Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (2): 362-373.doi: 10.12122/j.issn.1673-4254.2026.02.14
Zhaodie GENG1( ), Li HU1, Yunli DAI1, Ronggang LUO1, Tao XU1, Xuyang LIAO1, Zhiping YUAN2, Jianta WANG3, Ying XIAO1(
), Li HU1, Yunli DAI1, Ronggang LUO1, Tao XU1, Xuyang LIAO1, Zhiping YUAN2, Jianta WANG3, Ying XIAO1( )
)
Received:2025-04-30
Online:2026-02-20
Published:2026-03-10
Contact:
Ying XIAO
E-mail:3303068472@qq.com;yxhx20060725@126.com
Supported by:Zhaodie GENG, Li HU, Yunli DAI, Ronggang LUO, Tao XU, Xuyang LIAO, Zhiping YUAN, Jianta WANG, Ying XIAO. Cajanonic acid A derivative XJ-60 improves liver fibrosis in mice with non-alcoholic fatty liver disease by inhibiting the SP1/TGF-β/Smad3 signaling axis[J]. Journal of Southern Medical University, 2026, 46(2): 362-373.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.02.14
| Primer | Sequence(5´→3´) | Optimal Annealing Te-mperature (℃) |
|---|---|---|
| SP1 | F: TGCCAAATCTACTCCAGCA | 53.6 |
| R: AGCAAGGTGATGTTCCCA | ||
| FN | F: ATGTGGACCCCTCCTGATAGT | 57.3 |
| R: GCCCAGTGATTTCAGCAAAGG | ||
| Col-IV | F: TTTGGCTCGCCACCATAGAG | 60.04 |
| R: GCAGAGGCGAGCATCATAGT | ||
| TGF-β | F: CTCCCGTGGCTTCTAGTGC | 57.2 |
| R: GCCTTAGTTTGGACAGGATCTG | ||
| E-ca | F:CAGGTCTCCTCATGGCTTTGC | 57.7 |
| R:CTTCCGAAAAGAAGGCTGTCC | ||
| α-SMA | F: CCCAGACATCAGGGAGTAATGG | 56.1 |
| R: TCTATCGGATACTTCAGCGTCA | ||
| GAPDH | F: GGTTGTCTCCTGCGACTTCA | 57 |
| R: TGGTCCAGGGTTTCTTACTCC |
Tab.1 Primer sequence for RT-qPCR
| Primer | Sequence(5´→3´) | Optimal Annealing Te-mperature (℃) |
|---|---|---|
| SP1 | F: TGCCAAATCTACTCCAGCA | 53.6 |
| R: AGCAAGGTGATGTTCCCA | ||
| FN | F: ATGTGGACCCCTCCTGATAGT | 57.3 |
| R: GCCCAGTGATTTCAGCAAAGG | ||
| Col-IV | F: TTTGGCTCGCCACCATAGAG | 60.04 |
| R: GCAGAGGCGAGCATCATAGT | ||
| TGF-β | F: CTCCCGTGGCTTCTAGTGC | 57.2 |
| R: GCCTTAGTTTGGACAGGATCTG | ||
| E-ca | F:CAGGTCTCCTCATGGCTTTGC | 57.7 |
| R:CTTCCGAAAAGAAGGCTGTCC | ||
| α-SMA | F: CCCAGACATCAGGGAGTAATGG | 56.1 |
| R: TCTATCGGATACTTCAGCGTCA | ||
| GAPDH | F: GGTTGTCTCCTGCGACTTCA | 57 |
| R: TGGTCCAGGGTTTCTTACTCC |
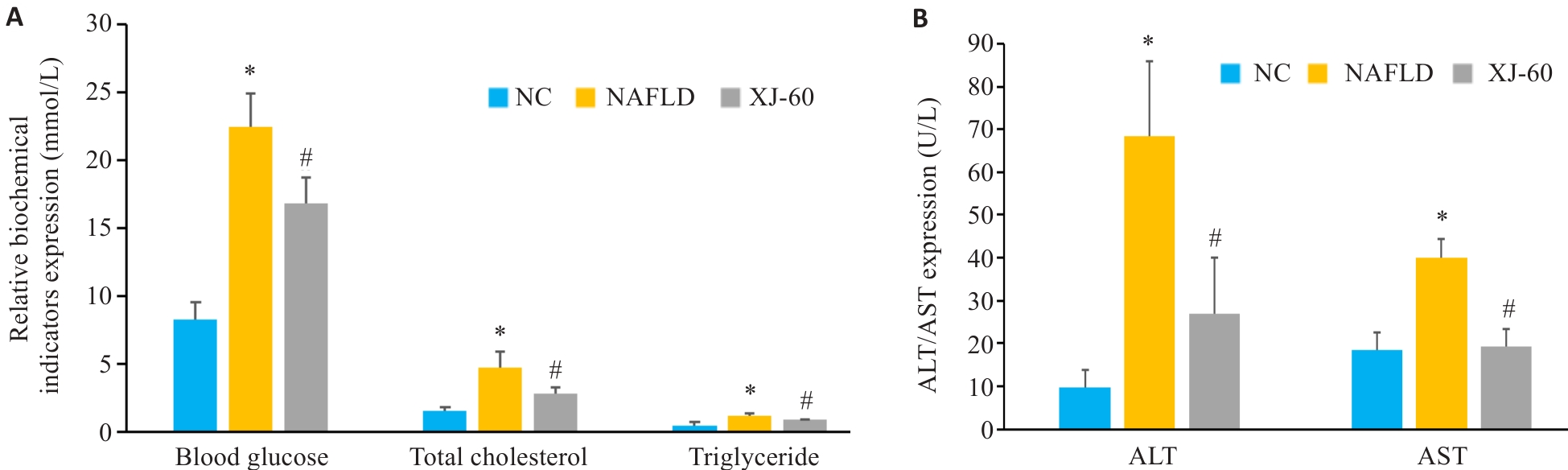
Fig.1 Levels of blood glucose, triglycerides and total cholesterol and expression levels of ALT and AST in the liver tissue of the mice in each group (n=6, Mean±SD). A: Levels of blood glucose, triglyceride and total cholesterol in different groups. B: Expression levels of ALT and AST in the liver tissue in different groups. *P<0.05 vs NC; #P<0.05 vs NAFLD.
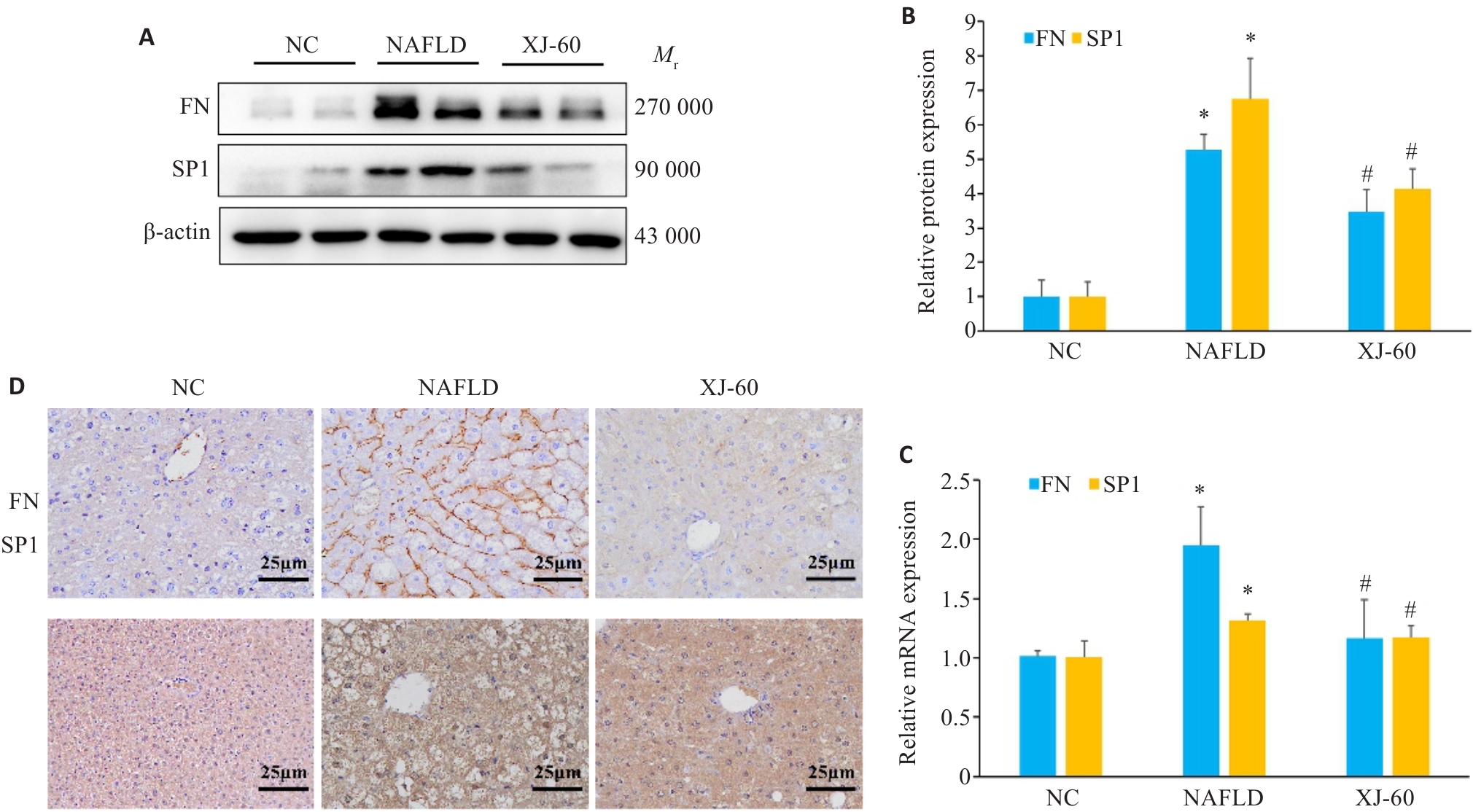
Fig.4 Protein levels of Sp1 and FN in the liver tissues of the mice in each group determined by Western blotting (n=6, Mean±SD). A, B: Western blotting for detecting protein expression levels of FN and SP1. C: RT-qPCR for detecting expressions of Sp1 and FN mRNA. D: Immunohistochemical staining for Sp1 and FN in the liver tissues of the mice in each group. *P<0.05 vs NC; #P<0.05 vs NAFLD.
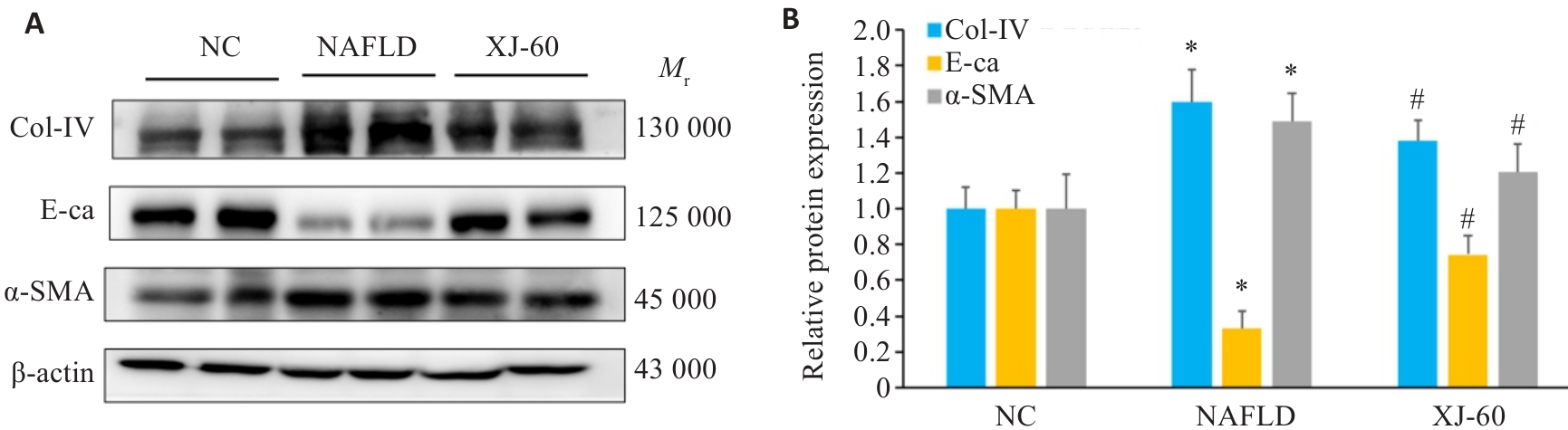
Fig.5 Expression levels of Col-IV, E-ca and α-SMA in the liver tissues of the mice in each group detected by Western blotting (n=6, Mean±SD). *P<0.05 vs NC; #P<0.05 vs NAFLD.
| Protein/Marker | Sp1 |
|---|---|
| Sp1 | 1.000 |
| E-ca | -0.767** |
| α-SMA | 0.777** |
Tab.2 Spearman rank correlation analysis
| Protein/Marker | Sp1 |
|---|---|
| Sp1 | 1.000 |
| E-ca | -0.767** |
| α-SMA | 0.777** |
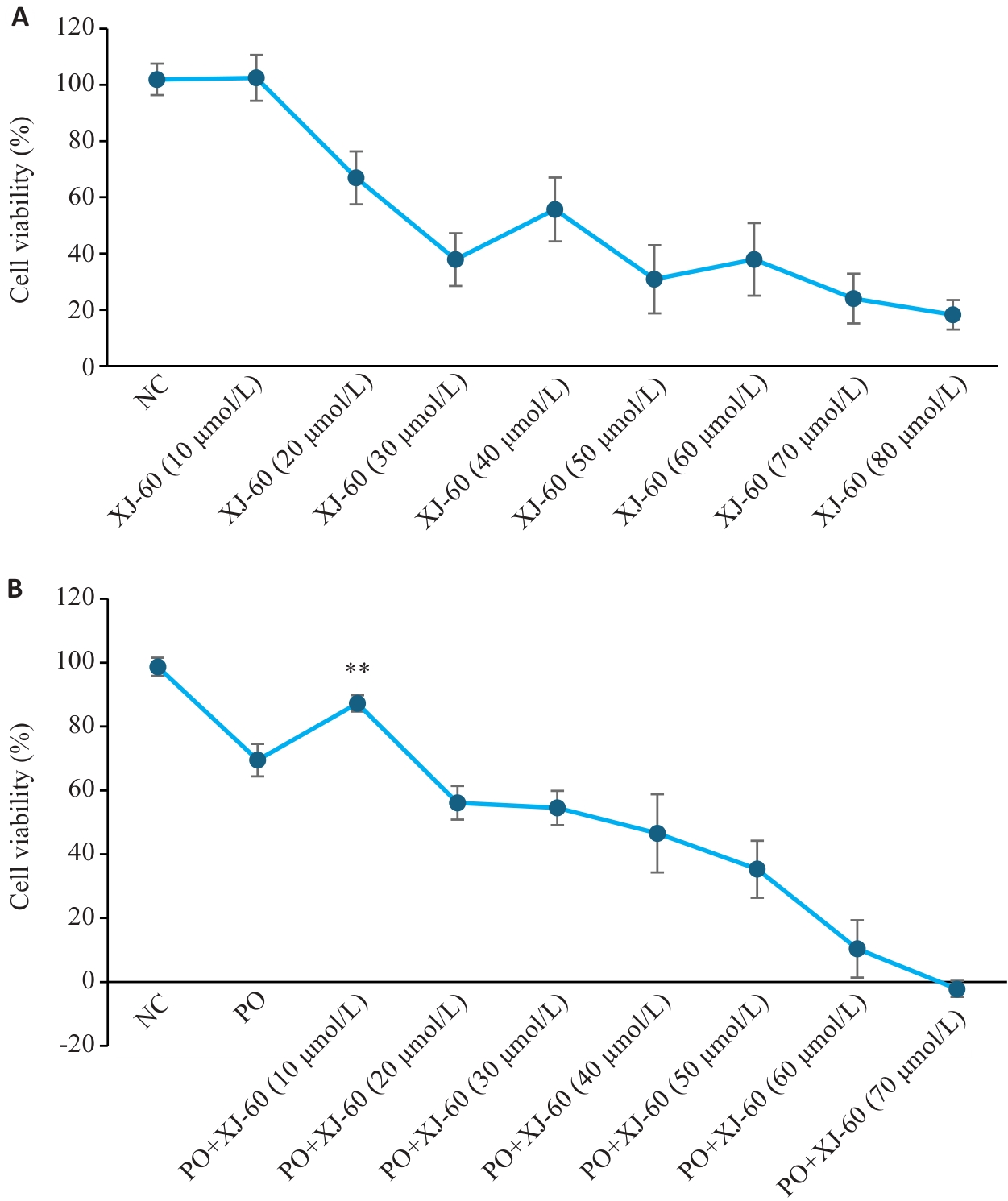
Fig.6 Comparison of cell survival rate of each group in CCK8 experiment (n=3, Mean±SD). A: Screening the safe concentration range of XJ-60 acting on AML12 cells without FFA intervention, P>0.05 vs NC. B: The optimal therapeutic concentration of XJ-60 acting on AML12 cells under FFA intervention. **P<0.01 vs PO.
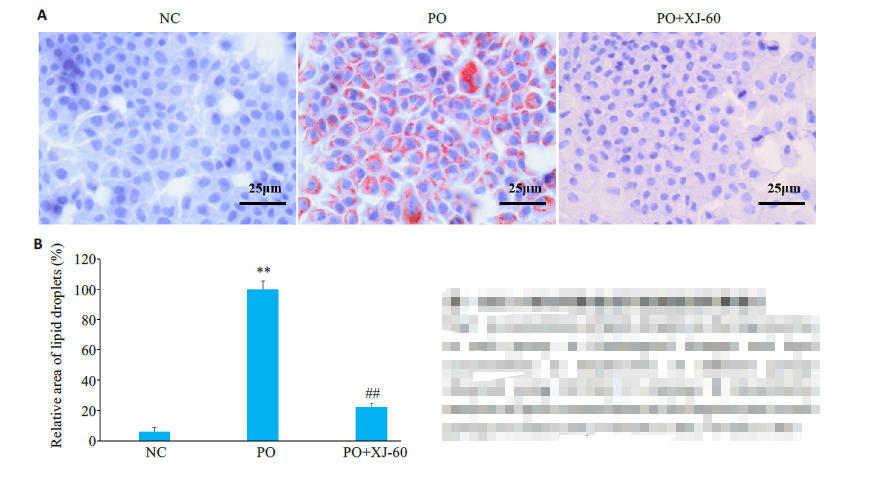
Fig.7 Accumulation of lipid droplets in the hepatocytesin each group. A: Pathological changes and lipidaccumulation in hepatocytes of different groupsobserved using Oil Red O staining. B: Quantitativeanalysis of lipid accumulation in the hepatocytes in eachgroup (n=3, Mean±SD), **P<0.01 vs NC; ##P<0.01 vs PO.
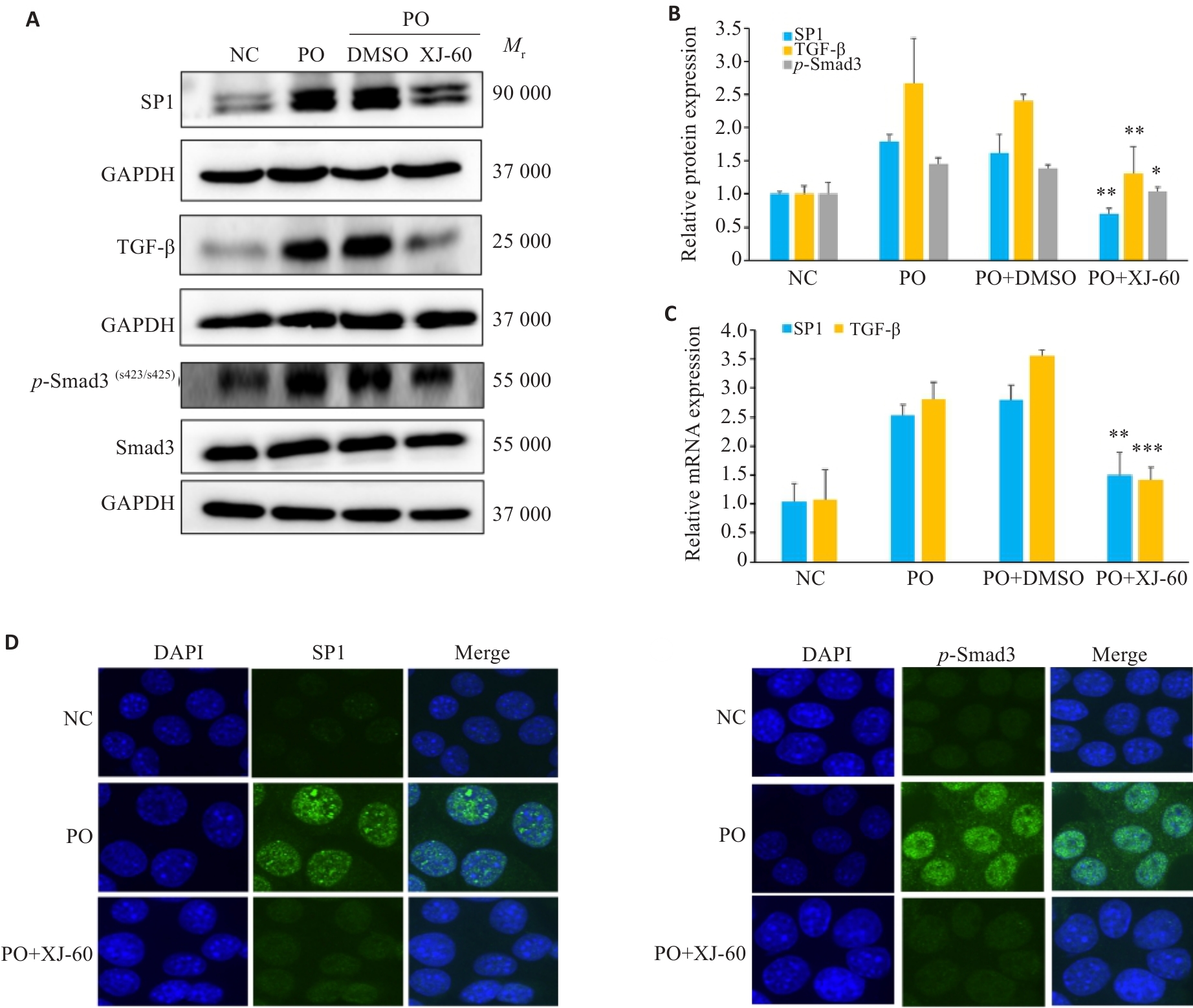
Fig.8 Effect of XJ-60 on expressions of SP1/TGF-β/Smad3 signaling pathway in FFA-induced AML12 cells (n=3, Mean±SD). A, B: Protein expression levels of SP1, TGF-β and p-Smad3 in AML12 cells in each group (*P<0.05, **P<0.01 vs PO+DMSO). C: mRNA expression levels of SP1, TGF-β and p-Smad3 in hepatocytes of each group (**P<0.01,***P<0.001 vs PO+DMSO). D: SP1 and p-Smad3 immunofluorescence staining of AML12 cells in each group (×1000).
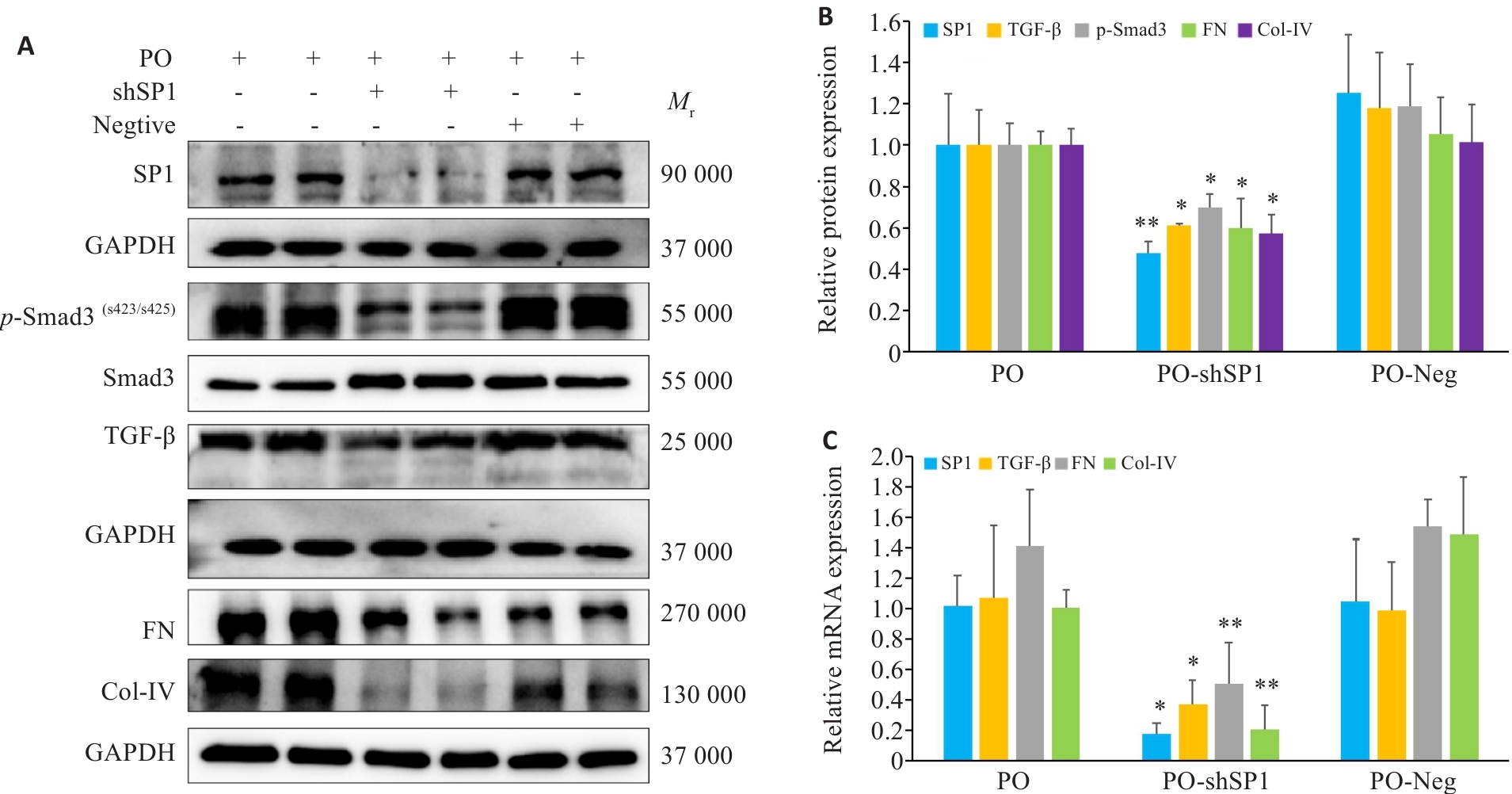
Fig.9 Changes in expressions of the protein markers in AML12 cells after SP1 knockdown (n=3, Mean±SD). A, B: Protein expression levels of FN, Col-IV, SP1, TGF‑β and p-Smad3 in AML12 cells (*P<0.05, **P<0.01 vs PO+Neg). C: mRNA expression levels of FN, Col-IV, SP1, TGF-β and p-Smad3 in AML12 cells in each group (*P<0.05, **P<0.01 vs PO+Neg).
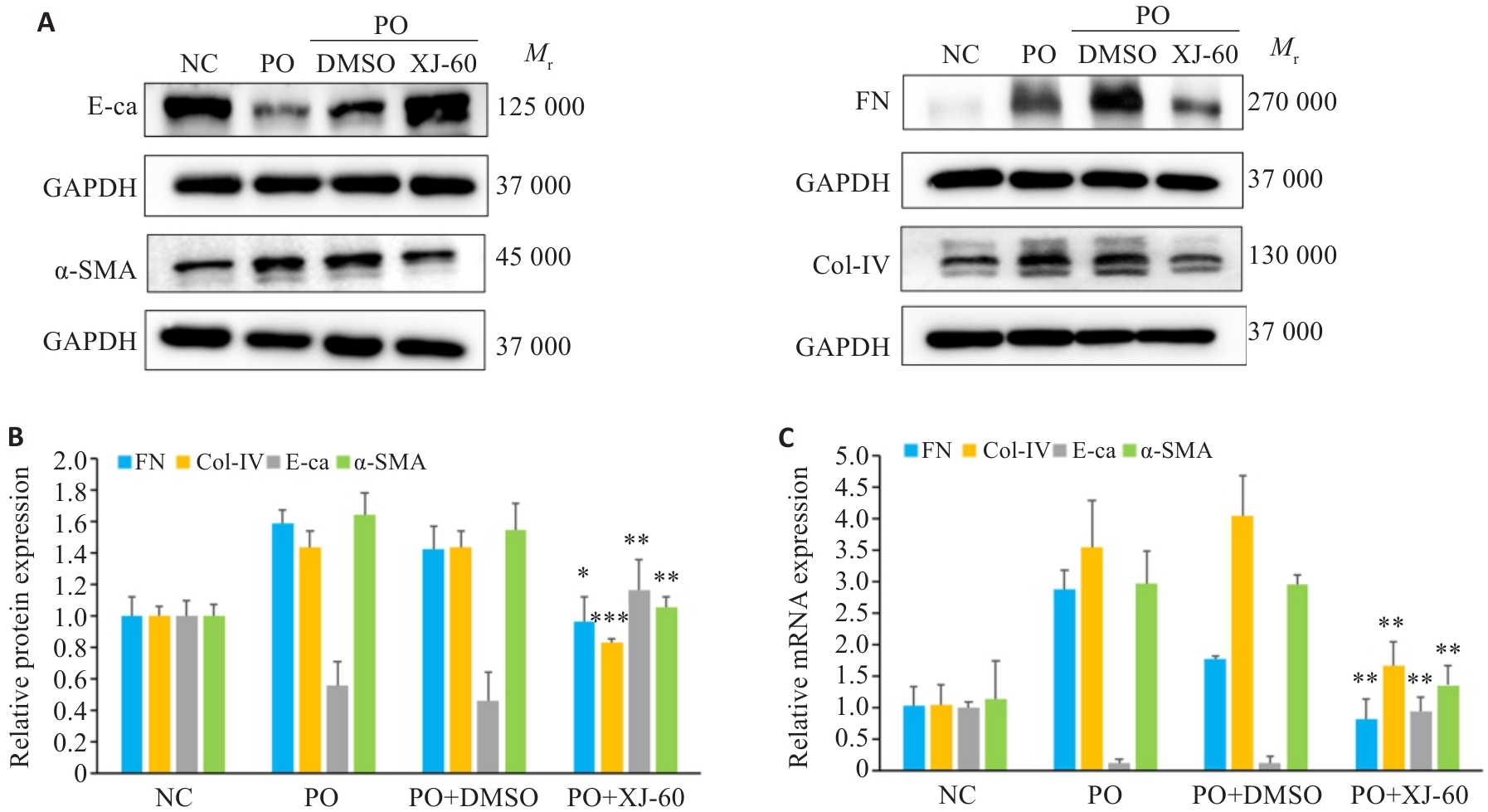
Fig.10 Effect of XJ-60 treatment on expressions of EMT and ECM markers in AML12 cells (n=3, Mean±SD). A, B: Protein expression levels of FN, Col-IV, E-ca and α-SMA in AML12 cells in each group (*P<0.05, **P<0.01, ***P<0.001 vs PO+DMSO). C: mRNA expression levels of FN, Col-IV, E-ca and α-SMA in hepatocytes of each group (**P<0.01 vs PO+DMSO).
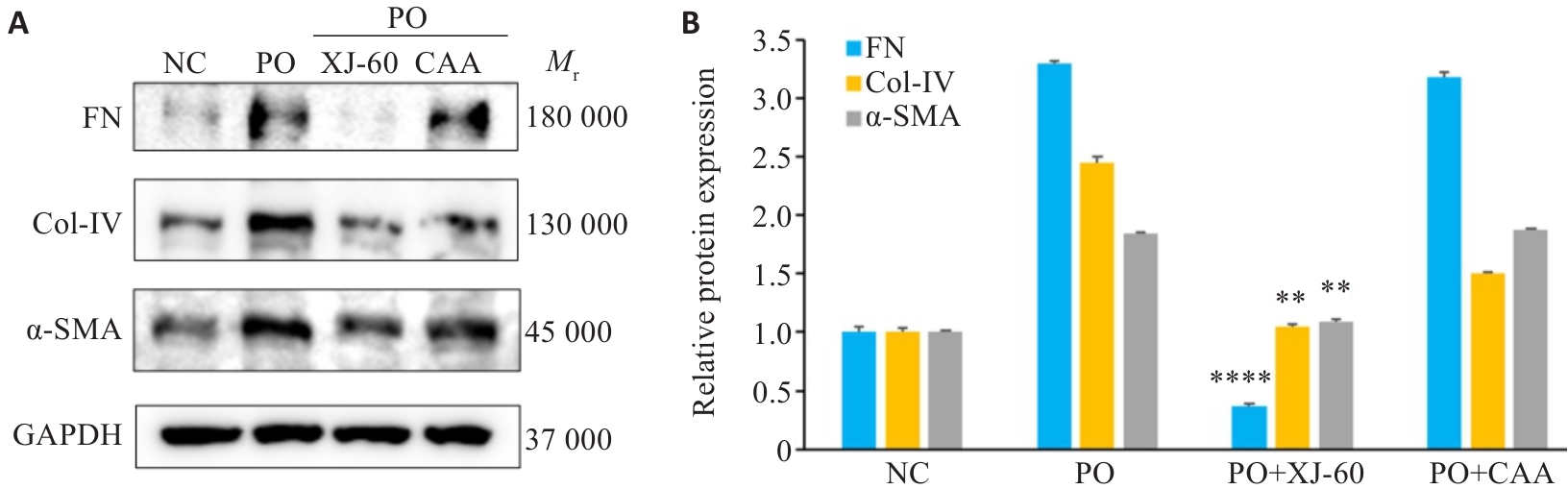
Fig.11 Effects of CAA and XJ-60 for improving fibrosis in AML12 cells (n=3, Mean±SD). A: Protein bands of FN, Col-IV and α-SMA in AML12 cells in each group. B: Relative protein expression levels. **P<0.01, ****P<0.0001 vs PO+CAA.
| [1] | Powell EE, Wong VW, Rinella M. Non-alcoholic fatty liver disease[J]. Lancet, 2021, 397(10290): 2212-24. doi:10.1016/s0140-6736(20)32511-3 |
| [2] | Brunt EM, Kleiner DE, Carpenter DH, et al. NAFLD: reporting histologic findings in clinical practice[J]. Hepatology, 2021, 73(5): 2028-38. doi:10.1002/hep.31599 |
| [3] | Rodríguez-Lara A, Rueda-Robles A, Sáez-Lara MJ, et al. From non-alcoholic fatty liver disease to liver cancer: microbiota and inflammation as key players[J]. Pathogens, 2023, 12(7): 940-55. doi:10.3390/pathogens12070940 |
| [4] | Zhou Y, Zhang L, Ma Y, et al. Secretome of senescent hepatic stellate cells favors malignant transformation from nonalcoholic steatohepatitis-fibrotic progression to hepatocellular carcinoma[J]. Theranostics, 2023, 13(13): 4430-48. doi:10.7150/thno.85369 |
| [5] | Martínez-Espinosa I, Serrato JA, Cabello-Gutiérrez C, et al. Mechanisms of microRNA regulation of the epithelial-mesenchymal transition (EMT) in lung cancer[J]. Life: Basel, 2024, 14(11): 1431. doi:10.3390/life14111431 |
| [6] | Nair B, Nath LR. Inevitable role of TGF‑β1 in progression of nonalcoholic fatty liver disease[J]. J Recept Signal Transduct Res, 2020, 40(3): 195-200. doi:10.1080/10799893.2020.1726952 |
| [7] | Chen C, Chen J, Wang Y, et al. Ganoderma lucidum polysaccharide inhibits HSC activation and liver fibrosis via targeting inflammation, apoptosis, cell cycle, and ECM-receptor interaction mediated by TGF-β/Smad signaling[J]. Phytomedicine, 2023, 110: 154626. doi:10.1016/j.phymed.2022.154626 |
| [8] | Xin X, Cheng X, Zeng F, et al. The role of TGF-β/SMAD signaling in hepatocellular carcinoma: from mechanism to therapy and prognosis[J]. Int J Biol Sci, 2024, 20(4): 1436-51. doi:10.7150/ijbs.89568 |
| [9] | Sun Y, Chen X, Chen L, et al. MFAP2 promotes HSCs activation through FBN1/TGF-β/Smad3 pathway[J]. J Cell Mol Med, 2023, 27(21): 3235-46. doi:10.1111/jcmm.17884 |
| [10] | Ding J, Fayyaz AI, Ding Y, et al. Role of specificity protein 1 (SP1) in cardiovascular diseases: pathological mechanisms and therapeutic potentials[J]. Biomolecules, 2024, 14(7): 807-21. doi:10.3390/biom14070807 |
| [11] | Zhou W, Fang J, Jia Q, et al. Transcription factor specificity protein (SP) family in renal physiology and diseases[J]. PeerJ, 2025, 13: e18820. doi:10.7717/peerj.18820 |
| [12] | Shen ZJ, Zhang YZ, Bu GK, et al. Renal denervation improves mitochondrial oxidative stress and cardiac hypertrophy through inactivating SP1/BACH1-PACS2 signaling[J]. Int Immunophar-macol, 2024, 141: 112778. doi:10.1016/j.intimp.2024.112778 |
| [13] | Liu X, Gong Q, Yang L, et al. microRNA-199a-5p regulates epithelial-to-mesenchymal transition in diabetic cataract by targeting SP1 gene[J]. Mol Med, 2020, 26(1): 122-222. doi:10.1186/s10020-020-00250-7 |
| [14] | Firneisz G. Non-alcoholic fatty liver disease and type 2 diabetes mellitus: the liver disease of our age[J]? World J Gastroenterol,2014, 20(27):9072-89. |
| [15] | Zeng J, Fan JG, Francque SM. Therapeutic management of metabolic dysfunction associated steatotic liver disease[J]. United European Gastroenterol J, 2024, 12(2): 177-86. doi:10.1002/ueg2.12525 |
| [16] | Mantovani A, Byrne CD, Targher G. Efficacy of peroxisome proliferator-activated receptor agonists, glucagon-like peptide-1 receptor agonists, or sodium-glucose cotransporter-2 inhibitors for treatment of non-alcoholic fatty liver disease: a systematic review[J]. Lancet Gastroenterol Hepatol, 2022, 7(4): 367-78. doi:10.1016/s2468-1253(21)00261-2 |
| [17] | Liu J, Wang LN. Peroxisome proliferator-activated receptor gamma agonists for preventing recurrent stroke and other vascular events in people with stroke or transient ischaemic attack[J]. Cochrane Database Syst Rev, 2023, 1: CD010693. doi:10.1002/14651858.cd010693.pub6 |
| [18] | Yang R, Wang L, Xie J, et al. Treatment of type 2 diabetes mellitus via reversing insulin resistance and regulating lipid homeostasis in vitro and in vivo using cajanonic acid A[J]. Int J Mol Med, 2018, 42(5): 2329-42. |
| [19] | Loomba R, Friedman SL, Shulman GI. Mechanisms and disease consequences of nonalcoholic fatty liver disease[J]. Cell, 2021, 184(10): 2537-64. doi:10.1016/j.cell.2021.04.015 |
| [20] | Nevens F, Andreone P, Mazzella G, et al. A placebo-controlled trial of obeticholic acid in primary biliary cholangitis[J]. N Engl J Med, 2016, 375(7): 631-43. doi:10.1056/nejmc1611913 |
| [21] | Kamata S, Honda A, Ishii I. Current clinical trial status and future prospects of PPAR-targeted drugs for treating nonalcoholic fatty liver disease[J]. Biomolecules, 2023, 13(8): 1264-85. doi:10.3390/biom13081264 |
| [22] | 王 璐. 树豆酮酸A调节血糖血脂的作用和机理研究[D]. 广州中医药大学, 2012. |
| [23] | 沈小玲, 王 璐, 胡英杰, 等.树豆酮酸A在制备糖尿病伴随症及高脂血症药物中的应用: CN201210119665.5[P].CN 102670576A[2025-12-11]. |
| [24] | 谢 洁. 树豆酮酸A改善胰岛素抵抗的作用研究[D].广州中医药大学, 2014. |
| [25] | 王一凡, 吕厚星, 李志阳, et al. 树豆酮酸A可减轻db/db小鼠非酒精性脂肪肝损伤 [J]. 中国生物化学与分子生物学报, 2024, 40(3): 373-82. |
| [26] | Yao Y, Luo ZP, Li HW, et al. P38γ modulates the lipid metabolism in non-alcoholic fatty liver disease by regulating theJAK-STATsignaling pathway[J]. FASEB J, 2023, 37(1): e22716. doi:10.1096/fj.202200939rr |
| [27] | Cai W, Wu S, Ming X, et al. IL6 Derived from Macrophages under Intermittent Hypoxia Exacerbates NAFLD by Promoting Ferroptosis via MARCH3-Led Ubiquitylation of GPX4 [J]. Advanced science (Weinheim, Baden-Wurttemberg, Germany), 2024, 11(41): e2402241. doi:10.1002/advs.202402241 |
| [28] | Zhang HC, Zhang GP, Zhang JY, et al. Transcription factor SP1 and oncoprotein PPP1R13L regulate nicotine-induced epithelial-mesenchymal transition in lung adenocarcinoma via a feedback loop[J]. Biochem Pharmacol, 2022, 206: 115344. doi:10.1016/j.bcp.2022.115344 |
| [29] | Xie J, Lin H, Jin F, et al. Jia Wei Qingxin Lotus Seed Drink ameliorates epithelial mesenchymal transition injury in diabetic kidney disease via inhibition of JMJD1C/SP1/ZEB1 signaling pathway[J]. Phytomedicine, 2024, 135: 156142. doi:10.1016/j.phymed.2024.156142 |
| [30] | Song LY, Huang YL, Liu L, et al. Meteorin-like alleviates hepatic steatosis by regulating hepatic triglyceride secretion and fatty acid oxidation[J]. Cell Rep, 2025, 44(2): 115246. doi:10.1016/j.celrep.2025.115246 |
| [31] | Li Y, Chen R, Wu J, et al. Salvianolic acid B protects against pulmonary fibrosis by attenuating stimulating protein 1-mediated macrophage and alveolar type 2 cell senescence[J]. Phytother Res, 2024, 38(2): 620-35. doi:10.1002/ptr.8070 |
| [32] | Zhu QQ, Yang XY, Zhang XJ, et al. EGCG targeting Notch to attenuate renal fibrosis via inhibition of TGFβ/Smad3 signaling pathway activation in streptozotocin-induced diabetic mice[J]. Food Funct, 2020, 11(11): 9686-95. doi:10.1039/d0fo01542c |
| [33] | Ren Q, Wang B, Guo F, et al. Natural flavonoid pectolinarigenin alleviated hyperuricemic nephropathy via suppressing TGFβ/SMAD3 and JAK2/STAT3 signaling pathways[J]. Front Pharmacol, 2021, 12: 792139. doi:10.3389/fphar.2021.792139 |
| [34] | Ding L, Li Y, Yang Y, et al. Wenfei buqi Tongluo formula against bleomycin-induced pulmonary fibrosis by inhibiting TGF-β/Smad3 pathway[J]. Front Pharmacol, 2021, 12: 762998. doi:10.3389/fphar.2021.762998 |
| [35] | Ding A, Bian YY, Zhang ZH. SP1/TGF‑β1/SMAD2 pathway is involved in angiogenesis during osteogenesis[J]. Mol Med Rep, 2020, 21(3): 1581-9. |
| [36] | Yang ZS, Zhang H, Yin MH, et al. TGF‑β1/Smad3 upregulates UCA1 to promote liver fibrosis through DKK1 and miR18a[J]. J Mol Med, 2022, 100(10): 1465-78. doi:10.1007/s00109-022-02248-6 |
| [37] | Wang J, Fan Z, Li J, et al. Transcription factor specificity protein 1-mediated Serine/threonine kinase 39 upregulation promotes the proliferation, migration, invasion and epithelial-mesenchymal transition of hepatocellular carcinoma cells by activating the transforming growth factor‑β1/Smad2/3 pathway[J]. Bioengineered, 2021, 12(1): 3566-77. doi:10.1080/21655979.2021.1947939 |
| [38] | Li YJ, Fan WG, Link F, et al. Transforming growth factor β latency: a mechanism of cytokine storage and signalling regulation in liver homeostasis and disease[J]. JHEP Rep, 2022, 4(2): 100397. doi:10.1016/j.jhepr.2021.100397 |
| [1] | Biyun LUO, Xin YI, Yijing CAI, Shiqing ZHANG, Peng WANG, Tong LI, Ken Kin Lam YUNG, Pingzheng ZHOU. Ching Shum Pills alleviates non-alcoholic fatty liver disease in mice by ameliorating lipid metabolism disorders [J]. Journal of Southern Medical University, 2025, 45(9): 1840-1849. |
| [2] | Du SHANG, Wen LI, Lihua CUI, Ming CHEN. Hugan Decoction alleviates non-alcoholic fatty liver disease in rats by activating the AMPK/m-TOR signaling pathway and reducing lipid synthesis [J]. Journal of Southern Medical University, 2025, 45(12): 2667-2678. |
| [3] | Qing SHI, Suye RAN, Lingyu SONG, Hong YANG, Wenjuan WANG, Hanlin LIU, Qi LIU. NLRP6 overexpression improves nonalcoholic fatty liver disease by promoting lipid oxidation and decomposition in hepatocytes through the AMPK/CPT1A/PGC1A pathway [J]. Journal of Southern Medical University, 2025, 45(1): 118-125. |
| [4] | Jing XIAO, Ying LI, Min FANG, Hong GONG, Wen LI, Chunyan ZHANG, Fangyao CHEN, Yan ZHANG, Tuo HAN. Triglyceride-glucose index in non-obese individuals: its association with and predictive value for non-alcoholic fatty liver disease [J]. Journal of Southern Medical University, 2024, 44(7): 1266-1271. |
| [5] | Zhi CUI, Cuijiao MA, Qianru WANG, Jinhao CHEN, Ziyang YAN, Jianlin YANG, Yafeng LÜ, Chunyu CAO. A recombinant adeno-associated virus expressing secretory TGF‑β type II receptor inhibits triple-negative murine breast cancer 4T1 cell proliferation and lung metastasis in mice [J]. Journal of Southern Medical University, 2024, 44(5): 818-826. |
| [6] | KONG Xiang, ZHANG Teng, ZHANG Yan, GAO Linxi, WANG Wen, WANG Mengyan, WANG Guodong, LÜ Kun. Overexpression of lncRNA HEM2M alleviates liver injury in mice with non-alcoholic fatty liver disease [J]. Journal of Southern Medical University, 2024, 44(1): 1-8. |
| [7] | DENG Guanghui, JIA Hui, LI Yunjia, LI Junjie, WU Chaofeng, SHI Hao, QIN Mengchen, ZHAO Jiamin, LIU Chang, LIAO Yuxin, GAO Lei. Erchen Decoction improves iron homeostasis in mice with non-alcoholic fatty liver disease by regulating iron transport capacity in the spleen [J]. Journal of Southern Medical University, 2023, 43(8): 1287-1296. |
| [8] | ZHANG Jia, XUE Wei, ZHANG Shujun, ZHU Yali, YANG Cheng, GAO Yue, SHI Lingfeng, HUANG Wenxiang. TSPAN8 is involved in lipid metabolism in non-alcoholic fatty liver disease in mice [J]. Journal of Southern Medical University, 2022, 42(5): 705-711. |
| [9] | . Role of TGF-β1/ILK/FSP1 signaling pathway in cyclosporin A-induced epithelialmesenchymal transition in cultured renal tubular epithelial cells [J]. Journal of Southern Medical University, 2019, 39(07): 804-. |
| [10] |
.
Effect of electro-acupuncture on hepatic Toll-like receptor 4 and nuclear factor κB expressions in rats with non-alcoholic fatty liver disease [J]. Journal of Southern Medical University, 2014, 34(11): 1584-. |
| [11] |
.
Therapeutic effect of saxagliptin in rat models of nonalcoholic fatty liver and type 2 diabetes [J]. Journal of Southern Medical University, 2014, 34(06): 862-. |
| [12] | . Effects of glucagon-like peptide-1 on liver oxidative stress, TNF-α and TGF-β1 in rats with non-alcoholic fatty liver disease [J]. Journal of Southern Medical University, 2013, 33(11): 1661-. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||