南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 570-581.doi: 10.12122/j.issn.1673-4254.2026.03.11
• 基础研究 • 上一篇
刘泽1( ), 毛樟坤2, 尤达3, 王俊杰4, 何咏梅1, 余伊雯2, 文志强2, 方会龙2(
), 毛樟坤2, 尤达3, 王俊杰4, 何咏梅1, 余伊雯2, 文志强2, 方会龙2( ), 何汶霞1(
), 何汶霞1( )
)
收稿日期:2025-08-16
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
方会龙,何汶霞
E-mail:liuze0113@xnu.edu.cn;fanghuilong@xnu.edu.cn;602743915@qq.com
作者简介:刘 泽,博士,副教授,E-mail: liuze0113@xnu.edu.cn
基金资助:
Ze LIU1( ), Zhangkun MAO2, Da YOU3, Junjie WANG4, Yongmei HE1, Yiwen YU2, Zhiqiang WEN2, Huilong FANG2(
), Zhangkun MAO2, Da YOU3, Junjie WANG4, Yongmei HE1, Yiwen YU2, Zhiqiang WEN2, Huilong FANG2( ), Wenxia HE1(
), Wenxia HE1( )
)
Received:2025-08-16
Online:2026-03-20
Published:2026-03-26
Contact:
Huilong FANG, Wenxia HE
E-mail:liuze0113@xnu.edu.cn;fanghuilong@xnu.edu.cn;602743915@qq.com
摘要:
目的 探究宽缨酮(EN)减轻缺血再灌注(IR)诱导的小鼠急性肾损伤(AKI)的作用机制。 方法 24只雄性C57BL/6J小鼠随机分为Sham、IR及EN低、高剂量组4组(6只/组)。双侧肾蒂IR处理构建AKI模型,EN低、高剂量组在术前2 d腹腔注射EN(0.25、1.0 mg·kg-1·d-1)直至组织取材前1 d。采用网络药理学分析EN作用通路和分子机制,采用分子对接分析EN与靶点的结合性能。检测小鼠肾功能、肾组织病理、肾小管损伤指标(KIM-1、NGAL)、p-STAT3、p-PI3K及p-JAK2的表达、EN与STAT3相互作用、炎症及线粒体生物发生及其功能。另将24只雄性C57BL/6J小鼠随机分为:Sham、IR、IR+EN和IR+EN+ML115(STAT3激动剂)组4组(6只/组),分析STAT3在EN减轻IR诱导的AKI中的作用机制。 结果 低、高剂量EN均减轻小鼠肾组织损伤、改善肾功能,包括降低血肌酐、尿素氮、肾小管损伤评分及下调KIM-1和NGAL的表达(P<0.05)。网络药理学分析发现EN核心靶点富集信号通路与炎症密切相关。低、高剂量EN均降低AKI小鼠肾组织炎症因子(IL-6、MCP-1、TNF-α)的水平及肾间质巨噬细胞浸润(P<0.05),增加肾组织ATP含量及线粒体DNA拷贝数(P<0.05),并上调PGC-1α、TFAM、Nrf2的基因表达以及PGC-1α、TOM20的蛋白表达(P<0.05)。基于分子对接结果筛选出STAT3和PI3K为EN减轻AKI的关键靶点。动物实验结果显示,EN抑制AKI小鼠肾组织p-STAT3和p-PI3K表达(P<0.05),但不影响p-JAK2的表达(P>0.05)。表面等离子共振实验证实EN特异性结合STAT3。ML115可逆转EN对AKI小鼠的肾功能、肾组织病理损伤、肾小管损伤及炎症反应的保护作用(P<0.05)。 结论 EN通过靶向抑制STAT3促进线粒体生物发生,减轻肾脏线粒体功能损伤及炎症反应,从而缓解IR诱导的AKI。
刘泽, 毛樟坤, 尤达, 王俊杰, 何咏梅, 余伊雯, 文志强, 方会龙, 何汶霞. 宽缨酮靶向抑制STAT3减轻线粒体功能障碍和炎症缓解急性肾损伤[J]. 南方医科大学学报, 2026, 46(3): 570-581.
Ze LIU, Zhangkun MAO, Da YOU, Junjie WANG, Yongmei HE, Yiwen YU, Zhiqiang WEN, Huilong FANG, Wenxia HE. Eurycomanone inhibits renal ischemia/reperfusion-induced mitochondrial dysfunction and inflammation in mice by binding to STAT3 to inhibit its phosphorylation[J]. Journal of Southern Medical University, 2026, 46(3): 570-581.
| Gene name | Forward (5′-3′) | Reverse (5′-3′) |
|---|---|---|
| NGAL | ATGTCACCTCCATCCTGGTCAG | GCCACTTGCACATTGTAGCTCTG |
| KIM-1 | ACATATCGTGGAATCACAACGAC | ACTGCTCTTCTGATAGGTGACA |
| TNF-α | CAGGCGGTGCCTATGTCTC | CGATCACCCCGAAGTTCAGTAG |
| MCP-1 | TAAAAACCTGGATCGGAACCAAA | GCATTAGCTTCAGATTTACGGGT |
| IL-6 | CTGCAAGAGACTTCCATCCAG | AGTGGTATAGACAGGTCTGTTGG |
| PGC-1α | TGAACGCACCTTAAGTGTGGAA | GGGTTATCTTGGTTGGCTTTATGA |
| TFAM | CACCCAGATGCAAAACTTTCAG | CTGCTCTTTATACTTGCTCACAG |
| Nrf2 | AAAGCACAGCCAGCACATTC | GGGATTCACGCATAGGAGCA |
| GAPDH | GGTGAAGGTCGGTGTGAACG | CTCGCTCCTGGAAGATGGTG |
| ND1 | GGATCCGAGCATCTTATCCA | GGTGGTACTCCCGCTGTAAA |
| S18 | TTCCAGCACATTTTGCGAGTA | CACGCCCTTAATGGCAGTGAT |
表1 基因引物序列
Tab.1 Primer sequence for qRT-PCR
| Gene name | Forward (5′-3′) | Reverse (5′-3′) |
|---|---|---|
| NGAL | ATGTCACCTCCATCCTGGTCAG | GCCACTTGCACATTGTAGCTCTG |
| KIM-1 | ACATATCGTGGAATCACAACGAC | ACTGCTCTTCTGATAGGTGACA |
| TNF-α | CAGGCGGTGCCTATGTCTC | CGATCACCCCGAAGTTCAGTAG |
| MCP-1 | TAAAAACCTGGATCGGAACCAAA | GCATTAGCTTCAGATTTACGGGT |
| IL-6 | CTGCAAGAGACTTCCATCCAG | AGTGGTATAGACAGGTCTGTTGG |
| PGC-1α | TGAACGCACCTTAAGTGTGGAA | GGGTTATCTTGGTTGGCTTTATGA |
| TFAM | CACCCAGATGCAAAACTTTCAG | CTGCTCTTTATACTTGCTCACAG |
| Nrf2 | AAAGCACAGCCAGCACATTC | GGGATTCACGCATAGGAGCA |
| GAPDH | GGTGAAGGTCGGTGTGAACG | CTCGCTCCTGGAAGATGGTG |
| ND1 | GGATCCGAGCATCTTATCCA | GGTGGTACTCCCGCTGTAAA |
| S18 | TTCCAGCACATTTTGCGAGTA | CACGCCCTTAATGGCAGTGAT |
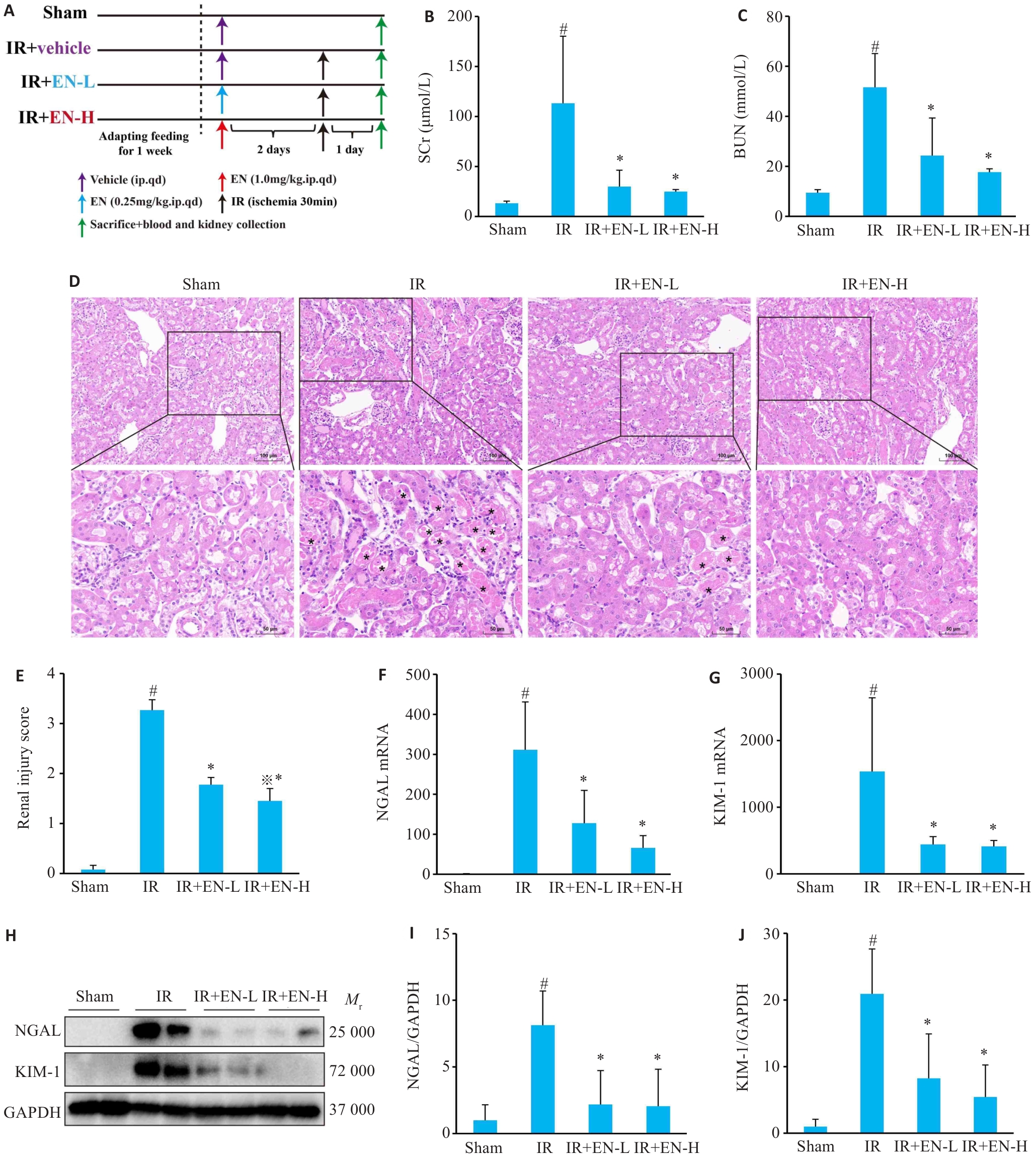
图1 EN减轻IR诱导的小鼠AKI
Fig.1 EN pretreatment alleviates ischemia/reperfusion (IR)-induced acute kidney injury (AKI) in mice. A: Schematic diagram of the animal study protocol. B, C: SCr and BUN levels in each group. D: HE staining showing the renal injury in the mice. The lower panels (Scale bar=50 μm) are magnified images of the boxed areas in the upper panels (Scale bar=100 μm). Asterisks indicate damaged tubules. E: Quantitative assessment of renal injury. F, G: qRT-PCR for detecting mRNA levels of KIM-1 and NGAL in the renal tissues. H-J: Western blotting for KIM-1 and NGAL expressions. #P<0.05 vs sham group, *P<0.05 vs IR group, ※P<0.05 vs IR+EN-L group (n=6).
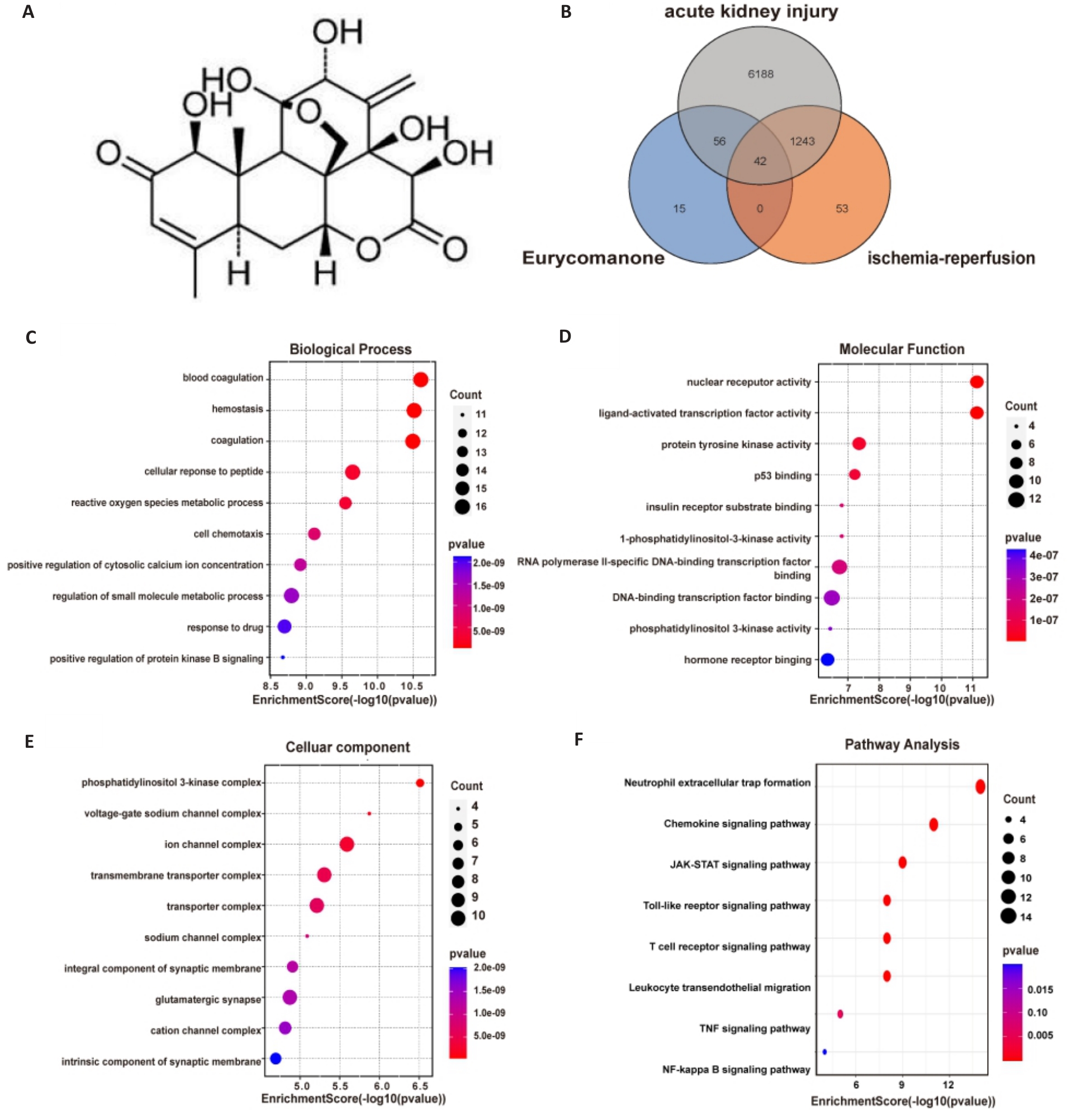
图2 交集靶点的GO和KEGG富集分析
Fig.2 GO and KEGG enrichment analysis of the intersection targets. A: Molecular structure of EN. B: Venn diagram of the EN targets, IR targets and AKI targets. C-E: Top 10 items for the biological process (BP), molecular functional (MF) and cellular component (CC) enrichment analysis in GO enrichment analysis. F: The top 8 inflammatory enrichment signaling pathways from KEGG pathway enrichment analysis.
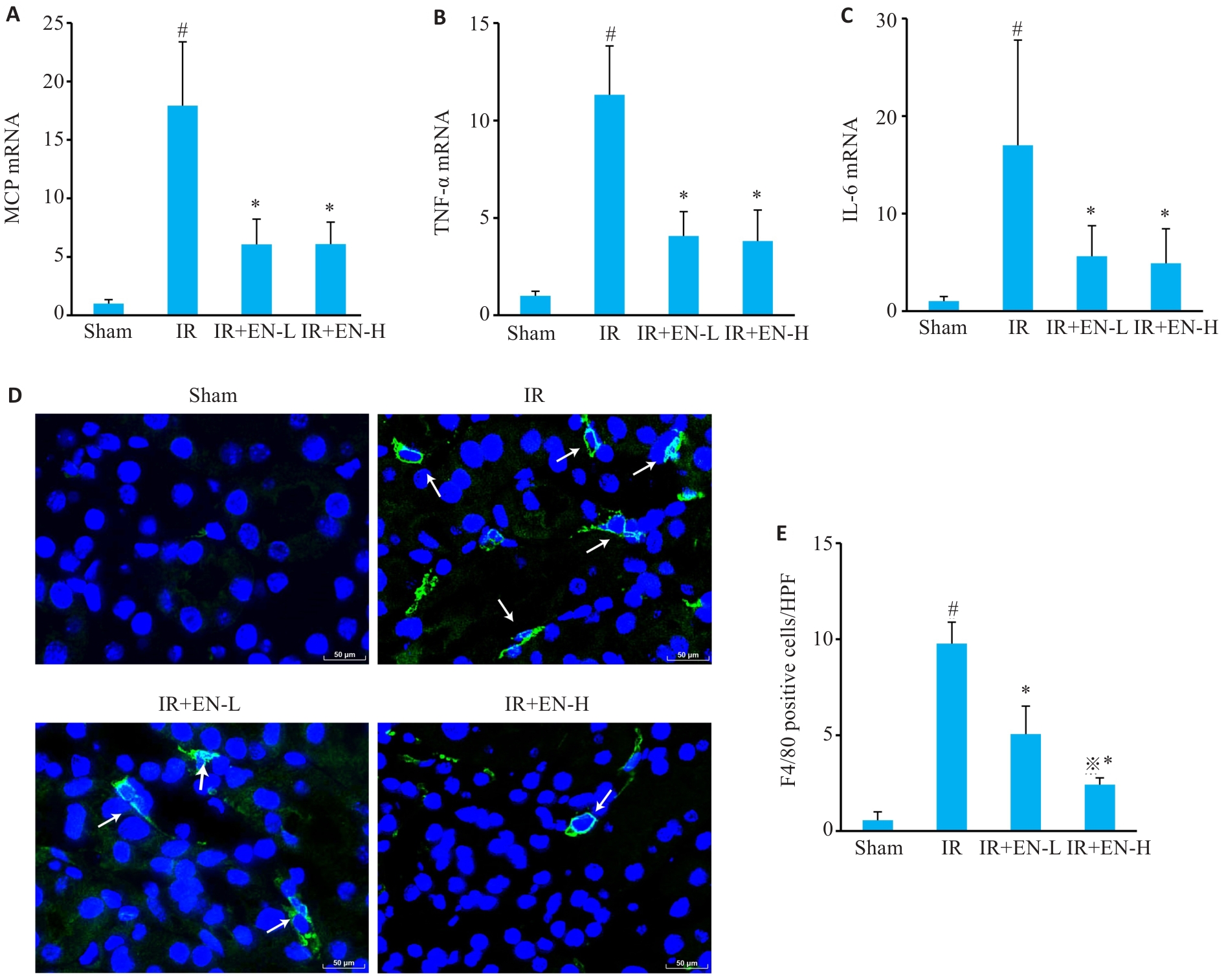
图3 EN抑制IR-AKI小鼠肾脏炎症
Fig.3 EN suppresses renal inflammation in mice with IR-induced AKI in mice. A-C: qRT-PCR for detecting mRNA levels of MCP-1, TNF‑α and IL-6. D, E: Immunofluorescence staining showed infiltration of F4/80-positvie cells (arrows) in mouse renal tissues (Scale bar=50 μm). #P<0.05 vs sham group, *P<0.05 vs IR group, ※P<0.05 vs IR+EN-L group (n=6).
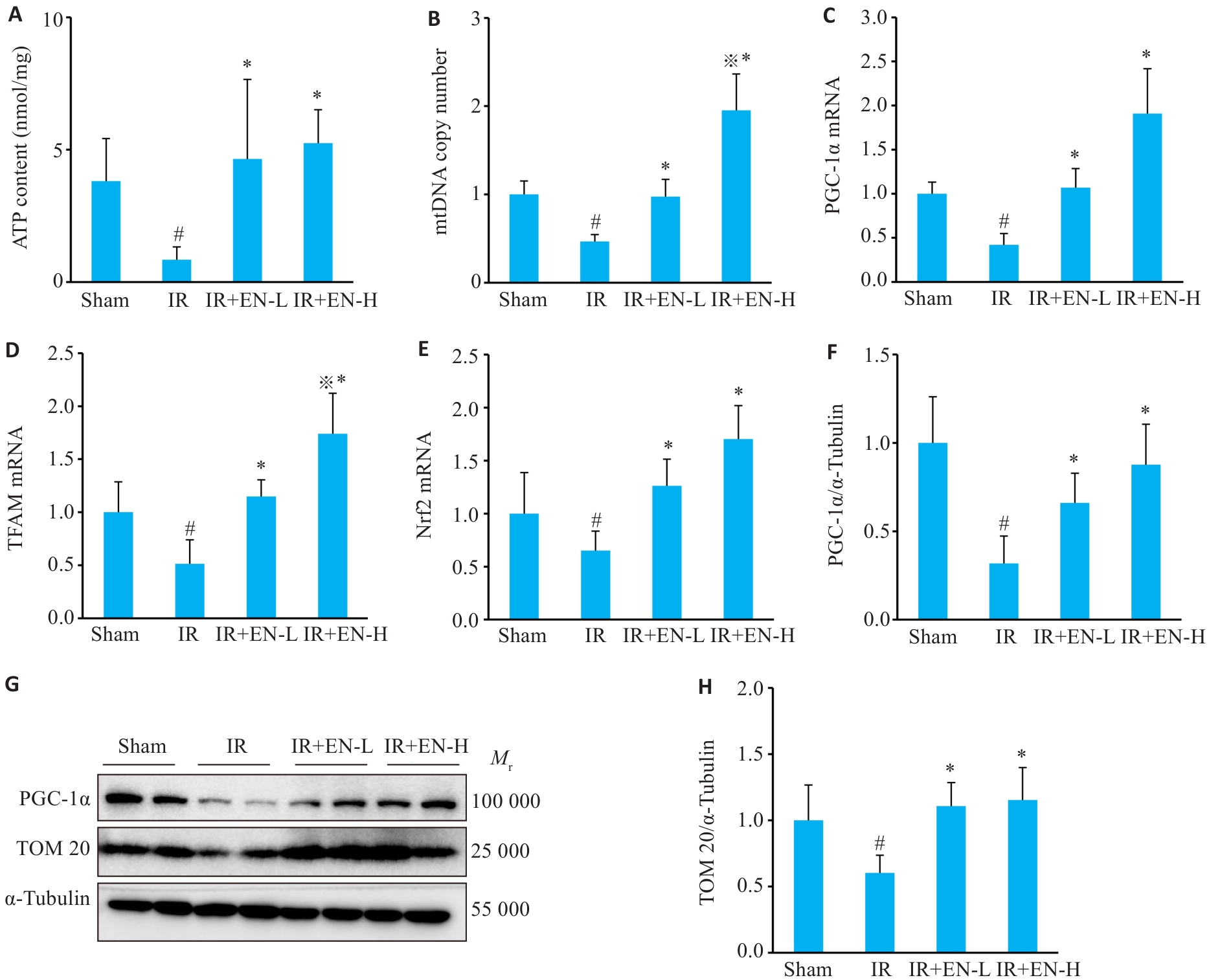
图4 EN通过促进线粒体生物发生改善IR-AKI小鼠肾脏线粒体功能
Fig.4 EN rescues renal mitochondrial function by promoting mitochondrial biogenesis in mice with IR-induced AKI. A: Quantification of ATP content. B: Quantification of mtDNA copy number. C-E: qRT-PCR for detecting mRNA level of PGC-1α, TFAM and Nrf2. F-H: Western blotting for PGC-1α and TOM 20 expressions. #P<0.05 vs sham group, *P<0.05 vs IR group, ※P<0.05 vs IR+EN-L group (n=6).
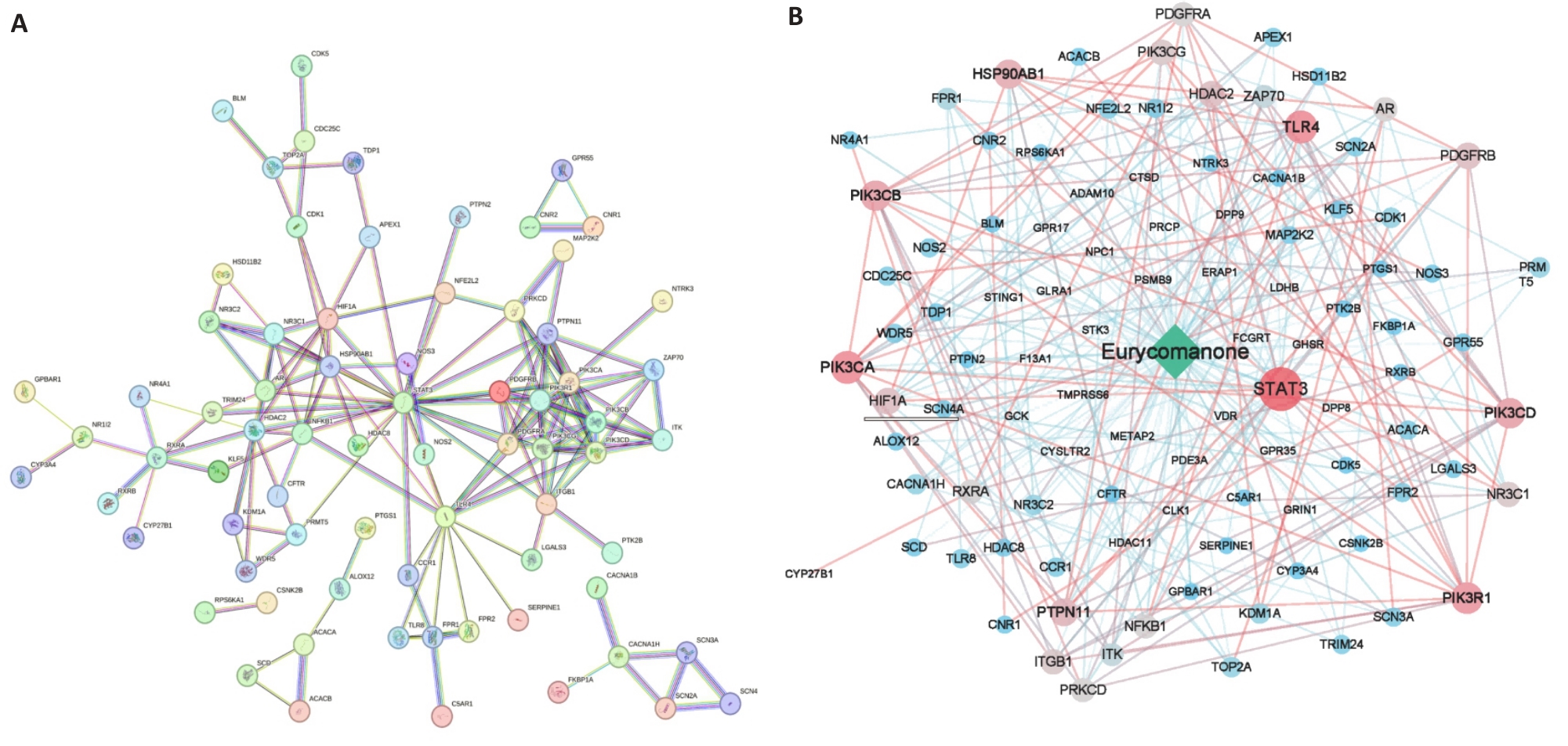
图5 PPI网络与C-T网络的构建
Fig.5 Construction of the protein-protein interaction (PPI) network and compound-target (C-T) network. A: The PPI network among the potential targets of EN against IR-induced AKI. B: The C-T network between EN and the potential targets. Square: EN compound; Circle: Predicted targets.
| Target | Score | Degree | RCSB ID |
|---|---|---|---|
| STAT3 | -7.7 | 24 | 6QHD |
| PIK3CA | -9.1 | 15 | 8BFU |
| TLR4 | -6.7 | 15 | 2Z62 |
| PIK3R1 | -6.8 | 14 | 1PHT |
| PIK3CB | -9.0 | 13 | 2Y2A |
| PIK3CD | -8.2 | 13 | 5IS5 |
| HSP90AB1 | -7.4 | 12 | 2NMQ |
| PTPN11 | -8.7 | 11 | 3O5X |
| HIF1A | -7.9 | 10 | 1H2K |
| HDAC2 | -9.2 | 10 | 4LXZ |
| PDGFRB | -6.7 | 10 | 1H9O |
| ITGB1 | -8.1 | 9 | 4DX9 |
| NR3C1 | -7.4 | 9 | 3BQD |
| PIK3CG | -8.6 | 9 | 2CHW |
| PDGFRA | -7.5 | 8 | 5GRN |
| RXRA | -7.3 | 8 | 4N5G |
| PRKCD | -7.0 | 8 | 1YRK |
| NFKB1 | -7.2 | 8 | 7LEQ |
| AR | -6.6 | 8 | 1E3G |
表2 EN与核心靶点分子对接结果
Tab.2 Molecular docking of EN with the core targets
| Target | Score | Degree | RCSB ID |
|---|---|---|---|
| STAT3 | -7.7 | 24 | 6QHD |
| PIK3CA | -9.1 | 15 | 8BFU |
| TLR4 | -6.7 | 15 | 2Z62 |
| PIK3R1 | -6.8 | 14 | 1PHT |
| PIK3CB | -9.0 | 13 | 2Y2A |
| PIK3CD | -8.2 | 13 | 5IS5 |
| HSP90AB1 | -7.4 | 12 | 2NMQ |
| PTPN11 | -8.7 | 11 | 3O5X |
| HIF1A | -7.9 | 10 | 1H2K |
| HDAC2 | -9.2 | 10 | 4LXZ |
| PDGFRB | -6.7 | 10 | 1H9O |
| ITGB1 | -8.1 | 9 | 4DX9 |
| NR3C1 | -7.4 | 9 | 3BQD |
| PIK3CG | -8.6 | 9 | 2CHW |
| PDGFRA | -7.5 | 8 | 5GRN |
| RXRA | -7.3 | 8 | 4N5G |
| PRKCD | -7.0 | 8 | 1YRK |
| NFKB1 | -7.2 | 8 | 7LEQ |
| AR | -6.6 | 8 | 1E3G |
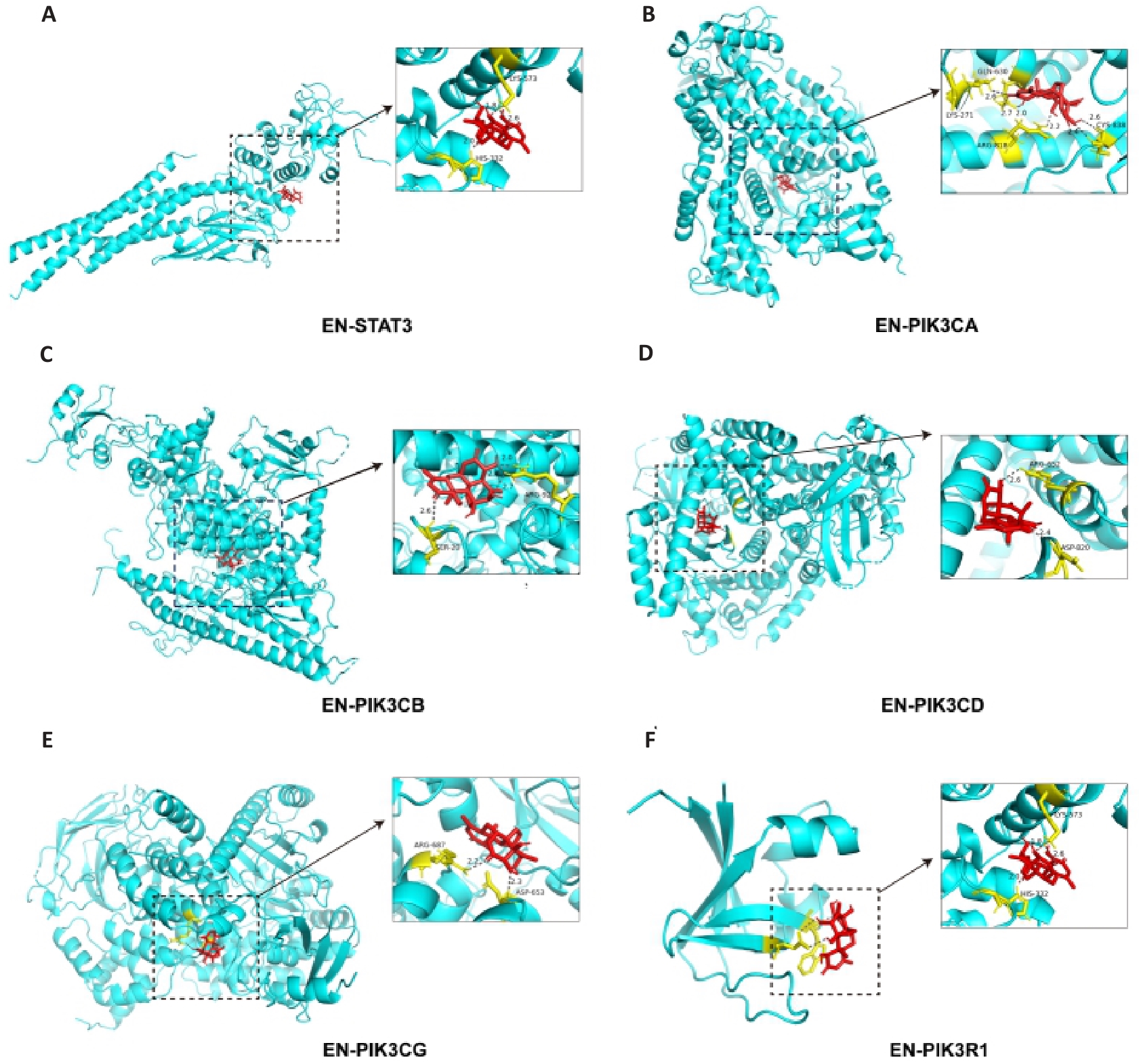
图6 EN与核心靶点的分子对接可视化
Fig.6 Visualization of molecular docking between the core targets and EN. A: Schematic diagram of molecular docking between EN and STAT3. B-F: Schematic diagram of molecular docking between EN with different subunits of PI3K, including PIK3CA, PIK3CB, PIK3CD, PIK3CG and PIK3R1.
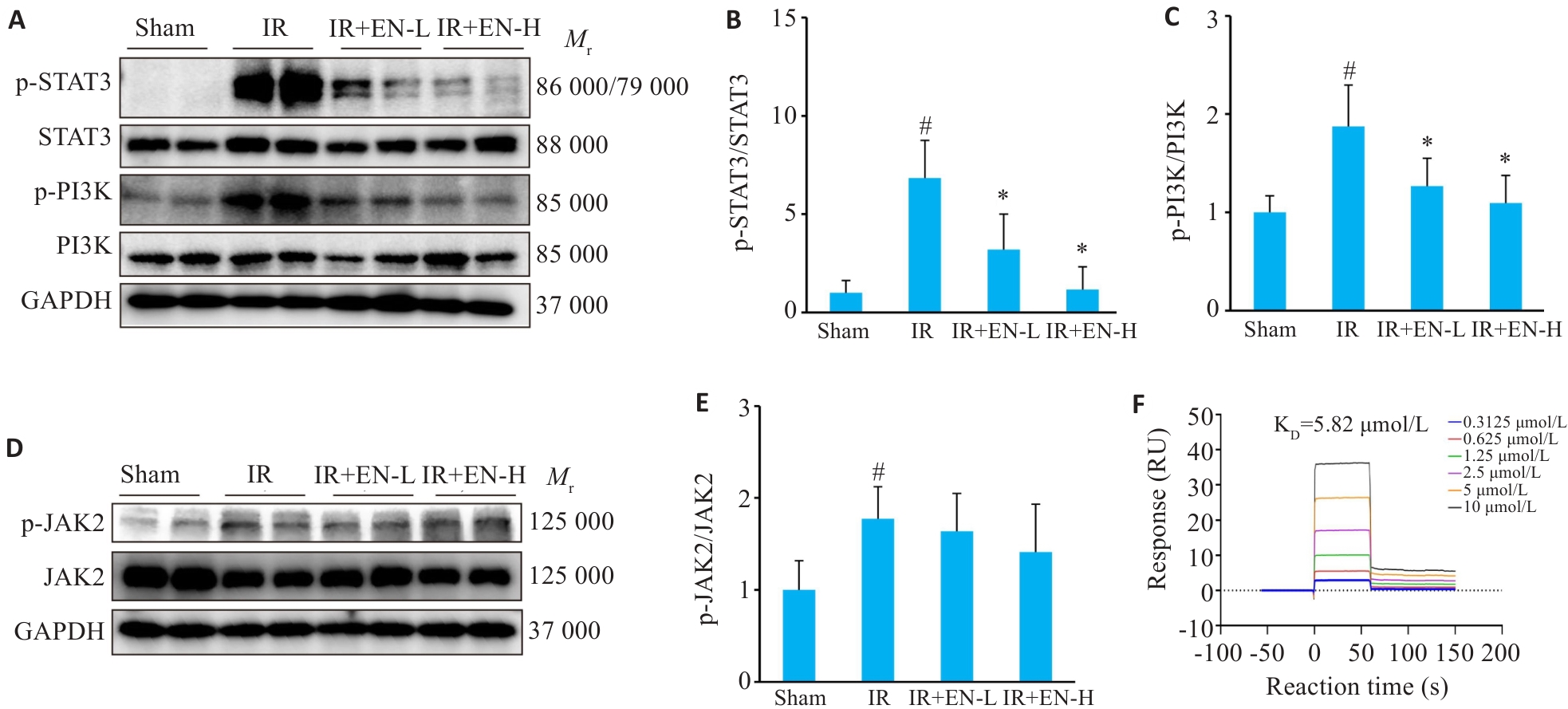
图7 EN直接靶向STAT3并抑制其磷酸化
Fig.7 EN suppresses phosphorylation of STAT3 by binding to STAT3. A-C: Western blotting for p-STAT3, STAT3, p-PI3K and PI3K expressions in the renal tissues. D-E: Western blotting for p-JAK2 and JAK2 expressions in the renal tissues. #P<0.05 vs sham group, *P<0.05 vs IR group, ※P<0.05 vs IR+EN-L group (n=6). F: SPR test using Biacore demonstrating the stable fit of the interaction between EN and STAT3.
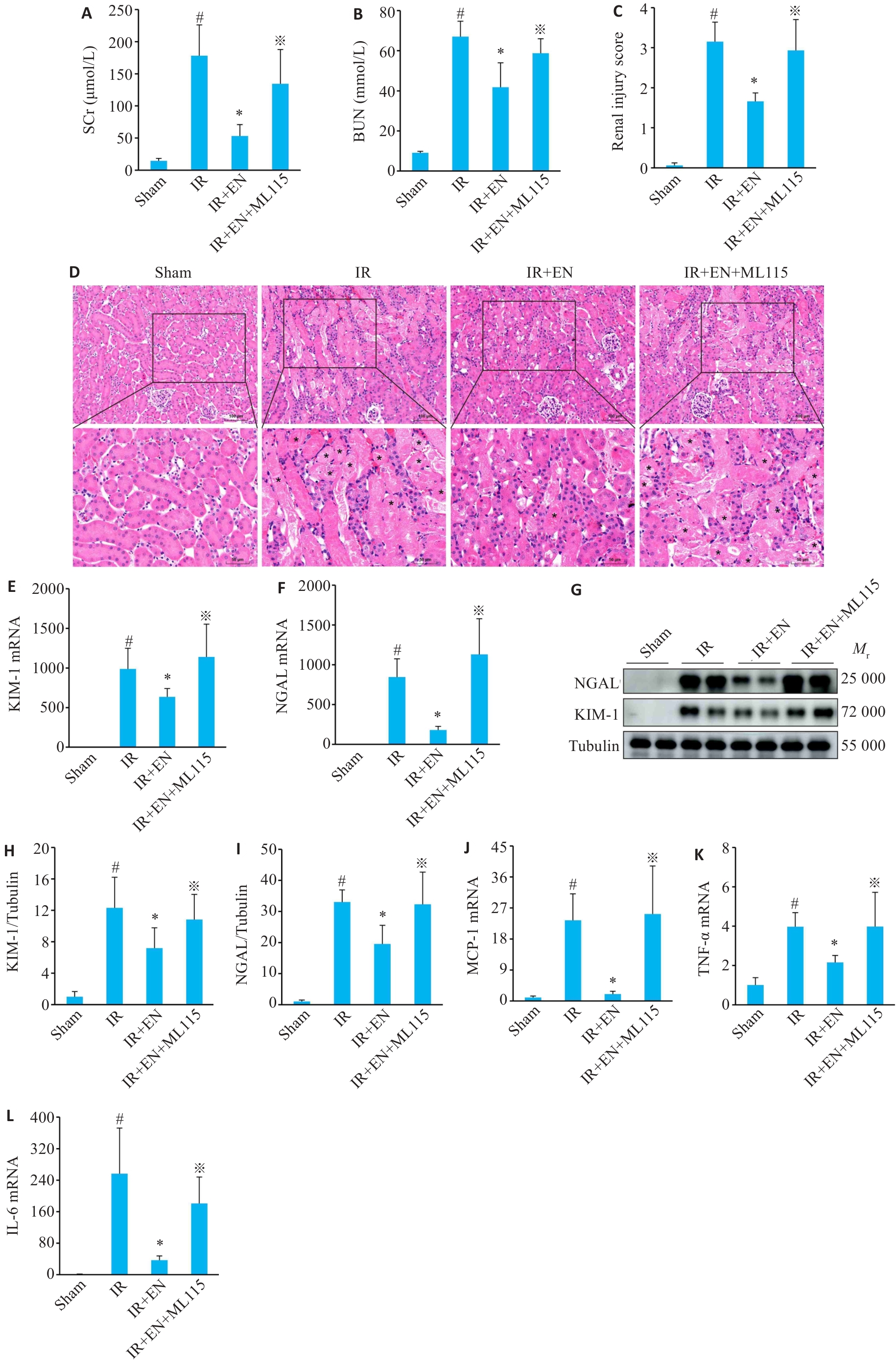
图8 STAT3激动剂ML115能够逆转EN对IR-AKI小鼠的肾损伤及炎症反应
Fig.8 STAT3 agonist ML115 reduces EN-mediated renal and anti-inflammatory protection in IR-AKI mice. A, B: SCr and BUN levels in each group. C: Quantitative assessment of renal injury. D: HE staining show the morphological injury. The lower panels (Scale bar=50 μm) are magnified images of the boxed areas in the upper panels (Scale bar=100 μm). Asterisks indicate damaged tubules. E, F: qRT-PCR for detecting mRNA levels of KIM-1 and NGAL in the renal tissues. G-I: Western blotting for KIM-1 and NGAL expressions. J-L: qRT-PCR for detecting mRNA levels of MCP-1, TNF-α and IL-6. #P<0.05 vs sham group, *P<0.05 vs IR group, ※P<0.05 vs IR+EN group (n=6).
| [1] | Kellum JA, Romagnani P, Ashuntantang G, et al. Acute kidney injury[J]. Nat Rev Dis Primers, 2021, 7(1): 52. doi:10.1038/s41572-021-00284-z |
| [2] | Niculae A, Gherghina ME, Peride I, et al. Pathway from acute kidney injury to chronic kidney disease: molecules involved in renal fibrosis[J]. Int J Mol Sci, 2023, 24(18): 14019. doi:10.3390/ijms241814019 |
| [3] | Chen LC, Yuan JX, Li H, et al. Trans-cinnamaldehyde attenuates renal ischemia/reperfusion injury through suppressing inflammation via JNK/p38 MAPK signaling pathway[J]. Int Immunopharmacol, 2023, 118: 110088. doi:10.1016/j.intimp.2023.110088 |
| [4] | Chen YM, Li ZX, Zhang HY, et al. Mitochondrial metabolism and targeted treatment strategies in ischemic-induced acute kidney injury[J]. Cell Death Discov, 2024, 10(1): 69. doi:10.1038/s41420-024-01843-5 |
| [5] | Ma HJ, Guo XZ, Cui SC, et al. Dephosphorylation of AMP-activated protein kinase exacerbates ischemia/reperfusion-induced acute kidney injury via mitochondrial dysfunction[J]. Kidney Int, 2022, 101(2): 315-30. doi:10.1016/j.kint.2021.10.028 |
| [6] | Vijayan A. Tackling AKI: prevention, timing of dialysis and follow-up[J]. Nat Rev Nephrol, 2021, 17(2): 87-8. doi:10.1038/s41581-020-00390-3 |
| [7] | Huang JW, Liang Y, Zhou LL. Natural products for kidney disease treatment: Focus on targeting mitochondrial dysfunction[J]. Front Pharmacol, 2023, 14: 1142001. doi:10.3389/fphar.2023.1142001 |
| [8] | Ahmad N, Samiulla DS, Teh BP, et al. Bioavailability of eurycomanone in its pure form and in a standardised Eurycoma longifolia water extract[J]. Pharmaceutics, 2018, 10(3): 90. doi:10.3390/pharmaceutics10030090 |
| [9] | Yunos NM, Wahab HA, Al-Thiabat MG, et al. In vitro and in silico analysis of the anticancer effects of eurycomanone and euryco-malactone from Eurycoma longifolia [J]. Plants, 2023, 12(15): 2827. doi:10.3390/plants12152827 |
| [10] | Ye GQ, Xu MT, Shu YH, et al. A quassinoid diterpenoid euryco-manone from Eurycoma longifolia jack exerts anti-cancer effect through autophagy inhibition[J]. Molecules, 2022, 27(14): 4398. doi:10.3390/molecules27144398 |
| [11] | Balan D, Chan KL, Murugan D, et al. Antiadipogenic effects of a standardized quassinoids-enriched fraction and eurycomanone from Eurycoma longifolia [J]. Phytother Res, 2018, 32(7): 1332-45. doi:10.1002/ptr.6065 |
| [12] | Zhong YT, Liao HB, Ye ZQ, et al. Eurycomanone stimulates bone mineralization in zebrafish larvae and promotes osteogenic differentiation of mesenchymal stem cells by upregulating AKT/GSK-3β/β-catenin signaling[J]. J Orthop Translat, 2023, 40: 132-46. doi:10.1016/j.jot.2023.05.006 |
| [13] | Liu Z, Li H, Su JQ, et al. Numb depletion promotes Drp1-mediated mitochondrial fission and exacerbates mitochondrial fragmentation and dysfunction in acute kidney injury[J]. Antioxid Redox Signal, 2019, 30(15): 1797-816. doi:10.1089/ars.2017.7432 |
| [14] | Zhao M, Wang YZ, Li L, et al. Mitochondrial ROS promote mitochondrial dysfunction and inflammation in ischemic acute kidney injury by disrupting TFAM-mediated mtDNA maintenance[J]. Theranostics, 2021, 11(4): 1845-63. doi:10.7150/thno.50905 |
| [15] | Chaudhary S, Kashani KB. Acute kidney injury management strategies peri-cardiovascular interventions[J]. Interv Cardiol Clin, 2023, 12(4): 555-72. doi:10.1016/j.iccl.2023.06.008 |
| [16] | McWilliam SJ, Wright RD, Welsh GI, et al. The complex interplay between kidney injury and inflammation[J]. Clin Kidney J, 2020, 14(3): 780-8. doi:10.1093/ckj/sfaa164 |
| [17] | Trang NTH, Trang TT, Nam NT. Cytokine inhibitory activity of eurycomanone in RAW 264.7 cells stimulated with viral-mimicking poly (I: C)[J]. Vietnam J Biotechnol, 2023, 21(3): 435-41. doi:10.15625/1811-4989/20291 |
| [18] | Hajjouli S, Chateauvieux S, Teiten MH, et al. Eurycomanone and eurycomanol from Eurycoma longifolia jack as regulators of signaling pathways involved in proliferation, cell death and inflammation[J]. Molecules, 2014, 19(9): 14649-66. doi:10.3390/molecules190914649 |
| [19] | Feng YS, Imam Aliagan A, Tombo N, et al. RIP3 translocation into mitochondria promotes mitofilin degradation to increase inflammation and kidney injury after renal ischemia-reperfusion[J]. Cells, 2022, 11(12): 1894. doi:10.3390/cells11121894 |
| [20] | Abu Shelbayeh O, Arroum T, Morris S, et al. PGC-1α is a master regulator of mitochondrial lifecycle and ROS stress response[J]. Antioxidants, 2023, 12(5): 1075. doi:10.3390/antiox12051075 |
| [21] | Ma LY, Hui JL, He GT, et al. DNAJC6 in acute kidney injury: a novel target for protecting renal tubular epithelial cells through PGC-1α-mediated mitochondrial homeostasis[J]. Exp Cell Res, 2025, 450(2): 114682. doi:10.1016/j.yexcr.2025.114682 |
| [22] | Yu JT, Fan S, Li XY, et al. Novel insights into STAT3 in renal diseases[J]. Biomed Pharmacother, 2023, 165: 115166. doi:10.1016/j.biopha.2023.115166 |
| [23] | Zhao XY, Zhang EF, Ren XF, et al. Edaravone alleviates cell apoptosis and mitochondrial injury in ischemia-reperfusion-induced kidney injury via the JAK/STAT pathway[J]. Biol Res, 2020, 53(1): 28. doi:10.1186/s40659-020-00297-0 |
| [24] | Liu Y, Wang L, Du Y, et al. Effects of apigenin pretreatment against renal ischemia/reperfusion injury via activation of the JAK2/STAT3 pathway[J]. Biomed Pharmacother, 2017, 95: 1799-808. doi:10.1016/j.biopha.2017.09.091 |
| [25] | Xu MJ, Feng DC, Wang H, et al. IL-22 ameliorates renal ischemia-reperfusion injury by targeting proximal tubule epithelium[J]. J Am Soc Nephrol, 2014, 25(5): 967-77. doi:10.1681/asn.2013060611 |
| [26] | Bilanges B, Posor Y, Vanhaesebroeck B. PI3K isoforms in cell signalling and vesicle trafficking[J]. Nat Rev Mol Cell Biol, 2019, 20(9): 515-34. doi:10.1038/s41580-019-0129-z |
| [27] | Liu C, Chen K, Wang HX, et al. Gastrin attenuates renal ischemia/reperfusion injury by a PI3K/Akt/bad-mediated anti-apoptosis signaling[J]. Front Pharmacol, 2020, 11: 540479. doi:10.3389/fphar.2020.540479 |
| [28] | Tang Z, Wang Y, Liu Y, et al. Salidroside inhibits renal ischemia/reperfusion injury-induced ferroptosis by the PI3K/AKT signaling pathway[J]. Exp Ther Med, 2023, 26(5): 507. doi:10.3892/etm.2023.12206 |
| [29] | Pace J, Paladugu P, Das B, et al. Targeting STAT3 signaling in kidney disease[J]. Am J Physiol Renal Physiol, 2019, 316(6): F1151-61. doi:10.1152/ajprenal.00034.2019 |
| [30] | Huang F, Wang X, Xiao GF, et al. Loganin exerts a protective effect on ischemia-reperfusion-induced acute kidney injury by regulating JAK2/STAT3 and Nrf2/HO-1 signaling pathways[J]. Drug Dev Res, 2022, 83(1): 150-7. doi:10.1002/ddr.21853 |
| [31] | Park JY, Yoo KD, Bae E, et al. Blockade of STAT3 signaling alleviates the progression of acute kidney injury to chronic kidney disease through antiapoptosis[J]. Am J Physiol Renal Physiol, 2022, 322(5): F553-72. doi:10.1152/ajprenal.00595.2020 |
| [32] | Ogura M, Uchida T, Terui Y, et al. Phase I study of OPB-51602, an oral inhibitor of signal transducer and activator of transcription 3, in patients with relapsed/refractory hematological malignancies[J]. Cancer Sci, 2015, 106(7): 896-901. doi:10.1111/cas.12683 |
| [33] | Okusaka T, Ueno H, Ikeda M, et al. Phase 1 and pharmacological trial of OPB-31121, a signal transducer and activator of transcription-3 inhibitor, in patients with advanced hepatocellular carcinoma[J]. Hepatol Res, 2015, 45(13): 1283-91. doi:10.1111/hepr.12504 |
| [1] | 杨剑明, 杨龙, 洪铠文, 耿贝贝, 赵满, 王耀光, 夏婷, 董津睿. 腹腔注射薤白碳量子点可改善顺铂诱导的小鼠急性肾损伤并修复线粒体功能[J]. 南方医科大学学报, 2026, 46(3): 505-512. |
| [2] | 闫爱丽, 罗梦瑶, 常晋瑞, 李新华, 朱娟霞. 橙皮素通过调控AMPK/NLRP3通路减轻阿霉素诱导的小鼠心肌毒性[J]. 南方医科大学学报, 2025, 45(9): 1850-1858. |
| [3] | 欧泽金, 李瀛, 陈诗, 王梓译, 何美仪, 陈志成, 唐侍豪, 孟晓静, 王致. 抑制铁死亡减轻敌草快引起的斑马鱼急性肾损伤的机制[J]. 南方医科大学学报, 2025, 45(8): 1743-1750. |
| [4] | 范正媛, 沈子涵, 李亚, 沈婷婷, 李高峰, 李素云. 补肺益肾方对香烟烟雾提取物诱导的人支气管上皮细胞损伤的保护作用及其机制[J]. 南方医科大学学报, 2025, 45(7): 1372-1379. |
| [5] | 夏冰, 彭进, 丁九阳, 王杰, 唐国伟, 刘国杰, 王沄, 万昌武, 乐翠云. ATF3通过NF-κB信号通路调控动脉粥样硬化斑块内的炎症反应[J]. 南方医科大学学报, 2025, 45(6): 1131-1142. |
| [6] | 牛民主, 殷丽霞, 乔通, 尹林, 张可妮, 胡建国, 宋传旺, 耿志军, 李静. 旱莲苷A通过调控JAK2/STAT3通路抑制M1型巨噬细胞极化改善葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2025, 45(6): 1297-1306. |
| [7] | 王子皓, 陶丽丽, 邹碧清, 安胜利. 重症监护病房急性肾损伤患者首次24 h动脉氧分压与死亡率相关:基于MIMIC-IV数据库[J]. 南方医科大学学报, 2025, 45(5): 1056-1062. |
| [8] | 张柳攀, 石晓彤, 李露兰, 师瑞, 安胜利, 曾振华. 合并急性肾损伤的危重症患者输注白蛋白后血清白蛋白水平与28 d死亡率的相关性[J]. 南方医科大学学报, 2025, 45(5): 1074-1081. |
| [9] | 张安邦, 孙秀颀, 庞博, 吴远华, 时靖宇, 张宁, 叶涛. 电针预处理通过调节肠道-大脑轴及Nrf2/HO-1信号通路抑制铁死亡减轻大鼠脑缺血再灌注损伤[J]. 南方医科大学学报, 2025, 45(5): 911-920. |
| [10] | 浦延鹏, 王震, 储浩然. 眼针疗法通过上调METTL3介导的m6A甲基化修饰促进脑皮层血管新生进而改善脑缺血再灌注损伤大鼠的神经功能[J]. 南方医科大学学报, 2025, 45(5): 921-928. |
| [11] | 张璐, 丁焕章, 许浩燃, 陈珂, 许博文, 杨勤军, 吴迪, 童佳兵, 李泽庚. 参芪补中方通过激活AMPK/SIRT1/PGC-1α改善COPD肺脾气虚证大鼠线粒体功能障碍[J]. 南方医科大学学报, 2025, 45(5): 969-976. |
| [12] | 卞芬兰, 倪诗垚, 赵鹏, 戚毛男星, 唐碧, 王洪巨, 康品方, 刘进军. 积雪草苷通过抑制NLRP3炎症体介导的细胞焦亡减轻大鼠心肌缺血再灌注损伤[J]. 南方医科大学学报, 2025, 45(5): 977-985. |
| [13] | 陈镝, 吕莹, 郭怡欣, 张怡荣, 王蕊璇, 周小若, 陈雨欣, 武晓慧. 双氢青蒿素可显著增强阿霉素诱导的三阴性乳腺癌细胞凋亡:基于负向调控STAT3/HIF-1α通路[J]. 南方医科大学学报, 2025, 45(2): 254-260. |
| [14] | 宾禹, 李子雯, 左素微, 孙思诺, 李敏, 宋佳茵, 林旭, 薛刚, 吴靖芳. 载脂蛋白C1高表达通过激活JAK2/STAT3信号通路促进甲状腺乳头状癌细胞的增殖并抑制凋亡[J]. 南方医科大学学报, 2025, 45(2): 359-370. |
| [15] | 原梦瑶, 阮湘涵, 李扬, 张婷, 郝春香, 李皓, 娄景盛, 曹江北, 刘艳红, 米卫东, 张晓莹. 术前血清镁水平与非心脏手术老年患者术后谵妄风险的关系:一项回顾性队列研究[J]. 南方医科大学学报, 2025, 45(12): 2616-2627. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||