Journal of Southern Medical University ›› 2026, Vol. 46 ›› Issue (2): 434-442.doi: 10.12122/j.issn.1673-4254.2026.02.21
Qiang WANG1( ), Zhixin CHAI1, Yulu DENG1, Zhiwei ZHANG1, Ying GONG1, Sheng GAO2(
), Zhixin CHAI1, Yulu DENG1, Zhiwei ZHANG1, Ying GONG1, Sheng GAO2( ), Pingfeng FENG1(
), Pingfeng FENG1( )
)
Received:2025-08-08
Online:2026-02-20
Published:2026-03-10
Contact:
Sheng GAO, Pingfeng FENG
E-mail:258114811@qq.com;xiuheyu@163.com;fengpf1978@163.com
Supported by:Qiang WANG, Zhixin CHAI, Yulu DENG, Zhiwei ZHANG, Ying GONG, Sheng GAO, Pingfeng FENG. cGAS-STING agonist cGAMP enhances natural killer cell-mediated cytotoxicity against gastric cancer cells[J]. Journal of Southern Medical University, 2026, 46(2): 434-442.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.j-smu.com/EN/10.12122/j.issn.1673-4254.2026.02.21
| Group | Post-modeling treatment method | Treatment basis |
|---|---|---|
| PBS | 肿瘤体积达 100 mm³ 后, 隔日腹腔注射 100 μL PBS, 持续至实验终点 | 作为空白对照,排除注射操作、溶剂(PBS) 对肿瘤生长的影响,明确后续治疗效应的特异性 |
| NK Cell | 肿瘤体积达 100 mm³ 后, 尾静脉回输 1×10⁷个 NK细胞; 隔日腹腔注射 20000 IU IL-2, 持续至实验终点 | IL-2 是 NK 细胞体外培养和体内存活的关键细胞因子, 可维持 NK 细胞活性,该组用于验证单独 NK 细胞对胃癌移植瘤的杀伤效果 |
| NK Cell+cGAMP | 肿瘤体积达 100 mm³ 后, 尾静脉回输 1×10⁷个 NK 细胞; 隔日腹腔注射 20000 IU IL-2; 隔日腹腔注射 1 μg/只 cGAMP, 持续至实验终点 | 基于前期体外实验中 cGAMP 对 NK 细胞的激活效应, 该组通过联合给药验证 cGAMP 是否能协同增强 NK 细胞的体内抗胃癌活性 |
Tab.1 Experimental Animal Treatment Groups (n=4)
| Group | Post-modeling treatment method | Treatment basis |
|---|---|---|
| PBS | 肿瘤体积达 100 mm³ 后, 隔日腹腔注射 100 μL PBS, 持续至实验终点 | 作为空白对照,排除注射操作、溶剂(PBS) 对肿瘤生长的影响,明确后续治疗效应的特异性 |
| NK Cell | 肿瘤体积达 100 mm³ 后, 尾静脉回输 1×10⁷个 NK细胞; 隔日腹腔注射 20000 IU IL-2, 持续至实验终点 | IL-2 是 NK 细胞体外培养和体内存活的关键细胞因子, 可维持 NK 细胞活性,该组用于验证单独 NK 细胞对胃癌移植瘤的杀伤效果 |
| NK Cell+cGAMP | 肿瘤体积达 100 mm³ 后, 尾静脉回输 1×10⁷个 NK 细胞; 隔日腹腔注射 20000 IU IL-2; 隔日腹腔注射 1 μg/只 cGAMP, 持续至实验终点 | 基于前期体外实验中 cGAMP 对 NK 细胞的激活效应, 该组通过联合给药验证 cGAMP 是否能协同增强 NK 细胞的体内抗胃癌活性 |
| Gene | Forward primers (5'to3') | Reverse primers (5'to3') |
|---|---|---|
| GAPDH | CTGTTCGACAGTCAGCCGCATC | GCGCCCAATACGACCAAATCCG |
| IFN-γ | TCGGTAACTGACTTGAATGTCCA | TCGCTTCCCTGTTTTAGCTGC |
| TNF-α | GAGGCCAAGCCCTGGTATG | CGGGCCGATTGATCTCAGC |
| GZMB | CCCTGGGAAAACACTCACACA | GCACAACTCAATGGTACTGTCG |
| IL-2 | AACTCCTGTCTTGCATTGCAC | GCTCCAGTTGTAGCTGTGTTT |
| Perforin | GACTGCCTGACTGTCGAGG | TCCCGGTAGGTTTGGTGGAA |
| NKG2D | CCTTGACCGAAAGTTACTGTGG | GGCTGGCATTTTGAGACATACAA |
| TIGIT | TGGTCGCGTTGACTAGAAAGA | GGGCTCCATTCCTCCTGTC |
Tab.2 qPCR primers
| Gene | Forward primers (5'to3') | Reverse primers (5'to3') |
|---|---|---|
| GAPDH | CTGTTCGACAGTCAGCCGCATC | GCGCCCAATACGACCAAATCCG |
| IFN-γ | TCGGTAACTGACTTGAATGTCCA | TCGCTTCCCTGTTTTAGCTGC |
| TNF-α | GAGGCCAAGCCCTGGTATG | CGGGCCGATTGATCTCAGC |
| GZMB | CCCTGGGAAAACACTCACACA | GCACAACTCAATGGTACTGTCG |
| IL-2 | AACTCCTGTCTTGCATTGCAC | GCTCCAGTTGTAGCTGTGTTT |
| Perforin | GACTGCCTGACTGTCGAGG | TCCCGGTAGGTTTGGTGGAA |
| NKG2D | CCTTGACCGAAAGTTACTGTGG | GGCTGGCATTTTGAGACATACAA |
| TIGIT | TGGTCGCGTTGACTAGAAAGA | GGGCTCCATTCCTCCTGTC |
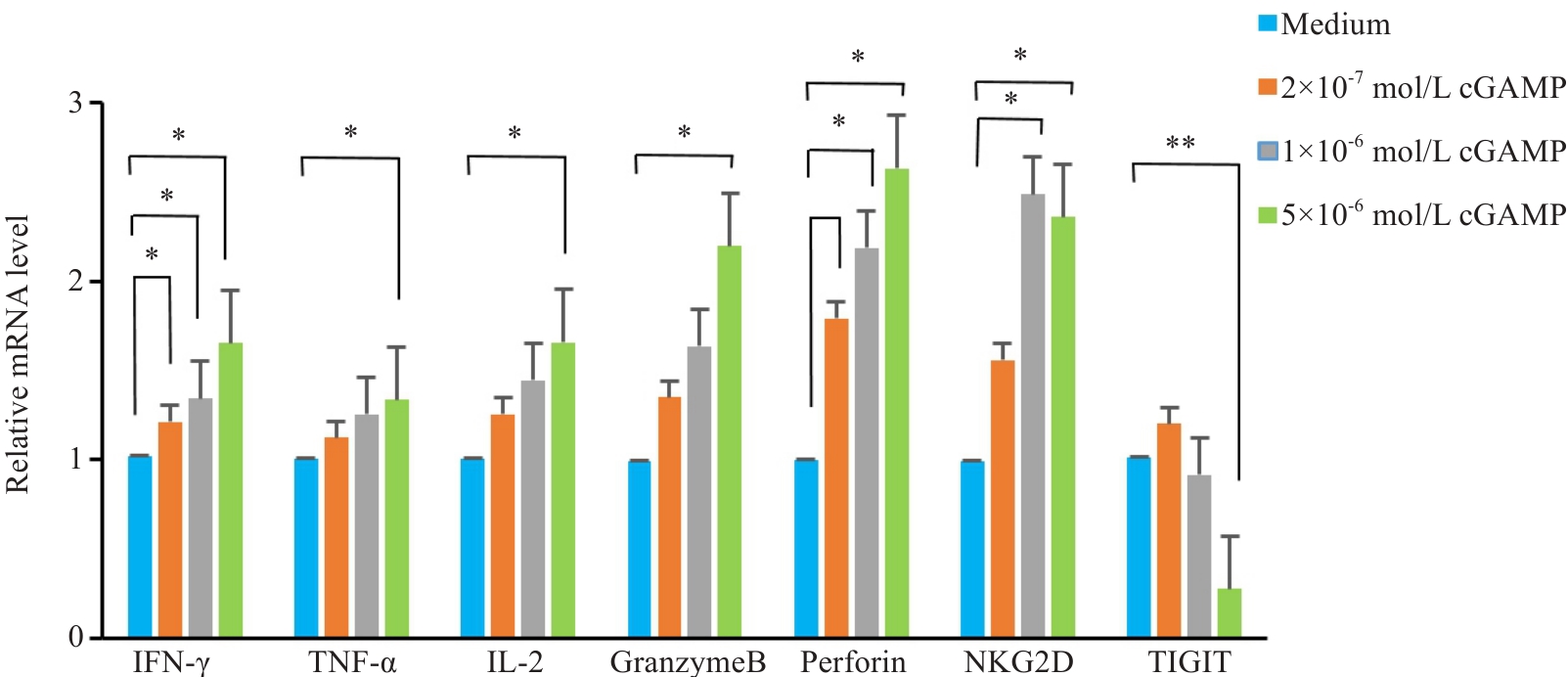
Fig.1 Effects of cGAS-STING agonist on mRNA expressions in NK-92 cells. Statistical analysis was done using one-way ANOVA with Tukey post-tests (n=3). *P<0.05, **P<0.01.
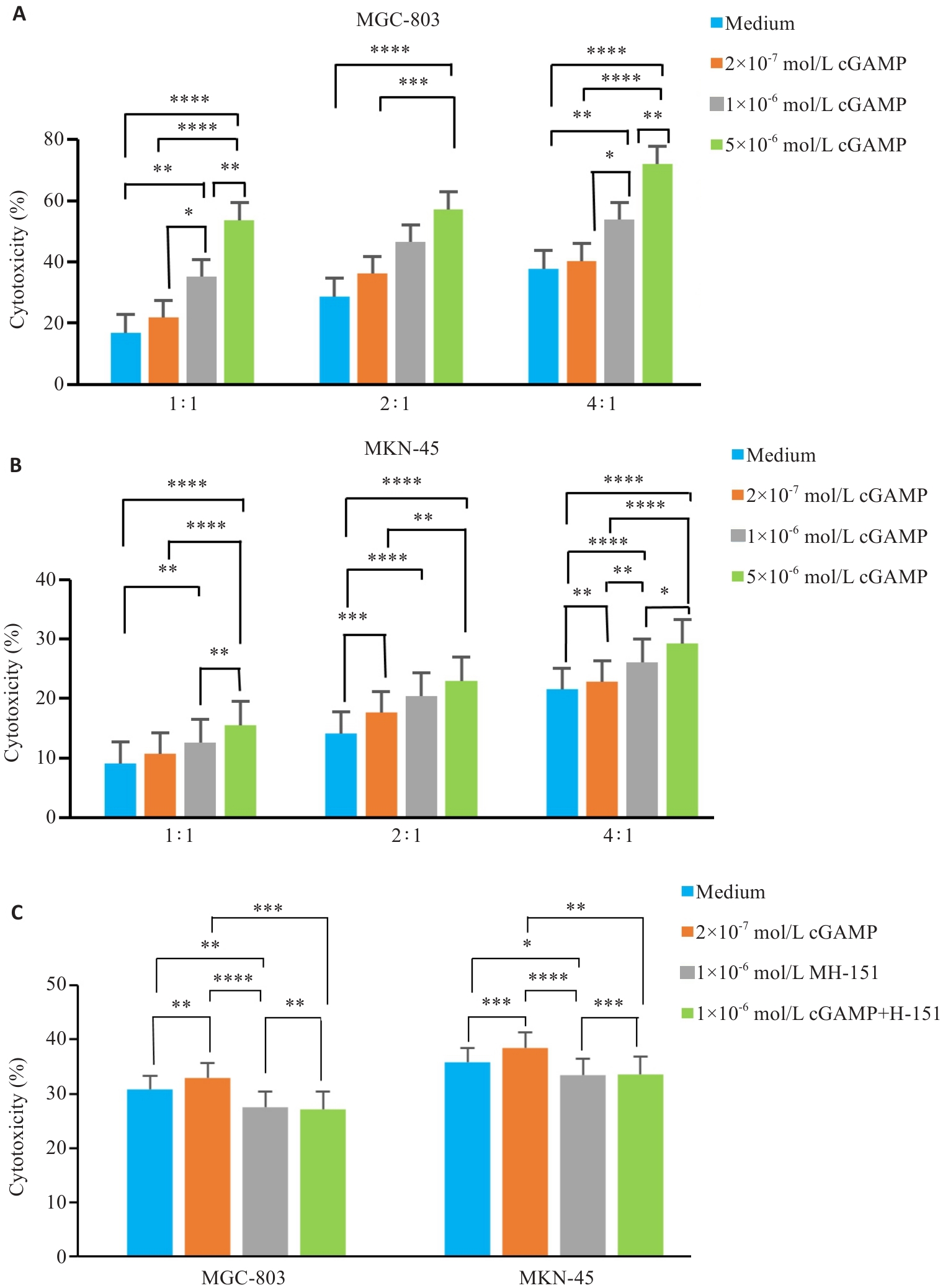
Fig.4 cGAS-STING agonist enhances cytotoxicity of NK-92 cells against gastric cancer cells in vitro. NK-92 cells pretreated with 1 μmol/L cGAMP showed significantly enhanced cytotoxicity against gastric cancer cells MGC-803 (A) and MKN-45 (B) at effector-to-target ratios of 1:1, 2:1 and 4:1. C: STING antagonist H-151 blockade assay, in which NK-92 cells were pretreated with 1×10-6 mol/L cGAMP alone or combined with 1×10-6 mol/L H-151 for 24 h, then co-cultured with MGC-803 or MKN-45 cells for another 4 h. Dead tumor cells were analyzed by flow cytometry. Data are presented as Mean±SD (n=3). *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
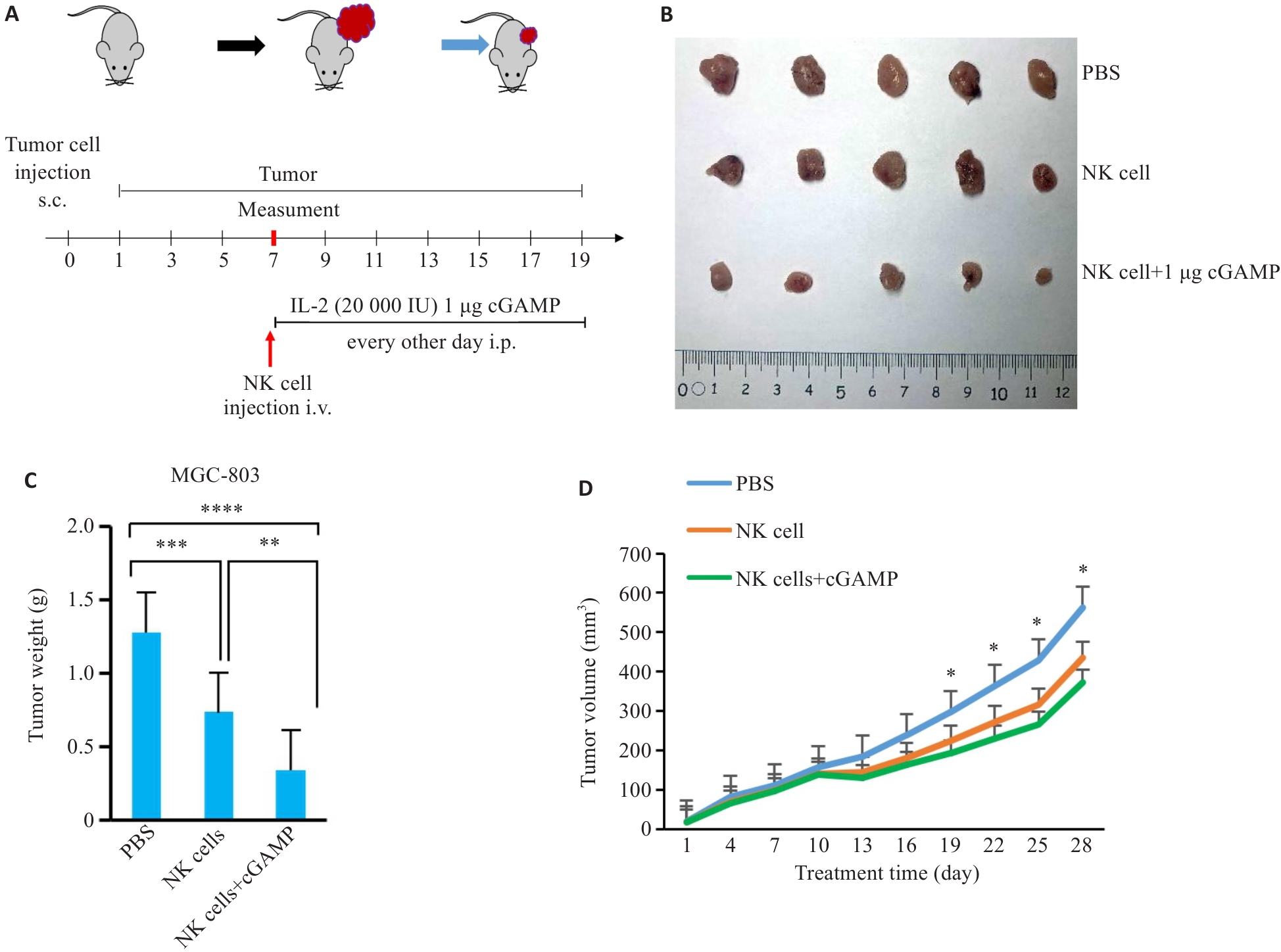
Fig.5 cGAS-STING agonist enhances NK-92 cell cytotoxicity against gastric cancer cells in nude mice. A: Process of tumor-bearing mouse model establishment and treatment process. B: Representative images of tumors at experimental endpoint showing obviously reduced tumor size in NK+cGAMP group. C: Bar graph of tumor weight at endpoint showing reduced tumor mass in NK+cGAMP group by 40% relative to that in NK-only group. D: Line graph of tumor volume in each group at endpoint. Data are presented as Mean±SD. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001.
| [1] | Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-49. doi:10.3322/caac.21660 |
| [2] | Joshi SS, Badgwell BD. Current treatment and recent progress in gastric cancer[J]. CA Cancer J Clin, 2021, 71(3): 264-79. doi:10.3322/caac.21657 |
| [3] | Smyth EC, Nilsson M, Grabsch HI, et al. Gastric cancer[J]. Lancet, 2020, 396(10251): 635-48. doi:10.1016/s0140-6736(20)31288-5 |
| [4] | Wagner AD, Syn NL, Moehler M, et al. Chemotherapy for advanced gastric cancer[J]. Cochrane Database Syst Rev, 2017, 8(8): CD004064. doi:10.1002/14651858.cd004064.pub4 |
| [5] | Fuchs CS, Doi T, Jang RW, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 trial[J]. JAMA Oncol, 2018, 4(5): e180013. doi:10.1001/jamaoncol.2018.0013 |
| [6] | Kang YK, Boku N, Satoh T, et al. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial[J]. Lancet, 2017, 390(10111): 2461-71. doi:10.1016/s0140-6736(17)31827-5 |
| [7] | Shitara K, Özgüroğlu M, Bang YJ, et al. Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial[J]. Lancet, 2018, 392(10142): 123-33. |
| [8] | Vivier E, Tomasello E, Baratin M, et al. Functions of natural killer cells[J]. Nat Immunol, 2008, 9(5): 503-10. doi:10.1038/ni1582 |
| [9] | Myers JA, Miller JS. Exploring the NK cell platform for cancer immunotherapy[J]. Nat Rev Clin Oncol, 2021, 18(2): 85-100. doi:10.1038/s41571-020-0426-7 |
| [10] | Liu SZ, Galat V, Galat Y, et al. NK cell-based cancer immunotherapy: from basic biology to clinical development[J]. J Hematol Oncol, 2021, 14(1): 7. doi:10.1186/s13045-020-01014-w |
| [11] | Bald T, Krummel MF, Smyth MJ, et al. The NK cell-cancer cycle: advances and new challenges in NK cell-based immunotherapies[J]. Nat Immunol, 2020, 21(8): 835-47. doi:10.1038/s41590-020-0728-z |
| [12] | Li T, Chen ZJ. The cGAS-cGAMP-STING pathway connects DNA damage to inflammation, senescence, and cancer[J]. J Exp Med, 2018, 215(5): 1287-99. doi:10.1084/jem.20180139 |
| [13] | Marcus A, Mao AJ, Lensink-Vasan M, et al. Tumor-derived cGAMP triggers a STING-mediated interferon response in non-tumor cells to activate the NK cell response[J]. Immunity, 2018, 49(4): 754-63.e4. doi:10.1016/j.immuni.2018.09.016 |
| [14] | Gong Y, Germeraad WTV, Zhang XL, et al. NKG2A genetic deletion promotes human primary NK cell anti-tumor responses better than an anti-NKG2A monoclonal antibody[J]. Mol Ther, 2024, 32(8): 2711-27. doi:10.1016/j.ymthe.2024.06.034 |
| [15] | Ablasser A, Schmid-Burgk JL, Hemmerling I, et al. Cell intrinsic immunity spreads to bystander cells via the intercellular transfer of cGAMP[J]. Nature, 2013, 503(7477): 530-4. doi:10.1038/nature12640 |
| [16] | Motwani M, Pesiridis S, Fitzgerald KA. DNA sensing by the cGAS-STING pathway in health and disease[J]. Nat Rev Genet, 2019, 20(11): 657-74. doi:10.1038/s41576-019-0151-1 |
| [17] | Corrales L, McWhirter SM, Dubensky TW Jr, et al. The host STING pathway at the interface of cancer and immunity[J]. J Clin Invest, 2016, 126(7): 2404-11. doi:10.1172/jci86892 |
| [18] | Vivier E, Raulet DH, Moretta A, et al. Innate or adaptive immunity? The example of natural killer cells[J]. Science, 2011, 331(6013): 44-9. doi:10.1126/science.1198687 |
| [19] | Orange JS. Formation and function of the lytic NK-cell immunological synapse[J]. Nat Rev Immunol, 2008, 8(9): 713-25. doi:10.1038/nri2381 |
| [20] | Ashkenazi A. Targeting death and decoy receptors of the tumour-necrosis factor superfamily[J]. Nat Rev Cancer, 2002, 2(6): 420-30. doi:10.1038/nrc821 |
| [21] | 龚 英, 艾丽飞热·艾麦提, 何宗忠. CD39小分子抑制剂ARL67156增强NK细胞对胃癌细胞的杀伤作用[J]. 南方医科大学学报, 2023, 43(12): 2006-14. |
| [22] | Lu L, Yang C, Zhou XY, et al. STING signaling promotes NK cell antitumor immunity and maintains a reservoir of TCF-1+ NK cells[J]. Cell Rep, 2023, 42(9): 113108. doi:10.1016/j.celrep.2023.113108 |
| [23] | Da YY, Liu YX, Hu Y, et al. STING agonist cGAMP enhances anti-tumor activity of CAR-NK cells against pancreatic cancer[J]. Oncoimmunology, 2022, 11(1): 2054105. doi:10.1080/2162402x.2022.2054105 |
| [24] | Zhang LL, Wei XB, Wang ZM, et al. NF-κB activation enhances STING signaling by altering microtubule-mediated STING trafficking[J]. Cell Rep, 2023, 42(3): 112185. doi:10.1016/j.celrep.2023.112185 |
| [25] | Bakhoum SF, Ngo B, Laughney AM, et al. Chromosomal instability drives metastasis through a cytosolic DNA response[J]. Nature, 2018, 553(7689): 467-72. doi:10.1038/nature25432 |
| [26] | Berger G, Knelson EH, Jimenez-Macias JL, et al. STING activation promotes robust immune response and NK cell-mediated tumor regression in glioblastoma models[J]. Proc Natl Acad Sci USA, 2022, 119(28): e2111003119. doi:10.1073/pnas.2111003119 |
| [27] | Wang QW, Bergholz JS, Ding LY, et al. STING agonism reprograms tumor-associated macrophages and overcomes resistance to PARP inhibition in BRCA1-deficient models of breast cancer[J]. Nat Commun, 2022, 13(1): 3022. doi:10.1038/s41467-022-30568-1 |
| [28] | Baird JR, Feng ZP, Xiao HD, et al. STING expression and response to treatment with STING ligands in premalignant and malignant disease[J]. PLoS One, 2017, 12(11): e0187532. doi:10.1371/journal.pone.0187532 |
| [29] | Chen XN, Meng FC, Xu YT, et al. Chemically programmed STING-activating nano-liposomal vesicles improve anticancer immunity[J]. Nat Commun, 2023, 14(1): 4584. doi:10.1038/s41467-023-40312-y |
| [1] | Qiao TANG, Chao ZHOU, Zhaofang BAI, Qing YAO, Simin CHEN, Xinru WEN, Zhaoyun HE, Jin ZHANG, Ruisheng LI, Man GONG. Liangxue Jiedu Huayu Formula improves liver function of mice with acute-on-chronic liver failure by inhibiting excessive activation of the cGAS-STING signaling pathway [J]. Journal of Southern Medical University, 2024, 44(12): 2291-2299. |
| [2] | YU Huilin, LIU Qian, GUO Yongzheng, XIA Yong, LUO Suxin. Palmitic acid suppresses autophagy in neonatal rat cardiomyocytes via the cGAS-STING-IRF3 pathway [J]. Journal of Southern Medical University, 2022, 42(1): 36-44. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||