南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (2): 385-393.doi: 10.12122/j.issn.1673-4254.2026.02.16
• • 上一篇
王秋丽1( ), 胥伟华1, 谷芳秋2, 王思勤3, 何韶衡2, 王君灵2,3(
), 胥伟华1, 谷芳秋2, 王思勤3, 何韶衡2, 王君灵2,3( )
)
收稿日期:2025-08-03
出版日期:2026-02-20
发布日期:2026-03-10
通讯作者:
王君灵
E-mail:18841609323@163.com;wangjuneling@163.com
作者简介:王秋丽,医师,硕士,E-mail: 18841609323@163.com
基金资助:
Qiuli WANG1( ), Weihua XU1, Fangqiu GU2, Siqin WANG3, Shaoheng HE2, Junling WANG2,3(
), Weihua XU1, Fangqiu GU2, Siqin WANG3, Shaoheng HE2, Junling WANG2,3( )
)
Received:2025-08-03
Online:2026-02-20
Published:2026-03-10
Contact:
Junling WANG
E-mail:18841609323@163.com;wangjuneling@163.com
Supported by:摘要:
目的 探讨变应原对变应性鼻炎(AR)患者血液CCR3+粒细胞亚型活化的影响。 方法 流式细胞术检测变应原对AR患者血液粒细胞群CCR3+CD203c-嗜酸性粒细胞和CCR3+CD203c+嗜碱性粒细胞活化分子CD63和CD203c表达水平的影响。多重免疫荧光分析和ELISA技术分别检测志愿者血浆IL-25和TSLP的水平,并分析其与嗜酸性粒细胞和嗜碱性粒细胞数量的相关性。转录组测序AR患者血液CD16-粒细胞,并富集分析其差异表达基因的功能和潜在机制。 结果 AR患者CCR3+粒细胞中嗜酸性粒细胞和嗜碱性粒细胞的比例增加(P<0.005),变应原诱导嗜碱性粒细胞的比例增加(P<0.005)。季节性AR(sAR)和常年性AR(pAR)而非皮肤点刺试验阴性的AR(nAR)患者血液粒细胞群中嗜酸性粒细胞CD63的平均荧光强度表达水平增强(P<0.005),变应原诱导pAR患者嗜酸性粒细胞和嗜碱性粒细胞CD63和CD203c平均荧光强度的表达水平增强(P<0.005)。AR患者升高的血浆IL-25和TSLP水平与增加的嗜酸性粒细胞和嗜碱性粒细胞的数量相关(均P<0.005)。皮肤点刺试验阳性AR患者CD16-粒细胞差异表达的3999个基因在蛋白合成、能量代谢和免疫功能等方面发生了潜在变化,内吞作用、吞噬体和碳代谢等通路可能参与上述变化。 结论 本研究深化了对血液粒细胞群中CCR3+嗜酸性粒细胞和嗜碱性粒细胞参与皮肤点刺试验阳性AR发病机制的理解,揭示了AR治疗的潜在靶点。
王秋丽, 胥伟华, 谷芳秋, 王思勤, 何韶衡, 王君灵. 变应原诱导常年性变应性鼻炎患者血液中CCR3+粒细胞亚群活化[J]. 南方医科大学学报, 2026, 46(2): 385-393.
Qiuli WANG, Weihua XU, Fangqiu GU, Siqin WANG, Shaoheng HE, Junling WANG. Allergens induce activation of blood CCR3+ granulocyte subsets in patients with perennial allergic rhinitis[J]. Journal of Southern Medical University, 2026, 46(2): 385-393.
| Population | Positive allergen | Sample size (n) | Age (year) | Medical history (year) | Onset age (year) |
|---|---|---|---|---|---|
| HC | —— | 30 | 34.5 (23.8-42.0) | —— | —— |
| nAR | —— | 14 | 35.5 (18.0-42.3) | 3.0 (2.0-6.3) | 30.5 (15.3-34.5) |
| sAR | ASE | 16 | 32.0 (24.5-39.8) | 4.0 (3.0-10.0) | 26.5 (13.3-30.8) |
| pAR | ASE+DPE+Others | 24 | 26.0 (19-44.5) | 3.5 (2.0-10.0) | 19.0 (14.0-38.8) |
| DPE+Cockroach+Others | 1 | 9.0 (9.0-9.0) | 6.0 (6.0-6.0) | 3.0 (3.0-3.0) | |
| Cockroach+Others | 4 | 38.5 (14.0-48.0) | 4.0 (1.3-12.8) | 34.0 (12.8-35.8) | |
| In total | 29 | 28.0 (18.5-44.0) | 4.0 (2.0-10.0) | 20.0 (11.5-35.5) | |
| ——: not applicable; Age, medical history and onset age were expressed as Median (P25-P75). HC: Healthy control; nAR: AR with negative skin prick test; sAR: Seasonal AR with positive skin prick test; pAR: Perennial AR with positive skin prick test. | |||||
表1 志愿者的基本信息
Tab.1 Basic information of volunteers
| Population | Positive allergen | Sample size (n) | Age (year) | Medical history (year) | Onset age (year) |
|---|---|---|---|---|---|
| HC | —— | 30 | 34.5 (23.8-42.0) | —— | —— |
| nAR | —— | 14 | 35.5 (18.0-42.3) | 3.0 (2.0-6.3) | 30.5 (15.3-34.5) |
| sAR | ASE | 16 | 32.0 (24.5-39.8) | 4.0 (3.0-10.0) | 26.5 (13.3-30.8) |
| pAR | ASE+DPE+Others | 24 | 26.0 (19-44.5) | 3.5 (2.0-10.0) | 19.0 (14.0-38.8) |
| DPE+Cockroach+Others | 1 | 9.0 (9.0-9.0) | 6.0 (6.0-6.0) | 3.0 (3.0-3.0) | |
| Cockroach+Others | 4 | 38.5 (14.0-48.0) | 4.0 (1.3-12.8) | 34.0 (12.8-35.8) | |
| In total | 29 | 28.0 (18.5-44.0) | 4.0 (2.0-10.0) | 20.0 (11.5-35.5) | |
| ——: not applicable; Age, medical history and onset age were expressed as Median (P25-P75). HC: Healthy control; nAR: AR with negative skin prick test; sAR: Seasonal AR with positive skin prick test; pAR: Perennial AR with positive skin prick test. | |||||
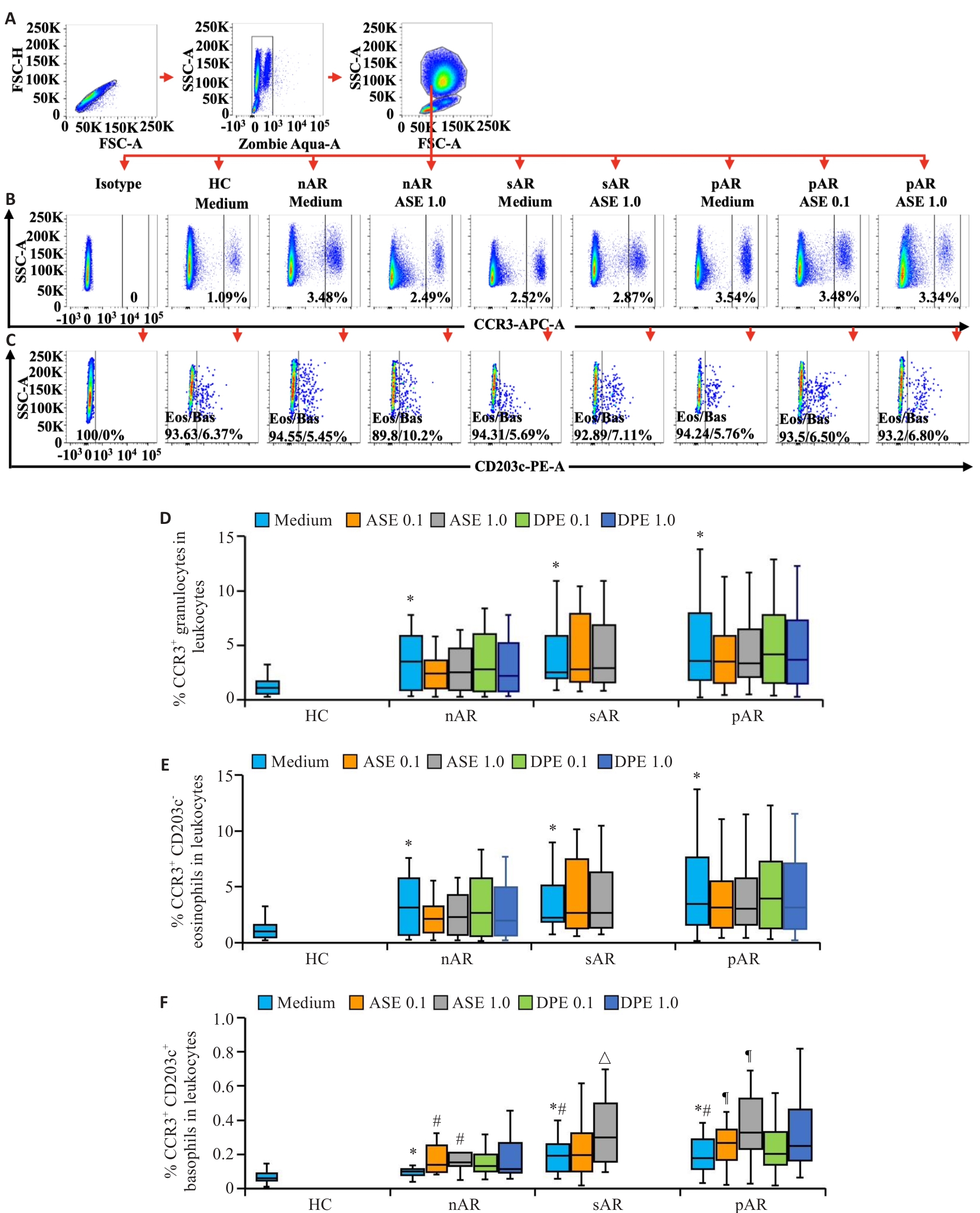
图1 AR患者血液粒细胞群中嗜酸性粒细胞、嗜碱性粒细胞的比例
Fig.1 Ratios of eosinophils and basophils in blood granulocytes of AR patients. A: Flow cytometry gating strategy for human blood granulocytes. B, C: Representative flow cytometry graphs of CCR3+ cells (B), CCR3+CD203c- eosinophils and CCR3+CD203c+ basophils (C) in granulocytes. D-F: Statistical graphs of the expression ratios of CCR3+ cells (D), eosinophils (E), basophils (F) in granulocytes. *P<0.05 vs HC, #P<0.05 vs nAR Medium, △P<0.05 vs sAR Medium, ¶P<0.05 vs pAR Medium.
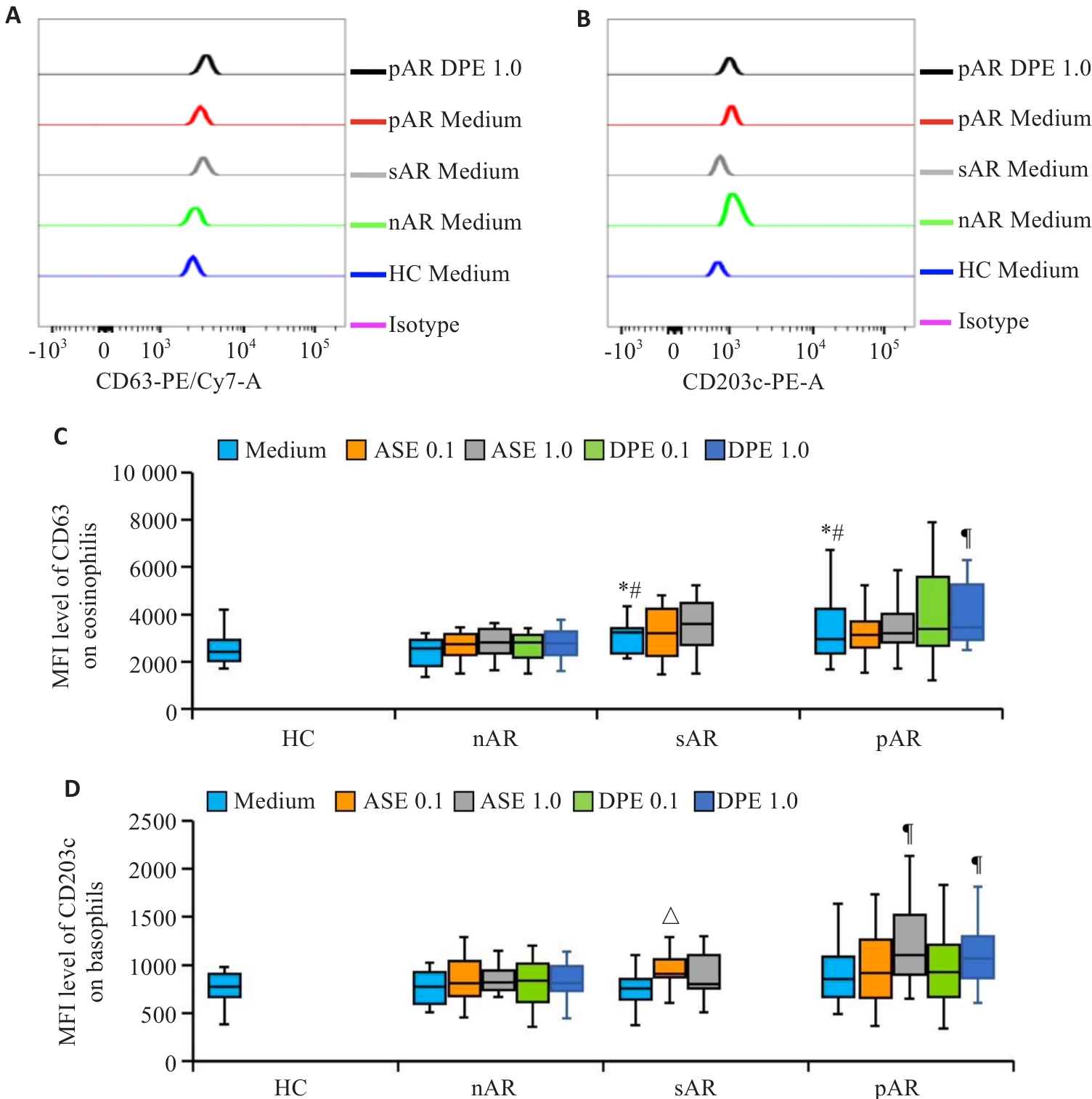
图2 变应原对AR患者血液粒细胞群中嗜酸性粒细胞CD63和嗜碱性粒细胞CD203c MFI表达水平的影响
Fig.2 Effects of allergens on MFI expression levels of CD63 on eosinophils and CD203c on basophils in blood granulocytes of AR patients. A, B: Flow cytometry for analyzing MFI expression levels of CD63 on eosinophils (A) and CD203c on basophils (B) in granulocytes. C, D: Statistical graph of the MFI expression levels of CD63on eosinophils (C) and CD203c on basophils (D). *P<0.05 vs HC, #P<0.05 vs nAR Medium, △P<0.05 vs sAR Medium, ¶P<0.05 vs pAR Medium.
| Group | Compound | IL-25 | TSLP | Eosinophil | Basophil |
|---|---|---|---|---|---|
| HC | IL-25 | —— | -0.343 | 0.165 | -0.056 |
| TSLP | -0.343 | —— | -0.401 | -0.231 | |
| nAR | IL-25 | —— | 0.741⁋ | 0.587⁋ | 0.630⁋ |
| TSLP | 0.741⁋ | —— | 0.582⁋ | 0.669⁋ | |
| sAR | IL-25 | —— | 0.577⁋ | 0.802⁋ | 0.895⁋ |
| TSLP | 0.577⁋ | —— | 0.590⁋ | 0.537⁋ | |
| pAR | IL-25 | —— | 0.772⁋ | 0.880⁋ | 0.601⁋ |
| TSLP | 0.772⁋ | —— | 0.666⁋ | 0.429⁋ | |
| ——: not applicable;⁋P<0.05. | |||||
表2 细胞因子水平与嗜酸性粒细胞和嗜碱性粒细胞数量间Spearman's秩相关系数(R)
Tab.2 Spearman's rank correlation coefficient (R) between cytokine levels and eosinophil and basophil counts in granulocytes
| Group | Compound | IL-25 | TSLP | Eosinophil | Basophil |
|---|---|---|---|---|---|
| HC | IL-25 | —— | -0.343 | 0.165 | -0.056 |
| TSLP | -0.343 | —— | -0.401 | -0.231 | |
| nAR | IL-25 | —— | 0.741⁋ | 0.587⁋ | 0.630⁋ |
| TSLP | 0.741⁋ | —— | 0.582⁋ | 0.669⁋ | |
| sAR | IL-25 | —— | 0.577⁋ | 0.802⁋ | 0.895⁋ |
| TSLP | 0.577⁋ | —— | 0.590⁋ | 0.537⁋ | |
| pAR | IL-25 | —— | 0.772⁋ | 0.880⁋ | 0.601⁋ |
| TSLP | 0.772⁋ | —— | 0.666⁋ | 0.429⁋ | |
| ——: not applicable;⁋P<0.05. | |||||
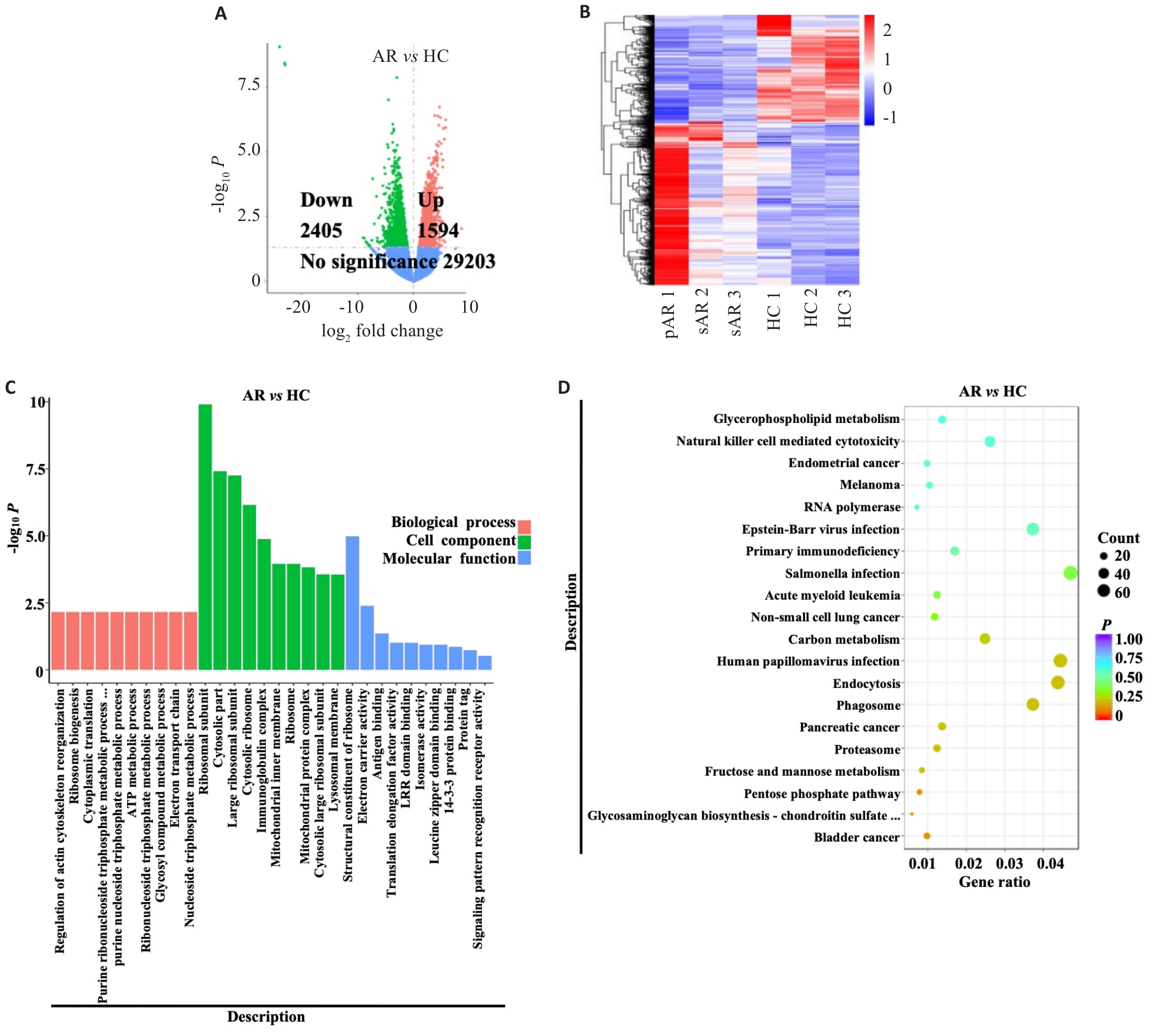
图5 转录组测序AR患者血液CD16-粒细胞
Fig.5 RNA-seq analysis of blood CD16- granulocytes of AR patients. A, B: Volcano plot (A) and cluster heatmap (B) of differential expression genes. C, D: GO (C) and KEGG (D) enrichment diagramsof differential expression genes.
| Protein name | Gene number | Gene name | log2 fold change (AR vs HC) |
|---|---|---|---|
| CCR3 | ENSG00000183625 | CCR3 | 2.55** |
| CD203c | ENSG00000154269 | ENPP3 | -0.44 |
| CD63 | ENSG00000135404 | CD63 | 2.34** |
| **P<0.01vs HC. | |||
表3 转录组分析CD16- 粒细胞中CCR3、CD203c和CD63 mRNA的表达水平
Tab.3 RNA-seq analysis of the expression levels of CCR3, ENPP3 and CD63 mRNA in CD16- granulocytes
| Protein name | Gene number | Gene name | log2 fold change (AR vs HC) |
|---|---|---|---|
| CCR3 | ENSG00000183625 | CCR3 | 2.55** |
| CD203c | ENSG00000154269 | ENPP3 | -0.44 |
| CD63 | ENSG00000135404 | CD63 | 2.34** |
| **P<0.01vs HC. | |||
| [1] | Wise SK, Damask C, Roland LT, et al. International consensus statement on allergy and rhinology: Allergic rhinitis-2023[J]. Int Forum Allergy Rhinol, 2023, 13(4): 293-859. |
| [2] | Milanese M, Ricca V, Canonica GW, et al. Eosinophils, specific hyperreactivity and occurrence of late phase reaction in allergic rhinitis[J]. Eur Ann Allergy Clin Immunol, 2005, 37(1): 7-10. |
| [3] | Xie H, Zhang HY, Chen D, et al. Increased CD123 + HLA-DR- granulocytes in allergic rhinitis and influence of allergens on expression of cell membrane markers[J]. Am J Rhinol Allergy, 2024, 38(5): 294-305. doi:10.1177/19458924241252456 |
| [4] | Xie H, Chen LP, Zhang HY, et al. Increased expressions of CD123, CD63, CD203c, and Fc Epsilon receptor I on blood leukocytes of allergic asthma[J]. Front Mol Biosci, 2022, 9: 907092. doi:10.3389/fmolb.2022.907092 |
| [5] | De Lucca GV. Recent developments in CCR3 antagonists[J]. Curr Opin Drug Discov Devel, 2006, 9(4): 516-24. |
| [6] | Ziegler SF, Roan F, Bell BD, et al. The biology of thymic stromal lymphopoietin (TSLP)[J]. Adv Pharmacol, 2013, 66: 129-55. doi:10.1016/b978-0-12-404717-4.00004-4 |
| [7] | Uguccioni M, MacKay CR, Ochensberger B, et al. High expression of the chemokine receptor CCR3 in human blood basophils. Role in activation by eotaxin, MCP-4, and other chemokines[J]. J Clin Invest, 1997, 100(5): 1137-43. doi:10.1172/jci119624 |
| [8] | Sénéchal S, Fahy O, Gentina T, et al. CCR3-blocking antibody inhibits allergen-induced eosinophil recruitment in human skin xenografts from allergic patients[J]. Lab Invest, 2002, 82(7): 929-39. doi:10.1097/01.lab.0000020417.13757.05 |
| [9] | Carmo LAS, Bonjour K, Ueki S, et al. CD63 is tightly associated with intracellular, secretory events chaperoning piecemeal degranulation and compound exocytosis in human eosinophils[J]. J Leukoc Biol, 2016, 100(2): 391-401. doi:10.1189/jlb.3a1015-480r |
| [10] | De Week AL, Sanz ML, Gamboa PM, et al. Diagnostic tests based on human basophils: more potentials and perspectives than pitfalls. II. technical issues[J]. J Investig Allergol Clin Immunol, 2008, 18(3): 143-55. doi:10.1159/000115885 |
| [11] | Celik GE, Guloglu Karatas D, Aydin O, et al. Diagnostic utility of basophil CD203c expression in β-lactam allergy[J]. Allergy Asthma Proc, 2023, 44(3): 200-7. doi:10.2500/aap.2023.44.230013 |
| [12] | Nakajima S, Kabata H, Kabashima K, et al. Anti-TSLP antibodies: Targeting a master regulator of type 2 immune responses[J]. Allergol Int, 2020, 69(2): 197-203. doi:10.1016/j.alit.2020.01.001 |
| [13] | Hong HY, Liao SM, Chen FH, et al. Role of IL-25, IL-33, and TSLP in triggering united airway diseases toward type 2 inflammation[J]. Allergy, 2020, 75(11): 2794-804. doi:10.1111/all.14526 |
| [14] | Wang YH, Angkasekwinai P, Lu N, et al. IL-25 augments type 2 immune responses by enhancing the expansion and functions of TSLP-DC-activated Th2 memory cells[J]. J Exp Med, 2007, 204(8): 1837-47. doi:10.1084/jem.20070406 |
| [15] | Sokol CL, Barton GM, Farr AG, et al. A mechanism for the initiation of allergen-induced T helper type 2 responses[J]. Nat Immunol, 2008, 9(3): 310-8. doi:10.1038/ni1558 |
| [16] | Bernstein DI, Schwartz G, Bernstein JA. Allergic rhinitis: mechanisms and treatment[J]. Immunol Allergy Clin North Am, 2016, 36(2): 261-78. doi:10.1016/j.iac.2015.12.004 |
| [17] | Eifan AO, Durham SR. Pathogenesis of rhinitis[J]. Clin Exp Allergy, 2016, 46(9): 1139-51. doi:10.1111/cea.12780 |
| [18] | 中华耳鼻咽喉头颈外科杂志编辑委员会鼻科组, 中华医学会耳鼻咽喉头颈外科学分会鼻科学组.中国变应性鼻炎诊断和治疗指南(2022年, 修订版)[J].中华耳鼻咽喉头颈外科杂志, 2022, 57(2): 106-29. |
| [19] | Jeffery PK, Haahtela T. Allergic rhinitis and asthma: inflammation in a one-airway condition[J]. BMC Pulm Med, 2006, 6(): S5. doi:10.1186/1471-2466-6-s1-s5 |
| [20] | Choi EA, Nah G, Chang WS, et al. Blood eosinophil related to maternal allergic rhinitis is associated with the incidence of allergic rhinitis in offspring: COCOA study[J]. BMC Pediatr, 2023, 23(1): 343. doi:10.1186/s12887-023-04156-1 |
| [21] | Linden M, Svensson C, Andersson M, et al. Circulating eosinophil/basophil progenitors and nasal mucosal cytokines in seasonal allergic rhinitis[J]. Allergy, 1999, 54(3): 212-9. doi:10.1034/j.1398-9995.1999.00756.x |
| [22] | Broide DH. Allergic rhinitis: pathophysiology[J]. Allergy Asthma Proc, 2010, 31(5): 370-4. doi:10.2500/aap.2010.31.3388 |
| [23] | Saito H, Howie K, Wattie J, et al. Allergen-induced murine upper airway inflammation: local and systemic changes in murine experimental allergic rhinitis[J]. Immunology, 2001, 104(2): 226-34. doi:10.1046/j.1365-2567.2001.01253.x |
| [24] | Shamji MH, Bellido V, Scadding GW, et al. Effector cell signature in peripheral blood following nasal allergen challenge in grass pollen allergic individuals[J]. Allergy, 2015, 70(2): 171-9. doi:10.1111/all.12543 |
| [25] | Wilson AM, Duong M, Crawford L, et al. An evaluation of peripheral blood eosinophil/basophil progenitors following nasal allergen challenge in patients with allergic rhinitis[J]. Clin Exp Allergy, 2005, 35(1): 39-44. doi:10.1111/j.1365-2222.2004.02072.x |
| [26] | Stone KD, Prussin C, Metcalfe DD. IgE, mast cells, basophils, and eosinophils[J]. J Allergy Clin Immunol, 2010, 125(2 ): S73-80. doi:10.1016/j.jaci.2009.11.017 |
| [27] | Gao YQ, Zhou JN, Wang ML, et al. Characteristics of upper respiratory tract rhinovirus in children with allergic rhinitis and its role in disease severity[J]. Microbiol Spectr, 2024, 12(7): e0385323. doi:10.1128/spectrum.03853-23 |
| [28] | Tyurin YA, Lissovskaya SA, Fassahov RS, et al. Cytokine profile of patients with allergic rhinitis caused by pollen, mite, and microbial allergen sensitization[J]. J Immunol Res, 2017, 2017: 3054217. doi:10.1155/2017/3054217 |
| [29] | Eggel A, Pennington LF, Jardetzky TS. Therapeutic monoclonal antibodies in allergy: targeting IgE, cytokine, and alarmin pathways[J]. Immunol Rev, 2024, 328(1): 387-411. doi:10.1111/imr.13380 |
| [30] | Andreasson LM, Dyhre-Petersen N, Hvidtfeldt M, et al. Airway hyperresponsiveness correlates with airway TSLP in asthma independent of eosinophilic inflammation[J]. J Allergy Clin Immunol, 2024, 153(4): 988-97. e11. doi:10.1016/j.jaci.2023.11.915 |
| [31] | Wang JL, Zhan MM, Gu FQ, et al. Diagnostic use of CCR3, CD63, CD203c and FcεRIα on blood leukocytes of allergic asthma and combined allergic rhinitis and asthma syndrome[J]. J Cell Mol Med, 2025, 29(12): e70594. doi:10.1111/jcmm.70594 |
| [32] | Thurau AM, Schylz U, Wolf V, et al. Identification of eosinophils by flow cytometry[J]. Cytometry, 1996, 23(2): 150-8. doi:10.1002/(sici)1097-0320(19960201)23:2<150::aid-cyto8>3.0.co;2-o |
| [33] | Isogai S, Hayashi M, Yamamoto N, et al. Upregulation of CD11b on eosinophils in aspirin induced asthma[J]. Allergol Int, 2013, 62(3): 367-73. doi:10.2332/allergolint.12-oa-0499 |
| [34] | Agis H, Füreder W, Bankl HC, et al. Comparative immuno-phenotypic analysis of human mast cells, blood basophils and monocytes[J]. Immunology, 1996, 87(4): 535-43. doi:10.1046/j.1365-2567.1996.493578.x |
| [35] | Papavasileiou S, Mo J, Boey D, et al. Single-cell omics-based characterization of human basophils reveals two transcriptionally distinct populations [J]. bioRxiv, 2025, 2025: 639059. doi:10.1111/all.70209 |
| [36] | Gibbs BF, Papenfuss K, Falcone FH. A rapid two-step procedure for the purification of human peripheral blood basophils to near homogeneity[J]. Clin Exp Allergy, 2008, 38(3): 480-5. doi:10.1111/j.1365-2222.2007.02919.x |
| [37] | 王君灵, 张慧云, 何韶衡. 人嗜碱性粒细胞胞膜蛋白在过敏诊断中的研究进展[J]. 中华临床免疫和变态反应杂志, 2024, 18(1): 88-95. |
| [38] | Gray N, Limberg MM, Wiebe D, et al. Differential upregulation and functional activity of S1PR1 in human peripheral blood basophils of atopic patients[J]. Int J Mol Sci, 2022, 23(24): 16117. doi:10.3390/ijms232416117 |
| [39] | Tsai SH, Takeda K. Regulation of allergic inflammation by the ectoenzyme E-NPP3 (CD203c) on basophils and mast cells[J]. Semin Immunopathol, 2016, 38(5): 571-9. doi:10.1007/s00281-016-0564-2 |
| [40] | Gachanja NN, Dorward DA, Rossi AG, et al. Assays of eosinophil apoptosis and phagocytic uptake[J]. Methods Mol Biol, 2021, 2241: 113-32. doi:10.1007/978-1-0716-1095-4_10 |
| [41] | Davies LC, Rice CM, McVicar DW, et al. Diversity and environmental adaptation of phagocytic cell metabolism[J]. J Leukoc Biol, 2019, 105(1): 37-48. doi:10.1002/jlb.4ri0518-195r |
| [42] | Schuijs MJ, Hammad H, Lambrecht BN. Professional and 'amateur' antigen-presenting cells in type 2 immunity[J]. Trends Immunol, 2019, 40(1): 22-34. doi:10.1016/j.it.2018.11.001 |
| [43] | Wu ZK, Liang XQ, Li MK, et al. Advances in the optimization of central carbon metabolism in metabolic engineering[J]. Microb Cell Fact, 2023, 22(1): 76. doi:10.1186/s12934-023-02090-6 |
| [1] | 赵锦燕, 彭娇, 林明和, 朱晓勤, 黄彬, 林久茂. 清解扶正颗粒通过抑制线粒体依赖的凋亡、激活AMPK-PGC-1α通路缓解5-氟尿嘧啶引起的骨骼肌损伤[J]. 南方医科大学学报, 2026, 46(1): 94-103. |
| [2] | 罗嘉纯, Sodnomjamts Batzaya, 高雪锋, 陈晶宇, 余政颖, 熊莎莎, 曹虹. Akkermansia muciniphila改善gp120转基因小鼠的肠-脑相互作用障碍[J]. 南方医科大学学报, 2025, 45(3): 554-565. |
| [3] | 姚宏, 刘珂娣, 刘澄曌, 李伟红, 代旗, 赵石, 丁子恒, 王鹤霏, 葛晓静, 卫培峰, 段佳林, 奚苗苗. 五谷虫通过抑制免疫应激-补体活化缓解咪喹莫特诱导的小鼠银屑病样皮肤损伤[J]. 南方医科大学学报, 2024, 44(11): 2121-2130. |
| [4] | 王 敏, 张 茜, 徐桂铃, 黄淑榆, 赵文驱, 梁健鹏, 黄俊文, 蔡绍曦, 赵海金. 健康人群和慢性阻塞性肺疾病患者的血清维生素D与血嗜酸性粒细胞计数的关系[J]. 南方医科大学学报, 2023, 43(5): 727-732. |
| [5] | 马金苗, 顼志兵, 朱 杰, 刘迎庆, 王青青, 奚希相, 张莉芬, 黄 静, 孝玲玲. 附萸汤改善心力衰竭大鼠的心室重构:基于抑制AMPK/mTOR通路介导的细胞自噬[J]. 南方医科大学学报, 2023, 43(3): 466-473. |
| [6] | 孙 柳, 焦沃尔, 孔勇刚, 杨长亮, 续 珊, 谯月龙, 陈始明. GATA3+Treg细胞在变应性鼻炎中的表达及意义[J]. 南方医科大学学报, 2023, 43(2): 280-286. |
| [7] | 杨金水, 万月华, 张江林, 朱 剑. 45例嗜酸性筋膜炎的临床特征、超声诊断及治疗随访的单中心回顾性研究[J]. 南方医科大学学报, 2023, 43(1): 145-152. |
| [8] | 杨淑銮, 赵文驱, 彭显如, 蓝紫涵, 黄俊文, 韩慧珊, 陈 颖, 蔡绍曦, 赵海金. 抑制TAK1可加重甲苯二异氰酸酯诱导的哮喘小鼠气道炎症[J]. 南方医科大学学报, 2022, 42(2): 181-189. |
| [9] | 刘 泽, 尤 达, 李 勇, 何咏梅, 李阿芳, 李 潘, 李春艳. Numb通过上调V1G1的表达激活近端肾小管细胞mTORC1信号通路[J]. 南方医科大学学报, 2022, 42(10): 1462-1469. |
| [10] | 徐 俊, 史嘉炜, 蔡欣玲, 黄盛娜, 李 刚, 徐 燕. 富心方通过调控c-Fos-NR4A1-p38通路改善人动脉血管内皮细胞缺氧引起的损伤[J]. 南方医科大学学报, 2021, 41(2): 200-209. |
| [11] | 王瑞茵,林江涛,王静茹,李春晓. 青蒿琥酯对哮喘小鼠嗜酸性粒细胞凋亡和Fas、Bcl-2基因蛋白 表达的影响[J]. 南方医科大学学报, 2020, 40(01): 93-98. |
| [12] | 熊传锋,齐杰莹,邓 蓉,谢丽芬,李长征,聂晓莉. 白芍总苷抑制小鼠T淋巴细胞体外增殖促进活化诱导细胞死亡[J]. 南方医科大学学报, 2020, 40(01): 118-124. |
| [13] | 金健,金大地. 利塞膦酸钠抑制大鼠骨髓内脂肪细胞分化及脂肪细胞核因子κB受体活化因子配体蛋白的表达[J]. 南方医科大学学报, 2019, 39(08): 987-. |
| [14] | 王万林,毛春,肖娟. 人重组蛋白PDCD5抑制胶原诱导性关节炎大鼠来源的淋巴细胞增殖和炎性细胞因子分泌并促进活化的 淋巴细胞凋亡[J]. 南方医科大学学报, 2019, 39(06): 627-. |
| [15] | 夏珺,郑明月,李灵杰,侯旭峰,曾位森. 共轭亚油酸改善肥胖糖尿病小鼠的糖脂代谢[J]. 南方医科大学学报, 2019, 39(06): 740-. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||