.01, ####P<0.0001 vs OGD/R.
| [1] | Zhao Z, Nelson AR, Betsholtz C, et al. Establishment and dysfunction of the blood-brain barrier[J]. Cell, 2015, 163(5): 1064-78. doi:10.1016/j.cell.2015.10.067 |
| [2] | Ding QQ, Liu SW, Yao YD, et al. Global, regional, and national burden of ischemic stroke, 1990-2019[J]. Neurology, 2022, 98(3): e279-90. doi:10.1212/wnl.0000000000013115 |
| [3] | Munji RN, Soung AL, Weiner GA, et al. Profiling the mouse brain endothelial transcriptome in health and disease models reveals a core blood-brain barrier dysfunction module[J]. Nat Neurosci, 2019, 22(11): 1892-902. doi:10.1038/s41593-019-0497-x |
| [4] | Cartier A, Hla T. Sphingosine 1-phosphate: Lipid signaling in pathology and therapy[J]. Science, 2019, 366(6463): eaar5551. doi:10.1126/science.aar5551 |
| [5] | Obinata H, Hla T. Sphingosine 1-phosphate and inflammation[J]. Int Immunol, 2019, 31(9): 617-25. doi:10.1093/intimm/dxz037 |
| [6] | van Doorn R, Lopes Pinheiro MA, Kooij G, et al. Sphingosine 1-phosphate receptor 5 mediates the immune quiescence of the human brain endothelial barrier[J]. J Neuroinflammation, 2012, 9: 133. doi:10.1186/1742-2094-9-133 |
| [7] | Di Pardo A, Castaldo S, Amico E, et al. Stimulation of S1PR5 with A-971432, a selective agonist, preserves blood-brain barrier integrity and exerts therapeutic effect in an animal model of Huntington's disease[J]. Hum Mol Genet, 2018, 27(14): 2490-501. doi:10.1093/hmg/ddy153 |
| [8] | 李星阅, 任自敬, 王 越, 等. S1PR5激动剂减轻H2O2诱导的脑微血管内皮细胞高通透性及氧化应激损伤[J]. 医学研究生学报, 2022, 35(8): 820-6. |
| [9] | Matsumoto N, Yamashita T, Shang JW, et al. Up-regulation of sphingosine-1-phosphate receptors and sphingosine kinase 1 in the peri-ischemic area after transient middle cerebral artery occlusion in mice[J]. Brain Res, 2020, 1739: 146831. doi:10.1016/j.brainres.2020.146831 |
| [10] | Cao CC, Dai L, Mu JY, et al. S1PR2 antagonist alleviates oxidative stress-enhanced brain endothelial permeability by attenuating p38 and Erk1/2-dependent cPLA2 phosphorylation[J]. Cell Signal, 2019, 53: 151-61. doi:10.1016/j.cellsig.2018.09.019 |
| [11] | Jiang XY, Andjelkovic AV, Zhu L, et al. Blood-brain barrier dysfunction and recovery after ischemic stroke[J]. Prog Neurobiol, 2018, 163/164: 144-71. doi:10.1016/j.pneurobio.2017.10.001 |
| [12] | Qiu Y, Shen JY, Jiang WL, et al. Sphingosine 1-phosphate and its regulatory role in vascular endothelial cells[J]. Histol Histopathol, 2022, 37(3): 213-25. |
| [13] | Qiao N, An ZH, Fu ZY, et al. Kinsenoside alleviates oxidative stress-induced blood-brain barrier dysfunction via promoting Nrf2/HO-1 pathway in ischemic stroke[J]. Eur J Pharmacol, 2023, 949: 175717. doi:10.1016/j.ejphar.2023.175717 |
| [14] | Prager B, Spampinato SF, Ransohoff RM. Sphingosine 1-phosphate signaling at the blood-brain barrier[J]. Trends Mol Med, 2015, 21(6): 354-63. doi:10.1016/j.molmed.2015.03.006 |
| [15] | Thompson J, Yang YR, Duval K, et al. Progressive disruption of sphingosine-1-phosphate receptor 1 correlates with blood-brain barrier leakage in a rat model of chronic hypoxic hypoperfusion[J]. Aging Dis, 2024, 16(2): 1099-119. |
| [16] | Xiang P, Chew WS, Seow WL, et al. The S1P2 receptor regulates blood-brain barrier integrity and leukocyte extravasation with implications for neurodegenerative disease[J]. Neurochem Int, 2021, 146: 105018. doi:10.1016/j.neuint.2021.105018 |
| [17] | Fan XH, Chen HP, Xu C, et al. S1PR3, as a core protein related to ischemic stroke, is involved in the regulation of blood-brain barrier damage[J]. Front Pharmacol, 2022, 13: 834948. doi:10.3389/fphar.2022.834948 |
| [18] | Wu JL, Liang Y, Fu PF, et al. Sphingosine-1-phosphate receptor 3 induces endothelial barrier loss via ADAM10-mediated vascular endothelial-cadherin cleavage[J]. Int J Mol Sci, 2023, 24(22): 16083. doi:10.3390/ijms242216083 |
| [19] | Hansen LN, Lohfink N, Vutukuri R, et al. Endothelial sphingosine-1-phosphate receptor 4 regulates blood-brain barrier permeability and promotes a homeostatic endothelial phenotype[J]. J Neurosci, 2022, 42(10): 1908-29. doi:10.1523/jneurosci.0188-21.2021 |
| [20] | Spampinato SF, Merlo S, Sano Y, et al. Protective effect of the sphingosine-1 phosphate receptor agonist siponimod on disrupted blood brain barrier function[J]. Biochem Pharmacol, 2021, 186: 114465. doi:10.1016/j.bcp.2021.114465 |
| [21] | Wang ZF, Higashikawa K, Yasui H, et al. FTY720 protects against ischemia-reperfusion injury by preventing the redistribution of tight junction proteins and decreases inflammation in the subacute phase in an experimental stroke model[J]. Transl Stroke Res, 2020, 11(5): 1103-16. doi:10.1007/s12975-020-00789-x |
| [22] | Yang CJ, Hawkins KE, Doré S, et al. Neuroinflammatory mechanisms of blood-brain barrier damage in ischemic stroke[J]. Am J Physiol Cell Physiol, 2019, 316(2): C135-53. doi:10.1152/ajpcell.00136.2018 |
| [23] | Gao CL, Xu YZ, Liang ZZ, et al. A novel PGAM5 inhibitor LFHP-1c protects blood-brain barrier integrity in ischemic stroke[J]. Acta Pharm Sin B, 2021, 11(7): 1867-84. doi:10.1016/j.apsb.2021.01.008 |
| [24] | Li Y, Liao J, Xiong LY, et al. Stepwise targeted strategies for improving neurological function by inhibiting oxidative stress levels and inflammation following ischemic stroke[J]. J Control Release, 2024, 368: 607-22. doi:10.1016/j.jconrel.2024.02.039 |
| [25] | Sun YY, Zhu HJ, Zhao RY, et al. Remote ischemic conditioning attenuates oxidative stress and inflammation via the Nrf2/HO-1 pathway in MCAO mice[J]. Redox Biol, 2023, 66: 102852. doi:10.1016/j.redox.2023.102852 |
| [26] | Martín-Montañez E, Pavia J, Valverde N, et al. The S1P mimetic fingolimod phosphate regulates mitochondrial oxidative stress in neuronal cells[J]. Free Radic Biol Med, 2019, 137: 116-30. doi:10.1016/j.freeradbiomed.2019.04.022 |
| [27] | Colombo E, Bassani C, De Angelis A, et al. Siponimod (BAF312) activates Nrf2 while hampering NFκB in human astrocytes, and protects from astrocyte-induced neurodegeneration[J]. Front Immunol, 2020, 11: 635. doi:10.3389/fimmu.2020.00635 |
| [28] | Chen HW, Chen K, Huang WJ, et al. Structure of S1PR2-hetero-trimeric G13 signaling complex[J]. Sci Adv, 2022, 8(13): eabn0067. doi:10.1126/sciadv.abn0067 |
| [29] | Bellezza I, Giambanco I, Minelli A, et al. Nrf2-Keap1 signaling in oxidative and reductive stress[J]. Biochim Biophys Acta Mol Cell Res, 2018, 1865(5): 721-33. doi:10.1016/j.bbamcr.2018.02.010 |
| [30] | Fan W, Chen HP, Li M, et al. NRF2 activation ameliorates blood-brain barrier injury after cerebral ischemic stroke by regulating ferroptosis and inflammation[J]. Sci Rep, 2024, 14(1): 5300. doi:10.1038/s41598-024-53836-0 |
| [1] | Anbang ZHANG, Xiuqi SUN, Bo PANG, Yuanhua WU, Jingyu SHI, Ning ZHANG, Tao YE. Electroacupuncture pretreatment alleviates cerebral ischemia-reperfusion injury in rats by inhibiting ferroptosis through the gut-brain axis and the Nrf2/HO-1 signaling pathway [J]. Journal of Southern Medical University, 2025, 45(5): 911-920. |
| [2] | Zhi GAO, Ao WU, Zhongxiang HU, Peiyang SUN. Bioinformatics analysis of oxidative stress and immune infiltration in rheumatoid arthritis [J]. Journal of Southern Medical University, 2025, 45(4): 862-870. |
| [3] | Pengwei HUANG, Jie CHEN, Jinhu ZOU, Xuefeng GAO, Hong CAO. Quercetin mitigates HIV-1 gp120-induced rat astrocyte neurotoxicity via promoting G3BP1 disassembly in stress granules [J]. Journal of Southern Medical University, 2025, 45(2): 304-312. |
| [4] | Yuming ZHANG, Shicheng XIA, Linlin ZHANG, Mengxi CHEN, Xiaojing LIU, Qin GAO, Hongwei YE. Protective effect of Lonicerae japonicae flos extract against doxorubicin-induced liver injury in mice [J]. Journal of Southern Medical University, 2024, 44(8): 1571-1581. |
| [5] | Zhijun REN, Jianxin DIAO, Yiting WANG. Xionggui Decoction alleviates heart failure in mice with myocardial infarction by inhibiting oxidative stress-induced cardiomyocyte apoptosis [J]. Journal of Southern Medical University, 2024, 44(7): 1416-1424. |
| [6] | Guoxin LIANG, Hongyue TANG, Chang GUO, Mingming ZHANG. MiR-224-5p overexpression inhibits oxidative stress by regulating the PI3K/Akt/FoxO1 axis to attenuate hypoxia/reoxygenation-induced cardiomyocyte injury [J]. Journal of Southern Medical University, 2024, 44(6): 1173-1181. |
| [7] | CHEN Guodong, LUO Suxin. Colchicine alleviates myocardial ischemia-reperfusion injury in mice by activating AMPK [J]. Journal of Southern Medical University, 2024, 44(2): 226-235. |
| [8] | LING Xuguang, XU Wenwen, PANG Guanlai, HONG Xuxing, LIU Fengqin, LI Yang. Tea polyphenols ameliorates acute lung injury in septic mice by inhibiting NLRP3 inflammasomes [J]. Journal of Southern Medical University, 2024, 44(2): 381-386. |
| [9] | SUN Xiaopeng, SHI Hang, ZHANG Lei, LIU Zhong, LI Kewei, QIAN Lingling, ZHU Xingyu, YANG Kangjia, FU Qiang, DING Hua. Exosomes from ectoderm mesenchymal stem cells inhibits lipopolysaccharide-induced microglial M1 polarization and promotes survival of H2O2-exposed PC12 cells by suppressing inflammatory response and oxidative stress [J]. Journal of Southern Medical University, 2024, 44(1): 119-128. |
| [10] | YE Hongwei, ZHANG Yuming, YUN Qi, DU Ruoli, LI Lu, LI Yuping, GAO Qin. Resveratrol alleviates hyperglycemia-induced cardiomyocyte hypertrophy by maintaining mitochondrial homeostasis via enhancing SIRT1 expression [J]. Journal of Southern Medical University, 2024, 44(1): 45-51. |
| [11] | MENG Nuo, YANG Huiying, CHEN Jinfeng, QIN Yingying, LEI Yu, HUANG Zhenning, TANG Guodu. Honokiol reduces oxidative stress by activating the SIRT3-MnSOD2 pathway to alleviate hypertriglyceridemia-induced acute pancreatitis in rats [J]. Journal of Southern Medical University, 2023, 43(3): 405-411. |
| [12] | CHEN Xing, WANG Kaiwan, CHU Dehai, ZHU Yu, ZHANG Wenbing, CAO Huiping, XIE Wenyu, LU Chuanhao, LI Xia. Forsythiaside B inhibits cerebral ischemia/reperfusion-induced oxidative stress injury in mice via the AMPK/DAF-16/FOXO3 pathway [J]. Journal of Southern Medical University, 2023, 43(2): 199-205. |
| [13] | ZHENG Dongxiao, CHEN Linlin, WEI Qihui, ZHU Ziran, LIU Zilue, JIN Lin, YANG Guanyu, XIE Xi. Fucoxanthin regulates Nrf2/Keap1 signaling to alleviate myocardial hypertrophy in diabetic rats [J]. Journal of Southern Medical University, 2022, 42(5): 752-759. |
| [14] | ZHU Yanyan, WANG Tongsheng, DAI Ning, DENG Mengyun, LIU Hongjuan, TONG Xiaohui, LI Li. Hyperoside protects mouse spermatocytes GC-2 cells from oxidative damage by activating the Keap1/Nrf2/HO-1 pathway [J]. Journal of Southern Medical University, 2022, 42(5): 673-680. |
| [15] | SHAO Xiaoli, YU Jiangyi, NI Weihui. Wogonoside alleviates high glucose-induced dysfunction of retinal microvascular endothelial cells and diabetic retinopathy in rats by up-regulating SIRT1 [J]. Journal of Southern Medical University, 2022, 42(4): 463-472. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||