南方医科大学学报 ›› 2025, Vol. 45 ›› Issue (4): 853-861.doi: 10.12122/j.issn.1673-4254.2025.04.21
张毅1,2( ), 沈昱1,2, 万志强1,2, 陶嵩1, 柳亚魁1, 王栓虎1(
), 沈昱1,2, 万志强1,2, 陶嵩1, 柳亚魁1, 王栓虎1( )
)
收稿日期:2024-09-26
出版日期:2025-04-20
发布日期:2025-04-28
通讯作者:
王栓虎
E-mail:2826348547@qq.com;knight01030103@126.com
作者简介:张 毅,在读硕士研究生,E-mail: 2826348547@qq.com
基金资助:
Yi ZHANG1,2( ), Yu SHEN1,2, Zhiqiang WAN1,2, Song TAO1, Yakui LIU1, Shuanhu WANG1(
), Yu SHEN1,2, Zhiqiang WAN1,2, Song TAO1, Yakui LIU1, Shuanhu WANG1( )
)
Received:2024-09-26
Online:2025-04-20
Published:2025-04-28
Contact:
Shuanhu WANG
E-mail:2826348547@qq.com;knight01030103@126.com
摘要:
目的 探究周期蛋白依赖性激酶抑制因子3(CDKN3)在胃癌中的表达情况,并分析其影响胃癌患者预后的潜在作用机制。 方法 纳入114例胃癌患者资料,分析CDKN3在胃癌组织中表达水平及其对胃癌患者术后5年生存率的影响;GO及KEGG富集分析预测CDKN3的生物学功能及可能的作用机制;通过慢病毒转染技术干预CDKN3的表达。采用Transwell、CCK-8、TUNEL染色、流式细胞术及Western blotting验证生物学功能。 结果 CDKN3在胃癌组织的表达水平显著高于癌旁组织(P<0.01),且CDKN3的表达水平与CEA、CA19-9、T分期及N分期相关(P<0.05)。单因素联合多因素分析显示,CDKN3高表达是影响胃癌患者术后5年生存率的独立风险因子(P<0.05)且CDKN3高表达对胃癌患者远期预后具有预判价值(P<0.01)。生物信息学富集分析提示CDKN3的功能可能与凋亡有关。Transwell实验结果显示,下调CDKN3可抑制胃癌细胞迁移、侵袭,上调则反之(P<0.01)。TUNEL染色结果显示,癌旁组织中细胞凋亡水平高于胃癌组织(P<0.01)。CCK-8和流式结果显示,降低CDKN3表达可抑制胃癌细胞的增殖,升高胃癌细胞凋亡率(P<0.05)。Western blotting结果显示过表达CDKN3后,p53、p21和促凋亡蛋白Bax表达降低,p-p65和抗凋亡蛋白Bcl-2表达升高(P<0.05)。 结论 CDKN3在胃癌组织中高表达并影响患者预后,其可能通过p53/NF-κB信号通路调控胃癌细胞凋亡并抑制其增殖、侵袭与迁移有关。
张毅, 沈昱, 万志强, 陶嵩, 柳亚魁, 王栓虎. CDKN3高表达促进胃癌细胞的迁移和侵袭:基于调控p53/NF-κB信号通路和抑制胃癌细胞凋亡[J]. 南方医科大学学报, 2025, 45(4): 853-861.
Yi ZHANG, Yu SHEN, Zhiqiang WAN, Song TAO, Yakui LIU, Shuanhu WANG. High expression of CDKN3 promotes migration and invasion of gastric cancer cells by regulating the p53/NF-κB signaling pathway and inhibiting cell apoptosis[J]. Journal of Southern Medical University, 2025, 45(4): 853-861.
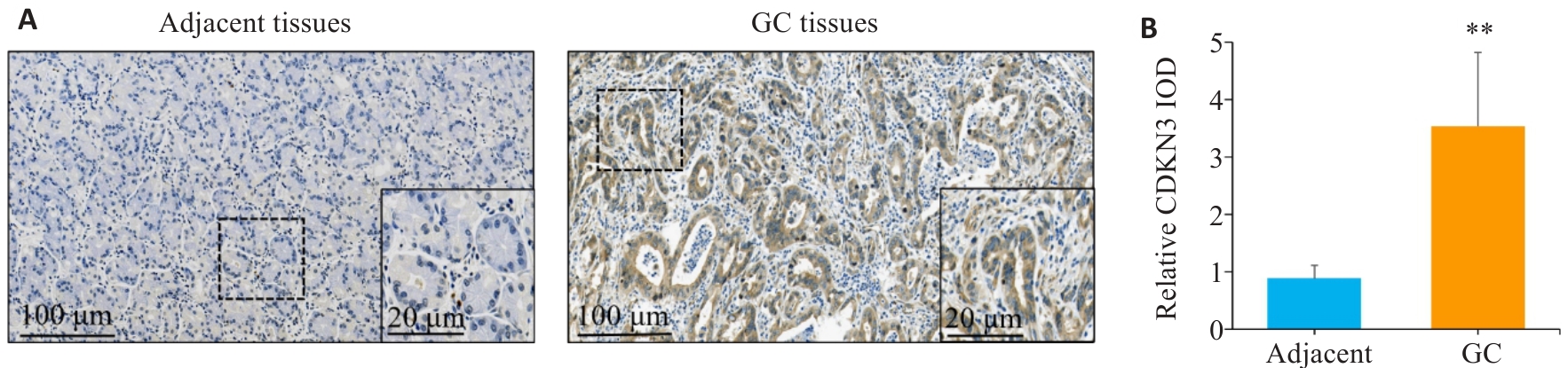
图1 CDKN3在胃癌及癌旁组织中的表达情况
Fig.1 Expression of CDKN3 in gastric cancer (GC) and adjacent tissues. A: Immunohistochemistry of CDKN3 in gastric cancer (GC) and adjacent tissues. B: Relative IOD values of CDKN3 expression. **P<0.01.
| Clinicopatholological parameters | n | CDKN3 | χ2 | P | |
|---|---|---|---|---|---|
| Low expression (n=57) | High expression (n=57) | ||||
| Gender | |||||
| Male | 81 | 43 (53.09%) | 38 (46.91%) | 1.066 | 0.302 |
| Female | 33 | 14 (42.42%) | 19 (57.58%) | ||
| Age (year) | |||||
| <60 | 46 | 22 (47.83%) | 24 (52.17%) | 0.146 | 0.703 |
| ≥60 | 68 | 35 (51.47%) | 33 (48.53%) | ||
| CEA (μg/L) | |||||
| <5 | 69 | 46 (66.67%) | 23 (33.33%) | 19.422 | <0.001 |
| ≥5 | 45 | 11 (24.44%) | 34 (75.56%) | ||
| CA19-9 (kU/L) | |||||
| <37 | 86 | 53 (61.63%) | 33 (38.37%) | 18.937 | <0.001 |
| ≥37 | 28 | 4 (14.29%) | 24 (85.71%) | ||
| Tumor size (cm) | |||||
| <5 | 67 | 38 (56.72%) | 29 (43.28%) | 2.932 | 0.087 |
| ≥5 | 47 | 19 (40.43%) | 28 (59.57%) | ||
| Histological type | |||||
| Adenocarcinoma | 104 | 53 (50.96%) | 51 (49.04%) | 0.438 | 0.508 |
| Other | 10 | 4 (40.00%) | 6 (60.00%) | ||
| Grade of differentiation | |||||
| Well | 10 | 4 (40.00%) | 6 (60.00%) | 4.001 | 0.135 |
| Moderate | 38 | 24 (63.16%) | 14 (36.84%) | ||
| Poor | 66 | 29 (43.94%) | 37 (56.06%) | ||
| T Stage | |||||
| T1-T2 | 37 | 24 (64.86%) | 13 (35.14%) | 4.842 | 0.028 |
| T3-T4 | 77 | 33 (42.86%) | 44 (57.14%) | ||
| N Stage | |||||
| N0-N1 | 71 | 41 (57.75%) | 30 (42.25%) | 4.518 | 0.034 |
| N2-N3 | 43 | 16 (37.21%) | 27 (62.79%) | ||
表1 CDKN3表达量与胃癌患者临床病理参数间的关系
Tab.1 Relationship between the expression level of CDKN3 in GC tissues and clinicopathological parameters of the patients
| Clinicopatholological parameters | n | CDKN3 | χ2 | P | |
|---|---|---|---|---|---|
| Low expression (n=57) | High expression (n=57) | ||||
| Gender | |||||
| Male | 81 | 43 (53.09%) | 38 (46.91%) | 1.066 | 0.302 |
| Female | 33 | 14 (42.42%) | 19 (57.58%) | ||
| Age (year) | |||||
| <60 | 46 | 22 (47.83%) | 24 (52.17%) | 0.146 | 0.703 |
| ≥60 | 68 | 35 (51.47%) | 33 (48.53%) | ||
| CEA (μg/L) | |||||
| <5 | 69 | 46 (66.67%) | 23 (33.33%) | 19.422 | <0.001 |
| ≥5 | 45 | 11 (24.44%) | 34 (75.56%) | ||
| CA19-9 (kU/L) | |||||
| <37 | 86 | 53 (61.63%) | 33 (38.37%) | 18.937 | <0.001 |
| ≥37 | 28 | 4 (14.29%) | 24 (85.71%) | ||
| Tumor size (cm) | |||||
| <5 | 67 | 38 (56.72%) | 29 (43.28%) | 2.932 | 0.087 |
| ≥5 | 47 | 19 (40.43%) | 28 (59.57%) | ||
| Histological type | |||||
| Adenocarcinoma | 104 | 53 (50.96%) | 51 (49.04%) | 0.438 | 0.508 |
| Other | 10 | 4 (40.00%) | 6 (60.00%) | ||
| Grade of differentiation | |||||
| Well | 10 | 4 (40.00%) | 6 (60.00%) | 4.001 | 0.135 |
| Moderate | 38 | 24 (63.16%) | 14 (36.84%) | ||
| Poor | 66 | 29 (43.94%) | 37 (56.06%) | ||
| T Stage | |||||
| T1-T2 | 37 | 24 (64.86%) | 13 (35.14%) | 4.842 | 0.028 |
| T3-T4 | 77 | 33 (42.86%) | 44 (57.14%) | ||
| N Stage | |||||
| N0-N1 | 71 | 41 (57.75%) | 30 (42.25%) | 4.518 | 0.034 |
| N2-N3 | 43 | 16 (37.21%) | 27 (62.79%) | ||
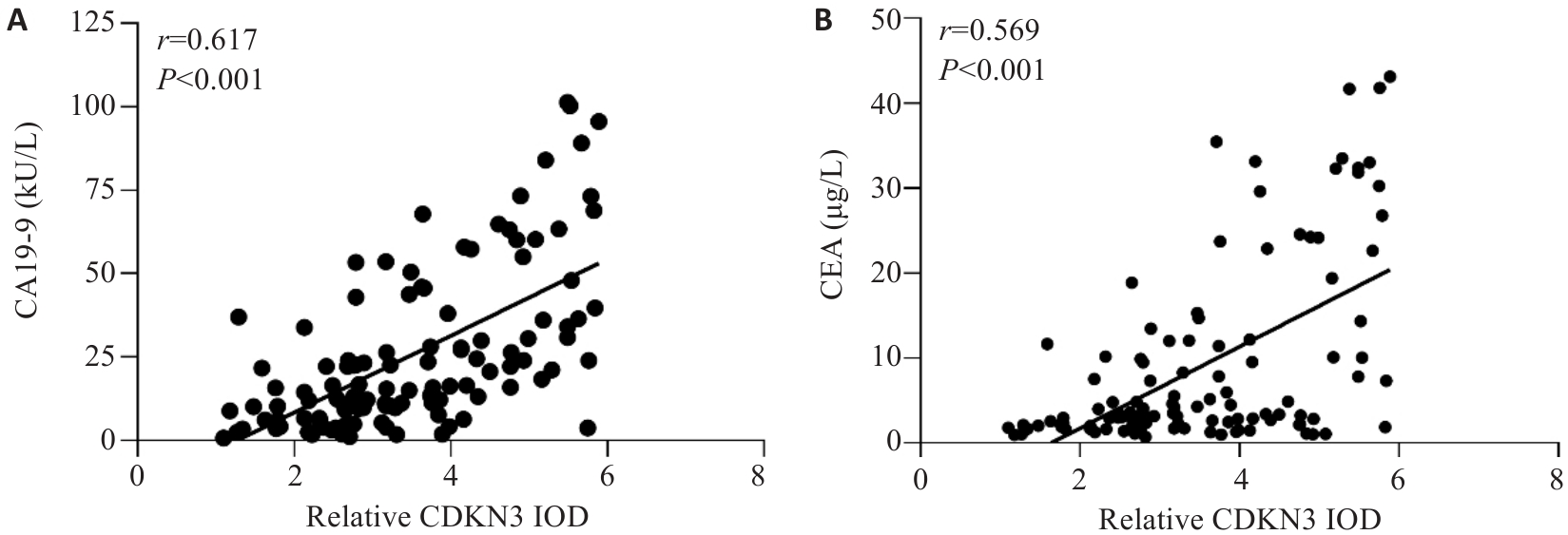
图2 胃癌组织中CDKN3表达水平与CA19-9及CEA的相关性分析
Fig.2 Correlation analysis between the expression level of CDKN3 in GC and the levels of CA19-9 and CEA in peripheral blood. A: Correlation analysis between CDKN3 and CA19-9. B: Correlation analysis between CDKN3 and CEA.
| Factors | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| Log-rank χ2 | P | HR | 95% CI | P | ||
| Gender (female vs male) | 0.108 | 0.742 | - | - | - | |
| Age (≥60 years vs <60 years) | 0.162 | 0.687 | - | - | - | |
| CDKN3 expression (high vs low) | 34.036 | <0.001 | 2.819 | 1.396-5.692 | 0.004 | |
| CEA(≥5 μg/L vs <5 μg/L) | 20.961 | <0.001 | 1.954 | 1.084-3.522 | 0.026 | |
| CA19-9 (≥37 kU/L vs <37 kU/L) | 22.695 | <0.001 | 1.847 | 1.048-3.257 | 0.034 | |
| Tumor size (≥5 cm vs <5 cm) | 6.139 | 0.013 | 0.978 | 0.552-1.730 | 0.938 | |
| Histological type (other vs adenocarcinoma) | 0.309 | 0.578 | - | - | - | |
| Grade of differentiation (well vs moderate vs poor) | 5.623 | 0.060 | - | - | - | |
| T Stage (T3-T4vs T1-T2) | 13.586 | <0.001 | 2.438 | 1.160-5.120 | 0.019 | |
| N Stage (N2-N3vs N0-N1) | 20.178 | <0.001 | 2.099 | 1.177-3.744 | 0.012 | |
表2 影响胃癌根治术后5年生存率的单因素及多因素分析
Tab.2 Univariate and multivariate analyses of the factors affecting the 5-year survival rate after radical gastrectomy for gastric cancer patients
| Factors | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| Log-rank χ2 | P | HR | 95% CI | P | ||
| Gender (female vs male) | 0.108 | 0.742 | - | - | - | |
| Age (≥60 years vs <60 years) | 0.162 | 0.687 | - | - | - | |
| CDKN3 expression (high vs low) | 34.036 | <0.001 | 2.819 | 1.396-5.692 | 0.004 | |
| CEA(≥5 μg/L vs <5 μg/L) | 20.961 | <0.001 | 1.954 | 1.084-3.522 | 0.026 | |
| CA19-9 (≥37 kU/L vs <37 kU/L) | 22.695 | <0.001 | 1.847 | 1.048-3.257 | 0.034 | |
| Tumor size (≥5 cm vs <5 cm) | 6.139 | 0.013 | 0.978 | 0.552-1.730 | 0.938 | |
| Histological type (other vs adenocarcinoma) | 0.309 | 0.578 | - | - | - | |
| Grade of differentiation (well vs moderate vs poor) | 5.623 | 0.060 | - | - | - | |
| T Stage (T3-T4vs T1-T2) | 13.586 | <0.001 | 2.438 | 1.160-5.120 | 0.019 | |
| N Stage (N2-N3vs N0-N1) | 20.178 | <0.001 | 2.099 | 1.177-3.744 | 0.012 | |
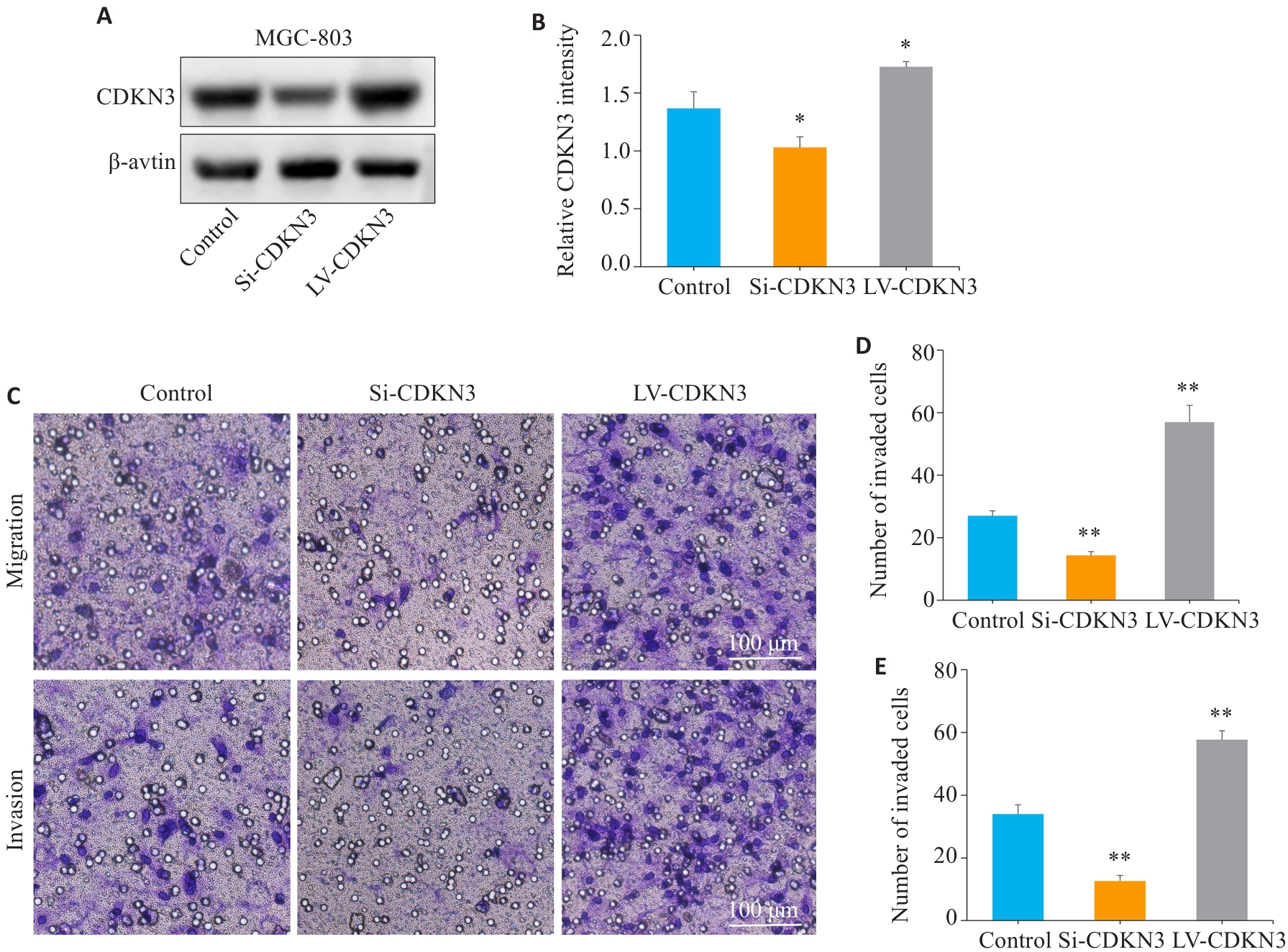
图5 CDKN3促进胃癌细胞的迁移与侵袭
Fig.5 CDKN3 overexpression promotes gastric cancer cell migration and invasion. A,B: The expression of CDKN3 protein after lentivirus transfection was detected by Western blotting. C:Transwell experiment for analyzing the impact of CDKN3 on the migration and invasion abilities of MGC803 cells. D: Quantitative analysis of the number of migrated cells. E: Quantitative analysis of the number of invaded cells. *P<0.05, **P<0.01 vs control.
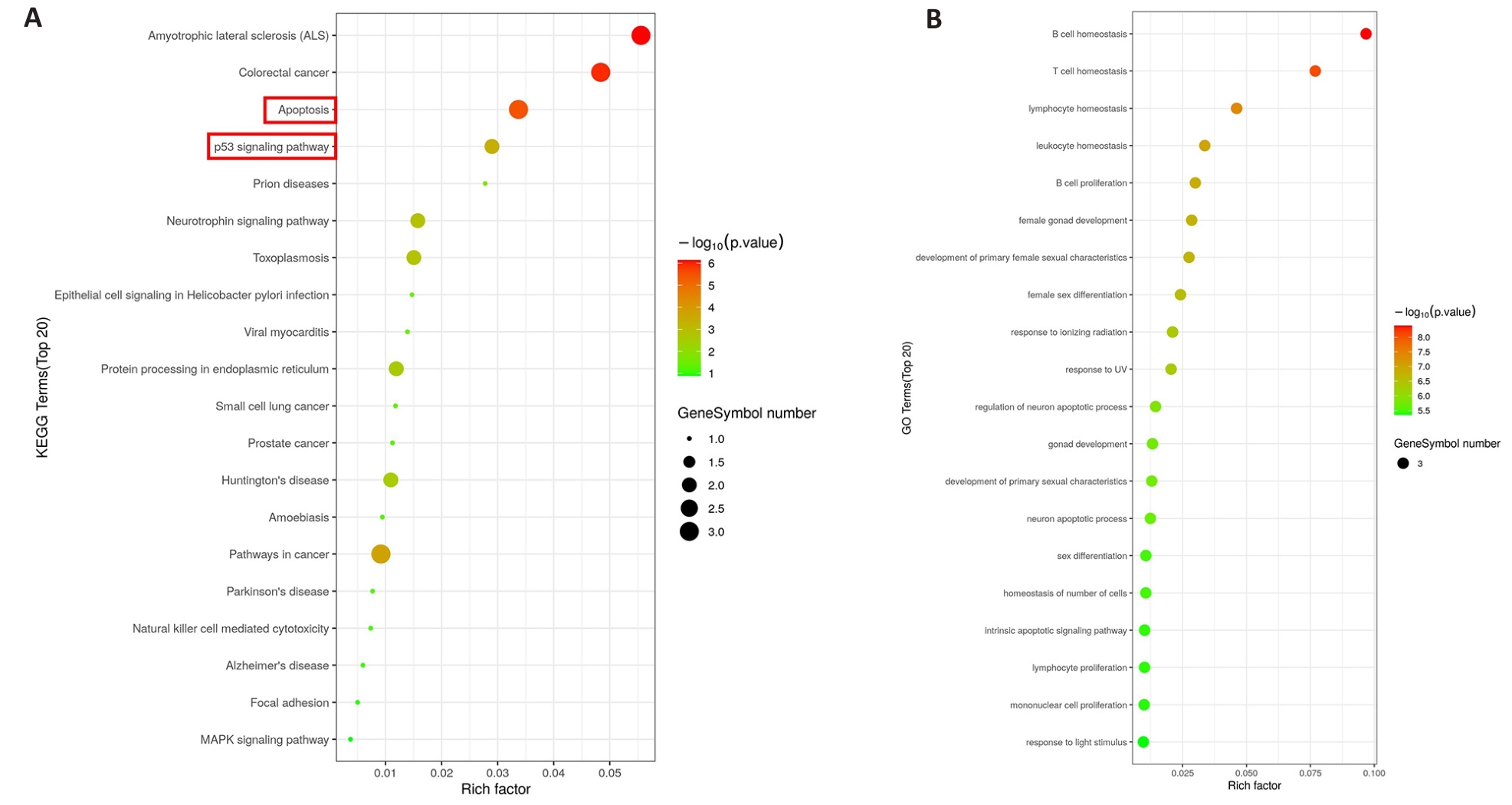
图6 CDKN3的KEGG和GO富集分析
Fig.6 KEGG and GO enrichment analysis of CDKN3. A: KEGG analysis of CDKN3 in gastric cancer. B: GO analysis of CDKN3 in gastric cancer.

图7 TUNEL染色检测胃癌及癌旁组织中细胞凋亡情况
Fig.7 TUNEL staining was used to detect cell apoptosis in gastric cancer and adjacent tissues. A,B: TUNEL staining and scoring of apoptosis in gastric cancer tissues sections and adjacent tissues sections. **P<0.01vs gastric cancer. GC: Gastric cancer.
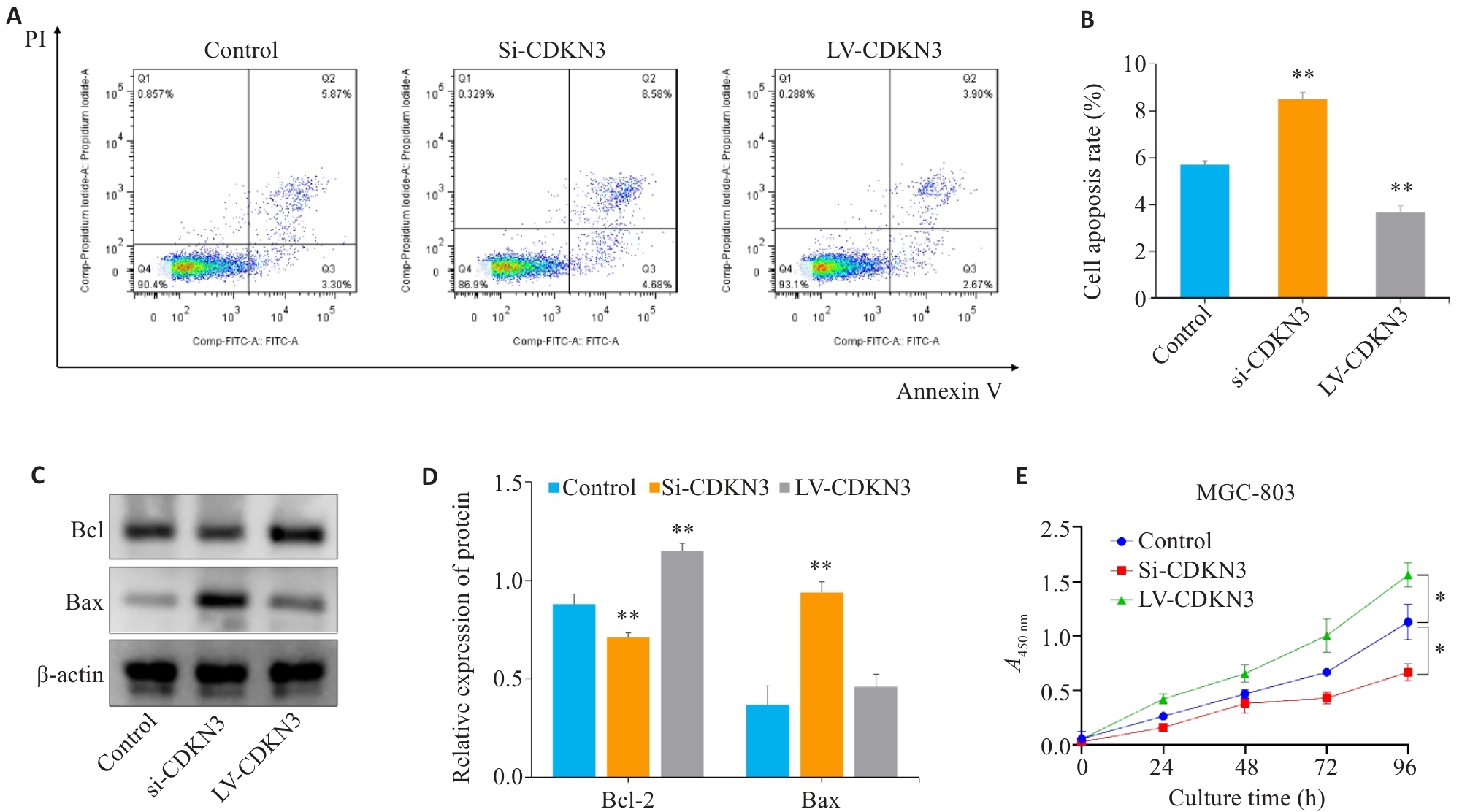
图8 CDKN3对胃癌细胞凋亡和增殖的影响
Fig.8 Effect of CDKN3 on apoptosis and proliferation of MGC803 cells. A,B: Cell apoptosis rate detected by flow cytometry after lentivirus transfection. C: Expression of Bcl-2 and Bax in MGC803 cells. D: Quantitative analysis of Bcl-2/Bax ratio. E: Effect of CDKN3 on proliferation of MGC-803 cells. *P<0.05, **P<0.01 vs control.
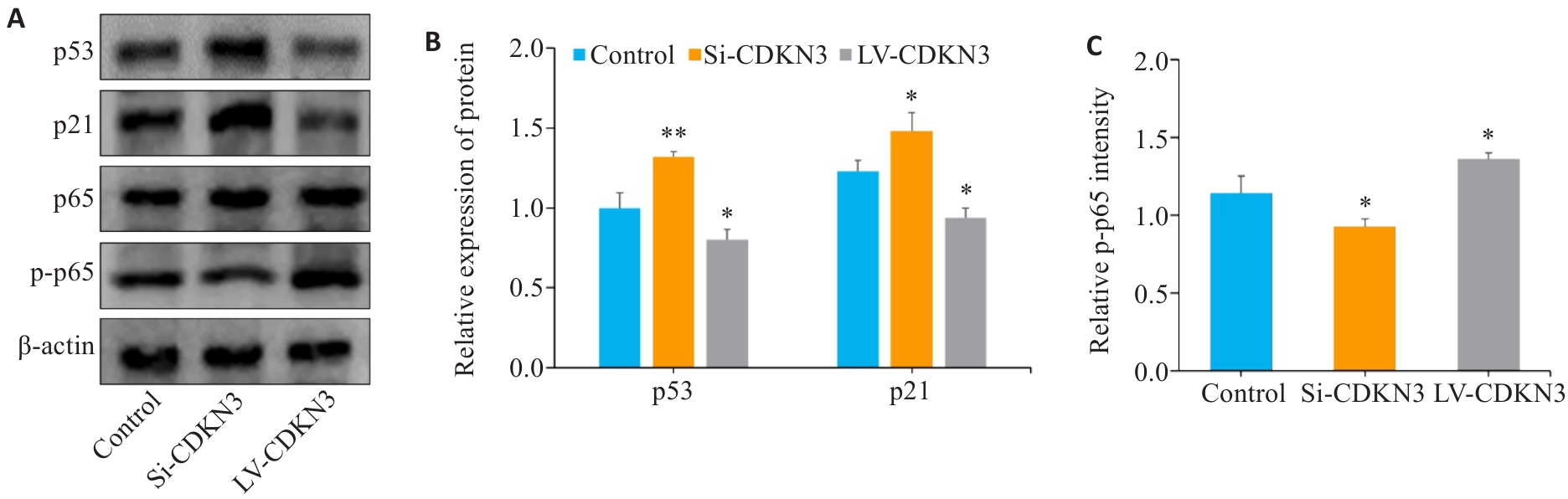
图9 CDKN3对胃癌细胞p53/NF-κB信号通路的影响
Fig.9 Effect of CDKN3 on p53/NF-κB signaling in MGC803 cells. A: Expression of p53, p21, p65 and p-p65 in MGC803 cells. B, C: Quantitative analysis of p53, p21, p65 and p-p65 ratio. *P<0.05, **P<0.01 vs control.
| 1 | Yang L, Ying X, Liu S, et al. Gastric cancer: Epidemiology, risk factors and prevention strategies[J]. Chin J Cancer Res, 2020, 32(6): 695-704. |
| 2 | Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2024, 74(3): 229-63. |
| 3 | Machlowska J, Baj J, Sitarz M, et al. Gastric cancer: epidemiology, risk factors, classification, genomic characteristics and treatment strategies[J]. Int J Mol Sci, 2020, 21(11): E4012. |
| 4 | Thrift AP, Wenker TN, El-Serag HB. Global burden of gastric cancer: epidemiological trends, risk factors, screening and prevention[J]. Nat Rev Clin Oncol, 2023, 20(5): 338-49. |
| 5 | Li WH, Zhang L, Wu YH. CDKN3 regulates cisplatin resistance to colorectal cancer through TIPE1[J]. Eur Rev Med Pharmacol Sci, 2020, 24(7): 3614-23. |
| 6 | Dai W, Miao H, Fang S, et al. CDKN3 expression is negatively associated with pathological tumor stage and CDKN3 inhibition promotes cell survival in hepatocellular carcinoma[J]. Mol Med Rep, 2016, 14(2): 1509-14. |
| 7 | Dai W, Fang S, Cai G, et al. CDKN3 expression predicates poor prognosis and regulates adriamycin sensitivity in hepatocellular carcinoma in vitro [J]. J Int Med Res, 2020, 48(7): 300060520936879. |
| 8 | Chang SL, Chen TJ, Lee YE, et al. CDKN3 expression is an independent prognostic factor and associated with advanced tumor stage in nasopharyngeal carcinoma[J]. Int J Med Sci, 2018, 15(10): 992-8. |
| 9 | Wang J, Che W, Wang W, et al. CDKN3 promotes tumor progression and confers cisplatin resistance via RAD51 in esophageal cancer[J]. Cancer Manag Res, 2019, 11: 3253-64. |
| 10 | 李苗苗, 王海啸, 陶国全. CDKN3基因在肝癌中的表达及其对肝癌细胞生长、细胞周期的影响[J]. 山西医科大学学报, 2016, 47(6): 496-501. |
| 11 | 朱 慧, 陆欢平, 李天佑, 等. CDKN3在口腔鳞状细胞癌中的预后价值及免疫细胞浸润分析[J]. 国际检验医学杂志, 2024, 45(11): 1302-1307. |
| 12 | 胡 赞, 孙 锐, 程方雄, 等. 胰腺癌中细胞周期依赖激酶抑制剂3的表达及意义[J]. 实用医学杂志, 2018, 34(20): 3403-5. |
| 13 | de Visser KE, Joyce JA. The evolving tumor microenvironment: From cancer initiation to metastatic outgrowth[J]. Cancer Cell, 2023, 41(3): 374-403. |
| 14 | Ma J, Zhou W, Yuan Y, et al. PSMD12 interacts with CDKN3 and facilitates pancreatic cancer progression[J]. Cancer Gene Ther, 2023, 30(8): 1072-83. |
| 15 | Srinivas V, Kitagawa M, Wong J, et al. The tumor suppressor Cdkn3 is required for maintaining the proper number of centrosomes by regulating the centrosomal stability of Mps1[J]. Cell Rep, 2015, 13(8): 1569-77. |
| 16 | Zhang CL, Shen Q, Gao MQ, et al. The role of Cyclin Dependent Kinase Inhibitor 3 (CDKN3) in promoting human tumors: Literature review and pan-cancer analysis[J]. Heliyon, 2024, 10(4): e26061. |
| 17 | Li Y, Ji S, Fu LY, et al. Knockdown of cyclin-dependent kinase inhibitor 3 inhibits proliferation and invasion in human gastric cancer cells[J]. Oncol Res, 2017, 25(5): 721-31. |
| 18 | Zhou Y, Bian S, Zhou X, et al. Single-cell multiomics sequencing reveals prevalent genomic alterations in tumor stromal cells of human colorectal cancer[J]. Cancer Cell, 2020, 38(6): 818-28. e5. |
| 19 | Cooper J, Giancotti FG. Integrin signaling in cancer: mechanotransduction, stemness, epithelial plasticity, and therapeutic resistance[J]. Cancer Cell, 2019, 35(3): 347-67. |
| 20 | Yan CS, Zheng L, Jiang ST, et al. Exhaustion-associated cholesterol deficiency dampens the cytotoxic arm of antitumor immunity[J]. Cancer Cell, 2023, 41(7): 1276-93.e11. |
| 21 | 米贤良, 魏子白, 杨长青. 基于生物信息学分析胃癌与癌旁组织中差异基因的表达[J]. 现代消化及介入诊疗, 2022, 27(11): 1419-23. |
| 22 | Ucaryilmaz Metin C, Ozcan G. Comprehensive bioinformatic analysis reveals a cancer-associated fibroblast gene signature as a poor prognostic factor and potential therapeutic target in gastric cancer[J]. BMC Cancer, 2022, 22(1): 692. |
| 23 | Pistritto G, Trisciuoglio D, Ceci C, et al. Apoptosis as anticancer mechanism: function and dysfunction of its modulators and targeted therapeutic strategies[J]. Aging: Albany NY, 2016, 8(4): 603-19. |
| 24 | Patriarca C, Pini GM, Conti G. Invasion and metastasis: a historical perspective[J]. Pathologica, 2020, 112(4): 229-33. |
| 25 | Park W, Wei S, Kim BS, et al. Diversity and complexity of cell death: a historical review[J]. Exp Mol Med, 2023, 55(8): 1573-94. |
| 26 | Bedoui S, Herold MJ, Strasser A. Emerging connectivity of programmed cell death pathways and its physiological implications[J]. Nat Rev Mol Cell Biol, 2020, 21(11): 678-95. |
| 27 | Vaghari-Tabari M, Ferns GA, Qujeq D, et al. Signaling, metabolism, and cancer: an important relationship for therapeutic intervention[J]. J Cell Physiol, 2021, 236(8): 5512-32. |
| 28 | Herriage HC, Huang YT, Calvi BR. The antagonistic relationship between apoptosis and polyploidy in development and cancer[J]. Semin Cell Dev Biol, 2024, 156: 35-43. |
| 29 | Hadian K, Stockwell BR. The therapeutic potential of targeting regulated non-apoptotic cell death[J]. Nat Rev Drug Discov, 2023, 22(9): 723-42. |
| 30 | Kumar S, Dorstyn L, Lim Y. The role of caspases as executioners of apoptosis[J]. Biochem Soc Trans, 2022, 50(1): 33-45. |
| 31 | Li MM, Yang JY, Li J, et al. Epiberberine induced p53/p21-dependent G2/M cell cycle arrest and cell apoptosis in gastric cancer cells by activating γ‑aminobutyric acid receptor‑β3[J]. Phytomedicine, 2024, 123: 155198. |
| 32 | 艾亚楠, 赵唯含. NF-κB信号通路在胃癌前病变中的发病机制及中西医治疗的研究进展[J]. 中国实验方剂学杂志, 2022, 28(24): 237-43. |
| 33 | Schäfer C, Göder A, Beyer M, et al. Class I histone deacetylases regulate p53/NF-κB crosstalk in cancer cells[J]. Cell Signal, 2017, 29: 218-25. |
| [1] | 常笑语, 张瀚文, 曹红亭, 侯玲, 孟鑫, 陶虹, 罗彦, 李光华. 热应激对大鼠胸主动脉内皮细胞生物钟基因 Bmal1和细胞周期蛋白表达水平的影响[J]. 南方医科大学学报, 2025, 45(7): 1353-1362. |
| [2] | 陈鑫源, 吴成挺, 李瑞迪, 潘雪芹, 张耀丹, 陶俊宇, 林才志. 双术汤通过P53/SLC7A11/GPX4通路诱导胃癌细胞铁死亡[J]. 南方医科大学学报, 2025, 45(7): 1363-1371. |
| [3] | 谢婷, 王云云, 郭婷, 袁春华. 雷氏大疣蛛多肽毒素组分通过激活促凋亡通路和协同作用抑制癌细胞增殖[J]. 南方医科大学学报, 2025, 45(7): 1460-1470. |
| [4] | 庞金龙, 赵新丽, 张振, 王豪杰, 周星琦, 杨玉梅, 李姗姗, 常小强, 李锋, 李娴. 皮肤黑色素瘤中MMRN2高表达促进肿瘤细胞的侵袭和迁移并与不良预后相关[J]. 南方医科大学学报, 2025, 45(7): 1479-1489. |
| [5] | 吴璇, 方家敏, 韩玮玮, 陈琳, 孙菁, 金齐力. 高表达PRELID1促进胃癌细胞上皮间质转化并与不良预后相关[J]. 南方医科大学学报, 2025, 45(7): 1535-1542. |
| [6] | 王康, 李海宾, 余靖, 孟源, 张虹丽. ELFN1高表达是结肠癌的预后生物标志物并促进结肠癌细胞的增殖转移[J]. 南方医科大学学报, 2025, 45(7): 1543-1553. |
| [7] | 侯鑫睿, 张振东, 曹明远, 杜予心, 王小平. 红景天苷靶向miR-1343-3p-OGDHL/PDHB糖代谢轴抑制胃癌细胞的体内外增殖[J]. 南方医科大学学报, 2025, 45(6): 1226-1239. |
| [8] | 杨毓甲, 杨丽芳, 吴雅玲, 段兆达, 于春泽, 吴春云, 于建云, 杨力. 大麻二酚经PERK-eIF2α-ATF4-CHOP通路减轻多重脑震荡大鼠的神经元内质网应激和凋亡[J]. 南方医科大学学报, 2025, 45(6): 1240-1250. |
| [9] | 曾玉梅, 李继科, 黄仲曦, 周毅波. 绒毛样蛋白VILL通过与LMO7蛋白相互作用抑制鼻咽癌细胞的增殖[J]. 南方医科大学学报, 2025, 45(5): 954-961. |
| [10] | 陈悦, 肖林雨, 任侣, 宋雪, 李静, 胡建国. 水晶兰苷通过抑制PI3K/AKT信号通路减少神经元凋亡改善脊髓损伤后小鼠的运动功能[J]. 南方医科大学学报, 2025, 45(4): 774-784. |
| [11] | 储菲, 陈孝华, 宋博文, 杨晶晶, 左芦根. 苏荠宁黄酮通过抑制PI3K/AKT信号通路拮抗肠上皮细胞凋亡改善小鼠实验性结肠炎[J]. 南方医科大学学报, 2025, 45(4): 819-828. |
| [12] | 岳雅清, 牟召霞, 王希波, 刘艳. Aurora-A过表达通过激活NF-κBp65/ARPC4信号轴促进宫颈癌细胞的侵袭和转移[J]. 南方医科大学学报, 2025, 45(4): 837-843. |
| [13] | 庆顺杰, 沈智勇. 过表达己糖激酶2通过激活JAK/STAT途径促进结直肠癌细胞的增殖、迁移和侵袭并调节肿瘤免疫微环境[J]. 南方医科大学学报, 2025, 45(3): 542-553. |
| [14] | 黄晴晴, 张文静, 张小凤, 王炼, 宋雪, 耿志军, 左芦根, 王月月, 李静, 胡建国. 高表达MYO1B促进胃癌细胞增殖、迁移和侵袭并与患者的不良预后有关[J]. 南方医科大学学报, 2025, 45(3): 622-631. |
| [15] | 李华莉, 宋婷, 刘嘉雯, 李永宝, 姜兆静, 窦文, 周凌宏. 预后导向的肺癌调强放疗计划优化新方法[J]. 南方医科大学学报, 2025, 45(3): 643-649. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||