南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 335-344.doi: 10.12122/j.issn.1673-4254.2026.02.11
• • 上一篇
李斌杰1,2( ), 周晓芳2, 朗晓猛3, 康欣3, 刘建平3(
), 周晓芳2, 朗晓猛3, 康欣3, 刘建平3( )
)
收稿日期:2025-04-30
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
刘建平
E-mail:344014450@qq.com;13603396653@163.com
作者简介:李斌杰,博士,E-mail: 344014450@qq.com
基金资助:
Binjie LI1,2( ), Xiaofang ZHOU2, Xiaomeng LANG3, Xin KANG3, Jianping LIU3(
), Xiaofang ZHOU2, Xiaomeng LANG3, Xin KANG3, Jianping LIU3( )
)
Received:2025-04-30
Online:2026-02-20
Published:2026-03-10
Contact:
Jianping LIU
E-mail:344014450@qq.com;13603396653@163.com
摘要:
目的 探讨泄浊解毒方调节Th17/Treg免疫平衡改善溃疡性结肠炎(UC)的作用机制。 方法 将SD大鼠建立UC模型后随机分为模型组、美沙拉秦组(Mesalamine组,0.36 g/kg)、泄浊解毒方高剂量组(XZJD-H组,26.64 g/kg)、泄浊解毒方中剂量组(XZJD-M组,13.32 g/kg)和泄浊解毒方低剂量组(XZJD-L组,6.66 g/kg),10只/组,另取10只大鼠设为空白组。空白组和模型组采用生理盐水灌胃,Mesalamine组和泄浊解毒方各组按照相应的药物和剂量分别灌胃14 d。HE染色观察结肠组织病理改变;ELISA检测血清白细胞介素-6(IL-6)、IL-10、IL-17表达水平;免疫组织化学法(IHC)检测结肠组织IL-6、IL-10、IL-17的表达;免疫荧光法检测结肠组织闭合蛋白、闭锁小带蛋白1(ZO-1)的表达;流式细胞术检测结肠组织中Th17和Treg细胞的比例;RT-PCR检测维甲酸相关孤儿受体(ROR-γt)、叉状头转录因子p3(Foxp3)mRNA表达水平;Western blotting检测结肠组织RORγt、Foxp3蛋白的表达。 结果 与空白组比较,模型组出现结肠黏膜损伤、大量炎性细胞浸润等表现,血清IL-6、IL-17含量和结肠组织IL-6、IL-17表达升高(P<0.001),血清IL-10含量和结肠组织IL-10表达降低(P<0.001),Occludin、ZO-1表达水平降低,Th17细胞比例升高(P<0.001),Treg细胞比例降低(P<0.001),RORγt mRNA和蛋白表达增高(P<0.01),Foxp3 mRNA和蛋白降低(P<0.01)。与模型组比较,泄浊解毒方组可改善UC大鼠结肠黏膜的炎症反应,降低血清IL-6、IL-17含量,升高IL-10含量(P<0.01),降低结肠组织IL-6、IL-17蛋白的表达(P<0.01),升高IL-10蛋白的表达(P<0.01),升高Occludin、ZO-1的表达,降低结肠组织Th17细胞百分比(P<0.001),升高Treg细胞百分比(P<0.001),降低RORγt mRNA和蛋白表达(P<0.05),升高Foxp3 mRNA和蛋白表达(P<0.05)。 结论 泄浊解毒方可改善UC大鼠结肠黏膜的病理损伤,其作用机制可能与调节Th17/Treg免疫平衡有关。
李斌杰, 周晓芳, 朗晓猛, 康欣, 刘建平. 泄浊解毒方通过调节Th17/Treg免疫平衡改善大鼠溃疡性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 335-344.
Binjie LI, Xiaofang ZHOU, Xiaomeng LANG, Xin KANG, Jianping LIU. Xiezhuo Jiedu Recipe improves ulcerative colitis in rats by regulating Th17/Treg immune balance[J]. Journal of Southern Medical University, 2026, 46(2): 335-344.
| Gene | Forward (5'→3') | Reverse (3'→5') |
|---|---|---|
| RORγt | ACCAACCTCTTCTCACGGG | CTTCCATTGCTCCTGCTTTC |
| Foxp3 | GCTTGTTTGCTGTGCGGAGA | TGGCATAGGTGAAAGGGGGC |
| GAPDH | ACAGCAACAGGGTGGTGGAC | TTTGAGGGTGCAGCGAACTT |
表1 引物序列
Tab.1 Primer sequences for RT-qPCR
| Gene | Forward (5'→3') | Reverse (3'→5') |
|---|---|---|
| RORγt | ACCAACCTCTTCTCACGGG | CTTCCATTGCTCCTGCTTTC |
| Foxp3 | GCTTGTTTGCTGTGCGGAGA | TGGCATAGGTGAAAGGGGGC |
| GAPDH | ACAGCAACAGGGTGGTGGAC | TTTGAGGGTGCAGCGAACTT |
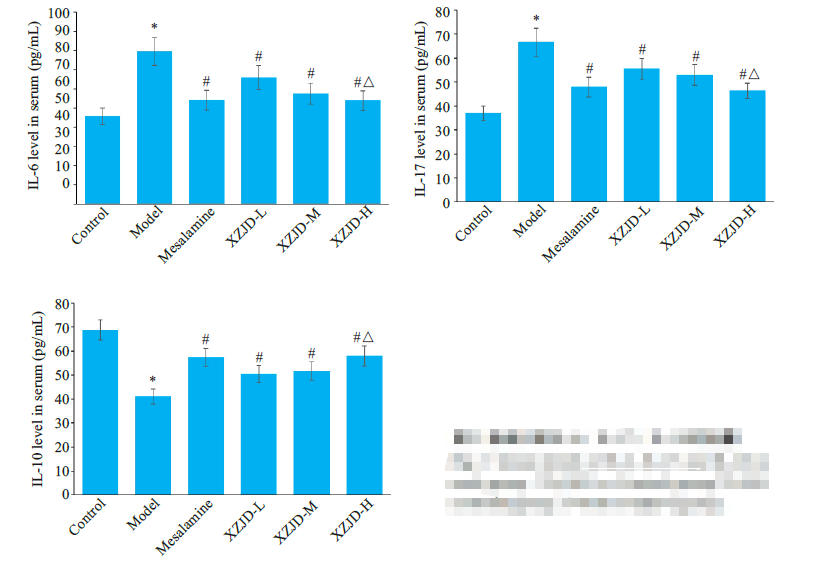
图2 各组大鼠IL-6、IL-17、IL-10的含量
Fig.2 Serum levels of IL-6, IL-17 and IL-10 ineach group. *P<0.001 vs Control group; #P<0.01vs Model group,△P<0.05 vs XZJD-L group.
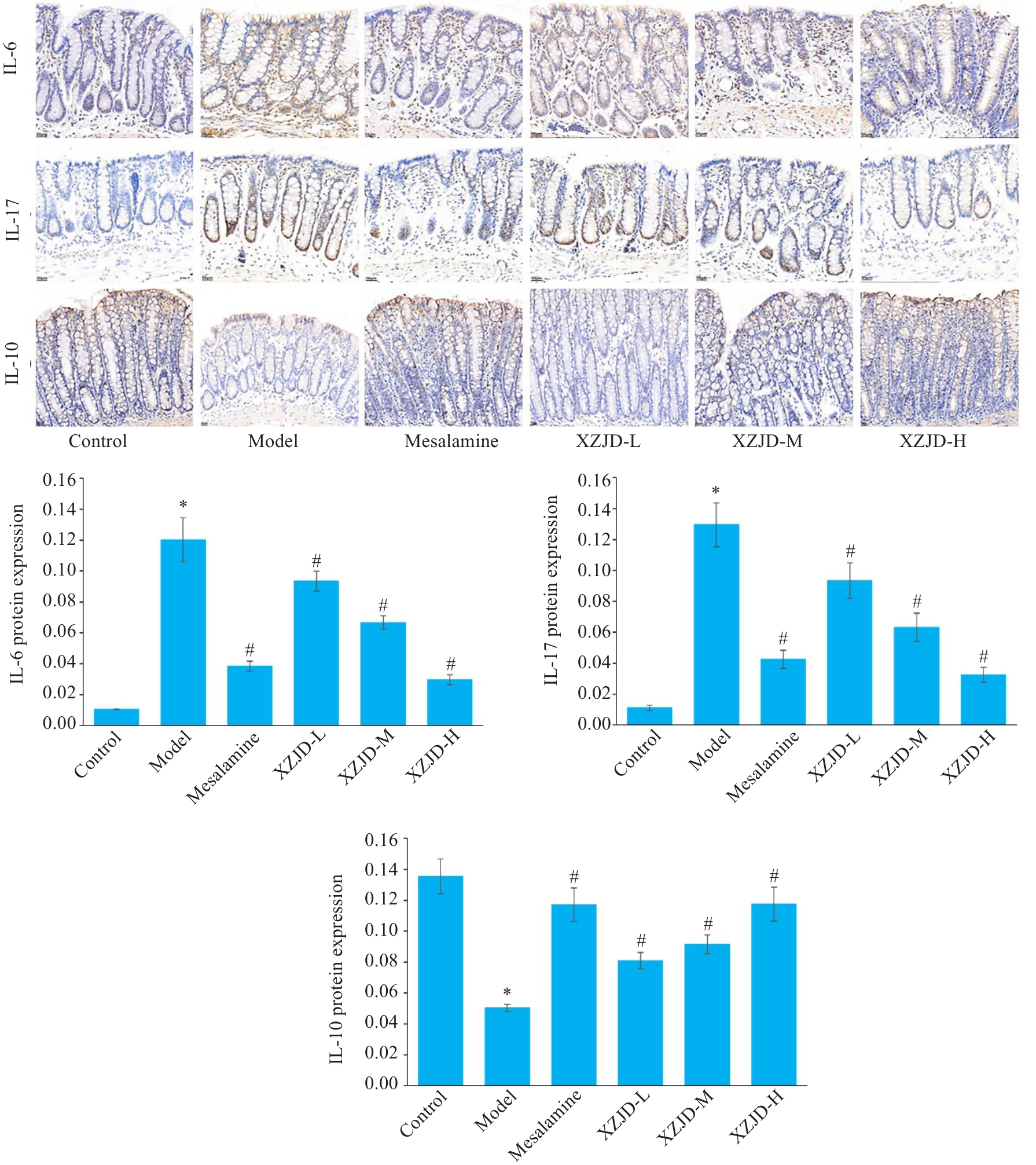
图3 各组大鼠结肠组织IL-6、IL-17、IL-10蛋白的表达
Fig.3 Immunohistochemical detection of expressions of IL-6, IL-17 and IL-10 proteins in the colon tissue of the rats in each group (Original magnification: ×400). *P<0.001 vs Control group; #P<0.01 vs Model group.
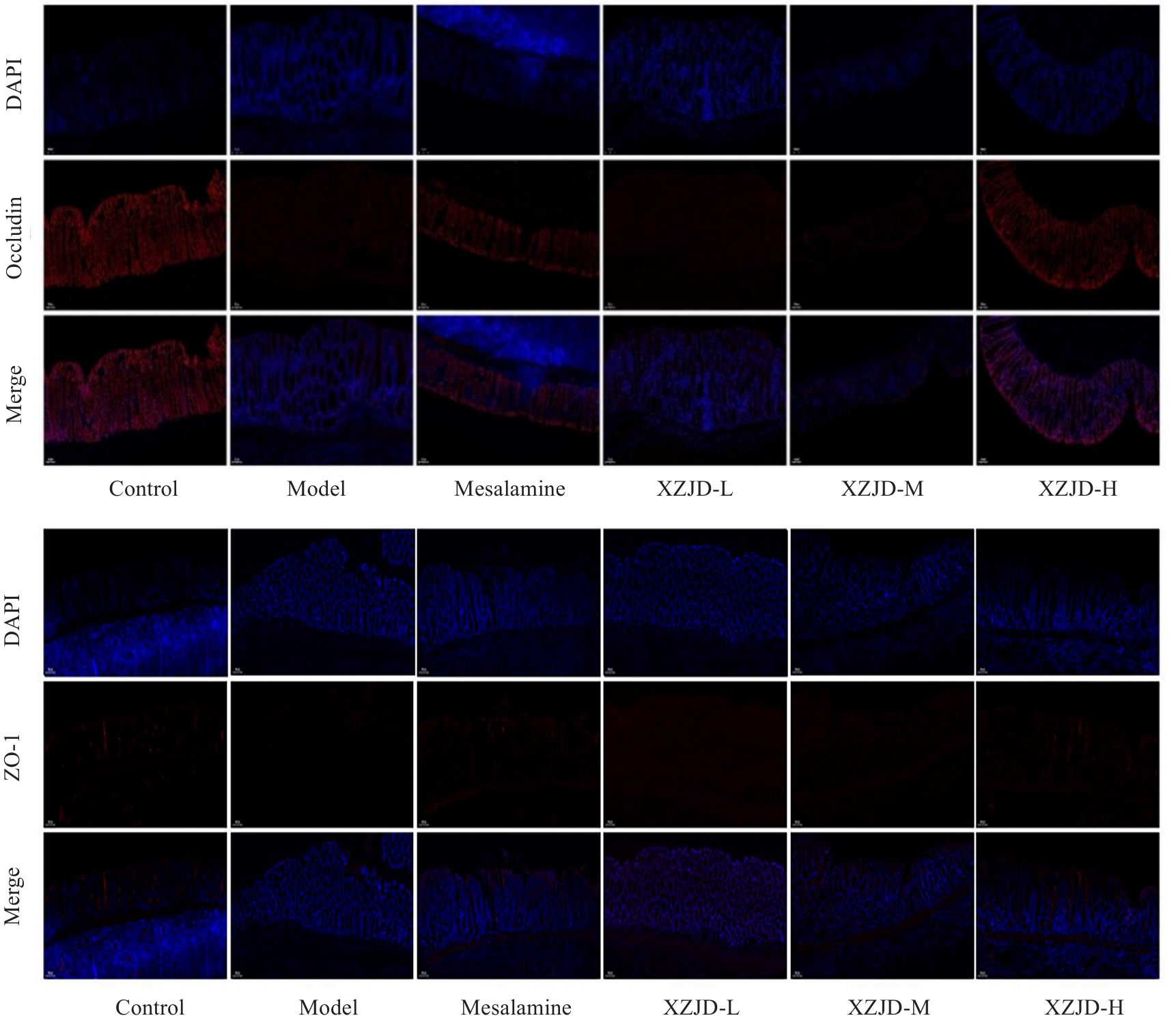
图4 各组大鼠结肠组织Occludin、ZO-1蛋白的比较
Fig.4 Expressions of occludin and ZO-1 proteins in the colon tissue of the rats in each group (DAPI nuclear staining, ×200).
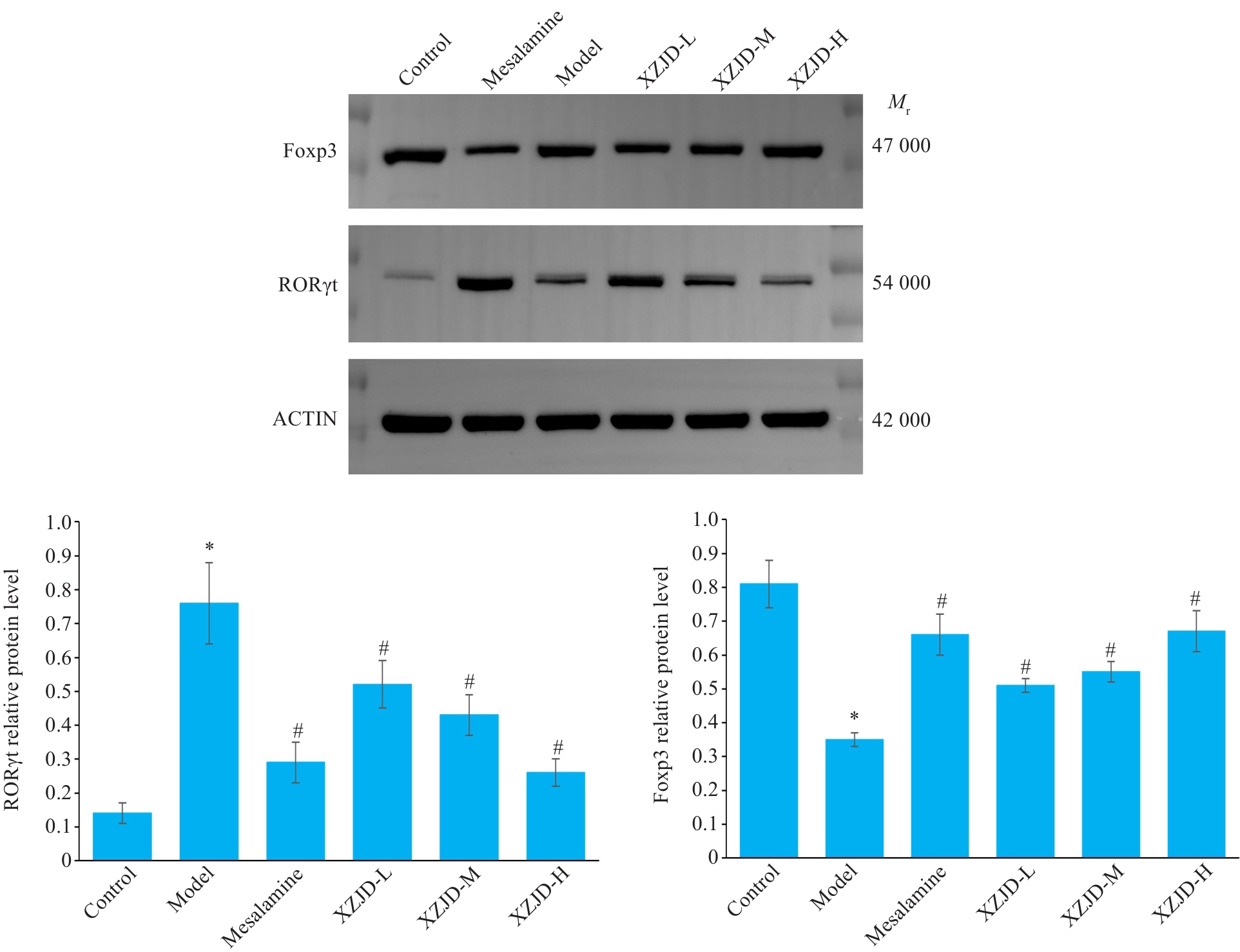
图7 大鼠结肠RORγt、Foxp3蛋白表达的比较
Fig.7 Expression levels of RORγt and Foxp3 proteins in rat colon tissues detected by Western blotting. *P<0.001 vs Control group; #P<0.01 vs Model group.
| [1] | Su L, Su Y, An Z, et al. Fermentation products of Danshen relieved dextran sulfate sodium-induced experimental ulcerative colitis in mice[J]. Sci Rep, 2021, 11(1): 16210. doi:10.1038/s41598-021-94594-7 |
| [2] | Ungaro R, Mehandru S, Allen PB, et al. Ulcerative colitis[J]. Lancet, 2017, 389(10080): 1756-70. doi:10.1016/s0140-6736(16)32126-2 |
| [3] | Nanki K, Fujii M, Shimokawa M, et al. Somatic inflammatory gene mutations in human ulcerative colitis epithelium[J]. Nature, 2020, 577(7789): 254-9. doi:10.1038/s41586-019-1844-5 |
| [4] | Abelson JS, Michelassi F, Mao J, et al. Higher surgical morbidity for ulcerative colitis patients in the era of biologics[J]. Ann Surg, 2018, 268(2): 311-7. doi:10.1097/sla.0000000000002275 |
| [5] | Jones GR, Lyons M, Plevris N, et al. IBD prevalence in Lothian, Scotland, derived by capture-recapture methodology[J]. Gut, 2019, 68(11): 1953-60. doi:10.1136/gutjnl-2019-318936 |
| [6] | Rubin DT, Ananthakrishnan AN, Siegel CA, et al. ACG clinical guideline: ulcerative colitis in adults[J]. Am J Gastroenterol, 2019, 114(3): 384-413. doi:10.14309/ajg.0000000000000152 |
| [7] | Chen R, Lai LA, Brentnall TA, et al. Biomarkers for colitis-associated colorectal cancer[J]. World J Gastroenterol, 2016, 22(35): 7882-91. doi:10.3748/wjg.v22.i35.7882 |
| [8] | Zhu YF, Yang SH, Zhao N, et al. CXCL8 chemokine in ulcerative colitis[J]. Biomed Pharmacother, 2021, 138: 111427. doi:10.1016/j.biopha.2021.111427 |
| [9] | Dubin PJ, Kolls JK. Th17 cytokines and mucosal immunity[J]. Immunol Rev, 2008, 226: 160-71. doi:10.1111/j.1600-065x.2008.00703.x |
| [10] | 曹 霞, 邱 榕, 陶云平, 等. 基于Th17/Treg平衡的调节探讨参苓白术散治疗溃疡性结肠炎的疗效及作用机制[J].中药材, 2025, (1):237-41. |
| [11] | Duan S, Cao Y, Chen P, et al. Circulating and intestinal regulatory T cells in inflammatory bowel disease: a systemic review and meta-analysis[J]. Int Rev Immunol, 2024, 43(2): 83-94. doi:10.1080/08830185.2023.2249525 |
| [12] | Josefowicz SZ, Lu LF, Rudensky AY. Regulatory T cells: mechanisms of differentiation and function[J]. Annu Rev Immunol, 2012, 30: 531-64. doi:10.1146/annurev.immunol.25.022106.141623 |
| [13] | Ueno A, Jeffery L, Kobayashi T, et al. Th17 plasticity and its relevance to inflammatory bowel disease[J]. J Autoimmun, 2018, 87: 38-49. doi:10.1016/j.jaut.2017.12.004 |
| [14] | 覃小燕, 韩晓玲, 张 媛, 等.基于网络药理学的女贞子抗骨质疏松作用机制研究[J].中国药学杂志, 2021, 56(5): 368-78. |
| [15] | 任 娟, 张 娜, 王 敏, 等. 基于网络药理学的砂仁镇痛作用机制研究[J].中国药学杂志, 2021, 56(9): 723-30. |
| [16] | Xiang SY, Zhao J, Lu Y, et al. Network pharmacology-based identification for therapeutic mechanism of Ling-Gui-Zhu-Gan decoction in the metabolic syndrome induced by antipsychotic drugs[J]. Comput Biol Med, 2019, 110: 1-7. doi:10.1016/j.compbiomed.2019.05.007 |
| [17] | Zhu BC, Zhang WT, Lu Y, et al. Network pharmacology-based identification of protective mechanism of Panax Notoginseng Saponins on aspirin induced gastrointestinal injury[J]. Biomed Pharmacother, 2018, 105: 159-66. doi:10.1016/j.biopha.2018.04.054 |
| [18] | Wei Y, Ren S, Wang R, et al. Based on network pharmacology to explore the potential bioactive compounds and mechanisms of Zuojin pill for the treatment of ulcerative colitis[J]. Evid Based Complement Alternat Med, 2021, 2021: 7567025. doi:10.1155/2021/7567025 |
| [19] | 李斌杰, 康 帅, 刘晓萌, 等. 基于网络药理学和实验验证探讨泄浊解毒方治疗溃疡性结肠炎的作用机制[J]. 中国药学杂志, 2023, 58(1):48-56. |
| [20] | 刘建平, 吴鹤伶, 郎晓猛, 等. 泄浊解毒方对溃疡性结肠炎大鼠血清IL-1β、IL-10及结肠黏膜CD14的影响[J]. 河北中医药学报, 2017,32(2): 1-4. |
| [21] | 康 欣, 刘建平, 任 杰, 等. 泄浊解毒方改善大鼠溃疡性结肠炎和调控巨噬细胞极化机制研究[J].免疫学杂志, 2024, 40(1): 65-71. |
| [22] | Morris GP, Beck PL, Herridge MS, et al. Hapten-induced model of chronic inflammation and ulceration in the rat colon[J]. Gastroenterology, 1989, 96(3): 795-803. doi:10.1016/s0016-5085(89)80079-4 |
| [23] | Sun M, He C, Wu W, et al. Hypoxia inducible factor-1α-induced interleukin-33 expression in intestinal epithelia contributes to mucosal homeostasis in inflammatory bowel disease[J]. Clin Exp Immunol, 2017, 187(3): 428-40. doi:10.1111/cei.12896 |
| [24] | 徐叔云. 药理实验方法学[M]. 3版. 北京: 人民卫生出版社, 2002. |
| [25] | Danese S, Fiocchi C. Ulcerative colitis[J]. N Engl J Med, 2011, 365(18): 1713-25. doi:10.1056/nejmra1102942 |
| [26] | Kakuta Y, Ichikawa R, Fuyuno Y, et al. An integrated genomic and transcriptomic analysis reveals candidates of susceptibility genes for Crohn's disease in Japanese populations[J]. Sci Rep, 2020, 10(1): 10236. doi:10.1038/s41598-020-66951-5 |
| [27] | 娄莹莹, 李佃贵, 霍永利, 等. 溃疡性结肠炎特色病机“浊毒损膜伤络”及其意义[J]. 中国中西医结合杂志, 2022, 42(6): 749-53. |
| [28] | 郎晓猛, 刘建平, 李佃贵, 等. 泄浊解毒方对溃疡性结肠炎大鼠结肠组织TLR2基因、蛋白表达的影响[J]. 中华中医药杂志, 2017, 32(2):838-40. |
| [29] | 李雪可. 泄浊解毒方联合美沙拉秦治疗UC患者临床疗效观察及对肠道菌群的影响[D]. 河北中医学院, 2021. |
| [30] | 康 欣, 刘建平, 李佃贵, 等.泄浊解毒方联合美沙拉嗪治疗溃疡性结肠炎90例临床疗效观察[J].世界中西医结合杂志, 2019, 14(1): 90-3. |
| [31] | Herndon JS, Vitta S, Weber FH. Marked eosinophilia in a 27-year-old woman with recent onset ulcerative colitis[J]. Gastroenterology, 2021, 160(1): 29-30. doi:10.1053/j.gastro.2020.08.030 |
| [32] | Fu X, Sun F, Sun F, et al. Aloperine protects mice against DSS-induced colitis by PP2A-mediated PI3K/Akt/mTOR signaling suppression[J]. Mediators Inflamm, 2017, 2017: 5706152. doi:10.1155/2017/5706152 |
| [33] | Afif W, Sandborn WJ, Faubion WA, et al. Risk factors for lymphoma in patients with inflammatory bowel disease: a case-control study[J]. Inflamm Bowel Dis, 2013, 19(7): 1384-9. doi:10.1097/mib.0b013e318281325e |
| [34] | Shahini A, Shahini A. Role of interleukin-6-mediated inflammation in the pathogenesis of inflammatory bowel disease: focus on the available therapeutic approaches and gut microbiome[J]. J Cell Commun Signal, 2023, 17(1): 55-74. doi:10.1007/s12079-022-00695-x |
| [35] | Machiels K, Joossens M, Sabino J, et al. A decrease of the butyrate-producing species Roseburia hominisand Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis[J]. Gut, 2014, 63(8): 1275-83. doi:10.1136/gutjnl-2013-304833 |
| [36] | Pankratz S, Ruck T, Meuth SG, et al. CD4+HLA-G(+) regulatory T cells: Molecular signature and pathophysiological relevance[J]. Hum Immunol, 2016, 77(9): 727-33. doi:10.1016/j.humimm.2016.01.016 |
| [37] | Jofra T, Galvani G, Cosorich I,et al.Experimental colitis in IL-10-deficient mice ameliorates in the absence of PTPN22[J]. Clin Exp Immunol, 2019, 197(3):263-75. doi:10.1111/cei.13339 |
| [38] | 于 斌,邓 力,张 丽,等.湿邪致病现代机理研究进展[J].广州中医药大学学报,2015,32(1):174-7. |
| [39] | 田同德,杨 峰,唐静雯,等.慢性炎性反应与胃癌前病变关系及中医对策探讨[J].中华中医药杂志,2016,31(2):359-63. |
| [40] | Wu H, Chen QY, Wang WZ, et al. Compound sophorae decoction enhances intestinal barrier function of dextran sodium sulfate induced colitis via regulating Notch signaling pathway in mice[J]. Biomed Pharmacother, 2021, 133: 110937. doi:10.1016/j.biopha.2020.110937 |
| [41] | Yin SJ, Yang HF, Tao Y, et al. Artesunate ameliorates DSS-induced ulcerative colitis by protecting intestinal barrier and inhibiting inflammatory response[J]. Inflammation, 2020, 43(2): 765-76. doi:10.1007/s10753-019-01164-1 |
| [42] | Chu XQ, Wang J, Chen GX, et al. Overexpression of microRNA-495 improves the intestinal mucosal barrier function by targeting STAT3 via inhibition of the JAK/STAT3 signaling pathway in a mouse model of ulcerative colitis[J]. Pathol Res Pract, 2018, 214(1): 151-62. doi:10.1016/j.prp.2017.10.003 |
| [43] | Yang F, Wang A, Zeng X, et al. Lactobacillus reuteri I5007 modulates tight junction protein expression in IPEC-J2 cells with LPS stimulation and in newborn piglets under normal conditions[J]. BMC Microbiol, 2015, 15: 32. doi:10.1186/s12866-015-0372-1 |
| [44] | Tatiya-Aphiradee N, Chatuphonprasert W, Jarukamjorn K. Immune response and inflammatory pathway of ulcerative colitis[J]. J Basic Clin Physiol Pharmacol, 2018, 30(1): 1-10. doi:10.1515/jbcpp-2018-0036 |
| [45] | Iacomino G, Rotondi Aufiero V, Iannaccone N, et al. IBD: Role of intestinal compartments in the mucosal immune response[J]. Immunobiology, 2020, 225(1): 151849. doi:10.1016/j.imbio.2019.09.008 |
| [46] | Gomez-Bris R, Saez A, Herrero-Fernandez B, et al. CD4 T-cell subsets and the pathophysiology of inflammatory bowel disease[J]. Int J Mol Sci, 2023, 24(3): 2696. doi:10.3390/ijms24032696 |
| [47] | Zhao Q, Duck LW, Huang F, et al. CD4+ T cell activation and concomitant mTOR metabolic inhibition can ablate microbiota-specific memory cells and prevent colitis[J]. Sci Immunol, 2020, 5(54): eabc6373. doi:10.1126/sciimmunol.abc6373 |
| [48] | Xiong XY, Cheng Z, Wu F, et al. Berberine in the treatment of ulcerative colitis: a possible pathway through Tuft cells[J]. Biomed Pharmacother, 2021, 134: 111129. doi:10.1016/j.biopha.2020.111129 |
| [49] | Gupta PK, Wagner SR, Wu Q, et al. Th17 cells are not required for maintenance of IL-17A-producing γδ T cells in vivo [J]. Immunol Cell Biol, 2017, 95(3): 280-6. doi:10.1038/icb.2016.94 |
| [50] | Thapa B, Pak S, Kwon HJ, et al. Decursinol angelate ameliorates dextran sodium sulfate-induced colitis by modulating type 17 helper T cell responses[J]. Biomol Ther: Seoul, 2019, 27(5): 466-73. doi:10.4062/biomolther.2019.004 |
| [51] | Ivanov II, McKenzie BS, Zhou L, et al. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells[J]. Cell, 2006, 126(6): 1121-33. doi:10.1016/j.cell.2006.07.035 |
| [52] | Chen X, Du Y, Lin XQ, et al. CD4+CD25+ regulatory T cells in tumor immunity[J]. Int Immunopharmacol, 2016, 34: 244-9. doi:10.1016/j.intimp.2016.03.009 |
| [53] | Yan JB, Luo MM, Chen ZY, et al. The function and role of the Th17/treg cell balance in inflammatory bowel disease[J]. J Immunol Res, 2020: 8813558. doi:10.1155/2020/8813558 |
| [54] | Barbi J, Pardoll DM, Pan F. Ubiquitin-dependent regulation of Foxp3 and Treg function[J]. Immunol Rev, 2015, 266(1): 27-45. doi:10.1111/imr.12312 |
| [55] | Nie J, Li YY, Zheng SG, et al. FOXP3+ Treg cells and gender bias in autoimmune diseases[J]. Front Immunol, 2015, 6: 493. doi:10.3389/fimmu.2015.00493 |
| [56] | Sheng Y, Wu T, Dai Y, et al. The effect of 6-gingerol on inflammatory response and Th17/Treg balance in DSS-induced ulcerative colitis mice[J]. Ann Transl Med, 2020, 8(7): 442. doi:10.21037/atm.2020.03.141 |
| [1] | 乔通, 尹林, 张可妮, 牛民主, 黄菊, 耿志军, 李静, 胡建国. 茯苓新酸A通过调节AMPK/mTOR介导的自噬来减轻葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2026, 46(1): 131-140. |
| [2] | 何榕茂, 方泽扬, 张芸芸, 吴友谅, 梁世秀, 计涛, 陈科全, 王斯琪. 铁死亡相关基因对溃疡性结肠炎具有诊断预测价值[J]. 南方医科大学学报, 2025, 45(9): 1927-1937. |
| [3] | 刘辰菲, 张玮, 曾尧, 梁艳, 王梦婷, 张明芳, 李新元, 王凤超, 杨燕青. 2,6-二甲氧基-1,4-苯醌通过抑制NLRP3炎症小体活化改善葡聚糖硫酸钠诱导的小鼠溃疡性结肠炎[J]. 南方医科大学学报, 2025, 45(8): 1654-1662. |
| [4] | 牛民主, 殷丽霞, 乔通, 尹林, 张可妮, 胡建国, 宋传旺, 耿志军, 李静. 旱莲苷A通过调控JAK2/STAT3通路抑制M1型巨噬细胞极化改善葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2025, 45(6): 1297-1306. |
| [5] | 申琳, 宋翠豪, 王聪敏, 高西, 安俊红, 李承新, 梁斌, 李霞. 溃疡性结肠炎并发坏疽性脓皮病患者发生营养风险的因素及预测模型[J]. 南方医科大学学报, 2025, 45(3): 514-521. |
| [6] | 殷丽霞, 牛民主, 张可妮, 耿志军, 胡建国, 李江艳, 李静. 升麻素抑制MAPK通路调节辅助性T细胞免疫平衡改善小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2025, 45(3): 595-602. |
| [7] | 黄晴晴, 杨晶晶, 姜雪凝, 张文静, 汪煜, 左芦根, 王炼, 王月月, 张小凤, 宋雪, 胡建国. 刺桐碱通过抑制肠上皮炎症反应并改善肠屏障功能缓解小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2025, 45(11): 2456-2465. |
| [8] | 赵娜, 沈梦迪, 赵睿, 奥迪, 骆泽谭, 张银亮, 徐志东, 范方田, 郑海伦. 血根碱通过调控Nrf2/NF-κB通路缓解小鼠溃疡性结肠炎[J]. 南方医科大学学报, 2024, 44(8): 1467-1475. |
| [9] | 于官正, 程炜强, 涂星, 张满, 李鸿, 聂娟. 隔山消治疗溃疡性结肠炎的机制:基于UPLC-QE-MS、网络药理学及代谢组学技术[J]. 南方医科大学学报, 2024, 44(8): 1485-1496. |
| [10] | 刘硕, 李静, 吴兴旺. Swertiamarin通过抑制肠上皮细胞细胞凋亡改善TNBS诱导的实验性结肠炎[J]. 南方医科大学学报, 2024, 44(8): 1545-1552. |
| [11] | 牛民主, 殷丽霞, 段婷, 黄菊, 李静, 耿志军, 胡建国, 宋传旺. 川续断皂苷VI通过抑制PI3K/AKT/NF-κB通路拮抗肠上皮细胞凋亡缓解TNBS诱导的小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2024, 44(12): 2335-2346. |
| [12] | 邱建国, 邱一桐, 李国荣, 张林生, 郑雪, 姚泳江, 王熙丹, 黄海阳, 张凤敏, 苏冀彦, 郑学宝, 黄晓其. 黄芩汤通过调控内质网应激减轻小鼠溃疡性结肠炎[J]. 南方医科大学学报, 2024, 44(11): 2172-2183. |
| [13] | 宋泽军, 董海滨, 马 娜, 任渝棠, 姜 泊. 改进的MAYO内镜评分对活动期溃疡性结肠炎疗效有较高的评估价值[J]. 南方医科大学学报, 2023, 43(7): 1204-1213. |
| [14] | 邵荣瑢, 杨 子, 张文静, 张 诺, 赵雅静, 张小凤, 左芦根, 葛思堂. 茯苓酸缓解小鼠克罗恩病:基于抑制PI3K/AKT信号通路拮抗肠上皮细胞凋亡[J]. 南方医科大学学报, 2023, 43(6): 935-942. |
| [15] | 宋泽军, 张明君, 任渝棠, 姜 泊. 改良Mayo内镜评分对溃疡性结肠炎有较高的评估价值[J]. 南方医科大学学报, 2022, 42(7): 997-1005. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||