南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 646-654.doi: 10.12122/j.issn.1673-4254.2026.03.18
• 基础研究 • 上一篇
冯鹏1,2( ), 王洁1, 褚江伟1, 王鹤霖1, 田永衍1,2, 张杰昌1, 王明露1, 王宇彤1
), 王洁1, 褚江伟1, 王鹤霖1, 田永衍1,2, 张杰昌1, 王明露1, 王宇彤1
收稿日期:2025-08-05
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
冯鹏
E-mail:sanzhichan@163.com
作者简介:冯 鹏,副教授,E-mail: sanzhichan@163.com
基金资助:
Peng FENG1,2( ), Jie WANG1, Jiangwei CHU1, helin WANG1, Yongyan TIAN1,2, Jiechang ZHANG1, Minglu WANG1, Yutong WANG1
), Jie WANG1, Jiangwei CHU1, helin WANG1, Yongyan TIAN1,2, Jiechang ZHANG1, Minglu WANG1, Yutong WANG1
Received:2025-08-05
Online:2026-03-20
Published:2026-03-26
Contact:
Peng FENG
E-mail:sanzhichan@163.com
摘要:
目的 探讨石菖蒲挥发油(VOA)通过TLR4/MyD88/NF-κB通路调控小胶质细胞(MG)抗抽动的机制。 方法 48只3周龄SD大鼠,随机分为空白组(n=8)和模型制备组(n=40),采用腹腔注射亚氨基二丙腈诱导TD模型,将模型制备组大鼠随机分为模型组、硫必利组(47.91 mg/kg)、VOA组(51.12 mg/kg)、TAK-242组(3 mg/kg)和VOA+TAK242组(51.12 mg/kg+3 mg/kg),每组8只,1次/d,干预28 d。干预结束后,评估大鼠行为学变化情况,采用尼氏染色观察纹状体神经元形态改变,ELISA法检测大鼠血清、纹状体中TNF-α、C1q、TGF-β、VEGF含量,RT-PCR、Western blotting法检测观察大鼠纹状体中TLR4、MyD88、NF-κB p65的mRNA和蛋白的表达情况,免疫细胞荧光染色检测NF-κB p65入核情况、MG特异性标志物Iba1、M1型、M2型标记蛋白CD86、CD206的表达水平。 结果 与模型组相比较,VOA组大鼠抽动样行为评分降低(P<0.01),纹状体神经元损伤改善;血清及纹状体中TNF-α、C1q水平降低(P<0.01)、TGF-β、VEGF水平升高(P<0.01);纹状体中TLR4、MyD88、NF-κB p65 mRNA和蛋白相对表达量水平降低(P<0.01),NF-κB p65 入核减少(P<0.05),Iba1、CD86相对荧光强度减弱(P<0.01),Iba1、CD206相对荧光强度增强(P<0.01)。 结论 VOA通过降低MG的活化及极化状态,进而抑制脑内炎症水平改善抽动症状,其作用机制可能与抑制TLR4/MyD88/NF-κB通路有关。
冯鹏, 王洁, 褚江伟, 王鹤霖, 田永衍, 张杰昌, 王明露, 王宇彤. 石菖蒲挥发油通过抑制TLR4/MyD88/NF-κB通路介导的小胶质细胞极化改善大鼠抽动障碍[J]. 南方医科大学学报, 2026, 46(3): 646-654.
Peng FENG, Jie WANG, Jiangwei CHU, helin WANG, Yongyan TIAN, Jiechang ZHANG, Minglu WANG, Yutong WANG. Volatile oil from Acorus tatarinowii Schott ameliorates tic disorder in rats by inhibiting TLR4/MyD88/NF-κB-mediated microglial polarization[J]. Journal of Southern Medical University, 2026, 46(3): 646-654.
| Degree rating | Stereotypic behavior | Motor behavior |
|---|---|---|
| 0 | No stereotyped behavior | Quiet or normal activity |
| 1 | Rotation behavior (clockwise or counterclockwise rotation motion) | Be overexcited |
| 2 | Excessive head and neck movement | Exploring behavior increases |
| 3 | Excessive vertical movement of the head and neck combined with rotational behavior | Keep running |
| 4 | Head to side swing, combined with excessive vertical movement of the head and neck | Constantly jumping and startled jumping |
表1 行为学评分表
Tab.1 Behavioral assessment form
| Degree rating | Stereotypic behavior | Motor behavior |
|---|---|---|
| 0 | No stereotyped behavior | Quiet or normal activity |
| 1 | Rotation behavior (clockwise or counterclockwise rotation motion) | Be overexcited |
| 2 | Excessive head and neck movement | Exploring behavior increases |
| 3 | Excessive vertical movement of the head and neck combined with rotational behavior | Keep running |
| 4 | Head to side swing, combined with excessive vertical movement of the head and neck | Constantly jumping and startled jumping |
| Primer | Forward | Reverse | Extension length (bp) |
|---|---|---|---|
| TLR4 | TATCGGTGGTCAGTGTGCTT | CTCGTTTCTCACCCAGTCCT | 167 |
| MyD88 | CCTGGGCACACATCTCAGTT | CTGGGGGCGGAATGTTTTTG | 220 |
| P65 | TGGATGAGTAATGCGTCCAGG | GGACTGGCGGTGGAGAATAA | 91 |
| GAPDH | TGATGGGTGTGAACCACGAG | AGTGATGGCATGGACTGTGG | 152 |
表2 PCR引物序列
Tab.2 Primer sequences for RT-PCR
| Primer | Forward | Reverse | Extension length (bp) |
|---|---|---|---|
| TLR4 | TATCGGTGGTCAGTGTGCTT | CTCGTTTCTCACCCAGTCCT | 167 |
| MyD88 | CCTGGGCACACATCTCAGTT | CTGGGGGCGGAATGTTTTTG | 220 |
| P65 | TGGATGAGTAATGCGTCCAGG | GGACTGGCGGTGGAGAATAA | 91 |
| GAPDH | TGATGGGTGTGAACCACGAG | AGTGATGGCATGGACTGTGG | 152 |
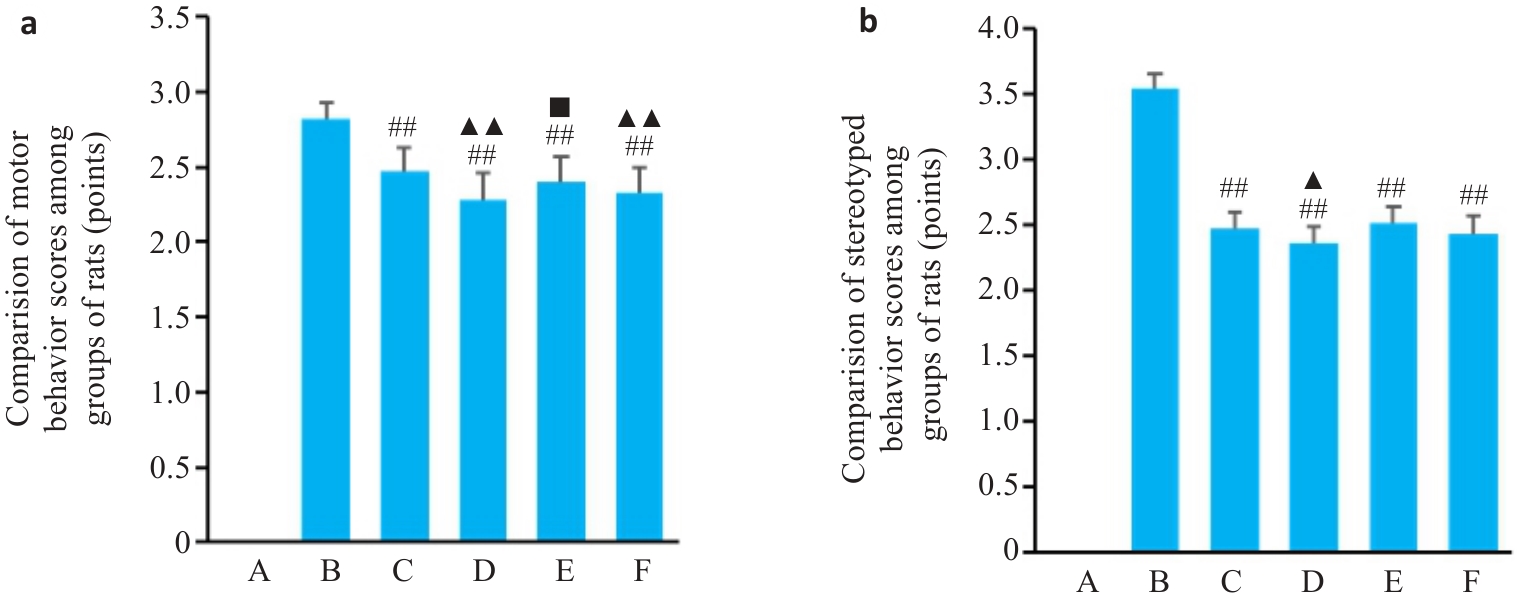
图1 第28天各组大鼠抽动行为评分
Fig1 Tic behavior scores of the rats in each group on day 28 (Mean±SD, n=8). a: motor behavior scores on day 28. b: Stereotyped behavior scores on day 28. A: Blank group; B: Model group; C: Tiapride group; D: VOA group; E: TAK-242 group; F: VOA+TAK-242 group. ##P<0.01 vs B group, ▲P<0.05, ▲▲P<0.01 vs C group, ■P<0.05 vs D group.
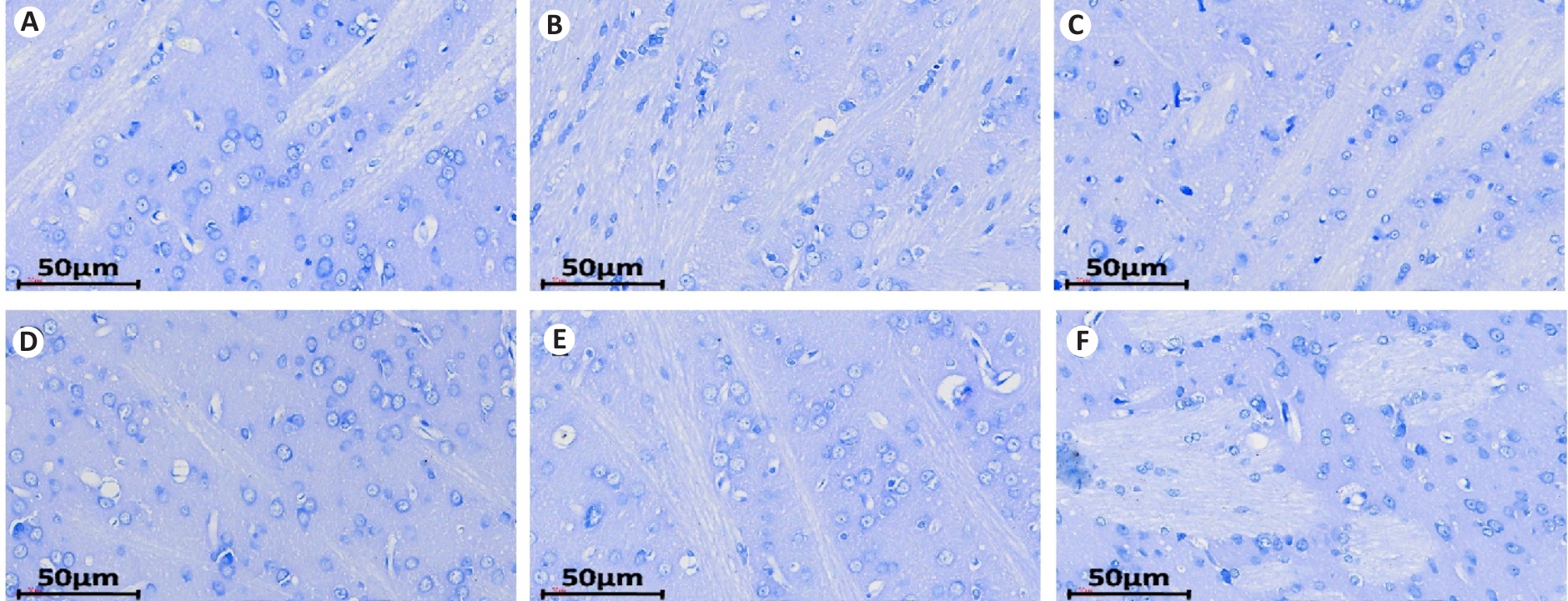
图2 各组大鼠纹状体尼氏染色
Fig.2 Nissl staining of the striatum of the rats in each group (Original magnification: ×200). A: Blank group; B: Model group; C: Tiapride group; D: VOA group; E: TAK-242 group; F: VOA+TAK-242 group.
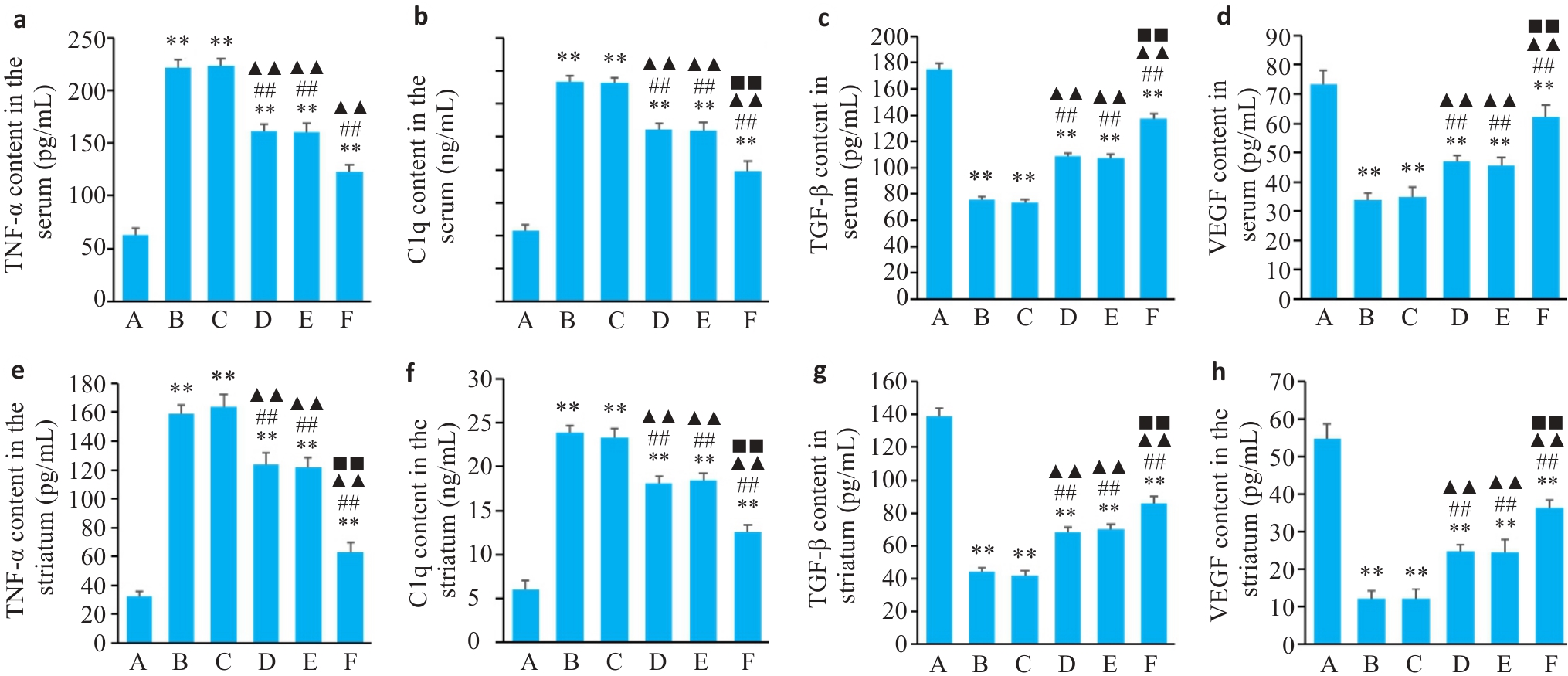
图3 各组血清、纹状体中TGF-β、VEGF、TNF-α、C1q含量水平
Fig.3 Levels of TNF-α (a, e), C1q (b, f), TGF-β (c, g) and VEGF (d, h) in the serum and striatum of the rats in different groups (Mean±SD, n=3). A: Blank group; B: Model group; C: Tiapride group; D: VOA group; E: TAK-242 group; F: VOA+TAK-242 group. **P<0.01 vs A group; ##P<0.01 vs B group; ▲▲P<0.01 vs C group; ■■P<0.01 vs D group.
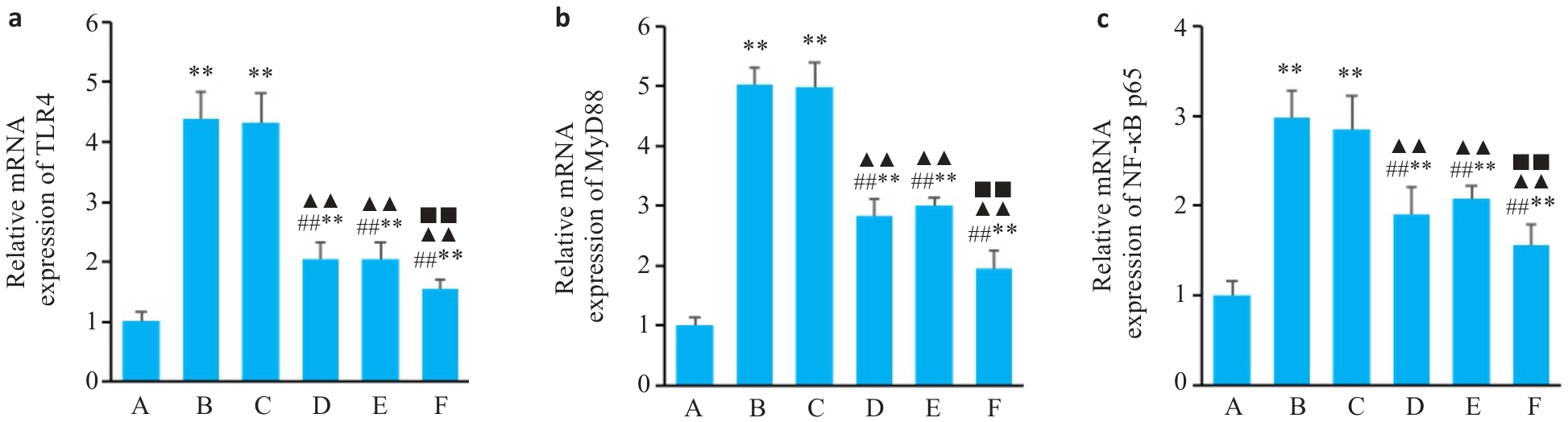
图4 各组大鼠纹状体中TLR4、MyD88、NF-κB mRNA相对表达
Fig.4 Relative expressions of TLR4 (a), NF-κB p65 (b) and MyD88 (c) mRNA in the striatum of the rats in each group. A: Blank group; B: Model group; C: Tiapride group; D: VOA group; E: TAK-242 group; F: VOA+TAK-242 group. n=3.**P<0.01 vs A group; ##P<0.01 vs B group; ▲▲P<0.01 vs C group; ■■P<0.01 vs D group.
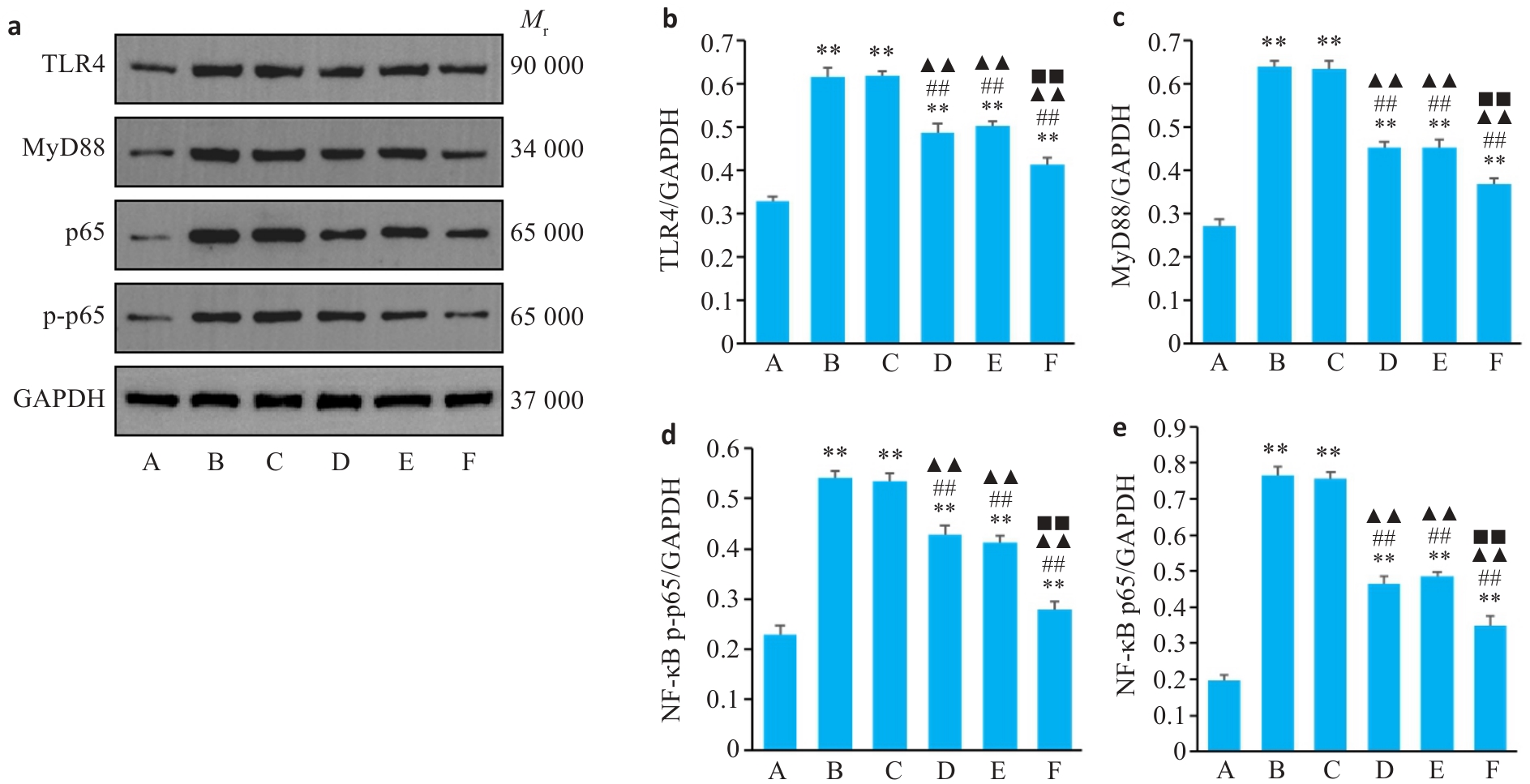
图5 Western blotting 检测各组大鼠纹状体中TLR4、MyD88、NF-κB p65、p-NF-κB p65蛋白相对表达
Fig.5 Relative expression of TLR4, MyD88, NF-κB p65 and p-NF-κB p65 proteins in the striatum of the rats in each group detected by Western blotting. a: Results of Western blotting of the proteins. b-e: Relative expressions of TLR4, MyD88, NF-κB p65 and p-NF-κB p65 proteins. A: Blank group; B: Model group; C: Tiapride group; D: VOA group; E: TAK-242 group; F: VOA+TAK-242 group. n=3. **P<0.01 vs A group; ##P<0.01 vs B group; ▲▲P<0.01 vs C group; ■■P<0.01 vs D group.
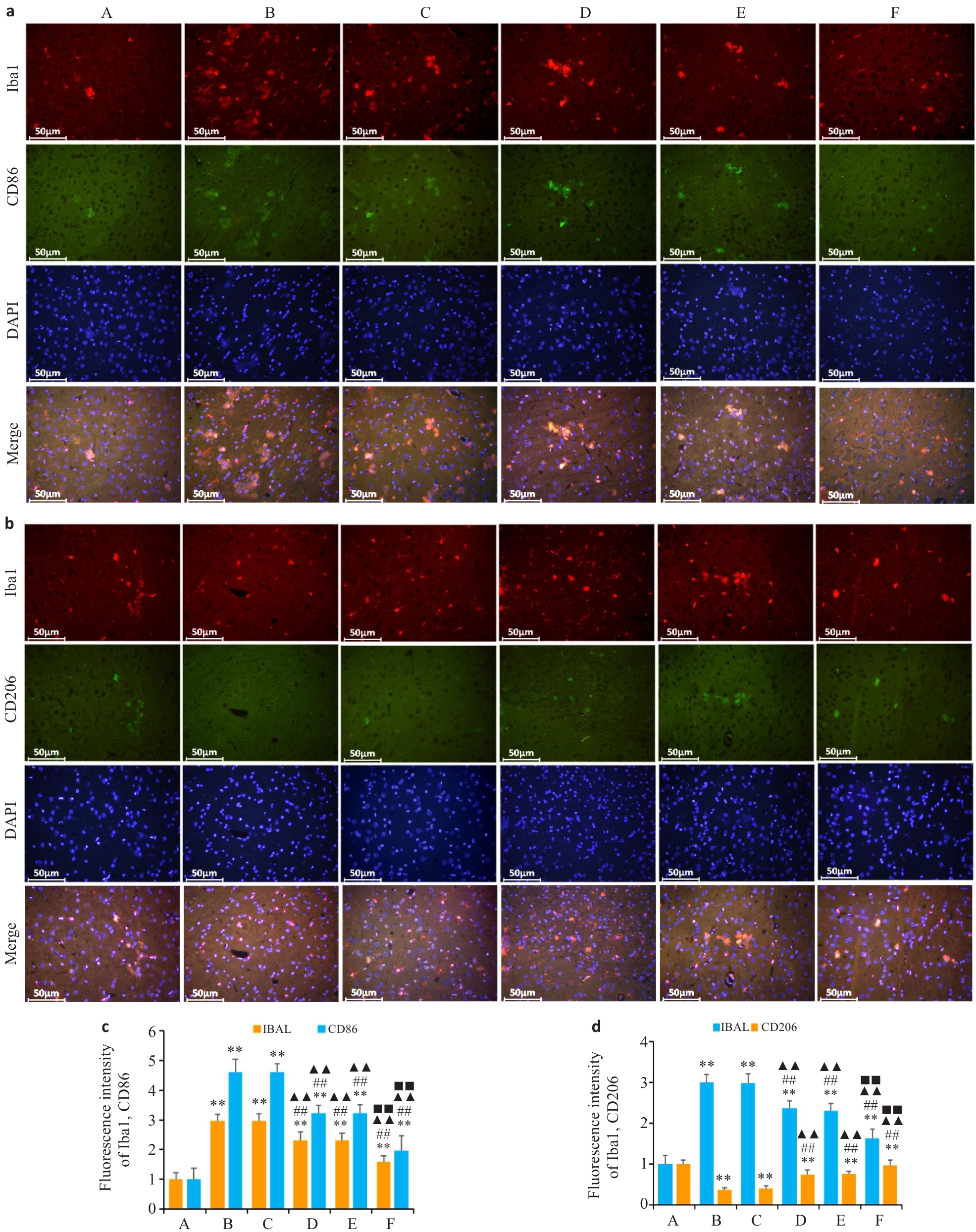
图7 各组大鼠纹状中MG活化及极化表达情况
Fig.7 Expression of microglial activation and polarization markers in the striatum in each group. a: Immuneofluorescence staining for Iba-1 (red), CD86 (green) and DAPI (blue). b: Immuneofluorescence staining for Iba-1 (red), CD206 (green) and DAPI (blue). c, d: Fluorescence intensity of Iba-1 and CD86. d: Fluorescence intensity of Iba-1 and CD206. n=3. **P<0.01 vs A group,##P<0.01 vs B group, ▲▲P<0.01 vs C group, ■■P<0.01 vs D group.
| [1] | Halvorsen MW. Tourette syndrome and chronic tic disorders[J]. Psychiatr Clin North Am, 2025, 48(2): 281-92. doi:10.1016/j.psc.2025.01.006 |
| [2] | Bai L, Jin M, Zhang Q, et al. Progress in research on nutrition, neuroinflammation and dopaminergic alterations in Tic disorders[J]. Front Pediatr, 2025, 13: 1526117. doi:10.3389/fped.2025.1526117 |
| [3] | Wu XN, Hao JJ, Jiang KY, et al. Neuroinflammation and pathways that contribute to Tourette syndrome[J]. Ital J Pediatr, 2025, 51(1): 63. doi:10.1186/s13052-025-01874-3 |
| [4] | Wang YF, Fasching L, Wu FN, et al. Interneuron loss and microglia activation by transcriptome analyses in the basal Ganglia of tourette disorder[J]. Biol Psychiatry, 2025, 98(3): 260-70. doi:10.1016/j.biopsych.2024.12.022 |
| [5] | Fang Q, Cai YH, Yang YT, et al. Curcumin attenuated neuroinflammation via the TLR4/MyD88/NF‑κB signaling way in the juvenile rat hippocampus following kainic acid-induced epileptic seizures[J]. Metab Brain Dis, 2024, 39(7): 1387-403. doi:10.1007/s11011-024-01401-z |
| [6] | 栾文燕. 菖蒲郁金汤加减治疗痰火扰神型儿童抽动障碍的临床观察[D]. 兰州: 甘肃中医药大学, 2024. |
| [7] | Chen L, Li JX, Zhang WH, et al. Antiepileptic effects of Acorus tatarinowii schott in a rat model of epilepsy: regulation of metabolic axes and gut microbiota[J]. Biology (Basel), 2025, 14(5): 488. doi:10.3390/biology14050488 |
| [8] | Huang JH, Xu ZW, Yu CS, et al. The volatile oil of Acorus tatarinowii Schott ameliorates Alzheimer's disease through improving insulin resistance via activating the PI3K/AKT pathway[J]. Phytomedicine, 2024, 135: 156168. doi:10.1016/j.phymed.2024.156168 |
| [9] | Feng P, Chen YH, Sun KX, et al. Volatile oil from Acori graminei Rhizoma affected the synaptic plasticity of rats with tic disorders by modulating dopaminergic and glutamatergic systems[J]. J Ethnopharmacol, 2024, 335: 118676. doi:10.1016/j.jep.2024.118676 |
| [10] | 丁艳琴, 冯 鹏, 王明露, 等. 石菖蒲挥发油对抽动障碍大鼠CX3CL1/CX3CR1信号轴及神经炎症的影响[J]. 中成药, 2025, 47(6): 1825-33. |
| [11] | 中华人民共和国药典:四部 [S]. 2020: 233. |
| [12] | Al Kadasah S, Al Mutairy A, Siddiquei M, et al. Pentoxifylline attenuates iminodipropionitrile-induced behavioral abnormalities in rats[J]. Behav Pharmacol, 2009, 20(4): 356-60. doi:10.1097/fbp.0b013e32832ec5ea |
| [13] | Dong JB, Liao Y, Wu BH. TAK-242 ameliorates epileptic symptoms in mice by inhibiting the TLR4/NF‑κB signaling pathway[J]. Ann Transl Med, 2022, 10(14): 795. doi:10.21037/atm-22-2707 |
| [14] | Johnson KA, Worbe Y, Foote KD, et al. Tourette syndrome: clinical features, pathophysiology, and treatment[J]. Lancet Neurol, 2023, 22(2): 147-58. doi:10.1016/s1474-4422(22)00303-9 |
| [15] | Chi S, Mok YE, Kang JE, et al. Cytokine levels reflect tic symptoms more prominently during mild phases[J]. BMC Neurosci, 2023, 24(1): 57. doi:10.1186/s12868-023-00830-3 |
| [16] | You HZ, Zhang J, Du YN, et al. Association of elevated plasma CCL5 levels with high risk for tic disorders in children[J]. Front Pediatr, 2023, 11: 1126839. doi:10.3389/fped.2023.1126839 |
| [17] | Wang YF, Fasching L, Wu FN, et al. Interneuron loss and microglia activation by transcriptome analyses in the basal Ganglia of tourette disorder[J]. Biol Psychiatry, 2025, 98(3): 260-70. doi:10.1016/j.biopsych.2024.12.022 |
| [18] | Takahashi N, Kato H, Nawa Y, et al. The role of inflammation in the development of tic symptoms in subjects with ADHD[J]. Brain Behav Immun Health, 2025, 45: 100981. doi:10.1016/j.bbih.2025.100981 |
| [19] | Tsetsos F, Yu DM, Sul JH, et al. Synaptic processes and immune-related pathways implicated in Tourette syndrome[J]. Transl Psychiatry, 2021, 11(1): 56. |
| [20] | Matuleviciute R, Akinluyi ET, TAOMuntslag, et al. Microglial contribution to the pathology of neurodevelopmental disorders in humans[J]. Acta Neuropathol, 2023, 146(5): 663-83. doi:10.1007/s00401-023-02629-2 |
| [21] | Lukens JR, Eyo UB. Microglia and neurodevelopmental disorders[J]. Annu Rev Neurosci, 2022, 45: 425-45. doi:10.1146/annurev-neuro-110920-023056 |
| [22] | Guo SR, Wang H, Yin YF. Microglia polarization from M1 to M2 in neurodegenerative diseases[J]. Front Aging Neurosci, 2022, 14: 815347. doi:10.3389/fnagi.2022.815347 |
| [23] | Wang XM, Liu XM, Chen LL, et al. The inflammatory injury in the striatal microglia-dopaminergic-neuron crosstalk involved in Tourette syndrome development[J]. Front Immunol, 2023, 14: 1178113. doi:10.3389/fimmu.2023.1178113 |
| [24] | Wang XM, Liu XM, Chen LL, et al. The inflammatory injury in the striatal microglia-dopaminergic-neuron crosstalk involved in Tourette syndrome development[J]. Front Immunol, 2023, 14: 1178113. doi:10.3389/fimmu.2023.1178113 |
| [25] | Sun JF, Lu LP, Lian YT, et al. Sodium butyrate attenuates microglia-mediated neuroinflammation by modulating the TLR4/MyD88/NF-κB pathway and microbiome-gut-brain axis in cardiac arrest mice[J]. Mol Brain, 2025, 18(1): 13. doi:10.1186/s13041-025-01179-w |
| [26] | Liu XM, Wang XM, Cao AH, et al. Immune function changes of the IDPN-induced Tourette syndrome rat model[J]. Int J Dev Neurosci, 2021, 81(2): 159-66. doi:10.1002/jdn.10085 |
| [27] | Tian MY, Huang JY, Xiao HR, et al. Characterization of 3, 3'-iminodipropionitrile (IDPN) damaged utricle transcriptome in the adult mouse utricle[J]. Front Mol Neurosci, 2024, 17: 1487364. doi:10.3389/fnmol.2024.1487364 |
| [28] | Huang JH, Xu ZW, Yu CS, et al. The volatile oil of Acorus tatarinowii Schott ameliorates Alzheimer's disease through improving insulin resistance via activating the PI3K/AKT pathway[J]. Phytomedicine, 2024, 135: 156168. doi:10.1016/j.phymed.2024.156168 |
| [29] | Chen L, Li JX, Zhang WH, et al. Antiepileptic effects of Acorus tatarinowii schott in a rat model of epilepsy: regulation of metabolic axes and gut microbiota[J]. Biology (Basel), 2025, 14(5): 488. doi:10.3390/biology14050488 |
| [30] | Liu XM, Wang XM, Cao AH, et al. Immune function changes of the IDPN-induced Tourette syndrome rat model[J]. Int J Dev Neurosci, 2021, 81(2): 159-66. doi:10.1002/jdn.10085 |
| [31] | Yin C, Zhang MY, Cheng L, et al. Melatonin modulates TLR4/MyD88/NF‑κB signaling pathway to ameliorate cognitive impairment in sleep-deprived rats[J]. Front Pharmacol, 2024, 15: 1430599. doi:10.3389/fphar.2024.1430599 |
| [32] | Borst K, Dumas AA, Prinz M. Microglia: immune and non-immune functions[J]. Immunity, 2021, 54(10): 2194-208. doi:10.1016/j.immuni.2021.09.014 |
| [33] | 桂建军, 孙晓东, 温 舒, 等. 白藜芦醇对帕金森病模型小鼠多巴胺能神经元的保护作用: 基于抑制TLR4/MyD88/NF-κB通路[J]. 南方医科大学学报, 2024, 44(2): 270-9. doi:10.12122/j.issn.1673-4254.2024.02.09 |
| [34] | 赵梦洁, 赵 琼, 杨翠玲, 等. 加味泻黄散对抽动障碍大鼠小胶质细胞极化及TLR4/MyD88/NF-κB通路的影响[J]. 中国实验方剂学杂志, 2025, 31(4): 10-8. |
| [1] | 夏金枝, 陈悦, 任侣, 李静, 宋雪, 陶露, 胡建国. 咖啡豆醇通过调控IκBα/NF-κB通路抑制小胶质细胞活化改善脊髓损伤后小鼠的运动功能[J]. 南方医科大学学报, 2025, 45(12): 2561-2572. |
| [2] | 孙一鸣, 徐昕冉, 卓雪瑞, 蔡慧, 王艳. C1q中和抗体可通过C1q/C3通路改善小鼠的产后抑郁样行为[J]. 南方医科大学学报, 2025, 45(10): 2111-2117. |
| [3] | 左涵珺, 段兆达, 王朝, 郭涛, 石金沙, 石浩龙, 李娟娟. 天麻素经PI3K/AKT通路改善新生大鼠缺氧缺血性脑损伤后小胶质细胞介导的炎症反应[J]. 南方医科大学学报, 2024, 44(9): 1712-1719. |
| [4] | 石金沙, 张皓南, 张幸霖, 石浩龙, 左涵珺, 郭涛, 王朝, 余航, 李娟娟. 天麻素通过调节CCR5/AKT信号传导缓解新生小鼠缺血缺氧后小胶质细胞介导的炎症反应[J]. 南方医科大学学报, 2024, 44(10): 1850-1857. |
| [5] | 孙晓鹏, 史 航, 张 磊, 刘 中, 李克威, 钱玲玲, 朱星宇, 杨康佳, 付 强, 丁 华. 外胚层间充质干细胞来源的外泌体通过控制炎症和氧化损伤减少M1型小胶质细胞并促进H2O2处理后PC12细胞的存活[J]. 南方医科大学学报, 2024, 44(1): 119-128. |
| [6] | 孙 洋, 许轶博, 肖林雨, 朱国庆, 李 静, 宋 雪, 徐 磊, 胡建国. 乙酰紫堇灵促进大鼠脊髓损伤后的功能恢复:基于调控EGFR/MAPK信号通路抑制小胶质细胞活化[J]. 南方医科大学学报, 2023, 43(6): 915-923. |
| [7] | 张嘉发, 杨灿洪, 张淑芬, 曹婷婷, 彭 瑞, 郭蔚泓, 严予苹, 谢淑婷, 彭晓佳, 吕田明, 黄添容. 莱菔硫烷通过下调MAPK/NF-κB信号通路逆转Aβ纤维介导的M1型小胶质细胞极化和神经炎症介导的神经干细胞程序性坏死[J]. 南方医科大学学报, 2023, 43(12): 2132-2138. |
| [8] | 陈丽旋, 黄定帮, 郑 刚, 孟晓静. 铅暴露加重Aβ1-42处理的小胶质细胞活化和小胶质细胞中的铜离子蓄积[J]. 南方医科大学学报, 2023, 43(10): 1752-1760. |
| [9] | 赵佳佳, 杨荷雨, 王招娣, 朱海丽, 谢 敏. ANA-12通过靶向阻断BDNF/TrkB信号通路降低大鼠的脊髓炎症和缓解病理性疼痛[J]. 南方医科大学学报, 2022, 42(2): 232-237. |
| [10] | 黄河灵, 高玉元, 聂 坤, 王丽娟. 巨噬细胞移动抑制因子介导MPP+/MPTP诱导的小胶质细胞NLRP3炎症小体的激活[J]. 南方医科大学学报, 2021, 41(7): 972-979. |
| [11] | 刘杨,蒋朴,徐颖. 脂多糖可致敏高浓度氧对新生小鼠未成熟脑的损伤[J]. 南方医科大学学报, 2014, 34(02): 214-. |
| [12] | 缪星宇,刘晓斌,岳青,秋楠,黄卫东,王继军,许彦钢,张越林,杨军,陈新林. 去铁胺对大鼠脑出血后小胶质细胞活化的抑制及其继发性神经损伤的保护作用[J]. 南方医科大学学报, 2012, 32(07): 970-. |
| [13] | 蒋朴,徐颖,胡良安,刘杨,邓世雄. 高浓度氧暴露对N9小胶质细胞功能的影响[J]. 南方医科大学学报, 2012, 32(01): 71-. |
| [14] | 颜广 ,刘伟 ,戴朝霞 ,王鹍 ,刘靖 ,赵灵凤 ,黄志斌 ,陈小辉 ,马宁 ,孟萍 ,许孟畅 ,温子龙 ,张文清. 一种斑马鱼原始造血髓系细胞突变体的研究[J]. 南方医科大学学报, 2011, 31(05): 755-. |
| [15] | 李锐; 彭宁; 杜芳; 李旭平; 乐卫东;. 表没食子儿茶素没食子酸酯的多巴胺能神经元保护作用(英文)[J]. 南方医科大学学报, 2006, 26(04): 376-380. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||