南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (4): 899-906.doi: 10.12122/j.issn.1673-4254.2026.04.18
• • 上一篇
陈潇煜1( ), 刘畅1, 杨绍杰1, 王娟2, 程平2, 尹红宇1, 王婧吉1(
), 刘畅1, 杨绍杰1, 王娟2, 程平2, 尹红宇1, 王婧吉1( ), 朱国旗2(
), 朱国旗2( )
)
收稿日期:2025-06-06
出版日期:2026-04-20
发布日期:2026-04-24
通讯作者:
王婧吉,朱国旗
E-mail:cxy199906@163.com;wangjingji@ahtcm.edu.cn;Guoqizhu@gmail.com
作者简介:陈潇煜,在读硕士研究生,E-mail: cxy199906@163.com
基金资助:
Xiaoyu CHEN1( ), Chang LIU1, Shaojie YANG1, Juan WANG2, Ping CHENG2, Hongyu YIN1, Jingji WANG1(
), Chang LIU1, Shaojie YANG1, Juan WANG2, Ping CHENG2, Hongyu YIN1, Jingji WANG1( ), Guoqi ZHU2(
), Guoqi ZHU2( )
)
Received:2025-06-06
Online:2026-04-20
Published:2026-04-24
Contact:
Jingji WANG, Guoqi ZHU
E-mail:cxy199906@163.com;wangjingji@ahtcm.edu.cn;Guoqizhu@gmail.com
Supported by:摘要:
目的 探究电针对单次长时间应激(SPS)小鼠恐惧记忆消退的影响及星形胶质细胞(AST)参与的潜在机制。 方法 将36只雄性C57BL/6J小鼠随机分为6组(6只/组):实验一分为对照组、模型(SPS)组、电针治疗(EA)组和帕罗西汀阳性药(PRX)组;实验二分为Saline组和CNO组,通过化学遗传病毒抑制小鼠海马AST。除对照组外,其余各组采用SPS法进行模型制备。造模7 d后,EA、Saline和CNO组取“百会”“足三里”进行电针干预,PRX组予帕罗西汀溶液灌胃,连续干预10 d。采用条件性恐惧实验和高架十字迷宫实验评价小鼠恐惧记忆消退和焦虑样行为,其中CNO组在检测开始前30 min予CNO腹腔注射;采用Western blotting检测小鼠海马GFAP和CX43的蛋白表达。 结果 与对照组比较,SPS组小鼠恐惧再暴露阶段3~15 min内的凝滞时间、消退测试阶段的凝滞时间增加(P<0.05),恐惧消退指数、进入开放臂的时间降低(P<0.01),GFAP的平均荧光强度和AST的分支数量在距离胞体40~60 μm降低(P<0.05),海马GFAP和CX43蛋白的表达降低(P<0.05)。与SPS组比较,EA组与PRX组小鼠恐惧再暴露阶段3~6 min内的凝滞时间、消退测试阶段的凝滞时间降低(P<0.05),恐惧消退指数、进入开放臂的时间增加(P<0.05),GFAP的平均荧光强度增加(P<0.001),海马CX43蛋白的表达增加(P<0.05);EA组海马AST的分支数量在距离胞体50 μm增加(P<0.05)。与Saline组比较,CNO组小鼠恐惧再暴露阶段3~9 min内和消退测试阶段的凝滞时间增加(P<0.05),恐惧消退指数和进入开放臂的时间降低(P<0.05)。 结论 电针“百会”“足三里”可通过激活海马AST改善SPS小鼠的恐惧记忆消退障碍和焦虑样行为。
陈潇煜, 刘畅, 杨绍杰, 王娟, 程平, 尹红宇, 王婧吉, 朱国旗. 电针“百会”“足三里”通过激活海马星形胶质细胞改善单次长时间应激小鼠恐惧记忆消退障碍[J]. 南方医科大学学报, 2026, 46(4): 899-906.
Xiaoyu CHEN, Chang LIU, Shaojie YANG, Juan WANG, Ping CHENG, Hongyu YIN, Jingji WANG, Guoqi ZHU. Electroacupuncture at Baihui and Zusanli improves fear memory extinction in single prolonged stress mice by activating hippocampal astrocytes[J]. Journal of Southern Medical University, 2026, 46(4): 899-906.
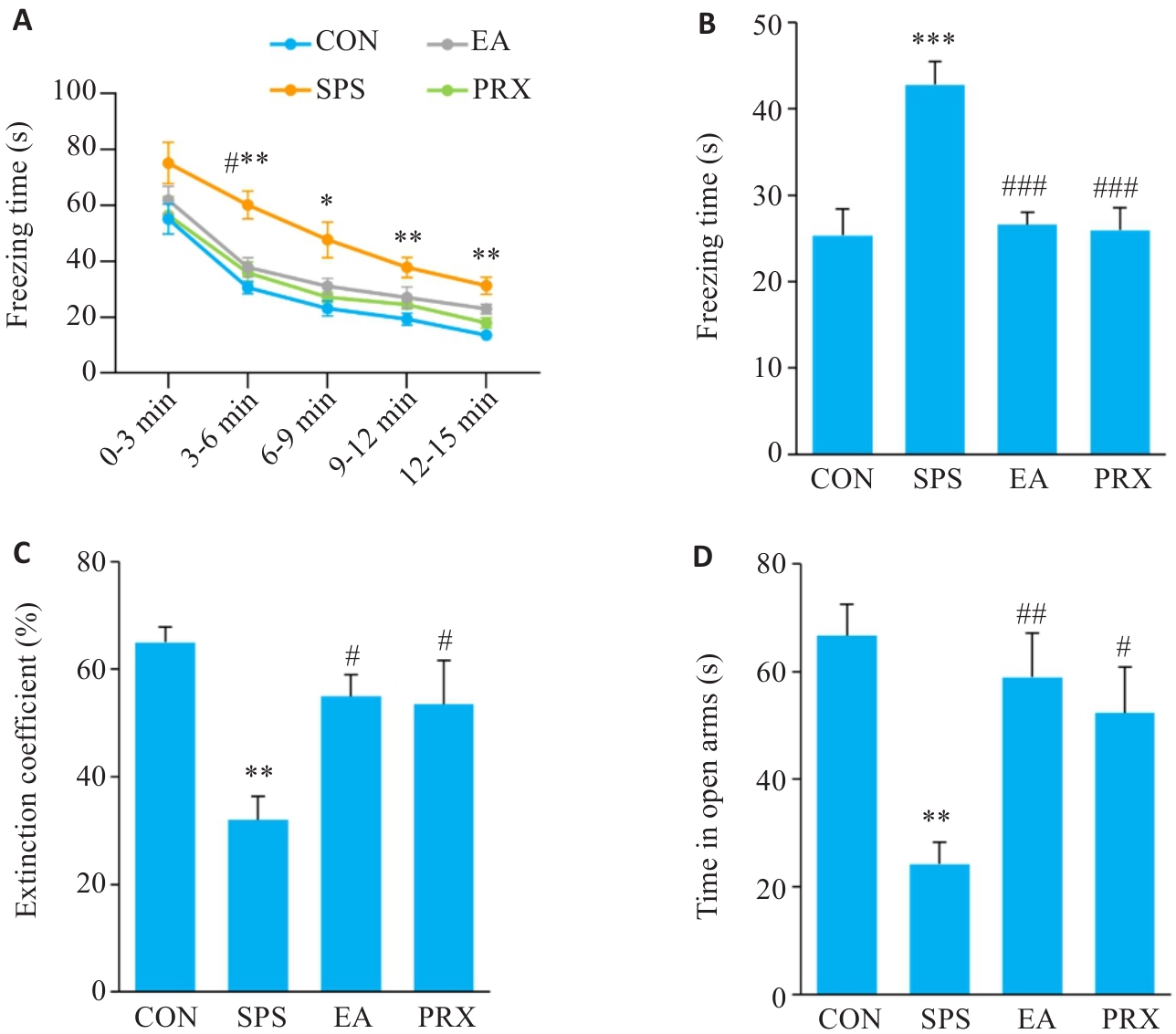
图2 电针能改善SPS小鼠的恐惧记忆消退障碍和焦虑样行为
Fig.2 EA alleviates abnormal fear memory extinction and anxiety-like behaviors in SPS mice (n=6). A: Freezing time of mice during the re-exposure phase. B: Freezing time of mice during the fear extinction phase. C: Extinction coefficient of mice in FCT. D: Time in open arms of mice. *P<0.05, **P<0.01, ***P<0.001 vs CON group; #P<0.05, ##P<0.01, ###P<0.001 vs SPS group. SPS: Single prolonged stress. EA: Electroacupuncture. PRX: Paroxetine positive drug.
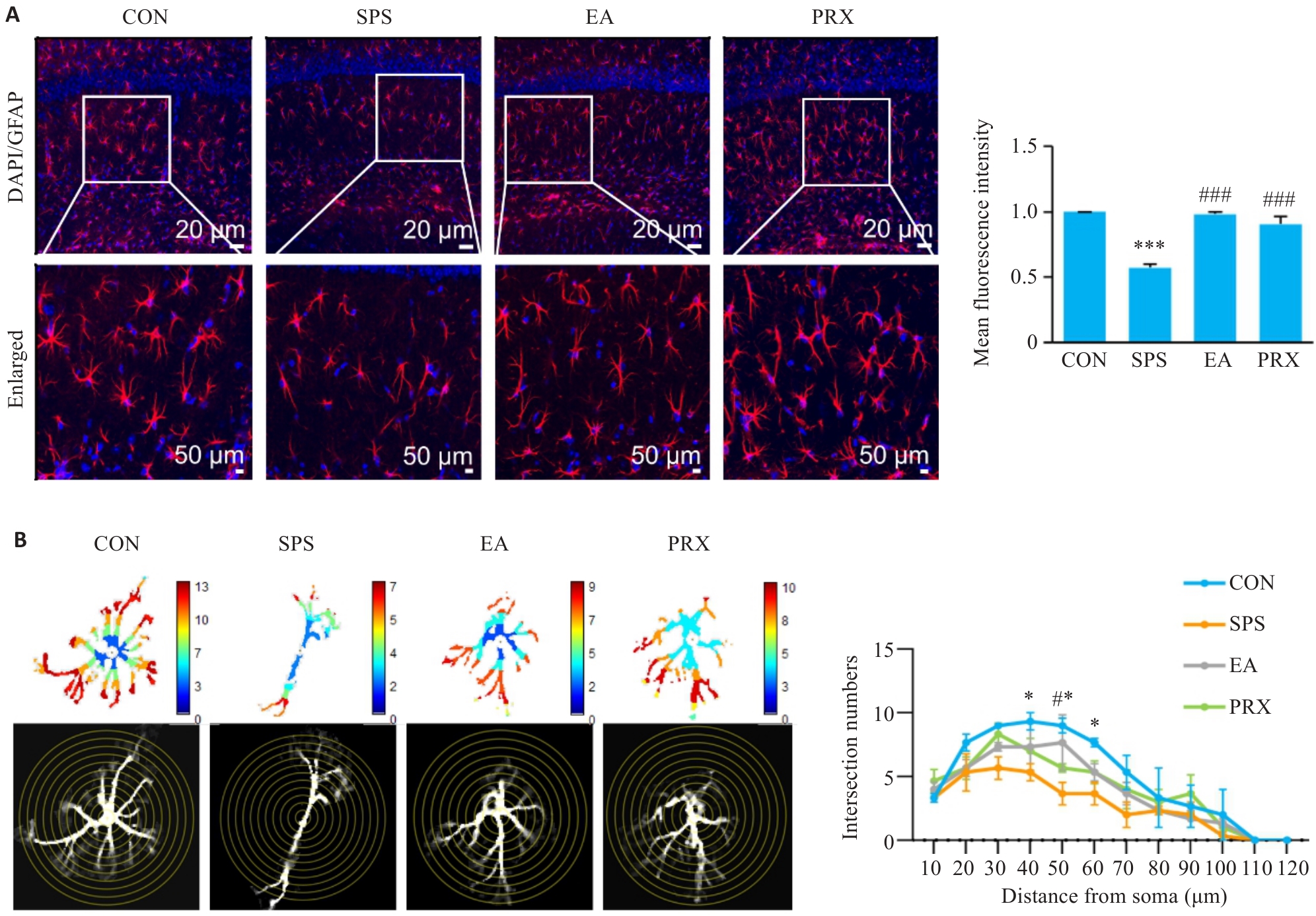
图3 电针能修复SPS小鼠海马GFAP下调和AST结构损伤
Fig.3 EA restores the down-regulation of GFAP and the structural damage of AST in the hippocampus of SPS mice (n=3). A: Immunofluorescence staining of GFAP (Nucleus: blue; AST: red; original magnification: ×200) and mean fluorescence intensity of GFAP in the hippocampus of the mice. B: Representative diagram and results of Sholl analysis. *P<0.05, ***P<0.001 vs CON group; #P<0.05, ###P<0.001 vs SPS group.
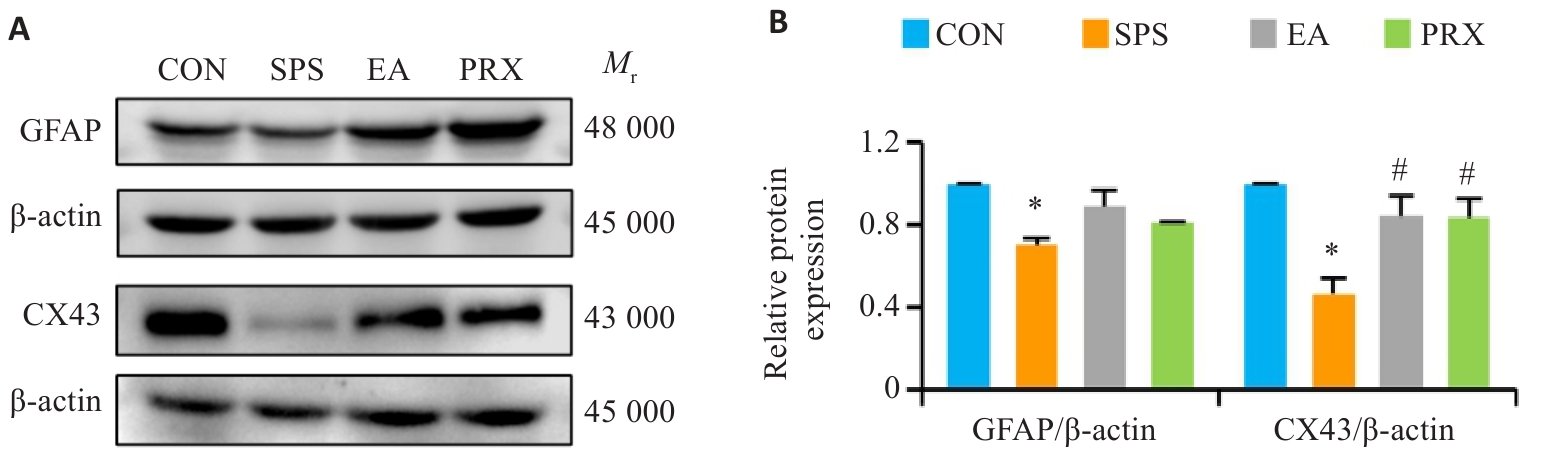
图4 电针能回调SPS小鼠海马GFAP和CX43蛋白的表达
Fig.4 EA restores the expression of GFAP and CX43 proteins in the hippocampus of SPS mice (n=3). A: Protein bands of GFAP and CX43 in Western blotting. B: Quantitative analysis of GFAP and CX43 expression levels. *P<0.05 vs CON group; #P<0.05 vs SPS group.
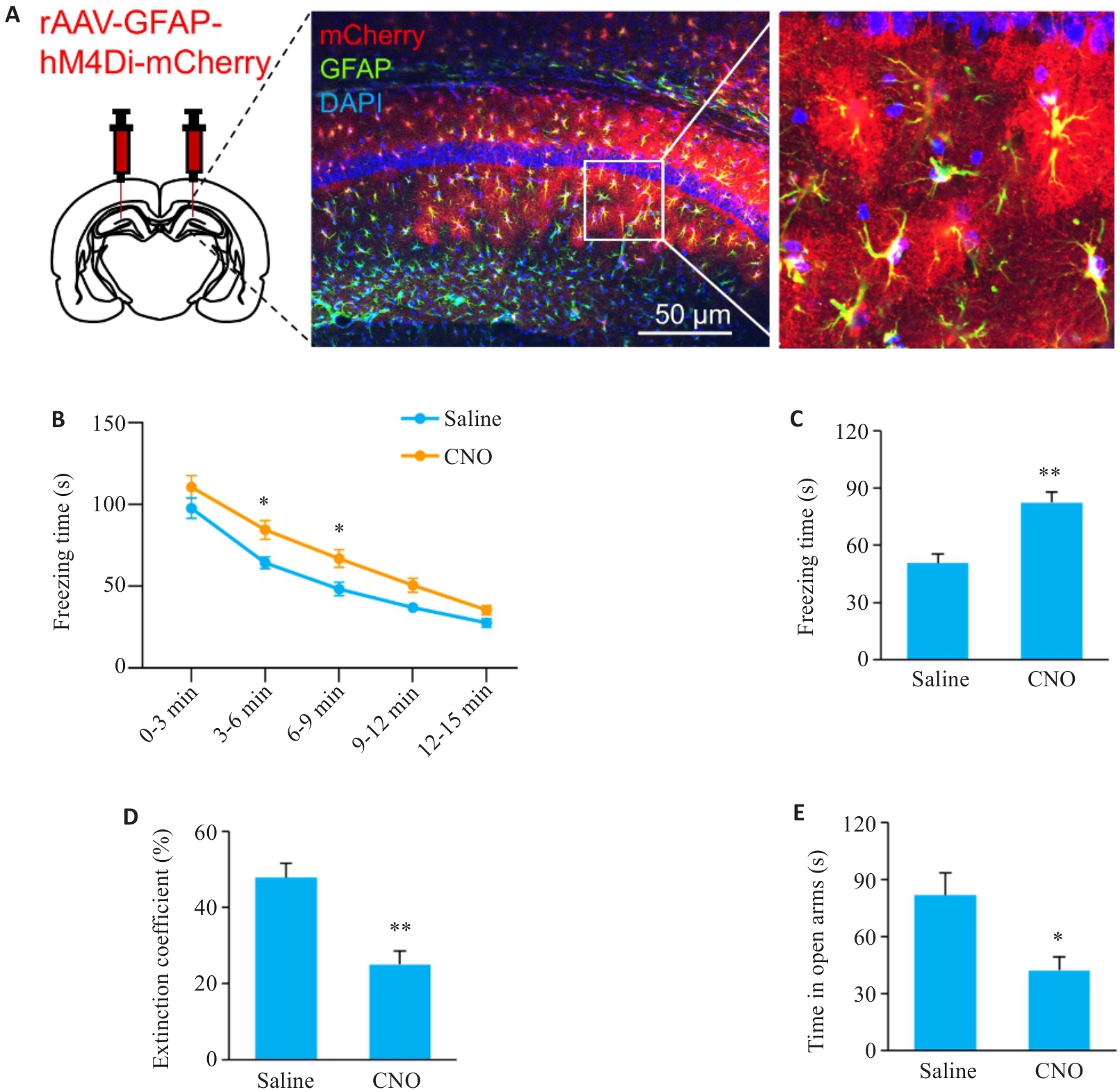
图5 抑制海马CA1区AST能阻断电针对SPS小鼠恐惧记忆消退障碍和焦虑样行为的改善作用
Fig. 5 Inhibition of astrocytes in hippocampal CA1 region of SPS mice blocks ameliorating effects of EA on abnormal fear memory extinction and anxiety-like behavior of SPS mice (n=6). A: Immunofluorescence staining for verifying virus injection (Nucleus: blue; AST: green; Virus: red; ×100). B: Freezing time of the mice during the re-exposure phase. C: Freezing time of mice during the fear extinction phase. D: Extinction coefficient of mice in FCT. E: Time in the open arms of the mice. *P<0.05, **P<0.01 vs Saline group.
| [1] | Shalev A, Cho D, Marmar CR. Neurobiology and treatment of posttraumatic stress disorder[J]. Am J Psychiatry, 2024, 181(8): 705-19. doi:10.1176/appi.ajp.20240536 |
| [2] | Schincariol A, Orrù G, Otgaar H, et al. Posttraumatic stress disorder (PTSD) prevalence: an umbrella review[J]. Psychol Med, 2024, 54(15): 4021-34. doi:10.1017/s0033291724002319 |
| [3] | Lewis C, Roberts NP, Gibson S, et al. Dropout from psychological therapies for post-traumatic stress disorder (PTSD) in adults: systematic review and meta-analysis[J]. Eur J Psychotraumatology, 2020, 11: 1709709. doi:10.1080/20008198.2019.1709709 |
| [4] | Tregub T, Lytvynenko M, Kukushkin V, et al. Pharmacology of posttraumatic stress disorder [J]. Georgian Med News, 2023, (342): 122-4. doi:10.1201/b22810-33 |
| [5] | Ben-Zion Z, Korem N, Spiller TR, et al. Longitudinal volumetric evaluation of hippocampus and amygdala subregions in recent trauma survivors[J]. Mol Psychiatry, 2023, 28(2): 657-67. doi:10.1038/s41380-022-01842-x |
| [6] | Fujikawa R, Ramsaran AI, Guskjolen A, et al. Neurogenesis-dependent remodeling of hippocampal circuits reduces PTSD-like behaviors in adult mice[J]. Mol Psychiatry, 2024, 29(11): 3316-29. doi:10.1038/s41380-024-02585-7 |
| [7] | Wang Y, Fu AKY, Ip NY. Instructive roles of astrocytes in hippocampal synaptic plasticity: neuronal activity-dependent regulatory mechanisms[J]. FEBS J, 2022, 289(8): 2202-18. doi:10.1111/febs.15878 |
| [8] | Bansal Y, Codeluppi SA, Banasr M. Astroglial dysfunctions in mood disorders and rodent stress models: consequences on behavior and potential as treatment target[J]. Int J Mol Sci, 2024, 25(12): 6357. doi:10.3390/ijms25126357 |
| [9] | Wang J, Cheng P, Qu Y, et al. Astrocytes and memory: implications for the treatment ofMemory-related disorders[J]. Curr Neuro-pharmacol, 2024, 22(13): 2217-39. doi:10.2174/1570159x22666240128102039 |
| [10] | Grant S, Colaiaco B, Motala A, et al. Acupuncture for the treatment of adults with posttraumatic stress disorder: a systematic review and meta-analysis[J]. J Trauma Dissociation, 2018, 19(1): 39-58. doi:10.1080/15299732.2017.1289493 |
| [11] | Hollifield M, Hsiao AF, Smith T, et al. Acupuncture for combat-related posttraumatic stress disorder: a randomized clinical trial[J]. JAMA Psychiatry, 2024, 81(6): 545. doi:10.1001/jamapsychiatry.2023.5651 |
| [12] | 王永蕊, 郑先丽, 严兴科. 针灸干预PTSD恐惧记忆的神经生物学机制研究进展 [J]. 世界科学技术-中医药现代化, 2023, 25(12): 4052-7. |
| [13] | Lin SS, Zhou B, Chen BJ, et al. Electroacupuncture prevents astrocyte atrophy to alleviate depression[J]. Cell Death Dis, 2023, 14(5): 343. doi:10.1038/s41419-023-05839-4 |
| [14] | Wang JW, Deng X, Jiang J, et al. Evaluation of electroacupuncture as a non-pharmacological therapy for astrocytic structural aberrations and behavioral deficits in a post-ischemic depression model in mice[J]. Front Behav Neurosci, 2023, 17: 1239024. doi:10.3389/fnbeh.2023.1239024 |
| [15] | Tang XR, Lin SM, Fang DW, et al. Efficacy and underlying mechanisms of acupuncture therapy for PTSD: evidence from animal and clinical studies[J]. Front Behav Neurosci, 2023, 17: 1163718. doi:10.3389/fnbeh.2023.1163718 |
| [16] | 陈潇煜, 刘 畅, 杜坤锐, 等. 电针对单次长时间应激小鼠恐惧记忆消退及睡眠时相的影响及机制研究 [J]. 中国针灸, 2024, 44(8): 923-30. |
| [17] | Verbitsky A, Dopfel D, Zhang NY. Rodent models of post-traumatic stress disorder: behavioral assessment[J]. Transl Psychiatry, 2020, 10: 132. doi:10.1038/s41398-020-0806-x |
| [18] | Wang J, Zhao PP, Cheng P, et al. Exploring the effect of Anshen Dingzhi prescription on hippocampal mitochondrial signals in single prolonged stress mouse model[J]. J Ethnopharmacol, 2024, 323: 117713. doi:10.1016/j.jep.2024.117713 |
| [19] | 中国针灸学会. 实验动物常用穴位名称与定位第3部分:小鼠 [J]. 针刺研究, 2021, 46(5): 445-6. |
| [20] | 朱秀玲, 王 薇, 宗永立, 等. 电针抑制PTSD对内侧额叶皮质BDNF的下调和GABAARa1的上调 [J]. 中国组织化学与细胞化学杂志, 2016, 25(1): 54-8. doi:10.16705/j.cnki.1004-1850.2016.01.010 |
| [21] | 姜 宁, 范琳犀, 杨玉洁,等. 金钗石斛提取物对慢性不可预见应激模型小鼠的抗抑郁作用 [J]. 生理学报, 2017, 69(2): 159-66. doi:10.13294/j.aps.2017.0006 |
| [22] | Lonsdorf TB, Merz CJ, Fullana MA. Fear extinction retention: is it what we think it is[J]? Biol Psychiatry, 2019, 85(12): 1074-82. doi:10.1016/j.biopsych.2019.02.011 |
| [23] | Codeluppi SA, Chatterjee D, Prevot TD, et al. Chronic stress alters astrocyte morphology in mouse prefrontal cortex[J]. Int J Neuropsychopharmacol, 2021, 24(10): 842-53. doi:10.1093/ijnp/pyab052 |
| [24] | 郑先丽, 严兴科, 姚小强, 等. 针刺治疗创伤后应激障碍的思路探讨 [J]. 中医研究, 2023, 36(12): 89-92. |
| [25] | 孙 英, 赵 琳, 朱 悦, 等. 基于关联规则及聚类分析探讨针灸治疗创伤后应激障碍选穴规律 [J]. 针灸临床杂志, 2023, 39(5): 50-6. |
| [26] | American Psychological Association Guideline Development Panel for the Treatment of PTSD in Adults). Summary of the clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults[J]. Am Psychol, 2019, 74(5): 596-607. doi:10.1037/amp0000473 |
| [27] | Bentefour Y, Rakibi Y, Bennis M, et al. Paroxetine treatment, following behavioral suppression of PTSD-like symptoms in mice, prevents relapse by activating the infralimbic cortex[J]. Eur Neuropsychopharmacol, 2016, 26(2): 195-207. doi:10.1016/j.euroneuro.2015.12.021 |
| [28] | Gao F, Wang J, Yang SJ, et al. Fear extinction induced by activation of PKA ameliorates anxiety-like behavior in PTSD mice[J]. Neuropharmacology, 2023, 222: 109306. doi:10.1016/j.neuropharm.2022.109306 |
| [29] | Roy DS, Kitamura T, Okuyama T, et al. Distinct neural circuits for the formation and retrieval of episodic memories[J]. Cell, 2017, 170(5): 1000-12.e19. doi:10.1016/j.cell.2017.07.013 |
| [30] | Kim YR, Lee M, Kim MS. Astrocytes in fear memory processing: molecular mechanisms across the amygdala-hippocampus-prefrontal cortex network[J]. Cells, 2025, 14(18): 1444. doi:10.3390/cells14181444 |
| [31] | WANG J, CHEN X, CHEN D, et al. Peroxisome Proliferator‑ activated receptor gamma coactivator‑1α deficiency in hippocampal astrocytes underlies enhanced fear memory retrieval in male posttraumatic stress disorder model mice[J]. MedComm, 2026, 7(3): e70671. doi:10.1002/mco2.70671 |
| [32] | Yang SJ, Qu Y, Wang J, et al. Anshen Dingzhi prescription in the treatment of PTSD in mice: Investigation of the underlying mechanism from the perspective of hippocampal synaptic function[J]. Phytomedicine, 2022, 101: 154139. doi:10.1016/j.phymed.2022.154139 |
| [33] | Xie P, Chen LX, Wang J, et al. Polysaccharides from Polygonatum cyrtonema Hua prevent post-traumatic stress disorder behaviors in mice: Mechanisms from the perspective of synaptic injury, oxidative stress, and neuroinflammation[J]. J Ethnopharmacol, 2024, 319: 117165. doi:10.1016/j.jep.2023.117165 |
| [34] | Zheng XX, Yang JY, Hou YW, et al. Prediction of clinical progression in nervous system diseases: plasma glial fibrillary acidic protein (GFAP)[J]. Eur J Med Res, 2024, 29(1): 51. doi:10.1186/s40001-023-01631-4 |
| [35] | Dong R, Han YQ, Jiang LH, et al. Connexin 43 gap junction-mediated astrocytic network reconstruction attenuates isoflurane-induced cognitive dysfunction in mice[J]. J Neuroinflammation, 2022, 19(1): 64. doi:10.1186/s12974-022-02424-y |
| [36] | Wu X, Li LJ, Zhou BL, et al. Connexin 43 regulates astrocyte dysfunction and cognitive deficits in early life stress-treated mice[J]. Exp Brain Res, 2023, 241(4): 1207-14. doi:10.1007/s00221-023-06587-9 |
| [37] | Wu CR, Li Y, He XR, et al. Chemogenetic activation of astrocytic Gi signaling promotes spinogenesis and motor functional recovery after stroke[J]. Glia, 2024, 72(6): 1150-64. doi:10.1002/glia.24521 |
| [38] | Lv XJ, Lv SS, Wang GH, et al. Glia-derived adenosine in the ventral hippocampus drives pain-related anxiodepression in a mouse model resembling trigeminal neuralgia[J]. Brain Behav Immun, 2024, 117: 224-41. doi:10.1016/j.bbi.2024.01.012 |
| [39] | Lin WT, Yu LY, Xu H, et al. Neuroinflammation-informed neuroimaging-transcriptomic signatures explaining acupuncture's therapeutic effects in chronic insomnia[J]. Chin Med, 2025, 20(1): 207. doi:10.1186/s13020-025-01236-5 |
| [40] | Cong L, Ding SK, Guo Y, et al. Acupuncture alleviates CSDS-induced depressive-like behaviors by modulating synaptic plasticity in vCA1[J]. Theranostics, 2025, 15(10): 4808-22. doi:10.7150/thno.106751 |
| [1] | 尹钦, 雷雨逍, 王晓澍, 杨刚, 唐兆华. 黄芪总苷通过抑制P38通路缓解氧糖剥夺/复氧后的星形胶质细胞水肿并降低AQP4表达[J]. 南方医科大学学报, 2026, 46(2): 286-292. |
| [2] | 张淑芬, 黄添容, 杨灿洪, 陈家镒, 吕田明, 张嘉发. 莱菔硫烷通过抑制Aβ42寡聚体激活的U87细胞中MAPK/NF-κB信号通路降低反应性星形胶质细胞介导的SH-SY5Y凋亡[J]. 南方医科大学学报, 2026, 46(1): 191-199. |
| [3] | 王堃, 左海燕, 张娇娇, 吴欣, 王文慧, 吴生兵, 周美启. 电针通过调控海马谷氨酸释放抑制HPA轴亢进从而改善急性心肌缺血大鼠的心肌损伤[J]. 南方医科大学学报, 2025, 45(8): 1599-1607. |
| [4] | 邓楚玉, 王雪莹, 甘立祥, 王大禹, 郑晓燕, 唐纯志. 电针足三里改善高脂血症小鼠的血脂紊乱:基于肠道菌群结构的改善[J]. 南方医科大学学报, 2025, 45(8): 1633-1642. |
| [5] | 唐东宁, 康赟赟, 何文杰, 夏青. 针康结合促进C57/BL6J小鼠脑缺血后星形胶质细胞转分化为神经元[J]. 南方医科大学学报, 2025, 45(7): 1434-1441. |
| [6] | 张安邦, 孙秀颀, 庞博, 吴远华, 时靖宇, 张宁, 叶涛. 电针预处理通过调节肠道-大脑轴及Nrf2/HO-1信号通路抑制铁死亡减轻大鼠脑缺血再灌注损伤[J]. 南方医科大学学报, 2025, 45(5): 911-920. |
| [7] | 郭舒婷, 曹福羊, 郭永馨, 李言响, 郝新宇, 张倬宁, 周志康, 仝黎, 曹江北. 激活下丘脑背内侧核区星形胶质细胞可加速七氟醚麻醉小鼠觉醒[J]. 南方医科大学学报, 2025, 45(4): 751-759. |
| [8] | 何存宝, 杨绍杰, 朱国旗. 4-(芳基乙炔基)-吡咯并[2,3-d]嘧啶通过抑制mGluR5调控ERK1/2-SGK1信号通路改善小鼠创伤后应激障碍[J]. 南方医科大学学报, 2025, 45(4): 765-773. |
| [9] | 黄鹏伟, 陈洁, 邹金虎, 高雪锋, 曹虹. 槲皮素促进应激颗粒G3BP1解聚改善HIV-1 gp120诱导的星形胶质细胞神经毒性[J]. 南方医科大学学报, 2025, 45(2): 304-312. |
| [10] | 马丹丹, 程洁, 张虹, 刘广, 宋凯. 电针通过Bcl-2/Bax/caspase-3信号通路修复海马线粒体损伤改善创伤后应激障碍大鼠的焦虑症状[J]. 南方医科大学学报, 2025, 45(11): 2375-2384. |
| [11] | 袁萍, 胡秀莉, 漆国佳, 代秀, 褚相远, 陈卫航, 石修权. 睡眠质量低下与创伤患者创伤后应激障碍的发生相关[J]. 南方医科大学学报, 2024, 44(6): 1166-1172. |
| [12] | 陈洁, 刘晨旭, 王春, 李丽, 陶伟婷, 徐婧茹, 唐红辉, 黄丽. 外源性瘦素通过上调星形胶质细胞GLT-1和GLAST的表达减轻小鼠脑缺血再灌注引起的谷氨酸兴奋毒性损伤[J]. 南方医科大学学报, 2024, 44(6): 1079-1087. |
| [13] | 满 豪, 王建伟, 吴 毛, 邵 阳, 杨俊锋, 李绍烁, 吕锦业, 周 悦. 脊髓康通过激活星形胶质细胞的YAP/PKM2信号轴促进脊髓损伤大鼠神经功能的恢复[J]. 南方医科大学学报, 2024, 44(4): 636-643. |
| [14] | 李言响, 郭永馨, 曹福羊, 郭舒婷, 薛丁豪, 周志康, 郝新宇, 仝 黎, 傅 强. 抑制dmPAG区谷氨酸能神经元可减轻创伤后应激障碍小鼠的过度防御反应[J]. 南方医科大学学报, 2024, 44(3): 420-427. |
| [15] | 林如辉, 夏金言, 马小涵, 李钻芳. 电针通过促进突触再生改善脑缺血再灌注损伤大鼠的学习记忆功能[J]. 南方医科大学学报, 2024, 44(12): 2317-2326. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||