Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (3): 489-496.doi: 10.12122/j.issn.1673-4254.2026.03.02
Zhuoning ZHANG1( ), Xinyu HAO1(
), Xinyu HAO1( ), Fuyang CAO2, Yongxin GUO1, Shuting GUO1, Chen CAI1, Weidong MI1(
), Fuyang CAO2, Yongxin GUO1, Shuting GUO1, Chen CAI1, Weidong MI1( ), Li TONG1(
), Li TONG1( )
)
Received:2025-09-09
Online:2026-03-20
Published:2026-03-26
Contact:
Weidong MI, Li TONG
E-mail:zhangzhuoning@aliyun.com;haoxinyu19980206@163.com;wwdd1962@163.com;tongli301@aliyun.com
Supported by:Zhuoning ZHANG, Xinyu HAO, Fuyang CAO, Yongxin GUO, Shuting GUO, Chen CAI, Weidong MI, Li TONG. Esketamine alleviates depression-like behaviors in mice with chronic restraint stress by activating glutamatergic neurons in the medial prefrontal cortex[J]. Journal of Southern Medical University, 2026, 46(3): 489-496.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.03.02
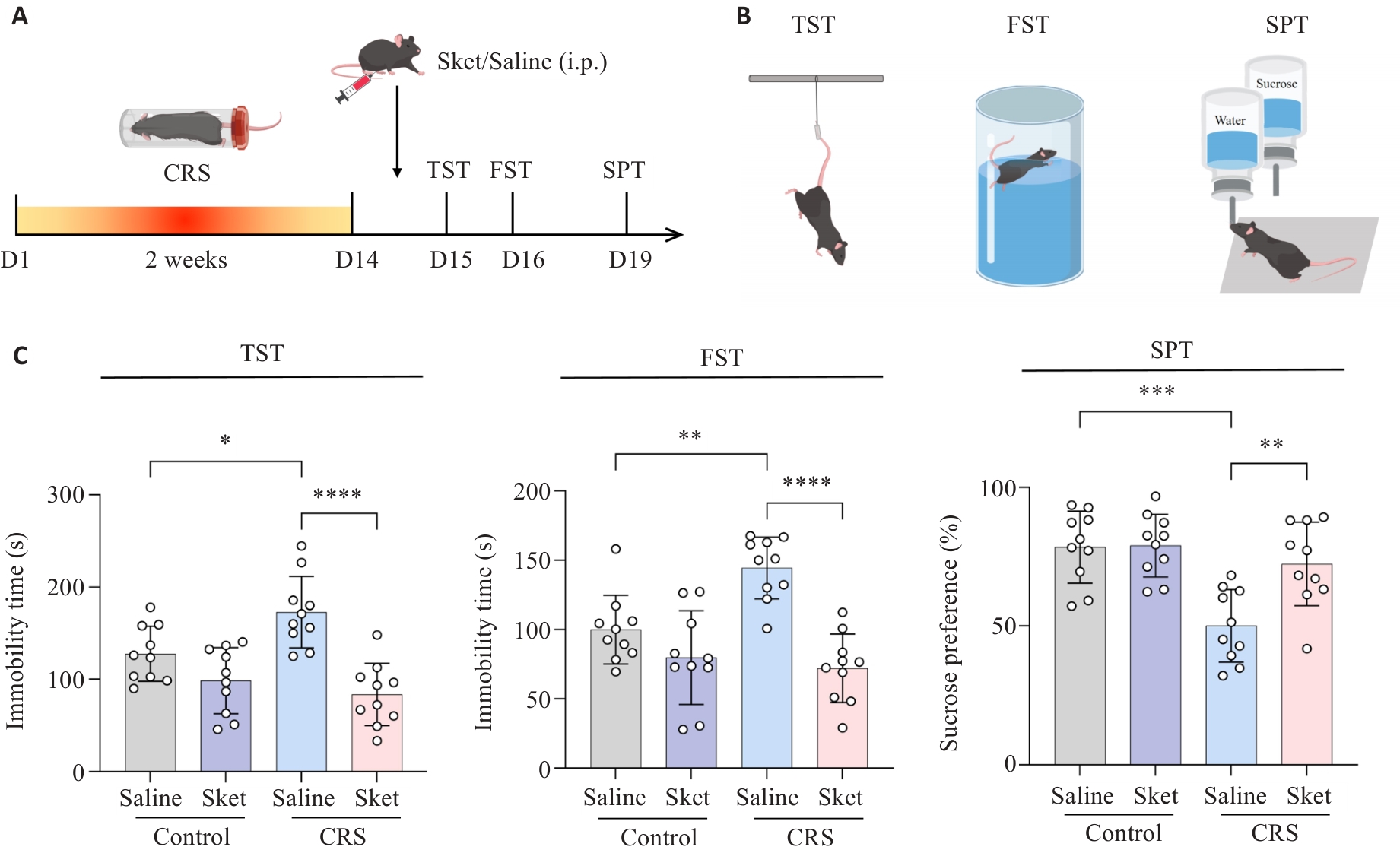
Fig.1 Effects of esketamine on depressive-like behaviors in mice with chronic restraint stress (CRS) (n=40). A: Flowchart showing the establishment of CRS models in mice and the schedule of behavioral tests. B: Tail Suspension Test (TST), Forced Swim Test (FST) and Sucrose Preference Test (SPT) of the mice for assessing depressive-like behaviors of the mice. C: Immobility time of the mice in TST and FST and sucrose preference in SPT. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
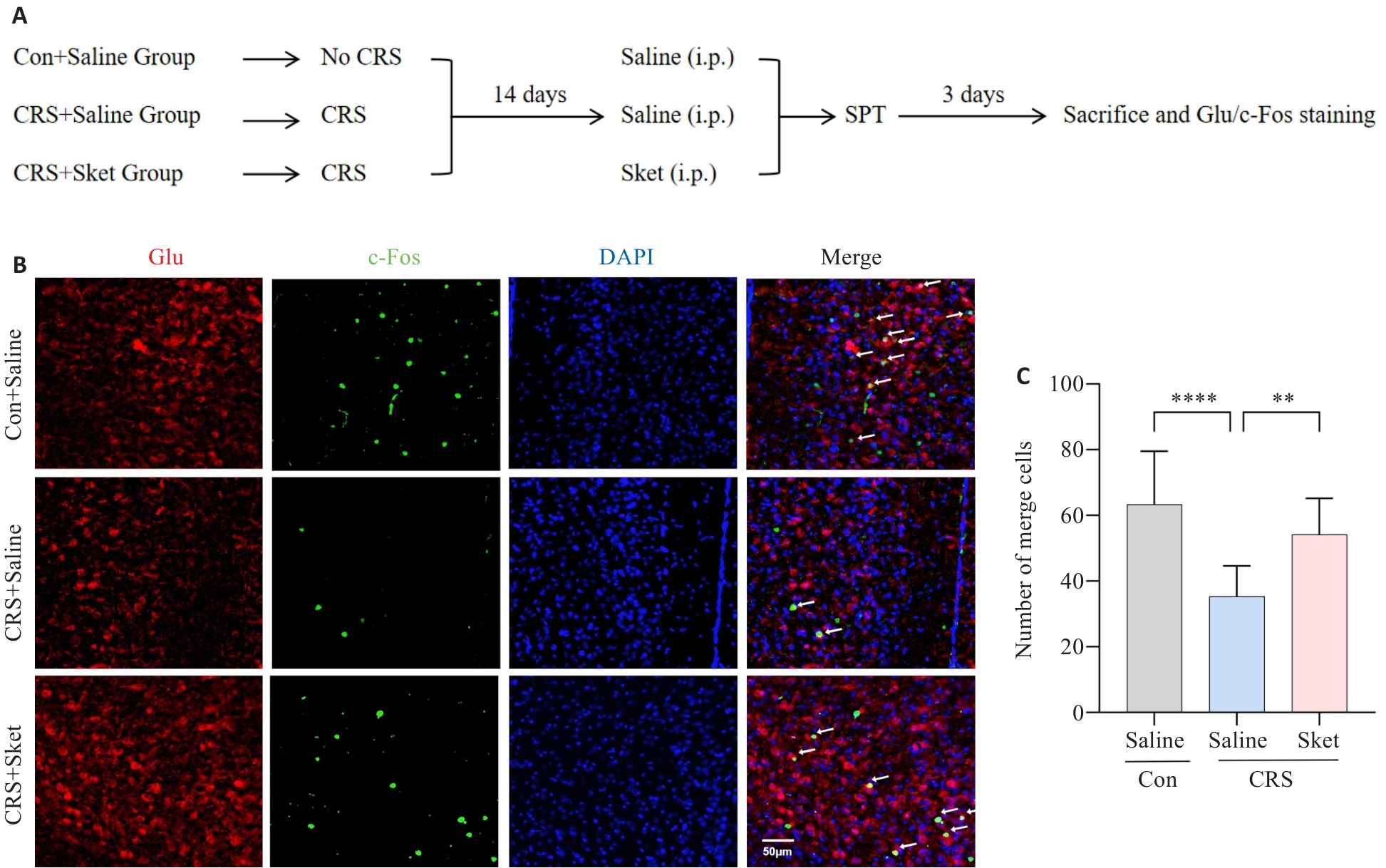
Fig.2 Esketamine activates CaMKII subtype glutamatergic neurons in the medial prefrontal cortex (mPFC) of CRS mice (n=15). A: Procedure of modeling and tissue collection. B: Representative images of c-Fos immunofluorescence staining and glutamatergic neurons in the mPFC. C: Statistical analysis of c-Fos neurons co-localized with glutamatergic neurons in the mPFC of the mice. **P<0.01, ****P<0.0001.
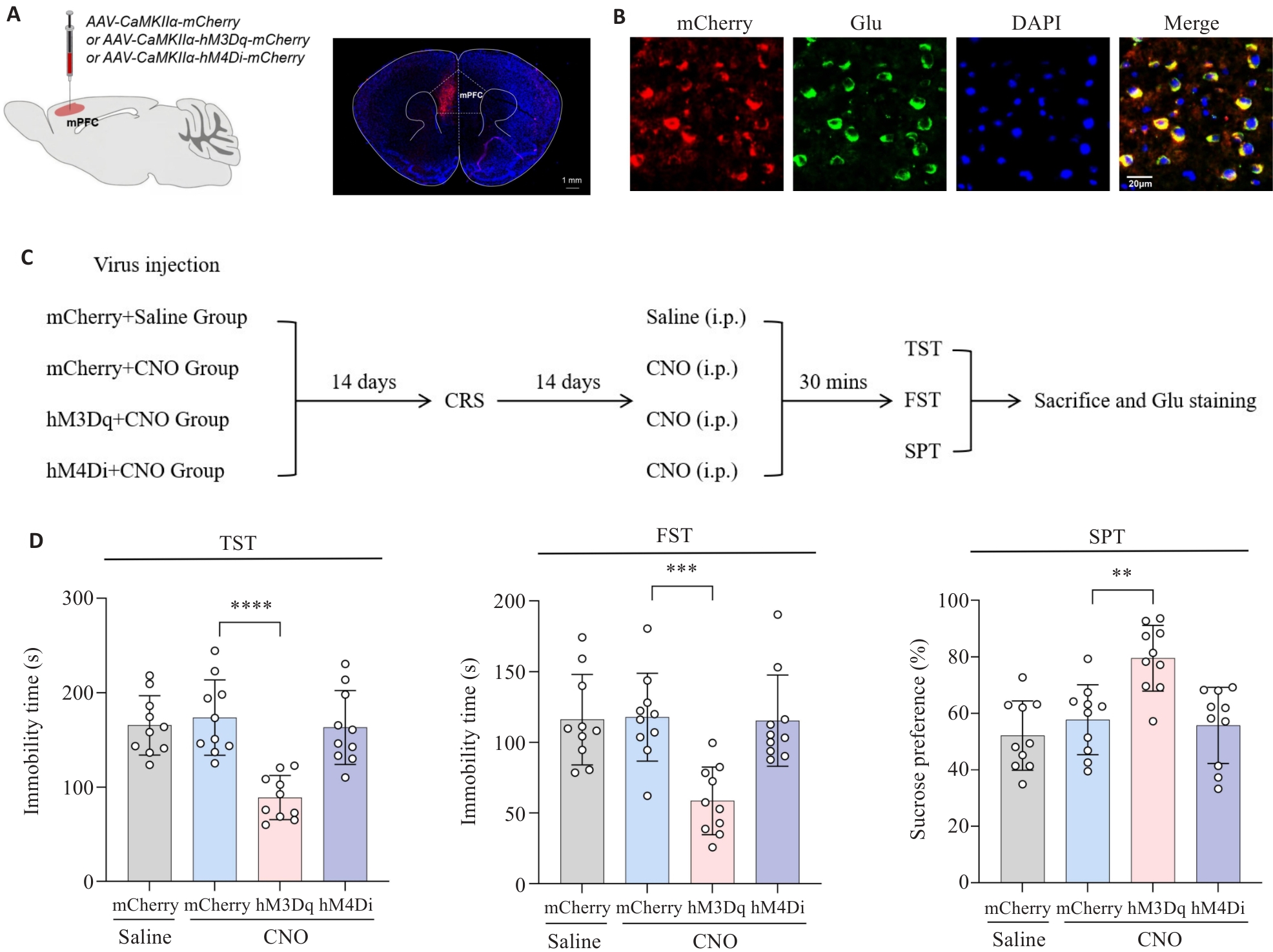
Fig.3 Effect of chemogenetic regulation of glutamatergic neuron activity in the mPFC on depressive-like behaviors of CRS mice (n=15). A: Illustration of virus injection and a coronal brain section showing virus expression in the mPFC. B: Expression of chemogenetic virus in the glutamatergic neurons. The mPFC was co-labeled with mCherry red fluorescence, Glu green immunofluorescence, and DAPI blue fluorescence. C: Procedure of virus injection and behavioral tests. D: Immobility time of the mice in TST/FST and sucrose preference in the SPT. **P<0.01, ***P<0.001, ****P<0.0001.
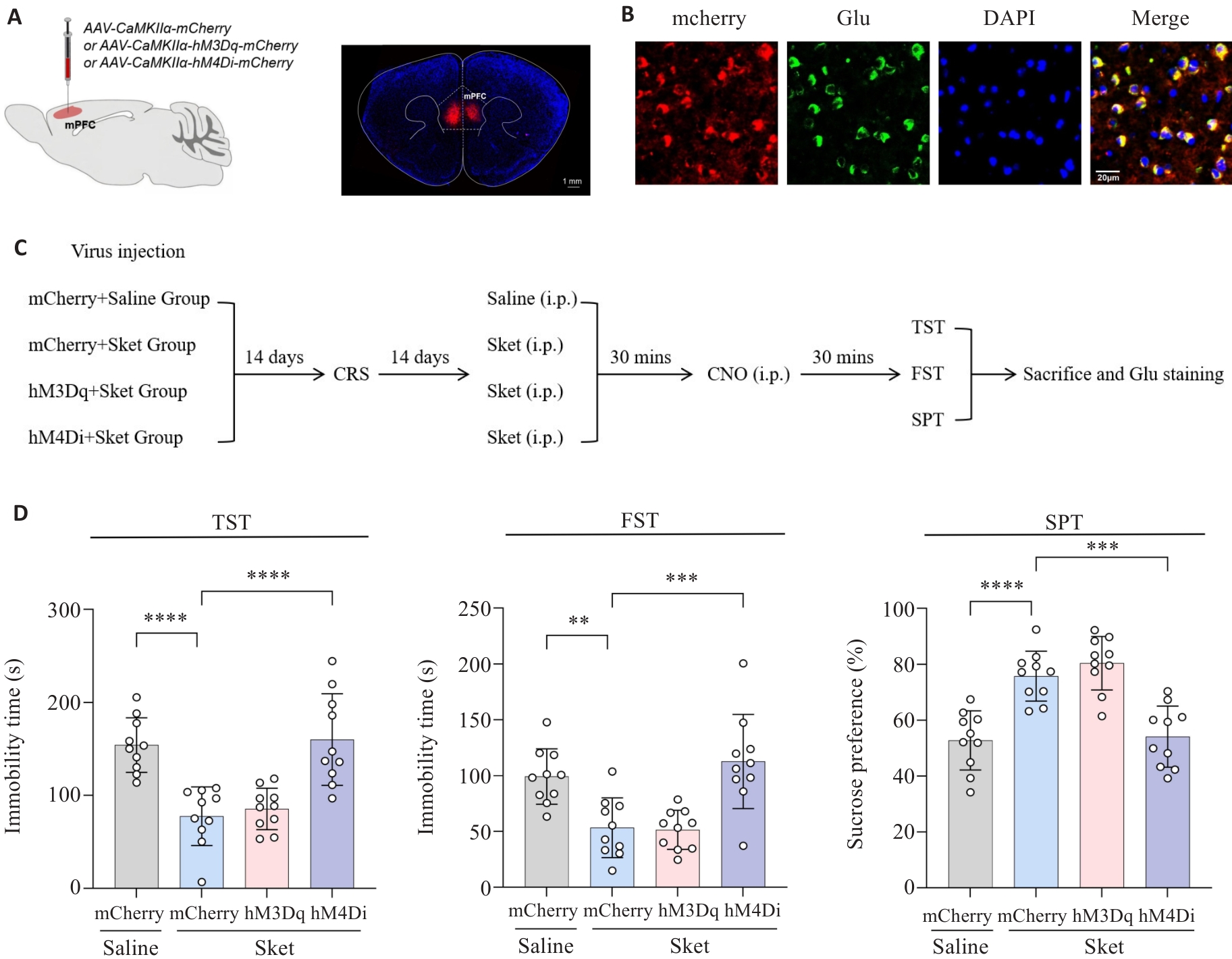
Fig.4 Esketamine alleviates depressive-like behaviors of the mice by activating CaMKIIα glutamatergic neurons in the mPFC (n=40). A: Illustration of a virus injection and a coronal brain section demonstrating virus expression in the mPFC. B: Expression of chemogenetic virus in the glutamatergic neurons. C: Procedure of virus injection and behavioral tests. D: Immobility time in the TST/FST and the sucrose preference in the SPT. **P<0.01, ***P<0.001, ****P<0.0001.
| [1] | Lu J, Xu XF, Huang YQ, et al. Prevalence of depressive disorders and treatment in China: a cross-sectional epidemiological study[J]. Lancet Psychiatry, 2021, 8(11): 981-90. doi:10.1016/s2215-0366(21)00251-0 |
| [2] | Kim JW, Suzuki K, Kavalali ET, et al. Ketamine: mechanisms and relevance to treatment of depression[J]. Annu Rev Med, 2024, 75: 129-43. doi:10.1146/annurev-med-051322-120608 |
| [3] | Anderer S. Esketamine monotherapy effective for treatment-resistant depression[J]. JAMA, 2025, 334(8): 661-2. doi:10.1001/jama.2025.10978 |
| [4] | 中华医学会精神医学分会抑郁障碍研究协作组,中国麻醉药品协会精神卫生分会. 艾司氯胺酮治疗抑郁症临床应用专家共识(2025)[J]. 中华精神科杂志, 2025, 58(6): 446-60. doi:10.3760/cma.j.cn113661-20250116-00026 |
| [5] | Bonaventura J, Lam S, Carlton M, et al. Pharmacological and behavioral divergence of ketamine enantiomers: implications for abuse liability[J]. Mol Psychiatry, 2021, 26(11): 6704-22. doi:10.1038/s41380-021-01093-2 |
| [6] | Qin H, Yu M, Han NJ, et al. Antidepressant effects of esketamine via the BDNF/AKT/mTOR pathway in mice with postpartum depression and their offspring[J]. Prog Neuro Psychopharmacol Biol Psychiatry, 2024, 132: 110992. doi:10.1016/j.pnpbp.2024.110992 |
| [7] | Sarrazin DH, Gardner W, Marchese C, et al. Prefrontal cortex molecular clock modulates development of depression-like phenotype and rapid antidepressant response in mice[J]. Nat Commun, 2024, 15(1): 7257. doi:10.1038/s41467-024-51716-9 |
| [8] | Hare BD, Duman RS. Prefrontal cortex circuits in depression and anxiety: contribution of discrete neuronal populations and target regions[J]. Mol Psychiatry, 2020, 25(11): 2742-58. doi:10.1038/s41380-020-0685-9 |
| [9] | Liu WZ, Zhang WH, Zheng ZH, et al. Identification of a prefrontal cortex-to-amygdala pathway for chronic stress-induced anxiety[J]. Nat Commun, 2020, 11(1): 2221. doi:10.1038/s41467-020-15920-7 |
| [10] | Kennedy SH, Konarski JZ, Segal ZV, et al. Differences in brain glucose metabolism between responders to CBT and venlafaxine in a 16-week randomized controlled trial[J]. Am J Psychiatry, 2007, 164(5): 778-88. doi:10.1176/appi.ajp.164.5.778 |
| [11] | Lajoie C, Levasseur MA, Paquet N. Complete normalization of severe brain 18F-FDG hypometabolism following electroconvulsive therapy in a major depressive episode[J]. Clin Nucl Med, 2013, 38(9): 735-6. doi:10.1097/rlu.0b013e31829b9bd9 |
| [12] | Lei Y, Wang J, Wang D, et al. SIRT1 in forebrain excitatory neurons produces sexually dimorphic effects on depression-related behaviors and modulates neuronal excitability and synaptic transmission in the medial prefrontal cortex[J]. Mol Psychiatry, 2020, 25(5): 1094-111. doi:10.1038/s41380-019-0352-1 |
| [13] | Son H, Baek JH, Go BS, et al. Glutamine has antidepressive effects through increments of glutamate and glutamine levels and glutamatergic activity in the medial prefrontal cortex[J]. Neuropharmacology, 2018, 143: 143-52. doi:10.1016/j.neuropharm.2018.09.040 |
| [14] | Chen JY, Wu K, Guo MM, et al. The PrLGlu→avBNSTGABA circuit rapidly modulates depression-like behaviors in male mice[J]. iScience, 2023, 26(10): 107878. doi:10.1016/j.isci.2023.107878 |
| [15] | Maes M, Almulla AF, You Z, et al. Neuroimmune, metabolic and oxidative stress pathways in major depressive disorder[J]. Nat Rev Neurol, 2025, 21(9): 473-89. doi:10.1038/s41582-025-01116-4 |
| [16] | Xu M, Zhang Z, Zhang Z, et al. The pathogenesis and medical treatment of depression: opportunity and challenge[J]. Neurol Int, 2025, 17(8): 120. doi:10.3390/neurolint17080120 |
| [17] | 尹一淑, 刘军莲, 王佳平, 等. 抑郁症相关发病机制研究进展[J].医学综述, 2022, 28(12): 2368-72. |
| [18] | Parekh PK, Johnson SB, Liston C. Synaptic mechanisms regulating mood state transitions in depression[J]. Annu Rev Neurosci, 2022, 45: 581-601. doi:10.1146/annurev-neuro-110920-040422 |
| [19] | Lammel S, Tye KM, Warden MR. Progress in understanding mood disorders: optogenetic dissection of neural circuits[J]. Genes Brain Behav, 2014, 13(1): 38-51. doi:10.1111/gbb.12049 |
| [20] | Belleau EL, Treadway MT, Pizzagalli DA. The impact of stress and major depressive disorder on hippocampal and medial prefrontal cortex morphology[J]. Biol Psychiatry, 2019, 85(6): 443-53. doi:10.1016/j.biopsych.2018.09.031 |
| [21] | Li SY, Gao MZ, Mou ZJ, et al. Advances in neurotransmitter-mediated prefrontal circuitry in depression[J]. Prog Neuro Psychopharmacol Biol Psychiatry, 2025, 141: 111475. doi:10.1016/j.pnpbp.2025.111475 |
| [22] | Howard DM, Adams MJ, Clarke TK, et al. Genome-wide meta-analysis of depression identifies 102 independent variants and highlights the importance of the prefrontal brain regions[J]. Nat Neurosci, 2019, 22(3): 343-52. doi:10.1038/s41593-018-0326-7 |
| [23] | Fan ZX, Chang JR, Liang YL, et al. Neural mechanism underlying depressive-like state associated with social status loss[J]. Cell, 2023, 186(3): 560-76.e17. doi:10.1016/j.cell.2022.12.033 |
| [24] | Yang Y, Cui Y, Sang K, et al. Ketamine blocks bursting in the lateral habenula to rapidly relieve depression[J]. Nature, 2018, 554(7692): 317-22. doi:10.1038/nature25509 |
| [25] | Ma S, Chen M, Jiang Y, et al. Sustained antidepressant effect of ketamine through NMDAR trapping in the LHb[J]. Nature, 2023, 622(7984): 802-9. doi:10.1038/s41586-023-06624-1 |
| [26] | Roth BL, Gibbons S, Arunotayanun W, et al. The ketamine analogue methoxetamine and 3- and 4-methoxy analogues of phencyclidine are high affinity and selective ligands for the glutamate NMDA receptor[J]. PLoS One, 2013, 8(3): e59334. doi:10.1371/journal.pone.0059334 |
| [27] | Leal GC, Bandeira ID, Correia-Melo FS, et al. Intravenous arketamine for treatment-resistant depression: open-label pilot study[J]. Eur Arch Psychiatry Clin Neurosci, 2021, 271(3): 577-82. doi:10.1007/s00406-020-01110-5 |
| [28] | Bandeira ID, Leal GC, Correia-Melo FS, et al. Arketamine for bipolar depression: Open-label, dose-escalation, pilot study[J]. J Psychiatr Res, 2023, 164: 229-34. doi:10.1016/j.jpsychires.2023.06.028 |
| [29] | Ji XM, Huang ZM, Zhou CM, et al. Esketamine alleviates depressive-like behavior in neuropathic pain mice through the METTL3-GluA1 pathway[J]. Cell Biol Toxicol, 2025, 41(1): 38. doi:10.1007/s10565-024-09975-1 |
| [30] | Hu N, Zheng YJ, Liu XR, et al. CircKat6b mediates the antidepressant effect of esketamine by regulating astrocyte function[J]. Mol Neurobiol, 2025, 62(2): 2587-600. doi:10.1007/s12035-024-04420-0 |
| [31] | Wang TY, Weng HD, Zhou HJ, et al. Esketamine alleviates postoperative depression-like behavior through anti-inflammatory actions in mouse prefrontal cortex[J]. J Affect Disord, 2022, 307: 97-107. doi:10.1016/j.jad.2022.03.072 |
| [1] | Jianxiong AN, Zhijia CHI, Caiqun ZHAO, Yongxiang LI, Ruoguo WANG, Yanan HU. Efficacy and safety of super electroconvulsive therapy for treatment-resistant depression: a retrospective analysis of 292 cases [J]. Journal of Southern Medical University, 2026, 46(1): 104-112. |
| [2] | Xia LV, Rong XIAO. Materialistic values and their association with depression in medical postgraduates: materialism leads to higher risk of depression [J]. Journal of Southern Medical University, 2025, 45(6): 1220-1225. |
| [3] | Meimei CHEN, Yang WANG, Huangwei LEI, Fei ZHANG, Ruina HUANG, Zhaoyang YANG. Construction of recognition models for subthreshold depression based on multiple machine learning algorithms and vocal emotional characteristics [J]. Journal of Southern Medical University, 2025, 45(4): 711-717. |
| [4] | Zhengtao GAO, Pingyan LIN, Bingcan ZHOU, Mingheng CHEN, Erqi LIU, Tianxiang LEI, Huixin NI, Haixin LIU, Yao LIN, Qian XU. Hypericin ameliorates stress-induced depressive-like behaviors in mice by modulating the CN-NFAT calcium signaling pathway in microglia [J]. Journal of Southern Medical University, 2025, 45(3): 506-513. |
| [5] | Ying LIU, Borui LI, Yongcai LI, Lubo CHANG, Jiao WANG, Lin YANG, Yonggang YAN, Kai QV, Jiping LIU, Gang ZHANG, Xia SHEN. Jiawei Xiaoyao Pills improves depression-like behavior in rats by regulating neurotransmitters, inhibiting inflammation and oxidation and modulating intestinal flora [J]. Journal of Southern Medical University, 2025, 45(2): 347-358. |
| [6] | Yiming SUN, Xinran XU, Xuerui ZHUO, Hui CAI, Yan WANG. C1q-neutralizing antibodies improves postpartum depressive-like behaviors in mice by regulating the C1q/C3 pathway [J]. Journal of Southern Medical University, 2025, 45(10): 2111-2117. |
| [7] | Xiaotao LIANG, Xiaoshan LIANG, Yifan XIONG, Shiru XIE, Xiaoyu ZHU, Wei XIE. Modified Chaihu Guizhi Decoction alleviates anxiety- and depression-like behaviors in mice with chronic unpredictable mild stress by inhibiting the JAK2/STAT3 signaling pathway [J]. Journal of Southern Medical University, 2025, 45(10): 2146-2159. |
| [8] | Mingzi OUYANG, Jiaqi CUI, Hui WANG, Zheng LIANG, Dajin PI, Liguo CHEN, Qianjun CHEN, Yingchao WU. Kaixinsan alleviates adriamycin-induced depression-like behaviors in mice by reducing ferroptosis in the prefrontal cortex [J]. Journal of Southern Medical University, 2024, 44(8): 1441-1449. |
| [9] | Yeming ZHANG, Yuanxiang ZHANG, Xuebin SHEN, Guodong WANG, Lei ZHU. MiRNA-103-3p promotes neural cell autophagy by activating Wnt/β-catenin signaling via targeting rab10 in a rat model of depression [J]. Journal of Southern Medical University, 2024, 44(7): 1315-1326. |
| [10] | Yiming SUN, Rong ZHANG, Ying MENG, Lei ZHU, Mingqiang LI, Zhe LIU. Coenzyme Q10 alleviates depression-like behaviors in mice with chronic restraint stress by down-regulating the pyroptosis signaling pathway [J]. Journal of Southern Medical University, 2024, 44(5): 810-817. |
| [11] | LI Yanxiang, GUO Yongxin, CAO Fuyang, GUO Shuting, XUE Dinghao, ZHOU Zhikang, HAO Xinyu, TONG Li, FU Qiang. Inhibition of glutamatergic neurons in the dorsomedial periaqueductal gray alleviates excessive defensive behaviors of mice with post-traumatic stress disorder [J]. Journal of Southern Medical University, 2024, 44(3): 420-427. |
| [12] | XIONG Yifan, LIANG Xiaoshan, LIANG Xiaotao, LI Weipeng, QIAN Yixiao, XIE Wei. Saikosaponin a alleviates pentylenetetrazol-induced acute epileptic seizures in mouse models of depression by suppressing microglia activation-mediated inflammation [J]. Journal of Southern Medical University, 2024, 44(3): 515-522. |
| [13] | YANG Yang, LIU Gang, OU Yi, LU Wenqi. Lung-protective effect of esketamine combined with distal limb ischemic preconditioning in elderly patients undergoing thoracoscopic radical surgery for lung cancer: a randomized controlled trial in 160 cases [J]. Journal of Southern Medical University, 2024, 44(3): 484-490. |
| [14] | LI Xinyi, LIU Yujie, DENG Kechong, HU Yikui. Modulating gut microbiota improves neurological function and depressive symptoms in rats with post-stroke depression [J]. Journal of Southern Medical University, 2024, 44(2): 405-410. |
| [15] | Dongbo LIU, Zewen CHEN, Yun WANG, Xinpeng LI, Pengyu ZHAO, Haoxian ZHENG. Neuronal plasticity changes in the central amygdala and prelimbic cortex network in mice with chronic unpredictable mild stress-induced depression [J]. Journal of Southern Medical University, 2024, 44(11): 2082-2091. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||