南方医科大学学报 ›› 2026, Vol. 46 ›› Issue (3): 523-531.doi: 10.12122/j.issn.1673-4254.2026.03.06
• 基础研究 • 上一篇
梁芷晴1,3( ), 潘富珍2,4, 邓利强4, 麦哲芬4, 马云2, 施传坚2,4(
), 潘富珍2,4, 邓利强4, 麦哲芬4, 马云2, 施传坚2,4( ), 付卫明1,3(
), 付卫明1,3( )
)
收稿日期:2025-09-11
出版日期:2026-03-20
发布日期:2026-03-26
通讯作者:
施传坚,付卫明
E-mail:liangzqing2025@163.com;shichuanjian2018@163.com;fuweiming76@smu.edu.cn
作者简介:梁芷晴,在读硕士研究生,E-mail: liangzqing2025@163.com
基金资助:
Zhiqing LIANG1,3( ), Fuzhen PAN2,4, Liqiang DENG4, Zhefen MAI4, Yun MA2, Chuanjian SHI2,4(
), Fuzhen PAN2,4, Liqiang DENG4, Zhefen MAI4, Yun MA2, Chuanjian SHI2,4( ), Weiming FU1,3(
), Weiming FU1,3( )
)
Received:2025-09-11
Online:2026-03-20
Published:2026-03-26
Contact:
Chuanjian SHI, Weiming FU
E-mail:liangzqing2025@163.com;shichuanjian2018@163.com;fuweiming76@smu.edu.cn
Supported by:摘要:
目的 探讨生脉散改善非小细胞肺癌奥希替尼耐药的作用和机制。 方法 制备大鼠含药血清,单用或联合奥希替尼处理人肺癌细胞A549检测细胞活力的变化,试剂盒检测细胞葡萄糖和乳酸含量变化,构建小鼠皮下肺癌模型,观察生脉散联合奥希替尼使用对奥希替尼体内抗肺癌活性的影响。Western blotting、RT-qPCR和免疫荧光实验检测生脉散和乳酸对Wnt/β-catenin/LDHA信号通路的影响。 结果 生脉散以浓度依赖性增加A549细胞对奥希替尼的药物敏感性,与单用奥希替尼组比较,生脉散联合奥希替尼抑制细胞活力(P<0.001),克隆形成(P<0.001)和肺癌细胞体内增长(P<0.05),生脉散呈剂量依赖性减少A549细胞葡萄糖和乳酸含量(P<0.05)。Western blotting结果显示生脉散抑制LDHA、β-catenin总蛋白、细胞质和细胞核β-catenin蛋白表达(P<0.01),而乳酸则激活β-catenin总蛋白、细胞核β-catenin和LDHA蛋白表达(P<0.001);免疫荧光结果显示乳酸激活β-catenin蛋白核积累,而生脉散抑制细胞核β-catenin蛋白表达;RT-qPCR结果显示乳酸激活Wnt/β-catenin下游靶基因(c-myc、CD44、Axin2、Oct3/4、Survivin和CCND1)的表达(P<0.05),而生脉散则抑制其表达(P<0.05)。 结论 生脉散抑制肺癌细胞乳酸/Wnt/β-catenin/LDHA通路,抑制糖酵解水平,改善非小细胞肺癌奥希替尼耐药。
梁芷晴, 潘富珍, 邓利强, 麦哲芬, 马云, 施传坚, 付卫明. 生脉散通过调控乳酸/Wnt/β-catenin/LDHA通路改善非小细胞肺癌奥希替尼耐药的作用机制[J]. 南方医科大学学报, 2026, 46(3): 523-531.
Zhiqing LIANG, Fuzhen PAN, Liqiang DENG, Zhefen MAI, Yun MA, Chuanjian SHI, Weiming FU. Shengmai San improves osimertinib resistance of non-small cell lung cancer cells by regulating the lactate/Wnt/β-catenin/LDHA pathway[J]. Journal of Southern Medical University, 2026, 46(3): 523-531.
| Gene name | Sequence (5' to 3') | |
|---|---|---|
| β-actin | Forward | GTCATTCCAAATATGAGATGCGT |
| Reverse | GCTATCACCTCCCCTGTGTG | |
| c-Myc | Forward | TTCGGGTAGTGGAAAACCAG |
| Reverse | CAGCAGCTCGAATTTCTTCC | |
| CD44 | Forward | TCAGAGGAGTAGGAGAGAGGAAAC |
| Reverse | GAAAAGTCAAAGTAACAATAACAGTGG | |
| Axin-2 | Forward | GGATCGCTCCTCTTGAAGGA |
| Reverse | CTTTCGCCAACCGTGGTT | |
| Survivin | Forward | CCACCGCATCTCTACATTCAAG |
| Reverse | CAAGTCTGGCTCGTTCTCAGTG | |
| Oct3/4 | Forward | TCGAGAACCGAGTGAGAGGC |
| Reverse | CACACTCGGACCACATCCTTC | |
| Cyclin D1 | Forward | CTGGAGGTCTGCGAGGAACA |
| Reverse | CCTTCATCTTAGAGGCCACGAA | |
| LDHA | Forward | ATGGCAACTCTAAAGGATCAGC |
| Reverse | CCAACCCCAACAACTGTAATCT |
表1 引物序列
Tab.1 Primer sequence for RT-qPCR
| Gene name | Sequence (5' to 3') | |
|---|---|---|
| β-actin | Forward | GTCATTCCAAATATGAGATGCGT |
| Reverse | GCTATCACCTCCCCTGTGTG | |
| c-Myc | Forward | TTCGGGTAGTGGAAAACCAG |
| Reverse | CAGCAGCTCGAATTTCTTCC | |
| CD44 | Forward | TCAGAGGAGTAGGAGAGAGGAAAC |
| Reverse | GAAAAGTCAAAGTAACAATAACAGTGG | |
| Axin-2 | Forward | GGATCGCTCCTCTTGAAGGA |
| Reverse | CTTTCGCCAACCGTGGTT | |
| Survivin | Forward | CCACCGCATCTCTACATTCAAG |
| Reverse | CAAGTCTGGCTCGTTCTCAGTG | |
| Oct3/4 | Forward | TCGAGAACCGAGTGAGAGGC |
| Reverse | CACACTCGGACCACATCCTTC | |
| Cyclin D1 | Forward | CTGGAGGTCTGCGAGGAACA |
| Reverse | CCTTCATCTTAGAGGCCACGAA | |
| LDHA | Forward | ATGGCAACTCTAAAGGATCAGC |
| Reverse | CCAACCCCAACAACTGTAATCT |
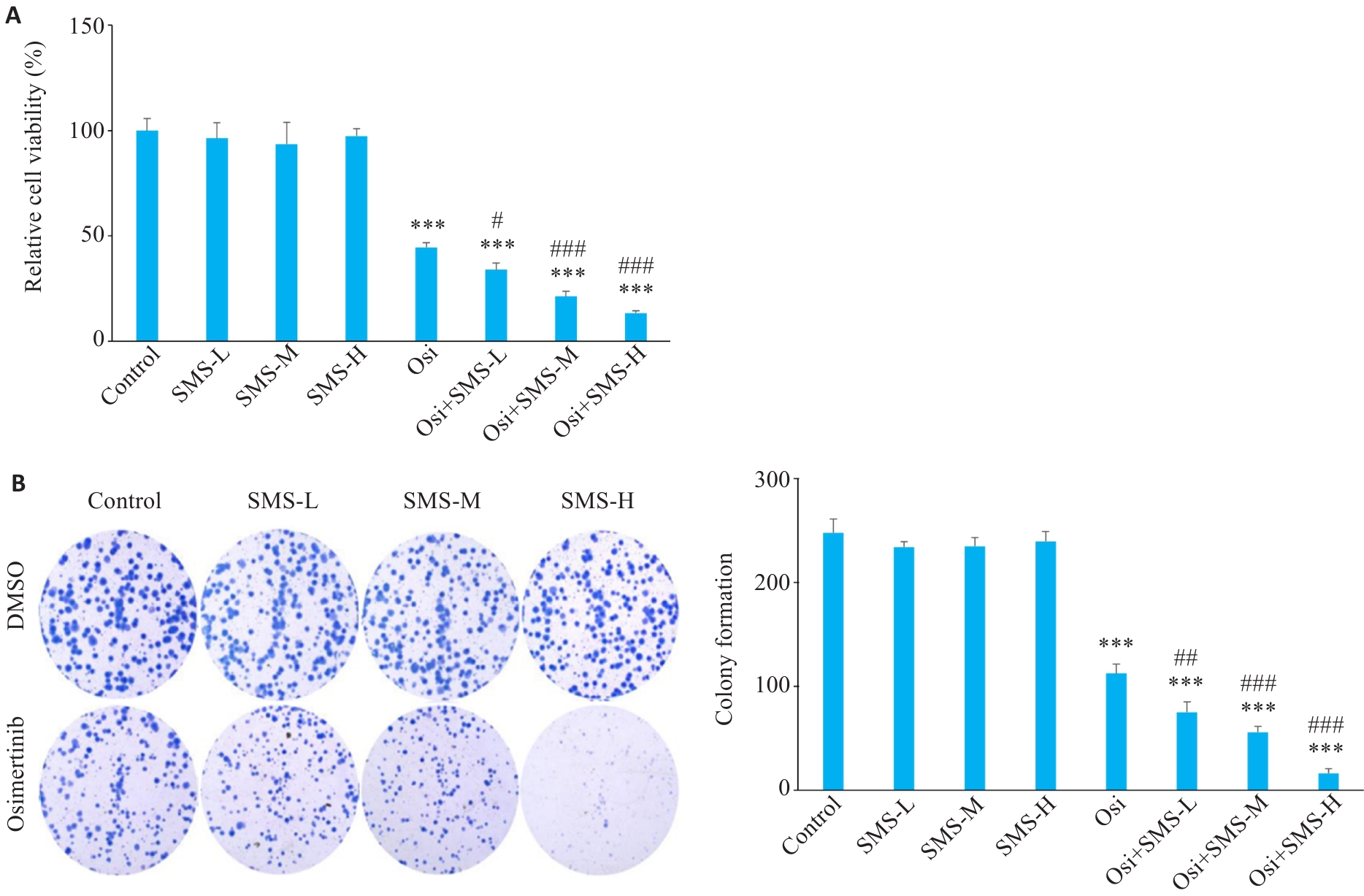
图1 549细胞存活率与克隆形成的能力
Fig.1 Viability and colony-forming ability of A549 cells with different treatments (Mean±SD, n=3). A: Viability of A549 cells treated with SMS, osimertinib, or osimertinib combined with various concentrations of SMS. B: Colony formation of A549 cells and quantitative analysis. ***P<0.001 vs Control group; #P<0.05, ##P<0.01, ###P<0.001 vs Osi group. SMS-L: SMS low-dose group; SMS-M: SMS medium-dose group; SMS-H: SMS high-dose group; Osi: Osimertinib group.
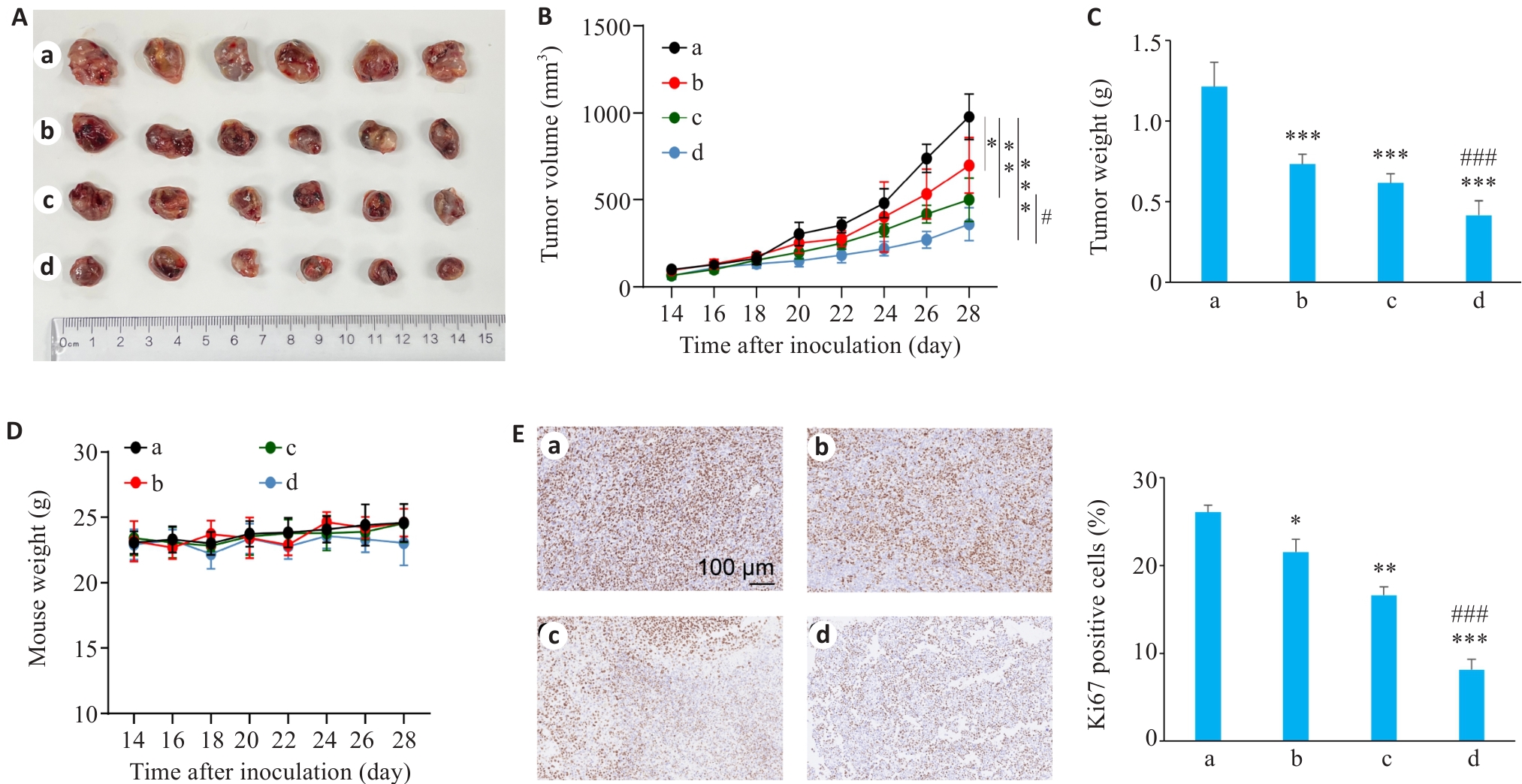
图2 动物实验体内肿瘤的相关情况
Fig.2 Assessment of xenograft growth in nude mice (Mean±SD, n=3). A-C: Representative images of xenograft tumors, growth curve and tumor weight in the tumor-bearing C57BL/6 mice in the 4 treatment groups. D: Changes in body weight of the mice in the 4 treatment groups. E: Immunohistochemistry for Ki67 in the tumor tissues in the 4 groups. *P<0.05, **P<0.01, ***P<0.001 vs Control group; #P<0.05, ###P<0.001 vs Osi group. a: Control group; b: SMS Medium-dose group; c: Osimertinib monotherapy group; d: SMS medium-dose combined with osimertinib group.
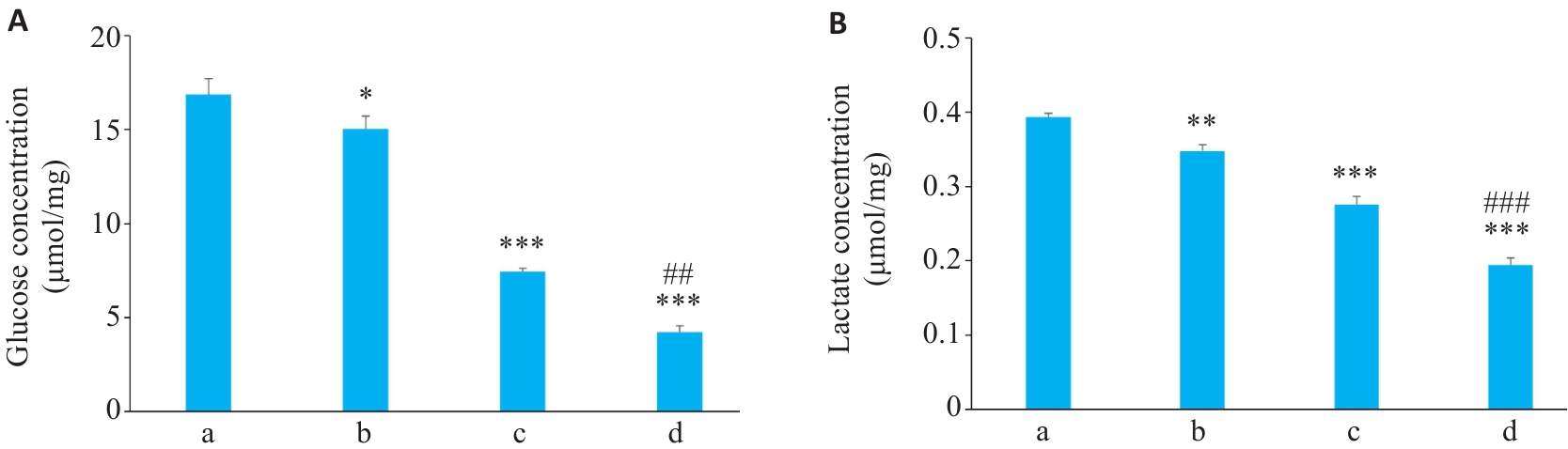
图3 各组细胞葡萄糖和乳酸浓度
Fig.3 Glucose (A) and lactate (B) concentrations A549 cells in each group (Mean±SD, n=3). *P<0.05, **P<0.01, ***P<0.001 vs Control group; ##P<0.01, ###P<0.001 vs Osi group. a: Control group; b: SMS Medium-dose group; c: Osimertinib monotherapy group; d: SMS medium-dose combined with osimertinib group.
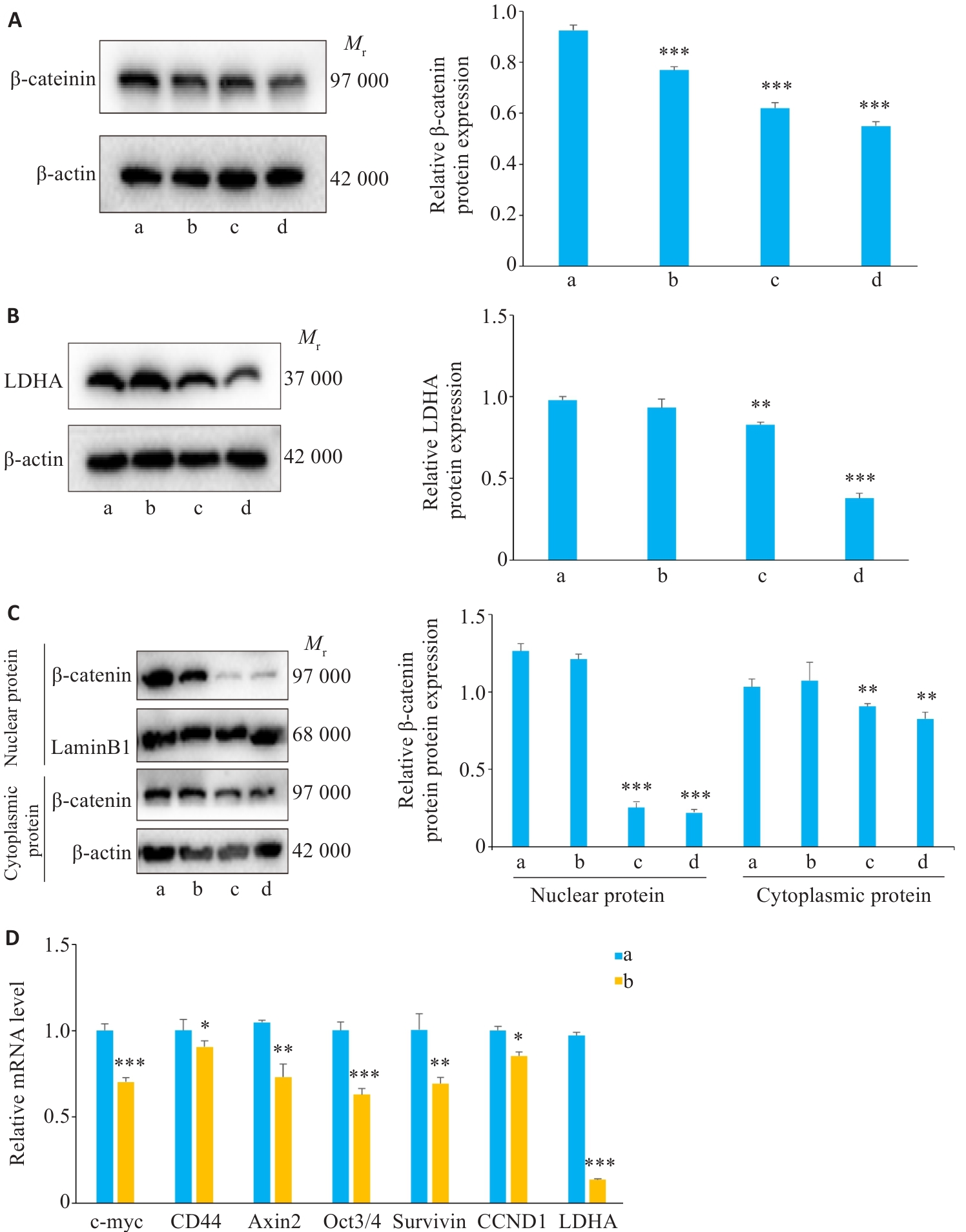
图4 各组Wnt/β-catenin/LDHA信号通路相关蛋白和基因表达情况比较
Fig.4 Protein and mRNA expressions of the Wnt/β-catenin/LDHA signaling pathway in A549 cells in different groups (Mean±SD, n=3). A, B: Western blotting of β-catenin and LDHA expression levels in A549 cells. C: Western blotting of β-catenin expression and quantitative analysis its cytoplasmic and nuclear fractions. D: mRNA expressions of Wnt/β-catenin pathway genes detected by by RT-qPCR. *P<0.05, **P<0.01,***P<0.001 vs Control group. a: Control group; b: SMS Medium-dose group; c: Osimertinib monotherapy group; d: SMS medium-dose combined with osimertinib group.
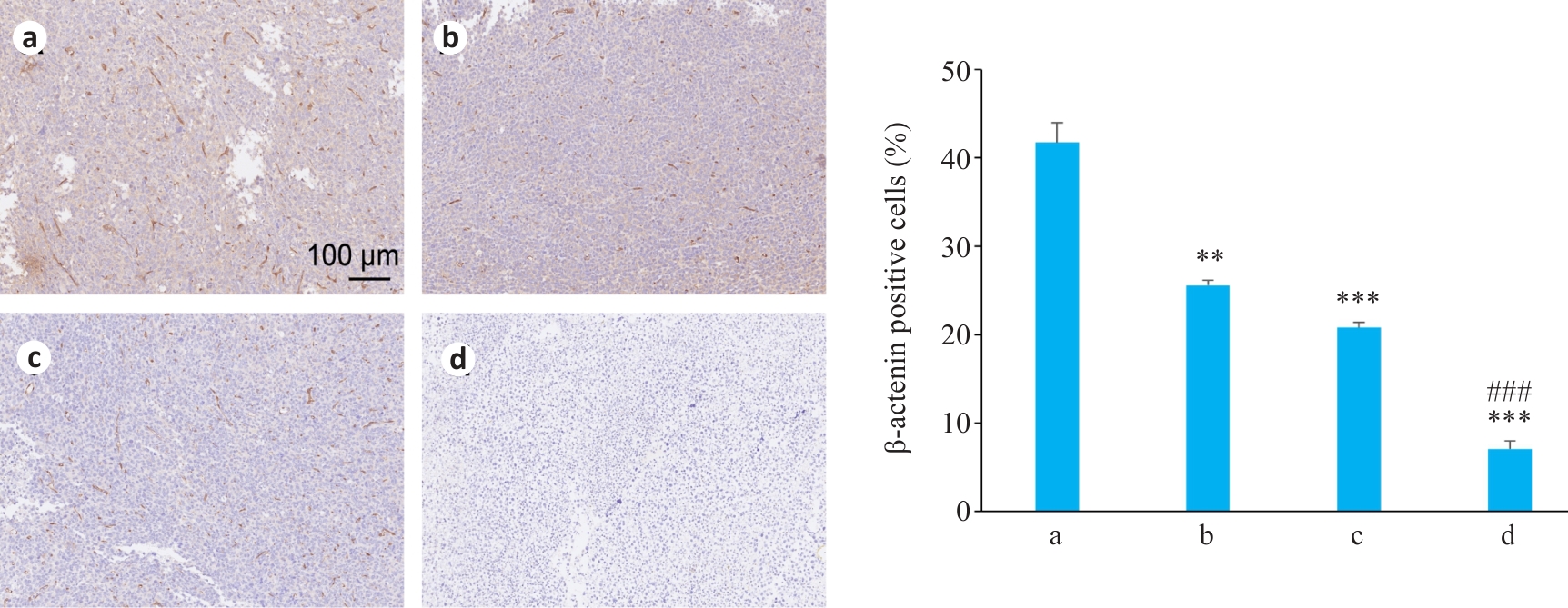
图5 各组体内肿瘤组织中β-catenin蛋白表达比较
Fig.5 Immunohistochemistry of β-catenin protein expression in the tumor tissues in different groups (Mean±SD, n=3). **P<0.01, ***P<0.001 vs Control group;###P<0.001 vs Osimertinib group. a: Control group; b: SMS Medium-dose group; c: Osimertinib monotherapy group; d: SMS medium-dose combined with osimertinib group.
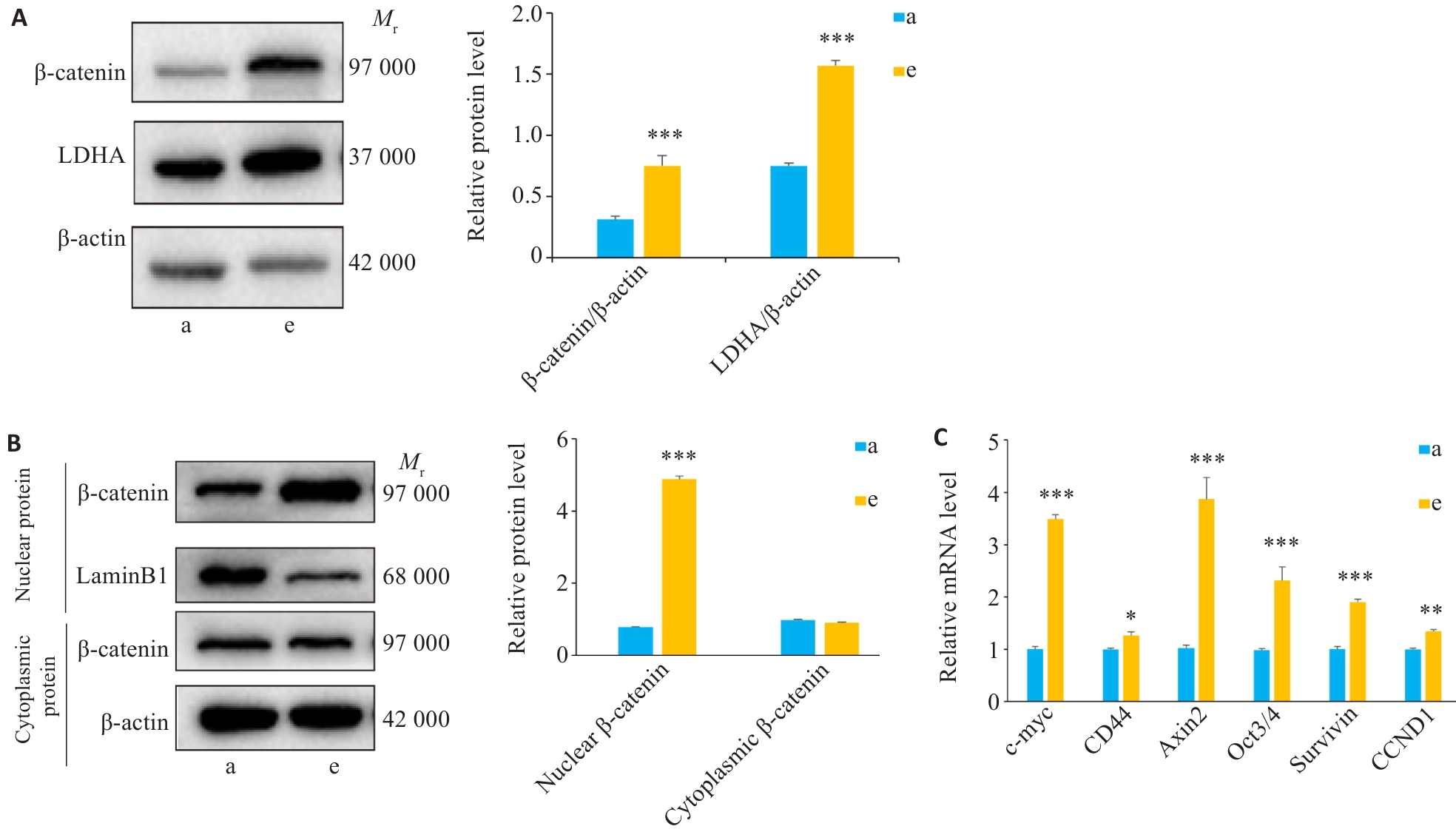
图6 各组Wnt/β-catenin/LDHA信号通路相关蛋白和基因表达情况比较
Fig.6 Expression of Wnt/β-catenin/LDHA pathway components in A549 cells with different treatments (Mean±SD, n=3). A: Western blotting of β‑catenin and LDHA expression in control and 10 mmol/L lactate-treated cells. B: Western blotting of cytoplasmic and nuclear fractions of β‑catenin in control and 10 mmol/L lactate-treated cells. C: mRNA expression of Wnt/β-catenin downstream targets detected by RT-qPCR. *P<0.05, **P<0.01, ***P<0.001 vs Control group. a: Control group; e: 10 mmol/L lactate treatment group.
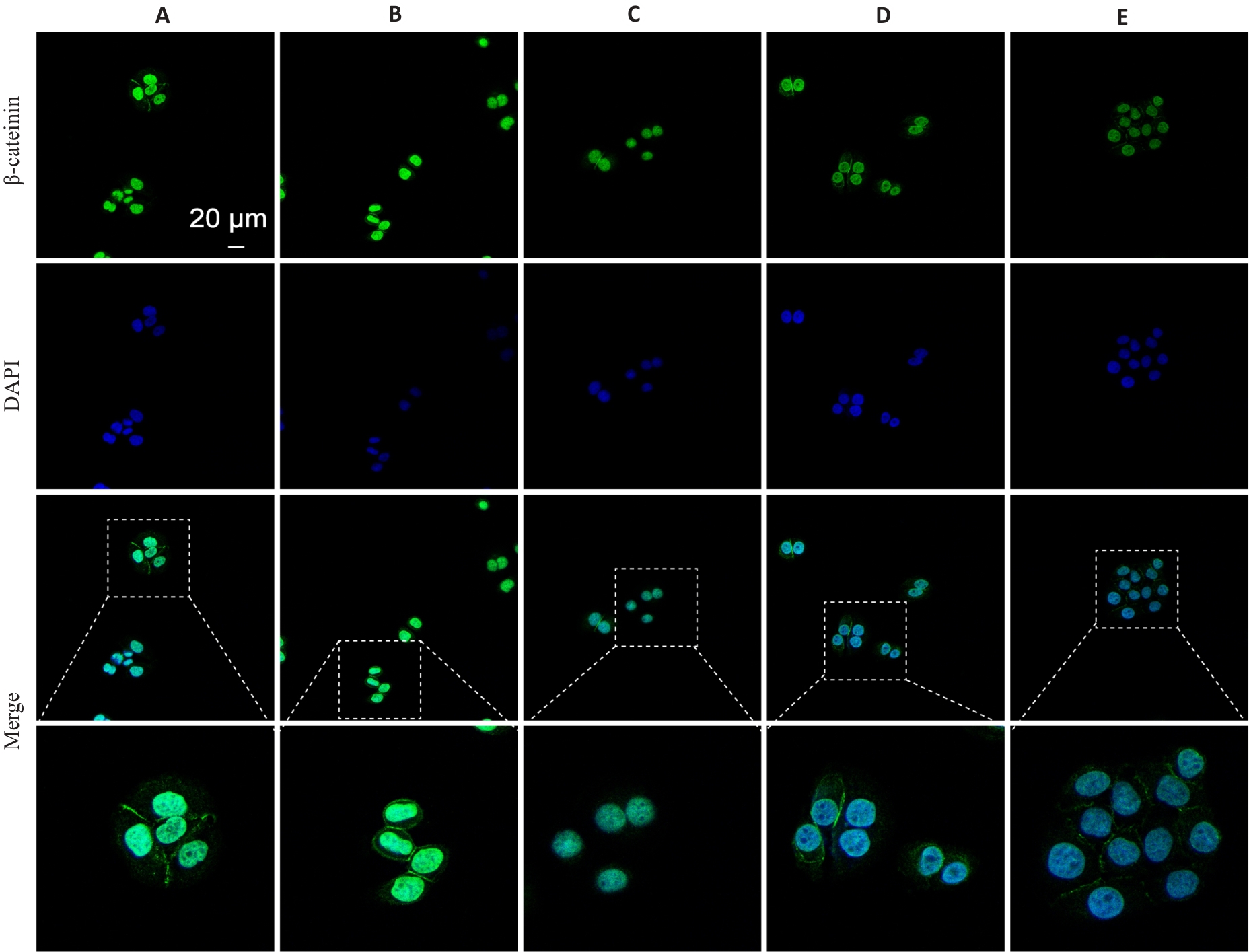
图7 细胞免疫荧光检测细胞β-catenin蛋白的变化
Fig.7 Immunofluorescence staining for detecting β-catenin expression in A549 cells. A: Control group; B: SMS-Medium-dose group; C: Osimertinib monotherapy group; D: SMS medium-dose combined with osimertinib group; E: 10 mmol/L lactate treatment group.
| [1] | Zhao DF, Li WZ, Li WY. Screening for lung cancer, overdiagnosis, and health care utilization: a nationwide population-based study[J]. J Thorac Oncol, 2025, 20(10): e114-5. doi:10.1016/j.jtho.2025.06.003 |
| [2] | Huang JL, Zhang XK, Zhang H, et al. Addressing clinical limitations of glutaminase inhibitors: novel strategies for osimertinib-resistant lung cancer by exploiting glutamine metabolic dependency[J]. Adv Sci (Weinh), 2025, 12(6): e2411479. doi:10.1002/advs.202411479 |
| [3] | Blaquier JB, Ortiz-Cuaran S, Ricciuti B, et al. Tackling osimertinib resistance in EGFR-mutant non-small cell lung cancer[J]. Clin Cancer Res, 2023, 29(18): 3579-91. doi:10.1158/1078-0432.ccr-22-1912 |
| [4] | Zhou Y, Huang SQ, Guo YZ, et al. Epigallocatechin gallate circumvents drug-induced resistance in non-small-cell lung cancer by modulating glucose metabolism and AMPK/AKT/MAPK axis[J]. Phytother Res, 2023, 37(12): 5837-53. doi:10.1002/ptr.7990 |
| [5] | Vallée A, Lecarpentier Y, Vallée JN. The key role of the WNT/ β‑catenin pathway in metabolic reprogramming in cancers under normoxic conditions[J]. Cancers (Basel), 2021, 13(21): 5557. doi:10.3390/cancers13215557 |
| [6] | Miao Z, Zhao XM, Liu X. Hypoxia induced β‑catenin lactylation promotes the cell proliferation and stemness of colorectal cancer through the Wnt signaling pathway[J]. Exp Cell Res, 2023, 422(1): 113439. doi:10.1016/j.yexcr.2022.113439 |
| [7] | 磨 炼, 慕晓艳, 李和根. 中药改善非小细胞肺癌患者EGFR-TKIs获得性耐药研究现状及其中医病机探析[J]. 上海中医药杂志, 2023, 57(4): 24-8. doi:10.16305/j.1007-1334.2023.2204044 |
| [8] | 郭禹航, 陈 虎, 苏 钊, 等. 肺癌的中医病机、防治及治法探析: 从六经辨证角度[J]. 亚太传统医药, 2024, 20(11): 199-203. doi:10.11954/ytctyy.202411039 |
| [9] | 吕承启, 牛蔚露, 刘学伟,等. 经典名方生脉散的古籍文献考证与现代临床应用[J].中国实验方剂学杂志,2025,31(22):223-34. |
| [10] | 孙永康,高宏,郑凯.基于“气血津液”理论探讨生脉散在治疗肺癌化疗后气阴两虚证中的作用[J].实用中医内科杂志,2025,39(07):112-5. |
| [11] | Fu K, Xie FC, Wang F, et al. Therapeutic strategies for EGFR-mutated non-small cell lung cancer patients with osimertinib resistance[J]. J Hematol Oncol, 2022, 15(1): 173. doi:10.1186/s13045-022-01391-4 |
| [12] | 赵齐林, 王 楠, 李亚霁, 等. Lnc-TMEM132D-AS1高表达明显降低非小细胞肺癌对奥希替尼的敏感性[J]. 南方医科大学学报, 2023, 43(2): 242-50. doi:10.12122/j.issn.1673-4254.2023.02.12 |
| [13] | 桑舒柳, 姜靖洁, 龚亚斌. 中医药联合EGFR-TKIs治疗NSCLC耐药的分子机制及研究进展[J]. 江西中医药, 2023, 54(11): 80-4. |
| [14] | 李 焕, 贾 妮. 论生脉散研究进展[J]. 辽宁中医药大学学报, 2020, 22(10): 190-3. |
| [15] | Sun MM, Li LL, Niu YJ, et al. PRMT6 promotes tumorigenicity and cisplatin response of lung cancer through triggering 6PGD/ENO1 mediated cell metabolism[J]. Acta Pharm Sin B, 2023, 13(1): 157-73. doi:10.1016/j.apsb.2022.05.019 |
| [16] | Wang S, Zhou LY, Ji N, et al. Targeting ACYP1-mediated glycolysis reverses lenvatinib resistance and restricts hepatocellular carcinoma progression[J]. Drug Resist Updat, 2023, 69: 100976. doi:10.1016/j.drup.2023.100976 |
| [17] | Cang S, Liu R, Jin W, et al. Integrated DIA proteomics and lipidomics analysis on non-small cell lung cancer patients with TCM syndromes[J]. Chin Med, 2021, 16(1): 126. doi:10.1186/s13020-021-00535-x |
| [18] | 王 敏, 王 媛, 孙 静, 等. 人参养荣汤对Lewis肺癌糖酵解途径相关酶LDH-A、HK2基因表达的影响[J]. 辽宁中医杂志, 2022, 49(1): 180-4. doi:10.13192/j.issn.1000-1719.2022.01.051 |
| [19] | 文艳萍, 姜 怡, 沈丽萍, 等. 基于有氧糖酵解探讨加味四君子汤合沙参麦冬汤提高EGFR-TKI耐药肺腺癌细胞顺铂敏感性的作用机制[J]. 中国实验方剂学杂志, 2025, 31(1): 39-46. |
| [20] | 余 功, 陈江涛, 胡 桥, 等. 清燥救肺汤对荷Lewis小鼠肺癌细胞糖酵解关键限速酶HK2, PFK2, PKM2的影响[J]. 中国实验方剂学杂志, 2020, 26(4): 54-8. doi:10.13422/j.cnki.syfjx.20191325 |
| [21] | Pachimatla AG, Fenstermaker R, Ciesielski M, et al. Survivin in lung cancer: a potential target for therapy and prevention-a narrative review[J]. Transl Lung Cancer Res, 2024, 13(2): 362-74. doi:10.21037/tlcr-23-621 |
| [22] | Ando T, Yamasaki J, Saya H, et al. CD44: a key regulator of iron metabolism, redox balance, and therapeutic resistance in cancer stem cells[J]. Stem Cells, 2025, 43(6): sxaf024. doi:10.1093/stmcls/sxaf024 |
| [23] | Lee H, Kim B, Park J, et al. Cancer stem cells: landscape, challenges and emerging therapeutic innovations[J]. Signal Transduct Target Ther, 2025, 10(1): 248. doi:10.1038/s41392-025-02360-2 |
| [24] | Zhang ZX, Westover D, Tang ZT, et al. Wnt/β‑catenin signaling in the development and therapeutic resistance of non-small cell lung cancer[J]. J Transl Med, 2024, 22(1): 565. doi:10.1186/s12967-024-05380-8 |
| [25] | Liu JQ, Xiao Q, Xiao JN, et al. Wnt/β-catenin signalling: function, biological mechanisms, and therapeutic opportunities[J]. Signal Transduct Target Ther, 2022, 7(1): 3. doi:10.1038/s41392-021-00762-6 |
| [26] | Wang MY, Zhou QL, Cao TP, et al. Lactate dehydrogenase A: a potential new target for tumor drug resistance intervention[J]. J Transl Med, 2025, 23(1): 713. doi:10.1186/s12967-025-06773-z |
| [27] | Chen ZZ, He QK, Lu TK, et al. mcPGK1-dependent mitochondrial import of PGK1 promotes metabolic reprogramming and self-renewal of liver TICs[J]. Nat Commun, 2023, 14(1): 1121. doi:10.1038/s41467-023-36651-5 |
| [28] | Li XL, Yang YY, Zhang B, et al. Lactate metabolism in human health and disease[J]. Signal Transduct Target Ther, 2022, 7(1): 305. doi:10.1038/s41392-022-01151-3 |
| [29] | Wu H, Yang JJ, Yang ZX, et al. Targeting the BCKDK/BCLAF1/MYC/HK2 axis to alter aerobic glycolysis and overcome Trametinib resistance in lung cancer[J]. Cell Death Differ, 2025, 32(12): 2210-24. doi:10.1038/s41418-025-01531-6 |
| [30] | Li J, Liu T, Zhao L, et al. Ginsenoside 20(S)‑Rg3 inhibits the Warburg effect through STAT3 pathways in ovarian cancer cells[J]. Int J Oncol, 2015, 46(2): 775-81. doi:10.3892/ijo.2014.2767 |
| [31] | He BC, Gao JL, Luo XJ, et al. Ginsenoside Rg3 inhibits colorectal tumor growth through the down-regulation of Wnt/ß-catenin signaling[J]. Int J Oncol, 2011, 38(2): 437-45. doi:10.3892/ijo.2010.858 |
| [32] | 白 晶. 麦冬皂苷B对人非小细胞肺癌A549裸鼠移植瘤糖酵解的抑制作用及机制研究[D]. 长春: 吉林大学, 2021. |
| [33] | Zhang SP, Li HX, Li LQ, et al. Ophiopogonin B inhibits migration and invasion in non-small cell lung cancer cells through enhancing the interaction between Axin and β‑catenin[J]. J Cancer, 2021, 12(20): 6274-84. doi:10.7150/jca.60066 |
| [34] | 王首星, 刘院刚, 温进平, 等. 五味子甲素调控miR-873-5p/G6PD轴对胃癌SGC-7901细胞活力、凋亡及有氧糖酵解的影响[J]. 中国中西医结合消化杂志, 2024, 32(7): 575-80, 585. |
| [1] | 朱昊坤, 郭延哺, 辛向军, 李朝阳, 周冬明. 基于异质图动态特征学习的药物重定位预测[J]. 南方医科大学学报, 2026, 46(2): 456-465. |
| [2] | 杨勤军, 朱虹宇, 高远, 杨程, 刘桐, 张璐, 童佳兵, 李泽庚. 桑麻止咳方改善感染后咳嗽大鼠气道炎症和敏感性:基于TRPV1-SP/CGRP和细胞焦亡途径[J]. 南方医科大学学报, 2025, 45(9): 1830-1839. |
| [3] | 黄启智, 谢戴鹏, 姚霖彤, 李洽轩, 吴少伟, 周海榆. 肿瘤微环境特异性CT影像组学标签预测非小细胞肺癌免疫治疗疗效[J]. 南方医科大学学报, 2025, 45(9): 1903-1918. |
| [4] | 郑子瑜, 杨夏颖, 吴圣杰, 张诗婕, 吕国荣, 柳培忠, 王珺, 何韶铮. 多特征融合的产时超声胎方位识别模型[J]. 南方医科大学学报, 2025, 45(7): 1563-1570. |
| [5] | 何丽君, 陈晓菲, 闫陈昕, 师林. 扶正化积汤治疗非小细胞肺癌的分子机制:基于网络药理学及体外实验验证[J]. 南方医科大学学报, 2025, 45(6): 1143-1152. |
| [6] | 计寰宇, 王蕊, 高盛祥, 车文刚. SG-UNet:基于全局注意力和自校准卷积增强的黑色素瘤分割模型[J]. 南方医科大学学报, 2025, 45(6): 1317-1326. |
| [7] | 李丹丹, 楚佳鑫, 闫妍, 徐文隽, 朱行春, 孙韵, 丁浩峰, 任丽, 朱博. 姜黄素通过下调HIF-1α通路抑制非小细胞肺癌脂质代谢[J]. 南方医科大学学报, 2025, 45(5): 1039-1046. |
| [8] | 李苏强, 王周阳, 产思贤, 周小龙. AConvLSTM U-Net:基于双向稠密连接和注意力机制的多尺度颌骨囊肿分割模型[J]. 南方医科大学学报, 2025, 45(5): 1082-1092. |
| [9] | 陈燕才, 张高峰, 李树波, 罗念祥, 张翼. 新型桥接钢板治疗RockwoodⅢ型肩锁关节脱位的生物力学性能[J]. 南方医科大学学报, 2025, 45(5): 1103-1112. |
| [10] | 宋添力, 王一民, 孙童, 刘绪, 黄胜, 冉云. 正肝方对二乙基亚硝胺诱导的肝癌大鼠的抗癌作用及机制:基于激活Hippo/YAP通路[J]. 南方医科大学学报, 2025, 45(4): 799-809. |
| [11] | 黄妍, 陈曦, 秦梦晨, 高磊. 活络效灵丹促进斑马鱼尾鳍再生的核心靶点与免疫调控机制[J]. 南方医科大学学报, 2025, 45(3): 494-505. |
| [12] | 任煜瀛, 黄凌霄, 杜方, 姚新波. 基于改进RT-DETR的多尺度特征融合的高效轻量皮肤病理检测方法[J]. 南方医科大学学报, 2025, 45(2): 409-421. |
| [13] | 付长龙, 陈若岚, 徐诗淇, 游锦欣, 林晴, 黄艳峰. 巴戟天多糖通过靶向lncRNA XIST调控糖酵解-焦亡延缓小鼠骨关节炎软骨细胞退变[J]. 南方医科大学学报, 2025, 45(12): 2541-2550. |
| [14] | 崔俊杰, 赖睿茵, 陈苏衡, 瞿珊珊, 廖阅, 马雪, 李玉兰. 过度劳累损伤小鼠心肌能量代谢稳态[J]. 南方医科大学学报, 2025, 45(12): 2598-2606. |
| [15] | 陈紫豪, 郭延哺, 宋胜利, 郭全明, 周冬明. 基于多层语义与拓扑融合的异质图方法提升药物-靶标相互作用预测性能[J]. 南方医科大学学报, 2025, 45(11): 2394-2404. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||