南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 423-433.doi: 10.12122/j.issn.1673-4254.2026.02.20
• • 上一篇
黄林林1( ), 郑旺1, 胡建国2,4, 宋雪3,4, 陶露3,4, 耿志军3,4, 李静2,4, 左芦根1,4, 葛思堂1,4(
), 郑旺1, 胡建国2,4, 宋雪3,4, 陶露3,4, 耿志军3,4, 李静2,4, 左芦根1,4, 葛思堂1,4( )
)
收稿日期:2025-06-09
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
葛思堂
E-mail:huangl1nl1n@163.com;gesitang@163.com
作者简介:黄林林,在读硕士研究生,E-mail: huangl1nl1n@163.com
基金资助:
Linlin HUANG1( ), Wang ZHENG1, Jianguo HU2,4, Xue SONG3,4, Lu TAO3,4, Zhijun GENG3,4, Jing LI2,4, Lugen ZUO1,4, Sitang GE1,4(
), Wang ZHENG1, Jianguo HU2,4, Xue SONG3,4, Lu TAO3,4, Zhijun GENG3,4, Jing LI2,4, Lugen ZUO1,4, Sitang GE1,4( )
)
Received:2025-06-09
Online:2026-02-20
Published:2026-03-10
Contact:
Sitang GE
E-mail:huangl1nl1n@163.com;gesitang@163.com
摘要:
目的 探讨天然植物提取物马鞭草苷(VE)对克罗恩病(CD)样结肠炎的改善作用及其机制。 方法 建立2,4,6-三硝基苯磺酸(TNBS)诱导的小鼠结肠炎模型和脂多糖(LPS)刺激的结肠类器官损伤模型。将50只C57BL/6小鼠随机分为5组:WT组、TNBS组,VE低、中、高剂量组(5、10、20 mg/kg),10只/组。TNBS模型通过25 mg/L TNBS乙醇溶液灌肠建立,VE组每日腹腔注射相应剂量药物,连续治疗7 d。结肠类器官取自C57BL/6小鼠结肠隐窝,培养至第10天分为对照组、LPS组(100 μg/mL LPS处理24 h)和LPS+VE组(5、10、20 μmol/L VE共处理)。通过疾病活动指数(DAI)、结肠组织病理学评分、脾指数及炎症因子水平(IL-6、IL-1β、TNF-α)评估VE的疗效;采用免疫荧光染色、Western blotting检测紧密连接蛋白(ZO-1、Claudin-1)的表达;利用Western blotting检测PI3K-AKT通路蛋白的表达变化;结合PI3K-AKT通路激动剂740 Y-P干预实验验证通路调控机制。 结果 VE缓解TNBS小鼠的结肠炎症状(降低DAI评分、结肠炎症评分及脾指数,缓解体质量下降和结肠缩短)(P<0.05),同时,VE干预可下调黏膜组织促炎因子(IL-6、IL-1β、TNF-α)的表达(P<0.05),上调ZO-1和Claudin-1蛋白表达(P<0.05),减少细菌移位(P<0.05)。机制上,VE可下调p-PI3K、p-AKT蛋白的表达(P<0.05),且其疗效可被PI3K-AKT激动剂(740 Y-P)逆转(P<0.05)。 结论 VE通过调控PI3K-AKT信号通路抑制肠道炎症,从而修复肠屏障功能,改善小鼠CD样结肠炎。
黄林林, 郑旺, 胡建国, 宋雪, 陶露, 耿志军, 李静, 左芦根, 葛思堂. 马鞭草苷通过抑制PI3K-AKT通路减轻肠上皮炎症改善小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2026, 46(2): 423-433.
Linlin HUANG, Wang ZHENG, Jianguo HU, Xue SONG, Lu TAO, Zhijun GENG, Jing LI, Lugen ZUO, Sitang GE. Verbenalin ameliorates intestinal inflammation and colitis in a mouse model of Crohn's disease by inhibiting the PI3K-AKT pathway[J]. Journal of Southern Medical University, 2026, 46(2): 423-433.
| Gene | Primer sequence (5'to 3') |
|---|---|
| TNF-α | F: CACGCTCTTCTGTCTACTGAACTTC |
| R: CTTGGTGGTTTGTGAGTGTGAGG | |
| IL-1β | F: AATCTCGCAGCAGCACATCAAC |
| R: AGGTCCACGGGAAAGACACAG | |
| IL-6 | F: GAGAGGAGACTTCACAGAGGATACC |
| R: TCATTTCCACGATTTCCCAGAGAAC | |
| GAPDH | F: AACTCCCACTCTTCCACCTTCG R: TCCACCACCCTGTTGCTGTAG |
表1 引物序列
Tab.1 Primer sequences for RT-qPCR
| Gene | Primer sequence (5'to 3') |
|---|---|
| TNF-α | F: CACGCTCTTCTGTCTACTGAACTTC |
| R: CTTGGTGGTTTGTGAGTGTGAGG | |
| IL-1β | F: AATCTCGCAGCAGCACATCAAC |
| R: AGGTCCACGGGAAAGACACAG | |
| IL-6 | F: GAGAGGAGACTTCACAGAGGATACC |
| R: TCATTTCCACGATTTCCCAGAGAAC | |
| GAPDH | F: AACTCCCACTCTTCCACCTTCG R: TCCACCACCCTGTTGCTGTAG |
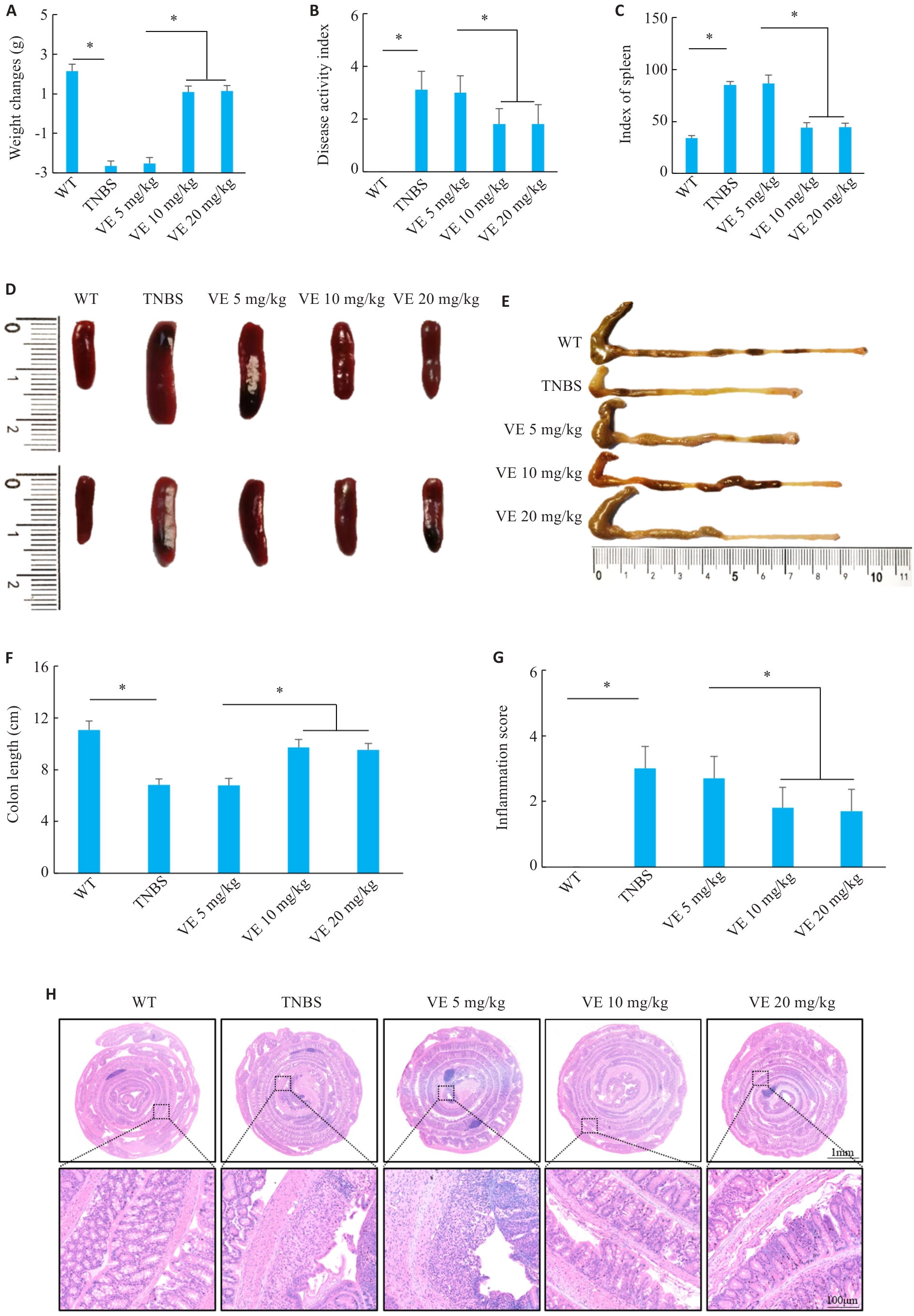
图1 VE干预对TNBS诱导的小鼠结肠炎症状的影响
Fig.1 Effects of VE treatment on TNBS-induced colitis symptoms in mice. A: Body weight changes. B: Disease activity index (DAI) scores. C: Spleen index. D: Spleen size. E: Gross morphology of mouse colon. F: Quantitative analysis of colon length. G: Histological inflammation score. H: HE staining of the colon tissue (n=10). *P<0.05.
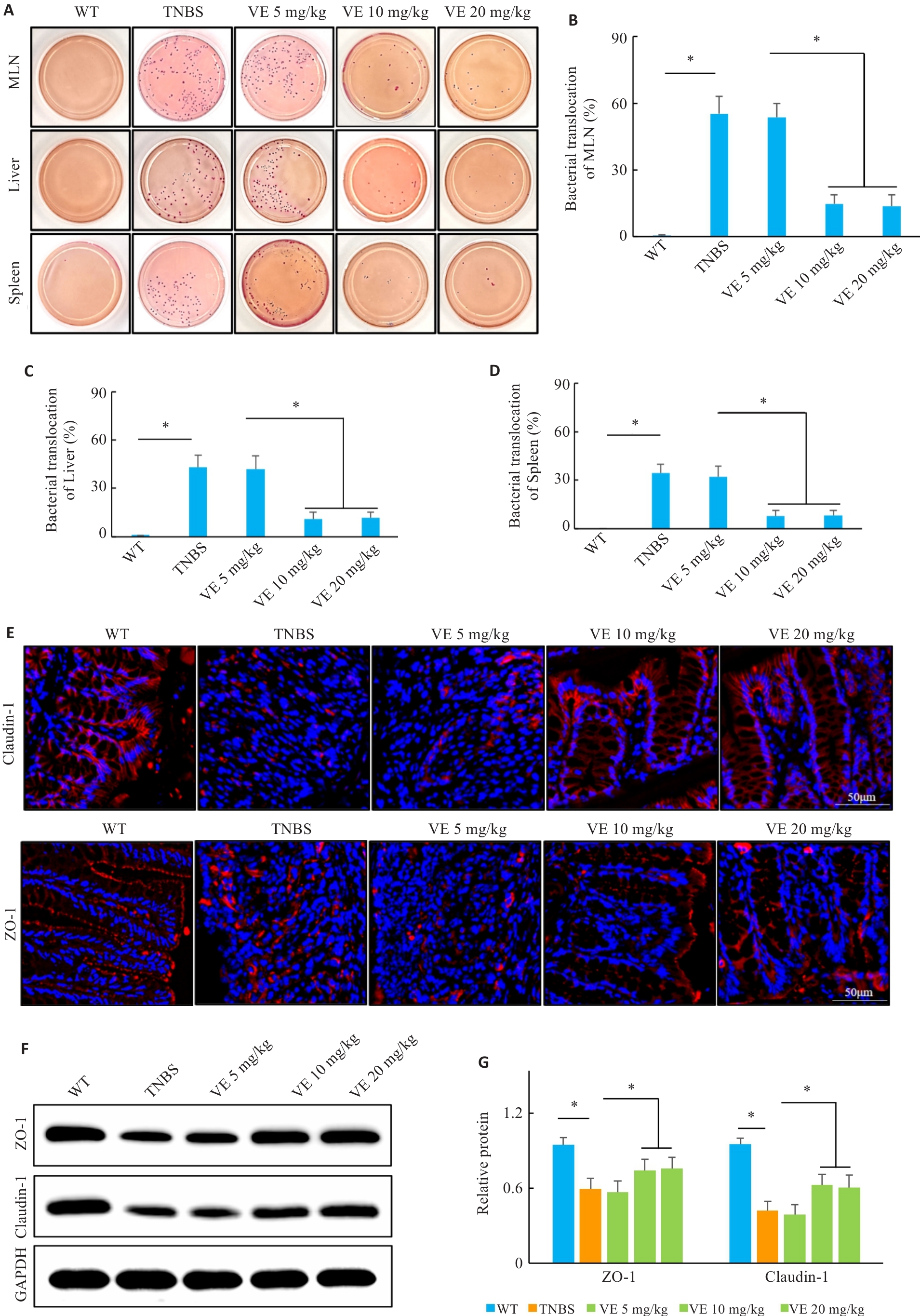
图2 VE干预对小鼠肠屏障功能和结构的影响
Fig.2 Effects of VE on intestinal barrier function and structure in mice. A-D: Bacterial culture and translocation rates in the lymph nodes, liver, and spleen. E: Immunofluorescence staining of ZO-1 and claudin-1 in colonic mucosa. F-G: Western blotting bands and quantitative analysis of ZO-1 and claudin-1 protein expression (n=10). *P<0.05.
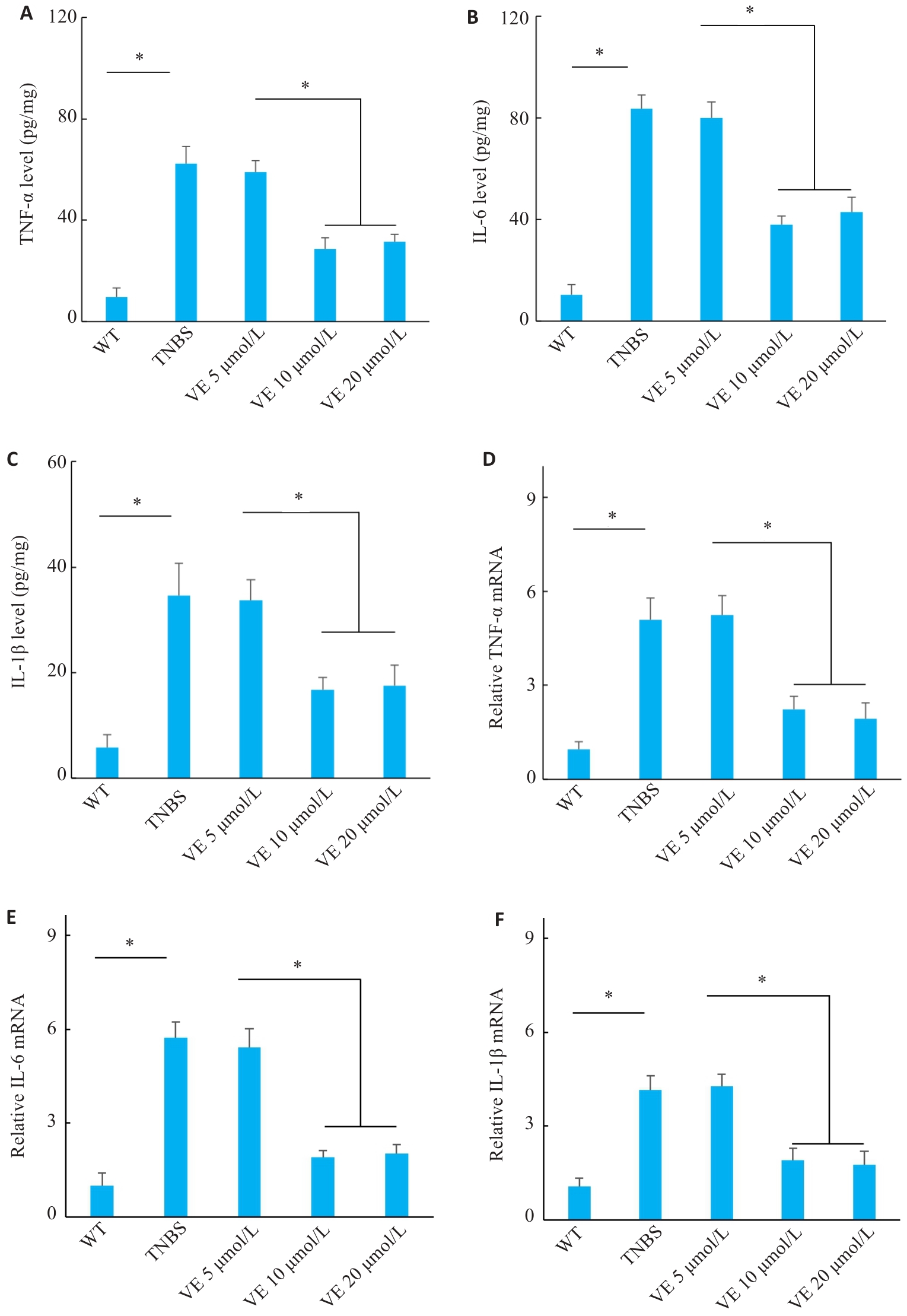
图3 VE干预对TNBS小鼠黏膜组织中的炎症介质水平的影响
Fig.3 Effects of VE on inflammatory mediator levels in colonic mucosa of TNBS-treated mice. A-C: ELISA detection of IL-6, IL-1β, and TNF-α protein levels. D-F: RT-qPCR analysis of IL-6, IL-1β, and TNF-α mRNA expression levels (n=10). *P<0.05.
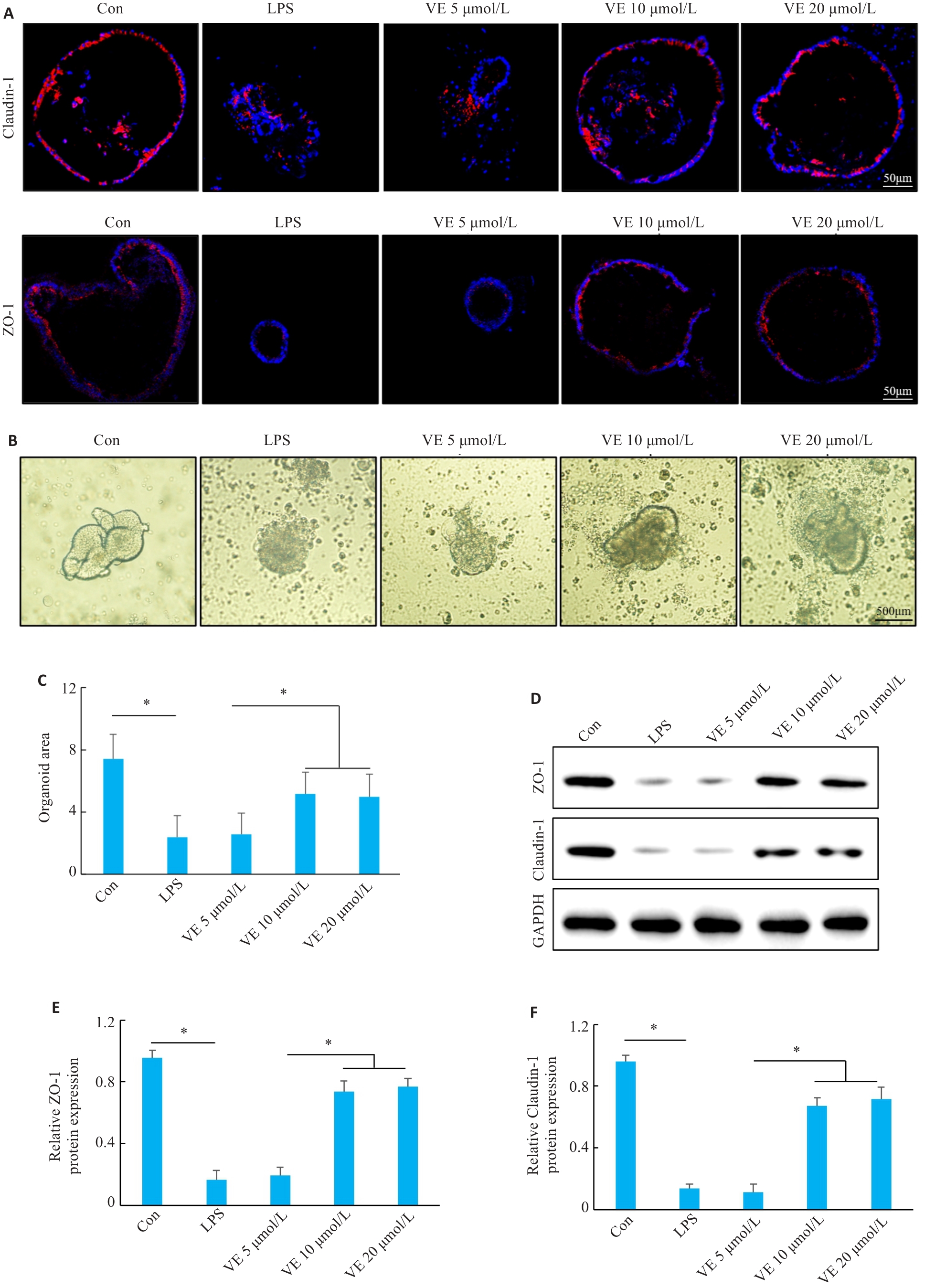
图4 VE干预对LPS诱导的结肠类器官肠屏障损伤的影响
Fig.4 Effects of VE on LPS-induced intestinal barrier injury in colonic organoids. A: Immunofluorescence staining of ZO-1 and claudin-1. B: Microscopic images of colonic organoids. C: Quantitative analysis of organoid area. D-F: Western blotting for analysis of ZO-1 and claudin-1 protein expression levels (n=3). *P<0.05.
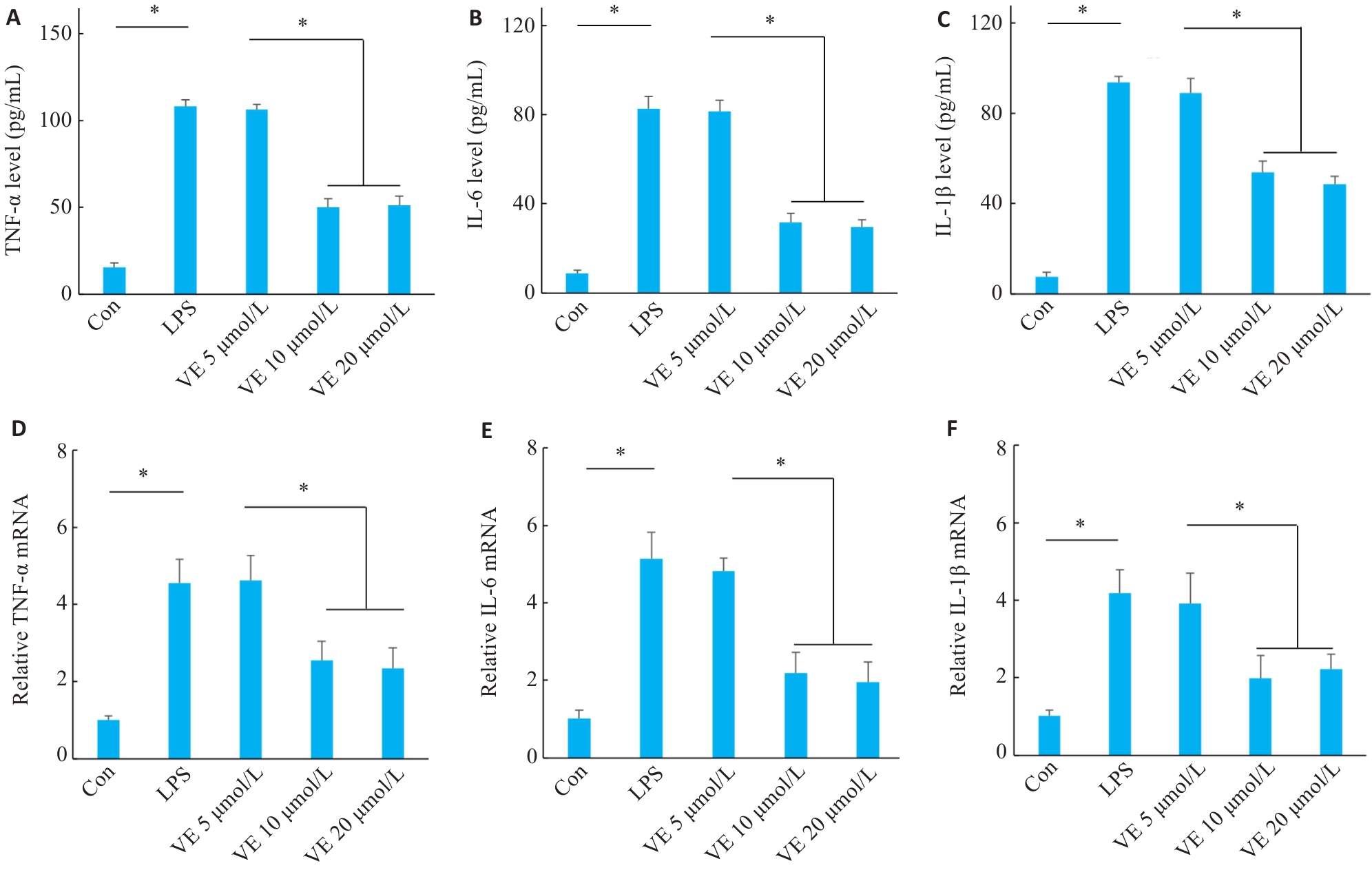
图5 VE干预对LPS诱导结肠类器官的炎症介质水平的影响
Fig.5 Effects of VE on inflammatory mediator levels in LPS-induced colonic organoids. A-C: ELISA detection of IL-6, IL-1β, and TNF-α protein levels. D-F: RT-qPCR analysis of IL-6, IL-1β, and TNF-α mRNA expression levels (n=3). *P<0.05.
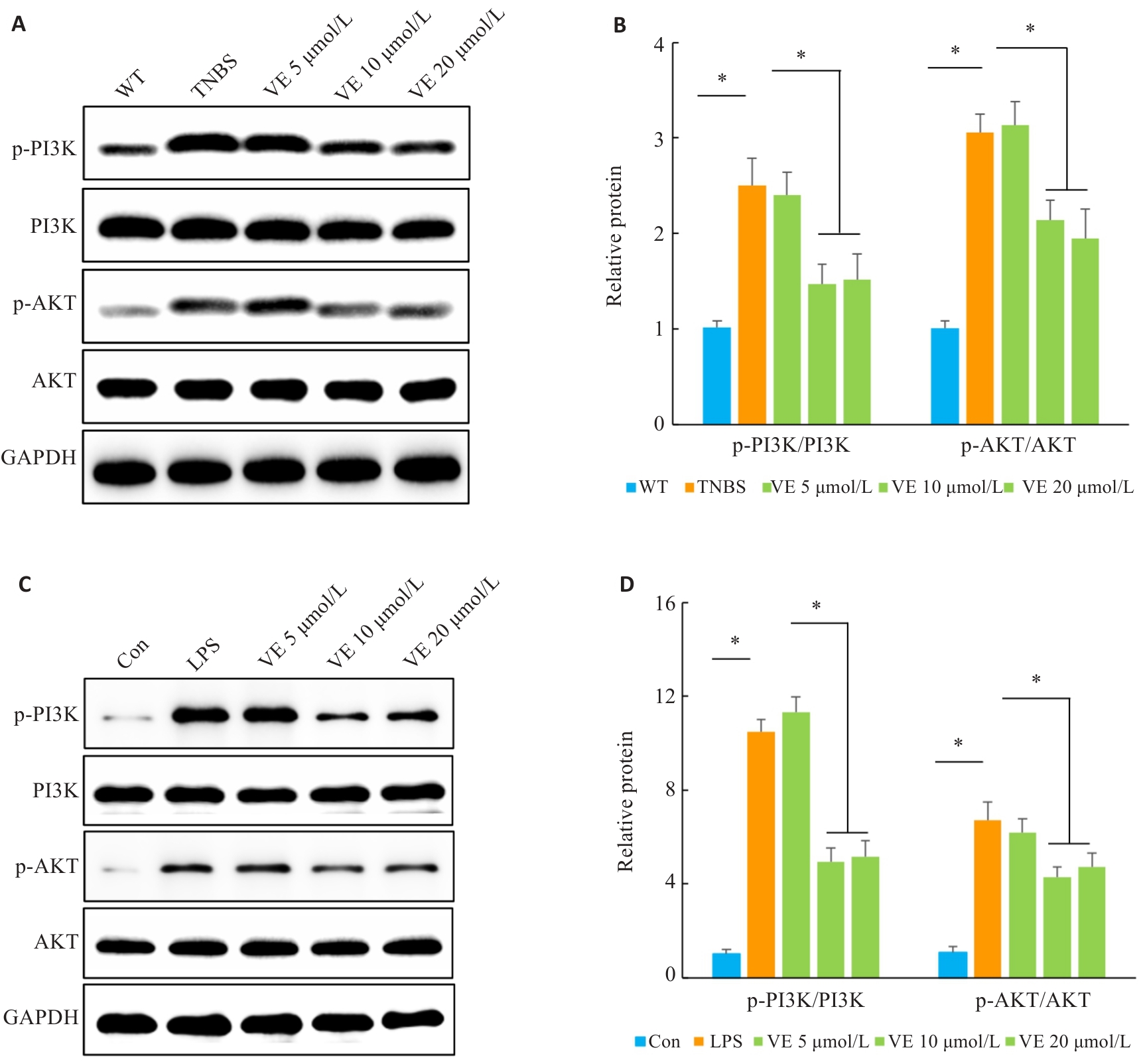
图6 VE干预对PI3K-AKT的信号通路活化的影响
Fig.6 Effects of VE on PI3K-AKT signaling pathway activation. A, B: Western blotting for analysis of p-PI3K/PI3K and p-AKT/AKT protein expressions in colon tissues of TNBS-treated mice (n=10). C, D: Western blotting for analysis of p-PI3K/PI3K and p-AKT/AKT protein expressions in LPS-induced colonic organoids (n=3). *P<0.05.
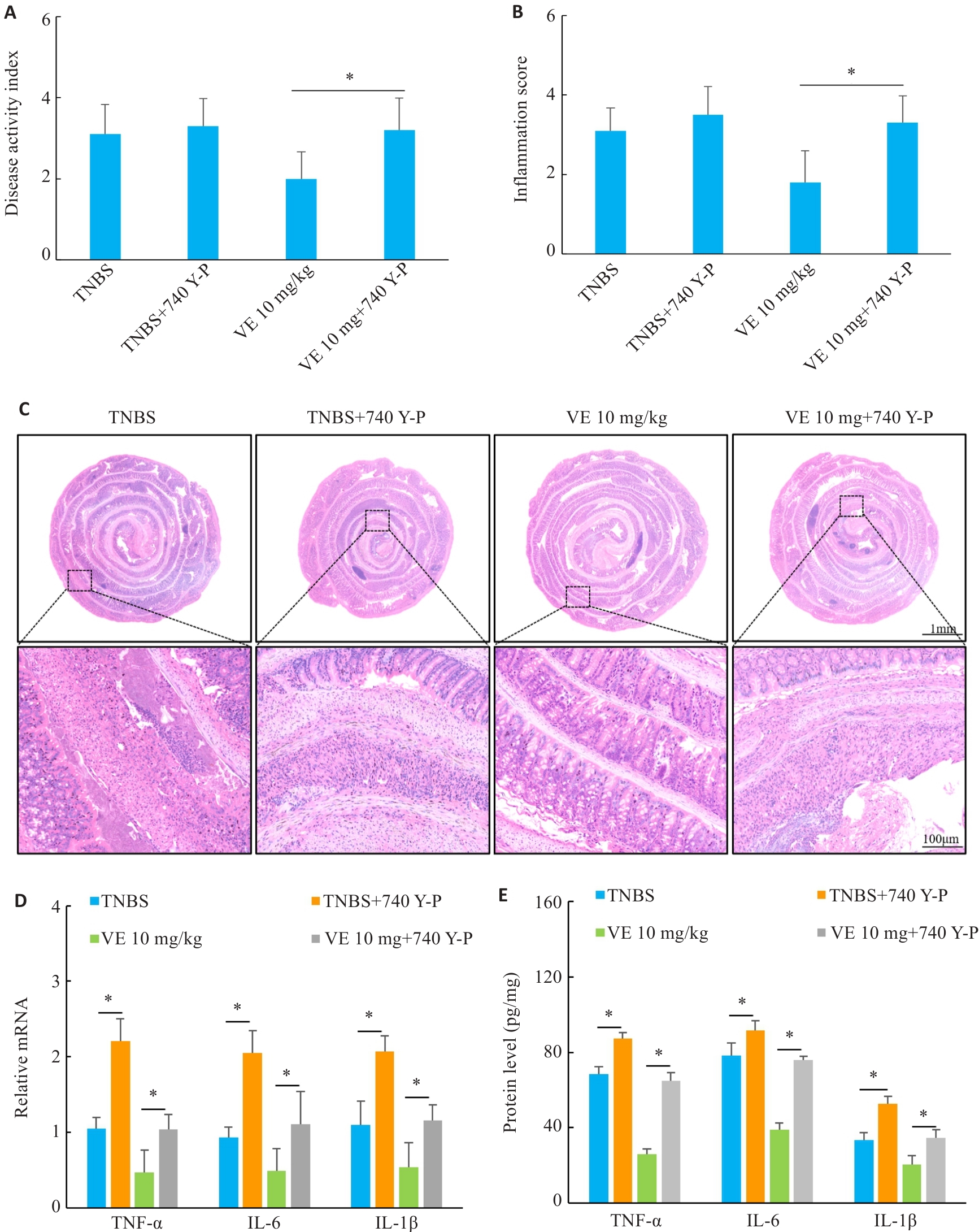
图7 VE通过抑制PI3K-AKT信号通路来影响小鼠克罗恩病样结肠炎
Fig.7 VE ameliorates colitis in mice by inhibiting the PI3K-AKT signaling pathway. A: DAI scores following 740 Y-P intervention. B: Histological inflammation scores. C: HE staining of the colon tissue. D-E: mRNA and protein expression levels of IL-6, IL-1β, and TNF-α in the colonic mucosa. n=10, *P<0.05.
| [1] | Wong SY, Estevinho MM, Heaney T, et al. Goblet cell loss linked to NOD2 and secondary resection in Crohn's disease is induced by dysbiosis and epithelial MyD88[J]. Cell Mol Gastroenterol Hepatol, 2025: 101533. doi:10.1016/j.jcmgh.2025.101533 |
| [2] | Palmela C, Chevarin C, Xu ZL, et al. Adherent-invasive Escherichia coli in inflammatory bowel disease[J]. Gut, 2018, 67(3): 574-87. doi:10.1136/gutjnl-2017-314903 |
| [3] | Liang WJ, Zhang W, Tian JY, et al. Advances in carbohydrate-based nanoparticles for targeted therapy of inflammatory bowel diseases: a review[J]. Int J Biol Macromol, 2024, 281(Pt 4): 136392. doi:10.1016/j.ijbiomac.2024.136392 |
| [4] | Pinelli M, Makdissi S, Scur M, et al. Peroxisomal cholesterol metabolism regulates Yap-signaling, which maintains intestinal epithelial barrier function and is altered in Crohn’s disease[J]. Cell Death Dis, 2024, 15(7): 536. doi:10.1038/s41419-024-06925-x |
| [5] | 李 静, 孙 洋, 熊心雨, 等. 樱黄素抑制TLR4/MyD88通路减轻肠上皮炎症反应改善小鼠克罗恩病样结肠炎[J]. 细胞与分子免疫学杂志, 2024, 40(3): 199-206. |
| [6] | Ma WY, Wang M, Chen JK, et al. Qingshu Yiqi decoction ameliorates exertional heat stroke-induced intestinal barrier injury via NF‑κB/MLC pathway and gut microbiota[J]. Phytomedicine, 2025, 143: 156723. doi:10.1016/j.phymed.2025.156723 |
| [7] | Kim HM, Kim YM. HMGB1: LPS delivery vehicle for caspase-11-mediated pyroptosis[J]. Immunity, 2018, 49(4): 582-4. doi:10.1016/j.immuni.2018.09.021 |
| [8] | Veloso PM, Machado R, Nobre C. Mesalazine and inflammatory bowel disease-From well-established therapies to progress beyond the state of the art[J]. Eur J Pharm Biopharm, 2021, 167: 89-103. doi:10.1016/j.ejpb.2021.07.014 |
| [9] | Kapizioni C, Desoki R, Lam D, et al. Biologic therapy for inflammatory bowel disease: real-world comparative effectiveness and impact of drug sequencing in 13 222 patients within the UK IBD BioResource[J]. J Crohns Colitis, 2024, 18(6): 790-800. |
| [10] | Alajmi A, Yuan YH, Solitano V, et al. 5-Aminosalicylates for non-surgical patients with active or quiescent Crohn's disease: an overview of systematic reviews (umbrella review)[J]. J Crohns Colitis, 2025, 19(5): jjaf069. doi:10.1093/ecco-jcc/jjaf069 |
| [11] | 李 艳, 刘 阳, 郑 敏, 等. 苍苓止泻口服液治疗儿童腹泻的系统评价[J]. 中国医院用药评价与分析, 2018, 18(1): 74-7, 82. |
| [12] | 朱鸿全. 桃红马鞭汤治疗子宫内膜异位症48例[J]. 陕西中医, 1998, 19(12): 532. |
| [13] | 赵益人. 马鞭草合剂治疗血尿[J]. 上海中医药杂志, 1979, 13(4): 26. |
| [14] | Wang YY, Wang X, Li YX, et al. Xuanfei Baidu Decoction reduces acute lung injury by regulating infiltration of neutrophils and macrophages via PD-1/IL17A pathway[J]. Pharmacol Res, 2022, 176: 106083. doi:10.1016/j.phrs.2022.106083 |
| [15] | Kang ZC, Jiang WL, Luan HY, et al. Cornin induces angiogenesis through PI3K-Akt-ENOS-VEGF signaling pathway[J]. Food Chem Toxicol, 2013, 58: 340-6. doi:10.1016/j.fct.2013.05.017 |
| [16] | Dong JH, Du CL, Xu CT, et al. Verbenalin attenuates hepatic damage and mitochondrial dysfunction in alcohol-associated steatohepatitis by regulating MDMX/PPARα‑mediated ferroptosis[J]. J Ethnopharmacol, 2023, 307: 116227. doi:10.1016/j.jep.2023.116227 |
| [17] | Zuo LG, Li J, Zhang XF, et al. Aberrant mesenteric adipose extracellular matrix remodelling is involved in adipocyte dysfunction in Crohn's disease: the role of TLR-4-mediated macrophages[J]. J Crohns Colitis, 2022, 16(11): 1762-76. doi:10.1093/ecco-jcc/jjac087 |
| [18] | Lukonin I, Serra D, Challet Meylan L, et al. Phenotypic landscape of intestinal organoid regeneration[J]. Nature, 2020, 586(7828): 275-80. doi:10.1038/s41586-020-2776-9 |
| [19] | Alula KM, Dowdell AS, LeBere B, et al. Interplay of gut microbiota and host epithelial mitochondrial dysfunction is necessary for the development of spontaneous intestinal inflammation in mice[J]. Microbiome, 2023, 11(1): 256. doi:10.1186/s40168-023-01686-9 |
| [20] | Zuo LG, Li J, Ge ST, et al. Bryostatin-1 ameliorated experimental colitis in Il-10-/- Mice by protecting the intestinal barrier and limiting immune dysfunction[J]. J Cell Mol Med, 2019, 23(8): 5588-99. doi:10.1111/jcmm.14457 |
| [21] | 李晴晴, 黄 菊, 孙 洋, 等. 乙酰紫堇灵通过抑制肠上皮细胞凋亡改善三硝基苯磺酸诱导的小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2023, 43(8): 1306-14. |
| [22] | Li YY, Wang XJ, Su YL, et al. Baicalein ameliorates ulcerative colitis by improving intestinal epithelial barrier via AhR/IL-22 pathway in ILC3s[J]. Acta Pharmacol Sin, 2022, 43(6): 1495-507. doi:10.1038/s41401-021-00781-7 |
| [23] | Dönder Y, Arikan TB, Baykan M, et al. Effects of quercitrin on bacterial translocation in a rat model of experimental colitis[J]. Asian J Surg, 2018, 41(6): 543-50. doi:10.1016/j.asjsur.2017.12.002 |
| [24] | Zuo LG, Geng ZJ, Song X, et al. Browning of mesenteric white adipose tissue in Crohn’s disease: a new pathological change and therapeutic target[J]. J Crohns Colitis, 2023, 17(8): 1179-92. doi:10.1093/ecco-jcc/jjad046 |
| [25] | Wirtz S, Popp V, Kindermann M, et al. Chemically induced mouse models of acute and chronic intestinal inflammation[J]. Nat Protoc, 2017, 12(7): 1295-309. doi:10.1038/nprot.2017.044 |
| [26] | Wei ZY, Ni X, Cui H, et al. Engeletin attenuates the inflammatory response via inhibiting TLR4-NFκB signaling pathway in Crohn's disease-like colitis[J]. J Ethnopharmacol, 2025, 336: 118733. doi:10.1016/j.jep.2024.118733 |
| [27] | Gao ZX, Yu XC, Su WL, et al. Atractylenolide-1 alleviates ulcerative colitis via restraining RhoA/ROCK/MLC pathway-mediated intestinal barrier dysfunction[J]. J Agric Food Chem, 2025, 73(21): 12690-701. doi:10.1021/acs.jafc.4c11976 |
| [28] | Liu HY, Li SC, Ogamune KJ, et al. Probiotic Lactobacillus johnsonii reduces intestinal inflammation and rebalances splenic treg/Th17 responses in dextran sulfate sodium-induced colitis[J]. Antioxidants (Basel), 2025, 14(4): 433. doi:10.3390/antiox14040433 |
| [29] | Leibovitzh H, Lee SH, Xue MY, et al. Altered gut microbiome composition and function are associated with gut barrier dysfunction in healthy relatives of patients with Crohn's disease[J]. Gastroenterology, 2022, 163(5): 1364-76.e10. doi:10.1053/j.gastro.2022.07.004 |
| [30] | Sato Y, Kanayama M, Nakajima S, et al. Sialyllactose enhances the short-chain fatty acid production and barrier function of gut epithelial cells via nonbifidogenic modification of the fecal microbiome in human adults[J]. Microorganisms, 2024, 12(2): 252. doi:10.3390/microorganisms12020252 |
| [31] | Spalinger MR, Schwarzfischer M, Niechcial A, et al. Loss of PTPN22 promotes intestinal inflammation by compromising granulocyte-mediated antibacterial defence[J]. J Crohns Colitis, 2021, 15(12): 2118-30. doi:10.1093/ecco-jcc/jjab098 |
| [32] | Hazime H, Ducasa GM, Santander AM, et al. DUOX2 activation drives bacterial translocation and subclinical inflammation in IBD-associated dysbiosis[J]. Gut, 2025, 74(10): 1589-601. doi:10.1136/gutjnl-2024-334346 |
| [33] | Zhang J, Zhao LL, He JY, et al. Protect effects of Perilla seed extract and its active ingredient luteolin against inflammatory bowel disease model via the PI3K/AKT signal pathway in vivo and in vitro [J]. Int J Mol Sci, 2025, 26(8): 3564. doi:10.3390/ijms26083564 |
| [34] | Qiao M, Xue TT, Zhu Y, et al. Polysaccharides from Cistanche deserticola mitigate inflammatory bowel disease via modulating intestinal microbiota and SRC/EGFR/PI3K/AKT signaling pathways[J]. Int J Biol Macromol, 2025, 308(Pt 2): 142452. doi:10.1016/j.ijbiomac.2025.142452 |
| [35] | Zhang SF, Zhong RQ, Zhou M, et al. Mechanisms of baicalin alleviates intestinal inflammation: role of M1 macrophage polarization and Lactobacillus amylovorus [J]. Adv Sci (Weinh), 2025, 12(21): e2415948. doi:10.1002/advs.202415948 |
| [36] | Geng ZJ, Zuo LG, Li J, et al. Ginkgetin improved experimental colitis by inhibiting intestinal epithelial cell apoptosis through EGFR/PI3K/AKT signaling[J]. FASEB J, 2024, 38(14): e23817. doi:10.1096/fj.202400211rr |
| [37] | Hu JY, Niu JJ, Jiang SS, et al. Qilian Jiechang Ning alleviates TNBS-induced ulcerative colitis in mice and segatella copri outer membrane vesicle-triggered inflammation in colon epithelial cells via the caspase-1/11-GSDMD pathways[J]. J Innate Immun, 2025, 17(1): 262-76. doi:10.1159/000545394 |
| [38] | Zhao HM, Xu R, Huang XY, et al. Curcumin improves regulatory T cells in gut-associated lymphoid tissue of colitis mice[J]. World J Gastroenterol, 2016, 22(23): 5374-83. doi:10.3748/wjg.v22.i23.5374 |
| [1] | 李斌杰, 周晓芳, 朗晓猛, 康欣, 刘建平. 泄浊解毒方通过调节Th17/Treg免疫平衡改善大鼠溃疡性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 335-344. |
| [2] | 尹林, 张可妮, 乔通, 牛民主, 殷丽霞, 刘馨悦, 耿志军, 李静, 胡建国. 藜芦酸通过激活Nrf2/HO-1信号通路减轻氧化应激缓解葡聚糖硫酸钠诱导的小鼠实验性结肠炎[J]. 南方医科大学学报, 2026, 46(2): 403-411. |
| [3] | 乔通, 尹林, 张可妮, 牛民主, 黄菊, 耿志军, 李静, 胡建国. 茯苓新酸A通过调节AMPK/mTOR介导的自噬来减轻葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2026, 46(1): 131-140. |
| [4] | 李嘉豪, 冼瑞婷, 李荣. 下调ACADM介导的脂毒性抑制雌激素受体阳性乳腺癌细胞的侵袭与转移[J]. 南方医科大学学报, 2025, 45(6): 1163-1173. |
| [5] | 牛民主, 殷丽霞, 乔通, 尹林, 张可妮, 胡建国, 宋传旺, 耿志军, 李静. 旱莲苷A通过调控JAK2/STAT3通路抑制M1型巨噬细胞极化改善葡聚糖硫酸钠诱导的小鼠结肠炎[J]. 南方医科大学学报, 2025, 45(6): 1297-1306. |
| [6] | 储菲, 陈孝华, 宋博文, 杨晶晶, 左芦根. 苏荠宁黄酮通过抑制PI3K/AKT信号通路拮抗肠上皮细胞凋亡改善小鼠实验性结肠炎[J]. 南方医科大学学报, 2025, 45(4): 819-828. |
| [7] | 殷丽霞, 牛民主, 张可妮, 耿志军, 胡建国, 李江艳, 李静. 升麻素抑制MAPK通路调节辅助性T细胞免疫平衡改善小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2025, 45(3): 595-602. |
| [8] | 黄菊, 殷丽霞, 牛民主, 耿志军, 左芦根, 李静, 胡建国. 紫花前胡苷通过抑制肠上皮细胞焦亡改善2,4,6-三硝基苯磺酸诱导的小鼠实验性结肠炎[J]. 南方医科大学学报, 2025, 45(2): 261-268. |
| [9] | 黄晴晴, 杨晶晶, 姜雪凝, 张文静, 汪煜, 左芦根, 王炼, 王月月, 张小凤, 宋雪, 胡建国. 刺桐碱通过抑制肠上皮炎症反应并改善肠屏障功能缓解小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2025, 45(11): 2456-2465. |
| [10] | 陶露, 陈悦, 黄林林, 郑旺, 宋雪, 项平, 胡建国. 珠子草素通过调控p38/JNK信号通路抑制肠上皮细胞凋亡保护肠屏障改善克罗恩病样肠炎[J]. 南方医科大学学报, 2025, 45(11): 2483-2495. |
| [11] | 刘硕, 李静, 吴兴旺. Swertiamarin通过抑制肠上皮细胞细胞凋亡改善TNBS诱导的实验性结肠炎[J]. 南方医科大学学报, 2024, 44(8): 1545-1552. |
| [12] | 席 进, 张 敏, 张永玉, 张 晨, 张雨路, 王 锐, 申 林, 李 静, 宋 雪. 上调KLF11可改善结肠炎模型小鼠的肠道炎症:基于抑制JAK2/STAT3信号通路[J]. 南方医科大学学报, 2024, 44(4): 765-772. |
| [13] | 牛民主, 殷丽霞, 段婷, 黄菊, 李静, 耿志军, 胡建国, 宋传旺. 川续断皂苷VI通过抑制PI3K/AKT/NF-κB通路拮抗肠上皮细胞凋亡缓解TNBS诱导的小鼠克罗恩病样结肠炎[J]. 南方医科大学学报, 2024, 44(12): 2335-2346. |
| [14] | 杨玉梅, 刘雪柔, 刘伟, 周星琦, 张振, 胡妍, 刘培培, 李娴, 刘浩, 李姗姗. 阿美替尼联合安罗替尼通过下调PI3K/AKT通路抑制非小细胞肺癌细胞的增殖[J]. 南方医科大学学报, 2024, 44(10): 1965-1975. |
| [15] | 邵荣瑢, 杨 子, 张文静, 张 诺, 赵雅静, 张小凤, 左芦根, 葛思堂. 茯苓酸缓解小鼠克罗恩病:基于抑制PI3K/AKT信号通路拮抗肠上皮细胞凋亡[J]. 南方医科大学学报, 2023, 43(6): 935-942. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||