南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (7): 1409-1422.doi: 10.12122/j.issn.1673-4254.2025.07.07
王立明1,2( ), 陈宏睿1,2, 杜燕1,2, 赵鹏1,2,3, 王玉洁1,2, 田燕歌1,2,3, 刘新光1,2,3(
), 陈宏睿1,2, 杜燕1,2, 赵鹏1,2,3, 王玉洁1,2, 田燕歌1,2,3, 刘新光1,2,3( ), 李建生1,2,3
), 李建生1,2,3
收稿日期:2025-03-24
出版日期:2025-07-20
发布日期:2025-07-17
通讯作者:
刘新光
E-mail:15956982550@163.com;xgliu2016@126.com
作者简介:王立明,硕士,E-mail: 15956982550@163.com
基金资助:
Liming WANG1,2( ), Hongrui CHEN1,2, Yan DU1,2, Peng ZHAO1,2,3, Yujie WANG1,2, Yange TIAN1,2,3, Xinguang LIU1,2,3(
), Hongrui CHEN1,2, Yan DU1,2, Peng ZHAO1,2,3, Yujie WANG1,2, Yange TIAN1,2,3, Xinguang LIU1,2,3( ), Jiansheng LI1,2,3
), Jiansheng LI1,2,3
Received:2025-03-24
Online:2025-07-20
Published:2025-07-17
Contact:
Xinguang LIU
E-mail:15956982550@163.com;xgliu2016@126.com
Supported by:摘要:
目的 明确益气滋肾方(YZF)的化学成分和入血成分,并阐明YZF改善慢性阻塞性肺疾病(COPD)炎症的整合作用机制与药效物质。 方法 通过超高效液相色谱串联四极杆轨道离子阱质谱(UHPLC-Q-Extractive-Orbitrap MS)鉴定YZF化学成分和入血成分。基于YZF入血成分构建YZF改善COPD的成分-疾病靶点网络,并进行富集分析筛选关键通路与作用靶标。通过香烟烟雾诱导构建小鼠COPD模型评价YZF抗炎作用及验证网络药理学富集到相关通路中关键蛋白的表达情况。50只C57BL/6J雄鼠随机分为对照组、模型组、YZF高、低剂量组和N-乙酰半胱氨酸组,10只/组。采用全身体积描记系统检测肺功能;HE染色法观察各组小鼠肺病理并进行肺泡和气道评估;ELISA法检测小鼠肺泡灌洗液中炎症因子IL-1β、IL-6、TNF-α含量;采用免疫组化法检测肺组织PI3K、Akt、p-Akt、p65和p-p65的表达水平。 结果 从YZF中共鉴定出156种化学成分,包括26种黄酮及黄酮苷类、27种生物碱类、11种皂苷类等成分;从含药血清样本中鉴定出43种原型入血成分。网络药理学分析获得YZF成分704个靶点和COPD 1199个疾病靶点;整合分析成分-疾病靶点后发现YZF缓解COPD可能与PI3K-Akt等信号通路相关;动物实验表明YZF可增加COPD模型小鼠的平均肺泡数和呼气峰流速(P<0.05);降低COPD模型小鼠的肺泡平均线性截距和支气管壁厚度和肺系数以及肺泡灌洗液的IL-1β、IL-6、TNF-α含量(P<0.01),并能够抑制肺组织PI3K、Akt、p-Akt、p65和p-p65蛋白的表达(P<0.01)。 结论 YZF能有效改善COPD症状,缓解COPD炎症,机制可能与其通过多成分-多靶点作用抑制PI3K/Akt/NF-κB通路相关。
王立明, 陈宏睿, 杜燕, 赵鹏, 王玉洁, 田燕歌, 刘新光, 李建生. 益气滋肾方通过抑制PI3K/Akt/NF-κB通路改善小鼠慢性阻塞性肺疾病的炎症反应[J]. 南方医科大学学报, 2025, 45(7): 1409-1422.
Liming WANG, Hongrui CHEN, Yan DU, Peng ZHAO, Yujie WANG, Yange TIAN, Xinguang LIU, Jiansheng LI. Yiqi Zishen Formula ameliorates inflammation in mice with chronic obstructive pulmonary disease by inhibiting the PI3K/Akt/NF-κB signaling pathway[J]. Journal of Southern Medical University, 2025, 45(7): 1409-1422.
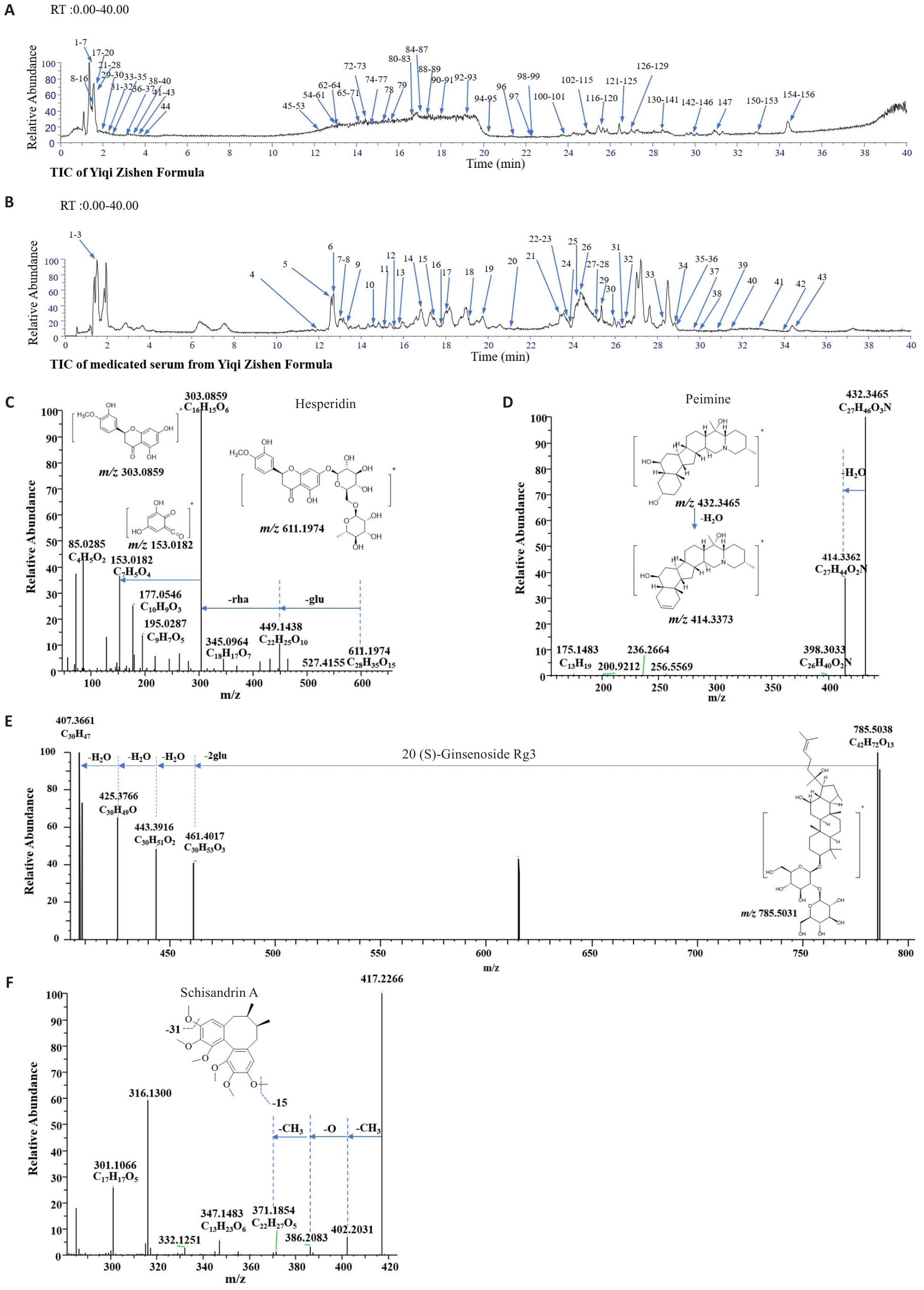
图1 益气滋肾方的化学成分与原型入血成分的鉴定
Fig.1 Identification of chemical components and prototype blood-absorbed constituents in Yiqi Zishen Formula (YZF). A: TIC of YZF in positive ion mode. B: TIC of prototype constituents of YZF in mediated rat serum in positive ion mode. C-F: MS/MS spectra and fragment assignment of hesperidin, peimine, 20(S)-ginsenoside Rg3, and schisandrin A, respectively.
| NO. | Compound | Retention time (min) | Formula | MS/MS | Error (ppm) |
|---|---|---|---|---|---|
| 1 | Synephrine | 1.6 | C9H13NO2 | 150.0917 | 1.20 |
| 2 | Histidinyl-asparagine | 1.62 | C10H13N4O3 | 192.0785; 120.0650; 103.0390 | -5.53 |
| 3 | Fructose-phenylalanine | 1.62 | C15H21NO7 | 310.1298; 292.1188 | -1.52 |
| 4 | Chlorogenic acid | 12.03 | C16H18O9 | 163.0389; 145.0285; 135.0439; 117.0336 | -6.19 |
| 5 | p-Coumaric acid O-glycoside (a) | 12.69 | C15H16O8 | 163.0390 | 1.24 |
| 6 | Schizonepetoside A | 12.72 | C16H26O7 | 169.1223 | -1.71 |
| 7 | Neostemonine or its isomer (a) | 13.15 | C18H25NO4 | 246.1492 | 0.50 |
| 8 | Croomine | 13.19 | C18H27NO4 | 248.1645 | -1.09 |
| 9 | Neostemonine or its isomer (b) | 13.43 | C18H25NO4 | 246.1491 | 0.50 |
| 10 | Verticine-N-Oxide | 14.55 | C27H45NO4 | 430.3311 | -1.44 |
| 11 | Diosmetin-6-C-glucoside | 15.06 | C22H22O11 | 445.1081; 397.0879; 367.0805 | -1.22 |
| 12 | Verticinone-N-Oxide or its isomer (a) | 15.51 | C27H43NO4 | 428.3118; 410.3059 | -1.60 |
| 13 | Verticinone-N-Oxide or its isomer (b) | 15.83 | C27H43NO4 | 428.3174; 410.3051 | -1.60 |
| 14 | Stemoninine or its isomer (a) | 16.78 | C22H31NO5 | 372.2172; 316.0912 | -2.08 |
| 15 | Stemoninine or its isomer (b) | 17.4 | C22H31NO5 | 372.2175; 316.0909 | -3.26 |
| 16 | Peimisine | 17.75 | C27H41O3N | 410.3039 | -1.85 |
| 17 | Meranzin | 18 | C15H16O4 | 189.0547 | -0.53 |
| 18 | Peimine | 19.18 | C27H45O3N | 414.3372 | -0.33 |
| 19 | TuberostemonineK/Neotuberostemonine | 19.73 | C22H33NO4 | 358.2334; 348.2545; 302.2115; 250.1802 | 0.21 |
| 20 | Peiminine | 21.25 | C27H43NO3 | 412.3218; 396.2900 | -0.91 |
| 21 | Peimine isomer | 23.46 | C27H45O3N | 414.3374 | -0.75 |
| 22 | Gomisin R | 23.74 | C22H24O7 | 383.1506; | -1.28 |
| 23 | Dihydrocapsaicin | 23.76 | C18H29NO3 | 290.2121;262.2171; 192.1391;136.0762;122.0602 | -0.90 |
| 24 | Peiminine isomer | 23.97 | C27H43NO3 | 412.3222 | -2.26 |
| 25 | Hapepunine | 24.31 | C28H47NO2 | 412.3602 | -1.44 |
| 26 | Fritillarizine/Puqiedinone | 24.4 | C27H43NO2 | 396.3281 | -1.08 |
| 27 | Gentrogenin or its isomer (a) | 25.1 | C27H40O4 | 411.2920 | 5.86 |
| 28 | Gomisin S | 25.2 | C23H30O7 | 401.1963; 369.1695 | -1.18 |
| 29 | Gentrogenin or its isomer (b) | 25.44 | C27H40O4 | 411.2888 | -3.86 |
| 30 | Methyl eugenol | 26.14 | C11H14O2 | 151.0752; 116.0011 | 0.16 |
| 31 | Schisandrol A | 26.43 | C24H32O7 | 415.2115; 400.1886; 384.1931; 369.1700; 338.1512 | -1.69 |
| 32 | Nobiletin | 26.65 | C21H22O8 | 388.1158; 373.0921; 358.0685; 355.0820; 327.0869; 211.0245 | -3.12 |
| 33 | Schisanhenol or its isomer (a) | 28.39 | C23H30O6 | 388.1881; 371.1856; 340.1665; 325.1430 | -2.42 |
| 34 | Schisantherin A | 28.85 | C30H32O9 | 415.1725; 268.9779; 91.0565 | -6.24 |
| 35 | Schisanhenol or its isomer (b) | 28.87 | C23H30O6 | 388.1876; 371.1868; 340.1668; 325.1438 | -2.42 |
| 36 | (-)-Gomisin L2 | 28.87 | C22H26O6 | 371.1465 | -1.69 |
| 37 | 9-Tetradecenoic acid | 29.8 | C14H26O2 | 209.1927 | -1.06 |
| 38 | Schisandrin A | 30.09 | C24H32O6 | 402.2043; 386.2090; 371.1852; 347.1491; 316.1307; 301.1073 | -1.23 |
| 39 | Schisandrin B | 30.88 | C23H28O6 | 386.1725; 370.1776; 355.1534; 300.0990; 285.0758 | -1.31 |
| 40 | Wuweizisu C | 31.53 | C22H24O6 | 285.0756; 257.0808; 227.0703; 212.0466 | 3.93 |
| 41 | Gondoic acid or its isomer (a) | 32.83 | C20H38O2 | 265.2524; 247.2425 | -0.57 |
| 42 | Gondoic acid or its isomer (b) | 33.96 | C20H38O2 | 293.2838 | -1.94 |
| 43 | Gondoic acid or its isomer (c) | 34.48 | C20H38O2 | 293.2824 | -3.41 |
表1 益气滋肾方原型入血成分鉴定
Tab.1 Exogenous blood components of Yiqi Zishen Formula in medicated rat serum
| NO. | Compound | Retention time (min) | Formula | MS/MS | Error (ppm) |
|---|---|---|---|---|---|
| 1 | Synephrine | 1.6 | C9H13NO2 | 150.0917 | 1.20 |
| 2 | Histidinyl-asparagine | 1.62 | C10H13N4O3 | 192.0785; 120.0650; 103.0390 | -5.53 |
| 3 | Fructose-phenylalanine | 1.62 | C15H21NO7 | 310.1298; 292.1188 | -1.52 |
| 4 | Chlorogenic acid | 12.03 | C16H18O9 | 163.0389; 145.0285; 135.0439; 117.0336 | -6.19 |
| 5 | p-Coumaric acid O-glycoside (a) | 12.69 | C15H16O8 | 163.0390 | 1.24 |
| 6 | Schizonepetoside A | 12.72 | C16H26O7 | 169.1223 | -1.71 |
| 7 | Neostemonine or its isomer (a) | 13.15 | C18H25NO4 | 246.1492 | 0.50 |
| 8 | Croomine | 13.19 | C18H27NO4 | 248.1645 | -1.09 |
| 9 | Neostemonine or its isomer (b) | 13.43 | C18H25NO4 | 246.1491 | 0.50 |
| 10 | Verticine-N-Oxide | 14.55 | C27H45NO4 | 430.3311 | -1.44 |
| 11 | Diosmetin-6-C-glucoside | 15.06 | C22H22O11 | 445.1081; 397.0879; 367.0805 | -1.22 |
| 12 | Verticinone-N-Oxide or its isomer (a) | 15.51 | C27H43NO4 | 428.3118; 410.3059 | -1.60 |
| 13 | Verticinone-N-Oxide or its isomer (b) | 15.83 | C27H43NO4 | 428.3174; 410.3051 | -1.60 |
| 14 | Stemoninine or its isomer (a) | 16.78 | C22H31NO5 | 372.2172; 316.0912 | -2.08 |
| 15 | Stemoninine or its isomer (b) | 17.4 | C22H31NO5 | 372.2175; 316.0909 | -3.26 |
| 16 | Peimisine | 17.75 | C27H41O3N | 410.3039 | -1.85 |
| 17 | Meranzin | 18 | C15H16O4 | 189.0547 | -0.53 |
| 18 | Peimine | 19.18 | C27H45O3N | 414.3372 | -0.33 |
| 19 | TuberostemonineK/Neotuberostemonine | 19.73 | C22H33NO4 | 358.2334; 348.2545; 302.2115; 250.1802 | 0.21 |
| 20 | Peiminine | 21.25 | C27H43NO3 | 412.3218; 396.2900 | -0.91 |
| 21 | Peimine isomer | 23.46 | C27H45O3N | 414.3374 | -0.75 |
| 22 | Gomisin R | 23.74 | C22H24O7 | 383.1506; | -1.28 |
| 23 | Dihydrocapsaicin | 23.76 | C18H29NO3 | 290.2121;262.2171; 192.1391;136.0762;122.0602 | -0.90 |
| 24 | Peiminine isomer | 23.97 | C27H43NO3 | 412.3222 | -2.26 |
| 25 | Hapepunine | 24.31 | C28H47NO2 | 412.3602 | -1.44 |
| 26 | Fritillarizine/Puqiedinone | 24.4 | C27H43NO2 | 396.3281 | -1.08 |
| 27 | Gentrogenin or its isomer (a) | 25.1 | C27H40O4 | 411.2920 | 5.86 |
| 28 | Gomisin S | 25.2 | C23H30O7 | 401.1963; 369.1695 | -1.18 |
| 29 | Gentrogenin or its isomer (b) | 25.44 | C27H40O4 | 411.2888 | -3.86 |
| 30 | Methyl eugenol | 26.14 | C11H14O2 | 151.0752; 116.0011 | 0.16 |
| 31 | Schisandrol A | 26.43 | C24H32O7 | 415.2115; 400.1886; 384.1931; 369.1700; 338.1512 | -1.69 |
| 32 | Nobiletin | 26.65 | C21H22O8 | 388.1158; 373.0921; 358.0685; 355.0820; 327.0869; 211.0245 | -3.12 |
| 33 | Schisanhenol or its isomer (a) | 28.39 | C23H30O6 | 388.1881; 371.1856; 340.1665; 325.1430 | -2.42 |
| 34 | Schisantherin A | 28.85 | C30H32O9 | 415.1725; 268.9779; 91.0565 | -6.24 |
| 35 | Schisanhenol or its isomer (b) | 28.87 | C23H30O6 | 388.1876; 371.1868; 340.1668; 325.1438 | -2.42 |
| 36 | (-)-Gomisin L2 | 28.87 | C22H26O6 | 371.1465 | -1.69 |
| 37 | 9-Tetradecenoic acid | 29.8 | C14H26O2 | 209.1927 | -1.06 |
| 38 | Schisandrin A | 30.09 | C24H32O6 | 402.2043; 386.2090; 371.1852; 347.1491; 316.1307; 301.1073 | -1.23 |
| 39 | Schisandrin B | 30.88 | C23H28O6 | 386.1725; 370.1776; 355.1534; 300.0990; 285.0758 | -1.31 |
| 40 | Wuweizisu C | 31.53 | C22H24O6 | 285.0756; 257.0808; 227.0703; 212.0466 | 3.93 |
| 41 | Gondoic acid or its isomer (a) | 32.83 | C20H38O2 | 265.2524; 247.2425 | -0.57 |
| 42 | Gondoic acid or its isomer (b) | 33.96 | C20H38O2 | 293.2838 | -1.94 |
| 43 | Gondoic acid or its isomer (c) | 34.48 | C20H38O2 | 293.2824 | -3.41 |
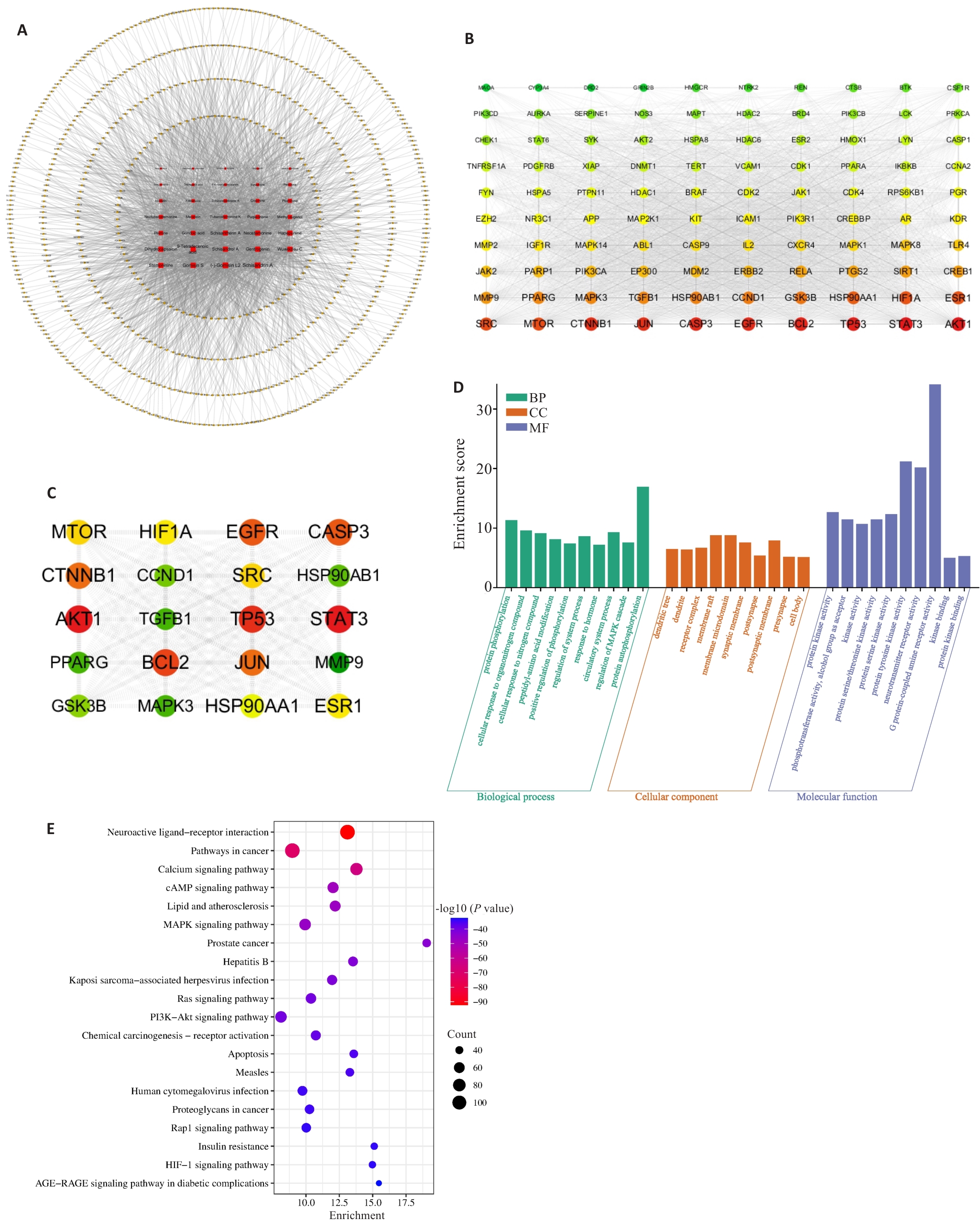
图 2 YZF活性成分的网络分析
Fig.2 Network analysis of active components in YZF. A: Network diagram of 34 components and their 704 associated targets. B: PPI network diagram of the top 100 targets. C: PPI network diagram of the top 20 targets. D: GO enrichment analysis of 704 targets of YZF components. E: KEGG enrichment analysis of 704 targets of YZF components.
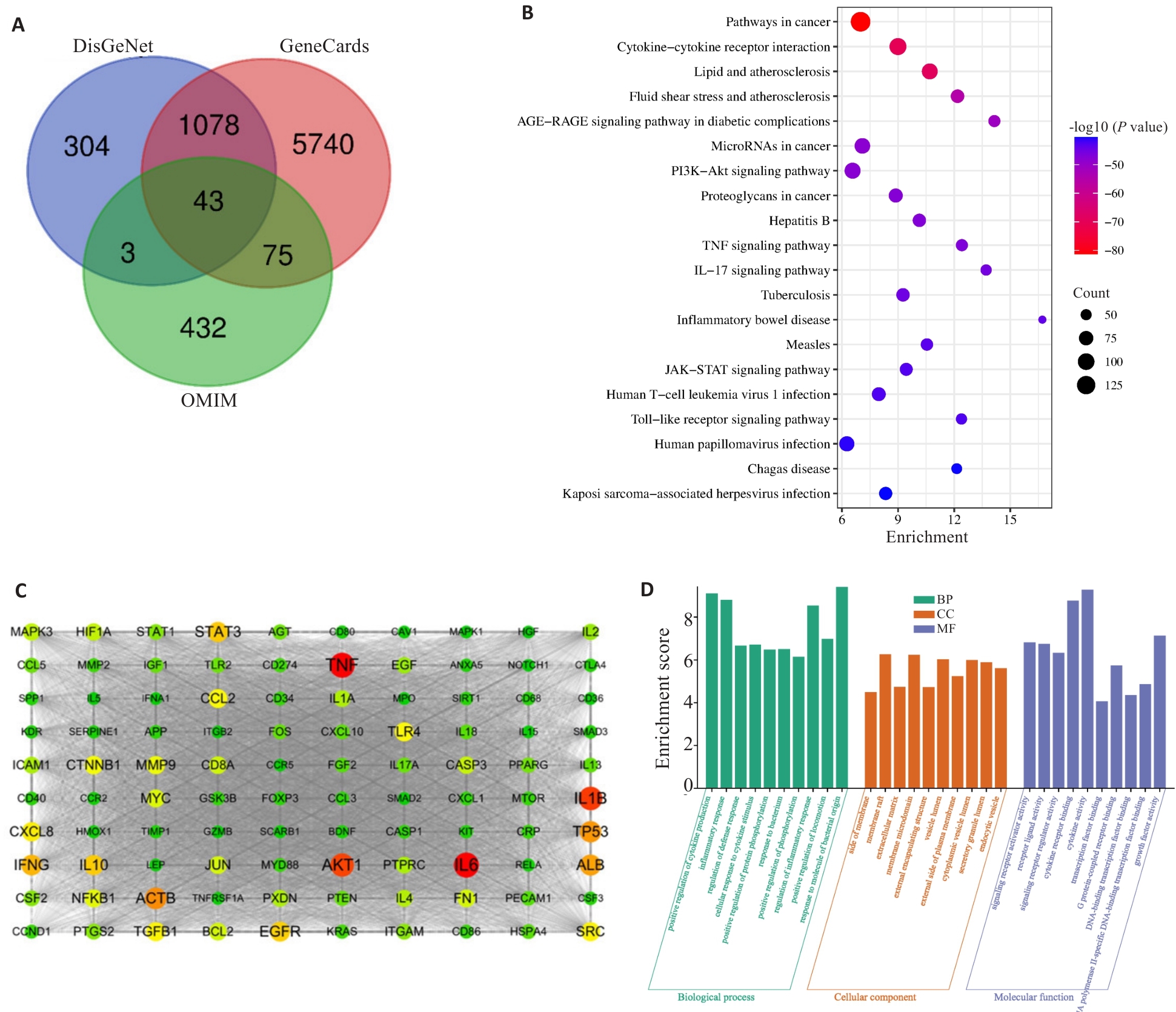
图 3 COPD靶点的网络分析
Fig.3 Network analysis of COPD Targets. A: Venn diagram of COPD targets from 3 disease databases. B: KEGG enrichment analysis of COPD disease targets. C: PPI network of the top 100 COPD targets. D: GO enrichment analysis of COPD disease targets.
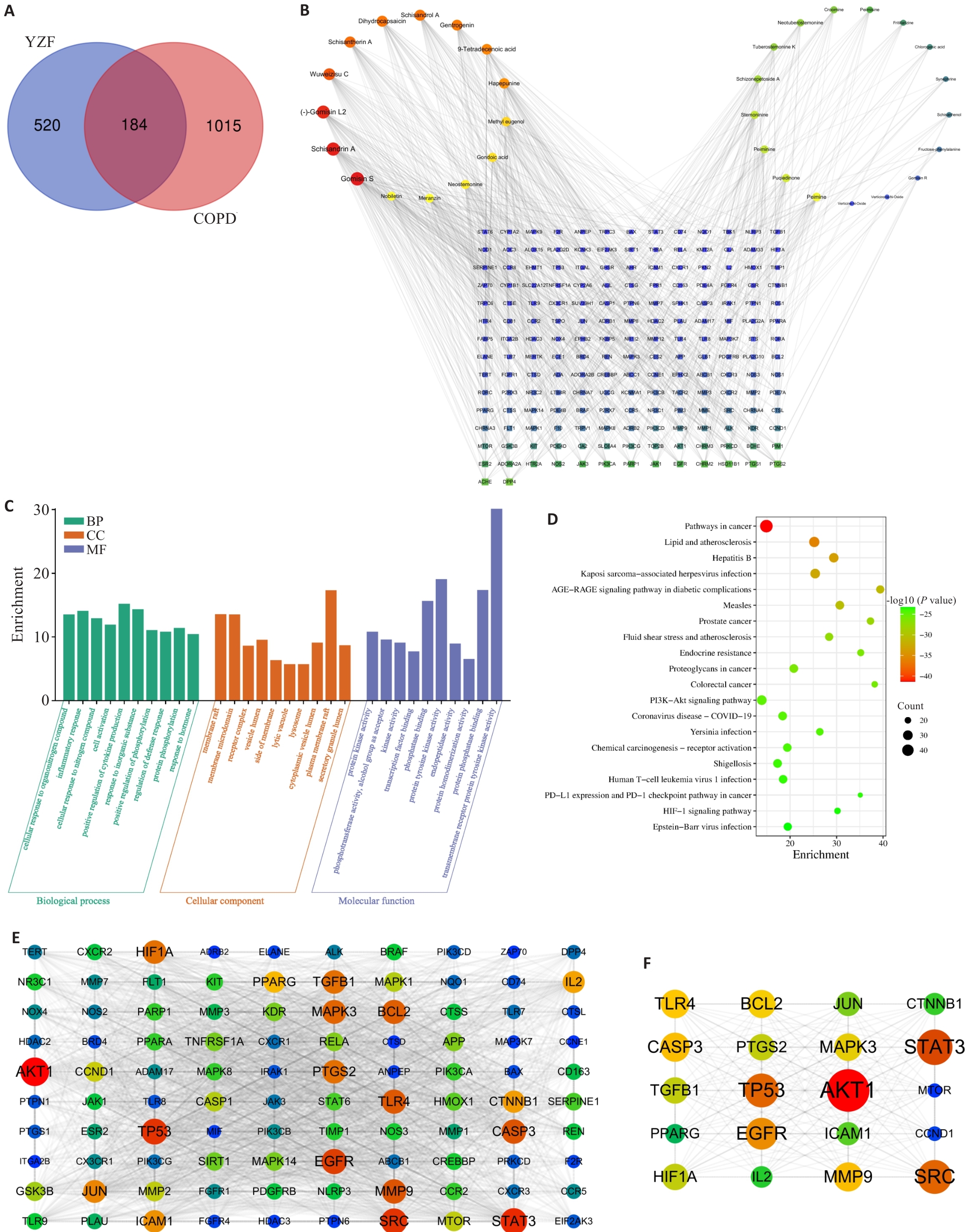
图4 YZF与COPD交集靶点的网络分析
Fig.4 Network analysis of shared targets between YZF and COPD. A: Venn Diagram of YZF targets and COPD disease targets. B: Component-target network construction. C: GO enrichment analysis of 184 intersection targets. D: KEGG enrichment analysis of 184 intersection targets. E: PPI network of the top 100 intersection targets. F: Core targets extracted from the 184 intersection targets.
| Ingredient | Degree | Closeness centrality | Betweenness centrality |
|---|---|---|---|
| Gomisin S | 46 | 0.4087 | 0.1222 |
| Schisandrin A | 45 | 0.4026 | 0.1143 |
| (-)-Gomisin L2 | 44 | 0.4011 | 0.1029 |
| Wuweizisu C | 38 | 0.3923 | 0.0869 |
| Schisantherin A | 35 | 0.3894 | 0.0765 |
| Dihydrocapsaicin | 34 | 0.3894 | 0.1012 |
| Schisandrol A | 34 | 0.3881 | 0.0748 |
| Gentrogenin | 33 | 0.3759 | 0.0782 |
| 9-Tetradecenoic acid | 32 | 0.3881 | 0.0940 |
| Hapepunine | 31 | 0.3853 | 0.0848 |
表2 YZF前10的活性成分(以度值排序)的拓扑学参数
Tab.2 Topological parameters of top 10 active components in YZF ranked by degree value
| Ingredient | Degree | Closeness centrality | Betweenness centrality |
|---|---|---|---|
| Gomisin S | 46 | 0.4087 | 0.1222 |
| Schisandrin A | 45 | 0.4026 | 0.1143 |
| (-)-Gomisin L2 | 44 | 0.4011 | 0.1029 |
| Wuweizisu C | 38 | 0.3923 | 0.0869 |
| Schisantherin A | 35 | 0.3894 | 0.0765 |
| Dihydrocapsaicin | 34 | 0.3894 | 0.1012 |
| Schisandrol A | 34 | 0.3881 | 0.0748 |
| Gentrogenin | 33 | 0.3759 | 0.0782 |
| 9-Tetradecenoic acid | 32 | 0.3881 | 0.0940 |
| Hapepunine | 31 | 0.3853 | 0.0848 |
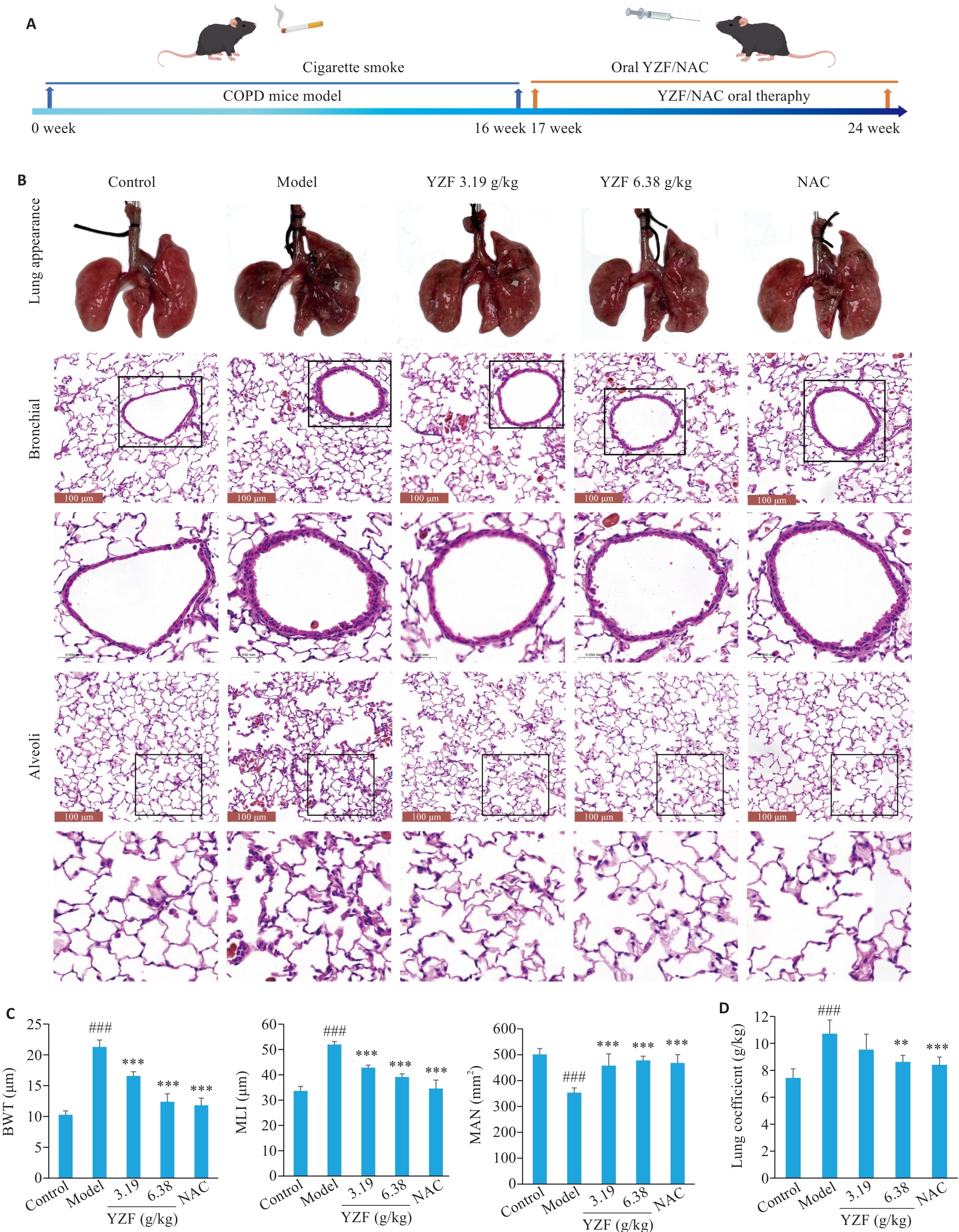
图5 动物实验流程及肺病理相关指标
Fig.5 Animal experimental workflow and lung pathology-related indicators. A: Timeline of COPD mouse modeling and interventions. B: Lung gross appearance and HE staining of airways and alveoli in each group. C: Quantitative analysis of airways and alveoli HE staining. D: Lung coefficient (ratio of lung weight to body weight) of the mice. n=6, **P<0.01, ***P<0.001 vs Model; ###P<0.001 vs Control. YZF: Yiqi Zishen Formula; NAC: N-Acetylcysteine.
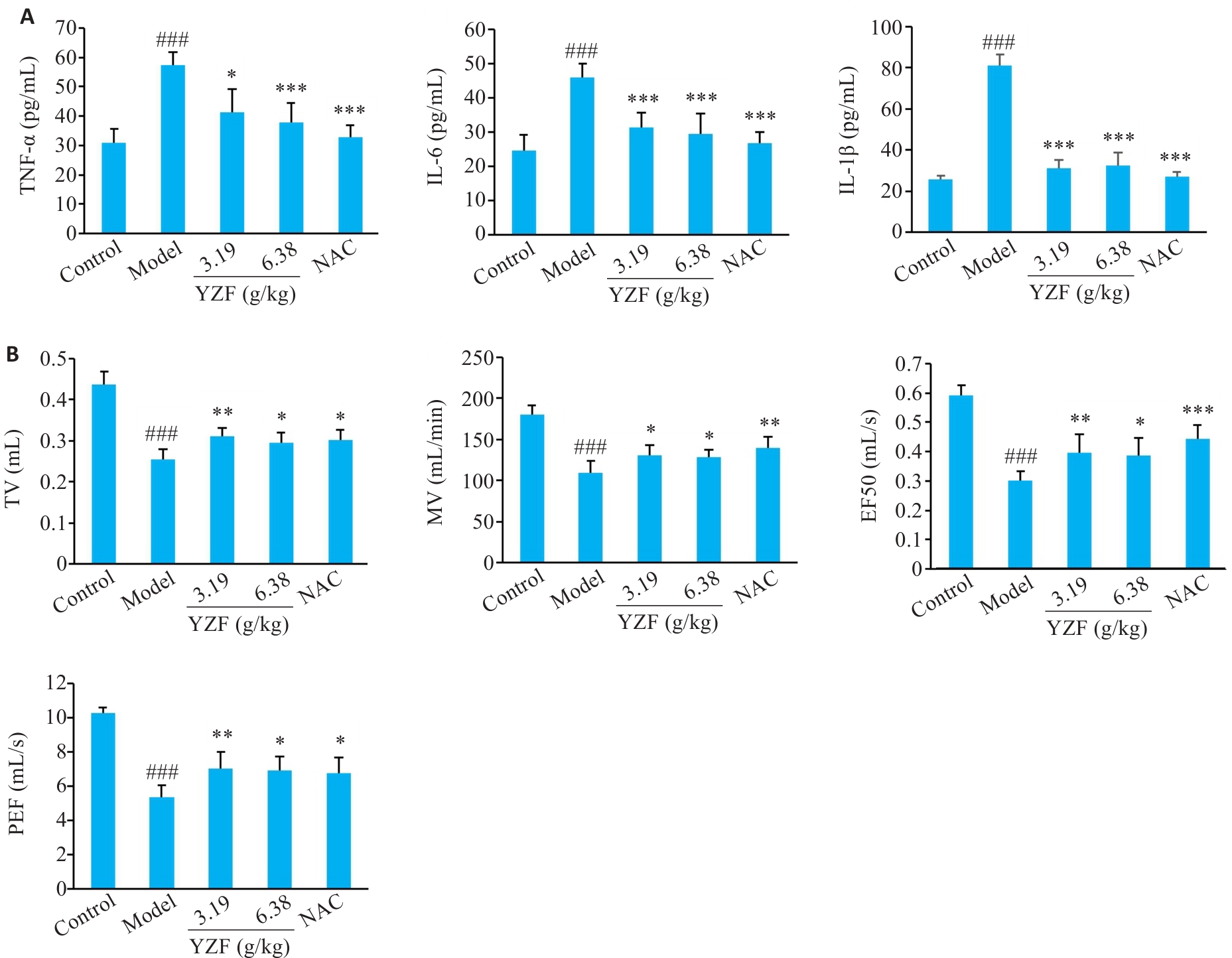
图 6 各组小鼠肺功能变化及肺部炎症因子表达情况
Fig.6 Changes in pulmonary function and expression of lung inflammatory cytokines in each group. A: ELISA analysis of inflammatory cytokines in each group. B: Pulmonary function in each group. n=6, *P<0.05, **P<0.01, ***P<0.001 vs Model; ###P<0.001 vs Control. YZF: Yiqi Zishen Formula.
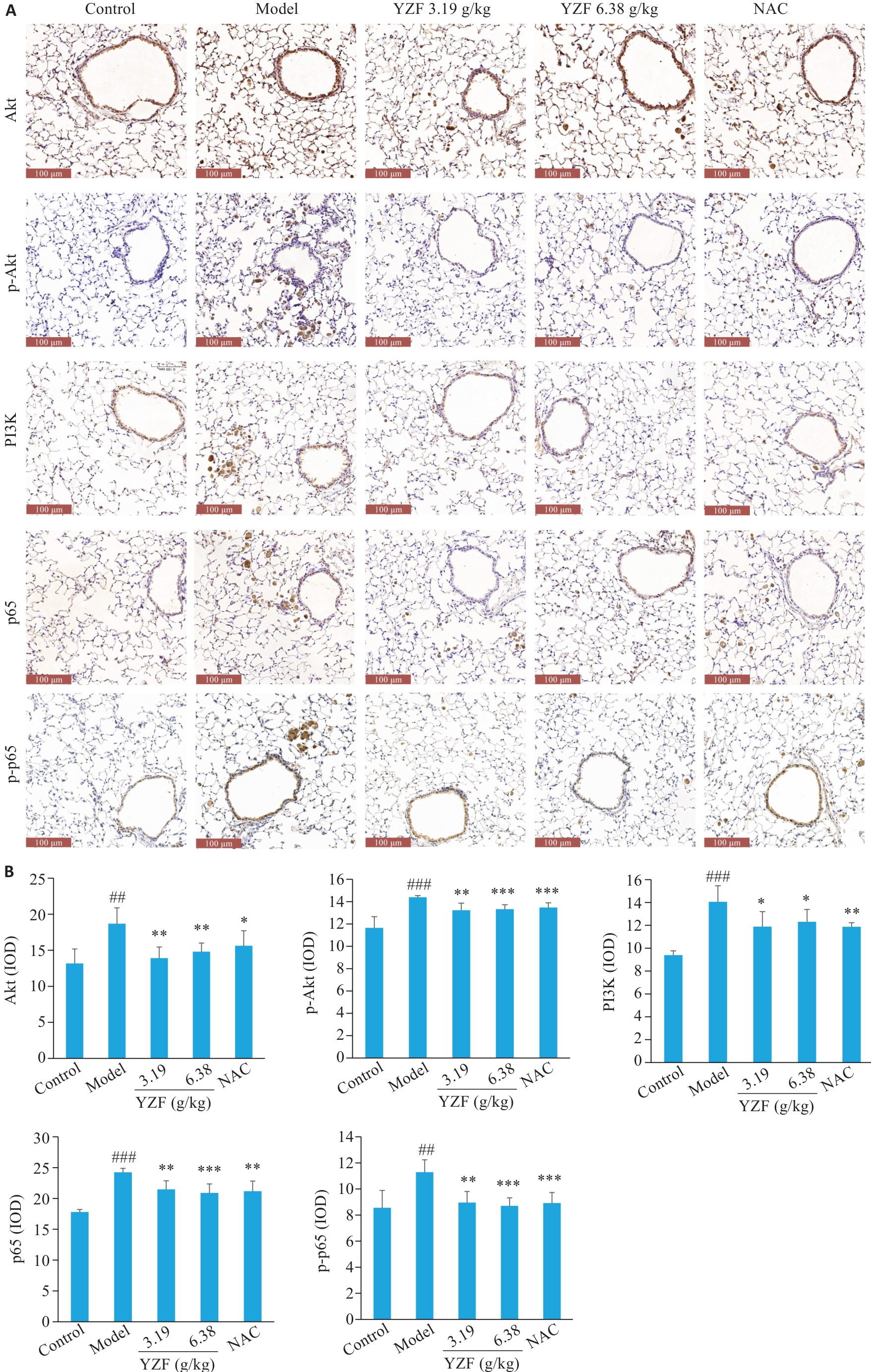
图7 PI3K-Akt相关通路蛋白的免疫组化及量化
Fig.7 Immunohistochemistry and quantitative analysis of PI3K-Akt pathway-related proteins. A: Immunohistochemical (IHC) staining images of PI3K, Akt, p-Akt, p65, and p-p65 in the lung tissue. B: Integrated optical density (IOD) analysis of protein expressions in IHC staining. n=6, *P<0.05, **P<0.01, ***P<0.001 vs Model; ##P<0.01, ###P<0.001 vs Control. YZF: Yiqi Zishen Formula.
| [1] | Agustí A, Celli BR, Criner GJ, et al. Global initiative for chronic obstructive lung disease 2023 report: gold executive summary[J]. Eur Respir J, 2023, 61(4): 2300239. doi:10.1183/13993003.00239-2023 |
| [2] | 丁莉莉, 冯淬灵, 赵克明. 慢性阻塞性肺疾病中医研究进展[J]. 辽宁中医药大学学报, 2023, 25(3): 164-8. |
| [3] | 李建生, 余学庆. 慢性阻塞性肺疾病中医分期分级防治策略[J]. 中医杂志, 2019, 60(22): 1895-9. |
| [4] | Wang MH, Li JS, Li SY, et al. Effects of comprehensive therapy based on traditional Chinese medicine patterns on older patients with chronic obstructive pulmonary disease: a subgroup analysis from a four-center, randomized, controlled study[J]. Front Med, 2014, 8(3): 368-75. doi:10.1007/s11684-014-0360-0 |
| [5] | 田燕歌, 李 亚, 李建生, 等. 调补肺肾法对COPD大鼠JAK/STAT信号转导的影响及远后效应[J]. 中国中药杂志, 2013, 38(1): 75-80. doi:10.4268/cjcmm20130117 |
| [6] | Qin YQ, Chen YL, Zhao P, et al. Tiaobu Feishen therapy inhibits inflammation induced by cigarette smoke extracts in a human monocyte/macrophage cell line[J]. J Tradit Chin Med, 2021, 41(3): 360-6. doi:10.19852/j.cnki.jtcm.2021.03.003 |
| [7] | Dong HR, Liu XF, Zheng WC, et al. Three Tiaobu Feishen formulae reduces cigarette smoke-induced inflammation in human airway epithelial cells[J]. J Tradit Chin Med, 2020, 40(3): 386-92. |
| [8] | Chen YL, Wu YS, Li JS, et al. Three Tiaobu Feishen therapies protect human alveolar epithelial cells against cigarette smoking and tumor necrosis factor: induced inflammation by nuclear factor-kappa B pathway[J]. J Tradit Chin Med, 2019, 39(1): 45-9. |
| [9] | 李高峰, 刘淑娟, 李 亚, 等. 调补肺肾三法通过抑制ERK1/2信号通路改善COPD大鼠气道黏液高分泌[J]. 中国实验动物学报, 2024, 32(4): 411-22. |
| [10] | 陈旺宇, 节笑笑, 陈甜甜, 等. 中药血清药物化学研究现状[J]. 中国医药科学, 2023, 13(4): 26-9, 86. |
| [11] | Arifin WN, Zahiruddin WM. Sample size calculation in animal studies using resource equation approach[J]. Malays J Med Sci, 2017, 24(5): 101-5. doi:10.21315/mjms2017.24.5.11 |
| [12] | 国家药典委员会. 中华人民共和国药典-三部: 2020年版[M]. 北京: 中国医药科技出版社, 2020. |
| [13] | Wu JY, Cai BR, Zhang A, et al. Chemical identification and antioxidant screening of Bufei yishen formula using an offline DPPH ultrahigh-performance liquid chromatography Q-extractive orbitrap MS/MS[J]. Int J Anal Chem, 2022, 2022: 1423801. doi:10.1155/2022/1423801 |
| [14] | Shao D, Liu XG, Wu JY, et al. Identification of the active compounds and functional mechanisms of Jinshui Huanxian formula in pulmonary fibrosis by integrating serum pharmacochemistry with network pharmacology[J]. Phytomedicine, 2022, 102: 154177. doi:10.1016/j.phymed.2022.154177 |
| [15] | 梅晓峰, 赵 鹏, 卢瑞龙, 等. 慢性阻塞性肺疾病小鼠模型制备方法的比较研究[J]. 中国病理生理杂志, 2022, 38(1): 178-85. doi:10.3969/j.issn.1000-4718.2022.01.023 |
| [16] | Ye JB, Ren G, Li WY, et al. Characterization and identification of prenylated flavonoids from Artocarpus heterophyllus lam. roots by quadrupole time-of-flight and linear trap quadrupole orbitrap mass spectrometry[J]. Molecules, 2019, 24(24): 4591. doi:10.3390/molecules24244591 |
| [17] | 慢性阻塞性肺疾病中西医结合管理专家共识写作组, 何权瀛, 冯淬灵. 慢性阻塞性肺疾病中西医结合管理专家共识(2023版)[J]. 中国全科医学, 2023, 26(35): 4359-71. |
| [18] | 徐砚通, 唐雅楠. 方剂“组-谱-效-机” 研究模式探讨[J]. 中国中药杂志, 2025, 50(3): 840-5. doi:10.19540/j.cnki.cjcmm.20241017.601 |
| [19] | 祁建宏, 董芳旭. 黄酮类化合物药理作用研究进展[J]. 北京联合大学学报, 2020, 34(3): 89-92. doi:10.16255/j.cnki.ldxbz.2020.03.014 |
| [20] | Hosawi S. Current update on role of hesperidin in inflammatory lung diseases: chemistry, pharmacology, and drug delivery approaches[J]. Life (Basel), 2023, 13(4): 937. doi:10.3390/life13040937 |
| [21] | Liu CL, Zhu XM, Aga EB, et al. Ebeiedinone and peimisine inhibit cigarette smoke extract-induced oxidative stress injury and apo-ptosis in BEAS-2B cells[J]. Cell Stress Chaperones, 2024, 29(6): 697-708. doi:10.1016/j.cstres.2024.10.001 |
| [22] | 李凡凡, 郑越超. 天然生物碱抗菌活性及机理研究[J]. 医学信息, 2021, 34(17): 49-52. doi:10.3969/j.issn.1006-1959.2021.17.013 |
| [23] | Wang LN, Zhang YX, Song ZM, et al. Ginsenosides: a potential natural medicine to protect the lungs from lung cancer and inflammatory lung disease[J]. Food Funct, 2023, 14(20): 9137-66. doi:10.1039/d3fo02482b |
| [24] | Zeng JM, Liao SD, Liang Z, et al. Schisandrin A regulates the Nrf2 signaling pathway and inhibits NLRP3 inflammasome activation to interfere with pyroptosis in a mouse model of COPD[J]. Eur J Med Res, 2023, 28(1): 217. doi:10.1186/s40001-023-01190-8 |
| [25] | Guo Q, Jin YZ, Chen XY, et al. NF-κB in biology and targeted therapy: new insights and translational implications[J]. Signal Transduct Target Ther, 2024, 9(1): 53. doi:10.1038/s41392-024-01757-9 |
| [26] | Liu YH, Kong HB, Cai HP, et al. Progression of the PI3K/Akt signaling pathway in chronic obstructive pulmonary disease[J]. Front Pharmacol, 2023, 14: 1238782. doi:10.3389/fphar.2023.1238782 |
| [27] | 杨 洋, 李竹英, 田春燕, 等. 中医药干预慢性阻塞性肺疾病相关信号通路的研究进展[J]. 中国实验方剂学杂志, 2024, 30(15): 244-55. |
| [28] | 张 钰, 涂 星, 张 燕, 等. 文王一支笔的抗炎作用机制: 基于网络药理学、分子对接及实验验证[J]. 南方医科大学学报, 2023, 43(3): 383-92. doi:10.12122/j.issn.1673-4254.2023.03.07 |
| [29] | 白莉敏, 吕 行, 任引刚, 等. miR-22-3p通过激活PTEN/PI3K/AKT/NF-κB信号通路加重慢性阻塞性肺疾病[J]. 现代生物医学进展, 2023, 23(10): 1835-42. |
| [30] | Thongin S, Den-Udom T, Uppakara K, et al. Beneficial effects of capsaicin and dihydrocapsaicin on endothelial inflammation, nitric oxide production and antioxidant activity[J]. Biomed Pharmacother, 2022, 154: 113521. doi:10.1016/j.biopha.2022.113521 |
| [31] | Dong Y, Zou YZ, Li T, et al. Schisandrol a alleviates allergic asthma in mice via regulating the NF-κB/IκBα and Nrf2/HO-1 signaling pathways[J]. J Med Food, 2025, 28(1): 28-37. doi:10.1089/jmf.2024.k.0117 |
| [32] | Chen JM, Li M, Chen R, et al. Gegen Qinlian standard decoction alleviated irinotecan-induced diarrhea via PI3K/AKT/NF-κB axis by network pharmacology prediction and experimental validation combination[J]. Chin Med, 2023, 18(1): 46. doi:10.1186/s13020-023-00747-3 |
| [33] | Kim JS, Yi HK. Schisandrin C enhances mitochondrial biogenesis and autophagy in C2C12 skeletal muscle cells: potential invo-lvement of anti-oxidative mechanisms[J]. Naunyn Schmiedebergs Arch Pharmacol, 2018, 391(2): 197-206. doi:10.1007/s00210-017-1449-1 |
| [34] | Qin YQ, Yang JF, Li HB, et al. Recent advances in the therapeutic potential of nobiletin against respiratory diseases[J]. Phyto-medicine, 2024, 128: 155506. doi:10.1016/j.phymed.2024.155506 |
| [1] | 陈鑫源, 吴成挺, 李瑞迪, 潘雪芹, 张耀丹, 陶俊宇, 林才志. 双术汤通过P53/SLC7A11/GPX4通路诱导胃癌细胞铁死亡[J]. 南方医科大学学报, 2025, 45(7): 1363-1371. |
| [2] | 范正媛, 沈子涵, 李亚, 沈婷婷, 李高峰, 李素云. 补肺益肾方对香烟烟雾提取物诱导的人支气管上皮细胞损伤的保护作用及其机制[J]. 南方医科大学学报, 2025, 45(7): 1372-1379. |
| [3] | 朱胤福, 李怡燃, 王奕, 黄颖而, 龚昆翔, 郝文波, 孙玲玲. 桂枝茯苓丸活性成分常春藤皂苷元通过抑制JAK2/STAT3通路抑制宫颈癌细胞的生长[J]. 南方医科大学学报, 2025, 45(7): 1423-1433. |
| [4] | 夏冰, 彭进, 丁九阳, 王杰, 唐国伟, 刘国杰, 王沄, 万昌武, 乐翠云. ATF3通过NF-κB信号通路调控动脉粥样硬化斑块内的炎症反应[J]. 南方医科大学学报, 2025, 45(6): 1131-1142. |
| [5] | 何丽君, 陈晓菲, 闫陈昕, 师林. 扶正化积汤治疗非小细胞肺癌的分子机制:基于网络药理学及体外实验验证[J]. 南方医科大学学报, 2025, 45(6): 1143-1152. |
| [6] | 王心恒, 邵小涵, 李童童, 张璐, 杨勤军, 叶卫东, 童佳兵, 李泽庚, 方向明. 平喘宁方通过调控HMGB1/Beclin-1轴介导的自噬改善患寒哮证大鼠的气道炎症[J]. 南方医科大学学报, 2025, 45(6): 1153-1162. |
| [7] | 李国永, 黎仁玲, 刘艺婷, 柯宏霞, 李菁, 王新华. 牛蒡子治疗小鼠病毒性肺炎后肺纤维化的机制:基于代谢组学、网络药理学和实验验证方法[J]. 南方医科大学学报, 2025, 45(6): 1185-1199. |
| [8] | 牛民主, 殷丽霞, 乔通, 尹林, 张可妮, 胡建国, 宋传旺, 耿志军, 李静. 旱莲苷A通过调控JAK2/STAT3通路抑制M1型巨噬细胞极化改善葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2025, 45(6): 1297-1306. |
| [9] | 管丽萍, 颜燕, 卢心怡, 李智峰, 高晖, 曹东, 侯晨曦, 曾靖宇, 李欣怡, 赵洋, 王俊杰, 方会龙. 复方积雪草减轻小鼠日本血吸虫引起的肝纤维化:通过调控TLR4/MyD88通路抑制炎症-纤维化级联反应[J]. 南方医科大学学报, 2025, 45(6): 1307-1316. |
| [10] | 唐培培, 谈勇, 殷燕云, 聂晓伟, 黄菁宇, 左文婷, 李玉玲. 调周滋阴方治疗早发性卵巢功能不全的疗效、安全性及作用机制[J]. 南方医科大学学报, 2025, 45(5): 929-941. |
| [11] | 张璐, 丁焕章, 许浩燃, 陈珂, 许博文, 杨勤军, 吴迪, 童佳兵, 李泽庚. 参芪补中方通过激活AMPK/SIRT1/PGC-1α改善COPD肺脾气虚证大鼠线粒体功能障碍[J]. 南方医科大学学报, 2025, 45(5): 969-976. |
| [12] | 梁晓涛, 熊一凡, 刘雪琪, 梁小珊, 朱晓煜, 谢炜. 活血疏风颗粒通过抑制TLR4/NF-κB通路改善慢性偏头痛小鼠的中枢敏化[J]. 南方医科大学学报, 2025, 45(5): 986-994. |
| [13] | 杨洋, 王凯, 柳鉴修, 周志谟, 贾雯, 吴思谋, 李金星, 何方, 程如越. 生命早期两歧双歧杆菌BD-1干预可缓解注意缺陷多动障碍雌性大鼠幼年期的多动行为[J]. 南方医科大学学报, 2025, 45(4): 702-710. |
| [14] | 朱正望, 王琳琳, 赵静涵, 马瑞雪, 余雨春, 蔡庆春, 王兵, 朱平生, 苗明三. 退黄合剂通过调控法尼醇X受体抑制NLRP3炎症小体改善α-萘异硫氰酸酯诱导的大鼠胆汁淤积[J]. 南方医科大学学报, 2025, 45(4): 718-724. |
| [15] | 冉念东, 刘杰, 徐剑, 张永萍, 郭江涛. 黑骨藤正丁醇萃取成分治疗大鼠阿尔茨海默病的药效学及作用机制[J]. 南方医科大学学报, 2025, 45(4): 785-798. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||