Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (2): 345-352.doi: 10.12122/j.issn.1673-4254.2026.02.12
Jinyan WEI1( ), Hairui ZHENG1, Yunteng ZHAO1, Yan YU1, Yingying XU2, Gang LI1,3(
), Hairui ZHENG1, Yunteng ZHAO1, Yan YU1, Yingying XU2, Gang LI1,3( )
)
Received:2025-06-26
Online:2026-02-20
Published:2026-03-10
Contact:
Gang LI
E-mail:2320925662@qq.com;lg@smu.edu.cn
Jinyan WEI, Hairui ZHENG, Yunteng ZHAO, Yan YU, Yingying XU, Gang LI. Establishment of an Epstein-Barr virus infection model using human nasal organoids[J]. Journal of Southern Medical University, 2026, 46(2): 345-352.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.02.12
| Sample number | Source | Reason for surgery |
|---|---|---|
| ZZH-9580 | Nasal polyps | CRSwNP |
| LHX-4635 | Nasal polyps | CRSwNP |
| XWJ-4024 | Nasal polyps | CRSwNP |
| XCY-8640 | Nasal polyps | CRSwNP |
| LJL-3202 | Nasal polyps | CRSwNP |
| LHB-4635 | Nasal polyps | CRSwNP |
| TRJ-8166 | Nasal polyps | CRSwNP |
Tab.1 Information of the nasal mucosa samples
| Sample number | Source | Reason for surgery |
|---|---|---|
| ZZH-9580 | Nasal polyps | CRSwNP |
| LHX-4635 | Nasal polyps | CRSwNP |
| XWJ-4024 | Nasal polyps | CRSwNP |
| XCY-8640 | Nasal polyps | CRSwNP |
| LJL-3202 | Nasal polyps | CRSwNP |
| LHB-4635 | Nasal polyps | CRSwNP |
| TRJ-8166 | Nasal polyps | CRSwNP |
| Antibody | Brand | Cat No. | dilution ratio |
|---|---|---|---|
| Anti-Cytokeratin 5 antibody | Abcam | ab75869 | 1:100 |
| Anti-Cytokeratin 14 antibody | Abcam | ab119695 | 1:100 |
| Anti-p63 antibody | Abcam | ab124762 | 1:100 |
| Anti-MUC5AC antibody | Abcam | ab198294 | 1:200 |
| Anti-beta Ⅳ tubulin antibody | Abcam | ab179509 | 1:200 |
| Anti-FoxJ1 antibody | Abcam | ab235445 | 1:2000 |
| COL4A2 Polyclonal antibody | Proteintech | 55131-1-AP | 1:300 |
| MYH9 Monoclonal antibody | Proteintech | 60233-1-IG | 1:200 |
| EPHA2 Monoclonal antibody | Proteintech | 66736-1-IG | 1:200 |
| Neuropilin 1/CD304 Recombinant antibody | Proteintech | 84429-5-RR | 1:300 |
| Anti-EBNA1 antibody | Abcam | ab316860 | 1:2000 |
| Goat anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 | Invitrogen | A11034 | 1:500 |
| Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 | Invitrogen | A11029 | 1:500 |
| HRP-conjugated Goat Anti-Rabbit/mouse IgG for IHC (ready to use) | Proteintech | PR30009 | No dilution needed |
Tab.2 Antibodies for immunofluorescence assay and immunohistochemistry
| Antibody | Brand | Cat No. | dilution ratio |
|---|---|---|---|
| Anti-Cytokeratin 5 antibody | Abcam | ab75869 | 1:100 |
| Anti-Cytokeratin 14 antibody | Abcam | ab119695 | 1:100 |
| Anti-p63 antibody | Abcam | ab124762 | 1:100 |
| Anti-MUC5AC antibody | Abcam | ab198294 | 1:200 |
| Anti-beta Ⅳ tubulin antibody | Abcam | ab179509 | 1:200 |
| Anti-FoxJ1 antibody | Abcam | ab235445 | 1:2000 |
| COL4A2 Polyclonal antibody | Proteintech | 55131-1-AP | 1:300 |
| MYH9 Monoclonal antibody | Proteintech | 60233-1-IG | 1:200 |
| EPHA2 Monoclonal antibody | Proteintech | 66736-1-IG | 1:200 |
| Neuropilin 1/CD304 Recombinant antibody | Proteintech | 84429-5-RR | 1:300 |
| Anti-EBNA1 antibody | Abcam | ab316860 | 1:2000 |
| Goat anti-Rabbit IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 | Invitrogen | A11034 | 1:500 |
| Goat anti-Mouse IgG (H+L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 | Invitrogen | A11029 | 1:500 |
| HRP-conjugated Goat Anti-Rabbit/mouse IgG for IHC (ready to use) | Proteintech | PR30009 | No dilution needed |
| Gene | Foward (5'→3') | Reverse (5'→3') |
|---|---|---|
| GAPDH | GGAGCGAGATCCCTCCAAAAT | GGCTGTTGTCATACTTCTCATGG |
| EPHA2 | CCCGATGAGATCACCGTCAG | GGCACCGATATCCTGGAAGG |
| NMHC-ⅡA | GAGCAAATGGGCCTGCT | TGTTGTCGGGCATGGA |
| NRP1 | ACCCAAGTGAAAAATGCGAATG | CCTCCAAATCGAAGTGAGGGTT |
| EBNA1 | GGTCGTGGACGTGGAGAAAA | GGTGGAGACCCGGATGATG |
| BALF5 | GAGCGATCTTGGCAATCTCT | TGGTCATGGATCTGCTAAACC |
Tab.3 Primer sequence for RT-qPCR
| Gene | Foward (5'→3') | Reverse (5'→3') |
|---|---|---|
| GAPDH | GGAGCGAGATCCCTCCAAAAT | GGCTGTTGTCATACTTCTCATGG |
| EPHA2 | CCCGATGAGATCACCGTCAG | GGCACCGATATCCTGGAAGG |
| NMHC-ⅡA | GAGCAAATGGGCCTGCT | TGTTGTCGGGCATGGA |
| NRP1 | ACCCAAGTGAAAAATGCGAATG | CCTCCAAATCGAAGTGAGGGTT |
| EBNA1 | GGTCGTGGACGTGGAGAAAA | GGTGGAGACCCGGATGATG |
| BALF5 | GAGCGATCTTGGCAATCTCT | TGGTCATGGATCTGCTAAACC |
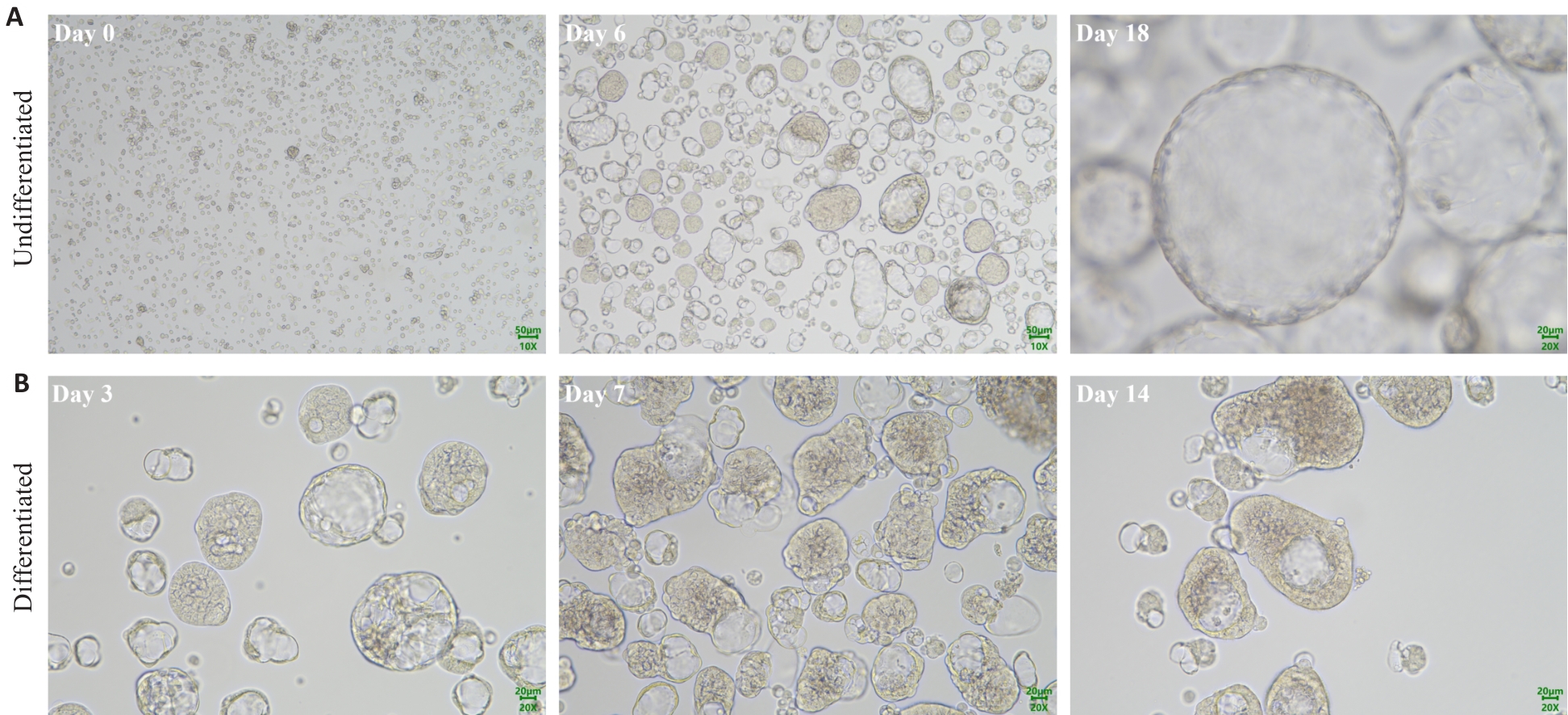
Fig.1 Phase-contrast microscopy of nasal organoids in culture. A: Representative images at different time points during the expansion of nasal organoids. B: Representative images of the nasal organoids on days 3, 7, and 14 during differentiation induction.
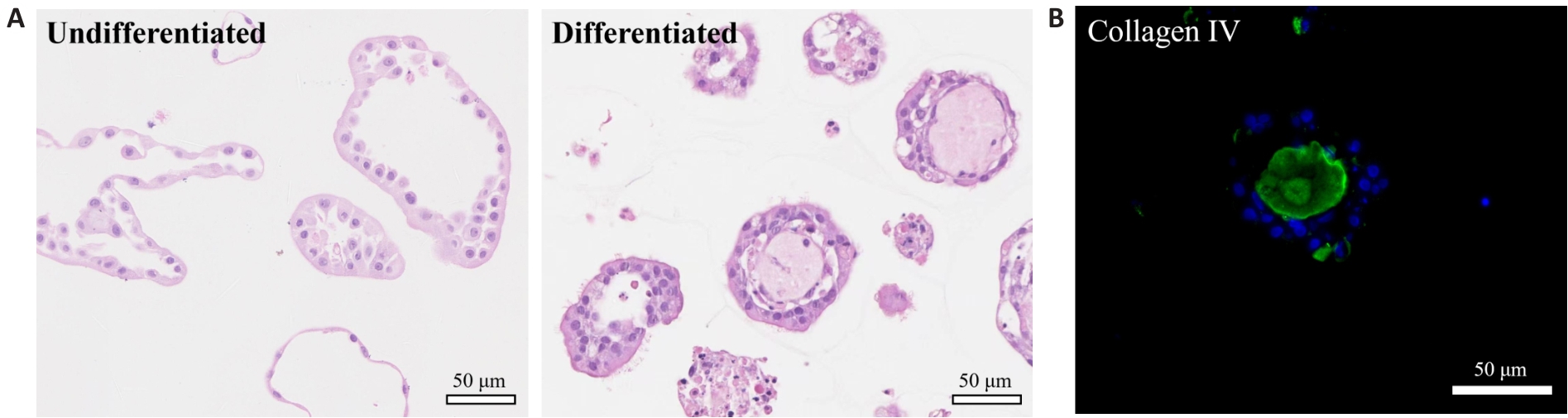
Fig.2 HE staining of the nasal organoids and immunofluorescence staining for the basal lamina marker collagen IV. A: HE staining of undifferentiated and differentiated nasal organoids. The differentiated nasal organoids exhibit a higher density of cilia on their apical surface. B: Immunofluorescence staining for collagen IV in differentiated nasal organoids.
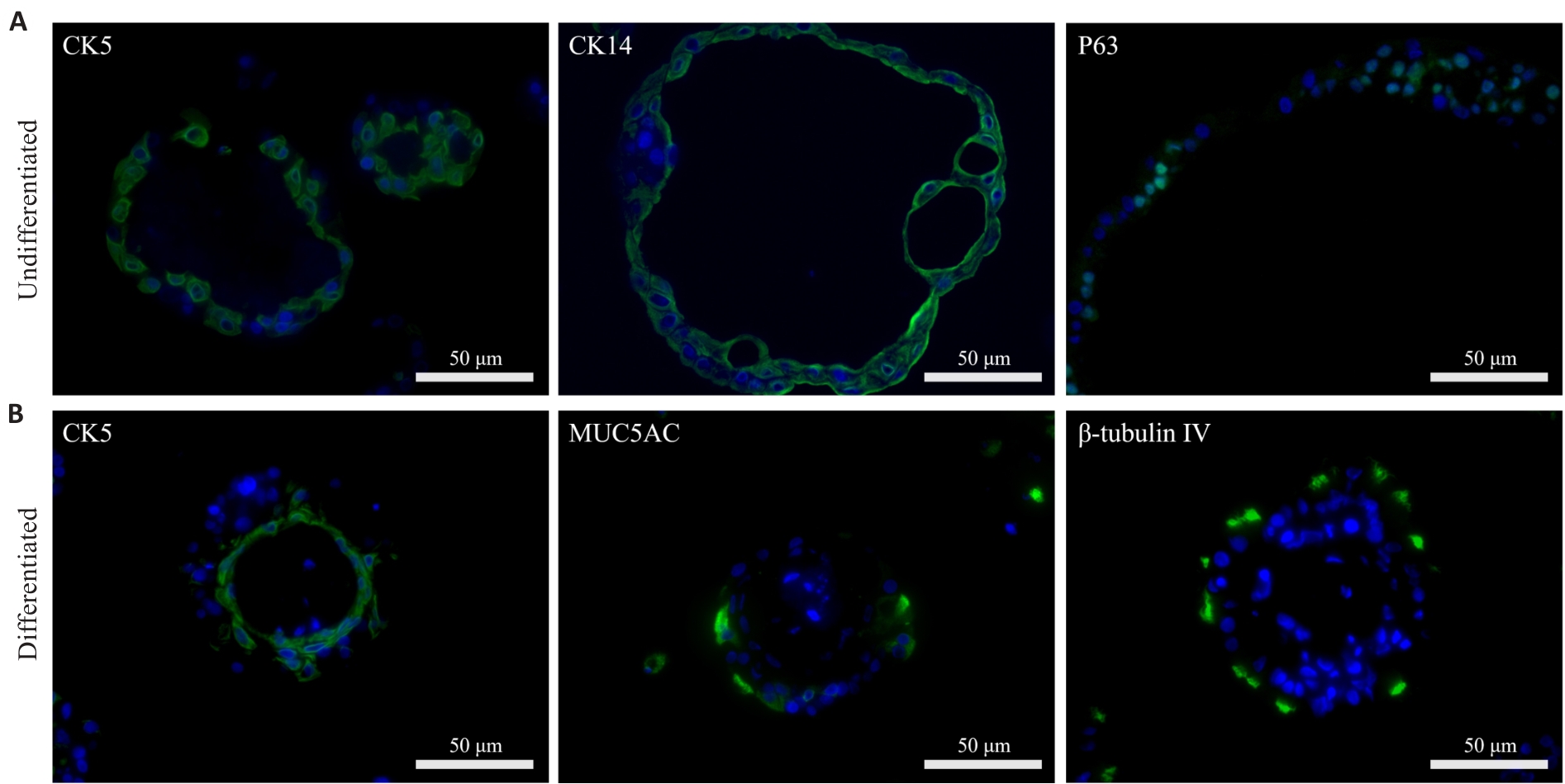
Fig.3 Immunofluorescence analysis of cellular composition of undifferentiated and differentiated nasal organoids. A: Representative images of undifferentiated organoids showing positive expressions of epithelial basal stem cell markers CK5, CK14, and p63. B: Representative images of differentiated organoids consisting of basal cells (CK5), goblet cells (MUC5AC), and ciliated cells (β-tubulin IV).
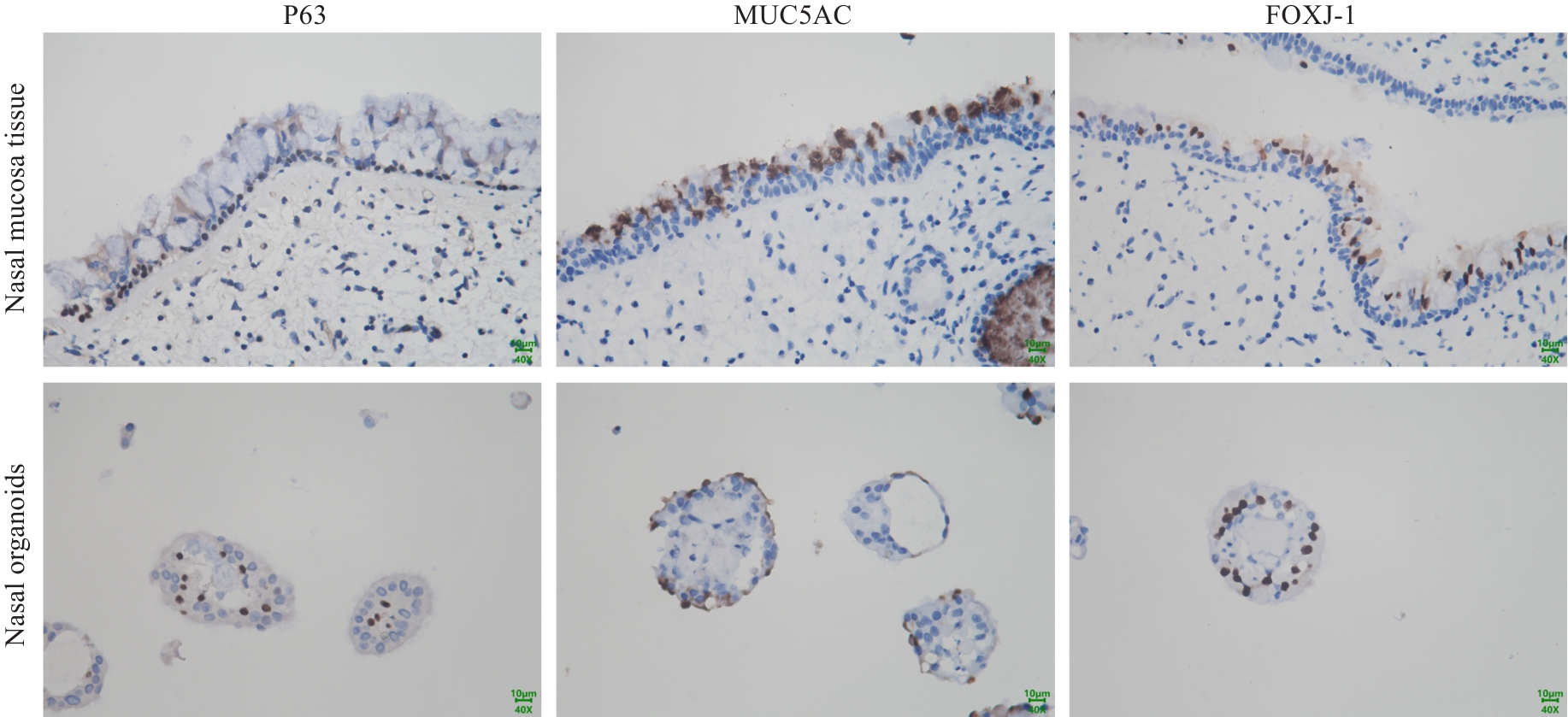
Fig.4 Immunohistochemical analysis of cellular composition of nasal mucosal tissue and organoids. Representative images of nasal mucosal tissue are shown in the upper panel and differentiated nasal organoids in the lower panel. The organoids exhibit comparable expression patterns to nasal mucosal tissue for P63 (basal cells), MUC5AC (goblet cells), and FOXJ-1 (ciliated cells).
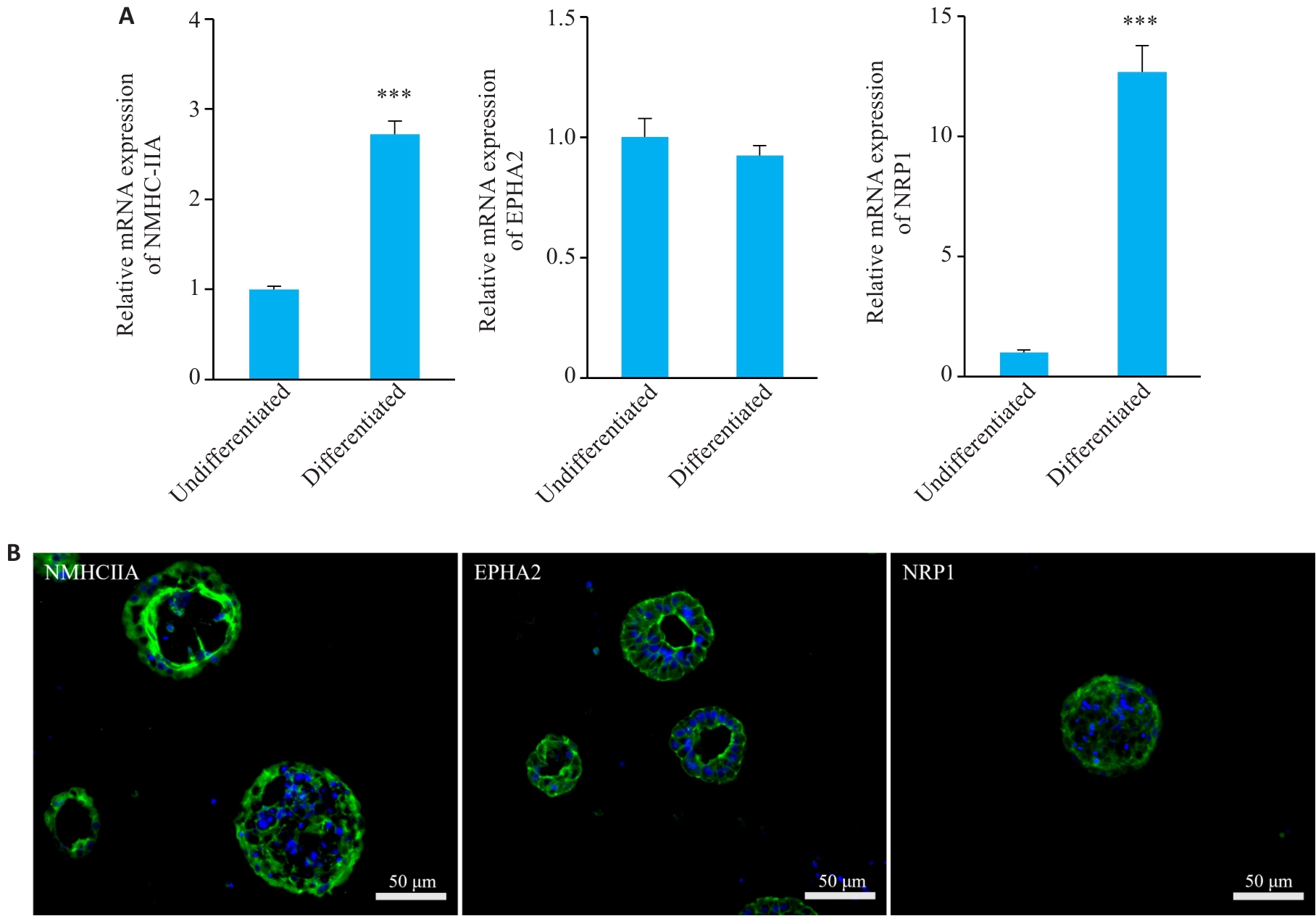
Fig.5 Differentiated nasal organoids exhibit high expressions of Epstein-Barr virus (EBV) receptors. A: Relative mRNA expressions of EBV receptors NMHC-IIA, EPHA2 and NRP1 in undifferentiated and differentiated nasal organoids. ***P<0.001 vs undifferentiated group. B: Immunofluorescence staining for EBV receptors NMHC-IIA, EPHA2 and NRP1 in differentiated nasal organoids (n=3).
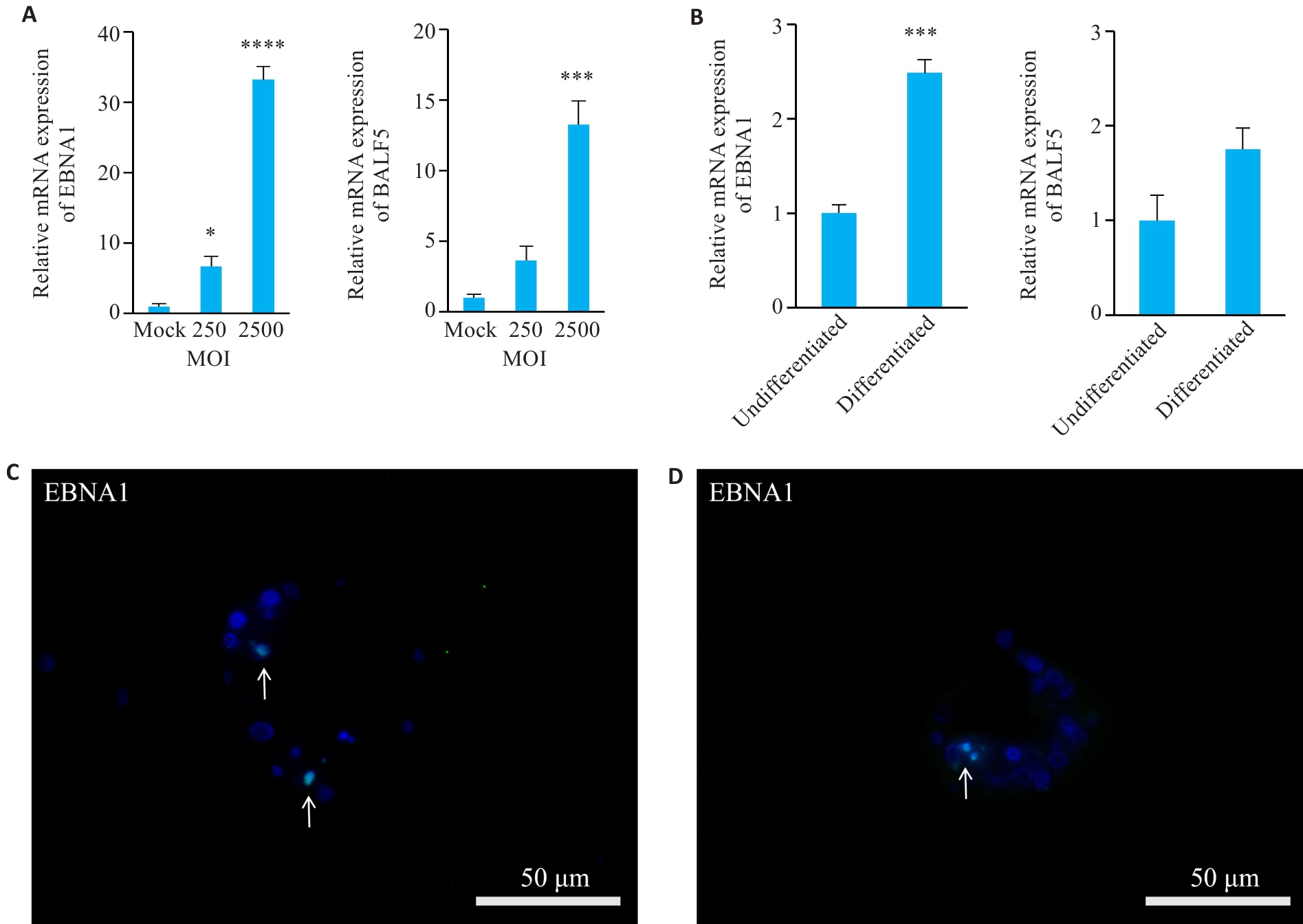
Fig.6 Establishment of EBV-infected nasal organoids. A, B: Relative mRNA expression levels of EBV-specific genes EBNA1 and BALF5 in differentiated organoids harvested at 72 h post-EBV infection at MOI=250 and 2500 (A; *P<0.05, ***P<0.001, ****P<0.0001 vs Mock group) and in both undifferentiated and differentiated organoids harvested 72 h after EBV infection at MOI=2500 (B; ***P<0.001 vs undifferentiated group). C, D: Immunofluorescence staining for EBNA1 undifferentiated (C) and differentiated (D) organoids with EBV infection at MOI=2500 (n=3).
| [1] | Alsaadawe M, Radman BA, Long JY, et al. Epstein Barr virus: a cellular hijacker in cancer[J]. Biochim Biophys Acta BBA Rev Cancer, 2024, 1879(6): 189218. doi:10.1016/j.bbcan.2024.189218 |
| [2] | Damania B, Kenney SC, Raab-Traub N. Epstein-Barr virus: Biology and clinical disease[J]. Cell, 2022, 185(20): 3652-70. doi:10.1016/j.cell.2022.08.026 |
| [3] | Schindele A, Holm A, Kraft S, et al. Cross-evaluating Epstein-Barr virus, human Papilloma virus, human cytomegalovirus and human adenovirus in nasal polyps and turbinate mucosa[J]. Acta Otolaryngol, 2025, 145(2): 164-7. doi:10.1080/00016489.2024.2445025 |
| [4] | Zaravinos A, Bizakis J, Spandidos DA. Prevalence of human Papilloma virus and human herpes virus types 1-7 in human nasal polyposis[J]. J Med Virol, 2009, 81(9): 1613-9. doi:10.1002/jmv.21534 |
| [5] | 谭学君, 姚文昊, 陈晓平, 等.三峡库区重庆万州段鼻息肉与HPV及EBV阳性表达的相关性研究[J].中国中西医结合耳鼻咽喉科杂志,2015, 23(1): 7-11. |
| [6] | Jia Z, Zhang D, Zhu L, et al. Animal models of human herpesvirus infection[J]. Animal Model Exp Med, 2025, 8(4): 615-28. doi:10.1002/ame2.12575 |
| [7] | Huang PY, Zeng TT, Li MQ, et al. Proteomic analysis of a nasopharyngeal carcinoma cell line and a nasopharyngeal epithelial cell line[J]. Tumori, 2015, 101(6): 676-83. doi:10.5301/tj.5000345 |
| [8] | Li HM, Man C, Jin Y, et al. Molecular and cytogenetic changes involved in the immortalization of nasopharyngeal epithelial cells by telomerase[J]. Int J Cancer, 2006, 119(7): 1567-76. doi:10.1002/ijc.22032 |
| [9] | Song LB, Zeng MS, Liao WT, et al. Bmi-1 is a novel molecular marker of nasopharyngeal carcinoma progression and immortalizes primary human nasopharyngeal epithelial cells[J]. Cancer Res, 2006, 66(12): 6225-32. doi:10.1158/0008-5472.can-06-0094 |
| [10] | Collett S, Torresi J, Silveira LE, et al. Investigating virus–host cell interactions: Comparative binding forces between hepatitis C virus-like particles and host cell receptors in 2D and 3D cell culture models[J]. J Colloid Interface Sci, 2021, 592: 371-84. doi:10.1016/j.jcis.2021.02.067 |
| [11] | Yan HHN, Chan AS, Lai FP, et al. Organoid cultures for cancer modeling[J]. Cell Stem Cell, 2023, 30(7): 917-37. doi:10.1016/j.stem.2023.05.012 |
| [12] | Zhu S, Chen D, Yang X, et al. Organoid models to study human infectious diseases[J]. Cell Prolif, 2025: e70004. doi:10.1111/cpr.70004 |
| [13] | 汪珂,于言,韩日,等.分化可控的人类鼻粘膜类器官模型的建立[J].南方医科大学学报,2022,42(06):868-77. |
| [14] | 于言,曹浚垣, 刘 蓉, 等.人鼻粘膜类器官冠状病毒感染模型可用于抗病毒药物的筛选和评价[J].南方医科大学学报,2024,44(11):2227-34. |
| [15] | Kaur S, Kaur I, Rawal P, et al. Non-matrigel scaffolds for organoid cultures[J]. Cancer Lett, 2021, 504: 58-66. doi:10.1016/j.canlet.2021.01.025 |
| [16] | Co JY, Margalef-Català M, Monack DM, et al. Controlling the polarity of human gastrointestinal organoids to investigate epithelial biology and infectious diseases[J]. Nat Protoc, 2021, 16(11): 5171-92. doi:10.1038/s41596-021-00607-0 |
| [17] | Chiu MC, Li C, Liu X, et al. Human nasal organoids model SARS-CoV-2 upper respiratory infection and recapitulate the differential infectivity of emerging variants[J]. mBio, 2022, 13(4): e0194422. doi:10.1128/mbio.01944-22 |
| [18] | Xia TL, Li XY, Wang XP, et al. N(6)‑methyladenosine-binding protein YTHDF1 suppresses EBV replication and promotes EBV RNA decay[J]. EMBO Rep, 2021, 22(4): e50128. doi:10.15252/embr.202050128 |
| [19] | Zhong LY, Xie C, Zhang LL, et al. Research landmarks on the 60th anniversary of Epstein-Barr virus[J]. Sci China Life Sci, 2025, 68(2): 354-80. doi:10.1007/s11427-024-2766-0 |
| [20] | 谭德重, 冯 源, 胡 月, 等. 病毒感染与鼻息肉关系的Meta分析[J].临床耳鼻咽喉头颈外科杂志,2018, 32(12): 910-6. |
| [21] | Wong KCW, Hui EP, Lo KW, et al. Nasopharyngeal carcinoma: an evolving paradigm[J]. Nat Rev Clin Oncol, 2021, 18(11): 679-95. doi:10.1038/s41571-021-00524-x |
| [22] | Tsang CM, Zhang G, Seto E, et al. Epstein-Barr virus infection in immortalized nasopharyngeal epithelial cells: regulation of infection and phenotypic characterization[J]. Int J Cancer, 2010, 127(7): 1570-83. doi:10.1002/ijc.25173 |
| [23] | Wang HB, Zhang H, Zhang JP, et al. Neuropilin 1 is an entry factor that promotes EBV infection of nasopharyngeal epithelial cells[J]. Nat Commun, 2015, 6: 6240. doi:10.3410/f.725353089.793504525 |
| [24] | Kozlowski MT, Crook CJ, Ku HT. Towards organoid culture without Matrigel [J]. Commun Biol, 2021, 4(1): 1387. doi:10.1038/s42003-021-02910-8 |
| [25] | Marchini A, Gelain F. Synthetic scaffolds for 3D cell cultures and organoids: applications in regenerative medicine[J]. Crit Rev Biotechnol, 2022, 42(3): 468-86. doi:10.1080/07388551.2021.1932716 |
| [26] | Capeling MM, Huang S, Childs CJ, et al. Suspension culture promotes serosal mesothelial development in human intestinal organoids[J]. Cell Rep, 2022, 38(7): 110379. doi:10.1016/j.celrep.2022.110379 |
| [27] | Kumar SV, Er PX, Lawlor KT, et al. Kidney micro-organoids in suspension culture as a scalable source of human pluripotent stem cell-derived kidney cells[J]. Development, 2019, 146(5): dev172361. doi:10.1242/dev.172361 |
| [28] | Wu H, Wang J, Liu S, et al. Large-scale production of expandable hepatoblast organoids and polarised hepatocyte organoids from hESCs under 3D static and dynamic suspension conditions[J]. Cell Prolif, 2025, 58(7): e70001. doi:10.1111/cpr.70001 |
| [29] | Stroulios G, Brown T, Moreni G, et al. Apical-out airway organoids as a platform for studying viral infections and screening for antiviral drugs[J]. Sci Rep, 2022, 12(1): 7673. doi:10.1038/s41598-022-11700-z |
| [30] | Zhao KY, Du YX, Cao HM, et al. The biological macromolecules constructed Matrigel for cultured organoids in biomedical and tissue engineering[J]. Colloids Surf B Biointerfaces, 2025, 247: 114435. doi:10.1016/j.colsurfb.2024.114435 |
| [31] | Bu GL, Xie C, Kang YF, et al. How EBV infects: the tropism and underlying molecular mechanism for viral infection[J]. Viruses, 2022, 14(11): 2372. doi:10.3390/v14112372 |
| [32] | Hayman IR, Temple RM, Burgess CK, et al. New insight into Epstein-Barr virus infection using models of stratified epithelium[J]. PLoS Pathog, 2023, 19(1): e1011040. doi:10.1371/journal.ppat.1011040 |
| [33] | Nawandar DM, Wang A, Makielski K, et al. Differentiation-dependent KLF4 expression promotes lytic Epstein-Barr virus infection in epithelial cells[J]. PLoS Pathog, 2015, 11(10): e1005195. doi:10.1371/journal.ppat.1005195 |
| [1] | Xue GONG, Yongyang FAN, Kaiyuan LUO, Yi YAN, Zhonghao LI. Construction of cardiac organoids derived from human induced pluripotent stem cells for cardiac disease modeling and drug evaluation [J]. Journal of Southern Medical University, 2025, 45(11): 2444-2455. |
| [2] | Yan YU, Junyuan CAO, Rong LIU, Minmin ZHOU, Jinyan WEI, Hairui ZHENG, Wei WANG, Gang LI. Development of a new platform for testing antiviral drugs using coronavirus-infected human nasal mucosa organoids [J]. Journal of Southern Medical University, 2024, 44(11): 2227-2234. |
| [3] | WANG Ke, YU Yan, HAN Ri, WANG Xianwen, ZHAO Yunteng, TANG Haocheng, LI Gang. Establishment of a culture system for human nasal mucosa organoids with controllable differentiation [J]. Journal of Southern Medical University, 2022, 42(6): 868-877. |
| [4] | . Effect of deoxycholic acid intervention on growth of ileum organoids derived from C57BL/6 mice [J]. Journal of Southern Medical University, 2017, 37(01): 6-. |
| [5] | . Effects of Epstein-Barr virus and cytomegalovirus infection on childhood acute lymphoblastic leukemia gene methylation [J]. Journal of Southern Medical University, 2013, 33(11): 1678-. |
| [6] | SONG Mei,GAO Jian-hua,YAN Xin,LIU Xiao-jun,CHEN Yang Department of Plastic Surgery,Nanfang Hospital,Southern Medical University,Guangzhou 510515,China. Establishment of immortalized B-lymphoblastoid cell lines of keloid pedigree and its karyotype analysis [J]. Journal of Southern Medical University, 2006, 26(12): 1760-1762. |
| [7] | LI Ang1,2, ZHAO Song1, HU Wei1, ZHAO Gao-feng1, ZHANG Xiao-shi2 1Department of Thoracic Surgery, First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, China; 2Cancer Institute, Oncology Center, Sun Yat-sen University, Guangzhou 510060, China. Cloning of A73 gene and its coding sequence analysis [J]. Journal of Southern Medical University, 2006, 26(06): 826-. |
| [8] | LIU Wei, HAN Hui-xia Department of Pathology, Southern Medical University, Guangzhou 510515, China. Expression of CD25~+ lymphocytes in nasopharyngeal carcinoma and its association with EBV infection [J]. Journal of Southern Medical University, 2006, 26(01): 94-97. |
| [9] | YAN Xin, GAO Jian-hua, SONG Mei, LIU Xiao-jun, CHEN Yang Department of Plastic Surgery, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China. Establishment of immortalized lymphoblastoid cell bank of a keloid pedigree [J]. Journal of Southern Medical University, 2006, 26(01): 22-24. |
| [10] | WU Wei-qiu1, HUANG Sheng-guang2, MA Zong1, ZHANG Yong-quan1, SHI Jian-bo3. Effects of irrigating solution of Sihuang on morphology and function of nasal mucosa following surgery for chronic sinusitis and nasal polyps [J]. Journal of Southern Medical University, 2005, 25(04): 424-427. |
| [11] | WANG Shuang, LÜ Li-chun, LI Hong, JIANG Pei-zhou, YAO Kai-tai. Difference in gene expression profile of human polymeric immunoglobulin receptor-transfected mouse nasopharyngeal epithelial cells before and after EBV infection [J]. Journal of Southern Medical University, 2004, 24(09): 1001-1005. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||