南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 513-522.doi: 10.12122/j.issn.1673-4254.2026.03.05
• 基础研究 • 上一篇
李彩霞1( ), 崔立华1, 曹婕2, 樊宇星2, 周雪颖3, 张淑坤1, 左艳洁3(
), 崔立华1, 曹婕2, 樊宇星2, 周雪颖3, 张淑坤1, 左艳洁3( )
)
收稿日期:2025-09-11
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
左艳洁
E-mail:licaixia2013 @163.com;zyjcz9886@163.com
作者简介:李彩霞,硕士,副研究员,硕士生导师,E-mail: licaixia2013 @163.com
基金资助:
Caixia LI1( ), Lihua CUI1, Jie CAO2, Yuxing FAN2, Xueying ZHOU3, Shukun ZHANG1, Yanjie ZUO3(
), Lihua CUI1, Jie CAO2, Yuxing FAN2, Xueying ZHOU3, Shukun ZHANG1, Yanjie ZUO3( )
)
Received:2025-09-11
Online:2026-03-20
Published:2026-03-26
Contact:
Yanjie ZUO
E-mail:licaixia2013 @163.com;zyjcz9886@163.com
摘要:
目的 探讨活血清解灵(HXQJL)通过调控沉默信息调节因子1(Sirt1)信号通路介导的自噬改善胆管结扎(BDL)诱导的肝纤维化。 方法 采用BDL处理SD大鼠,并随机分为:假手术组(Sham)、模型组(BDL)、活血清解灵低、高剂量组(HXQJL-L、HXQJL-H),Sirt1抑制剂Ex527组(EX527),活血清解灵联合Ex527组(HXQJL+EX527)。HE和天狼星红染色观察肝组织病变和胶原沉积变化;生化检测血清ALT、AST、ALP和GGT水平。RT-PCR 和Western blotting法分别检测α-SMA、FN、COL I、Atg 5、Beclin1、p62、LC3B和Sirt1 的表达。免疫荧光检测α-SMA、LC3B和Sirt1的表达。 结果 与Sham组比较,BDL组肝细胞损伤、炎细胞浸润及胶原沉积面积增多(P<0.05),血清中ALT、AST、ALP、GGT水平增加(P<0.05)。同时肝组织中α-SMA、FN、COL I、Atg 5和Beclin1表达增加, LC3B-II/I比值增加(P<0.05),而p62和Sirt1表达降低(P<0.05)。与BDL组相比,活血清解灵各剂量组肝细胞损伤、炎细胞浸润及胶原沉积面积减少(P<0.05),ALT、AST、TBA、ALP水平降低(P<0.05),肝组织中α-SMA、FN、COL I、Atg 5和Beclin1表达降低(P<0.05),LC3B-II/I比值降低,而p62和Sirt1表达增加(P<0.05);而Ex527组肝细胞损伤、炎细胞浸润及胶原沉积面积增多(P<0.05),ALT、AST、TBA、ALP水平升高(P<0.05),肝组织中α-SMA、FN、COL I、Atg 5和Beclin1表达升高,LC3B-II/I比值升高(P<0.05),而p62和Sirt1表达降低(P<0.05)。与HXQJL-H组相比, Ex527可以阻断活血清解灵对大鼠肝组织损伤、肝功能、肝纤维化和自噬的作用。 结论 活血清解灵通过调控Sirt1-自噬信号通路对胆管结扎诱导肝纤维化具有保护作用。
李彩霞, 崔立华, 曹婕, 樊宇星, 周雪颖, 张淑坤, 左艳洁. 活血清解灵通过调控Sirt1-自噬信号通路减轻胆管结扎诱导的大鼠肝纤维化[J]. 南方医科大学学报, 2026, 46(3): 513-522.
Caixia LI, Lihua CUI, Jie CAO, Yuxing FAN, Xueying ZHOU, Shukun ZHANG, Yanjie ZUO. Huoxue Qingjie Ling alleviates bile duct ligation-induced hepatic fibrosis in rats by regulating the Sirt1-autophagy signaling pathway[J]. Journal of Southern Medical University, 2026, 46(3): 513-522.
| Gene | Primer sequences (5'-3') (Forward) | Primer sequences (5'-3') (Reverse) |
|---|---|---|
| FN | GATTCTTCTGGCGTCTGCAC | GATTCTTCTGGCGTCTGCAC |
| COL I | GGATAGGGACTTGTGTGA | GCTGGAAGAGTGAAGAGG |
| α-SMA | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| Beclin-1 | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| Atg5 | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| LC3B | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| P62 | ACGACTGGACGCATTTGTCT | TGGTGGGAGATGTGGGATCA |
| Sirt 1 | ACCGAGACAACCTCCTGTTG | ATGGACCTATCCGTGGCCTT |
| GAPDH | AGATGGGTGAAGGTCGGTGTG | CTGGAAGATGGTGATGGGTT |
表1 引物序列
Tab.1 Primer sequences for qRT-PCR
| Gene | Primer sequences (5'-3') (Forward) | Primer sequences (5'-3') (Reverse) |
|---|---|---|
| FN | GATTCTTCTGGCGTCTGCAC | GATTCTTCTGGCGTCTGCAC |
| COL I | GGATAGGGACTTGTGTGA | GCTGGAAGAGTGAAGAGG |
| α-SMA | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| Beclin-1 | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| Atg5 | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| LC3B | AGGGAGTGATGGTTGGAATG | GATGATGCCGTGTTCTATCG |
| P62 | ACGACTGGACGCATTTGTCT | TGGTGGGAGATGTGGGATCA |
| Sirt 1 | ACCGAGACAACCTCCTGTTG | ATGGACCTATCCGTGGCCTT |
| GAPDH | AGATGGGTGAAGGTCGGTGTG | CTGGAAGATGGTGATGGGTT |
| Group | Dose (g/kg) | Body mass (g) | Liver mass (g) | Liver index (%) |
|---|---|---|---|---|
| Sham | 296.0±6.6 | 11.4±1.0 | 3.8±0.3 | |
| BDL | 198.1±11.81) | 17.9±1.81) | 9.0±0.81) | |
| HXQJL-L | 7.74 | 235.6±11.22) | 14.9±0.82) | 6.3±0.42) |
| HXQJL-H | 15.48 | 258.2±10.92)3) | 12.4±1.32)3) | 4.8±0.62) |
| EX527 | 0.005 | 163.6±8.82) | 21.4±1.02) | 13.1±1.02) |
| EX527+HXQJL | 0.005+15.48 | 204.0±13.04) | 17.5±2.14) | 8.6±0.94) |
表2 活血清解灵对BDL诱导肝纤维化大鼠体质量、肝脏质量和肝脏指数的影响
Tab.2 Effect of Huoxue Qingjie Ling (HXQJL) on body weight, liver mass and liver index in rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Body mass (g) | Liver mass (g) | Liver index (%) |
|---|---|---|---|---|
| Sham | 296.0±6.6 | 11.4±1.0 | 3.8±0.3 | |
| BDL | 198.1±11.81) | 17.9±1.81) | 9.0±0.81) | |
| HXQJL-L | 7.74 | 235.6±11.22) | 14.9±0.82) | 6.3±0.42) |
| HXQJL-H | 15.48 | 258.2±10.92)3) | 12.4±1.32)3) | 4.8±0.62) |
| EX527 | 0.005 | 163.6±8.82) | 21.4±1.02) | 13.1±1.02) |
| EX527+HXQJL | 0.005+15.48 | 204.0±13.04) | 17.5±2.14) | 8.6±0.94) |
| Group | Dose (g/kg) | ALT (U/L) | AST (U/L) | ALP (U/L) | GGT (U/L) | |
|---|---|---|---|---|---|---|
| Sham | 52.0±15.8 | 136.8±13.3 | 155±26.9 | 1.8±0.7 | ||
| BDL | 128.2±13.11) | 383.3±711) | 389.2±53.51) | 47.7±19.61) | ||
| HXQJL-L | 7.74 | 59.3±9.62) | 182.8±20.52) | 214.7±31.42) | 15.8±4.22) | |
| HXQJL-H | 15.48 | 53.6±7.42) | 141.9±28.12) | 179.4±57.62) | 10.1±4.92) | |
| EX527 | 0.005 | 169.7±16.42) | 490±107.82) | 577.5±70.92) | 78.2±7.72) | |
| EX527+HXQJL | 0.005+15.48 | 105.7±31.64) 289.7±101.34) | 361.2±54.24) | 41.3±14.44) | ||
表3 活血清解灵对BDL诱导肝纤维化大鼠血清ALT、AST、ALP和GGT水平的影响
Tab.3 Effect of HXQJL on serum levels of ALT, AST, ALP and GGT in rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | ALT (U/L) | AST (U/L) | ALP (U/L) | GGT (U/L) | |
|---|---|---|---|---|---|---|
| Sham | 52.0±15.8 | 136.8±13.3 | 155±26.9 | 1.8±0.7 | ||
| BDL | 128.2±13.11) | 383.3±711) | 389.2±53.51) | 47.7±19.61) | ||
| HXQJL-L | 7.74 | 59.3±9.62) | 182.8±20.52) | 214.7±31.42) | 15.8±4.22) | |
| HXQJL-H | 15.48 | 53.6±7.42) | 141.9±28.12) | 179.4±57.62) | 10.1±4.92) | |
| EX527 | 0.005 | 169.7±16.42) | 490±107.82) | 577.5±70.92) | 78.2±7.72) | |
| EX527+HXQJL | 0.005+15.48 | 105.7±31.64) 289.7±101.34) | 361.2±54.24) | 41.3±14.44) | ||
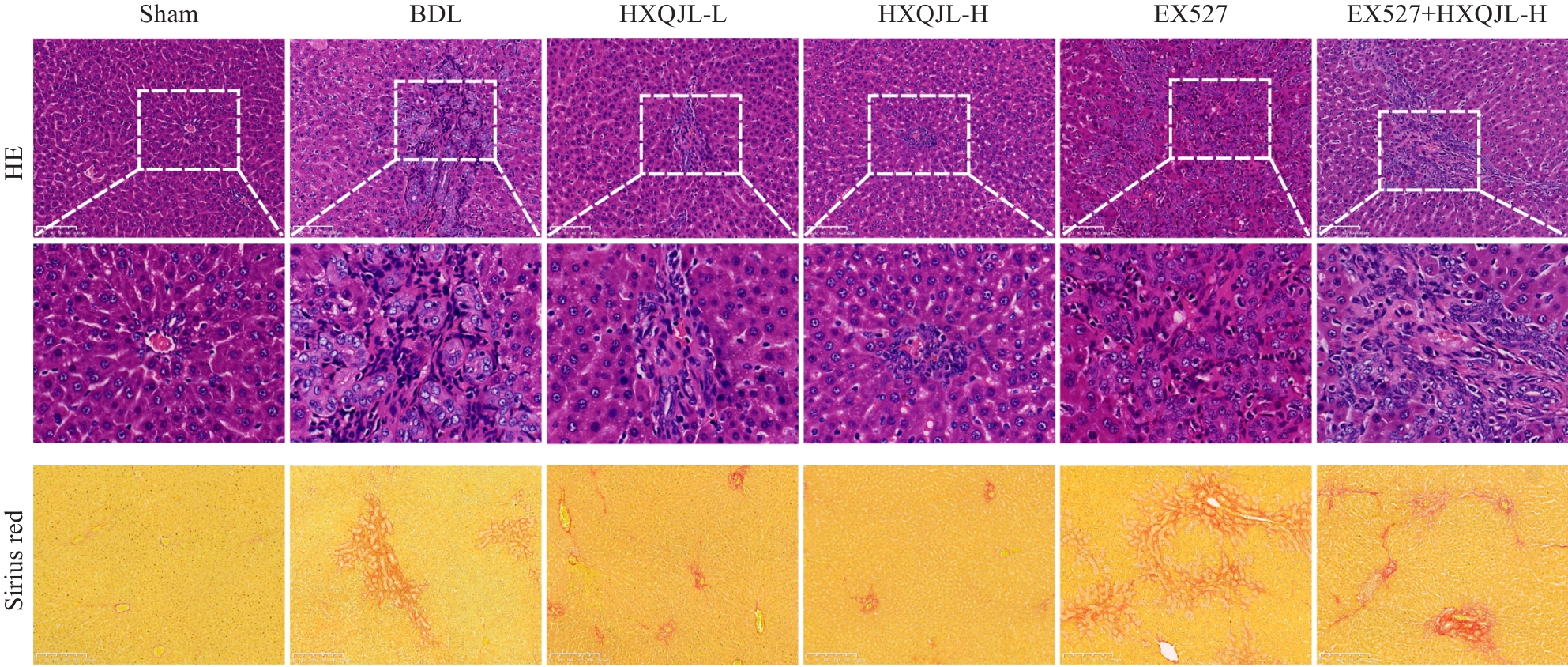
图1 活血清解灵对 BDL诱导的肝纤维化大鼠肝组织 HE 染色和天狼星红染色的影响
Fig.1 HE staining (Original magnification: ×200) and Sirius red staining (×100) of the liver tissue of rats in different groups.
| Group | Dose (g/kg) | Pathology score | Sirius red positive area (%) |
|---|---|---|---|
| Sham | 1.2±0.1 | 3.2±0.6 | |
| BDL | 3.9±0.31) | 47.5±7.81) | |
| HXQJL-L | 7.74 | 1.9±0.32) | 28.9±5.52) |
| HXQJL-H | 15.48 | 1.6±0.12) | 16.0±2.22) 3) |
| EX527 | 0.005 | 5.3±0.52) | 64.9±5.42) |
| EX527+HXQJL | 0.005+15.48 | 3.1±0.64) | 47.3±3.22) |
表4 活血清解灵对BDL诱导肝纤维化大鼠肝组织病理学损伤的影响
Tab.4 Effect of HXQJL on liver histopathological damage in rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Pathology score | Sirius red positive area (%) |
|---|---|---|---|
| Sham | 1.2±0.1 | 3.2±0.6 | |
| BDL | 3.9±0.31) | 47.5±7.81) | |
| HXQJL-L | 7.74 | 1.9±0.32) | 28.9±5.52) |
| HXQJL-H | 15.48 | 1.6±0.12) | 16.0±2.22) 3) |
| EX527 | 0.005 | 5.3±0.52) | 64.9±5.42) |
| EX527+HXQJL | 0.005+15.48 | 3.1±0.64) | 47.3±3.22) |
| Group | Dose (g/kg) | FN | COL I | α-SMA |
|---|---|---|---|---|
| Sham | 1.00±0.19 | 1.00±0.08 | 1.03±0.15 | |
| BDL | 2.84±0.621) | 11.87±3.151) | 4.62±1.121) | |
| HXQJL-L | 7.74 | 1.62±0.262) | 6.07±1.192) | 2.67±0.622) |
| HXQJL-H | 15.48 | 1.36±0.442) | 3.46±0.692) | 1.79±0.472) |
| EX527 | 0.005 | 5.36±1.062) | 18.96±3.362) | 8.56±1.392) |
| EX527+HXQJL | 0.005+15.48 | 3.08±1.404) | 13.79±4.924) | 3.91±1.54) |
表 5 活血清解灵对BDL诱导肝纤维化大鼠肝组织中FN、COL I和α-SMA mRNA表达的影响
Tab.5 Effect of HXQJL on mRNA level of FN, COL I and α-SMAin the liver tissues of rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | FN | COL I | α-SMA |
|---|---|---|---|---|
| Sham | 1.00±0.19 | 1.00±0.08 | 1.03±0.15 | |
| BDL | 2.84±0.621) | 11.87±3.151) | 4.62±1.121) | |
| HXQJL-L | 7.74 | 1.62±0.262) | 6.07±1.192) | 2.67±0.622) |
| HXQJL-H | 15.48 | 1.36±0.442) | 3.46±0.692) | 1.79±0.472) |
| EX527 | 0.005 | 5.36±1.062) | 18.96±3.362) | 8.56±1.392) |
| EX527+HXQJL | 0.005+15.48 | 3.08±1.404) | 13.79±4.924) | 3.91±1.54) |
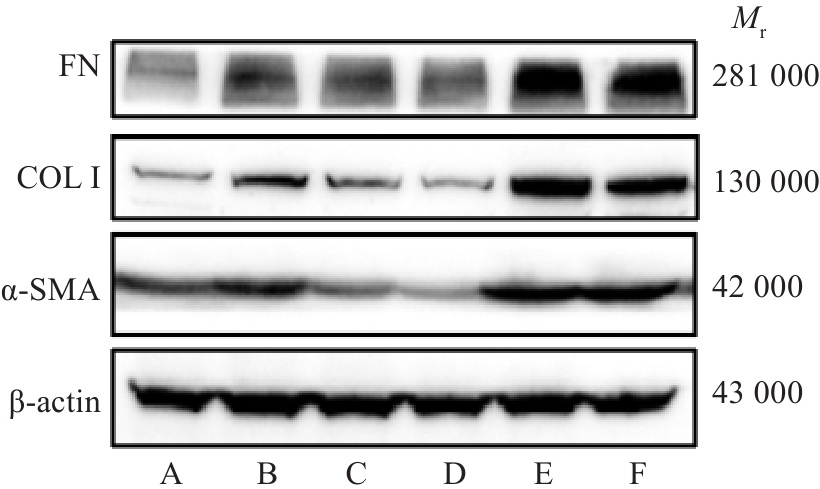
图2 HXQJL对 BDL诱导的肝纤维化大鼠肝组织中FN 、COL I和α-SMA蛋白表达的影响
Fig.2 Effect of HXQJL on expression of FN, COL I and α‑SMA protein in liver tissues in rats with BDL-induced liver fibrosis. A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Group | Dose (g/kg) | FN | COL I | α-SMA |
|---|---|---|---|---|
| Sham | 0.11±0.02 | 0.14±0.03 | 0.36±0.06 | |
| BDL | 0.36±0.021) | 0.38±0.011) | 0.75±0.071) | |
| HXQJL-L | 7.74 | 0.14±0.01 | 0.20±0.032) | 0.40±0.012) |
| HXQJL-H | 15.48 | 0.12±0.012) | 0.17±0.032) | 0.33±0.042) |
| EX527 | 0.005 | 2.33±0.172) | 1.12±0.112) | 1.22±0.102) |
| EX527+HXQJL | 0.005+15.48 | 1.44±0.134) | 0.70±0.064) | 1.00±0.024) |
表6 活血清解灵对BDL诱导肝纤维化大鼠肝组织中FN 、COL I和α-SMA蛋白表达的影响
Tab.6 Effect of HXQJL on protein expression levels of FN, COL I and α-SMAin the liver tissues of rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | FN | COL I | α-SMA |
|---|---|---|---|---|
| Sham | 0.11±0.02 | 0.14±0.03 | 0.36±0.06 | |
| BDL | 0.36±0.021) | 0.38±0.011) | 0.75±0.071) | |
| HXQJL-L | 7.74 | 0.14±0.01 | 0.20±0.032) | 0.40±0.012) |
| HXQJL-H | 15.48 | 0.12±0.012) | 0.17±0.032) | 0.33±0.042) |
| EX527 | 0.005 | 2.33±0.172) | 1.12±0.112) | 1.22±0.102) |
| EX527+HXQJL | 0.005+15.48 | 1.44±0.134) | 0.70±0.064) | 1.00±0.024) |
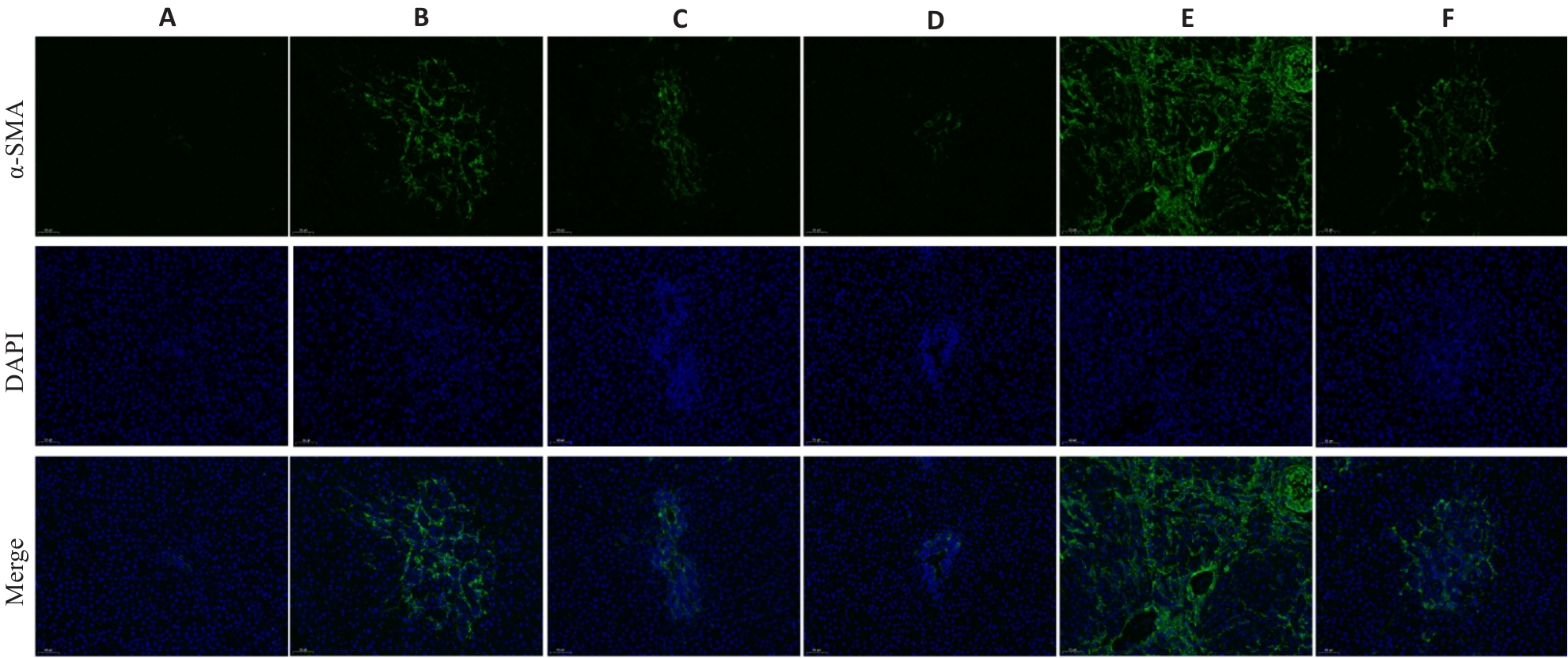
图3 活血清解灵对 BDL诱导的肝纤维化大鼠肝组织中α-SMA蛋白表达的影响
Fig.3 Effect of HXQJL onα-SMA expression in the liver tissues of rats with BDL-induced liver fibrosis(×200). A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Groups | Dose (g/kg) | Beclin-1 | Atg5 | LC3B | p62 |
|---|---|---|---|---|---|
| Sham | 1.01±0.08 | 1.00±0.20 | 1.00±0.17 | 1.00±0.14 | |
| BDL | 2.13±0.491) | 1.61±0.121) | 1.48±0.141) | 0.63±0.101) | |
| HXQJL-L | 7.74 | 1.11±0.082) | 1.09±0.182) | 0.92±0.162) | 0.83±0.182) |
| HXQJL-H | 15.48 | 1.03±0.182) | 0.85±0.202) | 0.83±0.252) | 0.98±0.142) |
| EX527 | 0.005 | 2.99±0.252) | 2.01±1.382) | 1.92±0.172) | 0.24±0.082) |
| EX527+HXQJL | 0.005+15.48 | 1.91±0.434) | 1.5±0.304) | 1.47±0.274) | 0.68±0.164) |
表7 活血清解灵对BDL诱导肝纤维化大鼠肝组织中Beclin-1、Atg5、LC3B 和 p62 mRNA 表达的影响
Tab.7 Effect of HXQJL on mRNA levels of Beclin-1, Atg5, LC3B and p62in the liver tissues of rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Groups | Dose (g/kg) | Beclin-1 | Atg5 | LC3B | p62 |
|---|---|---|---|---|---|
| Sham | 1.01±0.08 | 1.00±0.20 | 1.00±0.17 | 1.00±0.14 | |
| BDL | 2.13±0.491) | 1.61±0.121) | 1.48±0.141) | 0.63±0.101) | |
| HXQJL-L | 7.74 | 1.11±0.082) | 1.09±0.182) | 0.92±0.162) | 0.83±0.182) |
| HXQJL-H | 15.48 | 1.03±0.182) | 0.85±0.202) | 0.83±0.252) | 0.98±0.142) |
| EX527 | 0.005 | 2.99±0.252) | 2.01±1.382) | 1.92±0.172) | 0.24±0.082) |
| EX527+HXQJL | 0.005+15.48 | 1.91±0.434) | 1.5±0.304) | 1.47±0.274) | 0.68±0.164) |
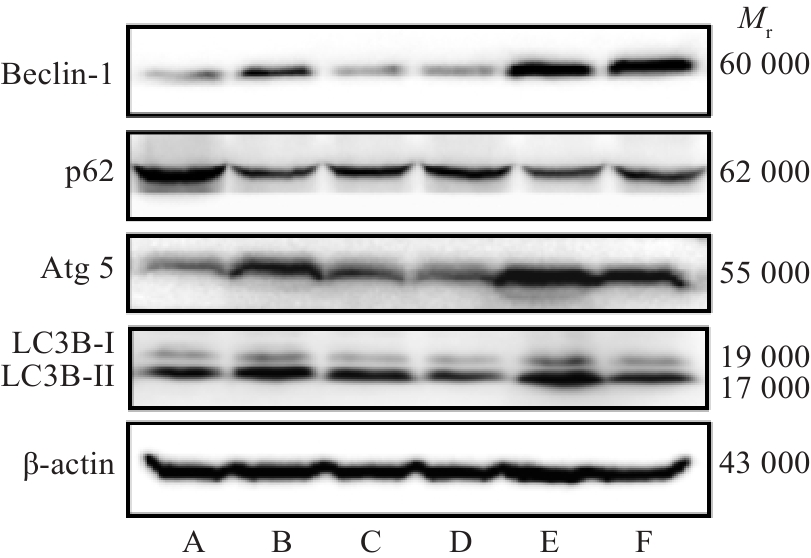
图4 活血清解灵对 BDL诱导的肝纤维化大鼠肝组织中Beclin-1、Atg5、LC3B 和p62蛋白表达的影响
Fig. 4 Effect of HXQJL on expressions of Beclin-1, Atg5, LC3B and p62 in the liver tissues of rats with BDL-induced liver fibrosis. A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Group | Dose (g/kg) | Beclin-1 | Atg5 | p62 | LC3B-II/LC3B-I |
|---|---|---|---|---|---|
| Sham | 0.48±0.01 | 0.60±0.04 | 1.25±0.11 | 1.81±0.01 | |
| BDL | 0.77±0.021) | 0.99±0.071) | 0.43±0.031) | 2.35±0.121) | |
| HXQJL-L | 7.74 | 0.52±0.032) | 0.73±0.032) | 0.57±0.03 | 2.14±0.022) |
| HXQJL-H | 15.48 | 0.49±0.022) | 0.67±0.042) | 0.83±0.072) | 1.83±0.022) |
| EX527 | 0.005 | 1.15±0.112) | 1.27±0.082) | 0.25±0.052) | 2.66±0.032) |
| EX527+HXQJL | 0.005+15.48 | 0.78±0.104) | 0.86±0.034) | 0.47±0.034) | 2.56±0.094) |
表8 活血清解灵对BDL诱导肝纤维化大鼠肝组织中Beclin-1、Atg5、LC3B 和 p62蛋白表达的影响
Tab.8 Effect of HXQJL on protein level of Beclin-1, Atg5, LC3B and p62in the liver tissues of rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Beclin-1 | Atg5 | p62 | LC3B-II/LC3B-I |
|---|---|---|---|---|---|
| Sham | 0.48±0.01 | 0.60±0.04 | 1.25±0.11 | 1.81±0.01 | |
| BDL | 0.77±0.021) | 0.99±0.071) | 0.43±0.031) | 2.35±0.121) | |
| HXQJL-L | 7.74 | 0.52±0.032) | 0.73±0.032) | 0.57±0.03 | 2.14±0.022) |
| HXQJL-H | 15.48 | 0.49±0.022) | 0.67±0.042) | 0.83±0.072) | 1.83±0.022) |
| EX527 | 0.005 | 1.15±0.112) | 1.27±0.082) | 0.25±0.052) | 2.66±0.032) |
| EX527+HXQJL | 0.005+15.48 | 0.78±0.104) | 0.86±0.034) | 0.47±0.034) | 2.56±0.094) |
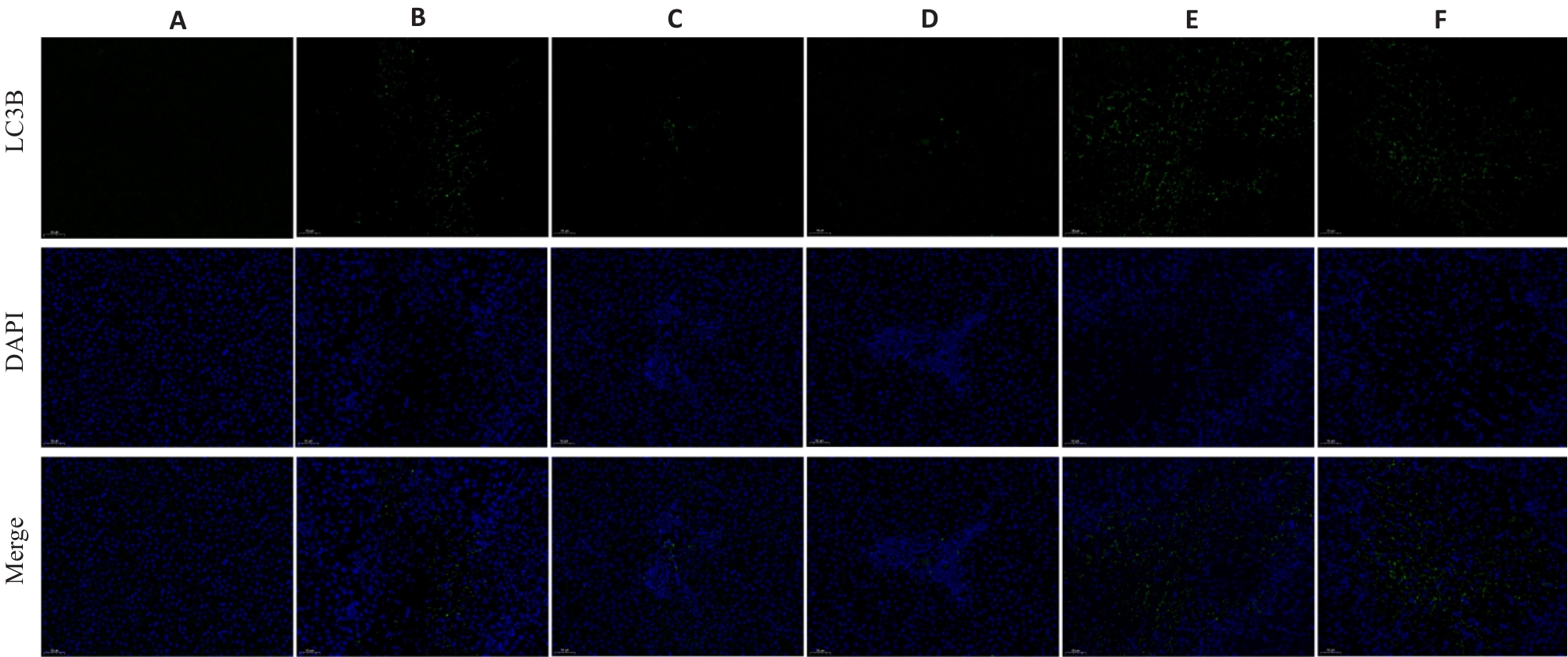
图 5 活血清解灵对 BDL诱导的肝纤维化大鼠肝组织中LC3B蛋白表达的影响
Fig.5 Effect of HXQJL on expressions of LC3B in the liver tissues of rats with BDL-induced liver fibrosis (×200). A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Group | Dose (g/kg) | Sirt 1 |
|---|---|---|
| Sham | 1.00±0.14 | |
| BDL | 0.60±0.041) | |
| HXQJL-L | 7.74 | 0.99±0.112) |
| HXQJL-H | 15.48 | 0.99±0.082) |
| EX527 | 0.005 | 0.28±0.082) |
| EX527+HXQJL | 0.005+15.48 | 0.51±0.154) |
表 9 活血清解灵对BDL诱导肝纤维化大鼠肝组织中Sirt 1mRNA 表达的影响
Tab.9 Effect of HXQJL on mRNA level ofSirt1in the liver tissues of the rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Sirt 1 |
|---|---|---|
| Sham | 1.00±0.14 | |
| BDL | 0.60±0.041) | |
| HXQJL-L | 7.74 | 0.99±0.112) |
| HXQJL-H | 15.48 | 0.99±0.082) |
| EX527 | 0.005 | 0.28±0.082) |
| EX527+HXQJL | 0.005+15.48 | 0.51±0.154) |
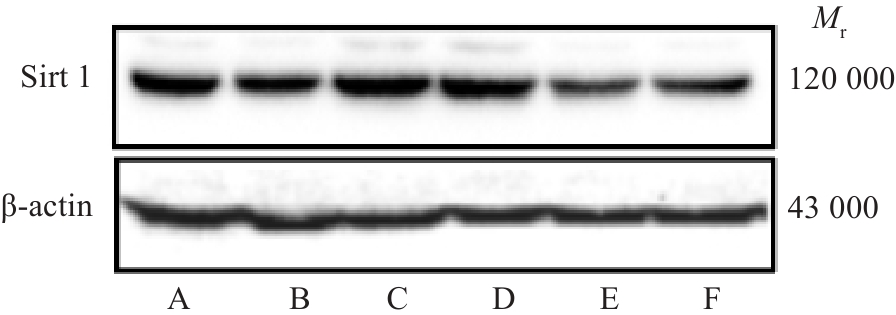
图 6 活血清解灵对 BDL诱导的肝纤维化大鼠肝组织中Sirt 1蛋白表达的影响
Fig.6 Effect of HXQJL on expression of Sirt 1 in liver tissues in rats with BDL-induced liver fibrosis. A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| Group | Dose (g/kg) | Sirt 1 |
|---|---|---|
| Sham | 1.14±0.10 | |
| BDL | 0.43±0.041) | |
| HXQJL-L | 7.74 | 1.09±0.042) |
| HXQJL-H | 15.48 | 1.13±0.102) |
| EX527 | 0.005 | 0.21±0.012) |
| EX527+HXQJL | 0.005+15.48 | 0.39±0.034) |
表10 活血清解灵对BDL诱导肝纤维化大鼠肝组织中Sirt 1蛋白表达的影响
Tab.10 Effect of HXQJL on protein level of Sirt1in the liver tissues of the rats with BDL-induced liver fibrosis (Mean±SD, n=8)
| Group | Dose (g/kg) | Sirt 1 |
|---|---|---|
| Sham | 1.14±0.10 | |
| BDL | 0.43±0.041) | |
| HXQJL-L | 7.74 | 1.09±0.042) |
| HXQJL-H | 15.48 | 1.13±0.102) |
| EX527 | 0.005 | 0.21±0.012) |
| EX527+HXQJL | 0.005+15.48 | 0.39±0.034) |
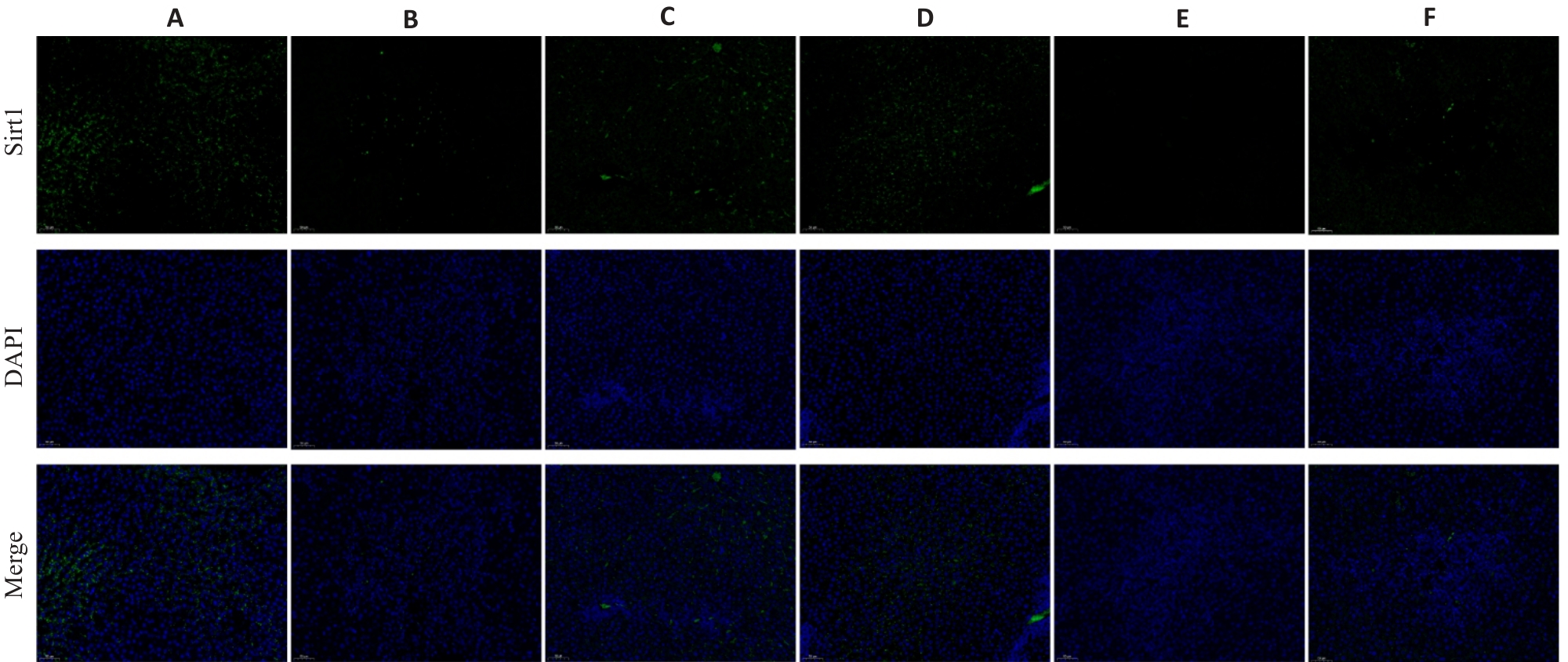
图 7 活血清解灵对 BDL诱导的肝纤维化大鼠肝组织中Sirt 1蛋白表达的影响
Fig.7 Effect of HXQJL on Sirt 1 expression in the liver tissues of the rats with BDL-induced liver fibrosis(×200). A: Sham group; B: BDL group; C: HXQJL-L group; D: HXQJL-H group; E: EX527 group; F: EX527+HXQJL group.
| [1] | Horn P, Tacke F. Metabolic reprogramming in liver fibrosis[J]. Cell Metab, 2024, 36(7): 1439-55. doi:10.1016/j.cmet.2024.05.003 |
| [2] | Filali-Mouncef Y, Hunter C, Roccio F, et al. The ménage à trois of autophagy, lipid droplets and liver disease[J]. Autophagy, 2022, 18(1): 50-72. doi:10.1080/15548627.2021.1895658 |
| [3] | Hernández-Gea V, Ghiassi-Nejad Z, Rozenfeld R, et al. Autophagy releases lipid that promotes fibrogenesis by activated hepatic stellate cells in mice and in human tissues[J]. Gastroenterology, 2012, 142(4): 938-46. doi:10.1053/j.gastro.2011.12.044 |
| [4] | Ma LQ, Fu RG, Duan ZY, et al. Sirt1 is essential for resveratrol enhancement of hypoxia-induced autophagy in the type 2 diabetic nephropathy rat[J]. Pathol Res Pract, 2016, 212(4): 310-8. doi:10.1016/j.prp.2016.02.001 |
| [5] | Gu LN, Tao XF, Xu YW, et al. Dioscin alleviates BDL- and DMN-induced hepatic fibrosis via Sirt1/Nrf2-mediated inhibition of p38 MAPK pathway[J]. Toxicol Appl Pharmacol, 2016, 292: 19-29. doi:10.1016/j.taap.2015.12.024 |
| [6] | 李锡晶, 沈洪昇, 王璐瑶, 等. 基于TGR5/NLRP3信号通路探讨活血清解灵对非酒精性脂肪性肝炎大鼠炎症反应的影响[J]. 中国现代应用药学, 2024, 41(10): 1324-31. doi:10.13748/j.cnki.issn1007-7693.20224076 |
| [7] | 无徐列明, 刘 平, 沈锡中, 等. 肝纤维化中西医结合诊疗指南(2019年版)[J]. 中国中西医结合杂志, 2019, 39(11): 1286-95. |
| [8] | 李婉华, 张桂贤, 沈洪昇, 等. 基于TGR5-NLRP3-Caspase1通路探讨活血清解灵对大鼠胆汁淤积性肝病的治疗作用[C]//第15届中国中西医结合学会基础理论专业委员会学术年会暨第二届广东省中西医结合学会转化医学专业委员会年会论文集. 广州, 2019: 47-8. |
| [9] | Cai YJ, Zheng Q, Sun R, et al. Recent progress in the study of Artemisiae Scopariae Herba (Yin Chen), a promising medicinal herb for liver diseases[J]. Biomed Pharmacother, 2020, 130: 110513. doi:10.1016/j.biopha.2020.110513 |
| [10] | Gijbels E, Pieters A, De Muynck K, et al. Rodent models of cholestatic liver disease: a practical guide for translational research[J]. Liver Int, 2021, 41(4): 656-82. doi:10.1111/liv.14800 |
| [11] | Ibrahim SH, Kamath BM, Loomes KM, et al. Cholestatic liver diseases of genetic etiology: Advances and controversies[J]. Hepatology, 2022, 75(6): 1627-46. doi:10.1002/hep.32437 |
| [12] | 王琳琳, 朱正望, 赵静涵, 等. 茵陈蒿汤对胆汁淤积性肝损伤的干预作用及调控机制[J/OL]. 中国实验方剂学杂志, 2025: 1-18. (2025-04-22). . |
| [13] | 向 杰, 邓敏贞, 李泽森, 等. 茵陈蒿汤方剂挥发油对肝纤维化小鼠的治疗作用及其机制[J]. 暨南大学学报: 自然科学与医学版, 2023, 44(6): 563-75. doi:10.11778/j.jdxb.20230125 |
| [14] | 朱晓东, 邸学士, 杨佳潞, 等. 基于网络药理学探讨茵陈蒿汤治疗肝纤维化的作用机制[J]. 中医药学报, 2021, 49(6): 29-36. doi:10.19664/j.cnki.1002-2392.210134 |
| [15] | 王 晶, 欧阳冰琛. 茵陈蒿汤防治肝脏疾病的药理作用及药动学研究进展[J]. 药物评价研究, 2021, 44(3): 628-37. doi:10.7501/j.issn.1674-6376.2021.03.026 |
| [16] | Cui XY, Zhang RH, Li YF, et al. Bie Jia Jian pill ameliorates BDL-induced cholestatic hepatic fibrosis in rats by regulating intestinal microbial composition and TMAO-mediated PI3K/AKT signaling pathway[J]. J Ethnopharmacol, 2025, 337(Pt 2): 118910. doi:10.1016/j.jep.2024.118910 |
| [17] | Xiao Z, Gao SQ, Li SS, et al. Taohong Siwu Decoction modulates glutathione metabolism to suppress hepatocyte ferroptosis and demonstrates anti-fibrotic effects in the liver[J]. J Ethnopharmacol, 2025, 350: 120025. doi:10.1016/j.jep.2025.120025 |
| [18] | Qu JR, Xue XY, Wang ZX, et al. Si-Wu-Tang attenuates liver fibrosis via regulating lncRNA H19-dependent pathways involving cytoskeleton remodeling and ECM deposition[J]. Chin J Nat Med, 2024, 22(1): 31-46. doi:10.1016/s1875-5364(24)60560-1 |
| [19] | Liu HM, Dong F, Li GQ, et al. Liuweiwuling tablets attenuate BDL-induced hepatic fibrosis via modulation of TGF-β/Smad and NF-κB signaling pathways[J]. J Ethnopharmacol, 2018, 210: 232-41. doi:10.1016/j.jep.2017.08.029 |
| [20] | Hou LS, Zhai XP, Zhang YW, et al. Targeted inhibition of autophagy in hepatic stellate cells by hydroxychloroquine: an effective therapeutic approach for the treatment of liver fibrosis[J]. Liver Int, 2024, 44(8): 1937-51. doi:10.1111/liv.15915 |
| [21] | Liu Y, Yao L, Liu YY, et al. Micheliolide alleviates hepatic fibrosis by inhibiting autophagy in hepatic stellate cells via the TrxR1/2-mediated ROS/MEK/ERK pathway[J]. Pharmaceuticals (Basel), 2025, 18(3): 287. doi:10.3390/ph18030287 |
| [22] | Wu LW, Zhang QH, Mo WH, et al. Quercetin prevents hepatic fibrosis by inhibiting hepatic stellate cell activation and reducing autophagy via the TGF-β1/Smads and PI3K/Akt pathways[J]. Sci Rep, 2017, 7: 9289. doi:10.1038/s41598-017-09673-5 |
| [23] | Ji J, Yu Q, Dai WQ, et al. Apigenin alleviates liver fibrosis by inhibiting hepatic stellate cell activation and autophagy via TGF-β 1/Smad3 and p38/PPAR α pathways[J]. PPAR Res, 2021, 2021: 6651839. doi:10.1155/2021/6651839 |
| [24] | Qin TT, Hasnat M, Wang ZW, et al. Geniposide alleviated bile acid-associated NLRP3 inflammasome activation by regulating SIRT1/FXR signaling in bile duct ligation-induced liver fibrosis[J]. Phytomedicine, 2023, 118: 154971. doi:10.1016/j.phymed.2023.154971 |
| [25] | Xia Y, Luo QH, Gao Q, et al. SIRT1 activation ameliorates Rhesus monkey liver fibrosis by inhibiting the TGF‑β/smad signaling pathway[J]. Chem Biol Interact, 2024, 394: 110979. doi:10.1016/j.cbi.2024.110979 |
| [26] | Zheng W, Guan F, Xu G, et al. FAT10 silencing prevents liver fibrosis through regulating SIRT1 expression in hepatic stellate cells[J]. Int J Med Sci, 2023, 20(4): 557-65. doi:10.7150/ijms.77367 |
| [27] | Lu HJ, Zhang RH, Zhang SK, et al. HSC-derived exosomal miR-199a-5p promotes HSC activation and hepatocyte EMT via targeting SIRT1 in hepatic fibrosis[J]. Int Immunopharmacol, 2023, 124(Pt B): 111002. doi:10.1016/j.intimp.2023.111002 |
| [28] | Yang L, Ao QF, Zhong QM, et al. SIRT1/IGFBPrP1/TGF β1 axis involved in cucurbitacin B ameliorating concanavalin A-induced mice liver fibrosis[J]. Basic Clin Pharmacol Toxicol, 2020, 127(5): 371-9. doi:10.1111/bcpt.13446 |
| [29] | Zhu J, Wang RW, Xu T, et al. Salvianolic acid a attenuates endoplasmic reticulum stress and protects against cholestasis-induced liver fibrosis via the SIRT1/HSF1 pathway[J]. Front Pharmacol, 2018, 9: 1277. doi:10.3389/fphar.2018.01277 |
| [30] | Patra S, Praharaj PP, Singh A, et al. Targeting SIRT1-regulated autophagic cell death as a novel therapeutic avenue for cancer prevention[J]. Drug Discov Today, 2023, 28(9): 103692. doi:10.1016/j.drudis.2023.103692 |
| [31] | Mei ZG, Huang YG, Feng ZT, et al. Electroacupuncture ameliorates cerebral ischemia/reperfusion injury by suppressing autophagy via the SIRT1-FOXO1 signaling pathway[J]. Aging (Albany NY), 2020, 12(13): 13187-205. doi:10.18632/aging.103420 |
| [1] | 乔通, 尹林, 张可妮, 牛民主, 黄菊, 耿志军, 李静, 胡建国. 茯苓新酸A通过调节AMPK/mTOR介导的自噬来减轻葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2026, 46(1): 131-140. |
| [2] | 徐嘉艺, 杨迪, 臧开来, 褚孟恩, 赵庆瑶, 李晴, 鲁森, 陈修丽, 李宁. EVA1A过表达通过调节脂质代谢和促进脂滴自噬改善非酒精性脂肪肝[J]. 南方医科大学学报, 2026, 46(1): 150-158. |
| [3] | 林心君, 何昱霖, 施红, 刘佳绣, 胡海霞. 石斛合剂通过调控Sirt3介导的线粒体自噬通路缓解大鼠糖尿病心肌病[J]. 南方医科大学学报, 2026, 46(1): 47-54. |
| [4] | 李玮怡, 江露, 张宗星, 陈丹, 包卓玛, 黄丽, 袁林. 强骨康疏方通过抑制HIF-1α/BNIP3自噬信号通路减少类风湿性关节炎大鼠的破骨细胞分化[J]. 南方医科大学学报, 2025, 45(7): 1389-1396. |
| [5] | 阳亭亭, 赵丽. 维生素A缺乏联合CCl4诱导制备稳定的小鼠慢性肝纤维化模型[J]. 南方医科大学学报, 2025, 45(7): 1527-1534. |
| [6] | 王心恒, 邵小涵, 李童童, 张璐, 杨勤军, 叶卫东, 童佳兵, 李泽庚, 方向明. 平喘宁方通过调控HMGB1/Beclin-1轴介导的自噬改善患寒哮证大鼠的气道炎症[J]. 南方医科大学学报, 2025, 45(6): 1153-1162. |
| [7] | 管丽萍, 颜燕, 卢心怡, 李智峰, 高晖, 曹东, 侯晨曦, 曾靖宇, 李欣怡, 赵洋, 王俊杰, 方会龙. 复方积雪草减轻小鼠日本血吸虫引起的肝纤维化:通过调控TLR4/MyD88通路抑制炎症-纤维化级联反应[J]. 南方医科大学学报, 2025, 45(6): 1307-1316. |
| [8] | 张璐, 丁焕章, 许浩燃, 陈珂, 许博文, 杨勤军, 吴迪, 童佳兵, 李泽庚. 参芪补中方通过激活AMPK/SIRT1/PGC-1α改善COPD肺脾气虚证大鼠线粒体功能障碍[J]. 南方医科大学学报, 2025, 45(5): 969-976. |
| [9] | 俞佳雯, 周薏, 钱春美, 穆蓝, 阙任烨. 铁过载诱导的小鼠肝纤维化过程影响巨噬细胞M2极化[J]. 南方医科大学学报, 2025, 45(4): 684-691. |
| [10] | 董妍妍, 张可敬, 储俊, 储全根. 抵当汤含药血清通过PI3K/Akt/mTOR信号通路增强高糖诱导的大鼠肾小球内皮细胞自噬[J]. 南方医科大学学报, 2025, 45(3): 461-469. |
| [11] | 廖茗, 钟文华, 张冉, 梁娟, 徐文陶睿, 万文珺, 吴超, 李曙. 源自蛇毒的蛋白C激活剂通过调控HIF-1α抑制BNIP3活性氧生成保护人脐静脉内皮细胞免受缺氧-复氧损伤[J]. 南方医科大学学报, 2025, 45(3): 614-621. |
| [12] | 花代平, 宣巧玉, 孙兰婷, 于庆生, 王琴, 王涛, 马麒颜, 杨文明, 汪瀚. Wilson 病患者 LncRNA Meg3 表达水平与肝纤维化指标的相关性分析[J]. 南方医科大学学报, 2025, 45(11): 2365-2374. |
| [13] | 易明, 罗烨, 吴露, 吴泽衡, 蒋翠平, 陈史钰, 柯晓. 黄芪甲苷通过抑制PINK1/Parkin通路调控细胞线粒体自噬减轻D-半乳糖诱导的内皮细胞衰老[J]. 南方医科大学学报, 2025, 45(11): 2427-2436. |
| [14] | 李思蒙, 陈建宁, 申思满, 刘望龙, 于丽丽, 张良清. 丹酚酸B通过抑制Sirt1蛋白降解促进心肌细胞线粒体功能稳态和改善缺血再灌注小鼠的心脏功能[J]. 南方医科大学学报, 2025, 45(10): 2062-2070. |
| [15] | 郭克磊, 李颖利, 宣晨光, 侯紫君, 叶松山, 李林运, 陈丽平, 韩立, 卞华. 益气养阴化浊通络方通过调控miR-21a-5p/FoxO1/PINK1介导的线粒体自噬减轻糖尿病肾病小鼠的足细胞损伤[J]. 南方医科大学学报, 2025, 45(1): 27-34. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||